Loss of Migratory Traditions Makes the Endangered Patagonian Huemul Deer a Year-Round Refugee in Its Summer Habitat
Abstract
:1. Introduction
2. Materials and Methods
2.1. Systematic Literature Review
2.2. Radiotelemetry
3. Results
3.1. Historical Distribution
3.2. Extent of Altitudinal Movements
3.3. Extent of Horizontal Movements and Size of Annual Home Ranges (MCP)
3.4. Processes of Migration and Geographic Expansion to New Habitat by Wild Cervids
4. Discussion
4.1. Historical Spatial Habitat Use
4.2. Historical Seasonal Habitat Use
4.3. Contemporary Spatiotemporal Habitat Use in the Protected Park Shoonem
4.4. Implications of Having Lost Migratory Traditions
4.5. Implications for Conservation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Total Data Points (N) | Minimal Separation (m) | Vital Status | |||||
|---|---|---|---|---|---|---|---|
| Cases | Summer | Winter | Spring/Fall | Horizontal | Altitudinal | Capture | Dead |
| female 1 | 3 | 6 | 4 | 122 | 68 | 2017 | unknown |
| female 2 | 2 | 3 | 1 | 2140 | 143 | 2017 | 2018 |
| female 3 | 8 | 7 | 5 | 0 | 0 | 2017 | alive |
| male 1 | 3 | 3 | 5 | 423 | 38 | 2017 | 2018 |
| male 2 | 7 | 7 | 1 | 89 | 71 | 2017 | 2020 |
| male 3 | 14 | 4 | 6 | 0 | 0 | 2017 | 2022 |
| GPS Fix * (n) | Area (ha) | Perimeter (m) | Overlap | Seasonal Displacement | |
|---|---|---|---|---|---|
| (a) Aug 13–Sept 30 | 437 | 163 | 5760 | 76 % | 490 m more south than (b) |
| (b) Oct 1–Dec 10 | 405 | 147 | 4810 | 85% | 235 m more north than (a) |
| (c) Dec 11–Mar 7 2022 | 833 | 158 | 5440 | no changes | |
| Total survey | 1675 | 190 | 6610 |
References
- Black-Decima, P.A.; Corti, P.; Díaz, N.; Fernandez, R.; Geist, V.; Gill, R.; Gizejewski, Z.; Jiménez, J.; Pastore, H.; Saucedo, C.; et al. Hippocamelus bisulcus. In The IUCN Red List of Threatened Species; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2016. [Google Scholar]
- Riquelme, C.; Estay, S.A.; López, R.; Pastore, H.; Soto-Gamboa, M.; Corti, P. Protected areas’ effectiveness under climate change: A latitudinal distribution projection of an endangered mountain ungulate along the Andes Range. PeerJ 2018, 6, e5222. [Google Scholar] [CrossRef] [PubMed]
- Smith-Flueck, J.M.; Flueck, W.T. Natural mortality patterns in a population of southern Argentina huemul (Hippocamelus bisulcus), an endangered Andean cervid. Zeits. Jagdwiss. 2001, 47, 178–188. [Google Scholar] [CrossRef]
- Putman, R.; Flueck, W.T. Intraspecific variation in biology and ecology of deer: Magnitude and causation. Anim. Prod. Sci. 2011, 51, 277–291. [Google Scholar] [CrossRef] [Green Version]
- Flueck, W.T.; Smith-Flueck, J.M. Recent advances in the nutritional ecology of the Patagonian huemul: Implications for recovery. Anim. Prod. Sci. 2011, 51, 311–326. [Google Scholar] [CrossRef]
- Grotta-Neto, F.; Duarte, J.M.B. Movements of Neotropical Forest Deer: What Do We Know? In Movement Ecology of Neotropical Forest Mammals: Focus on Social Animals; Reyna-Hurtado, R., Chapman, C., Eds.; Springer Nature: Cham, Switzerland, 2019; pp. 95–109. [Google Scholar]
- Peters, W.; Hebblewhite, M.; Mysterud, A.; Spitz, D.; Focardi, S.; Urbano, F.; Morellet, N.; Heurich, M.; Kjellander, P.; Linnell, J.D.C.; et al. Migration in geographic and ecological space by a large herbivore. Ecol. Monogr. 2017, 87, 297–320. [Google Scholar] [CrossRef] [Green Version]
- Xu, W.; Barker, K.; Shawler, A.; Van Scoyoc, A.; Smith, J.A.; Mueller, T.; Sawyer, H.; Andreozzi, C.; Bidder, O.R.; Karandikar, H.; et al. The plasticity of ungulate migration in a changing world. Ecology 2021, 102, e03293. [Google Scholar] [CrossRef]
- Howard, W.E. Innate and environmental dispersal of individual vertebrates. Am. Midl. Nat. 1960, 63, 152–161. [Google Scholar] [CrossRef]
- Bolger, D.T.; Newmark, W.D.; Morrison, T.A.; Doak, D.F. The need for integrative approaches to understand and conserve migratory ungulates. Ecol. Lett. 2008, 11, 63–77. [Google Scholar] [CrossRef]
- Festa-Bianchet, M. Learning to migrate. Science 2018, 361, 972–973. [Google Scholar] [CrossRef]
- Jesmer, B.R.; Merkle, J.A.; Goheen, J.R.; Aikens, E.O.; Beck, J.L.; Courtemanch, A.B.; Hurley, M.A.; McWhirter, D.E.; Miyasaki, H.M.; Monteith, K.L.; et al. Is ungulate migration culturally transmitted? Evidence of social learning from translocated animals. Science 2018, 361, 1023–1025. [Google Scholar] [CrossRef] [Green Version]
- Sawyer, H.; Merkle, J.A.; Middleton, A.D.; Dwinnell, S.P.H.; Monteith, K.L. Migratory plasticity is not ubiquitous among large herbivores. J. Anim. Ecol. 2019, 88, 450–460. [Google Scholar] [CrossRef]
- Frid, A. Habitat use by the endangered huemul (Hippocamelus bisculcus): Cattle, snow, and the problem of multiple causes. Biol. Conserv. 2001, 100, 261–267. [Google Scholar] [CrossRef]
- Joppa, L.N.; Pfaff, A. High and Far: Biases in the Location of Protected Areas. PLoS ONE 2009, 4, e8273. [Google Scholar] [CrossRef]
- Quevedo, P.; Von Hardenberg, A.; Pastore, H.; Álvarez, J.; Corti, P. Predicting the potential distribution of the endangered huemul deer Hippocamelus bisulcus in North Patagonia. Oryx 2017, 51, 315–323. [Google Scholar] [CrossRef] [Green Version]
- Kerley, G.I.H.; Kowalczyk, R.; Cromsigt, J.P.G.M. Conservation implications of the refugee species concept and the European bison: King of the forest or refugee in a marginal habitat? Ecography 2012, 35, 519–529. [Google Scholar] [CrossRef]
- Faurby, S.; Araujo, M.B. Anthropogenic range contractions bias species climate change forecasts. Nat. Clim. Change 2018, 8, 252–256. [Google Scholar] [CrossRef]
- Nüchel, J.; Bocher, P.K.; Xiao, W.; Zhu, A.X.; Svenning, J.C. Snub-nosed monkeys (Rhinopithecus): Potential distribution and its implication for conservation. Biodiv. Cons. 2018, 27, 1517–1538. [Google Scholar] [CrossRef] [Green Version]
- Britnell, J.A.; Lewis, R.N.; Elsner-Gearing, F.; Harvey, N.; Stanbrook, E.; Shultz, S. Species stereotypes as a result of unconscious research biases compromise conservation efficacy. Biol. Cons. 2021, 261, 109275. [Google Scholar] [CrossRef]
- Redford, K.H.; Eisenberg, J.F. Mammals of the Neotropics: The Southern Cone, Volume 2, Chile, Argentina, Uruguay, Paraguay; The University of Chicago Press: Chicago, IL, USA; London, UK, 1992. [Google Scholar]
- Aldridge, D.; Lopez, R.; Saucedo, C.; Vila, A.R. Los Ultimos Senderos del Huemul; Fundacion Hiunay: Santiago, Chile, 2007; ISBN 9789567667062. [Google Scholar]
- Vila, A.R.; Saucedo, C.; Aldridge, D.; Ramilo, E.; Corti, P. South Andean Huemul Hippocamelus bisulcus (Molina 1782). In Neotropical Cervidology; Duarte, J.M., González, S., Eds.; FUNEP: Jaboticabal, Brazil, 2010; pp. 89–100. [Google Scholar]
- Gonzalez, S.; Barbanti Duarte, J.M. Speciation, evolutionary history and conservation trends of neotropical deer. Mastozool. Neotrop. 2020, 27, 37–47. [Google Scholar] [CrossRef]
- Escobar Ruiz, E.M.; Smith, J.M.; Flueck, W.T. El Huemul—Shoonem: Madera que se Mueve/re, 2nd ed.; Biblioteca Popular “Dr. Enrique Perea”: Alto Río Senguer, Argentina, 2020. [Google Scholar]
- Díaz, N.I.; Smith-Flueck, J. The Patagonian Huemul. A Mysterious Deer on the Brink of Extinction; Literature of Latin America: Buenos Aires, Argentina, 2000. [Google Scholar]
- Donoso, D.; Iriarte, A.; Segura, B.; Tirado, M. Antecedentes de Huemul (Capitulo 1). In El Huemul de Aysén y Otros Rincones; Iriarte, A., Donoso, D.S., Segura, B., Tirado, M., Eds.; Ediciones Secretaría Regional Ministerial de Agricultura de la Región de Aysén y Flora & Fauna Chile Ltd.: Aysen, Chile, 2017; pp. 13–61. [Google Scholar]
- Smith-Flueck, J.M. La Ecología del Huemul (Hippocamelus bisulcus) en la Patagonia Andina de Argentina y Consideraciones sobre su Conservación. Ph.D Thesis, Universidad Nacional del Comahue, Bariloche, Argentina, 2003; pp. 1–361. [Google Scholar]
- Sauter, T. Revisiting extreme precipitation amounts over southern South America and implications for the Patagonian Icefields. Hydrol. Earth Syst. Sci. 2020, 24, 2003–2020. [Google Scholar] [CrossRef] [Green Version]
- Puig, S.; Rosi, M.I.; Videla, F.; Mendez, E. Summer and winter diet of the guanaco and food availability for a High Andean migratory population (Mendoza, Argentina). Mammal. Biol. 2011, 76, 727–734. [Google Scholar] [CrossRef]
- Kölliker, A.; Kühn, F.; Reichert, F.; Tomsen, A.; Witte, L. Patagonia. Resultado de las expediciones en 1910 a 1916; Sociedad Científica Alemana: Buenos Aires, Argentina, 1917; Volumes 1 and 2. [Google Scholar]
- Citino, S.B.; Bush, M.; Grobler, D.; Lance, W. Anesthesia of boma-captured Lichtenstein’s hartebeest (Sigmocerus lichtensteinii) with a combination of thiafentanil, medetomidine and ketamine. J. Wildl. Dis. 2002, 38, 457–462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muller, L.I.; Osborn, D.A.; Doherty, T.; Keel, M.K.; Miller, B.F.; Warren, R.J.; Mille, K.V. Optimal Medetomidine Dose When Combined with Ketamine and Tiletamine-zolazepam to Immobilize White-tailed Deer. J. Wildl. Dis. 2012, 48, 477–482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flueck, W.T.; Smith-Flueck, J.M. Age-independent osteopathology in skeletons of a south American cervid, the Patagonian huemul (Hippocamelus bisulcus). J. Wildl. Dis. 2008, 44, 636–648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- White, G.C.; Garrott, R.A. Analysis of Wildlife Radio-Tracking Data; Academic Press: San Diego, CA, USA, 1990. [Google Scholar]
- Harris, S.; Cresswell, W.J.; Forde, P.G.; Trewhella, W.J.; Woollard, T.; Wray, S. Home-range analysis using radio tacking data: A review of problems and techniques particularly as applied to the study of mammals. Mammal. Rev. 1990, 20, 97–123. [Google Scholar] [CrossRef]
- Börger, L.; Franconi, N.; de Michele, G.; Gantz, A.; Meschi, F.; Manica, A.; Lovari, S.; Coulson, T. Effects of sampling regime on the mean and variance of home range size estimates. J. Anim. Ecol. 2006, 75, 1393–1405. [Google Scholar] [CrossRef]
- Zweifel-Schielly, B.; Kreuzer, M.; Ewald, K.C.; Suter, W. Habitat selection by an Alpine ungulate: The significance of forage characteristics varies with scale and season. Ecography 2009, 32, 103–113. [Google Scholar] [CrossRef]
- Kropil, R.; Smolko, P.; Garaj, P. Home range and migration patterns of male red deer Cervus elaphus in Western Carpathians. Eur. J. Wildl. Res. 2015, 61, 63–72. [Google Scholar] [CrossRef] [Green Version]
- Kie, J.G.; Matthiopoulos, J.; Fieberg, J.; Powell, R.A.; Cagnacci, F.; Mitchell, M.S.; Gaillard, J.M.; Moorcroft, P.R. The home-range concept: Are traditional estimators still relevant with modern telemetry technology? Philos. Trans. R. Soc. B 2010, 365, 2221–2231. [Google Scholar] [CrossRef] [Green Version]
- Powell, R.A. Animal home ranges and territories and home range estimators. In Research Techniques in Animal Ecology: Controversies and Consequences; Boitani, L., Fuller, T., Eds.; Columbia University Press: New York, NY, USA, 2000. [Google Scholar]
- Bunnefeld, N.; Börger, L.; van Moorter, B.; Rolandsen, C.M.; Dettki, H.; Solberg, E.J.; Ericsson, G. A model-driven approach to quantify migration patterns: Individual, regional and yearly differences. J. Anim. Ecol. 2011, 80, 466–476. [Google Scholar] [CrossRef]
- Spitz, D.B.; Hebblewhite, M.; Stephenson, T.R.; German, D.W. How plastic is migratory behavior? Quantifying elevational movement in a partially migratory alpine ungulate, the Sierra Nevada bighorn sheep (Ovis canadensis sierrae). Can. J. Zool. 2018, 96, 1385–1394. [Google Scholar] [CrossRef] [Green Version]
- Morrison, T.A.; Merkle, J.A.; Hopcraft, J.G.C.; Aikens, E.O.; Beck, J.L.; Boone, R.B.; Courtemanch, A.B.; Dwinnell, S.P.; Fairbanks, W.S.; Griffith, B.; et al. Drivers of site fidelity in ungulates. J. Anim. Ecol. 2021, 90, 955–966. [Google Scholar] [CrossRef]
- Marshall, G.; Jonker, L. An introduction to descriptive statistics: A review and practical guide. Radiography 2010, 16, e1–e7. [Google Scholar] [CrossRef]
- Anchorena, A. Descripción Gráfica de la Patagonia y Valles Andinos; Compañia Sudamericana de Billetes de Banco: Buenos Aires, Argentina, 1902. [Google Scholar]
- Onelli, C. El huemul. Su patria: Su vida. Rev. Jardín Zool. Buenos Aires 1905, 1, 370–374. [Google Scholar]
- Bahre, C.J. Destruction of the Natural Vegetation of North-Central Chile; University of California Publications in Geography: Berkeley, CA, USA, 1979; Volume 23, pp. 1–117. ISBN 0-520-09594-4. [Google Scholar]
- Saavedra, B.; Simonetti, J.A. Archaeological evidence of Pudu pudu (Cervidae) in central Chile. Z. Saeugetierkunde 1991, 56, 252–253. [Google Scholar]
- Ale, A. A social economic formation of hunter-gatherers in the semiarid northern Chile: A revaluation of San Pedro Viejo of Pichasca site. Zaranda Ideas 2014, 11, 67–88. [Google Scholar]
- Merow, C.; Wilson, A.M.; Jetz, W. Integrating occurrence data and expert maps for improved species range predictions. Glob. Ecol. Biogeogr. 2017, 26, 243–258. [Google Scholar] [CrossRef]
- Travaini, A.; Zapata, S.C.; Bustamante, J.; Pedrana, J.; Zanón, J.I.; Rodríguez, A. Guanaco abundance and monitoring in Southern Patagonia: Distance sampling reveals substantially greater numbers than previously reported. Zool. Stud. 2015, 54, 23. [Google Scholar] [CrossRef] [Green Version]
- De Gea, G. El Ganado Lanar en la Argentina, 2nd ed.; Universidad Nacional de Río Cuarto: Río Cuarto, Argentina, 2007; ISBN 978-950-665-448-1. [Google Scholar]
- Uboldi, J.A.; Angeles, G.R.; Gentili, J.O.; Geraldi, A.M.; Melo, W.D.; Carbone, M.E. Geotecnologías del sur Argentino. Casos de Estudio; Departamento de Geografía y Turismo (TIG), Universidad Nacional del Sur: Bahía Blanca, Argentina, 2014. [Google Scholar]
- Flueck, W.T. Nutrition as an etiological factor causing diseases in endangered huemul deer. BMC Res. Notes 2020, 13, 276. [Google Scholar] [CrossRef]
- Merkle, J.A.; Sawyer, H.; Monteith, K.L.; Dwinnell, S.P.; Fralick, G.L.; Kauffman, M.J. Spatial memory shapes migration and its benefits: Evidence from a large herbivore. Ecol. Lett. 2019, 22, 1797–1805. [Google Scholar] [CrossRef]
- Aikens, E.O.; Kauffman, M.J.; Merkle, J.A.; Dwinnell, S.P.H.; Fralick, G.L.; Monteith, K.L. The greenscape shapes surfing of resource waves in a large migratory herbivore. Ecol. Lett. 2017, 20, 741–750. [Google Scholar] [CrossRef]
- Flueck, W.T. The effect of selenium on reproduction of black-tailed deer (Odocoileus hemionus columbianus) in Shasta County, California. Ph.D Thesis, University of California, Davis, CA, USA, 1989. [Google Scholar]
- Kauffman, M.J.; Copeland, H.E.; Berg, J.; Bergen, S.; Cole, E.; Cuzzocreo, M.; Dewey, S.; Fattebert, J.; Gagnon, J.; Gelzer, E.; et al. Ungulate Migrations of the Western United States; Report 2020–5101; U.S. Geological Survey Scientific Investigations: Reston, VA, USA, 2020; Volume 1. [Google Scholar]
- Gogan, P.J.P.; Klaver, R.W.; Olexa, E.M. Northern Yellowstone Mule Deer Seasonal Movement, Habitat Selection, and Survival Patterns. West. North Am. Nat. 2019, 79, 403–427. [Google Scholar] [CrossRef]
- Van de Kerk, M.; Larsen, R.T.; Olson, D.D.; Hersey, K.R.; McMillan, B.R. Variation in movement patterns of mule deer: Have we oversimplified migration? Mov. Ecol. 2021, 9, 44. [Google Scholar] [CrossRef] [PubMed]
- Haller, H. Der Rothirsch im Schweizerischen Nationalpark und dessen Umgebung. Eine alpine Population von Cervus elaphus zeitlich und räumlich dokumentiert. Natl. Forsch. Schweiz 2002, 91, 1–144. [Google Scholar]
- Howe, R.W.; Davis, G.J.; Mosca, V. The demographic significance of ‘sink’ populations. Biol. Conserv. 1990, 57, 239–255. [Google Scholar] [CrossRef]
- Remes, V. How can maladaptive habitat choice generate source-sink population dynamics? Oikos 2000, 91, 579–582. [Google Scholar] [CrossRef]
- Braunisch, V.; Bollmann, K.; Graf, R.F.; Hirzel, A.H. Living on the edge. Modelling habitat suitability for species at the edge of their fundamental niche. Ecol. Model. 2008, 214, 153–167. [Google Scholar] [CrossRef]
- Goss, R.J. Deer Antlers: Regeneration, Function and Evolution; Academic Press: New York, NY, USA, 1983. [Google Scholar]
- Tonko, J. Kawesqar travel narratives. Onomazein 2008, 18, 11–47. [Google Scholar]
- Anonymous. Excursión del gobernador del Chubut. Caras Caretas 1904, 7, 58. [Google Scholar]
- Moreno, F.P. Apuntes preliminares sobre una excursion a los territorios del Neuquén, Río Negro, Chubut y Santa Cruz. Rev. Mus. La Plata 1898, 8, 200–459. [Google Scholar]
- Steffen, H. Viajes de exploracion: Estudio en la Patagonia occidental 1892–1902. Anal. Univ. Chile 1910, 2, 1–419. [Google Scholar]
- Jiménez, J.; Guineo, G.; Corti, P.; Smith, J.A.; Flueck, W.T.; Vila, A.; Gizejewski, Z.; Gill, R.; McShea, B.; Geist, V. Hippocamelus bisulcus. In IUCN Red List of Threatened Species; IUCN: Gland, Switzerland, 2008. [Google Scholar]
- Curran, S.C. Expanding ecomorphological methods: Geometric morphometric analysis of Cervidae post-crania. J. Archaeol. Sci. 2012, 39, 1172–1182. [Google Scholar] [CrossRef]
- Curran, S.C. Exploring Eucladoceros ecomorphology using geometric morphometrics. Anat. Rec. 2015, 298, 291–313. [Google Scholar] [CrossRef]
- Flueck, W.T.; Smith-Flueck, J.M. Osteological comparisons of appendicular skeletons: A case study on Patagonian huemul deer and its implications for conservation. Anim. Prod. Sci. 2011, 51, 327–339. [Google Scholar] [CrossRef] [Green Version]
- Flueck, W.T. Functional limb anatomy in a refugee species: The endangered Patagonian huemul deer (Hippocamelus bisulcus). Anat. Histol. Embryol. 2021, 50, 411–416. [Google Scholar] [CrossRef]
- Barberena, R.; Méndez, C.; Mena, F.; Reyes, O. Endangered species, archaeology, and stable isotopes: Huemul (Hippocamelus bisulcus) isotopic ecology in central-western Patagonia (South America). J. Archaeol. Sci. 2011, 38, 2313–2323. [Google Scholar] [CrossRef]
- Caro, T. The adaptive significance of coloration in mammals. BioScience 2005, 55, 125–136. [Google Scholar] [CrossRef] [Green Version]
- Webb, S.D. Evolutionary history of New World Cervidae. In Antelopes, Deer, and Relatives; Vrba, E.S., Schaller, G.B., Eds.; Yale University Press: New York, NY, USA, 2000; pp. 38–64. [Google Scholar]
- Prichard, H.H. Field notes upon some of the larger mammals of Patagonia made between September 1900 and June 1901. Proc. Zool. Soc. Lond. 1902, 1, 272–277. [Google Scholar]
- Giai, A.G. Huemul, inofensivo venado de las soledades cordilleranas de la Patagonia. Chacra 1936, 6, 99–101. [Google Scholar]
- Grzimek, B. Grzimeks Tierleben: Enzyklopädie des Tierreichs—Säugetiere 3; Neue Schweizer Bibliothek: Zurich, Switzerland, 1973. [Google Scholar]
- Bürger, O. Aus der Wildnis des Huemul. Erlebnisse und Abenteuer unter den Kolonisten und Indianern Chiles; Verlag Deutsche Buchwerkstätten: Dresden, Germany, 1924. [Google Scholar]
- Goni, R.A.; Belardi, J.B.; Re, A.; Nuevo Delaunay, A.; Molinari, R.L.; Ferraro, L. Los grabados de la meseta del lago Strobel (Patagonia argentina) desde una perspectiva regional. In Actas del Primer Simposio Nacional de Arte Rupestre; Cusco Nov. 2004; Institut Francais d’Etudes Andines: Lima, Peru, 2007; pp. 427–438. [Google Scholar]
- Risenhoover, K.L.; Bailey, J.A.; Wakelyn, L.A. Assessing the Rocky Mountain Bighorn Sheep Management Problem. Wildl. Soc. Bull. 1988, 16, 346–352. [Google Scholar]
- Nagaike, T. Bark Stripping by Deer Was More Intensive on New Recruits than on Advanced Regenerants in a Subalpine Forest. Forests 2020, 11, 490. [Google Scholar] [CrossRef]
- Diaz, P.; Marqués, B.I.; Vila, A.R. Seasonal habitat use and selection of the endangered huemul deer (Hippocamelus bisulcus) in Patagonian Andes. Mammalia 2013, 77, 371–380. [Google Scholar] [CrossRef]
- Gill, R.; Saucedo, C.; Aldridge, D.; Morgan, G. Ranging behavior of huemul in relation to habitat and landscape. J. Zool. 2008, 274, 254–260. [Google Scholar] [CrossRef]
- Guineo, O.; Guineo Garay, R.; Garay, G. Conociendo al Huemul de Torres del Paine; La Prensa Austral: Punta Arenas, Chile, 2008. [Google Scholar]
- Garay, G.; Ortega, I.M.; Guineo, O. Social ecology of the huemul at Torres Del Paine National Park, Chile. Anal. Inst. Patagon. 2016, 44, 25–38. [Google Scholar] [CrossRef] [Green Version]
- Webb, S.L.; Dzialak, M.R.; Houchen, D.; Kosciuch, K.L.; Winstead, J.B. Spatial ecology of female Mule deer in an area proposed for wind energy development. West. N. Am. Nat. 2013, 73, 347–356. [Google Scholar] [CrossRef] [Green Version]
- Rau, J.A. Crecimiento poblacional de huemules del sur nativos y reintroducidos en la zona austral de Chile. In 4ta Reunión Chileno-Argentina sobre Estrategias de Conservación del Huemul; Acosta-Jamett, G., Ed.; CONAF and CODEFF: Santiago, Chile, 2003; pp. 43–45. [Google Scholar]
- Flueck, W.T.; Smith-Flueck, J.M. Troubling disease syndrome in endangered live Patagonian huemul deer (Hippocamelus bisulcus) from the Protected Park Shoonem: Unusually high prevalence of osteopathology. BMC Res. Notes 2017, 10, 739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clapperton, C.M. The glaciation of the Andes. Quat. Sci. Rev. 1983, 2, 83–155. [Google Scholar] [CrossRef]
- Flueck, W.T.; Smith-Flueck, J.M. Huemul heresies: Beliefs in search of supporting data. 2. Biological and ecological considerations. Anim. Prod. Sci. 2012, 52, 694–706. [Google Scholar] [CrossRef]
- Flueck, W.T. Elusive cranial lesions severely afflicting young endangered Patagonian huemul deer. BMC Res. Notes 2018, 11, 638. [Google Scholar] [CrossRef]
- Smith-Flueck, J.M.; Barrio, J.; Ferreyra, N.; Nuñez, A.; Tomas, N.; Guzman, J.; Flueck, W.T.; Hinojosa, A.; Vidal, F.; Garay, G.; et al. Advances in ecology and conservation of Hippocamelus species in South America. Anim. Prod. Sci. 2011, 51, 378–383. [Google Scholar] [CrossRef] [Green Version]
- Franklin, W.L. Biology, ecology, and relationship to man of the South American camelids. In Mammalian Biology in South America; Mares, M.A., Genoways, H.H., Eds.; Pymatuning Laboratory of Ecology, University Pittsburgh: Linesville, PA, USA, 1982; Volume 6, pp. 457–489. [Google Scholar]
- Mueller, T.; Olson, K.A.; Dressler, G.; Leimgruber, P.; Fuller, T.K.; Nicolson, C.; Novaro, A.J.; Bolgeri, M.J.; Wattles, D.; DeStefano, S.; et al. How landscape dynamics link individual- to population-level movement patterns: A multispecies comparison of ungulate relocation data. Glob. Ecol. Biogeogr. 2011, 20, 683–694. [Google Scholar] [CrossRef]
- Gelin, M.L.; Branch, L.C.; Thornton, D.H.; Novaro, A.J.; Gould, M.J.; Caragiulo, A. Response of pumas (Puma concolor) to migration of their primary prey in Patagonia. PLoS ONE 2017, 12, e0188877. [Google Scholar] [CrossRef] [Green Version]
- Franklin, W.L.; Bass, F.; Bonacic, C.F.; Cunazza, C.; Soto, N. Management of Patagonian guanaco in the grazing agroecosystem of southern Chile. Wildl. Soc. Bull. 1997, 25, 65–73. [Google Scholar]
- Bonavia, D. The South American Camelids: An Expanded and Corrected Edition; Monograph 64; Cotsen Institute of Archaeology Press, UCLA: Los Angeles, CA, USA, 2009; Available online: https://escholarship.org/uc/item/7xs9j2zs (accessed on 4 April 2022).
- Schroeder, N.M.; Matteucci, S.D.; Moreno, P.G.; Gregorio, P.; Ovejero, R.; Taraborelli, P.; Carmanchahi, P.D. Spatial and Seasonal Dynamic of Abundance and Distribution of Guanaco and Livestock: Insights from Using Density Surface and Null Models. PLoS ONE 2014, 9, e85960. [Google Scholar]
- Ladio, A.H.; Lozada, M. Summer cattle transhumance and wild edible plant gathering in a Mapuche community of northwestern Patagonia. Hum. Ecol. 2004, 32, 225–240. [Google Scholar] [CrossRef]
- Poole, K.G.; Mowat, G. Winter habitat relationships of deer and elk in the temperate interior mountains of British Columbia. Wildl. Soc. Bull. 2005, 33, 1288–1302. [Google Scholar] [CrossRef]
- Myburgh, J.; McGowan, K.; Davis, A. Veterinary Geology. In Practical Applications of Medical Geology; Siegel, M., Selinus, O., Finkelman, R., Eds.; Springer: Cham, Switzerland, 2021. [Google Scholar]
- Flueck, W.T. Consideraciones acerca de la calidad nutritiva de hábitat, hábitat óptimo, y evaluación de hábitat para huemul. In 4ta Reunión Chileno-Argentina Sobre Estrategias de Conservación del Huemul; Acosta-Jamett, G., Ed.; CONAF and CODEFF: Santiago, Chile, 2003; pp. 30–34. [Google Scholar]
- Landete-Castillejos, T.; Molina-Quilez, I.; Estevez, J.A.; Ceacero, F.; Garcia, A.J.; Gallego, L. Alternative hypothesis for the origin of osteoporosis: The role of Mn. Front. Biosci. Elite Ed. 2012, 4, 1385–1390. [Google Scholar] [CrossRef]
- Gambín, P.; Serrano, M.P.; Gallego, L.; García, A.; Cappelli, J.; Ceacero, F.; Landete-Castillejos, T. Does Cu supplementation affect the mechanical and structural properties and mineral content of red deer antler bone tissue? Animal 2017, 11, 1312–1320. [Google Scholar] [CrossRef] [Green Version]
- Bartlam-Brooks, H.L.; Beck, P.S.; Bohrer, G.; Harris, S. In search of greener pastures: Using satellite images to predict the effects of environmental change on zebra migration. J. Geophys. Res. Biogeosci. 2013, 118, 1427–1437. [Google Scholar] [CrossRef] [Green Version]
- Teitelbaum, C.S.; Fagan, W.F.; Fleming, C.H.; Dressler, G.; Calabrese, J.M.; Leimgruber, P.; Mueller, T. How far to go? Determinants of migration distance in land mammals. Ecol. Lett. 2015, 18, 545–552. [Google Scholar] [CrossRef]
- Monsarrat, S.; Jarvie, S.; Svenning, J.C. Anthropocene refugia: Integrating history and predictive modelling to assess the space available for biodiversity in a human-dominated world. Philos. Trans. R. Soc. B 2019, 374, 20190219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soga, M.; Gaston, K.J. Shifting baseline syndrome: Causes, consequences and implications. Front. Ecol. Environ. 2018, 16, 222–230. [Google Scholar] [CrossRef] [Green Version]
- Bar-Ilan, J.; Halevi, G. Retracted articles—The scientific version of fake news. In The Psychology of Fake News: Accepting, Sharing, and Correcting Misinformation; Greifeneder, R., Jaffé, M.E., Newman, E.J., Schwarz, N., Eds.; Taylor & Francis Group: New York, NY, USA, 2021. [Google Scholar]
- Novillo, A.; Ovejero, A.J.A.; Cristobal, L.; Ojeda, R.A. Alpine Mammals of South America. In Encyclopedia of the World’s Biomes; Goldstein, M.I., DellaSala, D.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 441–460. [Google Scholar]
- Cromsigt, J.P.G.M.; Kerley, G.I.H.; Kowalczyk, R. The difficulty of using species distribution modelling for the conservation of refugee species—The example of European bison. Div. Distrib. 2012, 18, 1253–1257. [Google Scholar] [CrossRef] [Green Version]
- Lea, J.M.D.; Kerley, G.I.H.; Hrabar, H.; Barry, T.J.; Shultz, S. Recognition and management of ecological refugees: A case study of the Cape mountain zebra. Biol. Conserv. 2016, 203, 207–215. [Google Scholar] [CrossRef]
- Kauffman, M.J.; Cagnacci, F.; Chamaillé-Jammes, S.; Hebblewhite, M.; Hopcraft, J.G.C.; Merkle, J.A.; Mueller, T.; Mysterud, A.; Peters, W.; Roettger, C.; et al. Mapping out a future for ungulate migrations. Limited mapping of migrations hampers conservation. Science 2021, 372, 566–569. [Google Scholar] [CrossRef]
- Lopez Alfonsin, M.A.; Bucetto, M.S. Endangered species and mechanisms for the recovery and conservation of biodiversity: A study on the viability of mechanisms and bureaucratic obstacles. LEX 2019, 17, 297–324. [Google Scholar]
- Doughty, C.E.; Roman, J.; Faurby, S.; Wolf, A.; Haque, A.; Bakker, E.S.; Malhi, Y.; Dunning, J.B.; Svenning, J.C. Global nutrient transport in a world of giants. Proc. Nat. Acad. Sci. USA 2018, 113, 868–873. [Google Scholar] [CrossRef] [Green Version]
- Monteith, K.L.; Bleich, V.C.; Stephenson, T.R.; Pierce, B.M.; Conner, M.M.; Kie, J.G.; Bowyer, R.T. Life-history characteristics of mule deer: Effects of nutrition in a variable environment. Wildl. Monogr. 2014, 186, 1–62. [Google Scholar] [CrossRef]
- Allred, W.J. Re-establishment of seasonal elk migration through transplanting. N. Am. Wildl. Conf. 1950, 15, 597–611. [Google Scholar]
- Stüwe, M.; Nievergelt, B. Recovery of Alpine ibex from near extinction: The result of effective protection, captive breeding, and reintroductions. Appl. Anim. Behav. Sci. 1991, 29, 379–387. [Google Scholar] [CrossRef]
- Gruell, G.E.; Papez, N.J. Movements of mule deer in northeastern Nevada. J. Wildl. Manag. 1963, 27, 414–427. [Google Scholar] [CrossRef]
- Haywood, D.D.; Brown, R.L.; Smith, R.H.; McCulloch, C.Y. Migration Patterns and Habitat Utilization by Kaibab Mule Deer; Arizona Game & Fish Department Research Branch: Phoenix, AZ, USA, 1987; pp. 1–44. [Google Scholar]
- Brown, C.G. Movement and migration patterns of mule deer in southeastern Idaho. J. Wildl. Manag. 1992, 56, 246–253. [Google Scholar] [CrossRef]
- Brooks, T.M.; Pimm, S.L.; Akçakaya, H.R.; Buchanan, G.M.; Buthchart, S.H.M.; Foden, W.; Hilton-Taylor, C.; Hoffmann, M.; Jenkins, C.N.; Joppa, L.; et al. Measuring terrestrial area of habitat (AOH) and its utility for the IUCN Red List. Trends Ecol. Evol. 2019, 24, 977–986. [Google Scholar] [CrossRef] [Green Version]
- Kerley, G.I.H.; te Beest, M.; Cromsigt, J.P.G.M.; Pauly, D.; Shultz, S. The Protected Area Paradox and refugee species: The giant panda and baselines shifted towards conserving species in marginal habitats. Conserv. Sci. Pract. 2020, 2, e203. [Google Scholar] [CrossRef]
- Adams, A.W. Migration. In Elk of North America. Ecology and Management; Thomas, J.W., Toweill, D.E., Eds.; Stackpole Books: Harrisburg, PA, USA, 1982; pp. 301–321. [Google Scholar]
- Agassiz, L. The 1871-1872 Hassler expedition. In Museum of Comparative Zoology; Occurrence Dataset, Version 162.229; Morris, P.J., Ed.; Harvard University: Cambridge, MA, USA, 1872. [Google Scholar]
- Aldenderfer, M.S. Montane Foragers: Asana and the South-Central Andean Archaic; University of Iowa Press: Iowa, IA, USA, 1998. [Google Scholar]
- Andersen, R. Habitat deterioration and the migratory behavior of moose (Alces alces L.) in Norway. J. Appl. Ecol. 1991, 28, 102–108. [Google Scholar] [CrossRef]
- Agencia CTyS-UNLaM/DICYT. Descubren los Restos Fósiles de Seis Ciervos Prehistóricos. 2021. Available online: www.dicyt.com (accessed on 7 October 2021).
- Armesto, J.J.; Manuschevich, D.; Mora, A.; Smith-Ramireza, C.; Rozzi, R.; Abarzúa, A.M.; Marquet, P.A. From the Holocene to the Anthropocene: A historical framework for land cover change in southwestern South America in the past 15,000 years. Land Use Policy 2010, 27, 148–160. [Google Scholar] [CrossRef]
- Aschero, C.A. Las escenas de caza en Cueva de las Manos: Una perspectiva regional (Santa Cruz, Argentina). In IFRAO Congress—Symposium: Pleistocene Art of the Americas (Pre-Acts) Austral; Talleres Gráficos Guillermo Kraft Ltda: Buenos Aires, Argentina, 2010. [Google Scholar]
- Avital, E.; Jablonka, E. Animal Traditions; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Battin, J. When good animals love bad habitats: Ecological traps and the conservation of animal populations. Cons. Biol. 2004, 18, 1482–1491. [Google Scholar] [CrossRef]
- Baumann, M.; Babota, C.; Schibler, J. Native or naturalized? Validating alpine chamois habitat models with archaeozoological data. Ecol. Appl. 2005, 15, 1096–1110. [Google Scholar] [CrossRef] [Green Version]
- Behm, E. Reise im südwestlichen Patagonien von J.T. Rogers und E. Ibar, 1877, nebst den Tagebüchern von A. de Viedma 1782 und J.H. Gardiner 1867. Petermanns Geogr. Mitteilungen 1880, 26, 47–64. [Google Scholar]
- Berger, J.; Wangchuk, T.; Briceno, C.; Vila, A.; Lambert, J.E. Disassembled Food Webs and Messy Projections: Modern Ungulate Communities in the Face of Unabating Human Population Growth. Front. Ecol. Evol. 2020, 8, 128. [Google Scholar] [CrossRef]
- Berger, J.; Steven, L.; Cain, S.L.; Berger, K.M. Connecting the dots: An invariant migration corridor links the Holocene to the present. Biol. Lett. 2006, 2, 528–531. [Google Scholar] [CrossRef]
- Brandborg, S.M. Life History and Management of the Mountain Goat in Idaho; Wildlife Bulletin No. 2; Department of Fish and Game: Boise, ID, USA, 1955; 142p.
- Bubenik, G.A.; Bubenik, A.B. Horns, Pronghorns, and Antlers; Springer: New York, NY, USA, 1990. [Google Scholar]
- Burmeister, C.V. Memoria Sobre el Territorio de Santa Cruz; Imprenta La Nación, Ministerio de Agricultura de la Republica Argentina: Buenos Aires, Argentina, 1901. [Google Scholar]
- Burmeister, H. The huemul. Nature 1873, 9, 82. [Google Scholar] [CrossRef]
- Burmeister, C.V. Nuevos datos sobre el territorio Patagonico de Santa Cruz. Rev. Museo Plata 1893, 4, 227–256, 338–352. [Google Scholar]
- Cabrera, A.; Yepes, J. Mamiferos Sudamericanos, 1st ed.; Compañia Argentina de Editores: Buenos Aires, Argentina, 1940; 370p. [Google Scholar]
- Carballo Marina, F.; Manzi, L.M.; Campan, P.A.; Belardi, J.B.; Tiberi, P.; Manero, A.; Saenz, J.L. Distribución del registro arqueológico en la cuenca del rio Gallegos (Santa Cruz): Linea de base y aporte a la preservación del patrimonio. In Arqueologia del Extremo sur del Continente Americano; Borrero, L.A., Franco, N., Eds.; Editorial Dunken: Buenos Aires, Argentina, 2008; pp. 175–225. [Google Scholar]
- Cardich, A.; Miotti, L. Recursos Faunisticos en la Economia de los Cazadores-Recolectores de Los Toldos (Provincia de Santa Cruz). Relaciones Soc. Argent. Antropol. 1983, 16, 269–273. [Google Scholar]
- Castellanos, A. Paleontología estratigráfica de los sedimentos neógenos de la Provincia de Córdoba. Publ Inst. Fisiogr. Geol. 1944, 23, 1–47. [Google Scholar]
- Church, G.E. A traveller in Patagonia. Nature 1903, 67, 321–322. [Google Scholar] [CrossRef]
- Clapperton, C.M. Nature of environmental changes in South America at the Last Glacial Maximum. Palaeogeogr. Paleoclimatol. Palaeoecol. 1993, 101, 189–208. [Google Scholar] [CrossRef]
- Claraz, M.G. Sur l’Equus bisulcus, de Molina. Revue et Magasin de Zoologie Pure et Apliquee 1864, 241–248. [Google Scholar]
- Coltorti, M.; Abbazzi, L.; Ferretti, M.P.; Iacumin, P.; Paredes Rios, F.; Pellegrini, M.; Pieruccini, P.; Rustioni, M.; Tito, G.; Rook, L. Last Glacial mammals in South America: A new scenario from the Tarija Basin (Bolivia). Naturwissenschaften 2007, 94, 288–299. [Google Scholar] [CrossRef]
- Conway, W. Act III in Patagonia: People and Wildlife; Island Press: Washington, DC, USA, 2005. [Google Scholar]
- Cox, G.E. Viaje a las Regiones Septentrionales de la Patagonia: 1862–1863; Imprenta Nacional: Santiago, Chile, 1863; 266p. [Google Scholar]
- Cruz, I.; Munoz, A.S.; Caracotche, M. A huemul (Hippocamelus bisulcus) antler artefact in archaeological deposits of the Atlantic coast. Implications for human mobility and species distribution. Magallania 2010, 38, 287–294. [Google Scholar] [CrossRef] [Green Version]
- da Silva, F.M.; da Silva Alves, R.; Franca Barreto, A.M.; Bezerra de Sá, F.; Borges Lins e Silva, A.C. A megafauna pleistocnica do estado de Pernambuco. Estudos Geologicos 2006, 16, 55–66. [Google Scholar]
- Dabbene, R. Sobre la existencia del huemul de Bolivia y Perú, Odocoileus (Hippocamelus) antisensis (Orb.) y del avestruz petiso, Rhea darwini Gould en el N.W. de la República Argentina. Anales del Museo Nacional Buenos Aires 1911, 14, 293–307. [Google Scholar]
- Dawilov. Coihue; Gmo. van Woerden & Cia: Buenos Aires, Argentina, 1926; 103p. [Google Scholar]
- de la Cruz, L. Descripción de la naturaleza de los terrenos que se comprenden en los Andes, poseídos por los Peguenches; y los demás espacios hasta el rio de Chadileubu. In Colección de Obras y Documentos Relativos a la Historia Antigua y Moderna de las Provincias del Río de la Plata; de Angelis, P., Ed.; Imprenta del Estado: Buenos Aires, Argentina, 1836; pp. 1–67. [Google Scholar]
- De Agostini, A.M. Andes Patagónicos: Viajes de Exploración a la Cordillera Patagónica, 1era Versión; Imprenta Gotelli: Buenos Aires, Argentina, 1941. [Google Scholar]
- De Agostini, A.M. Andes Patagónicos: Viajes de Exploración a la Cordillera Patagónica Austral; Talleres Gráficos Guillermo Kraft Ltda: Buenos Aires, Argentina, 1945; Volume 1, pp. 1–409. [Google Scholar]
- Diaz, N.I. Antecedentes sobre la historia natural de la taruca (Hippocamelus antisensis) y su rol en la economia Andina. Chungara 1995, 27, 45–55. [Google Scholar]
- Diaz, N.I. Changes in the range distribution of Hippocamelus bisulcus in Patagonia. Z. Säugetierkunde 1993, 58, 344–351. [Google Scholar]
- Díaz, N.I. The huemul (Hippocamelus bisulcus Molina, 1782): A historical perspective. In The Patagonian Huemul, a Mysterious Deer on the Brink of Extinction; Díaz, N.I., Smith-Flueck, J., Eds.; L.O.L.A.: Buenos Aires, Argentina, 2000; pp. 1–31. [Google Scholar]
- Díaz, N.I.; Prieto, A.; Bahamonde, G. Guanacos tímidos, huemules confiados: El límite occidental de los cazadores terrestres australes. Magallania 2007, 35, 133–138. [Google Scholar] [CrossRef] [Green Version]
- Eastman, C.R. Beginnings of American natural history. Amer. Museum J. 1915, 15, 349–355. [Google Scholar]
- Eisenberg, J.F. The contemporary Cervidae of Central and South America. In Antelopes, Deer, and Relatives; Vrba, E.S., Schaller, G.B., Eds.; Yale University Press: New York, NY, USA, 2000; pp. 189–202. [Google Scholar]
- Falkner, T. A Description of Patagonia and the Adjoining Parts of South America: Containing an Account of the Soil, Produce, the Religion, Government, and Some Particulars Relating to Falkland Islands; Hereford: London, UK, 1774; 144p. [Google Scholar]
- Fernandez, P.M.; Cruz, I.; Bautista Belardi, J.; de Nigris, M.; Muñoz, S. La explotación del huemul (Hippocamelus bisulcus, Molina 1782) en la Patagonia a lo largo del holoceno. Magallania 2016, 44, 187–209. [Google Scholar] [CrossRef] [Green Version]
- Fernández, O.A.; Busso, C.A. Arid and semi-arid rangelands: Two thirds of Argentina. In Proceedings from an International Workshop in Iceland; Arnalds, O., Archer, S., Eds.; Rala Report no. 200; Agricultural Research Institute: Reykjavik, Iceland, 1997; pp. 41–60. [Google Scholar]
- Fielder, P.C. Implications of selenium levels in Washington mountain goats, mule deer, and Rocky Mountain elk. Northwest Sci. 1986, 60, 15–20. [Google Scholar]
- Flint, R.F.; Fidalgo, F. Glacial Drift in the Eastern Argentine Andes between Latitude 41°10′ S. and Latitude 43°10′ S. Bull. Geol. Soc. America 1969, 80, 1043–1052. [Google Scholar] [CrossRef]
- Flueck, W.T. Spatio-temporal movements among red deer males, Cervus elaphus, introduced to Patagonia. In XXVIIth Congress of the International Union of Game Biologists; Pohlmeyer, K., Ed.; Hannover DSV-Verlag: Hamburg, Germany, 2005; pp. 330–332. [Google Scholar]
- Flueck, W.T. Exotic deer in southern Latin America: What do we know about impacts on native deer and on ecosystems? Biol. Invasions 2010, 12, 1909–1922. [Google Scholar] [CrossRef] [Green Version]
- Flueck, W.T. Osteopathology and selenium deficiency co-occurring in a population of endangered Patagonian huemul (Hippocamelus bisulcus). BMC Res. Notes 2015, 8, 330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flueck, W.T.; Smith-Flueck, J.M. Über das in Argentinien angesiedelte Rotwild (Cervus elaphus L., 1758): Verbreitung und Tendenzen. Zeits. Jagdwiss. 1993, 39, 153–160. [Google Scholar] [CrossRef]
- Flueck, W.T.; Smith-Flueck, J.M. Predicaments of endangered huemul deer, Hippocamelus bisulcus, in Argentina: A review. Europ. J. Wildl. Res. 2006, 52, 69–80. [Google Scholar] [CrossRef]
- Flueck, W.T.; Smith-Flueck, J.M. Huemul heresies: Beliefs in search of supporting data. 1. Historical and zooarcheological considerations. Anim. Prod. Sci. 2012, 52, 685–693. [Google Scholar] [CrossRef] [Green Version]
- Flueck, W.T.; Smith-Flueck, J.M. Radio marking the first group of endangered Patagonian huemul deer in Argentina. J. Neotrop. Mammal. 2018, 25, 461–465. [Google Scholar] [CrossRef]
- Flueck, W.T.; Smith-Flueck, J.M.; Mincher, B.J.; Winkel, L.H.E. Soil selenium levels corroborate direct evidence of selenium deficiency in endangered Patagonian huemul deer (Hippocamelus bisulcus). In Proceedings of the 8th International Deer Biology Congress; Ma, J., Zhang, M., Halbrook, R., Liu, B., Zhang, W., Eds.; Northeast Forestry University: Harbin, China, 2014; pp. 52–53. [Google Scholar]
- Frailey, D.; Campbell, K.E.; Wolff, R.G. Additions to the knowledge of Hippocamelus, Ctenomys, and Myocastor from the middle Pleistocene of the Tarija basin, Bolivia. Occas. Papers Museum Nat. Hist. Univ. Kansas 1980, 85, 1–14. [Google Scholar]
- Gay, C. Historia Fisica y Politica de Chile: Zoologia; Museo de Historia Natural de Santiago: Santiago, Chile, 1847; 495p. [Google Scholar]
- Gazzolo, C. Botanical composition of taruka (Hippocamelus antisensis) diet during rainy season in Huascaran national park, Peru. In Advances in Deer Biology; Bartos, L., Dusek, A., Kotrba, R., Bartosova, J., Eds.; Research Institute of Animal Production: Praha, Czech Republic, 2006; p. 216. [Google Scholar]
- Geist, V. Deer of the World; Stackpole Books: Pennsylvania, PA, USA, 1998; 421p. [Google Scholar]
- Gigoux, E.E. El huemul. Rev. Chilena Hist. Nat. 1929, 23, 573–582. [Google Scholar]
- Goni, R.A. Arqueologia de momentos tardios en el Parque Nacional Perito Moreno (Santa Cruz, Argentina). In Precirculados del IX Congreso Nacional de Arqueologia Argentina; Universidad de Buenos Aires: Buenos Aires, Argentina, 1988; pp. 140–151. [Google Scholar]
- Gonzalez, V.; Tapia, V. Manual bovino de carne. Boletín INIA 2017, 4, 1–173. [Google Scholar]
- Grosse, A. El huemul—Ciervo de los Andes y emblema del escudo Chileno. Condor 1949, 12, 10–12. [Google Scholar]
- Guérin, C.; Faure, M. The Cervidae (Mammalia, Artiodactyla) of the Upper Pleistocene/Lower Holocene deposits of the Serra da Capivara National Park Region (Piauý, Brazil). Geobios 2009, 42, 169–195. [Google Scholar] [CrossRef]
- Günther, A. Comments about Sclater and Cervus chilensis. In Proceedings of the Scientific Meetings of the Zoological Society of London; Messrs. Longmans, Green, Reader and Dyer: London, UK, 1875; pp. 44–46. [Google Scholar]
- Hatcher, J.B. Reports of the Princeton University Expeditions to Patagonia, 1896–1899. Vol. I: Narrative of the Expeditions. Geography of Southern Patagonia; E. Schweizerbart’sche Verlagshandlung: Stuttgart, Germany, 1903; 314p. [Google Scholar]
- Hauman, L. Étude phytogéographique de la Patagonie. Bulletin de la Société Royale de Botanique de Belgique 1926, 58, 105–179. [Google Scholar]
- Hershkovitz, P. The recent mammals of the neotropical region: A zoogeographic and ecological review. In Evolution, Mammals, and Southern Continents; Keast, A., Erk, F.C., Glass, B., Eds.; State University New York Press: New York, NY, USA, 1972; pp. 311–431. [Google Scholar]
- Hershkovitz, P. The recent mammals of the Neotropical region: A zoogeographic and ecological review. Quart. Rev. Biol. 1969, 44, 1–70. [Google Scholar] [CrossRef]
- Hoffstetter, R. La faune pleistocene de Tarija (Bolivie). Note preliminaire. Bulletin Muséum National d’Histoire Naturelle 1963, 35, 194–203. [Google Scholar]
- Honess, R.F.; Frost, N.M. A Wyoming bighorn sheep study. Wyoming Game Fish Depart. Bull. 1942, 1, 1–127. [Google Scholar]
- Horkheimer, H. Nahrung und Nahrungsgewinnung im Vorspanischen Peru; Colloquim Verlag: Berlin, Germany, 1960. [Google Scholar]
- Housse, P.R. Animales Salvajes de Chile en su Clasificación Moderna: Su Vida y Sus Costumbres; Ediciones de la Universidad de Chile: Santiago, Chile, 1953; 189p. [Google Scholar]
- Ibar Bruce, J. Aisen, Hombres y Naturaleza; Imprenta de la Armada: Valparaiso, Chile, 1973; pp. 1–164. [Google Scholar]
- Iglesias, R.E. El huemul. Montaña 1965, 7, 26–28. [Google Scholar]
- Jakopak, R.P.; LaSharr, T.N.; Dwinnell, S.P.H.; Fralick, G.L.; Monteith, K.L. Rapid acquisition of memory in a complex landscape by a mule deer. Ecology 2019, 100, e02854. [Google Scholar] [CrossRef]
- Johnson, L. Informe sobre una prospección arqueológica en magallanes. Ans. Inst. Pat. Punta Arenas 1976, 7, 87–94. [Google Scholar]
- Kolliker Frers, A. Das Waidwerk und die autochthonen Cerviden in Argentinien. In Parque Diana; Vogel, C.A., Ed.; Stefan Schwarz Verlag: München, Germany, 1969; pp. 25–31. [Google Scholar]
- Koprowski, J.L.; Krausman, P.R. International Wildlife Management: Conservation Challenges in a Changing World; Johns Hopkins University Press: Baltimore, MD, USA, 2019; 248p. [Google Scholar]
- Krieg, H. Biologische Reisestudien in Südamerika. V. Die chilenischen Hirsche. Zeits. Morphol. Ökol. Tiere 1925, 4, 585–597. [Google Scholar] [CrossRef]
- Krieg, H. Als Zoologe in Steppen und Wäldern Patagoniens; Bayerischer Landwirtschaftsverlag: Muenchen, Germany, 1940; 197p. [Google Scholar]
- Lacroix, F. Historia de la Patagonia, Tierra de Fuego, e Islas Malvinas; Imprenta del Liberal Barcelones: Barcelona, Spain, 1841. [Google Scholar]
- Laliberte, A.S.; Ripple, W.J. Range contractions of North American carnivores and ungulates. BioScience 2004, 54, 123–138. [Google Scholar] [CrossRef] [Green Version]
- Laming-Emperaire, A.; Lavallée, D.; Humbert, R. Le site de Marazzi en Terre de Feu. Objets et Mondes 1972, 12, 225–244. [Google Scholar]
- Latcham, R.E. Expedicion cientifica Macqueen al Aysen. Boletin del Museo Nacional 1935, 14, 7–31. [Google Scholar]
- Leopold, A.S.; Cain, S.A.; Cottam, C.; Gabrielson, I.N.; Kimball, T.L. Wildlife Management in the National Parks. Am. For. 1963, 4, 32–35, 61–63. [Google Scholar]
- Liebermann, J. Sobre la historia natural del huemul. Anal. Acad. Argent. Geogr. 1962, 6, 157–168. [Google Scholar]
- Lingle, S.; Wilson, W.F. Detection and avoidance of predators in white-tailed deer (Odocoileus virginianus) and mule deer (O. hemionus). Ethology 2001, 107, 125–147. [Google Scholar] [CrossRef] [Green Version]
- Lista, R. La Tierra del Fuego y sus habitantes. Boletin del Instituto Geografico Argentino 1881, 2, 109–114. [Google Scholar]
- Lydekker, R. The Deer of all Lands: A History of the Family Cervidae, Living and Extinct; R. Ward: London, UK, 1898; pp. 1–329. [Google Scholar]
- Lyman, R.L. Paleozoology in the service of conservation biology. Evol. Anthropol. 2006, 15, 11–19. [Google Scholar] [CrossRef]
- Lyras, G.A.; Giannakopoulou, A.; Lillis, T.; van der Geer, A.A.E. Paradise lost: Evidence for a devastating metabolic bone disease in an insular Pleistocene deer. Intern. J. Paleopathol. 2019, 24, 213–226. [Google Scholar] [CrossRef] [PubMed]
- MacDouall, J. Narratives of a Voyage to Patagonia and Terra del Fuego; Renshaw and Rush: London, UK, 1833; 320p. [Google Scholar]
- Machon, J.F.; Juarez, F.N. Patagonia 1892: Diario del Explorador Suizo Dr. Francisco Machón; Editorial Dunken: Buenos Aires, Argentina, 2013. [Google Scholar]
- Magalhaes, R.M.; Mello, M.G.; Bergqvist, L.P. Os cervidas pleistocenicos da regiao nordeste Brasileira. Anais da Academia Brasileira de Ciencias 1992, 64, 149–154. [Google Scholar]
- Magne de la Croix, P. El huemul. Caras y Caretas 1937, 40, 117. [Google Scholar]
- Mansur, M.E.; Piqué, R. Between the Forest and the Sea: Hunter-Gatherer Occupations in the Subantarctic Forests in Tierra del Fuego, Argentina. Arctic Anthropol. 2009, 46, 144–157. [Google Scholar] [CrossRef]
- Markgraf, V.; Kenny, R. Character of rapid vegetation and climate change during the late-Glacial in southernmost South America. In Past and Future Rapid Environmental Changes: Spatial and Evolutionary Responses to terrestrial Biota; Huntley, B., Ed.; Springer: Berlin, Germany, 1997; pp. 81–90. [Google Scholar]
- Marshall, L.G. Land Mammals and the Great American Interchange. Am. Sci. 1988, 76, 380–388. [Google Scholar]
- Massara Paletto, V.; Buono, G. Métodos de Evaluación de Pastizales en Patagonia Sur; Centro Regional Patagonia, Ediciones INTA: Buenos Aires, Argentina, 2020; 288p. [Google Scholar]
- Massone, M. Los paraderos tehuelches y proto-tehuelches en la costa del Estrecho de Magallanes. Anales del Instituto de la Patagonia 1984, 15, 27–42. [Google Scholar]
- McClure, M.F.; Bissonette, J.A.; Conover, M.R. Migratory strategies, fawn recruitment, and winter habitat use by urban and rural mule deer (Odocoileus hemionus). Europ. J. Wildl. Res. 2005, 51, 170–177. [Google Scholar] [CrossRef]
- Miller, S.; Rottman, J.; Taber, R.D. Dwindling and endangered ungulates of Chile: Vicugna, lama, Hippocamelus, and Pudu. Trans. N. Am. Wildl. Natural Res. Conf. 1973, 38, 55–67. [Google Scholar]
- Mincher, B.J.; Mionczynski, J.; Hnilicka, P.P.; Ball, R.D.; Houghton, T.X. Some aspects of geophagia in Wyoming big-horn sheep (Ovis canadensis). Europ. J. Wildl. Res. 2008, 54, 192–198. [Google Scholar] [CrossRef]
- Molina, J.I. The Geographical, Natural, and Civil History of Chili; Longman, Hurst, Rees, and Orme: London, UK, 1809; Volume 1. [Google Scholar]
- Moreira-Arce, D.; Pefiaranda, D.A.; Lopéz, R.; Stipicic, G.J.; Hidalgo-Hermoso, E.; Simonetti, J.A. Observations of a coastal population of huemul, Hippocamelus bisulcus (Artiodactyla: Cervidae) in Riesco lsland, Magallanes Region, Chile: A conservation opportunity. Mammalia 2021, 85, 291–295. [Google Scholar] [CrossRef]
- Morejohn, G.V.; Dailey, D.C. The identity and postcranial osteology of Odocoileus lucasi (Hay) 1927. Sierra Coll. Nat. Hist. Museum Bull. 2004, 1, 1–54. [Google Scholar]
- Moreno, F.P. Explorations in Patagonia. Geogr. J. 1899, 14, 241–269. [Google Scholar] [CrossRef] [Green Version]
- Moreno, P.I.; Villagran, C.; Marquet, P.A.; Marshall, L.G. Quaternary paleobiogeography of northern and central Chile. Rev. Chilena Hist. Nat. 1994, 67, 487–502. [Google Scholar]
- Moser, C.A. The Bighorn Sheep of Colorado: A Review of Colorado’s Bighorn Sheep Studies; Technical Publication No. 10; The Colorado Game and Fish Department: Denver, CO, USA, 1962; 49p. [Google Scholar]
- Musters, R.N. A year in Patagonia. J. Royal Geogr. Soc. London 1871, 41, 59–77. [Google Scholar] [CrossRef]
- Mysterud, A.; Loe, L.E.; Zimmermann, B.; Bischof, R.; Veiberg, V.; Meisingset, E. Partial migration in expanding red deer populations at northern latitudes—A role for density dependence? Oikos 2011, 120, 1817–1825. [Google Scholar] [CrossRef]
- Nelson, M.E.; Mech, L.D. Twenty-year home-range dynamics of a white-tailed deer matriline. Can. J. Zool. 1999, 77, 1128–1135. [Google Scholar] [CrossRef]
- Neveu-Lemaire, M.; Grandidier, G. Notes sur les Mammiféres des Hauts Plateaux de l’Amérique du Sud; Imprimerie Nationale: Paris, France, 1911; 127p. [Google Scholar]
- Onelli, C. Trepando los Andes; Compania Sud-Americana de Billetes de Banco: Buenos Aires, Argentina, 1904; 297p. [Google Scholar]
- Osgood, W.H. The journal of Wilfred Osgood: The Marshall Field Chilean Expedition of 1922-23. In Patterson, B.D. Field Museum of Natural History Bulletin 1983, 54, 28–33. [Google Scholar]
- Packard, F.M. An ecological study of the Bighorn sheep in Rocky Mountain National Park, Colorado. J. Mammal. 1946, 27, 3–28. [Google Scholar] [CrossRef]
- Paillan, J.T.; Tello, G.E. Los recursos naturales y culturales, 28 de Noviembre, Guer Aaike. In Santa Cruz: Su Importancia Turistica y Patrimonial; Informe Científico Técnico UNPA 4, ICT-UNPA-35-2012; Universidad Nacional de la Patagonia Austral: Rio Turbio, Argentina, 2012. [Google Scholar]
- Paula Couto, C. Paleontologia Brasileira (Mamíferos); Instituto Nacional do Livro: Rio de Janeiro, Brasil, 1953. [Google Scholar]
- Paula Couto, C. Tratado de Paleomastozoologia; Academia Brasileira de Ciencias: Rio de Janeiro, Brasil, 1979. [Google Scholar]
- Pefaur, J.; Hermosilla, W.; DiCastri, F.; Gonzalez, R.; Salinas, F. Estudio preliminar de mamíferos silvestres chilenos: Su distribución, valor económico e importancia zoonótica. Rev. Soc. Med. Vet. 1968, 18, 3–15. [Google Scholar]
- Pennant, T. History of Quadrupeds, 3rd ed.; B & J White: London, UK, 1793. [Google Scholar]
- Perez, A.E.; Batres, D.A. Los otros cazadores. Explotación de cérvidos en la Localidad Arqueológica Meliquina, Parque Nacional Lanin, República Argentina. In Zooarqueología hoy. Encuentros Hispano-Argentinos; Díez, J.C., Ed.; Universidad de Burgos: Burgos, Spain, 2008; pp. 89–107. [Google Scholar]
- Philippi, R.A. Über den Guemul von Molina. Archiv für Naturgeschichte 1857, 23, 135–136. [Google Scholar]
- Philippi, R.A. Zoología: Sinonimia del huemul. Anales de la Universidad de Chile 1873, 717–722. [Google Scholar]
- Philippi, R.A. El guemul de Chile. Anal. Museo Nac. Chile Primer Seccion Zool. 1892, 2, 1–9. [Google Scholar]
- Phoca-Cosmetatou, N. Site function and the ’ibex-site phenomenon’: Myth or reality? Oxford J. Archaeol. 2004, 23, 217–242. [Google Scholar] [CrossRef]
- Prichard, H.H. Through the Heart of Patagonia; D. Appleton and Co.: New York, NY, USA, 1902; 346p. [Google Scholar]
- Prichard, H.H. Hunting Camps in Wood and Wilderness; William Heinemann: London, UK, 1910; 274p. [Google Scholar]
- Prothero, D.R.; Foss, S.E. The Evolution of Artiodactyls; JHU Press: Baltimore, MD, USA, 2007. [Google Scholar]
- Pulliam, H.R. Sources, sinks, and population regulation. Am. Naturalist 1988, 132, 652–661. [Google Scholar] [CrossRef]
- Rabassa, J.; Coronato, A. Glaciations in Patagonia and Tierra del Fuego during the Ensenadan Stage/Age (Early Pleistocene-earliest Middle Pleistocene). Quaternary Intern. 2009, 210, 18–36. [Google Scholar] [CrossRef]
- Rabassa, J.; Coronato, A.; Martínez, O. Late Cenozoic glaciations in Patagonia and Tierra del Fuego: An updated review. Biol. J. Linnean Soc. 2011, 103, 316–335. [Google Scholar] [CrossRef] [Green Version]
- Ramirez Morales, F. Apuntes para una historia ecológica de Chile. Cuadernos de Historia 1991, 11, 149–196. [Google Scholar]
- Rasmussen, P.C. Geographic variation in morphology and allozymes of south american imperial shags. The Auk 1994, 111, 143–161. [Google Scholar] [CrossRef]
- Re, A.; Delaunay, A.N.; Ferraro, L. Grabados en la meseta del lago Strobel (provincia de Santa Cruz, Argentina), el sitio laguna del Faldeo Verde. Relaciones Soc. Argentina Antropol. 2005, 30, 245–256. [Google Scholar]
- Reichlen, H. Huemul in Fell’s Cave, Chile: Specimen MNHN-2M-MO-1988-211; Museum National d’Histoire Naturelle: Paris, France, 1959. [Google Scholar]
- Ren, J.Z.; Zhou, Z.Y.; Pan, B.; Chen, W. Selenium distribution in four grassland classes of China. In Selenium in Biology and Medicine; Comb, G.F., Spall-holz, J.E., Levander, O.A., Oldfield, J.E., Eds.; AVI Books: New York, NY, USA, 1987; pp. 769–774. [Google Scholar]
- Ringuelet, R.A. Temas de Ciencia Naturales; Museo de La Plata: La Plata, Argentina, 1946. [Google Scholar]
- Rosas, Y.M.; Peri, P.L.; Herrera, A.H.; Pastore, H.; Pastur, G.M. Modeling of potential habitat suitability of Hippocamelus bisulcus: Effectiveness of a protected areas network in Southern Patagonia. Ecol. Processes 2017, 6. [Google Scholar] [CrossRef]
- Roulin, M. Mémoire pour servir a l’histoire du tapir: Et description d’une espece nouvelle (le tapir pinchaque) appartenant aux hautes régions de la Cordillere des Andes. Mémoires des Savans Étrangers 1835, 6, 5–112. [Google Scholar]
- Rusconi, C. Animales Extinguidos de Mendoza y de la Argentina; Imprenta Oficial: Mendoza, Argentina, 1967. [Google Scholar]
- Ryan, S.J. The role of culture in conservation planning for small or endangered populations. Conserv. Biol. 2006, 20, 1321–1324. [Google Scholar] [CrossRef]
- Santos Gollan, J. Contribución al Conocimiento de los Mamíferos del Parque Nacional de Nahuel Huapi; Facultad de Ciencias Exactas y Naturales, Universidad de Buenos Aires: Buenos Aires, Argentina, 1946; 66p. [Google Scholar]
- Sawyer, H.; Middleton, A.D.; Hayes, M.M.; Kauffman, M.J.; Monteith, K.L. The extra mile: Ungulate migration distance alters the use of seasonal range and exposure to anthropogenic risk. Ecosphere 2016, 7, e01534. [Google Scholar] [CrossRef]
- Sclater, P.L. Remarks on Cervus chilensis and Cervus antisiensis. J. Nat. History Ser. 4 1873, 11, 213–214. [Google Scholar] [CrossRef] [Green Version]
- Sclater, P.L. On Cervus chilensis. Proc. Zool. Soc. Lond. 1875, 2, 44–47. [Google Scholar]
- Serret, A. Observaciones Preliminares de Huemul, Hippocamelus bisulcus, en el Lago Nansen del Parque Nacional Perito Moreno, Provincia Santa Cruz; Fundacion Vida Silvestre: Buenos Aires, Argentina, 1990; 23p. [Google Scholar]
- Sierralta, D. La microhistología de fecas para el estudio de dieta del huemul. In Huemul Ecology Research for Conservation Planning, Darwin Initiative ed.; Darwin Initiative: Cochrane, Chile, 2003. [Google Scholar]
- Siewert, C. Un viaje a Patagonia. Boletin Inst. Geogr. Argentino 1896, 17, 363–391. [Google Scholar]
- Silveira, M.J. Analisis e Interpretacion de los Restos Faunisticos de la Cueva Grande del Arroyo Feo. Relaciones Soc. Argent. Antropol. 1979, 13, 229–253. [Google Scholar]
- Simmonds, P.L. A Dictionary of Useful Animals and Their Products; E. & F.N. Spon: London, UK, 1883; 136p. [Google Scholar]
- Sinclaire, C. Daily life among the fishermen of the fog. In Fishermen of the fog: The Changos and Their Ancestors; Museo Chileno de Arte Precolombino: Santiago, Chile, 2009; pp. 41–48. [Google Scholar]
- Skottsberg, C. The wilds of Patagonia; Edward Arnold: London, UK, 1911; 336p. [Google Scholar]
- Stankowich, T. Tail-Flicking, Tail-Flagging, and Tail Position in Ungulates with Special Reference to Black-Tailed Deer. Ethology 2008, 114, 875–885. [Google Scholar] [CrossRef]
- Stankowich, T.; Coss, R.G. Effects of predator behavior and proximity on risk assessment by Columbian black-tailed deer. Behav. Ecol. 2006, 17, 246–254. [Google Scholar] [CrossRef]
- Steffen, H. Die Erforschung de Rio Puelo. Petermanns Geographischen Mitteilungen 1895, 41, 190–193. [Google Scholar]
- Steffen, H. Die chilenische Aisen Expedition. Verhandlungen der Gesellschaft für Erdkunde zu Berlin 1897, 24, 461–474. [Google Scholar]
- Steffen, H. Reisen in den Patagonischen Anden. Verhandlungen der Gesellschaft für Erdkunde zu Berlin 1900, 27, 194–220. [Google Scholar]
- Steward, J.H. Handbook of South American Indians. Volume 1. The Marginal Tribes; Smithsonian Institution: Washington, DC, USA, 1946. [Google Scholar]
- Stringham, S.F.; Rogers, L.L. Fear of Humans by Bears and Other Animals (Anthropophobia): How Much is Natural? J. Behav. 2017, 2, 1009. [Google Scholar]
- Tarifa, T.; Yensen, E. Mammals of Bolivian Polylepis woodlands. Revista Boliviana de Ecología y Conservación Ambiental 2001, 9, 29–44. [Google Scholar]
- Tatura, A.; del Valle, R.; Bianchi, M.; Outes, V.; Villarosa, G.; Niegodzisz, J.; Debaene, G. Late Pleistocene palaeolakes in the Andean and Extra-Andean Patagonia at mid-latitudes of South America. Quaternary Intern. 2002, 89, 135–150. [Google Scholar] [CrossRef]
- Teta, P.; Rodríguez, D. Mammalogy National Collection (MACNMa); Occurrence Dataset; Museo Argentino de Ciencias Naturales ’Bernardino Rivadavia’ (MACN): Caba, Argentina, 2020. [Google Scholar]
- Thirgood, S.J. The effect of sex, season and habitat availability on patterns of habitat use in fallow deer. J. Zool. Lond. 1995, 235, 645–659. [Google Scholar] [CrossRef]
- Torrejon, F. Variables geohistoricos en la evolucion del sistema economico Pehuenche durante el periodo colonial. Revista Universum 2001, 16, 219–236. [Google Scholar]
- Van Soest, P.J. Nutritional Ecology of the Ruminant; O & B Books, Inc.: Corvallis, OR, USA, 1982; 373p. [Google Scholar]
- Via, S.; Gomulkiewicz, R.; DeJong, G.; Scheiner, C.D.; Schlichting, S.M.; Van Tienderen, P.H. Adaptive phenotypic plasticity: Consensus and controversy. TREE 1995, 10, 212–217. [Google Scholar] [CrossRef]
- Vidaurre, F.G. Des Herrn Abts Vidaure Kurzgefasste, Geographische, Natürliche und Bürgerliche Geschichte des Königreichs Chile; Carl Ernst Bohn: Hamburg, Germany, 1782; 208p. [Google Scholar]
- von Colditz, R. Im Reiche des Kondor; Paul Parey: Berlin, Germany, 1925; 415p. [Google Scholar]
- von Thüngen, J.; Lanari, M.R. Profitability of sheep farming and wildlife management in Patagonia. Pastoralism 2010, 1, 274–290. [Google Scholar]
- Wagner, J.U. Die Säugthiere in Abbildungen nach der Natur; L.D. Weigel: Leipzig, Germany, 1855. [Google Scholar]
- Waterhouse, G.R. Mammalia. In The Zoology of the Voyage of H.M.S. Beagle, Under the Command of Captain Fitzroy, During the years 1832-1836; Smith, Elder and Co.: London, UK, 1839. [Google Scholar]
- Webb, S.D. A history of savanna vertebrates in the New World. Part II: South America and the Great Interchange. Annual Rev. Ecol. Evol. System. 1978, 9, 393–426. [Google Scholar] [CrossRef]
- Weber, A. Chiloe: Su Estado Actual, su Colonizacion, su Porvenir; Imprenta Mejia: Santiago, Chile, 1903; 194p. [Google Scholar]
- Wells, K.W.; Stangl, F.B. Superior size and antler development in populations of white-tailed deer (Odocoileus virginianus) from the North Texas rolling plains. Texas J. Sci. 2003, 55, 337–346. [Google Scholar]
- Wolffsohn, J.W. Notas sobre el huemul. Revista Chilena de Historia Natural 1910, 14, 227–234. [Google Scholar]
- Yockney, I.J.; Hickling, G.J. Distribution and diet of chamois (Rupicapra rupicapra) in Westland forests, South Island, New Zealand. N. Z. J. Ecol. 2000, 24, 31–38. [Google Scholar]



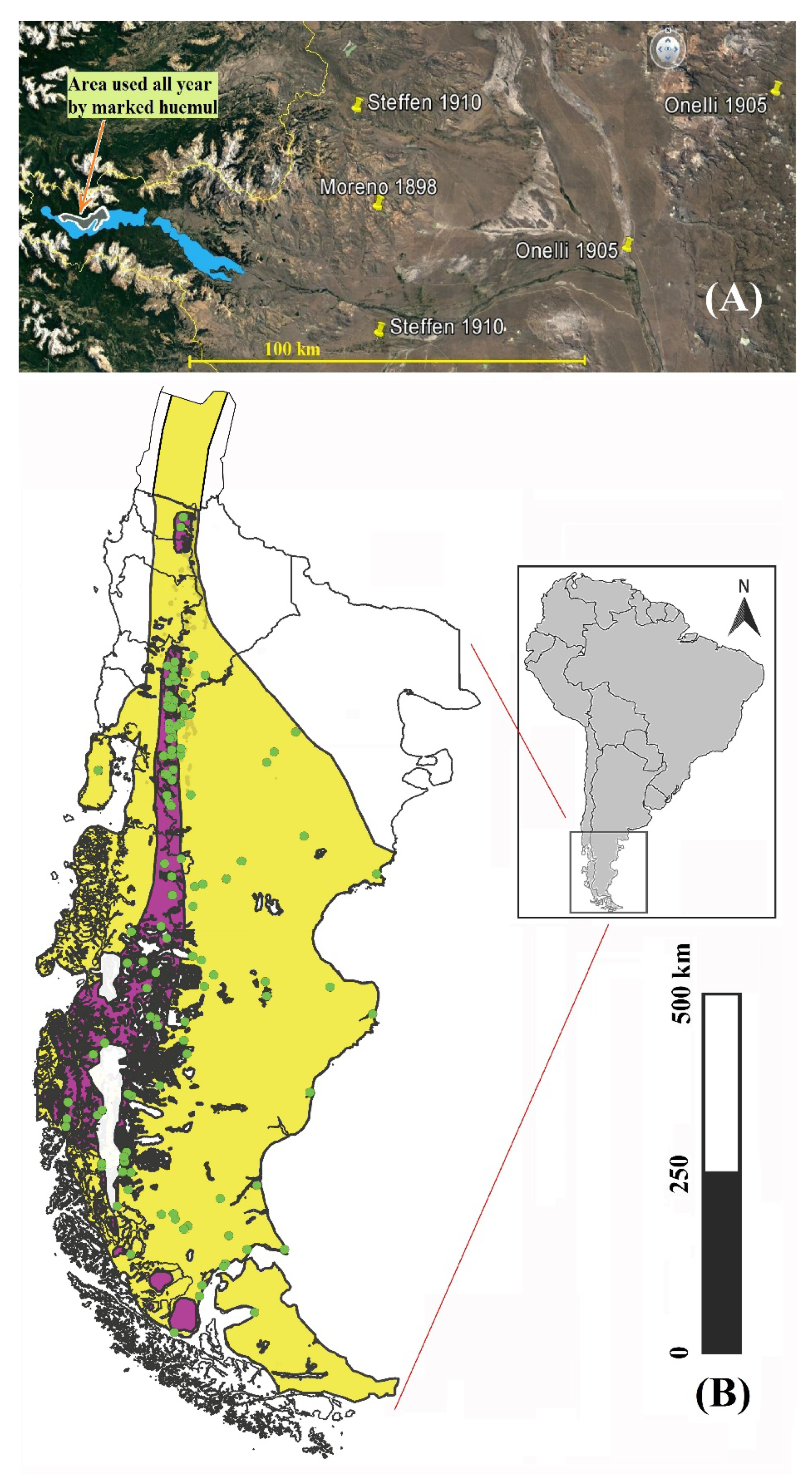
| Date Author(s) | Observations of Presence |
|---|---|
| 1521 and 1598 after Eastman 1915 | By Atlantic ports of San Julian and Desire, Atlantic |
| 1793 Pennant | By Port Desire, Atlantic Coast |
| 1833 MacDouall | By Gregory Bay, open area, 100 km from the nearest forest |
| 1835 Roulin | By Port San Julian, Atlantic Coast |
| 1863 Cox | Year-round resident populations on winter ranges, coexisting with guanaco |
| 1864 Claraz | Many guanacos coexisting with equally as many huemul in lowlands |
| 1871 Musters | Harvested huemul in open, treeless areas |
| 1875 Günther | Between Andean Mountain foothills and Patagonian mesas, reaching the Atlantic Coast |
| 1880 Behm | Seen in open area far from forests while hunting 45 km east of Chilean border |
| 1892 Philippi | Large groups during seasonal migration to lower areas |
| 1898 Lydekker | Migration down to the open grassland plains where they remain during winter |
| 1900 Steffen | His team ate huemul for weeks, working in open grasslands of foothills: coexistence with guanaco further east |
| 1901 Burmeister | Eighteen huemul (2 groups) in open grasslands, 220 km from nearest forest |
| 1902 Prichard | Groups with more than 100 huemul coexisting with guanaco during winter in valleys |
| 1903 Church | Grassland plains were the home of guanaco, huemul and ostriches |
| 1903 Hatcher | Harvesting huemul in the open 100 km from the nearest forest |
| 1904 Anonymous | The governor hunted huemul far east of continental divide 270 km from forests (includes photo) |
| 1905 Onelli | Near Choiquenilahue and between Senguer and Chubut rivers, 120 km from forests |
| 1911 Neveu-Lemaire | Reported winter migration down to valleys |
| 1923 Osgood | Harvesting huemul in steppe grasslands far from forest, coexisting with guanaco |
| 1925 von Colditz | Harvesting several huemul in steppe grasslands far from forest |
| 1929 Gigoux | Reported seasonal migration and formation of large herds in winter |
| 1936 Giai | Reported seasonal migration and formation of 50 or more huemul in winter |
| 1940 Krieg | Year-round resident huemul in low valleys |
| 1945 Agostini | Many guanacos and equally as many huemul in open grasslands |
| 1949 Grosse | Seasonal migrations and large herds in low valleys |
| 1962 Liebermann | Seasonal migrations in winter down to protected valleys and foothill areas |
| 1969 Kolliker Frers | Reported huemul still occurred in Patagonian open grasslands until 1850s |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flueck, W.T.; Smith-Flueck, J.A.M.; Escobar, M.E.; Zuliani, M.; Fuchs, B.; Geist, V.; Heffelfinger, J.R.; Black-Decima, P.; Gizejewski, Z.; Vidal, F.; et al. Loss of Migratory Traditions Makes the Endangered Patagonian Huemul Deer a Year-Round Refugee in Its Summer Habitat. Conservation 2022, 2, 322-348. https://doi.org/10.3390/conservation2020023
Flueck WT, Smith-Flueck JAM, Escobar ME, Zuliani M, Fuchs B, Geist V, Heffelfinger JR, Black-Decima P, Gizejewski Z, Vidal F, et al. Loss of Migratory Traditions Makes the Endangered Patagonian Huemul Deer a Year-Round Refugee in Its Summer Habitat. Conservation. 2022; 2(2):322-348. https://doi.org/10.3390/conservation2020023
Chicago/Turabian StyleFlueck, Werner T., Jo Anne M. Smith-Flueck, Miguel E. Escobar, Melina Zuliani, Beat Fuchs, Valerius Geist, James R. Heffelfinger, Patricia Black-Decima, Zygmunt Gizejewski, Fernando Vidal, and et al. 2022. "Loss of Migratory Traditions Makes the Endangered Patagonian Huemul Deer a Year-Round Refugee in Its Summer Habitat" Conservation 2, no. 2: 322-348. https://doi.org/10.3390/conservation2020023
APA StyleFlueck, W. T., Smith-Flueck, J. A. M., Escobar, M. E., Zuliani, M., Fuchs, B., Geist, V., Heffelfinger, J. R., Black-Decima, P., Gizejewski, Z., Vidal, F., Barrio, J., Molinuevo, S. M., Monjeau, A. J., Hoby, S., & Jiménez, J. E. (2022). Loss of Migratory Traditions Makes the Endangered Patagonian Huemul Deer a Year-Round Refugee in Its Summer Habitat. Conservation, 2(2), 322-348. https://doi.org/10.3390/conservation2020023







