1. Introduction
Snakebite envenoming has long represented a dramatic intersection of human vulnerability and ecological exposure. In contemporary times, it continues to pose a major threat to public health in many parts of the world, particularly in sub-Saharan Africa, South and Southeast Asia, and Latin America, where rural populations live in close proximity to habitats of venomous snakes [
1]. The World Health Organization has recently reclassified snakebite envenoming as a category A neglected tropical disease, a move that reflects not only its high morbidity and mortality burden (estimated at over 100,000 deaths annually) but also the severe underreporting and lack of reliable surveillance systems in affected regions [
2]. Despite the development of increasingly effective antivenoms, preventable deaths remain commonplace, largely due to socioeconomic barriers, logistical delays, and limitations in clinical infrastructure.
While the majority of the scientific literature focuses on the clinical and therapeutic aspects of envenomation, far less attention has been paid to the forensic perspective of snakebite fatalities. In many instances, deaths occur outside of medical settings and reach the attention of health authorities only through medico-legal investigations. The precise determination of cause of death in such cases is crucial not only for public health reporting, but also for legal accountability, insurance determinations, and the identification of systemic healthcare deficiencies. The variability in clinical presentation, ranging from rapid cardiovascular collapse due to neurotoxic envenoming to delayed renal or hepatic failure from hemotoxic/cytotoxic agents, further complicates diagnosis and underscores the need for a multidisciplinary post-mortem approach [
1,
2,
3,
4].
In this context, the autopsy emerges as a critical tool for clarifying the mechanisms underlying fatal envenomation. External examination may reveal fang marks, tissue edema, or necrosis, but it is the internal analysis, supported by histological and toxicological methods, that provides the most reliable evidence of venom-induced pathophysiology. Techniques such as immunohistochemistry, enzyme-linked immunosorbent assay (ELISA), and, more recently, liquid chromatography–tandem mass spectrometry (LC–MS/MS) allow for the detection and characterization of venom components in biological tissues and fluids, thereby enabling a more accurate attribution of cause of death. Moreover, forensic analysis can reveal otherwise occult comorbidities that contributed to the fatal outcome or identify signs of medical mismanagement, delayed intervention, or inappropriate treatment [
5,
6,
7].
The primary objective of this review is to synthesize the current forensic knowledge regarding autopsy-confirmed snakebite envenoming deaths, drawing on published case reports, retrospective studies, and systematic reviews that include post-mortem data. Through this synthesis, we aim to identify common diagnostic patterns, histopathological markers, and toxicological profiles associated with fatal envenomation and discuss the medico-legal implications of these findings. Ultimately, by focusing on the post-mortem perspective, an aspect largely neglected in previous epidemiological or clinical reviews, we intend to provide a framework for enhancing forensic accuracy and for reinforcing the scientific foundations of death certification in cases of suspected snakebite.
2. Materials and Methods
This scoping review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (PRISMA 2020 checklist provided as
Supplementary Material), with the aim of identifying all the available peer-reviewed literature reporting autopsy-confirmed deaths following snakebite envenoming. The review protocol was defined a priori to ensure methodological transparency and reproducibility.
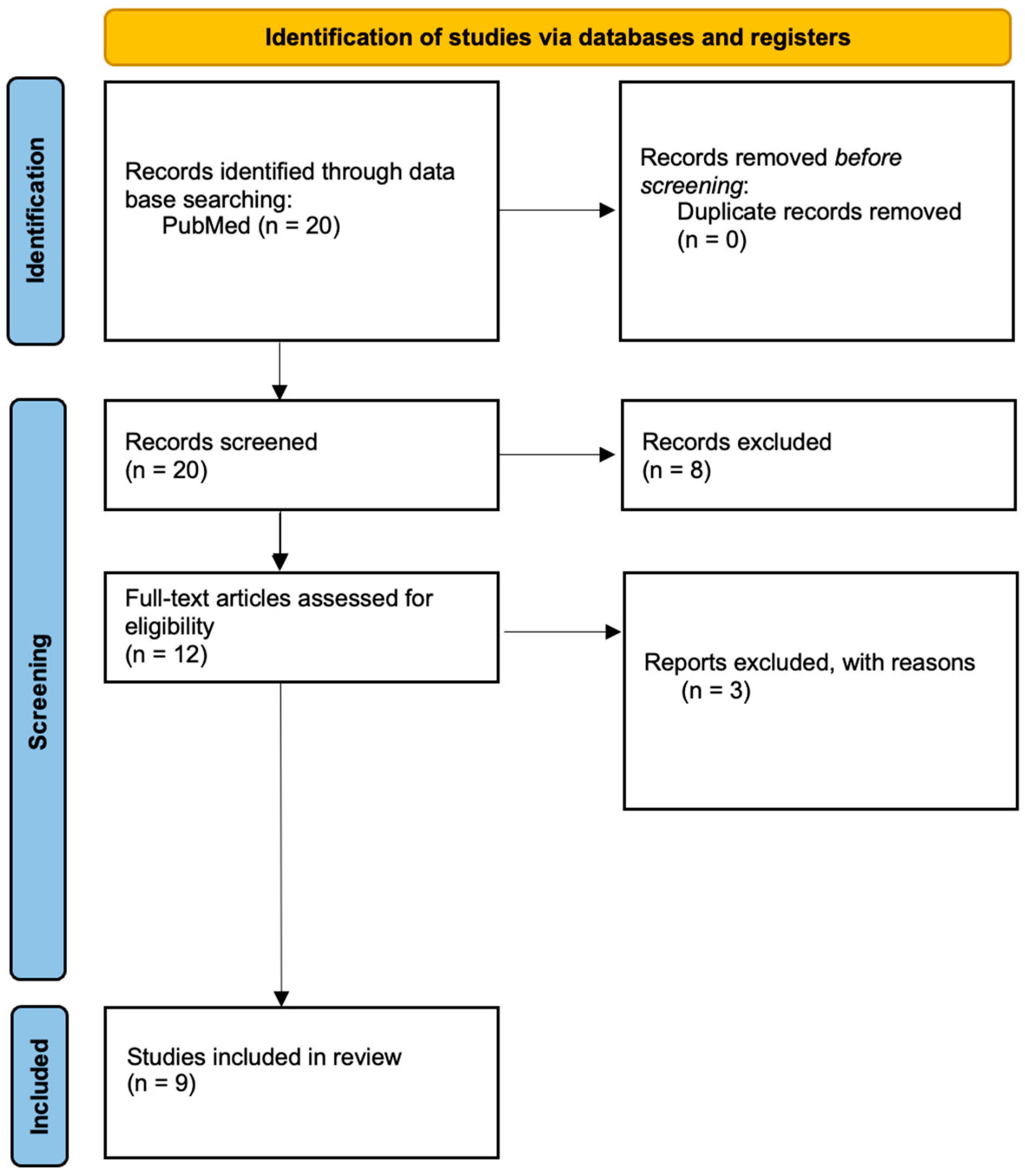
An extensive electronic search was performed using the databases PubMed and NCBI, covering publications up to March 2024. The search strategy was built using a combination of Medical Subject Headings (MeSH) and free-text terms designed to capture both the forensic and toxicological aspects of fatal snakebite cases. Specifically, the following query string was employed: “snake bite poisoning AND death AND forensics AND autopsy”. Filters were applied to restrict the results to human studies published in English. No temporal restrictions were imposed in order to ensure a comprehensive capture of the relevant literature.
Following database retrieval, all identified articles were screened at the title and abstract level to assess relevance. Studies were included only if they fulfilled the following criteria: (1) documentation of a fatal snakebite with forensic autopsy findings; (2) histopathological or toxicological data available for analysis; (3) the full-text article available in English; and (4) publication in a peer-reviewed scientific journal. Excluded from the review were studies that did not involve death certification, reports focusing on non-venomous snakebites or constriction deaths, articles lacking forensic detail, and studies in which autopsy data were absent or incomplete. Context-only records did not meet the primary inclusion criteria (e.g., absence of histology/toxicology or absence of envenoming) and were therefore excluded from the quantitative counts but discussed narratively to inform the forensic differential.
The selection process was conducted independently by two authors to minimize bias, and any disagreements regarding inclusion were resolved through discussion with a third reviewer. Of 20 records retrieved, 12 were assessed in full text and 9 fulfilled all eligibility criteria for inclusion in the primary synthesis. Among the selected publications, the majority were single-case reports, while two studies presented small case series or retrospective analyses with detailed post-mortem examinations (
Figure 1).
Data from the included studies were extracted using a structured form, which recorded key information such as geographic origin of the case, snake species involved, time between bite and death, autopsy findings, toxicological results, and the presence of comorbidities or predisposing conditions. No quantitative meta-analysis was performed due to the descriptive and heterogeneous nature of the data. Instead, the findings are presented as a narrative synthesis, emphasizing recurring forensic themes and medico-legal implications.
3. Results
The nine studies selected for this scoping review documented deaths attributed to snakebite envenoming and confirmed through autopsy findings. The literature spans multiple continents, including Asia, Africa, and the Americas, reflecting the global distribution of snakebite fatalities and the diversity of both the snake species involved and the forensic methodologies applied. While some of the included articles described single, well-documented cases, others reported series of deaths occurring in specific epidemiological contexts. Despite this heterogeneity, several recurring patterns and diagnostic features emerged from the analysis [
7,
8,
9,
10,
11,
12,
13,
14,
15,
16].
In six of the nine studies, the envenoming species was either identified directly through eyewitness reports and photographic evidence or inferred from the toxicological profile and the regional epidemiology of venomous snakes. The most frequently reported species included
Atractaspis corpulenta [
8],
Bothrops jararacussu [
9],
Deinagkistrodon acutus (Huang et al., 2021) [
10],
Bungarus caeruleus,
Bothrops lanceolatus (Malbranque et al., 2008), [
15] and
Laticauda colubrina, a species of sea snake [
16]. In the extensive review conducted by Feola et al. (2020) [
7], which included fifty-six autopsy-confirmed cases, the most commonly implicated families were Viperidae, Elapidae, and Lamprophiidae/Atractaspididae, demonstrating that these groups account for the majority of medically significant envenomings globally.
Across the cases examined, autopsies revealed a broad spectrum of lethal pathophysiological mechanisms. Venom-induced consumption coagulopathy (VICC) emerged as one of the most recurrent patterns in viperid envenoming (often previously labeled as DIC), consistent with the known procoagulant and hemorrhagic effects of their venoms. In three separate cases, acute renal failure was reported as either the primary cause of death or a significant contributing factor, with histological confirmation of acute tubular necrosis and glomerular microthrombi. These renal lesions were associated with venom-induced nephrotoxicity and hypoperfusion, and were most frequently described in bites from vipers and pit vipers, in agreement with the previous forensic and nephrotoxicological literature (Farooqui et al., 2016) [
13].
Acute respiratory failure was identified in at least two cases, particularly those involving elapid snakes such as kraits and cobras, whose neurotoxic venom disrupts neuromuscular transmission and causes rapid diaphragmatic paralysis. One case reported by Brunda et al. demonstrated evidence of complete respiratory arrest within hours of envenoming, with supportive histological findings of hypoxic–ischemic injury in the brainstem. Other lethal findings, albeit more sporadic, included hepatic necrosis, pulmonary edema, petechiae with asphyxial features, and encephalopathy, which were variably attributed to systemic effects of the venom or terminal pathophysiological cascades.
A unique case described by Benvenuti et al. documented a rupture of the mitral valve chordae tendineae following a
Bothrops jararacussu envenomation, highlighting a rare but potentially underrecognized cardiovascular complication [
9]. Similarly, a sea snake envenomation reported by Tiemensma and Byard was associated with rapid multisystem failure, including rhabdomyolysis and cardiac dysfunction, confirming that marine elapid venoms may exert profound systemic toxicity [
16].
Toxicological analysis was available in the majority of cases and revealed significant variability in methodology and sensitivity. Enzyme-linked immunosorbent assays (ELISA) were the most commonly employed tool for venom detection, with satisfactory specificity when appropriate reference antigens were used. Liquid chromatography–mass spectrometry (LC–MS/MS) was applied in a limited number of cases, but proved particularly useful in the differential identification of venom families and in the exclusion of alternative toxicological causes. The use of advanced immunoanalytical and chromatographic techniques enabled the correlation of autopsy findings with biochemical venom signatures, strengthening the diagnostic certainty in several of the reports reviewed.
From a methodological standpoint, most autopsies followed standardized forensic protocols, including full-body dissection, histological sampling of all major organs, and external documentation of the bite site. Fang marks were identified in nearly all cases, usually as paired puncture wounds associated with soft tissue edema, ecchymosis, or localized necrosis. Internal examination frequently revealed visceral congestion, hemorrhagic foci in the gastrointestinal tract and lungs, and microscopic evidence of vascular injury and inflammation. In the most comprehensive studies, immunohistochemistry was performed on tissue sections to localize venom components, providing visual confirmation of systemic dissemination and organ-specific venom effects. Although nine studies met our inclusion criteria (autopsy with histology and/or toxicology), this number is orders of magnitude lower than the estimated annual snakebite mortality globally. This discrepancy reflects limited availability and standardization of forensic investigations, not a low incidence of fatal envenoming. Accordingly, our review should be read as an appraisal of adequately investigated snakebite deaths and the quality of their autopsy/toxicology documentation, rather than as an estimate of epidemiologic burden (
Table 1).
Table 1.
(A)—Exposure and case descriptors. (B)—Clinicopathological and analytical findings; inferred cause of death.
Table 1.
(A)—Exposure and case descriptors. (B)—Clinicopathological and analytical findings; inferred cause of death.
| (A) |
|---|
| Reference | Type of Paper | Cases | Deaths | Family | Species
(Common/
Scientific) | Geographic Location |
|---|
| Feola et al., 2020 [7] | Systematic review | 56 | 56 | Viperidae; Elapidae; Lamprophiidae/Atractaspididae | – | Global (multiple regions) |
| Tilbury et al., 2016 [8] | Case report | 1 | 1 | Atractaspididae (Lamprophiidae clade) | Atractaspis corpulenta | South Africa |
| Benvenuti et al., 2003 [9] | Case report | 1 | 1 | Viperidae | Bothrops jararacussu | Brazil |
| Huang et al., 2021 [10] | Case report | 1 | 1 | Viperidae | Deinagkistrodon acutus | China |
| McCarty et al., 1989 [11] * | Case report | 1 | 1 | Non-venomous | Python reticulatus | USA (Hawaii) |
| Thein CM et al., 2019 [12] | Systematic review | 2069 | 84 | Viperidae | – | Myanmar |
| Farooqui et al., 2016 [13] | Retrospective study | 2539 | 54 | Viperidae; Elapidae (cobra) | – | India (Maharashtra) |
| Silva et al., 2013 [14] | Case report | 1 | 1 | Elapidae | Bungarus caeruleus | Sri Lanka |
| Malbranque et al., 2008 [15] | Case report | 1 | 1 | Viperidae | Bothrops lanceolatus | Martinique |
| Tiemensma et al., 2021 [16] | Case report | 1 | 1 | Elapidae | Laticauda colubrina | Oceania (Fiji/PNG) |
| (B) |
| Reference | Predominant Mechanism | Key Autopsy/Histology | Diagnostic Tools Used | Cause of Death (Inferred) |
| Feola et al., 2020 [7] | VICC/mixed | Cortical and glomerular necrosis; adrenal/pituitary hemorrhage; pulmonary edema; ATN; ARDS | Histopathology; ELISA; LC–MS/MS | VICC-related hemorrhage; multiorgan failure |
| Tilbury et al., 2016 [8] | Pulmonary edema/congestion | Severe pulmonary edema and congestion | Histology | Acute pulmonary failure |
| Benvenuti et al., 2003 [9] | VICC (hemorrhagic) | Pulmonary hemorrhage | Histopathology | Massive pulmonary hemorrhage |
| Huang et al., 2021 [10] | Cyto-/myotoxic with renal failure | Thigh muscle necrosis; renal tubular necrosis; hepatocyte necrosis | Histopathology | Multiorgan necrosis; renal failure |
| McCarty et al., 1989 [11] * | N/A (non-envenoming) | Asphyxial petechiae | Macroscopic exam | Asphyxia due to constriction |
| Thein CM et al., 2019 [12] | VICC; AKI | AKI (82/84); pituitary/adrenal hemorrhage | Histopathology (retrospective) | AKI; coagulopathy consistent with VICC |
| Farooqui et al., 2016 [13] | VICC or neurotoxicity (likely) | Undefined in records | Autopsy records (limited histology) | Hemorrhagic shock (VICC) or neurotoxicity (likely) |
| Silva et al., 2013 [14] | VICC + respiratory failure | Coagulopathy with respiratory failure | Histopathology; toxicology | VICC with respiratory failure |
| Malbranque et al., 2008 [15] | Thrombotic microangiopathy | Cerebral/myocardial/mesenteric infarctions; mitral chordae rupture | Histopathology; gross cardiac exam | TMA; myocardial infarction |
| Tiemensma et al., 2021 [16] | Neurotoxic paralysis ± rhabdomyolysis | Acute respiratory failure; features of rhabdomyolysis | Histopathology; gross exam | Neurotoxic respiratory paralysis with systemic toxicity |
4. Discussion
4.1. The Forensic Relevance of Autopsy in Snakebite Envenoming Deaths
The forensic investigation of snakebite envenoming deaths represents a crucial and underdeveloped area in the literature, despite its importance in death certification, public health surveillance, and medico-legal accountability. As demonstrated by Feola et al., the majority of snakebite fatalities reported in forensic practice show significant diagnostic challenges that demand an interdisciplinary approach combining scene analysis, clinical reconstruction, toxicological confirmation, and thorough autopsy [
7]. In regions where envenoming remains endemic, deaths frequently occur before the victim reaches a healthcare facility, thereby excluding the possibility of antemortem diagnosis. In such scenarios, autopsy not only serves a confirmatory function but becomes the primary investigative tool to establish the cause of death and to rule out alternative mechanisms, such as trauma, myocardial infarction, or asphyxia. The identification of external fang marks, often accompanied by localized hemorrhage and necrosis, provides initial support for envenomation; however, internal and microscopic findings interpreted with toxicology ultimately substantiate the lethal pathway.
4.2. Pathophysiological Mechanisms Revealed by Post-Mortem Analysis
The cases reviewed consistently revealed a diversity of organ-specific lesions corresponding to the pharmacodynamic profile of different venoms. For instance, viperid bites, such as those by
Bothrops jararacussu and
Deinagkistrodon acutus, were associated with hemorrhagic diathesis, fibrin/fibrinogen consumption, and renal cortical injury, reflecting a combination of endothelial damage, coagulopathy, and ischemic insult. The report by Benvenuti et al. described pulmonary hemorrhage as the primary cause of death, emphasizing how vascular leakage and alveolar damage may be terminal manifestations of viper envenoming [
9]. In contrast, neurotoxic envenomation by elapids such as
Bungarus caeruleus or
Laticauda colubrina produced fatalities through profound neuromuscular paralysis. The case documented by Tiemensma and Byard exemplified how sea snake venom induced fulminant systemic toxicity with rapid onset of rhabdomyolysis, hyperkalemia, and cardiac arrest—findings confirmed at autopsy through histopathology and toxicological correlation [
16]. Huang et al. described massive unilateral edema and hemorrhagic infarction following a bite by
Deinagkistrodon acutus, with post-mortem examination confirming acute renal failure and pulmonary congestion as terminal events [
10]. These data highlight the importance of detailed gross and microscopic examination of multiple organs to distinguish between primary venom effects and secondary terminal complications [
17,
18,
19,
20,
21,
22,
23].
At a molecular level, lethal outcomes reflect convergent pathways:
- −
Neurotoxicity (respiratory failure): postsynaptic three-finger α-neurotoxins antagonize nicotinic acetylcholine receptors; presynaptic β-neurotoxic PLA2 complexes disrupt vesicle release. Forensic correlates include hypoxic–ischemic brain injury with sparse gross findings. Beyond ELISA, targeted immunoaffinity-LC–MS/MS can detect proteotypic neurotoxin peptides in blood/vitreous; venom-specific immunohistochemistry on FFPE diaphragm/intercostal muscles can support tissue distribution when validated.
- −
Coagulopathy and endothelial injury (VICC): SVMPs and serine proteases activate prothrombin/factor X, deplete fibrinogen, and promote hyperfibrinolysis; SVMP-mediated endothelial damage drives capillary leak and hemorrhage. Histology often shows fibrin degradation with limited microthrombi compared with classic DIC. Ancillary tools include viscoelastic testing (if perimortem), SVMP zymography in tissue homogenates, and immunoaffinity-LC–MS/MS of protease peptides.
- −
Cytotoxic/myotoxic injury and rhabdomyolysis: PLA2 myotoxins and cytotoxins cause myonecrosis at the bite site with risk of compartment syndrome and sepsis; systemic rhabdomyolysis precipitates acute tubular necrosis. MALDI imaging can localize venom proteins in bite-site tissue; LC–MS/MS detects myotoxin peptides; CK/myoglobin (perimortem) are supportive.
- −
Cardiotoxicity/autonomic dysregulation: cardiotoxic peptides and vasoactive mediators provoke arrhythmias and profound hypotension; contraction band necrosis may be seen. Targeted MS panels can, where available, identify cardiotoxin peptides.
- −
Renal/endocrine injury: multifactorial ATN (myo/hemoglobinuria, direct nephrotoxicity, hypotension) and adrenal/pituitary hemorrhage are recurrent in viperid envenoming; kidney tissue may retain detectable venom longer than blood post-mortem, improving late detection.
4.3. The Role of Toxicology and Immunodetection in Confirming Envenomation
The forensic pathologist must not only identify morphological changes but also confirm the presence of venom within biological matrices. Immunoassay-based techniques, particularly ELISA, have been effectively employed to detect venom proteins in post-mortem blood and tissue samples [
18]. Brunda et al. demonstrated the utility of indirect competitive inhibition ELISA in quantifying
Bungarus caeruleus venom in fatal human cases, reinforcing its role in forensic identification when species documentation is lacking [
19]. However, ELISA sensitivity may decline post-mortem due to proteolysis and putrefaction, limiting its window of applicability. Complementary methods such as immunoaffinity LC–MS/MS enable proteotypic peptide detection and family/species-level attribution, while venom-specific immunohistochemistry on FFPE tissues can corroborate tissue distribution when fluids are unavailable. Where available, viscoelastic testing profiles support VICC characterization perimortem.
4.4. Interindividual Variability and Diagnostic Complexity
One of the most salient findings across the reviewed literature is the profound interindividual variability in both clinical course and post-mortem presentation of snakebite envenomation. Factors such as the amount of venom injected, the anatomical site of the bite, the interval between envenomation and death, and the presence of pre-existing conditions all contribute to this heterogeneity. Farooqui et al. [
13], in a retrospective analysis of fatal snakebites in Maharashtra, reported that time to hospital presentation and lack of antivenom administration were decisive predictors of mortality, yet autopsy findings ranged from minimal external signs to extensive visceral hemorrhage and organ failure. This variation reinforces the notion that no single pathological finding is pathognomonic of fatal envenoming; rather, the diagnosis must emerge from a synthesis of clinical, forensic, and toxicological data.
4.5. Diagnostic Limitations and the Importance of Contextual Integration
Despite the methodological advances in post-mortem toxicology and histopathology, the accurate diagnosis of death by snakebite envenoming continues to present notable limitations, particularly in settings lacking access to advanced analytical tools. In several of the reviewed cases, the forensic determination of death depended heavily on circumstantial evidence, such as eyewitness reports of the bite, ecological probability of venomous species in the region, or the presence of fang marks. However, as shown in the study by Silva et al., even these classic indicators can be misleading, as non-venomous or “dry” bites may leave similar local lesions, while severely envenomed patients may present without visible fang marks, especially in sea snake exposures or in delayed post-mortem examination [
15]. Therefore, a conclusive attribution of death to envenoming must rely on a convergence of data from scene investigation, external and internal autopsy findings, and, wherever possible, toxicological confirmation. Importantly, recognizing VICC aligns clinicopathological features with venom biochemistry and avoids interpretive errors.
4.6. Species-Specific Lethality and Comparative Pathology
A critical insight that emerges from the autopsy literature on snakebite envenoming is the marked variation in lethality and pathological expression across different snake species. This inter-species variability reflects not only differences in venom composition—ranging from neurotoxic peptides to hemotoxic enzymes—but also the anatomical and biochemical tropism of those toxins. For instance,
Bothrops species, particularly
B. jararacussu, are known for their potent hemorrhagic and procoagulant activity, as reflected in the case reported by Benvenuti et al., where coagulopathy consistent with VICC and massive pulmonary hemorrhage were the primary lethal mechanisms [
9]. In contrast,
Atractaspis corpulenta, though less frequently reported, can cause sudden death with minimal external findings, as observed in the South African case described by Tilbury et al., where histology revealed severe pulmonary congestion and edema without widespread systemic damage [
8]. Such differences necessitate a forensic approach that is not only anatomopathologically precise but also taxonomically informed, guiding targeted sampling and test selection.
4.7. The Medico-Legal Value of Snake Identification in Autopsy Cases
The correct identification of the offending snake species in post-mortem investigations carries substantial medico-legal implications. Not only does it enhance the accuracy of death certification, but it may also be essential for establishing liability, especially in occupational or environmental contexts. In rural and agricultural settings, where fatal envenomation may constitute a compensable work-related injury, species identification can help establish the foreseeability and preventability of exposure. The case reported by Silva et al. illustrates this point: a fatal
Bungarus caeruleus envenoming was confirmed post-mortem based on toxicological analysis and regional snake prevalence, enabling the court to link the death to professional risk factors and to determine jurisdictional responsibility [
14]. From a forensic perspective, species identification also aids in differentiating between venomous and non-venomous bites, as well as between accidental and intentional events. Although
Python reticulatus is non-venomous, the case reported by McCarty et al. (1989) [
11] involved fatal constriction leading to asphyxial petechiae, a scenario that might be misinterpreted in the absence of contextual data. Similarly, in rare cases involving exotic or non-native species—either through illegal captivity or zoos—accurate identification can help reconstruct the dynamics of exposure and assign responsibility for containment breaches or public safety lapses. Furthermore, species-level data collected through forensic autopsies can inform ecological surveillance systems and antivenom distribution strategies; deaths from rare or geographically unexpected species, as in the sea snake case described by Tiemensma and Byard, may signal shifts in species range, trade routes, or climate-driven migration patterns [
16].
4.8. Limitations of Current Autopsy Protocols in Snakebite-Related Deaths
Despite the crucial role of autopsy in identifying fatal envenomations, current forensic protocols often fall short of capturing the full pathophysiological complexity of these deaths. In many cases, particularly in low-resource settings, the autopsy is limited to a gross examination without adequate histological or toxicological sampling. This can lead to underreporting or misclassification of snakebite deaths, especially when external signs such as fang marks are ambiguous or absent, or when death occurs after a delayed progression of organ failure. The variability in autopsy standards across regions contributes to significant gaps in mortality data and impairs the establishment of consistent forensic criteria [
7,
8,
9,
10,
11,
12,
13,
14,
15,
16]. One critical deficiency lies in the absence of standardized sampling protocols for histological and biochemical analysis. Vital organs such as the kidneys, lungs, liver, and brain should be examined microscopically in all suspected cases, given the propensity of venoms to induce diffuse microvascular injury, necrosis, and edema. However, many reports—such as those in retrospective analyses by Farooqui et al. and Thein et al. (2019)—lack uniform detail on histopathological procedures, suggesting that tissue-level investigation may not have been consistently performed [
13]. Moreover, specialized techniques such as venom-specific immunohistochemistry or the use of LC–MS/MS for post-mortem venom identification remain confined to research contexts and are rarely applied in routine forensic practice. The collection and preservation of biological samples is also pivotal: venom proteins degrade post-mortem—especially under tropical conditions—so delays or inappropriate storage can compromise assay performance. Few laboratories hold comprehensive species-specific venom libraries, limiting confident attribution in biodiverse regions where multiple venomous species coexist [
17,
18,
19,
20,
21,
22,
23]. These limitations have direct medico-legal consequences, from “undetermined” certifications to lost compensation and missed public-health signals. Although nine studies, met our criteria, this is negligible compared with annual mortality estimates. The gap underscores scarcity of adequately investigated fatalities and constrained forensic capacity, rather than rarity of lethal envenoming. Finally, restricting to English may have excluded autopsy-based reports from high-burden regions, potentially underestimating the diversity of pathological patterns and toxicological approaches. Several regional databases (e.g., SciELO/LILACS, African Index Medicus, IndMED) could yield additional cases. Consequently, our synthesis should be interpreted as conservative with respect to geographic and methodological breadth.
4.9. Future Directions in Forensic Snakebite Investigation
The findings of this review underscore the need for a more structured and globally coordinated approach to the forensic study of snakebite deaths. The lack of standardized protocols for autopsy, toxicological sampling, and histological processing in these cases hinders both case-level certainty and cross-study comparability. As snakebite envenoming continues to affect some of the world’s most resource-constrained populations, efforts must focus on developing low-cost, field-deployable diagnostics—such as species-specific lateral flow assays or venom peptide panels—that can be used even in basic forensic settings. Moreover, the creation of international databases documenting autopsy-confirmed snakebite deaths, including toxicological profiles and species distribution, would offer an invaluable tool for both forensic scientists and public health authorities. The incorporation of geospatial, ecological, and clinical metadata could facilitate pattern recognition and early warning systems in high-risk regions. Cross-disciplinary collaboration between forensic pathologists, toxinologists, epidemiologists, and emergency medicine specialists will be essential to improving both the identification of fatal envenoming and the development of targeted preventive measures.
5. Conclusions
Snakebite envenoming remains a lethal yet often under-recognized cause of death, particularly in regions where medical infrastructure is inadequate and post-mortem investigations are scarce. This review demonstrates that autopsy, when coupled with histological and toxicological analysis, provides an indispensable means of confirming death due to envenoming and of characterizing the specific pathological mechanisms involved. From VICC-related hemorrhage and neurotoxic paralysis to rhabdomyolysis-associated renal failure and multi-organ injury, the forensic manifestations of snakebite deaths are diverse, often subtle, and require high diagnostic suspicion. Despite methodological progress, challenges persist in terms of diagnostic consistency, toxicological access, and standardization of investigative procedures. The reviewed literature makes clear that no single autopsy finding is universally diagnostic of fatal envenomation, and that the forensic confirmation of these deaths must rely on the integration of anatomical, biochemical, and circumstantial evidence. As such, forensic pathology occupies a central role in filling the diagnostic and epidemiological gaps left by clinical systems in snakebite-endemic regions. Strengthening the forensic investigation of these deaths is not only a matter of scientific rigor but a prerequisite for more accurate mortality surveillance and the development of public health interventions that are evidence-based and context-sensitive.