Monophyly or Homoplasy? The Relationships of a Rare New Species of Cambeva (Siluriformes: Trichomycteridae) from the Brazilian Atlantic Forest with a Bicolored Caudal Pattern †
Abstract
1. Introduction
2. Materials and Methods
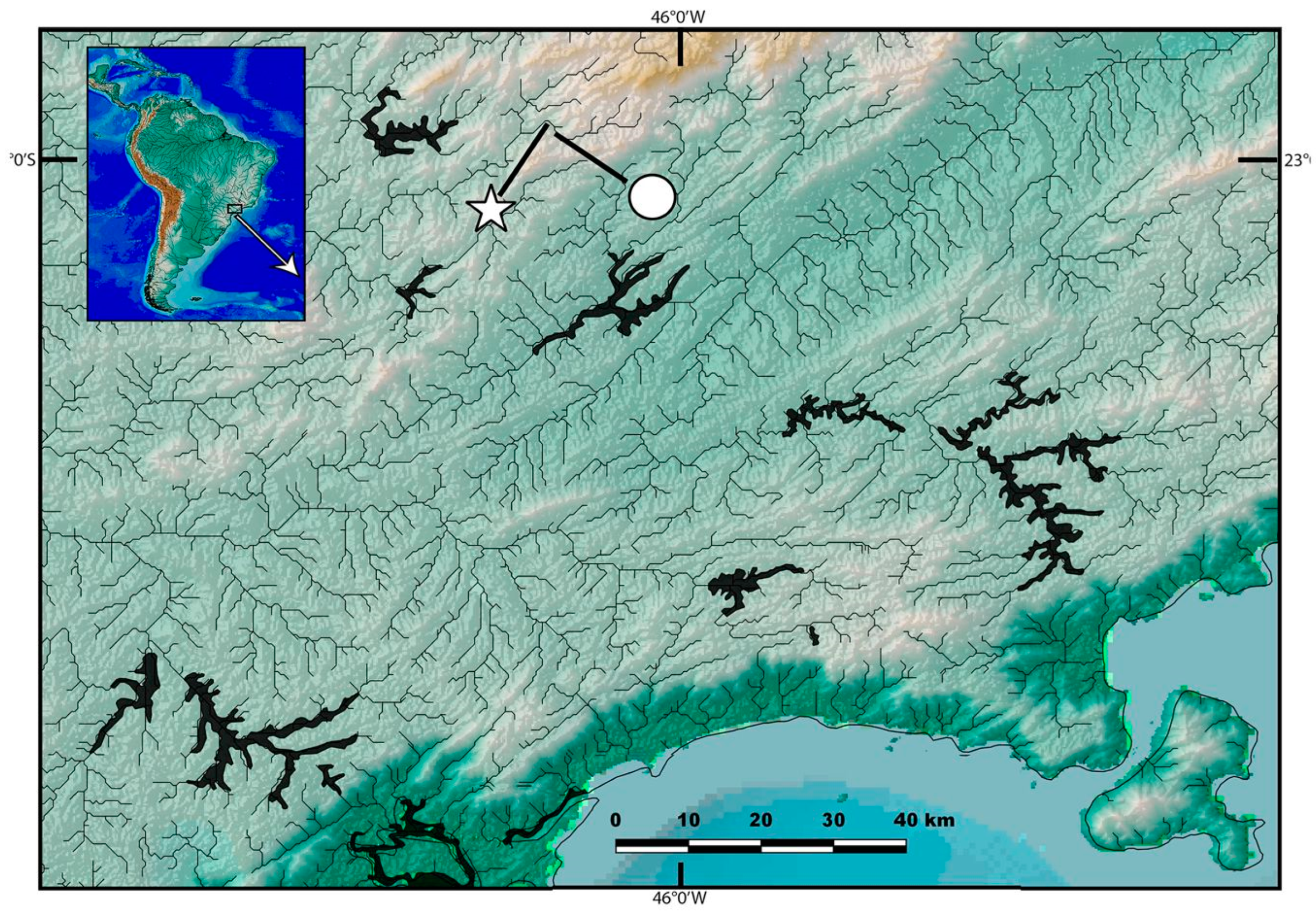
2.1. Specimens
2.2. Morphological Data
2.3. Molecular Phylogenetic Analysis
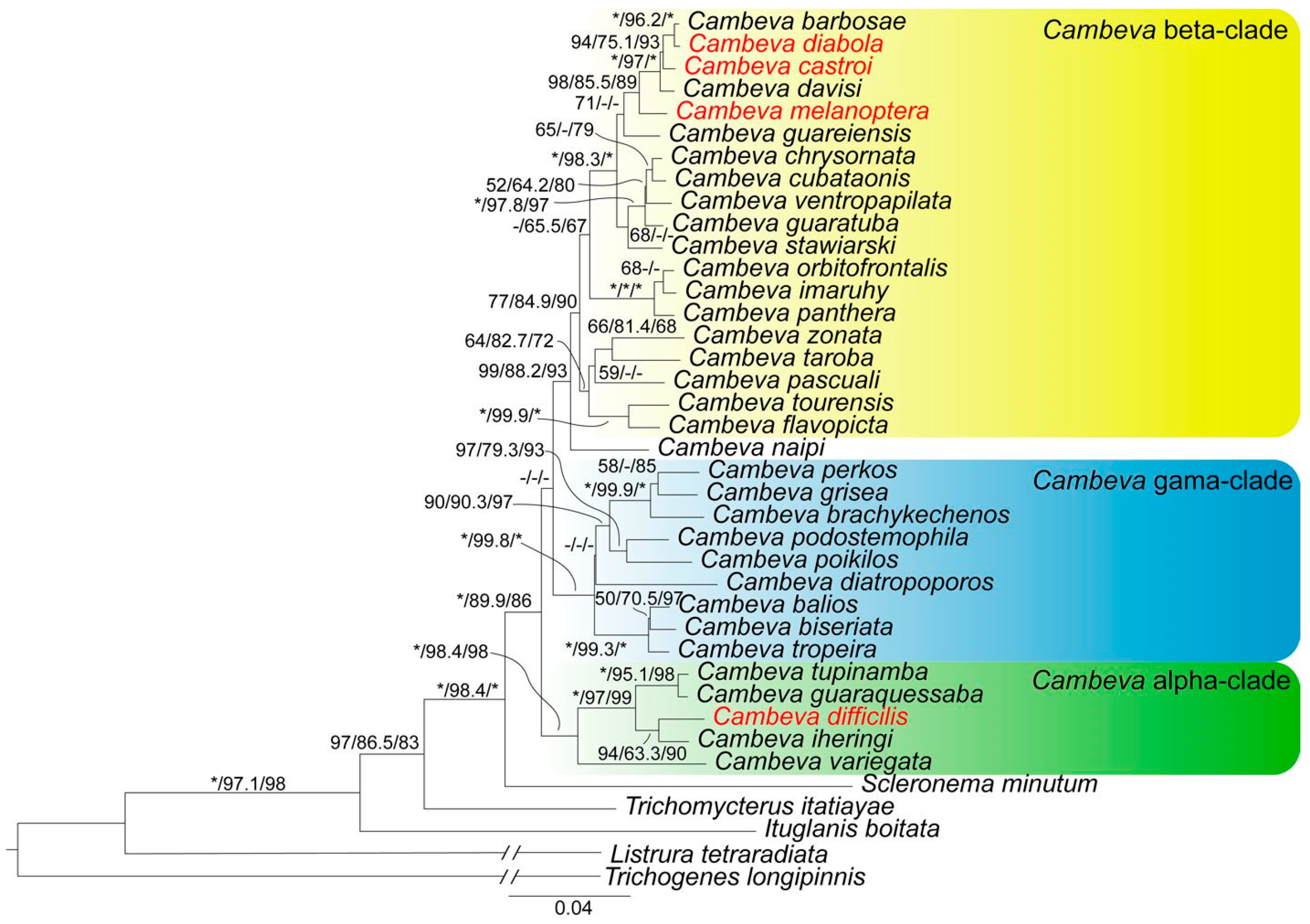
3. Results
3.1. Phylogenetic Relationships and Comparative Morphology
3.2. Taxonomic Accounts
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| COI | CYT | RAG2 | |
|---|---|---|---|
| Trichogenes longipinnis | OQ810037 | MK123704 | MF431117 |
| Listrura tetraradiata | JQ231083 | JQ231088 | MN385826 |
| Ituglanis boitata | OQ810038 | MK123706 | MK123758 |
| Trichomycterus itatiayae | MW671552 | MW679291 | KY858198 |
| Scleronema minutum | MK123685 | MK123707 | MK123759.1 |
| Cambeva variegata | KY857991 | OQ110805 | KY858211 |
| Cambeva zonata | MN995706 | KY858053 | — |
| Cambeva brachykechenos | MN995669 | MN995758 | — |
| Cambeva diatropoporos | MN995654 | KY858065 | KY858213 |
| Cambeva stawiarski | MN995720 | MN995779 | — |
| Cambeva perkos | MN995663 | MN995759 | KY858202 |
| Cambeva poikilos | MN995693 | MN995761 | — |
| Cambeva guaraquessaba | MN995662 | MN995749 | — |
| Cambeva naipi | MN995699 | MN995771 | — |
| Cambeva taroba | MN995708 | MN995757 | — |
| Cambeva tupinamba | MN995658 | MN995751 | — |
| Cambeva tropeira | MN995674 | MN995752 | — |
| Cambeva grisea | MN995671 | MN995760 | — |
| Cambeva imaruhy | MN995700 | MN995766 | OQ814191 |
| Cambeva orbitofrontalis | MN995703 | MN995764 | OQ814192 |
| Cambeva cubataonis | OQ810039 | OQ110814 | OQ110815 |
| Cambeva iheringi | GU701893 | KY858074 | KY858223 |
| Cambeva barbosae | MK123689 | OQ110808 | OQ110815 |
| Cambeva diabola | JN989258 | OQ110812 | — |
| Cambeva balios | OQ810040 | OQ814186 | OQ814193 |
| Cambeva chrysornata | MN995726 | OQ110810 | OQ110819 |
| Cambeva pascuali | MF034463 | OQ110811 | OQ110820 |
| Cambeva guaratuba | MN995721 | MN995792 | — |
| Cambeva panthera | OQ810041 | OQ814187 | OQ814194 |
| Cambeva flavopicta | OQ810042 | OQ814188 | OQ814195 |
| Cambeva podostemophila | OQ810043 | OQ814189 | OQ814196 |
| Cambeva tourensis | MN995697 | OQ814190 | OQ814197 |
| Cambeva davisi | MN995730 | MK123714 | MK123762 |
| Cambeva ventropapilata | PP319016 | OQ110807 | OQ110818 |
| Cambeva guareiensis | PP448192 | OQ110813 | OQ110821 |
| Cambeva melanoptera | PP496403 * | — | — |
| Cambeva difficilis | PP496404 * | PP496778 * | — |
Appendix B
| Partition | Base Pairs | Evolutive Model |
|---|---|---|
| COI 1st | 229 | TIM + G |
| COI 2nd | 228 | TRNEF + G |
| COI 3rd | 228 | F81 + I |
| CYTB 1st | 331 | K80 + I + G |
| CYTB 2nd | 331 | HKY + I + G |
| CYTB 3rd | 331 | TRN + G |
| RAG2 2 nd RAG2 3rd | 525 | TVMEF + G |
| RAG2 1st | 263 | HKY |
References
- Myers, N.; Mittermeir, R.A.; Mittermeir, C.G.; da Fonseca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Furtado, S.G.; Menini Neto, L. Diversity high up: A cloud forest of the Serra da Mantiqueira as a vascular epiphyte hotspot. Rodriguésia 2018, 69, 263–279. [Google Scholar] [CrossRef]
- da Silva, E.T.; Peixoto, M.A.A.; Leite, F.S.F.; Feio, R.N.; Garcia, P.C.A. Anuran Distribution in a Highly Diverse Region of the Atlantic Forest: The Mantiqueira Mountain Range in Southeastern Brazil. Herpetologica 2018, 74, 294–305. [Google Scholar] [CrossRef]
- Pereira, L.C.; Chautems, A.; Menini Neto, L. Biogeography and conservation of Gesneriaceae in the Serra da Mantiqueira, southeastern region of Brazil. Braz. J. Bot. 2020, 44, 239–248. [Google Scholar] [CrossRef]
- Costa, W.J.E.M.; Mattos, J.L.; Barbosa, M.A.; Vilardo, P.J.; Katz, A.M. High endemism in an endangered biodiversity hotspot: Phylogeny, taxonomy and distribution patterns of catfishes of the Psammocambeva alpha-clade (Siluriformes: Trichomycteridae) from the Rio Doce basin, Brazil. Fishes 2023, 8, 474. [Google Scholar] [CrossRef]
- de Pinna, M.C.C. Trichomycterus castroi, a new species of trichomycterid catfish from the Rio Iguaçu of southeastern Brazil (Teleostei, Siluriformes). Ichthyol. Explor. Freshw. 1992, 3, 89–95. [Google Scholar]
- Bockmann, F.A.; Casatti, L.; de Pinna, M.C.C. A new species of trichomycterid catfish from the Rio Paranapanema basin, southeastern Brazil (Teleostei: Siluriformes), with comments on the phylogeny of the family. Ichthyol. Explor. Freshw. 2004, 15, 225–242. [Google Scholar]
- Costa, W.J.E.M.; Abilhoa, V.; Dalcin, R.H.; Katz, A.M. A new catfish species of the genus Cambeva (Siluriformes: Trichomycteridae) from the Rio Iguaçu drainage, southern Brazil, with a remarkable unique colour pattern. J. Fish Biol. 2022, 101, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Costa, W.J.E.M.; Feltrin, C.R.M.; Mattos, J.L.M.; Amorim, P.F.A.; Katz, A.M. Phylogenetic relationships of new taxa support repeated pelvic fin loss in mountain catfishes from southern Brazil (Siluriformes: Trichomycteridae). Zool. Anz. 2023, 305, 82–90. [Google Scholar] [CrossRef]
- Costa, W.J.E.M.; Feltrin, C.R.M.; Mattos, J.L.O.; Dalcin, R.H.; Abilhoa, V.; Katz, A.M. Morpho-Molecular Discordance? Re-Approaching Systematics of Cambeva (Siluriformes: Trichomycteridae) from the Guaratuba-Babitonga-Itapocu Area, Southern Brazil. Fishes 2023, 8, 63. [Google Scholar] [CrossRef]
- Taylor, W.R.; Van Dyke, G.C. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium 1985, 9, 107–119. [Google Scholar]
- Costa, W.J.E.M. Description de huit nouvelles espèces du genre Trichomycterus (Siluriformes: Trichomycteridae), du Brésil oriental. Rev. Franç. D’aquariol. Herpetol. 1992, 18, 101–110. [Google Scholar]
- Costa, W.J.E.M.; Katz, A.M.; Mattos, J.L.O.; Amorim, P.F.; Mesquita, B.O.; Vilardo, P.J.; Barbosa, M.A. Historical review and redescription of three poorly known species of the catfish genus Trichomycterus from south-eastern Brazil (Siluriformes: Trichomycteridae). J. Nat. Hist. 2020, 53, 2905–2928. [Google Scholar] [CrossRef]
- Costa, W.J.E.M. Comparative osteology, phylogeny and classification of the eastern South American catfish genus Trichomycterus (Siluriformes: Trichomycteridae). Taxonomy 2021, 1, 160–191. [Google Scholar] [CrossRef]
- Kubicek, K.M. Developmental osteology of Ictalurus punctatus and Noturus gyrinus (Siluriformes: Ictaluridae) with a discussion of siluriform bone homologies. Verteb. Zool. 2022, 72, 661–727. [Google Scholar] [CrossRef]
- Arratia, G.; Huaquin, L. Morphology of the lateral line system and of the skin of diplomystid and certain primitive loricarioid catfishes and systematic and ecological considerations. Bonn Zool. Monogr. 1995, 36, 1–110. [Google Scholar]
- Villa-Verde, L.; Lazzarotto, H.; Lima, S.Q.M. A new glanapterygine catfish of the genus Listrura (Siluriformes: Trichomycteridae) from southeastern Brazil, corroborated by morphological and molecular data. Neotrop. Ichthyol. 2012, 10, 527–538. [Google Scholar] [CrossRef]
- Unmack, P.J.; Bennin, A.P.; Habi, E.M.; Victoriano, P.F.; Johnson, J.B. Impact of ocean barriers, topography, and glaciation on the phylogeography of the catfish Trichomycterus areolatus (Teleostei: Trichomycteridae) in Chile. Biol. J. Linn. Soc. 2009, 97, 876–892. [Google Scholar] [CrossRef]
- Barros, L.C.; Santos, U.; Cioffi, M.D.B.; Dergam, J.A. Evolutionary divergence among Oligosarcus spp. Ostariophysi, Characidae from the São Francisco and Doce River Basins: Oligosarcus solitarius Menezes, 1987 shows the highest rates of chromosomal evolution in the Neotropical region. Zebrafish 2015, 12, 102–110. [Google Scholar] [CrossRef]
- Ward, R.D.; Zemlak, T.S.; Innes, B.H.; Last, P.R.; Hebert, P.D. DNA barcoding Australia’s fish species. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2005, 360, 1847–1857. [Google Scholar] [CrossRef]
- Hardman, M.; Page, L.M. Phylogenetic relationships among bullhead catfishes of the genus Ameiurus (Siluriformes: Ictaluridae). Copeia 2003, 2003, 20–33. [Google Scholar]
- Costa, W.J.E.M.; Henschel, E.; Katz, A.M. Multigene phylogeny reveals convergent evolution in small interstitial catfishes from the Amazon and Atlantic forests (Siluriformes: Trichomycteridae). Zool. Scr. 2020, 49, 159–173. [Google Scholar] [CrossRef]
- Cramer, C.A.; Bonatto, S.L.; Reis, R.E. Molecular phylogeny of the Neoplecostominae and Hypoptopomatinae (Siluriformes: Loricariidae) using multiple genes. Mol. Phylog. Evol. 2011, 59, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis Version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef] [PubMed]
- Katz, A.M.; Barbosa, M.A.; Mattos, J.L.O.; Costa, W.J.E.M. Multigene analysis of the catfish genus Trichomycterus and description of a new South American trichomycterine genus (Siluriformes, Trichomycteridae). Zoosyst. Evol. 2018, 94, 557–566. [Google Scholar] [CrossRef]
- Ochoa, L.E.; Silva, G.S.C.; Costa e Silva, G.J.; Oliveira, C.; Datovo, A. New species of Trichomycterus (Siluriformes: Trichomycteridae) lacking pelvic fins from Paranapanema basin, southeastern Brazil. Zootaxa 2017, 4319, 550–560. [Google Scholar] [CrossRef]
- Donin, L.M.; Ferrer, J.; Carvalho, T.P. Taxonomical study of Trichomycterus (Siluriformes: Trichomycteridae) from the Ribeira de Iguape River basin reveals a new species recorded in the early 20th century. J. Fish Biol. 2020, 96, 886–904. [Google Scholar] [CrossRef]
- Ochoa, L.E.; Roxo, F.F.; DoNascimiento, C.; Sabaj, M.H.; Datovo, A.; Alfaro, M.; Oliveira, C. Multilocus analysis of the catfish family Trichomycteridae (Teleostei: Ostariophysi: Siluriformes) supporting a monophyletic Trichomycterinae. Mol. Phyl. Evol. 2017, 115, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Chenna, R.; Sugawara, H.; Koike, T.; Lopez, R.; Gibson, T.J.; Higgins, D.G.; Thompson, J.D. Multiple sequence alignment with the Clustal series of programs. Nucleic Acids Res. 2003, 31, 3497–3500. [Google Scholar] [CrossRef]
- Lanfear, R.; Frandsen, P.B.; Wright, A.M.; Senfeld, T.; Calcott, B. PartitionFinder 2: New methods for selecting partitioned models of evolution for molecular and morphological phylogenetic analyses. Mol. Biol. Evol. 2017, 34, 772–773. [Google Scholar] [CrossRef]
- Suchard, M.A.; Lemey, P.; Baele, G.; Ayres, D.L.; Drummond, A.J.; Rambaut, A. Bayesian phylogenetic and phylodynamic data integration using BEAST 1.10. Vir. Evol. 2018, 4, vey016. [Google Scholar] [CrossRef] [PubMed]
- Rambaut, A.; Drummond, A.J.; Xie, D.; Baele, G.; Suchard, M.A. Posterior summarisation in Bayesian phylogenetics using Tracer 1.7. Syst. Biol. 2018, 67, 901–904. [Google Scholar] [CrossRef] [PubMed]
- Minh, B.Q.; Schmidt, H.A.; Chernomor, O.; Schrempf, D.; Woodhams, M.D.; von Haeseler, A.; Lanfear, R. IQ-TREE 2: New models and efficient methods for phylogenetic inference in the genomic era. Mol. Biol. Evol. 2020, 37, 1530–1534. [Google Scholar] [CrossRef] [PubMed]
- Hoang, D.T.; Chernomor, O.; von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the ultrafast bootstrap approximation. Mol. Biol. Evol. 2018, 35, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Guindon, S.; Dufayard, J.-F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New algorithms and methods to estimate maximum-likelihood phylogenies: Assessing the performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Ferrer, J.; Malabarba, L.R. Systematic revision of the Neotropical catfish genus Scleronema (Siluriformes: Trichomycteridae), with descriptions of six new species from Pampa grasslands. Neotrop. Ichthyol. 2020, 18, e190081. [Google Scholar] [CrossRef]
- Stuart, Y.E. Divergent uses of “parallel evolution” during the history of The American Naturalist. Am. Nat. 2019, 193, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Schluter, D.; Nagel, L.M. Parallel speciation by natural-selection. Am. Nat. 1995, 146, 292–301. [Google Scholar] [CrossRef]
- Duponchelle, F.; Paradis, E.; Ribbink, A.J.; Turner, G.F. Parallel life history evolution in mouthbrooding cichlids from the African Great Lakes. Proc. Natl. Acad. Sci. USA 2008, 105, 15475–15480. [Google Scholar] [CrossRef]
- Balentine, C.M.; Bolnick, D.A. Parallel evolution in human populations: A biocultural perspective. Evol. Anthropol. 2022, 31, 302–316. [Google Scholar] [CrossRef]
- Bailey, S.F.; Rodrigue, N.; Kassen, R. The effect of selection environment on the probability of parallel evolution. Mol. Biol. Evol. 2015, 32, 1436–1448. [Google Scholar] [CrossRef]
- Costa, W.J.E.M.; Feltrin, C.R.M.; Katz, A.M. Two new remarkable and endangered catfish species of the genus Cambeva (Siluriformes, Trichomycteridae) from southern Brazil. Europ. J. Taxon. 2022, 794, 140–155. [Google Scholar] [CrossRef]
- Wosiacki, W.B.; de Pinna, M. Trichomycterus igobi, a new catfish species from the rio Iguaçu drainage: The largest head in Trichomycteridae (Siluriformes: Trichomycteridae). Neotrop. Ichthyol. 2008, 6, 17–23. [Google Scholar] [CrossRef]
- Wosiacki, W.B.; de Pinna, M. A new species of the Neotropical catfish genus Trichomycterus (Siluriformes: Trichomycteridae) representing a new body shape for the family. Copeia 2008, 2008, 273–278. [Google Scholar] [CrossRef]



| Holotype | Paratypes (n = 2) | |
|---|---|---|
| Standard length (SL) | 60.8 | 56.2–81.0 |
| Percentage of standard length | ||
| Body depth | 16.6 | 14.1–15.9 |
| Caudal peduncle depth | 11.5 | 9.8–10.4 |
| Body width | 14.6 | 11.8–12.0 |
| Caudal peduncle width | 6.2 | 4.0–5.0 |
| Pre-dorsal length | 57.3 | 56.5–60.0 |
| Pre-pelvic length | 53.7 | 56.7–56.7 |
| Dorsal fin base length | 13.1 | 13.4–14.9 |
| Anal fin base length | 8.6 | 8.5–10.6 |
| Caudal fin length | 18.4 | 14.0–19.7 |
| Pectoral fin length | 14.7 | 12.0–16.7 |
| Pelvic fin length | 11.9 | 8.6–12.4 |
| Head length | 23.1 | 21.5–22.6 |
| Percentage of head length | ||
| Head depth | 46.9 | 42.4–47.7 |
| Head width | 89.6 | 79.1–81.5 |
| Snout length | 43.5 | 45.0–45.2 |
| Interorbital width | 28.1 | 22.5–26.9 |
| Preorbital length | 16.8 | 16.3–19.2 |
| Eye diameter | 10.4 | 9.4–10.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Costa, W.J.E.M.; Feltrin, C.R.M.; Mattos, J.L.O.; Katz, A.M. Monophyly or Homoplasy? The Relationships of a Rare New Species of Cambeva (Siluriformes: Trichomycteridae) from the Brazilian Atlantic Forest with a Bicolored Caudal Pattern. Taxonomy 2024, 4, 341-353. https://doi.org/10.3390/taxonomy4020017
Costa WJEM, Feltrin CRM, Mattos JLO, Katz AM. Monophyly or Homoplasy? The Relationships of a Rare New Species of Cambeva (Siluriformes: Trichomycteridae) from the Brazilian Atlantic Forest with a Bicolored Caudal Pattern. Taxonomy. 2024; 4(2):341-353. https://doi.org/10.3390/taxonomy4020017
Chicago/Turabian StyleCosta, Wilson J. E. M., Caio R. M. Feltrin, José Leonardo O. Mattos, and Axel M. Katz. 2024. "Monophyly or Homoplasy? The Relationships of a Rare New Species of Cambeva (Siluriformes: Trichomycteridae) from the Brazilian Atlantic Forest with a Bicolored Caudal Pattern" Taxonomy 4, no. 2: 341-353. https://doi.org/10.3390/taxonomy4020017
APA StyleCosta, W. J. E. M., Feltrin, C. R. M., Mattos, J. L. O., & Katz, A. M. (2024). Monophyly or Homoplasy? The Relationships of a Rare New Species of Cambeva (Siluriformes: Trichomycteridae) from the Brazilian Atlantic Forest with a Bicolored Caudal Pattern. Taxonomy, 4(2), 341-353. https://doi.org/10.3390/taxonomy4020017






