Redefining the Taxonomic Boundaries of Genus Xanthomonas
Abstract
1. Introduction
2. Materials and Methods
2.1. Genome Access and Quality Assessment
2.2. Phylogenomic Investigation of the Members of the Order Lysobacterales (Xanthomonadales)
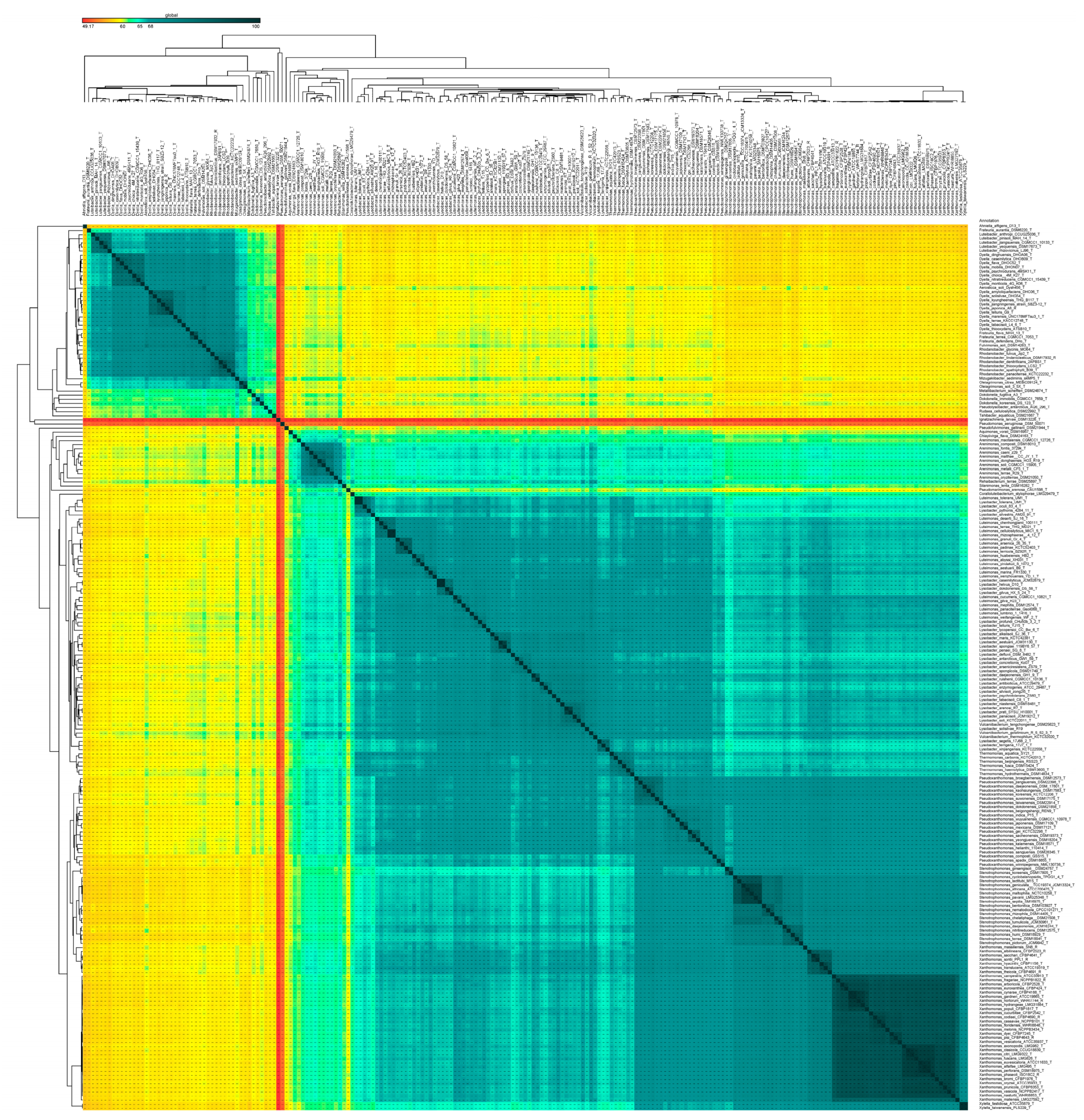
2.3. Taxonogenomic Assessment of the Members of the Order Lysobacterales (Xanthomonadales)
3. Results and Discussion
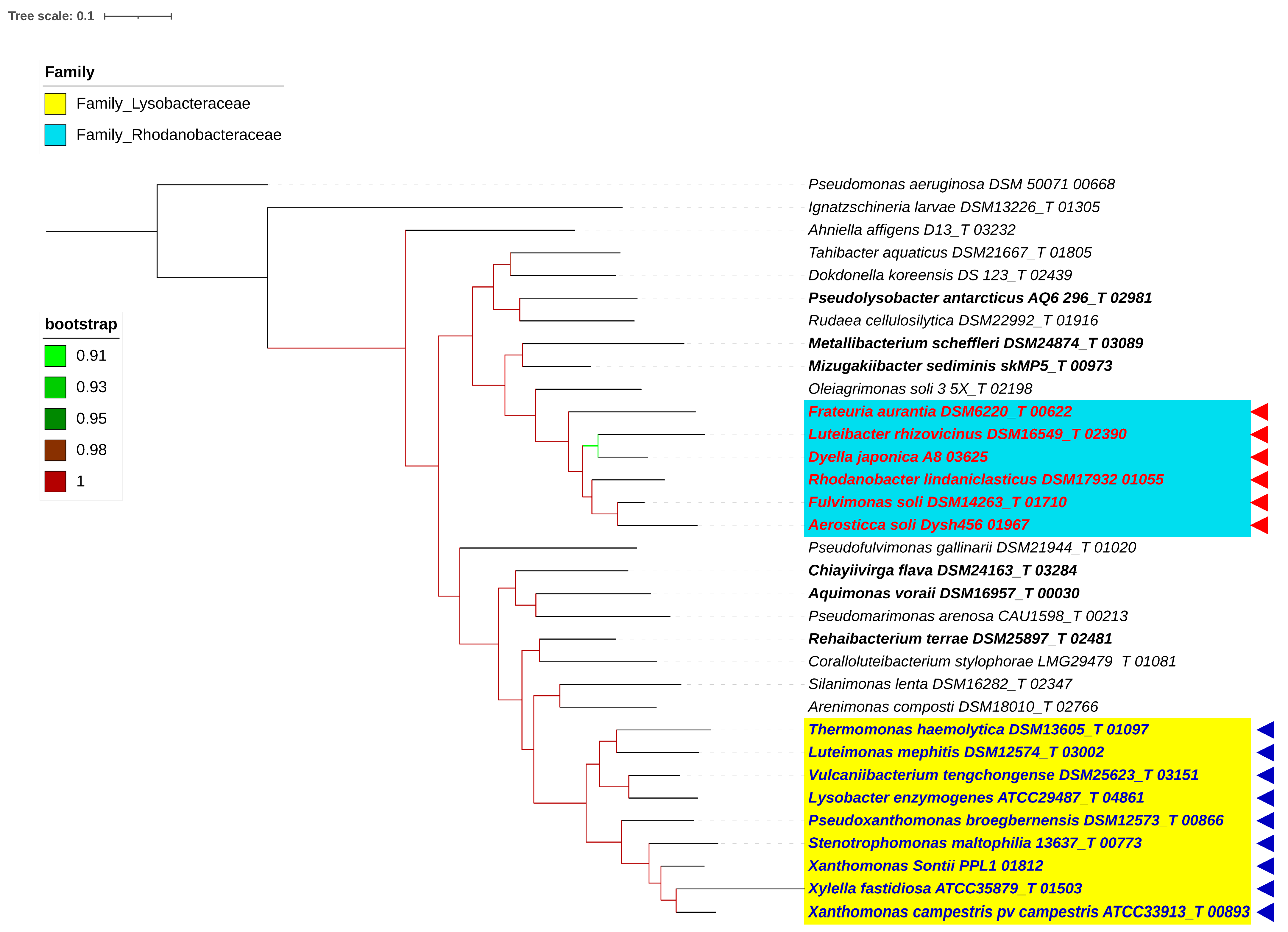
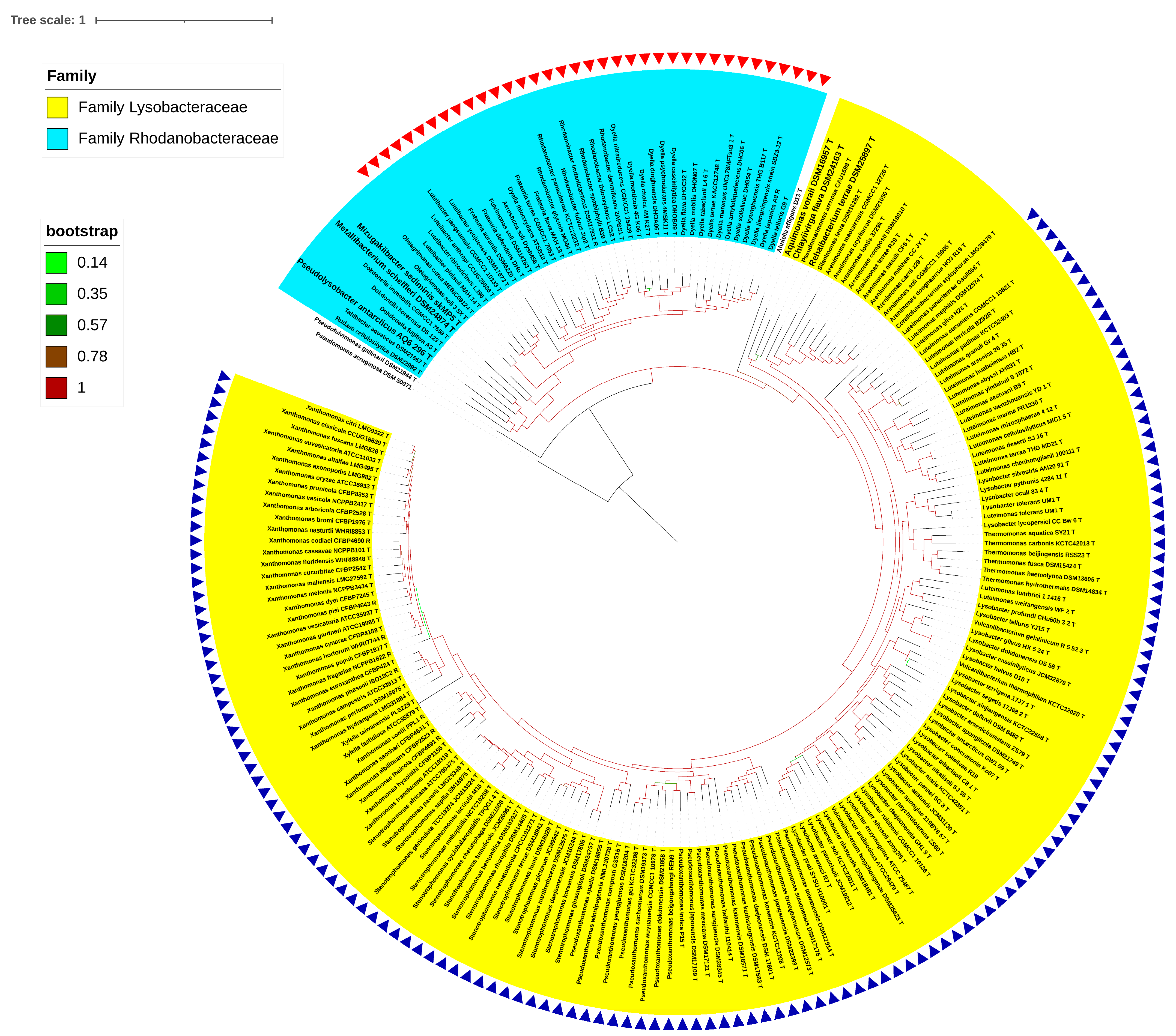
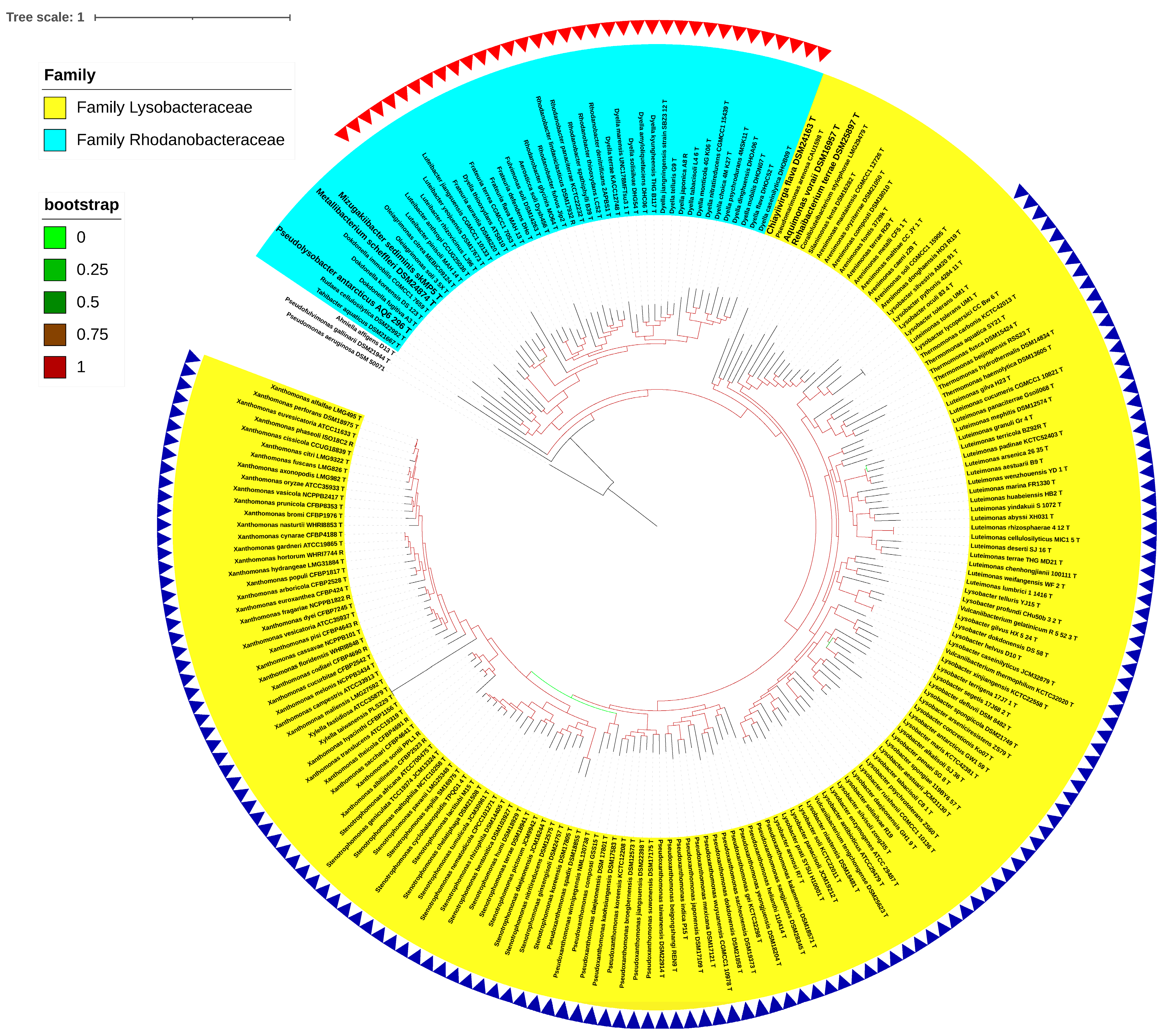
3.1. Phylogenomic Evaluation of the Families and Their Boundaries within the Order Lysobacterales (Xanthomonadales)
3.2. Reshuffling of Six Genera within the Families Lysobacteraceae and Rhodanobacteraceae
3.3. Phylo-Taxonogenomic Assessment of the Genera of Xanthomonas and Rhodanobacter within Their Respective Families
3.4. Taxonomic Revision of the Families Lysobacteraceae and Rhodanobacteraceae within the Order Lysobacterales in Light of the Phylo-Taxonogenomic Findings
3.5. Validating the Deep-Phylo-Taxonogenomic-Based Taxonomic Revelations at the Level of Genus with a Complete 16S rRNA Gene Sequence-Based Threshold
3.6. Emended Description of the Order Xanthomonadales (Saddler and Bradbury 2005a,b) [2]
- Synonym: Lysobacterales Christensen and Cook (1978) (Approved Lists 1980)
3.7. Emended Description of the Family Xanthomonadaceae Saddler et al. 2005
- N.L. fem. n. Xanthomonas, type genus of the family; L. fem. pl. n. suff. -aceae, ending to denote a family; N.L. fem. pl. n. Xanthomonadaceae, the Xanthomonas family.
- Synonym: Lysobacteraceae Christensen and Cook 1978 (Approved Lists 1980)
3.8. Description of the Family Frateuriaceae fam. nov.
- Synonym: Rhodanobacteraceae Naushad et al. 2015 [1].
3.9. Emended Description of the Genus Xanthomonas Dowson 1939 (Approved Lists 1980)
- Xan.tho.mo.nas. Gr. masc. adj. xanthos, yellow; L. fem. n. monas, unit, monad; N.L. fem. n. Xanthomonas, yellow monad.
- Emended description of Xanthomonas maltophilia = Stenotrophomonas maltophilia ((Hugh 1981) Swings et al. 1983).
- Emended description of Xanthomonas fastidiosa = Xylella fastidiosa Wells et al. 1987.
- Emended description of Xanthomonas broegbernensis = Pseudoxanthomonas broegbernensis Finkmann et al. 2000.
- Emended description of the genus Frateuria Swing et al. 1980.
- Frat.eur’i.a. N.L. fem. n. Frateuria, named after Joseph Frateur (1903–1974), the eminent Belgian microbiologist. The type species is Frateuria aurantia (ex Kondô and Ameyama 1958) Swings et al. 1980 [56].
- Synonyms: Aerosticca, Fulvimonas, Rhodanobacter, Dyella, and Luteibacter.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Naushad, S.; Adeolu, M.; Wong, S.; Sohail, M.; Schellhorn, H.E.; Gupta, R.S. A phylogenomic and molecular marker based taxonomic framework for the order Xanthomonadales: Proposal to transfer the families Algiphilaceae and Solimonadaceae to the order Nevskiales ord. nov. and to create a new family within the order Xanthomonadales, the family Rhodanobacteraceae fam. nov., containing the genus Rhodanobacter and its closest relatives. Antonie Van Leeuwenhoek 2015, 107, 467–485. [Google Scholar]
- Saddler, G.; Bradbury, J. Xanthomonadales ord. nov. In Bergey’s Manual of Systematic Bacteriology; validation of the publication of new names and new combinations previously effectively published outside the IJSEM, List; Springer: Boston, MA, USA, 2005; pp. 2235–2238. [Google Scholar]
- Midha, S.; Bansal, K.; Kumar, S.; Girija, A.M.; Mishra, D.; Brahma, K.; Laha, G.S.; Sundaram, R.M.; Sonti, R.V.; Patil, P.B. Pop-ulation genomic insights into variation and evolution of Xanthomonas oryzae pv. oryzae. Sci. Rep. 2017, 7, 40694. [Google Scholar] [CrossRef] [PubMed]
- Bansal, K.; Midha, S.; Kumar, S.; Patil, P.B. Ecological and evolutionary insights into Xanthomonas citri pathovar diversity. Appl. Environ. Microbiol. 2017, 83, e02993-16. [Google Scholar] [CrossRef] [PubMed]
- Midha, S.; Patil, P.B. Genomic insights into the evolutionary origin of Xanthomonas axonopodis pv. citri and its ecological rela-tives. Appl. Environ. Microbiol. 2014, 80, 6266–6279. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chatterjee, S.; Almeida, R.P.P.; Lindow, S. Living in two worlds: The plant and insect lifestyles of Xylella fastidiosa. Annu. Rev. Phytopathol. 2008, 46, 243–271. [Google Scholar] [CrossRef]
- Brooke, J.S. Stenotrophomonas maltophilia: An emerging global opportunistic pathogen. Clin. Microbiol. Rev. 2012, 25, 2–41. [Google Scholar] [CrossRef]
- Vauterin, L.; Yang, P.; Alvarez, A.; Takikawa, Y.; Roth, D.A.; Vidaver, A.K.; Stall, R.E.; Kersters, K.; Swings, J. Identification of non-pathogenic Xanthomonas strains associated with plants. Syst. Appl. Microbiol. 1996, 19, 96–105. [Google Scholar] [CrossRef]
- Singh, A.; Kumar, S.; Bansal, K.; Patil, P.B. Taxonomic and phylogenomic assessment identifies phyto-pathogenicity potential of Stenotrophomonas maltophilia complex. Phytopathology 2023. [Google Scholar] [CrossRef]
- Kumar, S.; Bansal, K.; Patil, P.P.; Patil, P.B. Phylogenomics insights into order and families of Lysobacterales. Access Microbiol. 2019, 1, e000015. [Google Scholar] [CrossRef]
- Bansal, K.; Kumar, S.; Patil, P.P.; Sharma, S.; Patil, P.B. Genomic data resource of type strains of genus Pseudoxanthomonas. Data Brief 2022, 42, 108145. [Google Scholar] [CrossRef]
- Yu, T.-T.; Zhou, E.-M.; Yin, Y.-R.; Yao, J.-C.; Ming, H.; Dong, L.; Li, S.; Nie, G.-X.; Li, W.-J. Vulcaniibacterium tengchongense gen. nov., sp. nov. isolated from a geothermally heated soil sample, and reclassification of Lysobacter thermophilus Wei et al. 2012 as Vulcaniibacterium thermophilum comb. nov. Antonie Van Leeuwenhoek 2013, 104, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, K.; Chohnan, S.; Ohashi, H.; Hirata, T.; Masaki, T.; Sakiyama, F. Purification, bacteriolytic activity, and specificity of β-lytic protease from Lysobacter sp. IB-9374. J. Biosci. Bioeng. 2003, 95, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Panthee, S.; Hamamoto, H.; Paudel, A.; Sekimizu, K. Lysobacter species: A potential source of novel antibiotics. Arch. Microbiol. 2016, 198, 839–845. [Google Scholar] [CrossRef]
- Zhang, Z.; Yuen, G.Y. Biological control of Bipolaris sorokiniana on tall fescue by Stenotrophomonas maltophilia strain C3. Phytopathology 1999, 89, 817–822. [Google Scholar] [CrossRef]
- Nalin, R.; Simonet, P.; Vogel, T.M.; Normand, P. Rhodanobacter lindaniclasticus gen. nov., sp. nov., a lindane-degrading bacte-rium. Int. J. Syst. Evol. Microbiol. 1999, 49, 19–23. [Google Scholar] [CrossRef]
- Mergaert, J.; Cnockaert, M.C.; Swings, J. Fulvimonas soli gen. nov., sp. nov., a gamma-proteobacterium isolated from soil after enrichment on acetylated starch plastic. Int. J. Syst. Evol. Microbiol. 2002, 52, 1285–1289. [Google Scholar]
- Iasur-Kruh, L.; Zahavi, T.; Barkai, R.; Freilich, S.; Zchori-Fein, E.; Naor, V. Dyella-like bacterium isolated from an insect as a potential biocontrol agent against grapevine yellows. Phytopathology 2018, 108, 336–341. [Google Scholar] [CrossRef]
- Fang, T.; Wang, H.; Huang, Y.; Zhou, H.; Dong, P. Oleiagrimonas soli gen. nov., sp. nov., a genome-sequenced gammaproteo-bacterium isolated from an oilfield. Int. J. Syst. Evol. Microbiol. 2015, 65, 1666–1671. [Google Scholar] [CrossRef]
- Green, S.J.; Prakash, O.; Jasrotia, P.; Overholt, W.A.; Cardenas, E.; Hubbard, D.; Tiedje, J.M.; Watson, D.B.; Schadt, C.W.; Brooks, S.C.; et al. Denitrifying bacteria from the genus Rhodanobacter dominate bacterial communities in the highly contaminated subsurface of a nuclear legacy waste site. Appl. Environ. Microbiol. 2012, 78, 1039–1047. [Google Scholar] [CrossRef]
- Campbell, B.J.; Polson, S.W.; Hanson, T.E.; Mack, M.C.; Schuur, E.A. The effect of nutrient deposition on bacterial communities in Arctic tundra soil. Environ. Microbiol. 2010, 12, 1842–1854. [Google Scholar] [CrossRef]
- Konstantinidis, K.T.; Rosselló-Móra, R.; Amann, R. Uncultivated microbes in need of their own taxonomy. ISME J. 2017, 11, 2399–2406. [Google Scholar] [CrossRef] [PubMed]
- Chun, J.; Oren, A.; Ventosa, A.; Christensen, H.; Arahal, D.R.; da Costa, M.S.; Rooney, A.P.; Yi, H.; Xu, X.-W.; De Meyer, S. Proposed minimal standards for the use of genome data for the taxonomy of prokaryotes. Int. J. Syst. Evol. Microbiol. 2018, 68, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Wittouck, S.; Salvetti, E.; Franz, C.M.; Harris, H.; Mattarelli, P.; O’Toole, P.W.; Pot, B.; Vandamme, P.; Walter, J. A taxonomic note on the genus Lactobacillus: Description of 23 novel genera, emended description of the genus Lactobacillus Bei-jerinck 1901, and union of Lactobacillaceae and Leuconostocaceae. Int. J. Syst. Evol. Microbiol. 2020, 70, 2782–2858. [Google Scholar] [CrossRef] [PubMed]
- Meehan, C.J.; Barco, R.A.; Loh, Y.-H.E.; Cogneau, S.; Rigouts, L. Reconstituting the genus Mycobacterium. Int. J. Syst. Evol. Mi-crobiol. 2021, 71, 004922. [Google Scholar] [CrossRef] [PubMed]
- Konstantinidis, K.T.; Tiedje, J.M. Prokaryotic taxonomy and phylogeny in the genomic era: Advancements and challenges ahead. Curr. Opin. Microbiol. 2007, 10, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Meier-Kolthoff, J.P.; Auch, A.F.; Klenk, H.-P.; Göker, M. Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinform. 2013, 14, 60. [Google Scholar] [CrossRef]
- Konstantinidis, K.T.; Tiedje, J.M. Towards a genome-based taxonomy for prokaryotes. J. Bacteriol. 2005, 187, 6258–6264. [Google Scholar] [CrossRef]
- Qin, Q.-L.; Xie, B.-B.; Zhang, X.-Y.; Chen, X.-L.; Zhou, B.-C.; Zhou, J.; Oren, A.; Zhang, Y.-Z. A proposed genus boundary for the prokaryotes based on genomic insights. J. Bacteriol. 2014, 196, 2210–2215. [Google Scholar] [CrossRef]
- Bansal, K.; Kumar, S.; Kaur, A.; Singh, A.; Patil, P.B. Deep phylo-taxono genomics reveals Xylella as a variant lineage of plant associated Xanthomonas and supports their taxonomic reunification along with Stenotrophomonas and Pseudoxanthomonas. Ge-nomics 2021, 113, 3989–4003. [Google Scholar] [CrossRef]
- Luo, C.; Rodriguez-r, L.M.; Konstantinidis, K.T. MyTaxa: An advanced taxonomic classifier for genomic and metagenomic sequences. Nucleic Acids Res. 2014, 42, e73. [Google Scholar] [CrossRef]
- Park, M.-J.; Kim, Y.J.; Park, M.; Yu, J.; Namirimu, T.; Roh, Y.-R.; Kwon, K.K. Establishment of genome based criteria for classi-fication of the family Desulfovibrionaceae and proposal of two novel genera, Alkalidesulfovibrio gen. nov. and Salidesulfovibrio gen. nov. Front. Microbiol. 2022, 13, 738205. [Google Scholar] [CrossRef] [PubMed]
- Wirth, J.S.; Whitman, W.B. Phylogenomic analyses of a clade within the roseobacter group suggest taxonomic reassignments of species of the genera Aestuariivita, Citreicella, Loktanella, Nautella, Pelagibaca, Ruegeria, Thalassobius, Thiobacimonas and Tropici-bacter, and the proposal of six novel genera. Int. J. Syst. Evol. Microbiol. 2018, 68, 2393–2411. [Google Scholar] [PubMed]
- Orata, F.D.; Meier-Kolthoff, J.P.; Sauvageau, D.; Stein, L.Y. Phylogenomic analysis of the gammaproteobacterial methanotrophs (order Methylococcales) calls for the reclassification of members at the genus and species levels. Front. Microbiol. 2018, 9, 3162. [Google Scholar] [CrossRef]
- Rosselló-Mora, R.; Amann, R. The species concept for prokaryotes. FEMS Microbiol. Rev. 2001, 25, 39–67. [Google Scholar] [CrossRef] [PubMed]
- Orata, F.D.; Kits, K.D.; Stein, L.Y. Complete genome sequence of methylomonas denitrificans strain FJG1, an obligate aerobic methanotroph that can couple methane oxidation with denitrification. Genome Announc. 2018, 6, e00276-18. [Google Scholar] [CrossRef]
- Parks, D.H.; Imelfort, M.; Skennerton, C.T.; Hugenholtz, P.; Tyson, G.W. CheckM: Assessing the quality of microbial genomes recovered from isolates, single cells, and metagenomes. Genome Res. 2015, 25, 1043–1055. [Google Scholar] [CrossRef]
- Seemann, T. Prokka: Rapid prokaryotic genome annotation. Bioinformatics 2014, 30, 2068–2069. [Google Scholar] [CrossRef]
- Asnicar, F.; Thomas, A.M.; Beghini, F.; Mengoni, C.; Manara, S.; Manghi, P.; Zhu, Q.; Bolzan, M.; Cumbo, F.; May, U. Precise phylogenetic analysis of microbial isolates and genomes from metagenomes using PhyloPhlAn 3.0. Nat. Commun. 2020, 11, 1–10. [Google Scholar] [CrossRef]
- Edgar, R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Price, M.N.; Dehal, P.S.; Arkin, A.P. FastTree 2–approximately maximum-likelihood trees for large alignments. PLoS ONE 2010, 5, e9490. [Google Scholar] [CrossRef]
- Bayliss, S.C.; Thorpe, H.A.; Coyle, N.M.; Sheppard, S.K.; Feil, E.J. PIRATE: A fast and scalable pangenomics toolbox for clus-tering diverged orthologues in bacteria. Gigascience 2019, 8, giz119. [Google Scholar] [CrossRef] [PubMed]
- Saha, P.; Krishnamurthi, S.; Mayilraj, S.; Prasad, G.; Bora, T.C.; Chakrabarti, T. Aquimonas voraii gen. nov., sp. nov., a novel gammaproteobacterium isolated from a warm spring of Assam, India. Int. J. Syst. Evol. Microbiol. 2005, 55, 1491–1495. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.-H.; Lai, W.-A.; Lin, S.-Y.; Hameed, A.; Shahina, M.; Shen, F.-T.; Zhu, Z.-L.; Young, L.-S.; Young, C.-C. Chiayiivirga flava gen. nov., sp. nov., a novel bacterium of the family Xanthomonadaceae isolated from an agricultural soil, and emended description of the genus Dokdonella. Int. J. Syst. Evol. Microbiol. 2013, 63, 3293–3300. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.-T.; Yao, J.-C.; Yin, Y.-R.; Dong, L.; Liu, R.-F.; Ming, H.; Zhou, E.-M.; Li, W.-J. Rehaibacterium terrae gen. nov., sp. nov. isolated from a geothermally heated soil sample. Int. J. Syst. Evol. Microbiol. 2013, 63, 4058–4063. [Google Scholar] [CrossRef] [PubMed]
- Kojima, H.; Tokizawa, R.; Fukui, M.J. Mizugakiibacter sediminis gen. nov., sp. nov., isolated from a freshwater lake. Int. J. Syst. Evol. Microbiol. 2014, 64, 3983–3987. [Google Scholar] [CrossRef]
- Ziegler, S.; Waidner, B.; Itoh, T.; Schumann, P.; Spring, S.; Gescher, J.J. Metallibacterium scheffleri gen. nov., sp. nov., an alka-linizing gammaproteobacterium isolated from an acidic biofilm. Int. J. Syst. Evol. Microbiol. 2013, 63, 1499–1504. [Google Scholar] [CrossRef]
- Wei, Z.; Wu, T.; Huang, Y.; Zhu, G.; Zhang, Y.; Geng, Y.; Peng, F.J. Pseudolysobacter antarcticus gen. nov., sp. nov., isolated from soil in Fildes Peninsula, Antarctica. Int. J. Syst. Evol. Microbiol. 2020, 70, 1861–1867. [Google Scholar] [CrossRef]
- Wells, J.M.; Raju, B.C.; Hung, H.-Y.; Weisburg, W.G.; Mandelco-Paul, L.; Brenner, D.J. Xylella fastidiosa gen. nov., sp. nov: Gram-negative, xylem-limited, fastidious plant bacteria related to Xanthomonas spp. Int. J. Syst. Evol. Microbiol. 1987, 37, 136–143. [Google Scholar] [CrossRef]
- Finkmann, W.; Altendorf, K.; Stackebrandt, E.; Lipski, A. Characterization of N2O-producing Xanthomonas-like isolates from biofilters as Stenotrophomonas nitritireducens sp. nov., Luteimonas mephitis gen. nov., sp. nov. and Pseudoxanthomonas broeg-bernensis gen. nov., sp. nov. Int. J. Syst. Evol. Microbiol. 2000, 50, 273–282. [Google Scholar] [CrossRef]
- Palleroni, N.J.; Bradbury, J.F. Stenotrophomonas, a new bacterial genus for Xanthomonas maltophilia (Hugh 1980) Swings et al. 1983. Int. J. Syst. Evol. Microbiol. 1993, 43, 606–609. [Google Scholar] [CrossRef] [PubMed]
- Christensen, P.; Cook, F. Lysobacter, a new genus of nonfruiting, gliding bacteria with a high base ratio. Int. J. Syst. Evol. Mi-crobiol. 1978, 28, 367–393. [Google Scholar] [CrossRef]
- Watanabe, M.; Kojima, H.; Fukui, M. Aerosticca soli gen. nov., sp. nov., an aerobic gammaproteobacterium isolated from crude oil-contaminated soil. Arch. Microbiol. 2020, 202, 1069–1076. [Google Scholar] [CrossRef] [PubMed]
- Johansen, J.E.; Binnerup, S.J.; Kroer, N.; Mølbak, L. Luteibacter rhizovicinus gen. nov., sp. nov., a yellow-pigmented gammapro-teobacterium isolated from the rhizosphere of barley (Hordeum vulgare L.). Int. J. Syst. Evol. Microbiol. 2005, 55, 2285–2291. [Google Scholar] [CrossRef] [PubMed]
- Swings, J.; Gillis, M.; Kersters, K.; De Vos, P.; Gosselé, F.; De Ley, J. Frateuria, a new genus for “Acetobacter aurantius”. Int. J. Syst. Evol. Microbiol. 1980, 30, 547–556. [Google Scholar] [CrossRef]
- Yarza, P.; Yilmaz, P.; Pruesse, E.; Glöckner, F.O.; Ludwig, W.; Schleifer, K.-H.; Whitman, W.B.; Euzéby, J.; Amann, R.; Rossel-ló-Móra, R. Uniting the classification of cultured and uncultured bacteria and archaea using 16S rRNA gene sequences. Nat. Rev. Microbiol. 2014, 12, 635–645. [Google Scholar] [CrossRef]
- Dowson, W.J. On the systematic position and generic names of the Gram-negative bacterial plant pathogens. Zentralblatt Bakte-riol. Parasitenkd. Infekt. Hyg. 1939, 100, 177–193. [Google Scholar]
- Tindall, B.J.; Rosselló-Móra, R.; Busse, H.-J.; Ludwig, W.; Kämpfer, P.J. Notes on the characterization of prokaryote strains for taxonomic purposes. Int. J. Syst. Evol. Microbiol. 2010, 60, 249–266. [Google Scholar] [CrossRef]
- Saddler, G.; Bradbury, J. Family I. Xanthomonadaceae. In Bergey’s Manual of Systematic Bacteriology, Volume 2 (The Proteobacteria), Part B (The Gammaproteobacteria); Springer: Berlin/Heidelberg, Germany, 2005. [Google Scholar]
- Chen, W.-M.; Xie, P.-B.; Tang, S.-L.; Sheu, S.-Y. Coralloluteibacterium stylophorae gen. nov., sp. nov., a new member of the family Lysobacteraceae isolated from the reef-building coral Stylophora sp. Arch. Microbiol. 2018, 200, 473–481. [Google Scholar] [CrossRef]
- Kwon, S.-W.; Kim, B.-Y.; Weon, H.-Y.; Baek, Y.-K.; Go, S.-J. Arenimonas donghaensis gen. nov., sp. nov., isolated from seashore sand. Int. J. Syst. Evol. Microbiol. 2007, 57, 954–958. [Google Scholar] [CrossRef]
- Lee, E.M.; Jeon, C.O.; Choi, I.; Chang, K.-S.; Kim, C.-J. Silanimonas lenta gen. nov., sp. nov., a slightly thermophilic and alkaliphilic gammaproteobacterium isolated from a hot spring. Int. J. Syst. Evol. Microbiol. 2005, 55, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Weon, H.-Y.; Yoo, S.-H.; Kim, Y.-J.; Lee, C.-M.; Kim, B.-Y.; Jeon, Y.-A.; Hong, S.-B.; Anandham, R.; Kwon, S.-W. Rudaea cellulosilytica gen. nov., sp. nov., isolated from soil. Int. J. Syst. Evol. Microbiol. 2009, 59, 2308–2312. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.-H.; Kang, S.-J.; Oh, T.-K. Dokdonella koreensis gen. nov., sp. nov., isolated from soil. Int. J. Syst. Evol. Microbiol. 2006, 56, 145–150. [Google Scholar] [CrossRef]
- Makk, J.; Homonnay, Z.G.; Kéki, Z.; Lejtovicz, Z.; Márialigeti, K.; Spröer, C.; Schumann, P.; Tóth, E.M. Tahibacter aquaticus gen. nov., sp. nov., a new gammaproteobacterium isolated from the drinking water supply system of Budapest (Hungary). Syst. Appl. Microbiol. 2011, 34, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Busse, H.-J.; Kämpfer, P.; Moore, E.; Nuutinen, J.; Tsitko, I.; Denner, E.; Vauterin, L.; Valens, M.; Rosselló-Mora, R.; Salki-noja-Salonen, M.S. Thermomonas haemolytica gen. nov., sp. nov., a gamma-proteobacterium from kaolin slurry. Int. J. Syst. Evol. Microbiol. 2002, 52, 473–483. [Google Scholar] [CrossRef] [PubMed][Green Version]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bansal, K.; Kumar, S.; Singh, A.; Chaudhary, A.; Patil, P.B. Redefining the Taxonomic Boundaries of Genus Xanthomonas. Taxonomy 2023, 3, 452-465. https://doi.org/10.3390/taxonomy3040026
Bansal K, Kumar S, Singh A, Chaudhary A, Patil PB. Redefining the Taxonomic Boundaries of Genus Xanthomonas. Taxonomy. 2023; 3(4):452-465. https://doi.org/10.3390/taxonomy3040026
Chicago/Turabian StyleBansal, Kanika, Sanjeet Kumar, Anu Singh, Arushi Chaudhary, and Prabhu B. Patil. 2023. "Redefining the Taxonomic Boundaries of Genus Xanthomonas" Taxonomy 3, no. 4: 452-465. https://doi.org/10.3390/taxonomy3040026
APA StyleBansal, K., Kumar, S., Singh, A., Chaudhary, A., & Patil, P. B. (2023). Redefining the Taxonomic Boundaries of Genus Xanthomonas. Taxonomy, 3(4), 452-465. https://doi.org/10.3390/taxonomy3040026





