3.1. Comparison of External Morphology
Dorsum. The dorsum of mature
P. parasitica encountered in this region is comparatively smoother (PP-4a) (
Figure 2), without longitudinal rows of prominent tubercles so diagnostic of
P. multilineata from the same region. However, tubercles, specifically those in intermediate position, are detectable in young
P. parasitica. As discussed below, the integrity of these structures is lost during maturation.
Note on terminology: The term ‘tubercle’ is used in this paper in preference to ‘papilla’ without comment with regard to cytology or function. Tubercle as used herein refers to pointed or rounded structures well raised from the tegument and potentially on any annulus. In the P. rugosa clade they are typically found in a regular pattern on the dorsum. In this paper ‘smooth dorsum’ refers to the absence of distinct tubercles, or at most bearing only relatively fewer diminutive structures of little overall import. The term sensillum refers to a simple sensory receptor in the epidermis.
Head. The head of P. parasitica is rounded with a broad oral sucker, whereas the head and oral sucker of P. multilineata is noticeably more attenuated. Thus, the width of the oral sucker in relation to body length of relaxed adults is 10.4% (N = 4) in P. parasitica but only 7.5% (N = 3) in P. multilineata. Furthermore, the ratio of length/width of the oral sucker is 58.2% (N = 4) in P. parasitica and 75.7% (N = 4) in P. multilineata.
Annulation. Annuli of the neck segments of P. parasitica are perceptibly foreshortened, i.e., detectably less subdivided, compared to P. multilineata. Thus, in the former species from segment V to as far as segments VII (PP-1), VIII (PP-4a) or IX (PP-2, PP-3), respectively, the first two annuli (a1 + a2) of each respective segment are barely divided, whereas in P. multilineata these serially homologous neck segments are clearly divided. Based on limited data, similar foreshortening may also occur in the caudal region in that segment XXV in P. parasitica appears to be uni-annulate, whereas this segment is consistently bi-annulate in P. multilineata.
The noticeably greater subdivisions in the head and tail segments of P. multilineata are interpreted functionally as manifestation of greater extensibility of P. multilineata compared to the seemingly less mobile P. parasitica.
Caudal sucker. The caudal sucker of P. parasitica is relatively larger and more substantial than that of P. multilineata. Thus, the width of the caudal sucker in relation to body length of relaxed adults is 17.3% (N = 4) in P. parasitica but only 10.8% (N = 18) in P. multilineata. The more substantial caudal sucker in P. parasitica is interpreted functionally as more of a holdfast organ rather than one of locomotion as suggestive in the case of P. multilineata. Interestingly, the caudal sucker of the former species functions amply after death, if pressure is initially applied.
Ventral stripes. The venter of
P. parasitica typically bears 8 to 10 prominent longitudinal stripes along its entire length, each comprised of distinctive green chromatophores (
Figure 3A, sg). These ventral stripes can be useful for field identification. However, they are not reliable for taxonomic purposes in that they fade or disappear under certain conditions of preservation (PP-1, PP-2, PP-3) (
Figure 3B,C), especially in ethanol.
Colour change. P. parasitica is capable of conspicuous colour change. In this study this phenomenon was observed only once in which a recently captured mature individual (PP-4a) changed within minutes from dark brown to brilliant green and cream. The latter colour was retained after fixation and preservation (
Figure 1A and
Figure 2A). Colour change was not observed to any degree in
P. multilineata from the Outer Banks [
9].
Placobdellid head pattern. The ‘placobdellid head pattern’ of
P. parasitica is basically similar to that which characterises
P. multilineata but the two species differ in detail. In
P. parasitica this head pattern is comprised of a light-coloured, short rod mid-dorsally in segments IV and V, with an indentation at VIa1 + a2, before expanding widely toward the margins at annulus VIa3 (
Figure 2A–C, pp). This medial rod appears to be comprised of homogeneous cellular constituents distinguishable from the cream-coloured patterns posterior to segment VI. In
P. multilineata, the pattern typically consists of three consecutive, slightly contiguous cream-coloured patches in IV, V and VI, and as a rule the cream colour of annulus VIa3 does not extend exaggeratedly toward the margins as consistently seen in
P. parasitica, at least in this study.
Metameric marginal pattern. The ‘metameric marginal pattern’ along the full length of the body of
P. parasitica is basically similar to that so characteristic of
P. multilineata. However, that of
P. parasitica differs slightly in that the two contiguous light annuli (a3, a1) of each segment at the margins are comprised of numerous small cells which typically extend medially to about the paramarginal position (
Figure 2A–C, ma). On the other hand, those of
P. multilineata are comprised of fewer ‘colossal’ cells (plus a contiguous unpigmented zone), and are mostly confined to the margins [
9] (figs 6A–10A).
Mid-dorsal cream-coloured pattern. Adults of
P. parasitica in the Outer Banks display a characteristically variable, cream-coloured pattern at the dorsal mid-line from segment VII to XXIVa3 (
Figure 2). This design is derivable from a notional longitudinal stripe in which the cream colour expands or recedes in respective annuli to form a pattern which is more or less unique to the individual. Unlike typical
P. multilineata there is no longitudinal brown stripe at the dorsal midline of
P. parasitica.
Intermediate row of cream-coloured spots. The dorsum of adult
P. parasitica from the Outer Banks displays a longitudinal array of cream-coloured spots in intermediate position, extending from segment VI to XXIV (PP-4a) (
Figure 2A). They are most evident in anterior segments VIII–X, and again in caudal segments XXII–XXIV, but lose integrity in mid-body segments in some specimens (PP-2) (
Figure 2B). These intermediate spots are on the a2 annulus and accordingly are serially homologous to the intermediate tubercles so diagnostic of
P. multilineata. The intermediate spots in adult
P. parasitica lack distinct tubercles. However, some spots that have a slightly raised centre comprised of a disorganised concentration of cells.
Interestingly, the dorsal pigmentation of P. parasitica from the Outer Banks is not strictly bilaterally symmetrical, especially evident in mid-body segments where spots may occur irregularly in paramedial or paramarginal positions. This adds to the camouflaged individuality of each specimen. Of developmental interest, much of the constituent pigment is confined strictly to a respective annulus, implying some sort of localised mechanism at work.
Pre-anal segments XXV–
XXVII. The three pre-anal segments XXV–XXVII of
P. parasitica lack prominent paired tubercles so characteristic of
P. multilineata [
9] (fig. 11A). Instead, in the homologous position corresponding to the pre-anal tubercles in
P. multilineata there are three pairs of cream-coloured spots in
P. parasitica (
Figure 2A). Each bears a slightly raised cellular centre, but no structural tubercles. The cream-colour of each spot is centred on these central cells.
3.2. Comparison of Internal Morphology
Length and position of proboscis. In
P. parasitica the proboscis is long and tapering with its tip positioned in situ anterior to the ‘brain’ in all specimens examined (
Figure 8). The position of the posterior end of the proboscis varies among the Outer Banks specimens, from IX/X (PP-1, 36.5 mm), X (PP-2, 22.0 mm) to X/XI (PP-4a, 39.8 mm).
Crook at posterior end of proboscis. In the largest specimen in this study (PP-4a) the posterior end of the proboscis is distinctly crooked along the dorso-ventral axis (
Figure 6), whereas in the remaining specimens the proboscis is straight, without a crook (
Figure 7, pc). Significantly, in
P. multilineata the posterior end of the proboscis is crooked in virtually all specimens, and is diagnostic of that species. Furthermore, the end of proboscis appears to be noticeably further back in
P. multilineata [
9] (fig. 4, ‘slightly anterior to g. XI’).
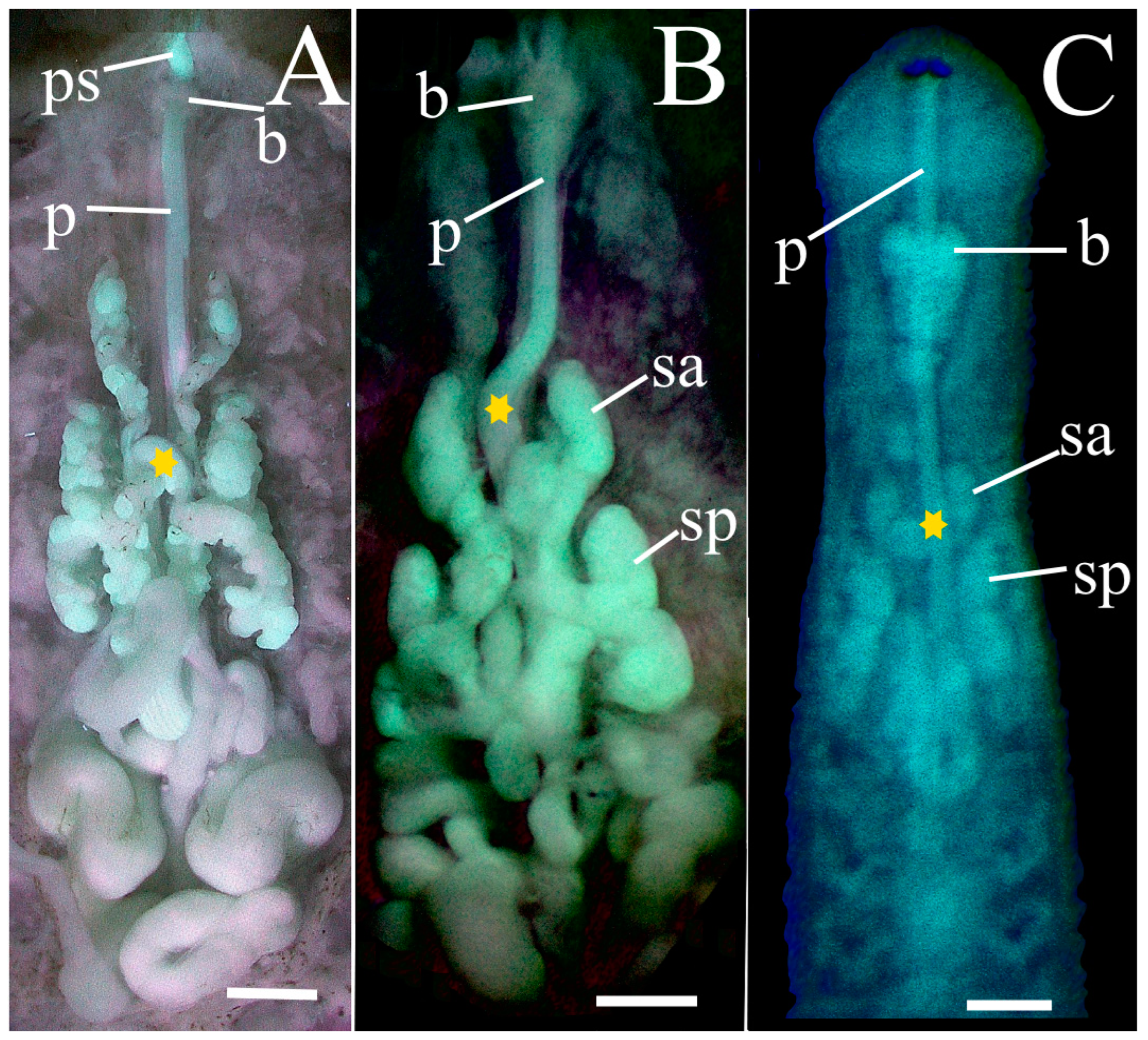
Asymmetrical deflection of proboscis. The proboscis of
P. parasitica, at least in Outer Banks specimens examined, consistently has a peculiar asymmetrical deflection at segment IX. In the mature specimen (PP-4a) this deflection constitutes an improbable fully formed 360° loop of the proboscis in this segment (
Figure 6A, star;
Figure 7, pad). In less mature specimens this deflection is less developed, appearing either as a sizeable curve (PP-2) (
Figure 6B, star;
Figure 8, pad), or simply as a slight deviation (PP-1) (
Figure 4, star). In contrast, the proboscis of all
P. multilineata examined is straight all along its length, and lacks an asymmetrical deflection.
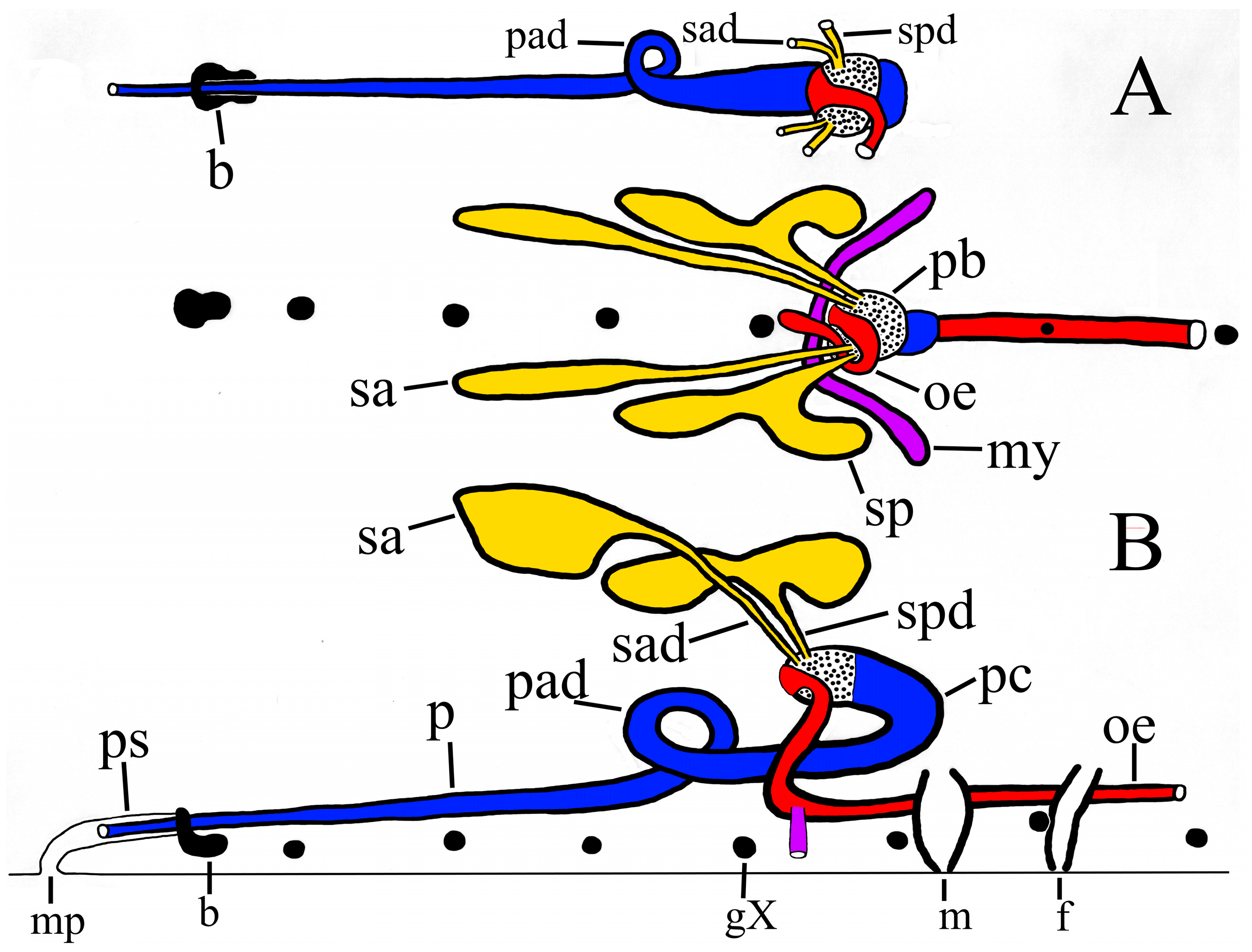
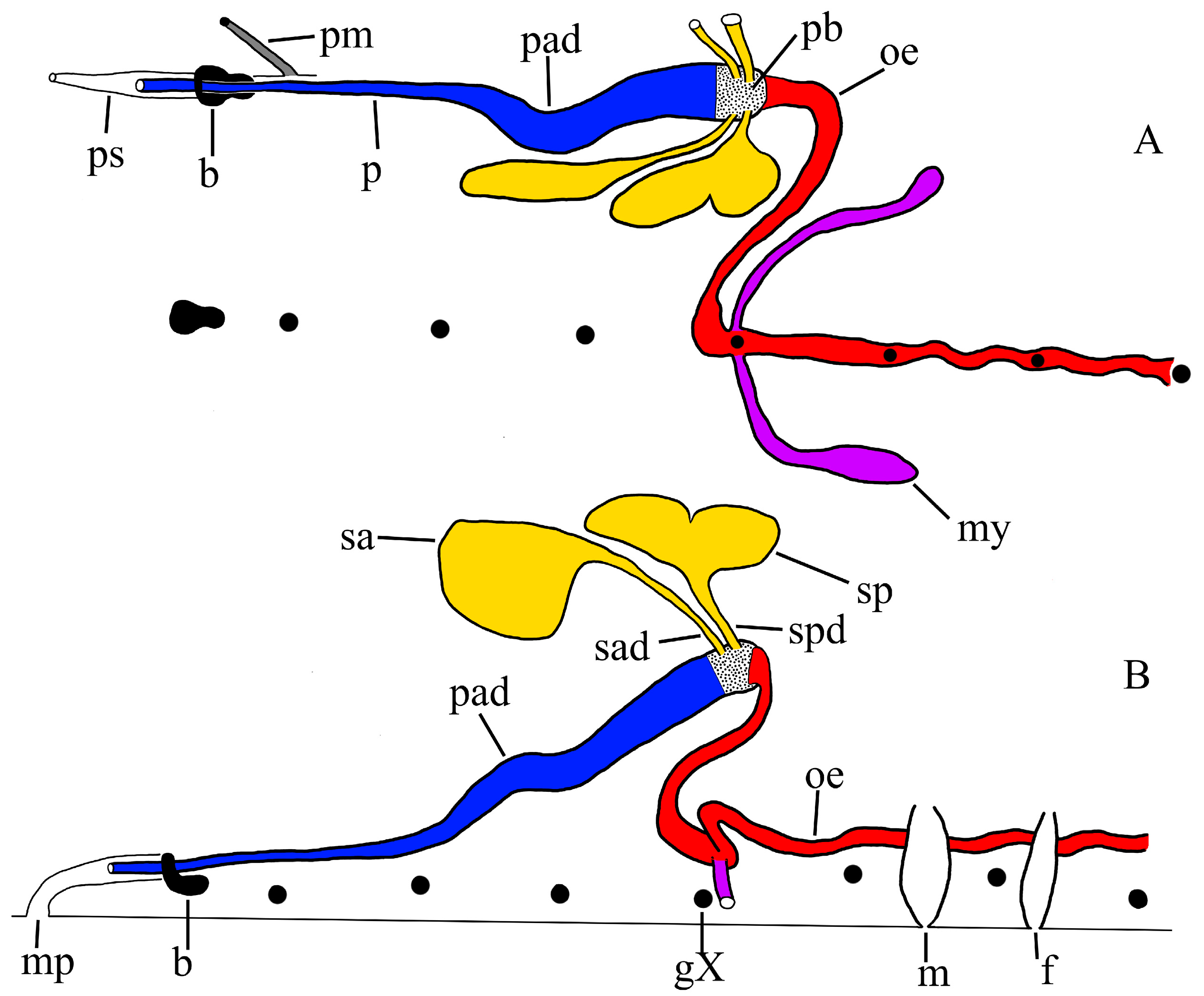
Compact salivary glands. All dissected specimens of P. parasitica in this paper have two bilateral pairs of compact salivary glands, each with an individual ductule (i.e., no common ductule) which leads into the base of the proboscis.
Note on terminology: Salivary ‘gland’ is used in this paper for convenience. They are not true glands with a lumen typical of vertebrate glands. The leech salivary gland is actually an aggregation of large unicellular exocrine cells each of which has an exceedingly long sub-cellular process leading from the soma into the proboscis, as demonstrated in this species by Moser and Desser (1995) [
6]. These individual processes from each gland together form a clearly recognisable bundle. For convenience each multi-cellular bundle is simply called ‘ductule’ in this paper.
Independent salivary ductules. The respective ductules of the anterior and posterior salivary glands of
P. parasitica enter the base of the proboscis independently or at most barely abutting (
Figure 7, sad, spd), whereas the ductules of
P. multilineata join a measurable distance from the proboscis to form a clear and unmistakable common ductule [
9] (fig. 4, sb). This taxonomically important distinction alone appears to distinguish
P. parasitica from its tuberculated counterpart
P. rugosa clade [
6,
9].
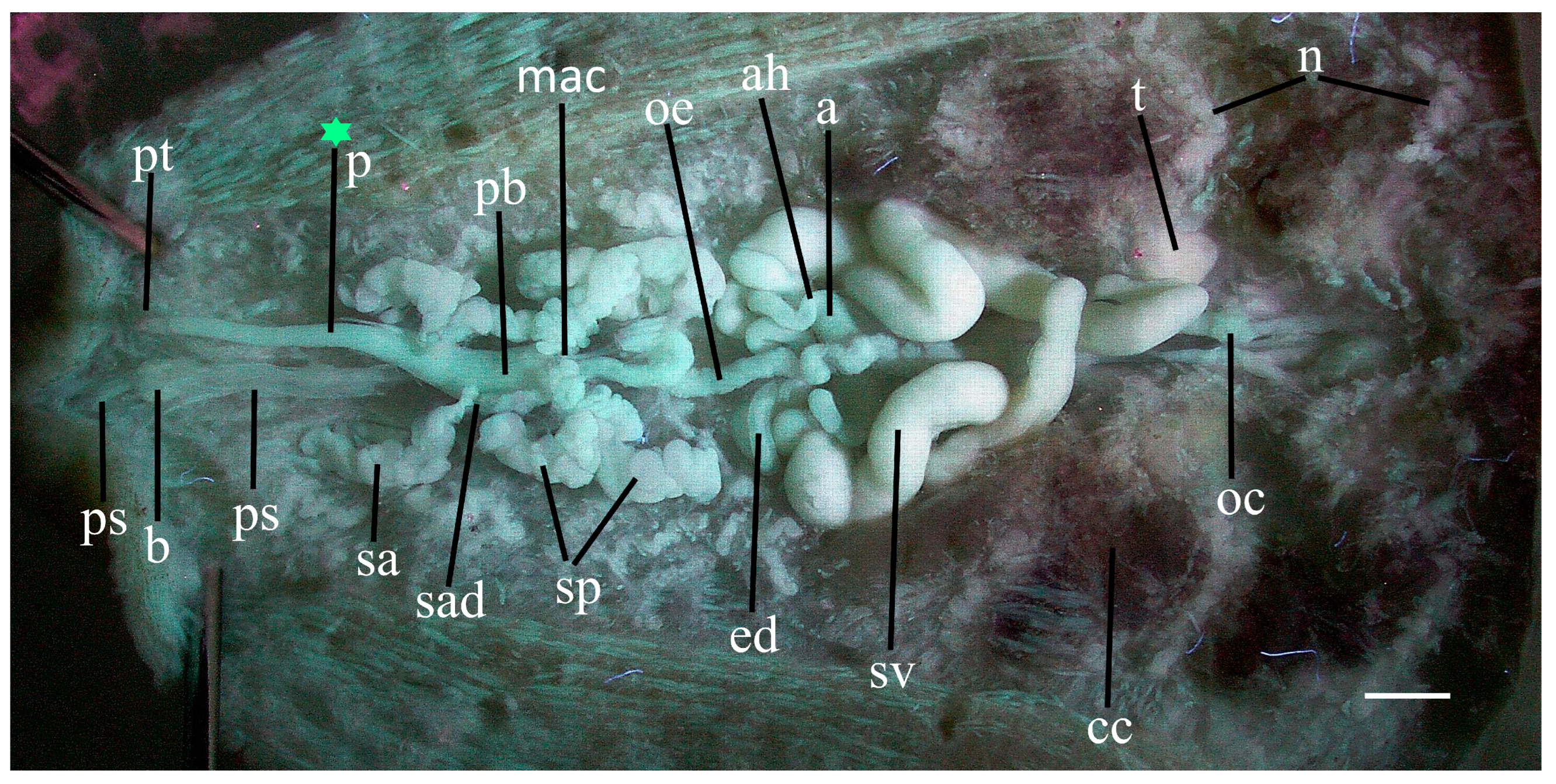
Median annectant lobe. Dissected specimen PP-1 (36.5 mm) reveals a peculiar accumulation of about 25–30 salivary cells at the mid-line at about IX/X. These cells together constitute a delicate median lobe which more or less connects the two posterior salivary glands in this specimen (
Figure 4, mac). A similar annectant lobe may also occur in the other two dissected specimens of
P. parasitica. However, due to its location at the mid-line it may have been cut inadvertently in order to view the underlying structures. This median annectant lobe appears not to occur in
P. multilineata, but this should be re-examined.
Larger and less numerous salivary cells. The salivary glands of
P. parasitica contain significantly fewer but larger cells than those of
P. multilineata, at least in Outer Banks specimens. Each salivary gland of
P. parasitica contains on the order of only 50 cells, typically 175 µm in diameter, but up to 385 µm. Those of
P. multilineata are much more numerous but smaller, up to only about 100 µm. The posterior gland of
P. parasitica is ‘squatty’ and confined to its segment, extending only to just beyond g. X (
Figure 4,
Figure 6 and
Figure 7, sp), whereas the posterior gland of
P. multilineata is typically elongated and extends as far as XI/XII [
9] (fig. 4, sp).
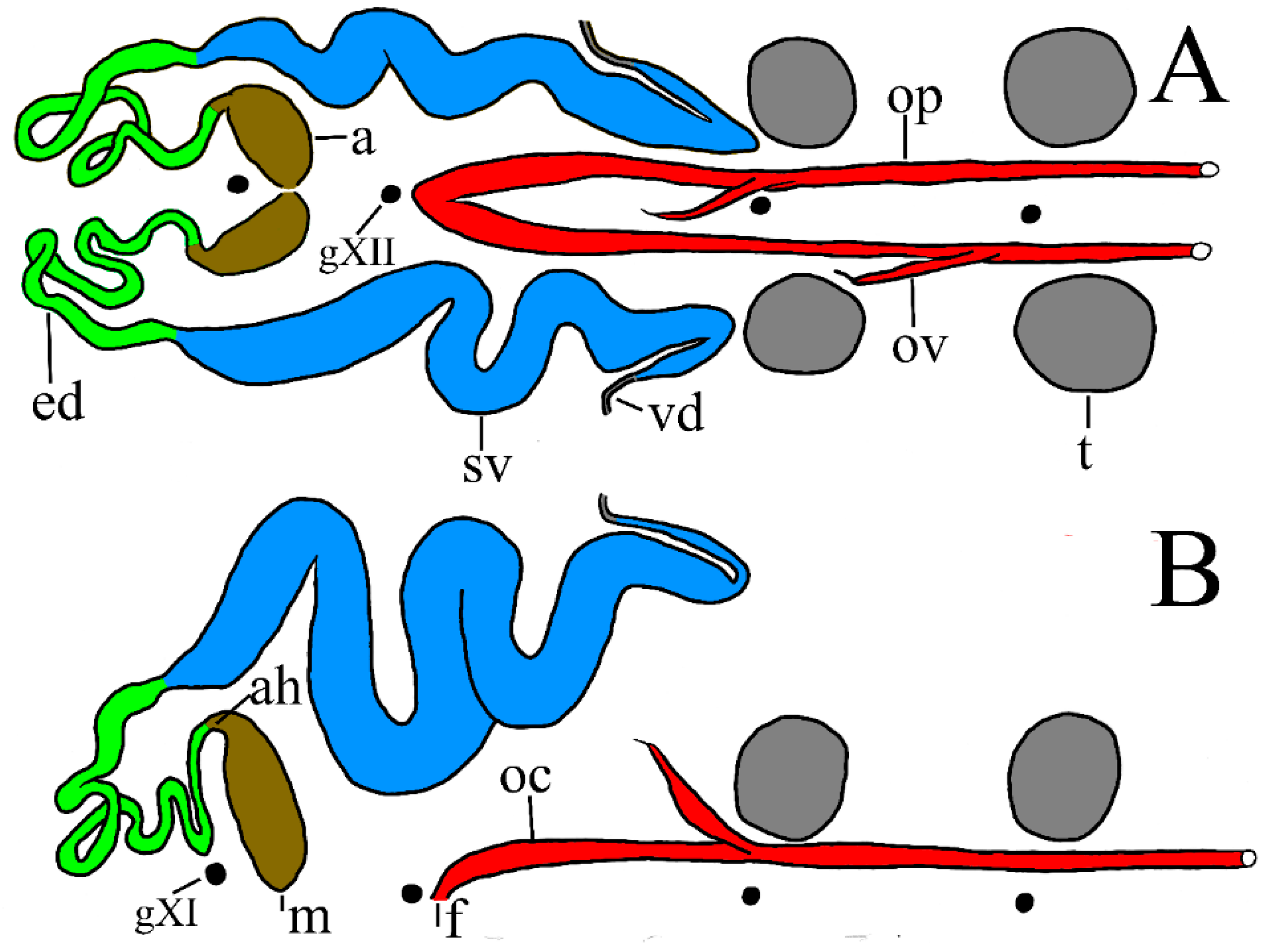
Reproductive systems (
Figure 9). Differences between the male anatomy of
P. parasitica and
P. multilineata are noted in the various dissections, but owing to the physiologically plastic nature of these structures taxonomic significance requires further scrutiny. In
P. parasitica the narrow, tubular ejaculatory duct is confined to segment XI and joins the seminal vesicle at about level of the atrium (PP-4a). In one mature specimen (PP-1) the ejaculatory duct extends a little more anterior, to X/XI. In
P. multilineata the ejaculatory duct is longer and joins the seminal vesicle at about the level of g. XII [
9] (fig. 5, ed). The seminal vesicles of mature
P. parasitica are particularly prominent and swollen (
Figure 4, sv), and typically observable through the ventral tegument (
Figure 3A). The apparent juncture between the ejaculatory duct and seminal vesicle in
P. parasitica is at level of the atrium (i.e., behind g. XI), consistently more anterior than the much less swollen seminal vesicles of
P. multilineata (i.e., level of g. XII). In both species the seminal vesicle joins the vas deferens just anterior to level of g. XIII. No significant differences between species were noted in the female reproductive system.
Based on limited information, the resting length of gravid/brooding individuals of
P. parasitica averaged 38.2 mm (range 36.5–39.8 mm; N = 2), and closely resembled that of
P. multilineata in the same region (mean, 36.6 mm; range, 28.0–45.0 mm; N = 23) [
9].
Mycetomes. The mycetomes of
P parasitica are more substantial than those of
P. multilineata. The flattened ductus is relatively wide throughout its length, and typically has a perceptible bulbous ending (
Figure 7 and
Figure 8, my).
Nephridia. Inexplicably, the nephridia of
P parasitica are relatively larger and more conspicuous (
Figure 4, n) than those of
P multilineata.
3.3. Taxonomic Diagnosis of P. parasitica from Outer Banks
The foregoing section describes external and internal feature of P. parasitica collected from the Outer Banks region, with particular attention to individual variability and differences from the outgroup species P. multilineata from the same locality. This methodology identifies those characters which occur in all adult individuals of P. parasitica, but do not occur in the P. rugosa clade. Taken together these invariant characters constitute a formal diagnosis which defines P. parasitica, at least from the Outer Banks.
Placobdella parasitica (Say, 1824) (Outer Banks).
Diagnosis (post-hatchling). Dorsum is essentially smooth without conspicuous, metameric tubercles. Dorsum displays a bold, irregular and highly variable cream-coloured pigment pattern. The long, tapering proboscis has a peculiar asymmetrical deflection at segment IX. The salivary ductules from the anterior and posterior salivary glands enter the base of the proboscis independently (or at most barely abutting each other). In other words, the respective ductules do not merge to form a clear and unmistakable common ductule a measurable distance from the base of the proboscis as is the case for P. multilineata. In addition, a sometimes useful but taxonomically unreliable character for identifying P. parasitica is the presence of 8–10 broad blue-green ventral stripes.
Hatchlings of P. parasitica are difficult to distinguish externally from P. multilineata in that both possess discrete white pigment patches at intervals, as well as small, but distinct tubercles.
3.4. Morphological Homogeneity of P. parasitica throughout United States and Canada
The current study is the first description of P. parasitica from the southern part of the immense range of this species. In this context, the question is addressed whether specimens from the Outer Banks are anatomically similar to specimens from northern parts of its range. Towards this end, this section critically reviews what is known regarding morphological features of P. parasitica from disparate localities, namely, Minnesota (type locality), Ontario, Illinois and Massachusetts. These four northern localities represent distances of 1000 to 2100 km from the Outer Banks.
P. parasitica from Minnesota (type locality).
P. parasitica has been recently redescribed from its type locality at Lily Lake, Waseka County, Minnesota [
7]. This redescription was based on a neotype plus nine other mature individuals from Minnesota, one of which was dissected and illustrated. In general, it agrees with the account of this species from the Outer Banks. However, some relatively minor discrepancies, omissions or clarifications are noted below.
P. parasitica from Ontario. In contrast to the scarcity of
P. parasitica in the warm swamps of the Outer Banks, this species abounds in the cold lakes of the Algonquin Provincial Park in southeast Ontario [
22,
23]. It is taxonomically fortuitous that the ultrastructure of the proboscis complex of this species was investigated as part of a morphological study [
6]. By way of summary, the proboscis and salivary glands of five specimens of
P. parasitica from Ontario were remarkably similar to that of Outer Banks specimens. Of taxonomic relevance for the current study these authors also meticulously described the proboscis complex of the allied tuberculated turtle leech
P. rugosa, also from Ontario.
P. parasitica from Illinois. Moore [
21] gave the first internal description of this species under the name
P. parasitica. He did not give numbers of specimens examined nor specific locations, other than from ‘Illinois’. He specifically commented on the large size of this ‘tortoise’ leech, citing that some individuals could extend to over 100 mm (4 inches). Moore was also the first to compare the external and internal anatomy of
P. parasitica and
P. rugosa, from Illinois.
P. parasitica from Massachusetts. The first internal description of
P. parasitica was based on five specimens from Charles River near Boston, Massachusetts, under the name
Clepsine plana [
19,
20]. The latter name is now recognised as a junior synonym for
P. parasitica [
7]. Presciently, Whitman was the first to illustrate the taxonomically important proboscis complex of this species [
19](fig. 5), [
24](fig. 23.4). However, in contrast to
P. parasitica presented in this paper, the proboscis of the Boston specimen was clearly illustrated as straight, without a bend or deflection in segment IX, nor with a dorso-ventral crook at its posterior end. These discrepancies may be an error or oversight. However, Whitman was a meticulous researcher, and even made a specific point about his attention to detail, and the accuracy of his reproductions [
20] (p. 411).
External features. Specimens from all five localities displayed a dorsum with a bold irregular, cream-coloured or yellowish pigment pattern, and a relatively smooth dorsum (never linear rows of large metameric tubercles, as in the
P. rugosa clade). This external uniformity of
P. parasitica was further corroborated by exemplary photographs of additional specimens from Minnesota and Connecticut [
8] (figs 1 and 2).
Size. The largest specimen of P. parasitica encountered in the Outer Banks (PP-4a, resting length, 39.8 mm) was notably smaller than those found in Minnesota (59.1 mm), Massachusetts (60.0 mm) and Illinois (extended length, 100 mm). This apparent discrepancy may simply be sampling bias.
Ventral stripes. Specimens of
P. parasitica from Minnesota [
7] (fig. 4), Massachusetts [
20] (fig. 2) and the Outer Banks (
Figure 3A) displayed 8–10 prominent bluish or greenish longitudinal stripes on the venter. Moore [
21] did not specifically mention ventral stripes in specimens from Illinois, but much later he relied on ventral longitudinal stripes in a popular and influential key for identifying this species [
24] (p. 550).
By way of caution, while such distinctive stripes are an undeniably useful guide for identifying this species, their presence or absence is not taxonomically sound for diagnosis, especially after preservation (
Figure 3B,C). Furthermore, very similar ventral stripes appeared to occur in at least one other nominal species of
Placobdella [
25] (p. 69). Moreover, interpreting ventral pigmentation can be subjective and misleading. For example, ventral stripes of some individuals of
P. multilineata may broadly overlap those found in
P. parasitica [
5] (fig. 10C).
Pre-anal segments. An apparent discrepancy between Minnesota (type locality) and Outer Banks specimens lies in the pre-anal segments (XXV–XXVII). In some Minnesota specimens three pairs of pre-anal tubercles (‘papillae’) were reported in these segments. However, there is reason to believe the relevant illustration may show instead three pairs of cream-coloured spots in these segments [
7] (fig. 3A). Such spots also occurred in Outer Banks specimens, but they were not tubercles in that each spot encompassed slightly disorganised bumps at most. In other words, the presence of distinct pre-anal tubercles is not diagnostic of
P. parasitica.
Internal Features
Proboscis complex. Specimens from all five disparate localities reviewed herein possessed two pairs of compact salivary glands and a long slender proboscis whose narrow tip typically extended anterior to the ‘brain’.
Length and position of proboscis. The posterior extension of the proboscis varied somewhat between localities. In most cases, the end of the proboscis was positioned at X (Illinois, Massachusetts, Ontario), whereas in the type locality it was recorded as posterior to X/XI (Minnesota). The position of the posterior end of the proboscis also varied within the Outer Banks population, and accordingly is not considered taxonomically significant.
Crook of posterior end of the proboscis. In the current study, a dorso-ventral crook of the posterior end of the proboscis occurred in only one specimen (PP-4a) encountered in the Outer Banks (
Figure 7, pc), the other specimens lack such a crook (
Figure 8). Interestingly, a crook also occurred in a specimen from the type locality in Minnesota [
7] (fig. 5) and, less demonstrably, from Ontario [
6] (fig. 4). No such crook was recorded from Illinois nor from Massachusetts. Owing to its variability, the presence or absence of a dorso-ventral crook is not considered taxonomically significant. On the other hand, such a crook occurred in nearly all specimens of
P. multilineata from the Outer Banks [
9] (fig. 4).
Asymmetrical bend or deflection of the proboscis at segment IX. This peculiar, possibly unique feature was characteristic of all post-hatching specimens of
P. parasitica examined from the Outer Banks. Significantly, it was also found in specimens from the type locality, as illustrated in an excellent dissection from Minnesota in which a veritable loop was present [
7] (fig. 5). Similarly, a distinct asymmetrical bend of the proboscis also occurred in specimens from Ontario [
6] (fig. 4).
Although such a deflection was not recorded from Illinois nor from Massachusetts, it must be considered that with further data this previously overlooked feature of the proboscis may be diagnostic of post-hatching
P. parasitica throughout its range. In this context, it is taxonomically important that this morphological character appeared to be absent in hatchlings of
P. parasitica from the Outer Banks (
Figure 5C), and significantly in the
P. rugosa clade [
6] (fig. 3), [
9] (fig. 4).
Independent salivary ductules. In all post-hatchling specimens examined from the Outer Banks, the ductules from the anterior and posterior salivary glands, respectively, entered the base of the proboscis independently, or nearly so. This taxonomically important feature may be present in the specimen illustrated from Ontario [
6] (fig. 4) but it could not be confirmed unequivocally in specimens from Minnesota, Illinois nor Massachusetts. However, J. P. Moore the doyen of American hirudinology, illustrated what appears to be independent salivary ductules in
P parasitica [
24] (fig. 23.4).
Conversely, none of the specimens of
P. parasitica from any of the localities, including the Outer Banks, have anterior and posterior salivary ductules which join at a measurable distance from the proboscis to form a clear and unmistakable common ductule which enters the base of the proboscis. This latter configuration is characteristic of the
P. rugosa clade [
9] (fig. 4), [
6] (fig. 3, arrowhead, their
ornata) and reliably sets these tuberculated forms apart from
P. parasitica.
Median annectant salivary lobe. Moore [
21] was first to describe a peculiar-overlooked feature of the salivary complex of
P. parasitica from Illinois. ‘Very frequently, but apparently not invariably’, a median lobe of salivary tissue links the two posterior salivary glands. A very similar annectant lobe had been illustrated earlier by Whitman [
19] (fig. 4) without comment from a specimen from Massachusetts. Finally, at least one specimen from the Outer Banks displayed what appears to be a similar median lobe (
Figure 4, mac). This possibly unique annectant lobe is not diagnostic of
P. parasitica in that it appears not to be present in all specimens from Illinois nor from the Outer Banks. Of possible taxonomic significance, however, such a median annectant lobe was not present in
P. rugosa from Illinois [
21] nor in
P. multilineata from the Outer Banks [
9]. Interestingly, in the current study a similar aggregation of annectant cells occurs in the eyes of
P. parasitica (
Figure 5D, mac).
Reproductive systems. Unfortunately, details of the reproductive systems from the four northern localities were inadequate for comparison with those from the Outer Banks. The nature of the ejaculatory duct and seminal vesicle from the type locality of
P. parasitica could be particularly informative [
7] (fig. 6).
By way of summary, morphological features of
P. parasitica from these four geographically disparate localities were remarkably similar to those described herein from the Outer Banks, with only minor discrepancies or omissions. Therefore, evidence from comparative anatomy suggests that the smooth turtle leech
P. parasitica, is morphologically homogeneous throughout the immense range of this species. This finding concurs well with a parallel study of low molecular diversity of this species [
8]. Parenthetically, a molecular phylogenetic study of
P rugosa documents a similar low diversity [
12,
13].
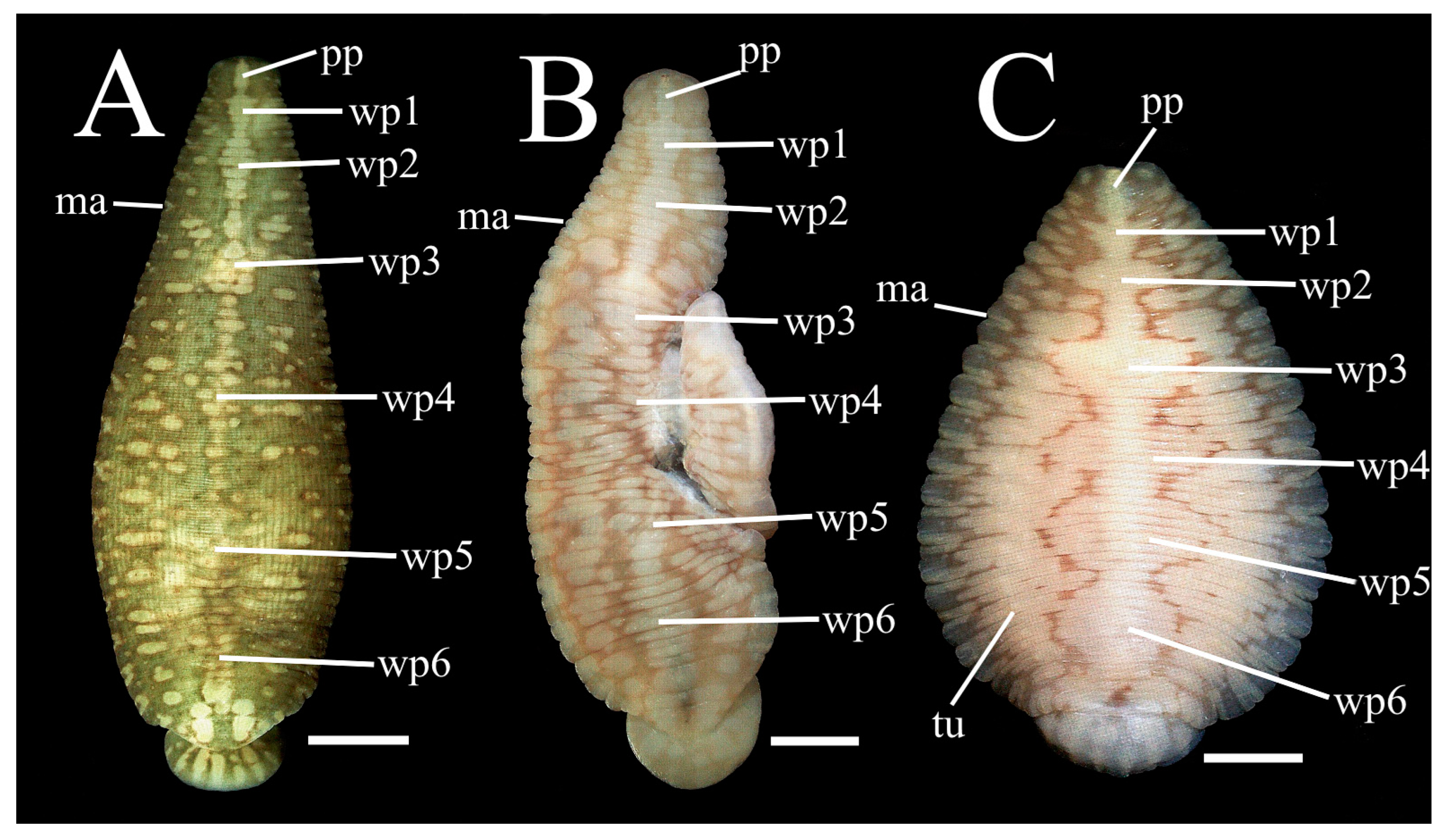
3.5. Similarity of Hatchlings of P. parasitica and P. multilineata
This section focuses on morphological aspects of early development of
P. parasitica, with unexpected results. It was found that in terms of tuberculation and pigmentation hatchlings of
P. parasitica more closely resemble
P. multilineata than adults of their own species (
Figure 10). In fact, it is remarkably difficult to distinguish these two species at the hatchling stage. Dorsal tubercles, so characteristic of
P. multilineata at all life stages, also occur in hatchlings of
P. parasitica, but in this species these structures do not develop and accordingly obfuscate during maturation (
Figure 1,
Figure 11 and
Figure 12). Furthermore, hatchlings of both
P. parasitica and
P. multilineata display very similar white pigment patches at discrete mid-dorsal intervals, but this pigment pattern loses overall integrity during maturation in
P. parasitica (
Figure 1 and
Figure 10).
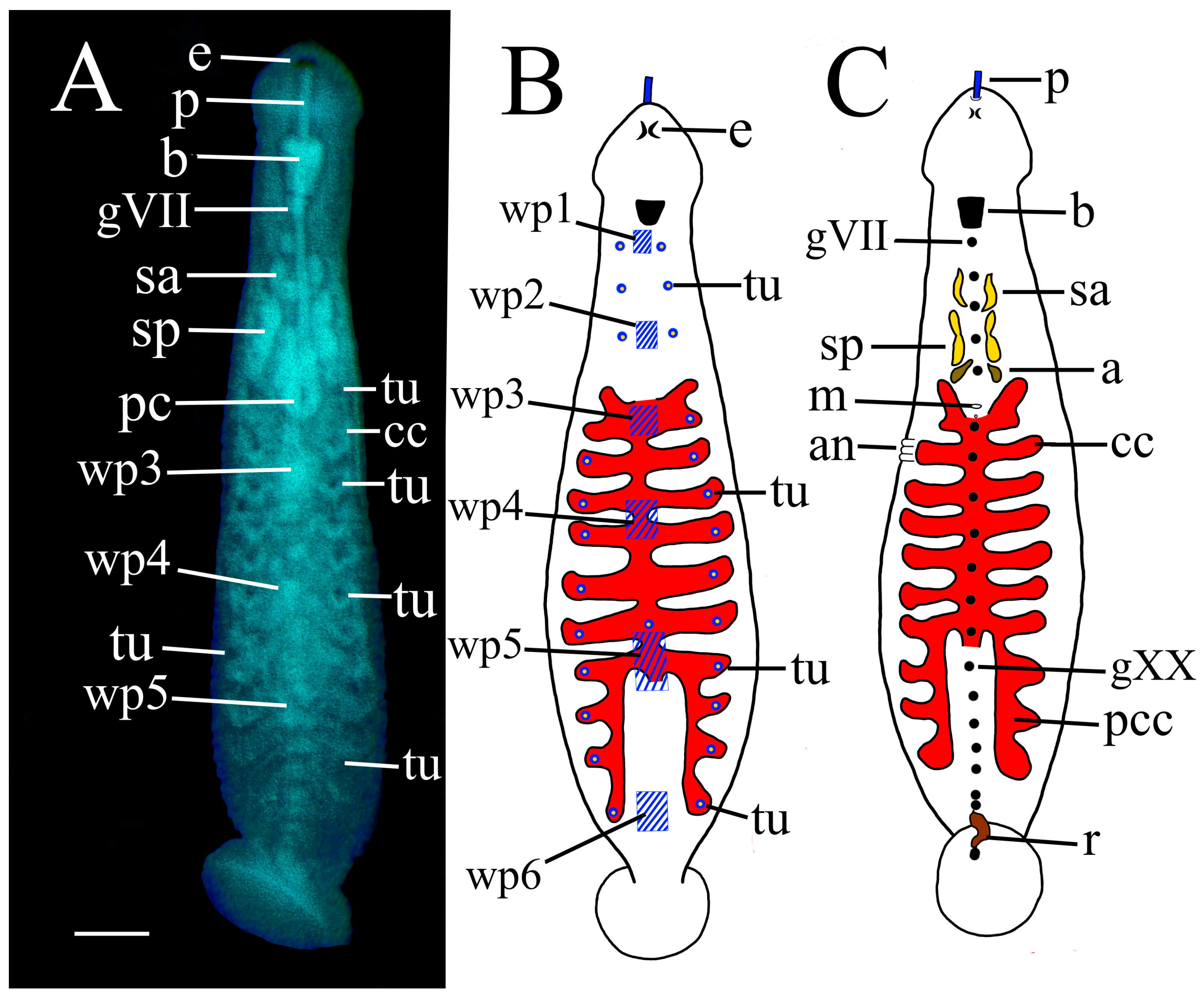
Tubercle obfuscation in
P. parasitica versus prominence in
P. multilineata. Of considerable morphological significance, hatchlings of
P. parasitica differ from adults in having small but distinct dorsal tubercles (
Figure 11, tu). These are in longitudinal rows in intermediate position on the a2 annulus in segments XIII–XXII, inclusively (
Figure 12, tu). Furthermore, these align near the respective tips of the underlying crop caeca, a feature seen in young and adult
P. multilineata [
9] (fig.15). In contrast, in adult
P. parasitica tubercles are substantially obliterated or lacking altogether (
Figure 2A,B). In younger, juvenile
P. parasitica there are still some discernible tubercle-like protuberances in intermediate position on the a2 annulus in segments IX–XXIV, inclusively (
Figure 2C, tu).
Whereas the tubercles of
P. parasitic become more indeterminate with age (
Figure 1A), those of
P. multilineata become more prominent (
Figure 1B). This is interpreted as the first example of character displacement in the Hirudinea [
26,
27]. This reflects a significant taxonomic distinction between smooth and tuberculated turtle leeches throughout much of North America as discussed below.
White pigment patches of hatchling
P. parasitica and
P. multilineata diverge during development. Hatchlings of
P. parasitica invariable display six white pigment patches (
Figure 10, wp) not discretely evident in the adult of this species. In hatchlings, these white patches are invariably centred at the midline at segments VIa3 (nuchal), IXa2 (pre-genital), XIIa2 (genital), XV/XVI (mid-crop), XVIII/XIX (pre-intestine) and XXIIa1 (anal), respectively (
Figure 11 and
Figure 12). During growth and development these same patches in
P. parasitica, but not
P. multilineata, widen into irregular cream-coloured expanses on either side of the mid-line (
Figure 2C). With further maturation these may become broadly confluent anteriorly and posteriorly, but they remain centred at the same ordinal locations as the respective patches of the hatchling. In mature adults this underlying pattern may become obscured or lost entirely, especially in mid-body segments XI–XXII (
Figure 2A,B). In other words, the characteristically variable dorsal pigment pattern of adult
P. parasitica (39.8 mm) is derivable during development from a more organised discrete condition in the hatchling (6.0 mm). The pigment pattern of the juvenile (
Figure 2C, 12.8 mm) is clearly intermediate between that of the hatchling and the adult.
These white pigment patches in hatchling
P. parasitica closely resemble in position and number ‘white shields’ described elsewhere in hatchling and post-hatchling
P. multilineata [
9] (fig. 8), with two notable differences. The position of the last or anal patch appears to differ between the two species. In
P. parasitica it is centred at XXIIa1, whereas in
P. multilineata it is more posterior at XXIII-XXIV.
In addition, in my earlier description of
P. multilineata only five, rather than six, white shields were recounted [
9]. In retrospect these actually correspond in
P. parasitica to white patches 2 to 6, inclusively. This is because the first white patch in
P. multilineata was misinterpreted as part of the placobdellid head pattern. This is an opportunity to correct this discrepancy and bring the two species into closer harmony, i.e., both species have six white patches with the first one at XVI-VII. This discrepancy was anticipated in the
P. multilineata study in that the white patch in the nuchal region of VI-VII was observed to be ‘comprised of different and less durable kind of pigmentation’ [
9] (p. 14).
In the context of the challenging task of separating the two turtle leech species at an early hatchling stage, there appears to be a morphologically useful difference in detail of white patches 1 and 6, respectively, as discussed briefly above. There may also be a species difference in the nature of the cells which make up the white patches, being diffused and rounded aggregations in
P. parasitica (
Figure 11C,D), but compact, linear and organised in
P. multilineata [
9] (fig. 8).