Study of Weak-Acid-Dissociable and Free Cyanide Oxidation by Ozone Injection into Gold Mine Pulp
Abstract
1. Introduction
1.1. Generalities of Cyanide
1.2. INCO Process
2. Materials and Methods
3. Results
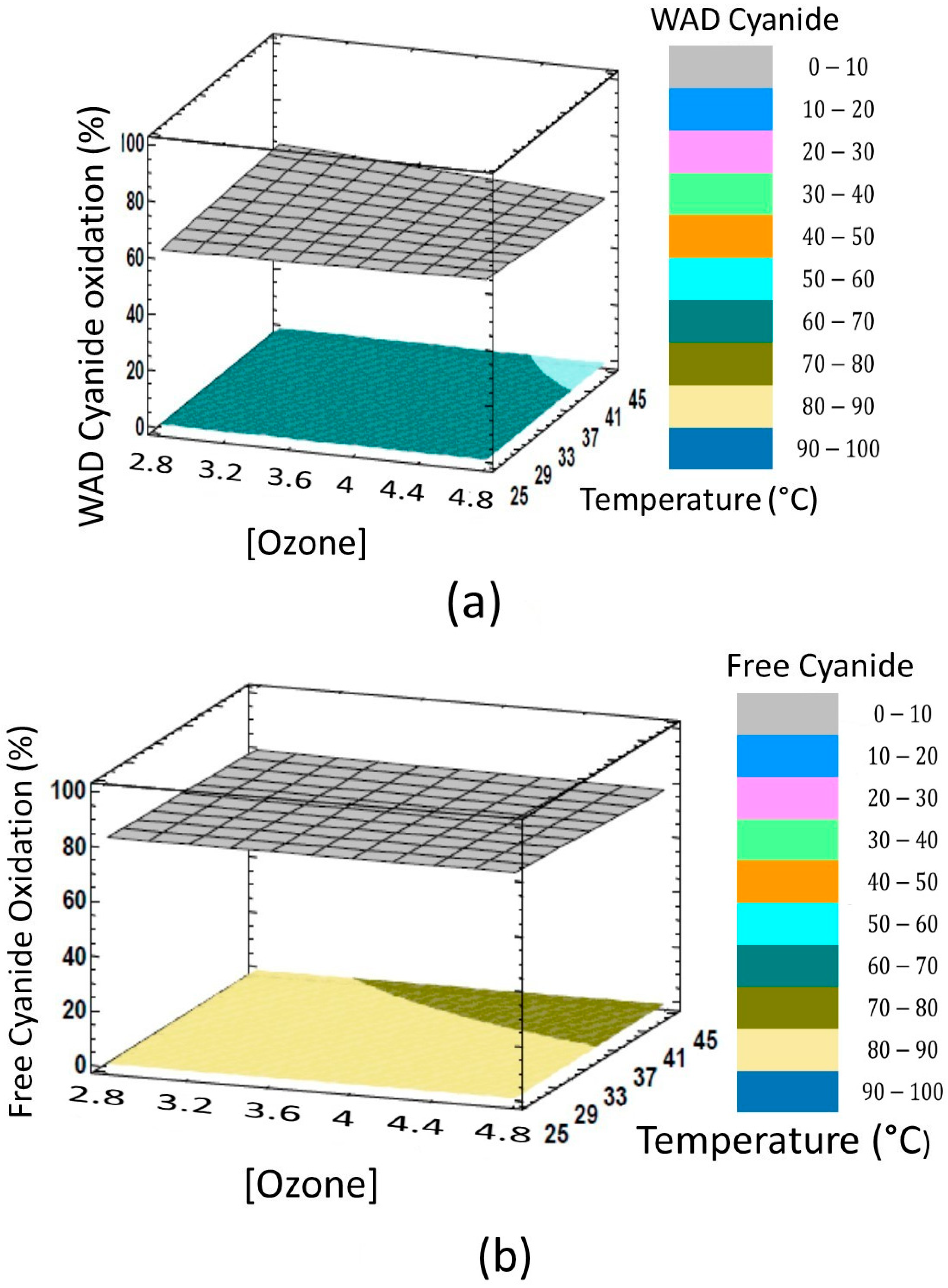
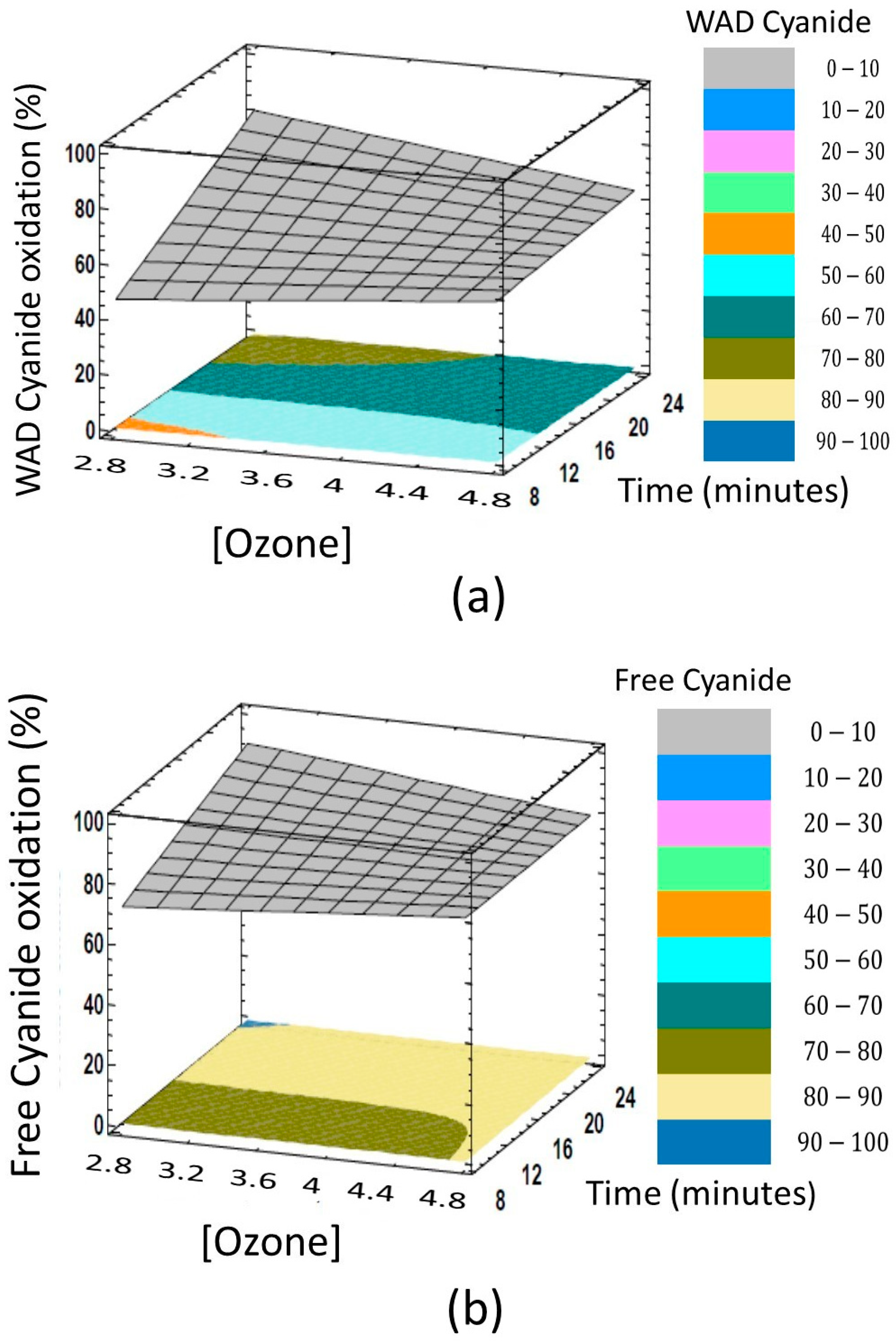
3.1. Volumetric Plots
3.2. Results in Surface Plots
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- David, A.D.; Rajat, S.G.; George, M.W.C. Cyanide in Water and Soil, Chemistry, Risk, and Management; Taylor & Francis Group: Abingdon, UK, 2006; ISBN 978-1-56670-666-7. [Google Scholar]
- Danielle, H.; Paul, B.; Coby, J. Cyanide detoxification of gold cyanidation tails and process streams. Miner. Process. Extr. Metall. 2012, 121, 228–236. [Google Scholar] [CrossRef]
- Julio César, P.D.; Óscar Fabián, H.C. Comportamiento electroquímico del Cianuro. Ing. Y Desarro. 2008, 24, 63–76. [Google Scholar]
- Edison, G.P.; Paola Andrea, C.M.; Claudia, C.J.; y Tomás Felipe, P.V. Oxidación Fotocatalítica de Cianuro; Universidad EAFIT: Medellín, Colombia, 2005; ISSN 1692-0694. [Google Scholar]
- Gurol, M.D.; Bremen, W.M.; Holden, T.E. Oxidation of Cyanide in Industrial Wastewater by Ozone. Environ. Prog. 1985, 4, 46–51. [Google Scholar] [CrossRef]
- Joseph, T.B.; Stephen, D.E.; David, A.D. Development of a Plant Uptake Model for Cyanide. Int. J. Phytoremediat. 2006, 8, 25–43. [Google Scholar] [CrossRef] [PubMed]
- Ricardo, L.L. Destrucción de Cianuro en Efluentes de la Industria Minera Mediante el Empleo de Reductores Químicos; Catálisis Industrial S.A. de C.V.: Tizayuca, México, 2024; pp. 285–290. Available online: https://www.ocmal.org/wp-content/uploads/2017/03/destruccion_de_cianuro.pdf (accessed on 27 January 2026).
- Terry, I.M.; Michael, M.B.; Adrian, S. Chemistry and Treatment of Cyanidation Wastes. In Treatment and Recovery of Cyanide; Chapter 6; Mining Journal Books Ltd.: London, UK, 2001; pp. 326–333. [Google Scholar]
- Patricia, G.L. Degradación de Cianuros Mediante Oxidación Química en Efluentes Industriales. Master’s Thesis, Departamento de Química Orgánica e Inorgánica, Universidad de Oviedo, Oviedo, Spain, 2012. [Google Scholar]
- Roth, J.A.; Sullivan, D.E. Solubility of ozone in water. Ing. Eng. Chem. Fundam. 1981, 20, 137–140. [Google Scholar] [CrossRef]
- Bin, A.K. Ozone Solubility in Liquids. Ozone Sci. Eng. J. Int. Ozone Assoc. 2006, 28, 67–75. [Google Scholar] [CrossRef]
- Piervandi, Z. Pretreatment of refractory gold minerals by ozonation before the cyanidation process: A review. J. Environ. Chem. Eng. 2023, 11, 109013. [Google Scholar] [CrossRef]
- Freitas, J.; Horta, D. Impacts of cyanide in gold mining and cyanide removal methodologies in liquid waste from gold processing. Rev. De Metal. 2023, 59, e247. [Google Scholar] [CrossRef]
- Jiménez-Prieto, Y.J.; Esperanza-Pérez, G.; Ramírez-González, S.; Alomas-Vicente, I. Assessment of Technological alternatives for cyanides waste waters management in gold ores processing plant. Rev. Cuba. De Química Habana Cuba 2020, 32, 218–231. [Google Scholar]
- Pandey, N.; Dutta, S.; Biswas, P. Cyanide mitigation at steel metallurgical process’s effluent. Water Air Soil Pollut. 2023, 234, 682. [Google Scholar] [CrossRef]
- Mamelkina, M.A.; Herraiz-Carbone, M.; Cotillas, S.; Lacasa, E.; Saez, C.; Tuunila, R.; Sillanpaa, M.; Hakkinen, A.; Rodrigo, M.A. Treatment of mining wastewater polluted with cyanide by coagulation processes: A mechanistic study. Sep. Purif. Technol. 2020, 237, 116345. [Google Scholar] [CrossRef]
- Schneider, C.L.; Matiolo, E.; Neumann, R.; Gomes, O.F.M. Beneficiamento de Minérios. Recursos Minerais no Brasil: Problemas e Desafios; Academia Brasileira de Ciên-cias: Rio de Janeiro, Brazil, 2019; pp. 257–262. Available online: https://www.abc.org.br/IMG/pdf/doc-7006.pdf (accessed on 27 January 2026).
- Zhang, H.; Zhang, Y.; Qiao, T.; Hu, S.; Liu, J.; Zhu, R.; Yang, K.; Li, S.; Zhang, L. Study on ultrasonic enhanced ozone oxidation of cyanide-containing wastewater. Sep. Purif. Technol. 2022, 303, 122258. [Google Scholar] [CrossRef]
- Botz, M.M.; Mudder, T.I.; Akcil, A.U. Chapter 35—Cyanide treatment: Physical, chemical, and biological processes. In Gold Ore Processing, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 619–645. [Google Scholar]
- Krylova, L.N.; Seliverstov, A.F.; Ryabtsev, D.A. Cleaning of industrial waters containing cyanides by using a combination of ozone and hydrogen peroxide. Metallurgist 2021, 64, 1331–1339. [Google Scholar] [CrossRef]






| Phase | Classification | Compound |
|---|---|---|
| Liquid (water) | Free cyanide | , |
| Metal–cyanide complexes | Weak complexes , , , , Strong complexes , , , | |
| Cyanate, thiocyanate | , | |
| Organocyanides | Nitriles, cyanohydrins |
| Mining Unit in the World | CNTOT (mg/L) | Reagent Usage (g/g) CHTOT | |||
|---|---|---|---|---|---|
| Before | After | SO2 | Lime | Cu2+ | |
| Colosseum | 374 | 0.4 | 4.6 | 0.12 | 0.04 |
| Ketza River | 150 | 5.0 | 6.0 | 0 | 0.30 |
| Equity | 175 | 2.3 | 3.4 | 0 | 0.03 |
| Casa Berardi | 150 | 1.0 | 4.5 | - | 0.10 |
| Westmin Premier | 150 | <0.2 | 5.8 | - | 0.12 |
| Golden Bear | 205 | 0.3 | 2.8 | - | - |
| Variable (Factors) | Units | Value to Be Evaluated (Levels) | ||
|---|---|---|---|---|
| Minimum | Medium | Maximum | ||
| O3/O2 flow | SCFM | 3 | 6 | 9 |
| pH | - | 8 | 9 | 10 |
| Temperature | °C | 25 | 35 | 45 |
| NH4HSO3 concentration | kg/ton | 1 | 1.5 | 2 |
| Reaction time | Minutes | 8 | 16 | 24 |
| Group of 10 Samples | [%] | [%] |
|---|---|---|
| 1 | 65.0555 ± 5.092 | 89.5088 ± 3.149 |
| 2 | 70.7480 ± 6.401 | 92.7790 ± 3.535 |
| 3 | 68.4090 ± 5.556 | 88.4700 ± 5.876 |
| 4 | 61.2150 ± 8.510 | 64.9540 ± 7.576 |
| 5 | 65.8910 ± 8.092 | 81.0210 ± 6.734 |
| 6 | 72.1350 ± 10.101 | 88.1150 ± 4.891 |
| Factor | Low | High | Optimal Values for WAD Cyanide Oxidation | Optimal Values for Free Cyanide Oxidation |
|---|---|---|---|---|
| [Ozone] Volts | 2.8 | 4.8 | 2.8 | 2.8 |
| O3/O2 flow SCFM | 3.0 | 9.0 | 9.0 | 9.0 |
| pH | 8.0 | 10.0 | 10.0 | 8.0 |
| Temperature °C | 25.0 | 45.0 | 25.0 | 25.0 |
| [NH4HSO3] kg/ton | 1.0 | 2.0 | 2.0 | 1.0 |
| Time (minutes) | 8.0 | 24.0 | 24.0 | 24.0 |
| Variable (Factors) | WAD Cyanide | Free Cyanide |
| O3/O2 (flow rate) |  |  |
| pH |  |  |
| Temperature |  |  |
| NH4HSO3 concentration |  |  |
| Reaction time |  |  |
 —high impact;
—high impact;  —low impact.
—low impact.Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
López, C.M.; Landeros, S.O.; Hernández, H.H.; Pérez, E.A.; Arteaga, C.E.; González, A.S.; Hernández, J.M. Study of Weak-Acid-Dissociable and Free Cyanide Oxidation by Ozone Injection into Gold Mine Pulp. Mining 2026, 6, 9. https://doi.org/10.3390/mining6010009
López CM, Landeros SO, Hernández HH, Pérez EA, Arteaga CE, González AS, Hernández JM. Study of Weak-Acid-Dissociable and Free Cyanide Oxidation by Ozone Injection into Gold Mine Pulp. Mining. 2026; 6(1):9. https://doi.org/10.3390/mining6010009
Chicago/Turabian StyleLópez, Coraquetzali Magdaleno, Saúl Ortiz Landeros, Héctor Herrera Hernández, Eugenia Aldeco Pérez, Carlos Estrada Arteaga, Antonia Sandoval González, and Jorge Morales Hernández. 2026. "Study of Weak-Acid-Dissociable and Free Cyanide Oxidation by Ozone Injection into Gold Mine Pulp" Mining 6, no. 1: 9. https://doi.org/10.3390/mining6010009
APA StyleLópez, C. M., Landeros, S. O., Hernández, H. H., Pérez, E. A., Arteaga, C. E., González, A. S., & Hernández, J. M. (2026). Study of Weak-Acid-Dissociable and Free Cyanide Oxidation by Ozone Injection into Gold Mine Pulp. Mining, 6(1), 9. https://doi.org/10.3390/mining6010009










