Cancer Therapy-Related Left Ventricular Dysfunction: Are There New Gatekeepers?
Abstract
1. Introduction
2. Cancer Therapy and Cardiotoxicity: An Overview
| Treatments | Mechanism of Cardiotoxicity | CRTCD*/LV Systolic Dysfunction | References |
|---|---|---|---|
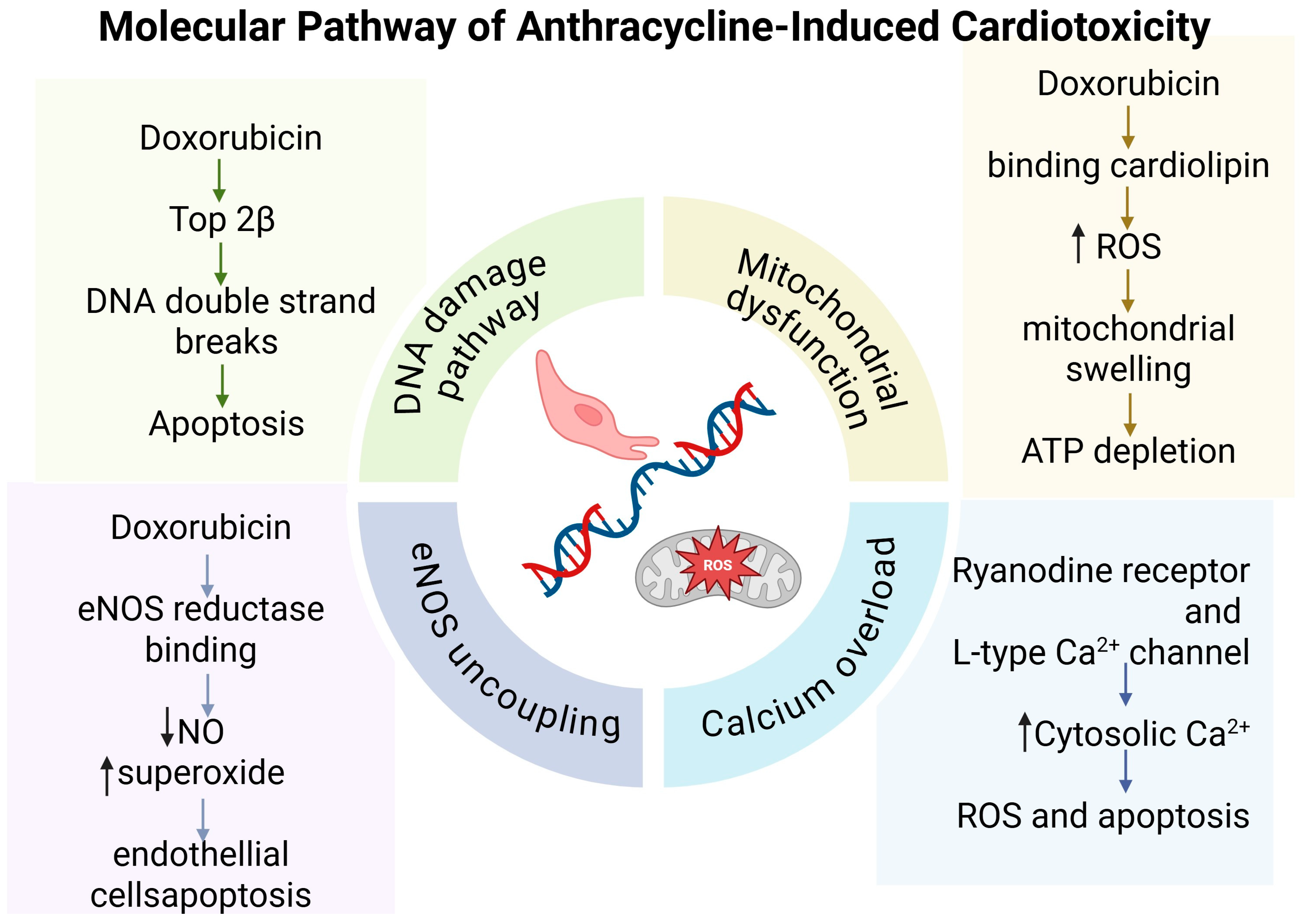
| Anthracyclines | Top2β inhibition: DNA breaks; ROS production; mitochondrial dysfunction; calcium dysregulation; apoptosis; eNOS uncoupling. | Cumulative and dose-dependent; occurs years after therapy. | [17,22,24] |
| HER2-Targeted Therapies | Oxidative stress; suppression of autophagy; mitochondrial gene expression alteration. | Typically reversible; increased risk when combined with anthracyclines. | [7,31,32] |
| Proteasome Inhibitors | UPP inhibition: proteotoxic stress; autophagy suppression; vascular dysfunction; ROS and inflammation. | HFpEF, subclinical diastolic dysfunction; higher risk in elderly or those with comorbidities. | [39,40,41] |
| Tyrosine Kinase Inhibitors | ER stress; ROS burst and lipid peroxidation; upregulation of pro-inflammatory and fetal gene programs. | LV dysfunction; cardiac remodeling associated with long-term use. | [48,49,50] |
| MEK/BRAF Inhibitors | MAPK pathway inhibition: LV dysfunction; reversibility upon dose reduction. | LV dysfunction; underreported due to patient selection. | [51,52] |
| Immune Checkpoint Inhibitors | T CD4+ and CD8+ activation; autoantibody generation; cytokine storm; PD-L1 pathway suppression; molecular mimicry: myocarditis, fibrosis, HF. | Myocarditis, HF; rare but often severe or fatal. | [55,56] |
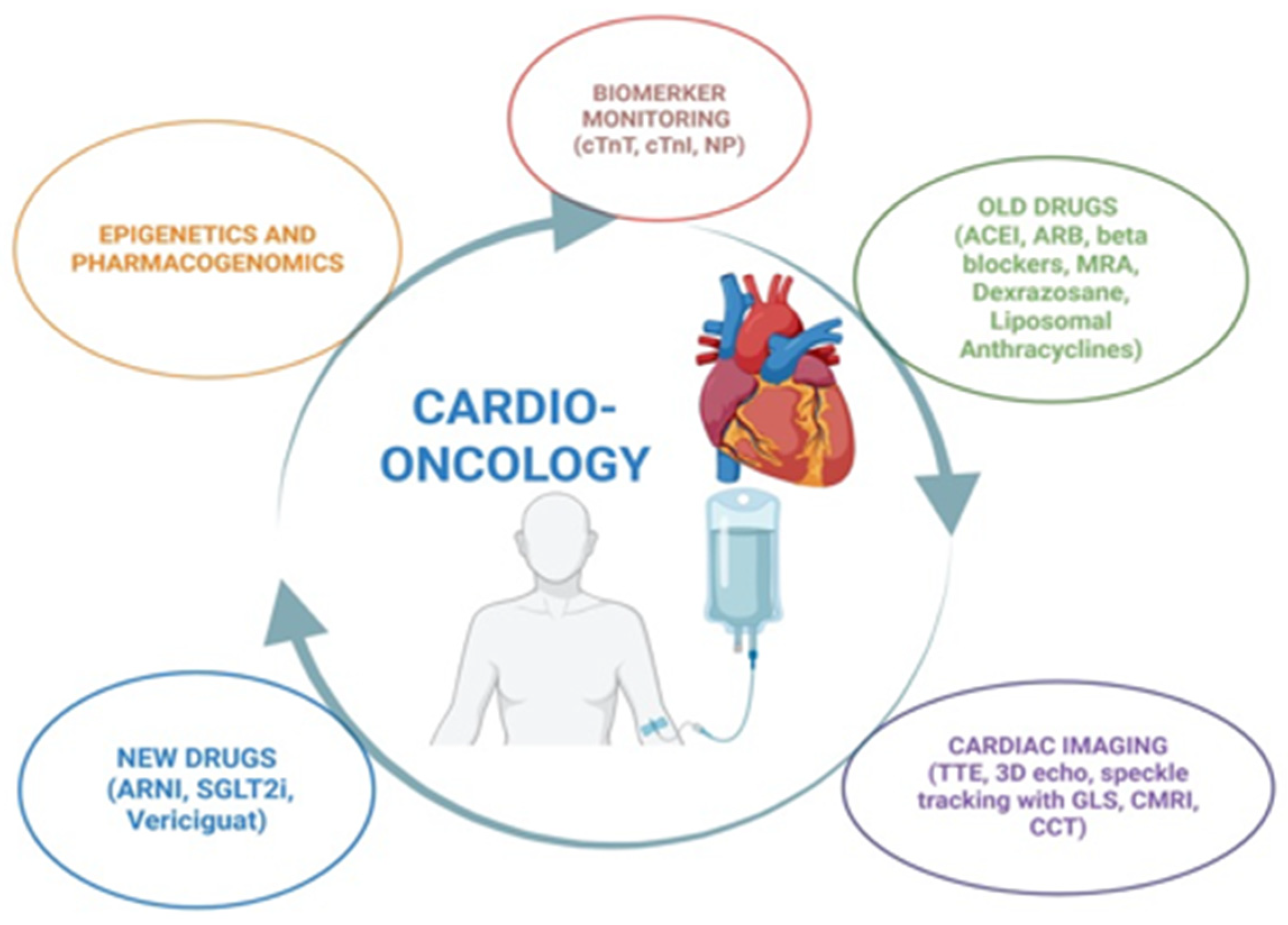
3. The “Old Gatekeepers”: Early Detection, Biomarkers, and Risk Stratification of CVDs
4. The “Old Gatekeepers”: Drugs Which Protect Against Ventricular Dysfunction
4.1. Angiotensin-Converting Enzyme Inhibitors and Angiotensin Receptor Blockers
4.2. Beta-Blockers
4.3. Mineralcorticoid Receptor Antagonist
4.4. Dexrazoxane and Liposomal Anthracyclines
4.5. Statins
5. The “New Gatekeepers”: Drugs Which Protect Against Ventricular Dysfunction
5.1. Angiotensin Receptor-Neprilysin Inhibitors
5.2. Sodium–Glucose Cotransporter Inhibitors-2
5.3. Soluble Guanylate Cyclase Stimulators
6. Future Direction: Pharmacogenomics and Epigenetics
7. Future Direction: Gene Therapy
8. Conclusions
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
Abbreviations
| ACB | ATP-binding cassette |
| ACEIs | angiotensin-converting enzyme inhibitors |
| ADP | adenosine diphosphate |
| AMPKa | AMP-activated protein kinase a |
| ARBs | angiotensin receptor blockers |
| ARNI | angiotensin receptor-neprilysin inhibitor |
| Atg7 | autophagy-related protein 7 |
| ATP | adenosine tri-phosphate |
| β1ARs | β1-adrenergic receptors |
| BCR-ABL | breakpoint cluster region–Abelson leukemia |
| BNP | B-type natriuretic peptide |
| BRAF | v-Raf murine sarcoma viral oncogene homolog B1 |
| CCT | cardiac computed tomography |
| CD4+ | cluster of differentiation 4+ |
| CD8+ | cluster of differentiation 8+ |
| cGMP | cyclic guanosine monophosphate |
| CMR | cardiac magnetic resonance |
| cTnI | cardiac troponin I |
| cTnT | cardiac troponin T |
| CTRCD | cancer therapy-related cardiac dysfunction |
| DNA | deoxyribonucleic acid |
| ECV | extracellular volume |
| EF | ejection fraction |
| eNOS | endothelial nitric oxide synthase |
| EGFR | endothelial growth factor receptor |
| ER | endoplasmic reticulum |
| ERK | extracellular signal-regulated kinase |
| FT | feature tracking |
| GLS | global longitudinal strain |
| HER2 | human epidermal growth factor receptor 2 |
| HF | heart failure |
| HFA-ICOS | Heart Failure Association–International Cardio-Oncology Society |
| HFpEF | heart failure with preserved ejection fraction |
| hiPSC-CMs | induced pluripotent stem cell-derived cardiomyocytes |
| HSF-1 | heat shock factor 1 |
| Hsp 25 | heat shock protein 25 |
| ICIs | immune checkpoint inhibitors |
| IL-6 | interleukin-6 |
| IRE1α- | inositol-requiring transmembrane kinase/endoribonuclease 1α-spliced |
| LV | left ventricle |
| MAP1LC3 | microtubule-associated protein 1 light chain 3 |
| MAPK | mitogen-activated protein kinase |
| MEK | mitogen-activated protein kinase kinase |
| MM | multiple myeloma |
| MRA | mineralcorticoid receptor antagonist |
| MRI | magnetic resonance imaging |
| mTOR | mechanistic target of rapamycin |
| mTORC1 | mechanistic target of rapamycin complex1 |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| NLP3 | NOD-like receptor protein 3 |
| NO | nitric oxide |
| NT-ptoBNP | N-terminal pro-BNP |
| O2− | superoxide |
| p53 | protein 53 |
| PDL-1 | programmed death ligand-1 |
| PIs | proteasome inhibitors |
| PP2A | phosphoprotein phosphatase 2A |
| PPRA | peroxisome proliferator-activated receptor |
| RAAS | renin–angiotensin aldosterone system |
| RAF | rapid accelerated fibrosarcoma |
| RV | right ventricle |
| RCR | respiratory control ratio |
| RNA | ribonucleic acid |
| ROS | oxygen reactive species |
| sGC | soluble guanylate cyclase |
| SGLT2 inhibitors | sodium–glucose cotransporter inhibitors-2 |
| SLC | soluble carrier transporters |
| SOD | superoxide dismutase |
| TBARS | thiobarbituric acid reactive substances |
| TKIs | tyrosine kinase inhibitors |
| TNFα | tumor necrosis factor-α |
| Top2β | topoisomerase 2β |
| TTE | trans-thoracic echocardiogram |
| UlK1 | Unc-51-like autophagy-activating kinase 1 |
| UPP | ubiquitin proteasome |
| VEGF | vascular endothelial growth factor |
| XBP1 | X-box binding protein 1 |
References
- Haj-Yehia, E.; Michel, L.; Mincu, R.I.; Rassaf, T.; Totzeck, M. Prevention of cancer-therapy related cardiac dysfunction. Curr. Heart Fail. Rep. 2025, 22, 9. [Google Scholar] [CrossRef]
- Desai, A.; Reddy, N.K.; Subbiah, V. Top advances of the year: Precision oncology. Cancer 2023, 129, 1634–1642. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, R.C.; Antoniou, A.C.; Fruk, L.; Rosenfeld, N. The future of early cancer detection. Nat. Med. 2022, 28, 666–677. [Google Scholar] [CrossRef]
- Lyon, A.R.; López-Fernández, T.; Couch, L.S.; Asteggiano, R.; Aznar, M.C.; Bergler-Klein, J.; Boriani, G.; Cardinale, D.; Cordoba, R.; Cosyns, B.; et al. 2022 ESC Guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS). Eur. Heart J. Cardiovasc. Imaging 2022, 43, e333–e465, Erratum in Eur. Heart J. Cardiovasc. Imaging 2023, 44, e98.. [Google Scholar]
- Koene, R.J.; Prizment, A.E.; Blaes, A.; Konety, S.H. Shared Risk Factors in Cardiovascular Disease and Cancer. Circulation 2016, 133, 1104–1114. [Google Scholar] [CrossRef]
- Bodai, B.I.; Tuso, P. Breast cancer survivorship: A comprehensive review of long-term medical issues and lifestyle recommendations. Perm. J. 2015, 19, 48–79. [Google Scholar] [CrossRef]
- Lenneman, C.G.; Sawyer, D.B. Cardio-oncology: An update on cardiotoxicity of cancer-related treatment. Circ. Res. 2016, 118, 1008–1020. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Cardona, J.A.; Ray, J.; Carver, J.; Zaha, V.; Cheng, R.; Yang, E.; Mitchell, J.D.; Stockerl-Goldstein, K.; Kondapalli, L.; Dent, S.; et al. Cardio-Oncology Leadership Council. Cardio-Oncology Education and Training: JACC Council Perspectives. J. Am. Coll. Cardiol. 2020, 19, 2267–2281. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Contaldi, C.; D’Aniello, C.; Panico, D.; Zito, A.; Calabrò, P.; Di Lorenzo, E.; Golino, P.; Montesarchio, V. Cancer-Therapy-Related Cardiac Dysfunction: Latest Advances in Prevention and Treatment. Life 2025, 15, 471. [Google Scholar] [CrossRef]
- Octavia, Y.; Tocchetti, C.G.; Gabrielson, K.L.; Janssens, S.; Crijns, H.J.; Moens, A.L. Doxorubicin-induced cardiomyopathy: From molecular mechanisms to therapeutic strategies. J. Mol. Cell. Cardiol. 2012, 52, 1213–1225. [Google Scholar] [CrossRef]
- Jain, D. Cardiotoxicity of doxorubicin and other anthracycline derivatives. J. Nucl. Cardiol. 2000, 7, 53–62. [Google Scholar] [CrossRef]
- Lipshultz, S.E.; Lipsitz, S.R.; Mone, S.M.; Goorin, A.M.; Sallan, S.E.; Sanders, S.P.; Orav, E.J.; Gelber, R.D.; Colan, S.D. Female sex and higher drug dose as risk factors for late cardiotoxic effects of doxorubicin therapy for childhood cancer. N. Engl. J. Med. 1995, 332, 1738–1744. [Google Scholar] [CrossRef] [PubMed]
- Capranico, G.; Tinelli, S.; Austin, C.A.; Fisher, M.L.; Zunino, F. Different patterns of gene expression of topoisomerase II isoforms in differentiated tissues during murine development. Biochim. Biophys. Acta 1992, 1132, 43–48. [Google Scholar] [CrossRef]
- Biancaniello, T.; Meyer, R.A.; Wong, K.Y.; Sager, C.; Kaplan, S. Doxorubicin cardiotoxicity in children. J. Pediatr. 1980, 97, 45–50. [Google Scholar] [CrossRef]
- Hershman, D.L.; McBride, R.B.; Eisenberger, A.; Tsai, W.Y.; Grann, V.R.; Jacobson, J.S. Doxorubicin, cardiac risk factors, and cardiac toxicity in elderly patients with diffuse B-cell non-Hodgkin’s lymphoma. J. Clin. Oncol. 2008, 26, 3159–3165. [Google Scholar] [CrossRef] [PubMed]
- Martin, E.; Thougaard, A.V.; Grauslund, M.; Jensen, P.B.; Bjorkling, F.; Hasinoff, B.B.; Tjørnelund, J.; Sehested, M.; Jensen, L.H. Evaluation of the topoisomerase II-inactive bisdioxopiperazine ICRF-161 as a protectant against doxorubicin-induced cardiomyopathy. Toxicology 2008, 255, 72–79. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, X.; Bawa-Khalfe, T.; Lu, L.-S.; Lyu, Y.L.; Liu, L.F.; Yeh, E.T.H. Identification of the molecular basis of doxorubicin-induced cardiotoxicity. Nat. Med. 2012, 18, 1639–1642. [Google Scholar] [CrossRef]
- Doroshow, J.H.; Locker, G.Y.; Myers, C.E. Enzymatic defenses of the mouse heart against reactive oxygen metabolites: Alterations produced by doxorubicin. J. Clin. Investig. 1980, 65, 128–135. [Google Scholar] [CrossRef]
- Schlame, M.; Rua, D.; Greenberg, M.L. The biosynthesis and functional role of cardiolipin. Prog. Lipid Res. 2000, 39, 257–288. [Google Scholar] [CrossRef] [PubMed]
- Ventura-Clapier, R.; Garnier, A.; Veksler, V. Energy metabolism in heart failure. J. Physiol. 2004, 555, 1–13. [Google Scholar] [CrossRef]
- Pecoraro, M.; Pala, B.; Di Marcantonio, M.C.; Muraro, R.; Marzocco, S.; Pinto, A.; Mincione, G.; Popolo, A. Doxorubicin-induced oxidative and nitrosative stress: Mitochondrial connexin 43 is at the crossroads. Int. J. Mol. Med. 2020, 46, 1197–1209. [Google Scholar] [CrossRef] [PubMed]
- Vásquez-Vivar, J.; Martasek, P.; Hogg, N.; Masters, B.S.; Pritchard, K.A., Jr.; Kalyanaraman, B. Endothelial nitric oxide synthase-dependent superoxide generation from adriamycin. Biochemistry 1997, 36, 11293–11297. [Google Scholar] [CrossRef] [PubMed]
- Kalivendi, S.V.; Kotamraju, S.; Zhao, H.; Joseph, J.; Kalyanaraman, B. Doxorubicin-induced apoptosis is associated with increased transcription of endothelial nitric-oxide synthase: Effect of antiapoptotic antioxidants and calcium. J. Biol. Chem. 2001, 276, 47266–47276. [Google Scholar] [CrossRef] [PubMed]
- Deng, S.; Kruger, A.; Kleschyov, A.; Kalinowski, L.; Daiber, A.; Wojnowski, L. Gp91phox-containing NAD(P)H oxidase increases superoxide formation by doxorubicin and NADPH. Free. Radic. Biol. Med. 2007, 42, 466–473. [Google Scholar] [CrossRef]
- Minotti, G.; Menna, P.; Salvatorelli, E.; Cairo, G.; Gianni, L. Anthracyclines: Molecular advances and pharmacologic developments in antitumor activity and cardiotoxicity. Pharmacol. Rev. 2004, 56, 185–229. [Google Scholar] [CrossRef]
- Boudoulas, K.D.; Triposkiadis, F.; Gumina, R.; Addison, D.; Iliescu, C.; Boudoulas, H. Cardiovascular Disease, Cancer, and Multimorbidity Interactions: Clinical Implications. Cardiology 2022, 147, 196–206. [Google Scholar] [CrossRef]
- Kalivendi, S.V.; Konorev, E.A.; Cunningham, S.; Vanamala, S.K.; Kaji, E.H.; Joseph, J.; Kalyanaraman, B. Doxorubicin activates nuclear factor of activated T-lymphocytes and Fas ligand transcription: Role of mitochondrial reactive oxygen species and calcium. Biochem. J. 2005, 389, 527–539. [Google Scholar] [CrossRef]
- Saeki, K.; Obi, I.; Ogiku, N.; Shigekawa, M.; Imagawa, T.; Matsumoto, T. Doxorubicin directly binds to the cardiac-type ryanodine receptor. Life Sci. 2002, 70, 2377–2389. [Google Scholar] [CrossRef]
- Vedam, K.; Nishijima, Y.; Druhan, L.J.; Khan, M.; Moldovan, N.I.; Zweier, J.L.; Ilangovan, G. Role of heat shock factor-1 activation in the doxorubicin-induced heart failure in mice. Am. J. Physiol. Heart Circ. Physiol. 2010, 298, H1832–H1841. [Google Scholar] [CrossRef]
- Kim, S.-C.; Stice, J.P.; Chen, L.; Jung, J.S.; Gupta, S.; Wang, Y.; Baumgarten, G.; Trial, J.; Knowlton, A.A. Extracellular heat shock protein 60, cardiac myocytes, and apoptosis. Circ. Res. 2009, 105, 1186–1195. [Google Scholar] [CrossRef]
- Bregni, G.; Galli, G.; Gevorgyan, A.; de Braud, F.; Di Cosimo, S. Trastuzumab cardiac toxicity: A problem we put our heart into. Tumori J. 2015, 102, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Mohan, N.; Jiang, J.; Dokmanovic, M.; Wu, W.J. Trastuzumab-mediated cardiotoxicity: Current understanding, challenges, and frontiers. Antib. Ther. 2018, 1, 13–17. [Google Scholar] [CrossRef]
- Ewer, M.S.; Ewer, S.M. Cardiotoxicity of anticancer treatments. Nat. Rev. Cardiol. 2015, 12, 547–558, Erratum in Nat. Rev. Cardiol. 2015, 12, 620. https://doi.org/10.1038/nrcardio.2015.133. [Google Scholar] [CrossRef] [PubMed]
- Riccio, G.; Coppola, C.; Piscopo, G.; Capasso, I.; Maurea, C.; Esposito, E.; De Lorenzo, C.; Maurea, N. Trastuzumab and target-therapy side effects: Is still valid to differentiate anthracycline Type I from Type II cardiomyopathies? Hum. Vaccines Immunother. 2016, 12, 1124–1131. [Google Scholar] [CrossRef]
- ElZarrad, M.K.; Mukhopadhyay, P.; Mohan, N.; Hao, E.; Dokmanovic, M.; Hirsch, D.S.; Shen, Y.; Pacher, P.; Wu, W.J.; Peng, T. Trastuzumab alters the expression of genes essential for cardiac function and induces ultrastructural changes of cardiomyocytes in mice. PLoS ONE 2013, 8, e79543, Erratum in PLoS ONE 2014, 9. https://doi.org/10.1371/annotation/3ba18ef8-8c9c-45ab-9bc5-ad571a54a28c. [Google Scholar] [CrossRef]
- Mohan, N.; Shen, Y.; Endo, Y.; ElZarrad, M.K.; Wu, W.J. Trastuzumab, but Not Pertuzumab, Dysregulates HER2 Signaling to Mediate Inhibition of Autophagy and Increase in Reactive Oxygen Species Production in Human Cardiomyocytes. Mol. Cancer Ther. 2016, 15, 1321–1331. [Google Scholar] [CrossRef]
- Yu, A.F.; Singh, J.C.; Wang, R.; Liu, J.E.; Eaton, A.; Oeffinger, K.C.; Steingart, R.M.; Hudis, C.A.; Dang, C.T. Cardiac Safety of Dual Anti-HER2 Therapy in the Neoadjuvant Setting for Treatment of HER2-Positive Breast Cancer. Oncologist 2017, 22, 642–647. [Google Scholar] [CrossRef]
- Pohl, C.; Dikic, I. Cellular quality control by the ubiquitin-proteasome system and autophagy. Science 2019, 366, 818–822. [Google Scholar] [CrossRef]
- Manasanch, E.E.; Orlowski, R.Z. Proteasome inhibitors in cancer therapy. Nat. Rev. Clin. Oncol. 2017, 14, 417–433. [Google Scholar] [CrossRef]
- Sundaravel, S.H.; I Marar, R.; A Abbasi, M.; Baljevic, M.; Stone, J.R. Bortezomib-Induced Reversible Cardiomyopathy: Recovered With Guideline-Directed Medical Therapy. Cureus 2021, 13, e20295. [Google Scholar] [CrossRef] [PubMed]
- Papanagnou, E.-D.; Terpos, E.; Kastritis, E.; Papassideri, I.S.; Tsitsilonis, O.E.; Dimopoulos, M.A.; Trougakos, I.P. Molecular responses to therapeutic proteasome inhibitors in multiple myeloma patients are donor-, cell type- and drug-dependent. Oncotarget 2018, 9, 17797–17809. [Google Scholar] [CrossRef]
- Efentakis, P.; Kremastiotis, G.; Varela, A.; Nikolaou, P.-E.; Papanagnou, E.-D.; Davos, C.H.; Tsoumani, M.; Agrogiannis, G.; Konstantinidou, A.; Kastritis, E.; et al. Molecular mechanisms of carfilzomib-induced cardiotoxicity in mice and the emerging cardioprotective role of metformin. Blood 2019, 133, 710–723. [Google Scholar] [CrossRef]
- Lettino, M.; Mascherbauer, J.; Nordaby, M.; Ziegler, A.; Collet, J.P.; Derumeaux, G.; Hohnloser, S.H.; Leclercq, C.; E O’NEill, D.; Visseren, F.; et al. Cardiovascular disease in the elderly: Proceedings of the European Society of Cardiology-Cardiovascular Round Table. Eur. J. Prev. Cardiol. 2022, 29, 1412–1424. [Google Scholar] [CrossRef]
- Bringhen, S.; Milan, A.; D’AGostino, M.; Ferri, C.; Wäsch, R.; Gay, F.; Larocca, A.; Offidani, M.; Zweegman, S.; Terpos, E.; et al. Prevention, monitoring and treatment of cardiovascular adverse events in myeloma patients receiving carfilzomib A consensus paper by the European Myeloma Network and the Italian Society of Arterial Hypertension. J. Intern. Med. 2019, 286, 63–74. [Google Scholar] [CrossRef]
- Waxman, A.J.; Clasen, S.; Hwang, W.T.; Garfall, A.; Vogl, D.T.; Carver, J.; O’Quinn, R.; Cohen, A.D.; Stadtmauer, E.A.; Ky, B.; et al. Carfilzomib-Associated Cardiovascular Adverse Events: A Systematic Review and Meta-analysis. JAMA Oncol. 2018, 4, e174519. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Dasgupta, S.; Gong, Y.; Shah, U.A.; Fradley, M.G.; Cheng, R.K.; Roy, B.; Guha, A. Cardiotoxicity as an adverse effect of immunomodulatory drugs and proteasome inhibitors in multiple myeloma: A network meta-analysis of randomized clinical trials. Hematol. Oncol. 2022, 40, 233–242. [Google Scholar] [CrossRef]
- Georgiopoulos, G.; Makris, N.; Laina, A.; Theodorakakou, F.; Briasoulis, A.; Trougakos, I.P.; Dimopoulos, M.-A.; Kastritis, E.; Stamatelopoulos, K. Cardiovascular Toxicity of Proteasome Inhibitors: Underlying Mechanisms and Management Strategies: JACC CardioOncology State-of-the-Art Review. JACC CardioOncol. 2023, 5, 1. [Google Scholar] [CrossRef]
- Wang, H.; Wang, Y.; Li, J.; He, Z.; Boswell, S.A.; Chung, M.; You, F.; Han, S. Three tyrosine kinase inhibitors cause cardiotoxicity by inducing endoplasmic reticulum stress and inflammation in cardiomyocytes. BMC Med. 2023, 21, 147. [Google Scholar] [CrossRef]
- Wang, H.; Sheehan, R.P.; Palmer, A.C.; Everley, R.A.; Boswell, S.A.; Ron-Harel, N.; Ringel, A.E.; Holton, K.M.; Jacobson, C.A.; Erickson, A.R.; et al. Adaptation of Human iPSC-Derived Cardiomyocytes to Tyrosine Kinase Inhibitors Reduces Acute Cardiotoxicity via Metabolic Reprogramming. Cell Syst. 2019, 8, 412–426.e7. [Google Scholar] [CrossRef] [PubMed]
- Richards, C.J.; Je, Y.; Schutz, F.A.; Heng, D.Y.; Dallabrida, S.M.; Moslehi, J.J.; Choueiri, T.K. Incidence and risk of congestive heart failure in patients with renal and nonrenal cell carcinoma treated with sunitinib. J. Clin. Oncol. 2011, 29, 3450–3456. [Google Scholar] [CrossRef] [PubMed]
- Glen, C.; Adam, S.; McDowell, K.; Waterston, A.; Tan, Y.Y.; Petrie, M.C.; Coats, C.J.; Lang, N.N. Cardiotoxicity of BRAF/MEK Inhibitors: A Longitudinal Study Incorporating Contemporary Definitions and Risk Scores. JACC CardioOncol. 2023, 5, 628–637. [Google Scholar] [CrossRef]
- Banks, M.; Crowell, K.; Proctor, A.; Jensen, B.C. Cardiovascular Effects of the MEK Inhibitor, Trametinib: A Case Report, Literature Review, and Consideration of Mechanism. Cardiovasc. Toxicol. 2017, 17, 487–493. [Google Scholar] [CrossRef]
- Agostinetto, E.; Eiger, D.; Lambertini, M.; Ceppi, M.; Bruzzone, M.; Pondé, N.; Plummer, C.; Awada, A.H.; Santoro, A.; Piccart-Gebhart, M.; et al. Cardiotoxicity of immune checkpoint inhibitors: A systematic review and meta-analysis of randomised clinical trials. Eur. J. Cancer 2021, 148, 76–91. [Google Scholar] [CrossRef] [PubMed]
- Haanen, J.B.; Robert, C. Immune Checkpoint Inhibitors. Prog. Tumor Res. 2015, 42, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Infante, N.; Ramírez-Flores, Y.A.; Castillo, E.C.; Lozano, O.; García-Rivas, G.; Torre-Amione, G. A Systematic Review of the Mechanisms Involved in Immune Checkpoint Inhibitors Cardiotoxicity and Challenges to Improve Clinical Safety. Front. Cell Dev. Biol. 2022, 10, 851032. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Gerber, D.E. Autoimmunity, checkpoint inhibitor therapy and immune-related adverse events: A review. Semin. Cancer Biol. 2020, 64, 93–101. [Google Scholar] [CrossRef]
- Grabie, N.; Lichtman, A.H.; Padera, R. T cell checkpoint regulators in the heart. Cardiovasc. Res. 2019, 115, 869–877. [Google Scholar] [CrossRef]
- Hanna, A.; Frangogiannis, N.G. Inflammatory Cytokines and Chemokines as Therapeutic Targets in Heart Failure. Cardiovasc. Drugs Ther. 2020, 34, 849–863. [Google Scholar] [CrossRef]
- Lyon, A.R.; Dent, S.; Stanway, S.; Earl, H.; Brezden-Masley, C.; Cohen-Solal, A.; Tocchetti, C.G.; Moslehi, J.J.; Groarke, J.D.; Bergler-Klein, J.; et al. Baseline cardiovascular risk assessment in cancer patients scheduled to receive cardiotoxic cancer therapies: A position statement and new risk assessment tools from the Cardio-Oncology Study Group of the Heart Failure Association of the European Society of Cardiology in collaboration with the International Cardio-Oncology Society. Eur. J. Heart Fail. 2020, 22, 1945–1960. [Google Scholar] [CrossRef]
- Michel, L.; Mincu, R.I.; Mahabadi, A.A.; Settelmeier, S.; Al-Rashid, F.; Rassaf, T.; Totzeck, M. Troponins and brain natriuretic peptides for the prediction of cardiotoxicity in cancer patients: A meta-analysis. Eur. J. Heart Fail. 2020, 22, 350–361. [Google Scholar] [CrossRef]
- Porter, T.R.; Mulvagh, S.L.; Abdelmoneim, S.S.; Becher, H.; Belcik, J.T.; Bierig, M.; Choy, J.; Gaibazzi, N.; Gillam, L.D.; Janardhanan, R.; et al. Clinical Applications of Ultrasonic Enhancing Agents in Echocardiography: 2018 American Society of Echocardiography Guidelines Update. J. Am. Soc. Echocardiogr. 2018, 31, 241–274. [Google Scholar] [CrossRef]
- Mousavi, N.; Tan, T.C.; Ali, M.; Halpern, E.F.; Wang, L.; Scherrer-Crosbie, M. Echocardiographic parameters of left ventricular size and function as predictors of symptomatic heart failure in patients with a left ventricular ejection fraction of 50-59% treated with anthracyclines. Eur. Heart J. Cardiovasc. Imaging. 2015, 16, 977–984. [Google Scholar] [CrossRef][Green Version]
- Ali, M.T.; Yucel, E.; Bouras, S.; Wang, L.; Fei, H.-W.; Halpern, E.F.; Scherrer-Crosbie, M. Myocardial Strain Is Associated with Adverse Clinical Cardiac Events in Patients Treated with Anthracyclines. J. Am. Soc. Echocardiogr. 2016, 29, 522–527.e3. [Google Scholar] [CrossRef]
- Manganaro, R.; Marchetta, S.; Dulgheru, R.; Ilardi, F.; Sugimoto, T.; Robinet, S.; Cimino, S.; Go, Y.Y.; Bernard, A.; Kacharava, G.; et al. Echocardiographic reference ranges for normal non-invasive myocardial work indices: Results from the EACVI NORRE study. Eur. Heart J. Cardiovasc. Imaging 2019, 20, 582–590. [Google Scholar] [CrossRef]
- Bellenger, N.G.; Burgess, M.I.; Ray, S.G.; Lahiri, A.; Coats, A.J.; Cleland, J.G.; Pennell, D.J. Comparison of left ventricular ejection fraction and volumes in heart failure by echocardiography, radionuclide ventriculography and cardiovascular magnetic resonance; are they interchangeable? Eur. Heart J. 2000, 21, 1387–1396. [Google Scholar] [CrossRef] [PubMed]
- Romano, S.; Judd, R.M.; Kim, R.J.; Kim, H.W.; Klem, I.; Heitner, J.; Shah, D.J.; Jue, J.; White, B.E.; Shenoy, C.; et al. Association of Feature-Tracking Cardiac Magnetic Resonance Imaging Left Ventricular Global Longitudinal Strain With All-Cause Mortality in Patients With Reduced Left Ventricular Ejection Fraction. Circulation 2017, 135, 2313–2315. [Google Scholar] [CrossRef] [PubMed]
- Thavendiranathan, P.; Shalmon, T.; Fan, C.-P.S.; Houbois, C.; Amir, E.; Thevakumaran, Y.; Somerset, E.; Malowany, J.M.; Urzua-Fresno, C.; Yip, P.; et al. Comprehensive Cardiovascular Magnetic Resonance Tissue Characterization and Cardiotoxicity in Women With Breast Cancer. JAMA Cardiol. 2023, 8, 524–534. [Google Scholar] [CrossRef]
- Farhad, H.; Staziaki, P.V.; Addison, D.; Coelho-Filho, O.R.; Shah, R.V.; Mitchell, R.N.; Szilveszter, B.; Abbasi, S.A.; Kwong, R.Y.; Scherrer-Crosbie, M.; et al. Characterization of the Changes in Cardiac Structure and Function in Mice Treated With Anthracyclines Using Serial Cardiac Magnetic Resonance Imaging. Circ. Cardiovasc. Imaging 2016, 9, 12. [Google Scholar] [CrossRef]
- Maffei, E.; Messalli, G.; Martini, C.; Nieman, K.; Catalano, O.; Rossi, A.; Seitun, S.; Guaricci, A.I.; Tedeschi, C.; Mollet, N.R.; et al. Left and right ventricle assessment with Cardiac CT: Validation study vs. Cardiac MR. Eur. Radiol. 2012, 22, 1041–1049. [Google Scholar] [CrossRef]
- Sharma, A.; Einstein, A.J.; Vallakati, A.; Arbab-Zadeh, A.; Mukherjee, D.; Lichstein, E. Meta-analysis of global left ventricular function comparing multidetector computed tomography with cardiac magnetic resonance imaging. Am. J. Cardiol. 2014, 113, 731–738. [Google Scholar] [CrossRef] [PubMed]
- Knuuti, J.; Wijns, W.; Saraste, A.; Capodanno, D.; Barbato, E.; Funck-Brentano, C.; Prescott, E.; Storey, R.F.; Deaton, C.; Cuisset, T.; et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur. Heart J. 2020, 41, 407–477, Erratum in Eur. Heart J. 2020, 41, 4242. https://doi.org/10.1093/eurheartj/ehz825. [Google Scholar] [CrossRef]
- Sueta, D.; Kidoh, M.; Oda, S.; Egashira, K.; Yamamoto, E.; Kaikita, K.; Matsushita, K.; Yamamoto, Y.; Hirai, T.; Tsujita, K. Usefulness of Cardiac Computed Tomography in the Diagnosis of Anti-Cancer Therapy-Related Cardiac Dysfunction-Consistency With Magnetic Resonance Imaging. Circ. J. 2021, 85, 393–396. [Google Scholar] [CrossRef] [PubMed]
- El-Aziz, M.A.A.; Othman, A.I.; Amer, M.; El-Missiry, M.A. Potential protective role of angiotensin-converting enzyme inhibitors captopril and enalapril against adriamycin-induced acute cardiac and hepatic toxicity in rats. J. Appl. Toxicol. 2001, 21, 469–473. [Google Scholar] [CrossRef] [PubMed]
- Vaynblat, M.; Shah, H.R.; Bhaskaran, D.; Ramdev, G.; Davis, W.J., 3rd; Cunningham, J.N., Jr.; Chiavarelli, M. Simultaneous angiotensin converting enzyme inhibition moderates ventricular dysfunction caused by doxorubicin. Eur. J. Heart Fail. 2002, 4, 583–586. [Google Scholar] [CrossRef]
- Cernecka, H.; Ochodnicka-Mackovicova, K.; Kucerova, D.; Kmecova, J.; Nemcekova, V.; Doka, G.; Kyselovic, J.; Krenek, P.; Ochodnicky, P.; Klimas, J. Enalaprilat increases PPARβ/δ expression, without influence on PPARα and PPARγ, and modulate cardiac function in sub-acute model of daunorubicin-induced cardiomyopathy. Eur. J. Pharmacol. 2013, 714, 472–477. [Google Scholar] [CrossRef]
- Boucek, R.J., Jr.; Steele, A.; Miracle, A.; Atkinson, J. Effects of angiotensin-converting enzyme inhibitor on delayed-onset doxorubicin-induced cardiotoxicity. Cardiovasc. Toxicol. 2003, 3, 319–330. [Google Scholar] [CrossRef]
- Blaes, A.H.; Gaillard, P.; Peterson, B.A.; Yee, D.; Virnig, B. Angiotensin converting enzyme inhibitors may be protective against cardiac complications following anthracycline chemotherapy. Breast Cancer Res. Treat. 2010, 122, 585–590. [Google Scholar] [CrossRef]
- Lee, S.; Alsamarrai, A.; Xiao, A.; Wang, T.K.M. Prevention of anthracycline and trastuzumab-induced decline in left ventricular ejection fraction with angiotensin-converting enzyme inhibitors or angiotensin receptor blocker: A narrative systematic review of randomised controlled trials. Intern. Med. J. 2024, 54, 1254–1263. [Google Scholar] [CrossRef]
- Cardinale, D.; Colombo, A.; Sandri, M.T.; Lamantia, G.; Colombo, N.; Civelli, M.; Martinelli, G.; Veglia, F.; Fiorentini, C.; Cipolla, C.M.; et al. Prevention of high-dose chemotherapy-induced cardiotoxicity in high-risk patients by angiotensin-converting enzyme inhibition. Circulation 2006, 114, 2474–2481. [Google Scholar] [CrossRef]
- Oliveira, P.J.; Bjork, J.A.; Santos, M.S.; Leino, R.L.; Froberg, M.; Moreno, A.J.; Wallace, K.B. Carvedilol-mediated antioxidant protection against doxorubicin-induced cardiac mitochondrial toxicity. Toxicol. Appl. Pharmacol. 2004, 2, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Santos, D.L.; Moreno, A.J.; Leino, R.L.; Froberg, M.K.; Wallace, K.B. Carvedilol protects against doxorubicin-induced mitochondrial cardiomyopathy. Toxicol. Appl. Pharmacol. 2002, 185, 218–227. [Google Scholar] [CrossRef]
- Carrasco, R.; Ramirez, M.C.; Nes, K.; Schuster, A.; Aguayo, R.; Morales, M.; Ramos, C.; Hasson, D.; Sotomayor, C.G.; Henriquez, P.; et al. Prevention of doxorubicin-induced Cardiotoxicity by pharmacological non-hypoxic myocardial preconditioning based on Docosahexaenoic Acid (DHA) and carvedilol direct antioxidant effects: Study protocol for a pilot, randomized, double-blind, controlled trial (CarDHA trial). Trials 2020, 21, 137. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Whaley-Connell, A.T.; Habibi, J.; Rehmer, J.; Rehmer, N.; Patel, K.; Hayden, M.; DeMarco, V.; Ferrario, C.M.; Ibdah, J.A.; et al. Mineralocorticoid receptor antagonism attenuates vascular apoptosis and injury via rescuing protein kinase B activation. Hypertension 2009, 53, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Varghese, S.S.; Eekhoudt, C.R.; Jassal, D.S. Mechanisms of anthracycline-mediated cardiotoxicity and preventative strategies in women with breast cancer. Mol. Cell Biochem. 2021, 476, 3099–3109. [Google Scholar] [CrossRef] [PubMed]
- Gabizon, A.; Shmeeda, H.; Barenholz, Y. Pharmacokinetics of pegylated liposomal Doxorubicin: Review of animal and human studies. Clin. Pharmacokinet. 2003, 42, 419–436. [Google Scholar] [CrossRef] [PubMed]
- Acar, Z.; Kale, A.; Turgut, M.; Demircan, S.; Durna, K.; Demir, S.; Meriç, M.; Ağaç, M.T. Efficiency of atorvastatin in the protection of anthracycline-induced cardiomyopathy. J. Am. Coll. Cardiol. 2011, 58, 988–989. [Google Scholar] [CrossRef] [PubMed]
- Riad, A.; Bien, S.; Westermann, D.; Becher, P.M.; Loya, K.; Landmesser, U.; Kroemer, H.K.; Schultheiss, H.P.; Tschöpe, C. Pretreatment with statin attenuates the cardiotoxicity of Doxorubicin in mice. Cancer Res. 2009, 69, 695–699. [Google Scholar] [CrossRef]
- Abdel-Qadir, H.; Bobrowski, D.; Zhou, L.; Austin, P.C.; Calvillo-Argüelles, O.; Amir, E.; Lee, D.S.; Thavendiranathan, P. Statin Exposure and Risk of Heart Failure After Anthracycline- or Trastuzumab-Based Chemotherapy for Early Breast Cancer: A Propensity Score–Matched Cohort Study. J. Am. Heart Assoc. 2021, 10, e018393. [Google Scholar] [CrossRef]
- Shahid, I.; Yamani, N.; Ali, A.; Kumar, P.; Figueredo, V.; Unzek, S.; Mookadam, F. Meta-analysis Evaluating the Use of Statins to attenuate Cardiotoxicity in Cancer Patients receiving Anthracyclines and Trastuzumab-based Chemotherapy. Am. J. Cardiol. 2021, 156, 142–145. [Google Scholar] [CrossRef]
- Sobiborowicz-Sadowska, A.M.; Kamińska, K.; Cudnoch-Jędrzejewska, A. Neprilysin Inhibition in the Prevention of Anthracycline-Induced Cardiotoxicity. Cancers 2023, 15, 312. [Google Scholar] [CrossRef]
- Hu, F.; Yan, S.; Lin, L.; Qiu, X.; Lin, X.; Wang, W. Sacubitril/valsartan attenuated myocardial inflammation, fibrosis, apoptosis and promoted autophagy in doxorubicin-induced cardiotoxicity mice via regulating the AMPKα-mTORC1 signaling pathway. Mol Cell Biochem. 2025, 480, 1891–1908, Erratum in Mol Cell Biochem. 2025, 480, 3217. https://doi.org/10.1007/s11010-024-05159-x. [Google Scholar] [CrossRef] [PubMed]
- Mecinaj, A.; Vinje-Jakobsen, V.; Ngo, D.T.M.; Sverdlov, A.L.; Myhre, P.L. The SARAH trial: More evidence on the role of neurohormonal blockers in prevention of anthracycline-induced cardiotoxicity. Heart Fail. Rev. 2025, 30, 627–631. [Google Scholar] [CrossRef]
- Dabour, M.S.; George, M.Y.; Daniel, M.R.; Blaes, A.H.; Zordoky, B.N. The Cardioprotective and Anticancer Effects of SGLT2 Inhibitors: JACC: CardioOncology State-of-the-Art Review. JACC CardioOncol. 2024, 6, 159–182. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Qadir, H.; Carrasco, R.; Austin, P.C.; Chen, Y.; Zhou, L.; Fang, J.; Su, H.M.H.; Lega, I.C.; Kaul, P.; Neilan, T.G.; et al. The Association of Sodium-Glucose Cotransporter 2 Inhibitors With Cardiovascular Outcomes in Anthracycline-Treated Patients With Cancer. JACC CardioOncol. 2023, 5, 318–328. [Google Scholar] [CrossRef]
- Daniele, A.J.; Gregorietti, V.; Costa, D.; López-Fernández, T. Use of EMPAgliflozin in the prevention of CARDiotoxicity: The EMPACARD-PILOT trial. Cardiooncology 2024, 10, 58. [Google Scholar] [CrossRef]
- Quagliariello, V.; Berretta, M.; Bisceglia, I.; Giacobbe, I.; Iovine, M.; Giordano, V.; Arianna, R.; Barbato, M.; Izzo, F.; Maurea, C.; et al. The sGCa Vericiguat Exhibit Cardioprotective and Anti-Sarcopenic Effects through NLRP-3 Pathways: Potential Benefits for Anthracycline-Treated Cancer Patients. Cancers 2024, 16, 1487. [Google Scholar] [CrossRef]
- Mir, A.; Badi, Y.; Bugazia, S.; Nourelden, A.Z.; Fathallah, A.H.; Ragab, K.M.; Alsillak, M.; Elsayed, S.M.; Hagrass, A.I.; Bawek, S.; et al. Efficacy and safety of cardioprotective drugs in chemotherapy-induced cardiotoxicity: An updated systematic review & network meta-analysis. Cardiooncology 2023, 9, 10. [Google Scholar] [CrossRef] [PubMed]
- Fonoudi, H.; Jouni, M.; Cejas, R.B.; Magdy, T.; Blancard, M.; Ge, N.; Shah, D.A.; Lyra-Leite, D.M.; Neupane, A.; Gharib, M.; et al. Functional Validation of Doxorubicin-Induced Cardiotoxicity-Related Genes. JACC CardioOncol. 2024, 6, 38–50. [Google Scholar] [CrossRef]
- Muckiene, G.; Vaitiekus, D.; Zaliaduonyte, D.; Bartnykaite, A.; Plisiene, J.; Zabiela, V.; Juozaityte, E.; Jurkevicius, R. The Impact of Polymorphisms in ATP-Binding Cassette Transporter Genes on Anthracycline-Induced Early Cardiotoxicity in Patients with Breast Cancer. J. Cardiovasc. Dev. Dis. 2023, 10, 232. [Google Scholar] [CrossRef]
- Visscher, H.; Ross, C.J.; Rassekh, S.R.; Sandor, G.S.; Caron, H.N.; Van Dalen, E.C.; Kremer, L.C.; Van Der Pal, H.J.; Rogers, P.C.; Rieder, M.J.; et al. Validation of variants in SLC28A3 and UGT1A6 as genetic markers predictive of anthracycline-induced cardiotoxicity in children. Pediatr. Blood Cancer. 2013, 60, 1375–1381. [Google Scholar] [CrossRef]
- Zemmour, H.; Planer, D.; Magenheim, J.; Moss, J.; Neiman, D.; Gilon, D.; Korach, A.; Glaser, B.; Shemer, R.; Landesberg, G.; et al. Non-invasive detection of human cardiomyocyte death using methylation patterns of circulating DNA. Nat. Commun. 2018, 9, 1443. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Hu, W.; Song, Z.-P.; Chen, Y.-G.; Zhang, D.-D.; Wang, C.-Q. Resveratrol-induced autophagy promotes survival and attenuates doxorubicin-induced cardiotoxicity. Int. Immunopharmacol. 2016, 32, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Du, J.; Meng, H.; Liu, F.; Yang, N.; Deng, S.; Wan, H.; Ye, D.; Song, E.; Zeng, H. Targeting autophagy with SAR405 alleviates doxorubicin-induced cardiotoxicity. Cell Biol. Toxicol. 2023, 39, 3255–3267. [Google Scholar] [CrossRef] [PubMed]
| Drug/Class | Mechanism of Protection | Key Evidence |
|---|---|---|
| ACE Inhibitors | ↓ ROS, ↑ SOD, mitochondrial support, PPARα modulation, renin–angiotensin system balance. | [4,72,73,74,75,76,77,78] |
| Angiotensin Receptor Blockers | Antioxidant effects. | [4] |
| Beta-Blockers (Carvedilol) | β-arrestin-biased signaling, ↓ apoptosis, mitochondrial respiration preservation. | [79,80,81] |
| Mineralcorticoid Receptoragonists | Aldosterone receptor blockade, antioxidant effects. | [82] |
| Dexrazoxane | Iron chelation, Top2β inhibition, prevention of DNA damage, and ROS generation. | [83] |
| Liposomal Anthracyclines | ↓ cardiac exposure to anthracycline, prolonged circulation, ↓ ROS, and apoptosis. | [84,85] |
| Statins | ↓ oxidative stress, ↑ SOD2, ↓ heart inflammation as evidenced by ↓ TNF-α expression. | [86,87,88,89] |
| Angiotensin Receptor-Neprilysin Inhibitors | AMPKα–mTORC1 modulation, ↓ oxidative stress and inflammation, ↓ autophagy. | [86] |
| Sodium–Glucose Cotransporter Inhibitors | ↓ ER stress, ↑ autophagy/ketogenesis, antioxidant/anti-inflammatory effects, ↑ survival. | [87,88,89,90] |
| Soluble Guanylate Cyclase Stimulators | ↑ cGMP, ↓ inflammation, mitochondrial support, vasodilation. | [91,92] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piscione, M.; Di Marcantonio, M.C.; Pala, B.; Mincione, G. Cancer Therapy-Related Left Ventricular Dysfunction: Are There New Gatekeepers? BioChem 2025, 5, 25. https://doi.org/10.3390/biochem5030025
Piscione M, Di Marcantonio MC, Pala B, Mincione G. Cancer Therapy-Related Left Ventricular Dysfunction: Are There New Gatekeepers? BioChem. 2025; 5(3):25. https://doi.org/10.3390/biochem5030025
Chicago/Turabian StylePiscione, Mariagrazia, Maria Carmela Di Marcantonio, Barbara Pala, and Gabriella Mincione. 2025. "Cancer Therapy-Related Left Ventricular Dysfunction: Are There New Gatekeepers?" BioChem 5, no. 3: 25. https://doi.org/10.3390/biochem5030025
APA StylePiscione, M., Di Marcantonio, M. C., Pala, B., & Mincione, G. (2025). Cancer Therapy-Related Left Ventricular Dysfunction: Are There New Gatekeepers? BioChem, 5(3), 25. https://doi.org/10.3390/biochem5030025









