Abstract
Epitranscriptome refers to post-transcriptional modifications to RNA and their associated regulatory factors that can govern changes in an organism’s cells in response to various environmental stimuli. Recent studies have recognized over 170 distinct chemical signatures in RNA, and the list keeps expanding. These modifications are hypothesized to have roles beyond simply fine-tuning the structure and function of RNA, as studies have linked them to various infectious and noninfectious diseases in humans. Dedicated cellular machinery comprising of RNA-binding proteins (RBPs) that can write, erase, and read these modifications drives the regulation of the epitranscriptomic code, and as such influences RNA metabolism and homeostasis. Equally, perturbations in the function of RBPs may disrupt RNA processing, further implicating them in pathogenesis. As such, the mechanisms underlying RNA modifications and their association with RBPs are emerging areas of interest within the field of biomedicine. This review focuses on understanding epitranscriptomic modifications, their effects on RNA–RBPs interactions, and their influence on cellular processes.
1. Introduction
RNA molecules fulfill critical roles in many cellular processes, from cell division and differentiation to regulating the chromatin state and gene expression. Their perceived role as “inert mediators” between DNA and proteins has changed drastically as we continue to recognize their enormous diversity. Although somewhere around a third of the human genome is transcribed into precursor mRNAs, after the removal of introns, approximately 2% of the genomic sequence ends up integrated into mature mRNAs. Beyond the well-established protein-coding genes, the rest of the genome is transcribed into noncoding (nc) RNAs [1,2]. These transcripts with no apparent coding capacity were once viewed as “junk”, but now they are renowned as an essential and tightly controlled part of the transcriptome.
Diverse chemical groups attached covalently to RNA add another layer of intricacy to coding and noncoding transcripts by increasing the chemical intricacy of RNA’s building blocks [3]. In general, epitranscriptomic modifications can be classified as reversible and irreversible. Among the first group, we can include the methylation of different ribonucleotides, i.e., N6-methyladenosine (m6A), 1-methyladenosine (m1A), 3-methylcytidine (m3C), and 7-methylguanosine (m7G), which impart a positive electrostatic charge to the RNA base. The nonreversible modifications include adenosine-to-inosine (A-to-I) editing, isomerization of uridine to pseudouridine (Ψ), splicing, and polyadenylation. This approximate classification can quickly change as we discover new mechanisms that may drive the reversal of chemical signatures that we previously regarded as enduring. Additionally, the repertoire of RNA modifications continues to expand beyond what we accepted as “transcript specific”. For example, recent studies report the presence of glycan modification of surface RNAs, which was previously known to modify only proteins and lipids [4].
Epitranscriptomic modifications regulate nearly all aspects of RNA metabolism, from transcription and translation to splicing, subcellular localization, and stability. They hold a powerful grip over the transcriptome and provide a swift and cost-effective way to change its composition and function in response to various stimuli. One way this decidable task is accomplished is by influencing RNA interactions with RNA-binding proteins (RBPs). It has been estimated that more than 3000 RBPs exist in human cells [5]. Although the function of the majority of them is unknown, it is now clear that many RBPs play a role in multiple steps of post-transcriptional RNA processing. In general, three main RBPs that are associated with RNA epitranscriptomic modifications have been identified, and these include enzymes that deposit (writers), remove (erasers), and recognize (readers) a given chemical group. However, not all RNA chemical groups have assigned erasers or readers. Thus, targeted proteomic methods that would be used for comprehensive identification and quantification of epitranscriptomic regulators and effectors are needed [6].
The epitranscriptomic RBPs interaction with RNA targets can occur directly, for instance, through RBPs that specifically recognize the RNA modification, or indirectly, since modifications can affect the RNA secondary structure and consequently the binding of proteins that recognize such structures. Both coding and ncRNAs are constantly in contact with various RBPs. While a subset of ‘house-keeping’ RBPs might be constitutively and ubiquitously active, many RBPs have more constrained expression patterns, or their RNA-binding activity may be regulated.
This review discusses our knowledge of epitranscriptomic modifications and their effects on RNA–RBPs interactions. Using exemplary ncRNAs and epitranscriptomic signatures, we highlight the molecular mechanisms that drive the modifications, their role in influencing local RNA structure, the biogenesis of specific ncRNAs, and the recruitment of modification-specific RBPs that fulfill the phenotypic outcome of the signature.
2. N6-Methyladenosine (m6A)
The methyl group at position 6 of adenine (Figure 1), referred to as N6-methyladenosine (m6A), was first described in the mRNA of liver cancer cells [7]. It is one of the most prevalent and conserved chemical signatures in coding and ncRNAs. M6A has been shown to regulate many aspects of RNA biology, including splicing [8], stability [9], export [10], translation [11], and RNA–protein interactions [12]. The primary mechanism by which this chemical signature exerts its function is through interaction with RNA-binding proteins that can be considered m6A-associated RNA-binding proteins (m6A RBPs).
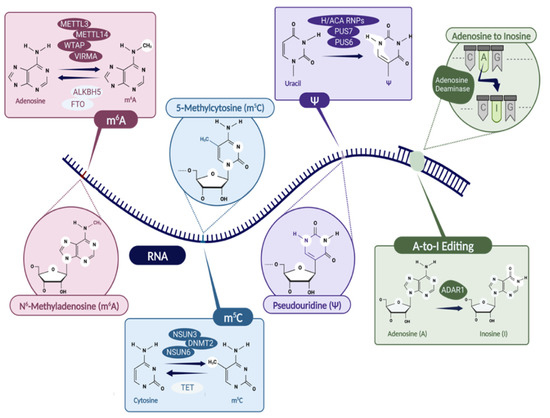
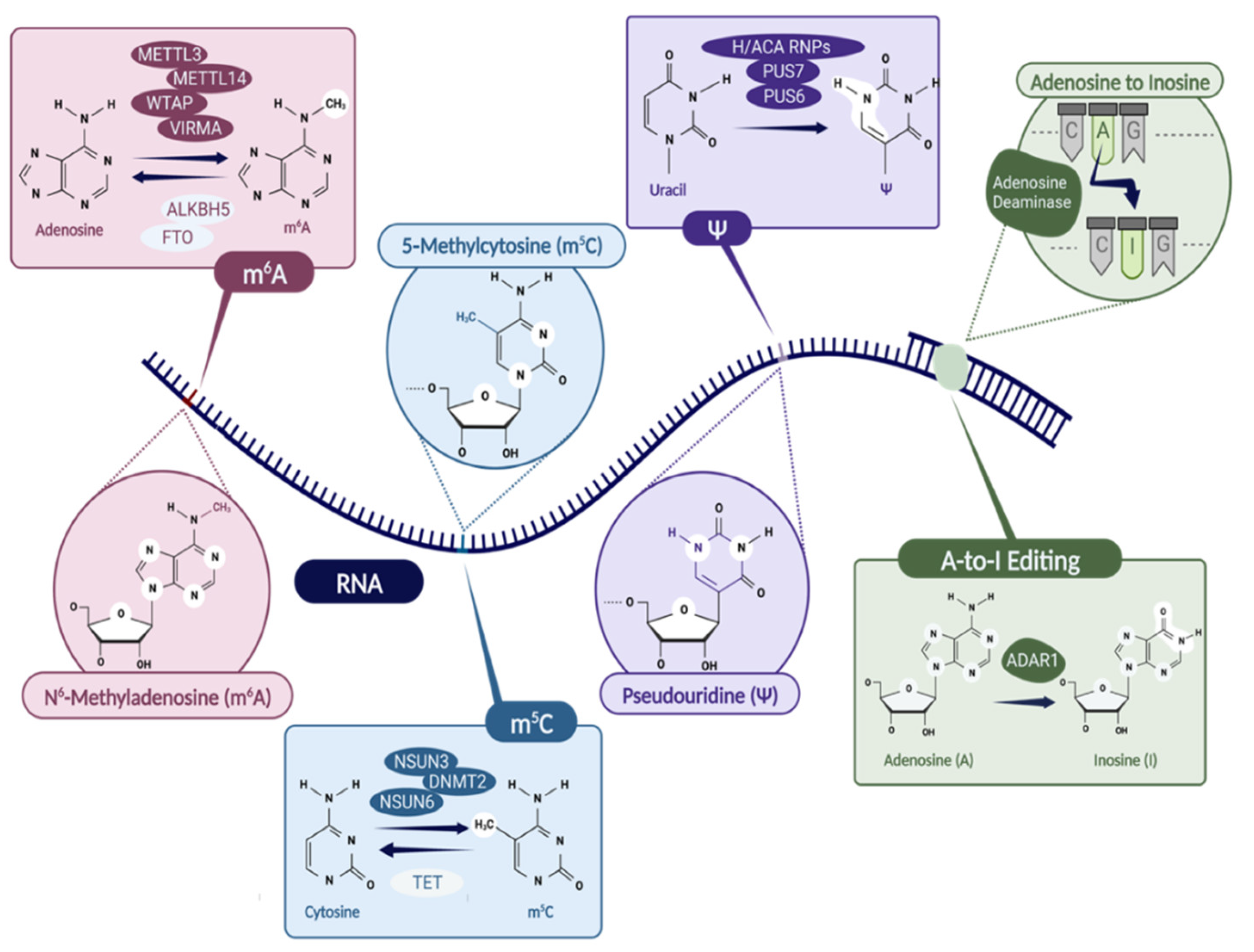
Figure 1.
The list of epitranscriptomic modifications discussed in the manuscript, i.e., N6-methyladenosine (m6A), 5-methyladenosine (m5C), pseudouridine (Ψ), and A-to-I editing. The enzymes involved in writing and erasing the modifications are indicated. Specifically, m6A writers: METTL3/METTL14 complex, WTAP, VIRMA; m6A erasers: ALKBH5, FTO; m5C writers: NSUN3, DNMT2, NSUN6; m5C eraser: TET; Ψ writers: H/ACA RNPs, PUS7, PUS6; ADARs are responsible for A-to-I editing. Created with BioRender.com.
The installation of m6A is mediated by a complex of enzymes, dubbed writers, comprising methyltransferase-like 3 (METTL3), methyltransferase-like 14 (METTL14), and Wilms tumor 1-associated protein (WTAP) forming the catalytic core, and additional components, such as VIR-like m6A methyltransferase-associated (VIRMA) and RNA-binding motif protein 15 (RBM15). Emerging evidence suggests that VIRMA recruits the catalytic core (METTL3/WTAP/METTL14) for the sequence-specific methylation of A to m6A [13]. Recent studies suggest that ZC3H13 is another component of the m6A writer complex [14]. These RBPs catalyze m6A addition onto specific RNA sequences containing the DRm6ACH motif, where D = A, G, or U; R = A or G; H = A, C, or U [15]. Other methyltransferases, including METTL16 and METTL5, have also been shown to deposit m6A by recognizing specific RNA structures and/or consensus motifs [16,17]. Mendel et al. described the crystal structure of METTL16, revealing a methyltransferase domain with an extra N-terminal domain that together form a deep-cut groove essential for RNA binding [18]. Van Tran et al. showed that METTL5 forms a heterodimeric complex with a known methyltransferase activator, tRNA methyltransferase activator subunit 11-2 (TRMT112), to gain metabolic stability. They proposed a model, in which METTL5 in complex with TRMT112, extrudes the target adenosine from a double-stranded RNA before the addition of the methyl group [19].
Two m6A demethylases, dubbed erasers, i.e., fat mass and obesity-associated (FTO) and alpha-ketoglutarate-dependent dioxygenase, known as AlkB homolog 5 (ALKBH5), are responsible for removing m6A from target RNAs, offering a dynamic regulation of m6A status [20]. FTO and ALKBH5 belong to the nonheme Fe(II)-2-oxoglutarate (2OG)-dependent dioxygenase AlkB family, which repairs N-alkylated nucleobases via oxidative demethylation. They have a core catalytic domain containing a double-stranded beta-helix (DSBH) domain for RNA and DNA demethylation [21].
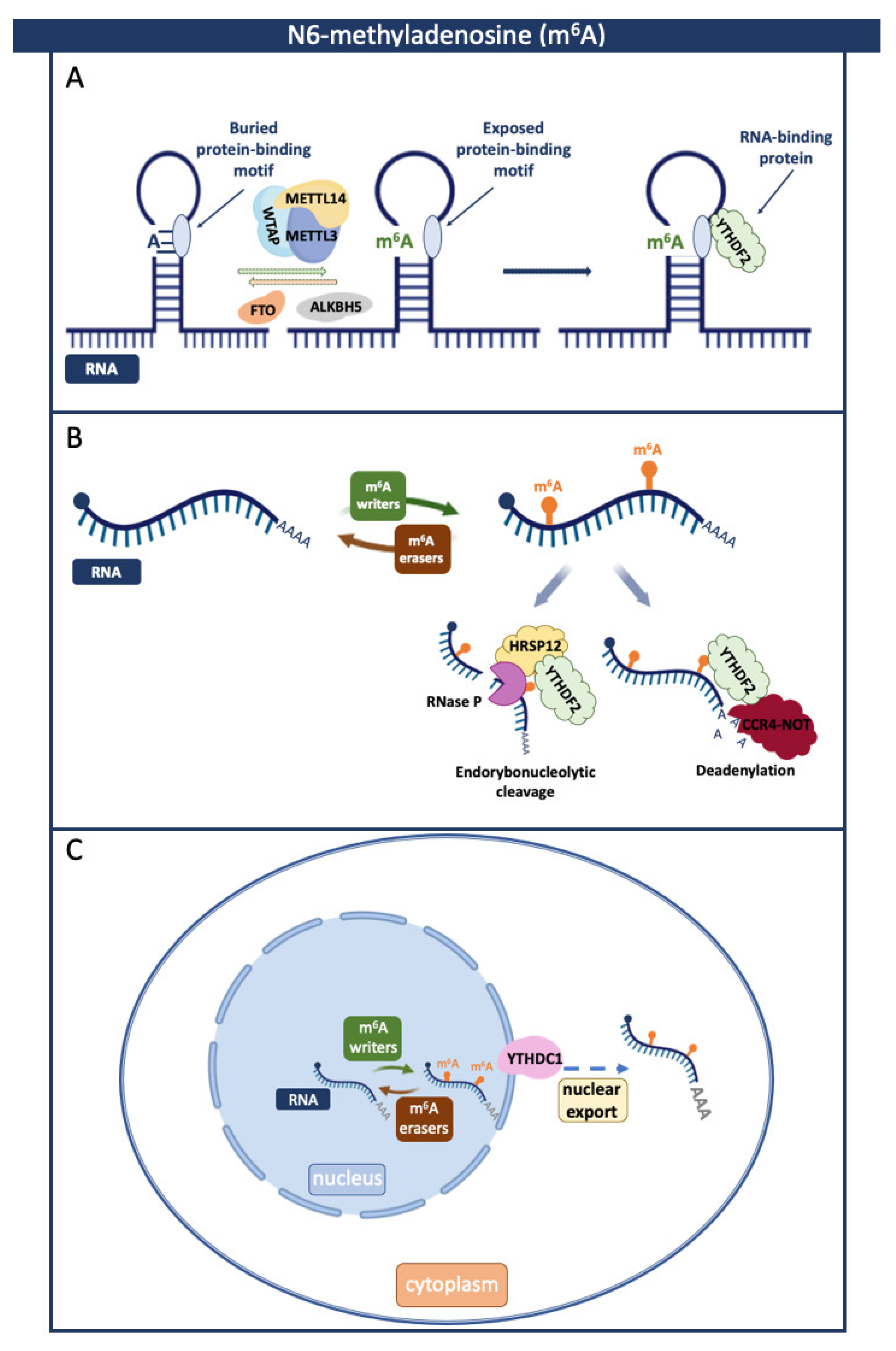
Another significant group of m6A regulatory proteins includes readers, which recognize and convey the m6A phenotypic effects. The YT521-B homology (YTH) domain family comprises five YTH proteins that directly bind m6A through an aromatic cage and interact with the nucleotides that precede and follow the signature [22]. Specifically, YTHDF1 recognizes modified mRNAs, supporting their efficient translation. It can also regulate RNA half-life by promoting mRNA degradation via interaction with Argonaute RISC catalytic component 2 (AGO2) [23]. Additionally, the regulation of RNA stability is the primary role of YTHDF2. By directly interacting with the YTHDF2 N-terminal region and the SH domain of the CNOT1 (CCR4-NOT Transcription Complex Subunit 1), YTHDF2 recruits the carbon catabolite repression 4 (CCR4)–negative on TATA-less (NOT) complex, leading to the deadenylation of modified RNAs and their subsequent degradation (Figure 2) [24]. Moreover, in the presence of heat-responsive protein 12 (HRSP12), YTHDF2 elicits endoribonucleolytic cleavage via the YTHDF2–HRSP12–RNase P/MRP complex (Figure 2) [25]. Through cooperation with m6A readers mentioned above, YTHDF3 affects the translation and decay of methylated RNAs [26]. YTH domain-containing 1 (YTHDC1), on the other hand, regulates the splicing of modified RNAs, while YTH domain-containing 2 (YTHDC2) is essential for escape from exonuclease SOX-induced RNA decay during Kaposi’s sarcoma-associated herpesvirus (KSHV) infection [27].
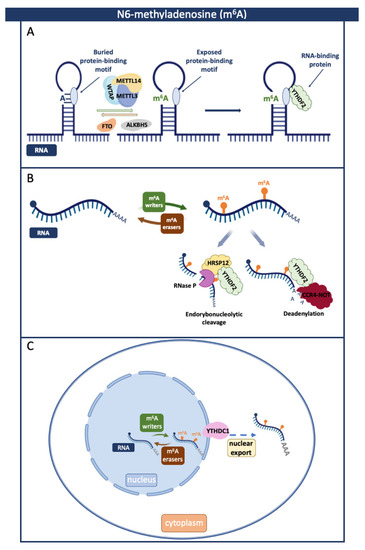
Figure 2.
Roles of N6-methyladenosine (m6A) and associated RBPs in regulating RNA biology. M6A is a dynamic and reversible process coordinated by writers, readers, and erasers that can affect interactions with proteins by modulating RNAs’ structure (A), stability (B), and subcellular location (C). (A) m6A affects the structure of RNA and enables the binding of RBPs. (B) In the presence of heat-responsive protein 12 (HRSP12), YTHDF2 elicits endoribonucleolytic cleavage via the YTHDF2–HRSP12–RNase P/MRP complex. By directly interacting with the YTHDF2 and the CNOT1, YTHDF2 recruits the CCR4–NOT complex, leading to the deadenylation of m6A-modified RNAs and its subsequent degradation. (C) YTHDC1 recognizes m6A-modified RBA and facilitates its transport to the cytoplasm. Created with BioRender.com.
M6A readers also include the heterogeneous nuclear ribonucleoproteins (HNRNPs) family. These enzymes play a significant role in RNA splicing, mRNA stabilization, and transcriptional and translational regulation. It has been shown that a member of this family, i.e., HNRNPC, enhances the metastasis of pancreatic ductal adenocarcinoma (PDAC) cells, and its expression is associated with poor prognosis for patients. Mechanistically, HNRNPC recognizes and binds m6A sites of TATA-box-binding protein-associated factor 8 (TAF8) pre-mRNA and leads to exon skipping, which causes the upregulation of the prometastasis isoform [28]. RNA-binding protein immunoprecipitation revealed that HNRNPC bound to TAF8 by recognizing the RNA-binding motifs around its exon 8.
Among m6A readers, IGF2BPs constitute a highly conserved RNA-binding protein family that affects a variety of processes in the RNA lifecycle, including cellular location, stability, and metabolism. The insulin-like growth factor 2 mRNA-binding proteins (IGF2BPs) regulate m6A-modified mRNA translation and stability [29,30,31]. Huang et al. reported that the knockdown of IGF2BPs globally downregulates gene expression [29]. Further analysis revealed that IGF2BPs regulate gene expression in an m6A-dependent manner by recognizing m6A sites and binding METTL3 and METTL14 [29].
M6A readers also include eukaryotic translation initiation factor 3 (eIF3), proline-rich coiled-coil 2A (PRRC2A), staphylococcal nuclease, and Tudor domain-containing 1 (SND1). The eIF3 reads m6A on 80S ribosomal ®RNA and, as a result, promotes translation elongation [31]. PRRC2A is an essential player in oligodendrocyte specification and myelination by regulating the stability of m6A-modified oligodendrocyte transcription factor 2 (Olig2) mRNA in mice [32]. The SND1 stabilizes the ORF50 transcript encoded by KSHV, promoting viral replication [33].
These m6A effector proteins facilitate both temporal and spatial RNA regulation, where writers work in the nucleus to introduce the modification, which various readers recognize in the nucleus and cytoplasm, influencing the biological fate of their target RNAs. We discuss the following examples of m6A-specific readers to illustrate their broad impact on RNA metabolism, structure, function, and regulation.
A growing body of evidence indicates that m6A can alter the structure of ncRNAs (Figure 2) and affect their interactions with effector proteins [34,35]. HNRNPG is comprised of an N-terminal globular RNA recognition motif of 90 amino acids, followed by 300 amino acids of a low-complexity sequence, with serine, arginine, glycine, and proline accounting for two-thirds of the amino acid residues. The low-complexity sequence’s C-terminal 58 amino acids have been shown to bind an RNA hairpin with an A/G-rich motif. Liu et al. identified heterogeneous nuclear ribonucleoprotein G (HNRNPG) as a protein that binds m6A-modified metastasis-associated lung adenocarcinoma transcript 1 (MALAT1) through its C-terminal low-complexity region [36]. In the same study, they showed that m6A influences the secondary structure of the MALAT1 hairpin, resulting in increased accessibility of HNRNPG to its low-complexity purine-rich binding motif. Increased HNRNPG pulldown from the nuclear extract was seen when the m6A site in the MALAT1 hairpin was changed to G, C, or U. Interestingly, the authors found over thirteen thousand high-confidence HNRNPG-bound m6A sites across the human transcriptome and demonstrated that HNRNPG and m6A work together to regulate gene expression. Moreover, Jones et al. described the influence of m6A on the Xist A-repeat AUCG tetraloop (Jones et al., 2022). Using isothermal calorimetry (ITC), the authors demonstrated that the (m6A)UC nucleotides are recognized by the YTH domain of YTHDC1 when the motif is present in a single-stranded conformation.
M6A-associated RBPs can also influence RNAs’ half-life (Figure 2). An excellent example of this phenomenon is DIAPH1 Antisense RNA 1 (DIAPH-AS1), whose dysregulation contributes to nasopharyngeal carcinoma growth and metastasis. A recent study revealed that m6A modification of DIAPH-AS1 is mediated by WTAP and that the protein also influences RNA stability [37]. A knockdown of WTAP drastically reduced DIAPH-AS1 levels, while the constructs containing the mutated m6A residues did not affect DIAPH1-AS1 expression. The authors analyzed the half-life of DIAPH1-AS1 in WTAP-silenced cells using an actinomycin D chase experiment and showed that silencing WTAP increased DIAPH1-AS1 turnover by about 25–50%. Furthermore, using an RNA pulldown assay with mass spectrometry and RNA immunoprecipitation (RIP), Li et al. identified that insulin-like growth factor 2 mRNA binding protein 2 (IGF2BP2) recognizes DIAPH1-AS1 m6A and supports its stabilization via an m6A-dependent manner. The authors showed that overexpression of IGF2BP2 can revert reduced DIAPH1-AS1 expression in cells with WTAP knockdown. They concluded that the m6A reader IGF2BP2 interprets WTAP-mediated m6A present on DIAPH1-AS1 and maintains its stability.
Another example of m6A RBPs regulating ncRNA stability is represented by promoter-associated ncRNA-D (pncRNA-D) lncRNA, a transcript expressed from the promoter region of the cyclin D1 (CCND1) gene. Yoneda et al. found that pncRNA-D expression is induced by osmotic stress and can inhibit CCND1 expression by interacting with RBPs translocated in liposarcoma/fused in sarcoma (TLS/FUS). Further analysis revealed that pncRNA-D is highly m6A-modified, and YTHDC1 is responsible for binding m6A sites on pncRNA-D. The m6A methylation shortens pncRNA-D lifetime and affects interaction with TLS [38].
M6A also influences RNAs’ subcellular location (Figure 2). RP11 lncRNA, which is highly expressed in colorectal cancer (CRC) cells, has been shown to regulate gene expression by interaction with the m6A reader, hnRNPA2B1 [34]. Although overexpression of METTL3 had no significant effect on RP11 stability, it led to RP11 upregulation, indicating m6A as a positive regulator of this lncRNA in CRC cells. Subcellular fractionation analysis showed that METTL3 overexpression markedly increases the association of RP11 with chromatin.
Accumulating evidence has demonstrated that m6A is widespread in circular RNAs (circRNAs). Zhou et al. developed a computational pipeline that they combined with rRNA depletion and m6A immunoprecipitation and found thousands of common and cell-type-specific m6A-modified circRNAs [39]. The authors also uncovered an unexpected connection between m6A circRNAs and mRNA turnover, where the half-life of modified mRNA encoded by the parent genes of m6A-circRNAs was significantly reduced compared to genes not coding for circRNAs.
Studies have indicated that m6A can modulate circRNAs subcellular localization and function by affecting their interactions with RBPs. The m6A-modified, oncogenic circNSUN2 is exported to the cytoplasm by YTHDC1 in CRC cells. In the cytoplasm, circNSUN2 increases the stability of high-mobility-group AT-hook 2 (HMGA2) mRNA through the association with m6A reader protein, IGF2BP2, which leads to enhanced CRC cell aggressiveness [40]. A similar phenomenon was observed in yet another type of malignancy, i.e., hepatocellular carcinoma (HCC), in which the cytoplasmic location of m6A-modified circHPS5 is mediated by YTHDC1. Furthermore, the circHPS5 increases HMGA2 expression and accelerates HCC cell tumorigenesis [41].
The aforementioned m6A reader, IGF2BP2, has also been shown to modulate the action of another circRNA, circARGHAP12, which plays a critical role in cervical cancer (CC). By interacting with IGF2BP2, circARGHAP12 regulates the stability of Forkhead Box M1 (FOXM1) mRNA and promotes CC progression [42].
M6A modification also decorates microRNAs (miRNAs), a class of small ncRNA molecules that regulate gene expression by pairing with target mRNA [43]. M6A modification has been shown to affect miRNA biogenesis [44]. Cleavage by a microprocessor complex composed of the RNA-binding protein DGCR8 Microprocessor Complex Subunit (DGCR8) and the ribonuclease type III DROSHA represents an essential regulatory step in miRNA biogenesis. Alarcón et al. found that METTL3 methylates primary (pri) miRNAs, marking them for recognition and processing by DGCR8 [45]. Similarly, METTL3 depletion decreased DGCR8’s ability to bind to pri-miRNAs, prompting a global decrease of mature miRNAs and a simultaneous buildup of unprocessed pri-miRNAs. Moreover, Bhat et al. reported that the m6A mark can change pri-miRNAs’ secondary structure and affect the recruitment of the microprocessor complex [46]. A growing body of research shows that the interaction between m6A modification and miRNAs influences cancer progression by affecting the biogenesis or stability of miRNAs [44].
3. 5-Methylcytosine (m5C)
With a recognized regulatory role in RNA folding and gene expression, 5-methylcytosine (m5C) is characterized by the presence of a methyl group at the C5 carbon of cytosine (C) (Figure 1) [47]. Though it constitutes a localized structural change, adding a methyl group to the nucleobase influences its chemical properties and, thus, its associations with RBPs [48].
The installation and removal of m5C sites are facilitated by specific methyltransferases and demethylases, respectively [47]. The m5C writers commonly include a variety of proteins that fall under the NOP/Sun (NSUN) RNA methyltransferase classification, including NSUN3 and NSUN6 [48]. The capacity of these enzymes to catalyze the methylation of cytosine is partially mediated by the presence of several cysteines in their active site [48]. The thiolate side chain of cysteine temporarily binds to the C6 carbon of the cytosine, activating the conjugated heterocyclic ring and encouraging the formation of a covalent bond between the C5 cytosine carbon and the methyl group of an S-adenosylmethionine (SAM) cofactor [48]. DNA methyltransferases 2 (DNMT2) is another m5C writer that shares similarities with NSUN proteins concerning its interactions with RNA and the mechanism of cytosine methylation [48]. In addition to writers, erasers catalyze the removal of methyl groups from modified cytidine [47]. The most prevalent cytosine demethylases are the ten-eleven translocation (TET) proteins; several specific forms of this enzyme, including TET2, are known to participate in the oxidation of m5C present in an assortment of RNAs [49]. Through their role in m5C demethylation, these proteins have the potential to influence RNA structure, stability, and thus RNA–RBPs interactions [47].
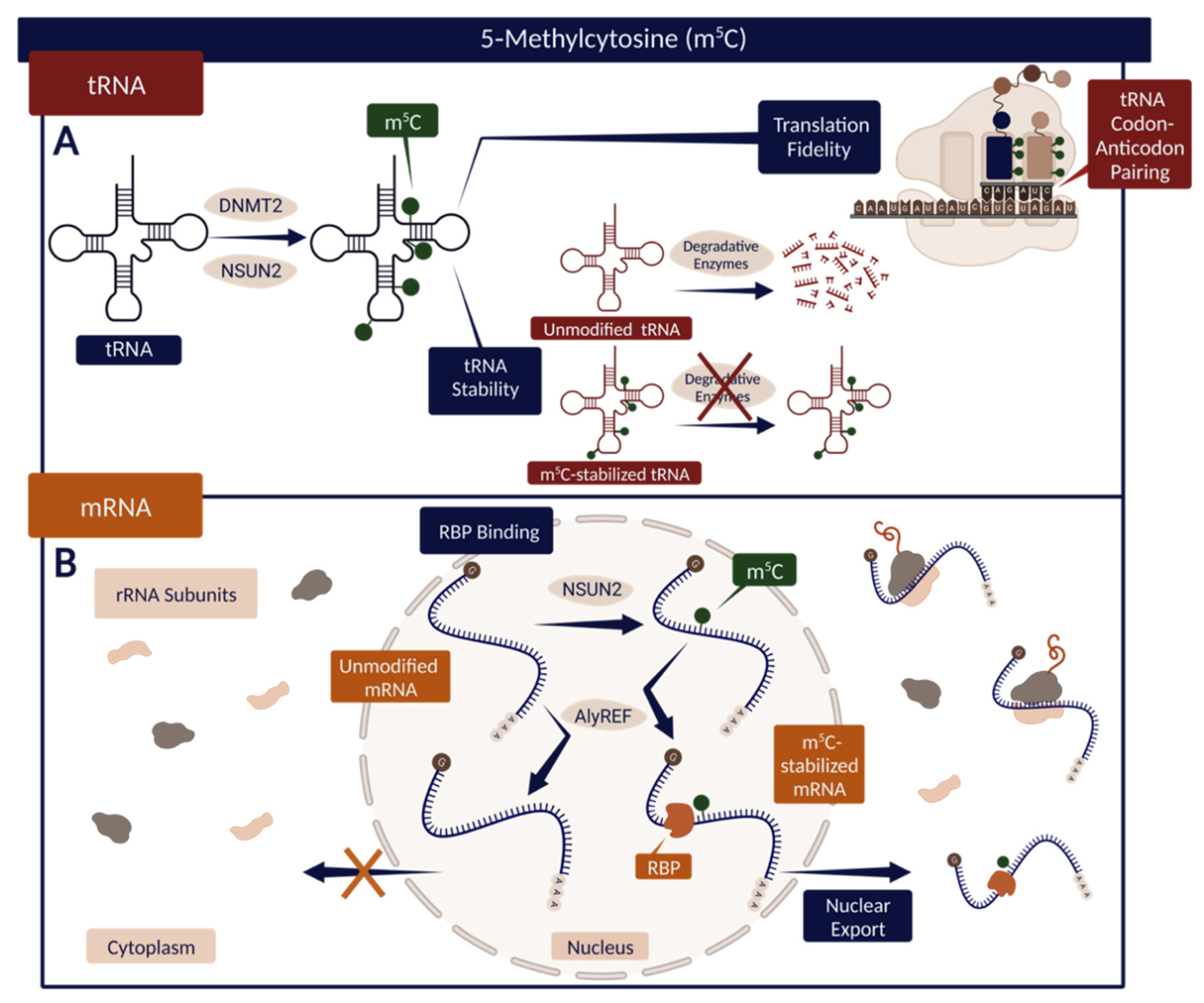
Though not as abundant as m6A, m5C has been identified in various RNAs, including rRNA, tRNA, mRNA, and lncRNA [47]. With consequential roles in maintaining the function and stability of tRNA specifically, m5C modifications have been located at various positions along its structure (Figure 3) [50]. One study of tRNALeu indicates that the m5C at position 34 (m5C34) within the anticodon sequence may influence translation through modulation of codon–anticodon pairing [50]. Furthermore, additional sites within different tRNA sequences are believed to act in association with other epitranscriptomic modifications to protect tRNA against degradation, in some cases interfering with endonucleolytic cleavage and contributing to overall tRNA stability (Figure 3) [50].
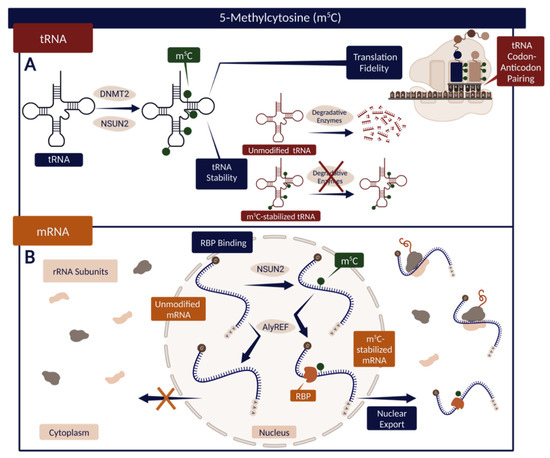
Figure 3.
(A) The effect of m5C modification on tRNA stability and translation accuracy. Specific m5C modulates tRNA codon–anticodon pairing and thus translation fidelity. m5C also promotes tRNA stability, in some cases preventing endonucleolytic cleavage and subsequent tRNA degradation. (B) The function of m5C in mRNA interaction with RBPs. m5C-stabilized mRNA can bind AlyREF—an RBP that supports mRNA nuclear export. Created with BioRender.com.
A separate study illustrates the effects of m5C on RNA association with RBPs, indicating that the silencing of NSUN2, and thus the reduction of m5C, correlates with a decreased binding affinity for Aly/REF Export Factor (ALYREF), a protein that facilitates the transportation of mRNA from the nucleus to the cytoplasm [51]. The binding capacity of ALYREF is predictably dependent upon the identity of its amino acid constituents; specifically, the authors of a recent study established that a single lysine residue at position 171 (K171) of ALYREF contributes substantially to its observed interactions with m5C-modified RNA transcripts [51]. Upon mutation of the K171 residue, the binding affinity of ALYREF for modified RNA decreased appreciably, resulting in a reduced ratio of protein bound to free oligonucleotides compared with the control [51]. This example depicts how the regulation of RNA–RBPs binding associated with m5C modifications can impact protein synthesis and, thus, gene expression.
Another study suggests that m5C-modified RNAs are found at higher concentrations in cancerous tumor cells of the lung when compared with noncancerous cells [47].
Here, m5C is believed to modulate the binding of a tumor-associated antigen known as Y-Box-Binding Protein 1 (YBX1) [47]. This RBP functions as a reader of m5C-modified mRNA within the cytoplasm through its cold-shock domain (CSD) [47]. More precisely, the indole ring of the tryptophan (W) residue at position 65 (W65) facilitates recognition and interaction with RNA modification [47]. Due to its capacity to stabilize the transcripts with which it binds and recruit certain other stability-enhancing RBPs, YBX1 is believed to play a notable role in cancer through its association with specific m5C-modified oncogenes, e.g., HDGF [47]. In effect, the capacity of m5C to regulate the binding of this RBP draws attention to its diverse role and potential implications in the context of human health.
4. Pseudouridine (Ψ)
Much like the epitranscriptomic modifications described above, pseudouridine (Ψ) plays an instrumental role in mediating RNA–RBP interactions. RNA pseudouridylation refers to the irreversible isomerization of the canonical base uridine to Ψ (Figure 1) [52]. Two distinct mechanisms, RNA-dependent and RNA-independent, guide pseudouridylation [53]. The RNA-dependent pseudouridylation necessitates the presence of a highly specialized RNA–RBP complex composed of box H/ACA small nucleolar (sno) RNA in conjunction with several accessory and catalytic proteins, including glycine–arginine-rich protein 1 (GAR1), dyskerin (DKC1), nucleolar protein 10 (NOP10), and nonhistone chromosome protein 2 (NHP2) [52,53]. Pseudouridylation by box H/ACA RNPs occurs in the nucleus and requires the collective function of all four RBPs; though the catalytic domain of DKC1 is ultimately responsible for the isomerization of uridine, the remaining core proteins are similarly essential [52]. NHP2 and NOP10 bind to the upper portion of the “hairpin-hinge–hairpin-tail structure” of the box H/ACA snoRNA, stabilizing interactions between the catalytic RNP and the RNA substrate to allow for isomerization of the target uridine [52]. Finally, GAR1 is the only constituent RBP that does not specifically interact with box H/ACA snoRNA; rather, it associates with DKC1, performing a vital function in “substrate turnover” [52]. In contrast, RNA-independent pseudouridylation involves a stand-alone pseudouridine synthase (PUS) catalyst [52,53]. Various enzymes under this classification are known to exist, i.e., PUS1-10, each with a similar fundamental shape that enables it to interact with a particular RNA sequence and/or structure [52]. This “conserved catalytic domain” gives rise to analogous mechanisms of catalysis observed among different classes of PUS enzymes [52]. A particular assembly of secondary structures enables PUS to interact with and stabilize a diverse array of RNA substrates while simultaneously employing its aspartate (D) amino acid, among other residues that comprise the active site [52]. Pseudouridylation by pseudouridine synthases involves the rotation of the uracil (U) nucleotide base and the transformation of an N-glycosidic bond to a stronger C-glycosidic bond [54]. Though seemingly small, this configurational modification of the nucleoside and the installation of Ψ maintains a powerful grip over RNA–RBP interactions [52]. In effect, pseudouridylation influences RBP binding through alterations to the chemical properties of modified RNA regions that impact the protein-binding affinity and site accessibility due to changes in RNA secondary structure [55]. In particular, the existence of a novel secondary amine at the N1 position of a Ψ nucleoside allows for the formation of an additional hydrogen bond, a contributing factor to the elevated stability of pseudouridylated RNA when compared with its unmodified counterpart [54]. Moreover, Ψ-containing RNA sequences are known to have increased base stacking interactions, increasing the rigidity of the RNA structure [55].
Research indicates that Ψ is integral in modulating RBP activity in coding and ncRNAs [54]. One study details the effects of Ψ on the ability of the specific serine arginine (SR)-rich RBP, RBSR1, to bind to the 3’ untranslated region (UTR) of Valyl-tRNA synthetase (VaIRS) [54]. A comparison was drawn between RBSR1 binding to VaIRS 3’ UTR with and without Ψ to determine how pseudouridylation affects RBP interactions with the RNA target. Binding affinity was measured quantitatively in terms of the relative dissociation constant (Kd) for pseudouridylated and unmodified RNA sequences. It was concluded that the presence of Ψ inhibits RBSR1 binding, decreasing binding affinity (higher Kd) and reducing RBP interactions at these sites [54].
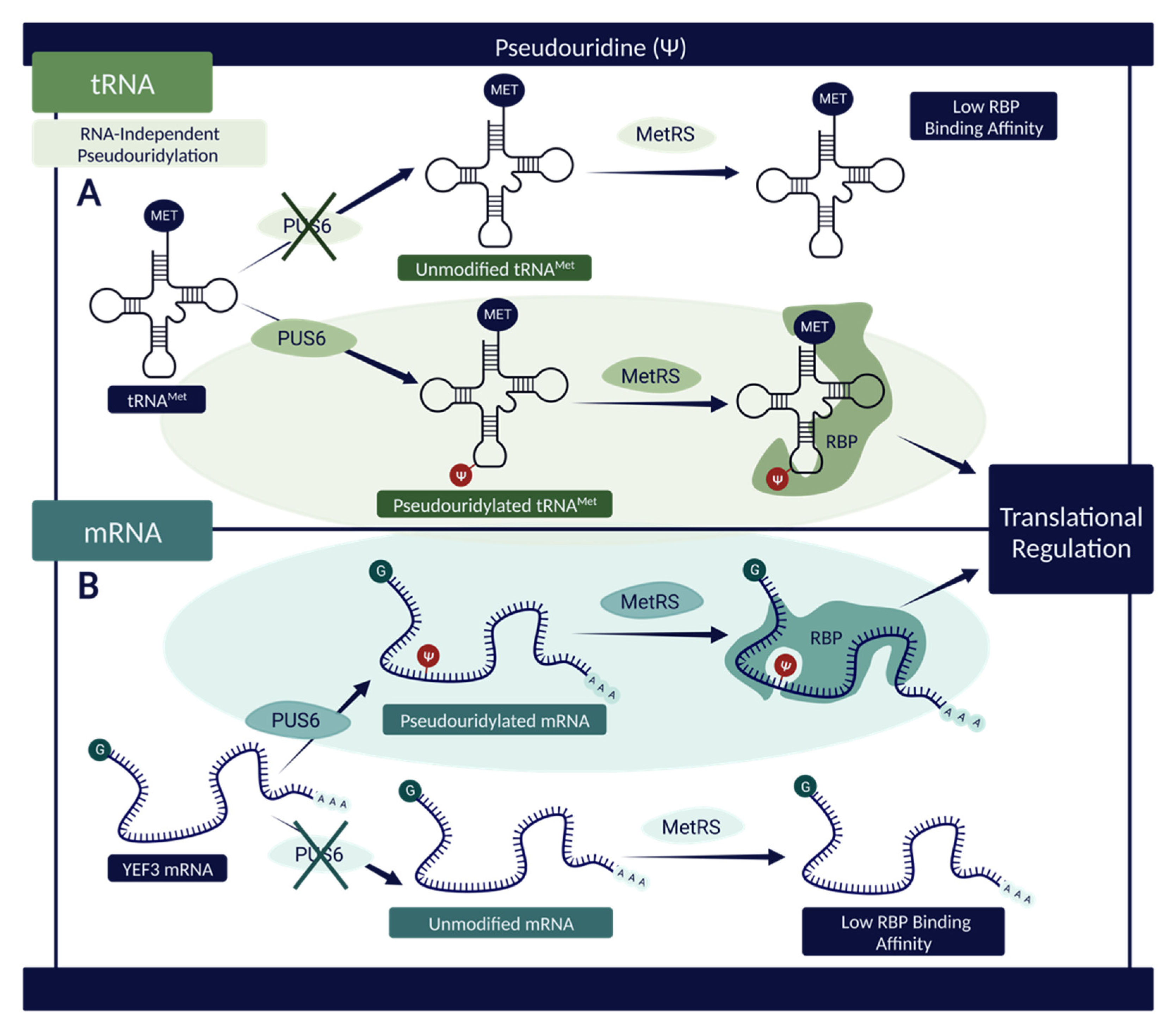
A separate study highlights a correlation between the presence of PUS6—the catalyzing agent of tRNAMet and YEF3 mRNA pseudouridylation—and the ability of these RNAs to bind to a reader protein known as methionine aminoacyl tRNAMet synthetase (MetRS) that influences translational regulation [53]. The data suggests that a knockout of PUS6 and a subsequent reduction in RNA pseudouridylation is directly associated with decreased binding of MetRS proteins at known sites within tRNAMet and YEF3 mRNA (Figure 4). This conclusion indicates that Ψ modification present within these RNA sequences influences their chemical properties and structure in such a way as to increase binding affinity, facilitate RBP interactions, and allow for effective regulation of translation via MetRS binding [53].
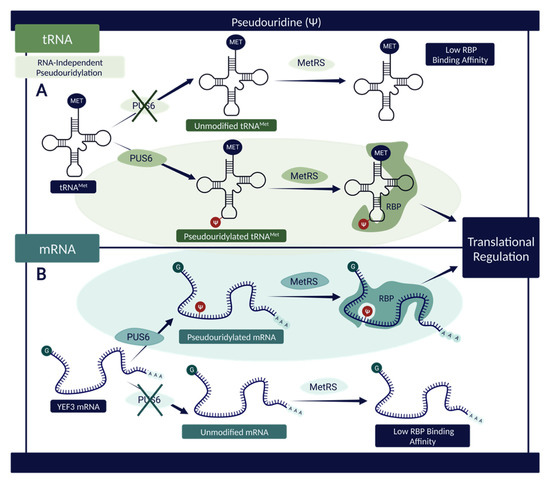
Figure 4.
The influence of Ψ on RNAs and their propensity to interact with RBPs. (A) Pseudouridylated tRNAMet demonstrates a higher binding affinity with MetRS—an RBP associated with translational regulation—when compared with its unmodified counterpart, suggesting a connection between PUS6 and the RNA–protein interactions [53]. (B) A similar effect can be observed with respect to pseudouridylated mRNA. In the presence of PUS6, the structure of YEF3 mRNA is modified, permitting elevated interactivity with MetRS [53]. In both instances (tRNAMet and YEF3 mRNA), Ψ sites increase RBP binding and subsequently aid in translational regulation due to the nature of MetRS [53]. Created with BioRender.com.
Research pertaining to a particular class of small ncRNAs known as Transfer-RNA-Derived Fragments (tRFs) further suggests that Ψ modulates RBP binding and, consequently, fundamental cellular processes such as protein synthesis [56]. Of recent interest due to their speculated association with specific diseases and cancers (i.e., myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML)), tRFs have been found to interact with certain RBPs in a “Ψ-dependent manner” [56]. A recent study explores the function of PUS7-catalyzed pseudouridylation of mTOG sequences—a particular class of tRFs that incorporates 5’ oligo-guanines. It was determined that pseudouridylation of mTOG elevates the binding affinity (comparatively small Kd) for certain domains of polyadenylate-binding protein cytoplasmic 1 (PABPC1), a protein thought to influence translation–initiation complex (eIF4F) stability [56].
Although the degree to which pseudouridylation influences RNA protein interactions is mainly dependent on the nature of specific RNAs and their associated RBPs, such modifications have been shown to consistently influence RNA stability, structure, an affinity for RBPs, and, thus, the regulation of an array of coding and ncRNAs. However, much remains to be discovered with respect to this epitranscriptomic modification and its impact on the binding of regulatory RBPs [55].
5. N1-Methyladenosine (m1A)
N1-methyladenosine (m1A) refers to a reversible epitranscriptomic modification that involves the addition of a methyl group to the N1 nitrogen of adenosine (A) [57]. This alteration produces a chemical change, i.e., the nitrogen of the heterocyclic aromatic ring loses electron density, resulting in a positively charged nucleoside at neutral pH [58]. Accordingly, m1A influences the electrostatic interactions of RNA transcripts, altering their three-dimensional structure and impacting their capacity to associate with RBPs through increased or decreased availability of binding sites [58]. Moreover, the positive charge generated by this modification has the potential to either disrupt or facilitate RBP binding depending on the favorability of electrostatic interactions between modified RNA and specific residues at the protein interface [58].
Although present in multiple RNAs, including rRNA and mRNA, m1A modifications are often discussed in the context of tRNA [59]. Such focus is predominantly due to the invariant presence of m1A at position 58 of tRNA [59]. This site is known to support tRNA folding and stability, with its absence leaving transcripts susceptible to degradation [59]. Additionally, regulation of this highly conserved modification by associated writer and eraser enzymes represents a novel “mechanism of translation control” [59].
In tRNA, m1A sites are installed by tRNA methyltransferases (TRMTs), including TRMT61A and TRMT6 for human cytoplasmic tRNAs and TRMT61B for human mitochondrial (mt-) tRNAs [60]. The capacity of several of these TRMTs (i.e., TRMT6 and TRMT61A) to methylate certain positions along specific mRNA transcripts has also been observed [57]. In addition to writers, m1A is modulated by two known demethylases: ALKBH1 and ALKBH3 [57]. These erasers are members of the AlkB homolog (ALKBH) protein family, and they facilitate the removal of the methyl group from the N1 nitrogen at m1A sites, altering the chemical properties, binding capacity, and functionality of the substrate RNAs [59]. Despite this knowledge, much remains to be discovered regarding m1A installation and removal, the enzymes involved in these dynamic processes, how these proteins differ based on transcript identity, and their precise catalytic mechanisms.
To elucidate the supposed role of m1A on translation, one study sought to determine the effect of reduced ALKBH1 expression and, specifically, its influence over the presence of an initiator tRNA (tRNAiMet) [59]. The authors found that a knockdown of this m1A eraser in HeLa cells was accompanied by an “up to 3-fold” increase in tRNAiMet compared with cells exhibiting normal ALKBH1 expression [59]. Because m1A demethylation in tRNA is associated with increased vulnerability to degradation, the reduced rate of m1A removal by ALKH1 likely allowed these transcripts to maintain a more stable structure, thus increasing tRNAiMet and enhancing translation initiation [59]. Ultimately, these findings provide compelling evidence to suggest that m1A may be a key regulator of translation and, consequently, gene expression through control over tRNA stability [59].
As with the other epitranscriptomic modifications discussed in this review, m1A allows interactions between modified RNA transcripts and reader proteins [57]. Akin to m6A, m1A sites promote binding with certain RBPs from the YTH-domain-protein family; however, m6A- and m1A-containing RNA differ in their relative affinities for these specific enzymes [57]. A recent study confirms that m1A sites are recognized by two YTHDF proteins: YTHDF1 and YTHDF2 [57]. To determine the degree to which m1A enhances YTHDF protein binding, an experiment that involved both modified and unmethylated RNA transcripts was performed [57]. The data established that unmodified RNAs express a markedly reduced binding affinity for YTHDF1 and YTHDF2 compared with m1A-modified RNA, as evidenced by higher Kd values [57]. Overall, this increased interaction of m1A-containing mRNAs with YTHDF2 was found to diminish transcript stability and increase degradation [57]. The results of this investigation illustrate the potential regulatory capacity of m1A modifications and yet another facet of their multifold role in gene expression through the modulation of mRNA turnover [57].
6. 5-Hydroxymethylcytosine (hm5C)
Another known RNA modification that is formed by the oxidation of m5C by the ten-eleven translocation (TET) family of enzymes is 5-hydoxymethylcytosine (hm5C) [61]. Originally, hm5C was found in wheat seedlings, though it was subsequently also identified in mammalian tissues [62,63,64]. Delatte et al. revealed that hm5C preferentially marks polyadenylated RNA [65]. In the same study, they showed that hydroxymethylation can favor mRNA translation. A growing body of evidence indicates that hm5C is an important regulator of gene expression in brain tissues [65,66]. However, the biological functionalities of this modification and how hm5C affects RNA and protein interactions are still largely unknown due to the lack of information on its distribution. Understanding these aspects can provide significant insights related to the field of epitranscriptomics.
In order to achieve this goal, several techniques have been developed. In addition to sequencing techniques that allow broad identification of hm5C, machine learning-based models and algorithms were applied [64,67]. For instance, Liu et al. developed iRNA5hmC, the first computational method for identifying RNA hm5C residues that allow making predictions based on RNA primary sequence without any other experimental knowledge [64].
7. A-to-I RNA Editing
RNA editing refers to a broad range of RNA-level alterations introduced during or after transcription that may alter the edited RNA molecule’s ability to code. This mechanism is an essential player in generating transcriptome and proteome diversity. However, when deregulated, it can contribute to various states of disease. One of the best-characterized RNA editing mechanisms is A-to-I RNA editing, which converts adenosines to inosines in double-stranded RNA (dsRNA) (Figure 1). This phenomenon is catalyzed by members of the adenosine deaminase acting on RNA (ADAR) family, initially identified in Xenopus laevis cells [68]. ADAR proteins are classified into three types in mammals: adenosine deaminase RNA-specific (ADAR1), adenosine deaminase RNA-specific B1 (ADARB1), and adenosine deaminase RNA-specific B2 (ADAR3). Each ADAR protein contains a conserved C-terminal deaminase between one and three dsRNA-binding domains (dsRBDs) that recognize and bind dsRNA [69]. ADAR enzymes prefer adenosines within a specific local sequence context (e.g., 5′-UAG-3′). They can be highly selective for specific adenosines within more complex secondary structures. RNA editing has been shown to affect many fundamental biological processes. In addition to mRNA, it also occurs in ncRNA, including lncRNAs, miRNAs, and circRNAs, leading to changes in structure that may affect transcripts’ stability and function.
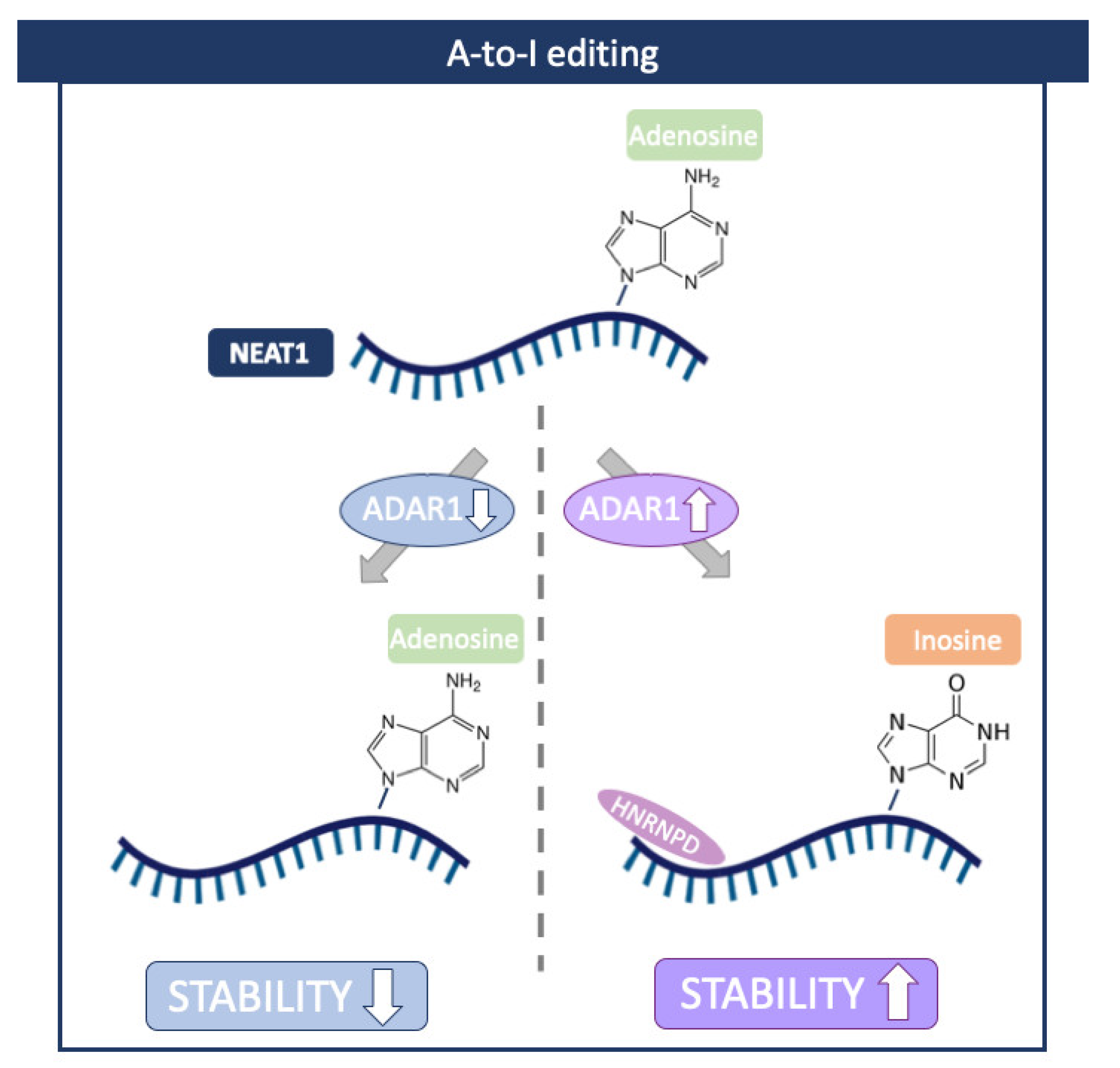
Nuclear-Enriched Abundant Transcript 1 (NEAT1) is a well-studied lncRNA that plays an essential role in cellular stress responses [70]. This lncRNA is frequently overexpressed in cancers, and its higher expression has been shown to correlate with decreased rates of survival in patients [71,72]. In addition, a recent study reported an increased level of NEAT1 in atherosclerotic cardiovascular disease [73]. Interestingly, in this case, NEAT stability was controlled by ADAR1 in an RNA-editing-dependent manner. Mechanistically, ADAR1 facilitated the binding of Heterogeneous Nuclear Ribonucleoprotein D (HNRNPD), which is required for NEAT1 stability (Figure 5) [73].
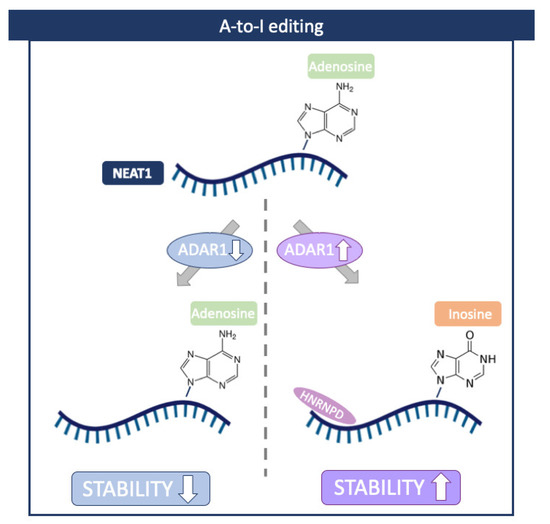
Figure 5.
A-to-I editing in NEAT1 lncRNA. ADAR1 facilitates the binding of Heterogeneous Nuclear Ribonucleoprotein D (HNRNPD), which is required for NEAT1 stability. Created with BioRender.com.
Another example of lncRNA affected by the ADAR-mediated RNA editing mechanism is prostate cancer antigen 3 (PCA3), the most specific prostate cancer biomarker [74]. PCA3 is an intronic antisense lncRNA embedded within intron 6 of tumor suppressor, Prune Homolog 2 with BCH Domain (PRUNE2) gene. Salameh et al. reported that oncogenic PCA3 binds PRUNE2 pre-mRNA and controls its expression level. Moreover, the authors showed that ADAR proteins associate with PRUNE2/PCA3 dsRNA and regulate PRUNE2 and PCA3 levels via A-to-I RNA editing [75].
As mentioned above, A-to-I editing also occurs in miRNAs and may significantly affect their biogenesis and function. Yang et al. reported that editing of the precursor (pre-) miRNA-142 inhibited its processing by ribonuclease III (DROSHA) [76]. It has also been shown that editing by ADAR enzymes regulates miRNA stability. Paul et al. analyzed human miRNAs across different tissues and observed higher stability in A-to-I edited miRNAs compared to their unedited forms [77]. They showed that all the miRNAs that gained stability after editing had mispaired cytosines on the pre-miRNA hairpin structure.
Interestingly, it has been recently revealed that the ADAR enzymes can affect circRNA biogenesis. Shen et al. created ADAR1/2 mutants depleted of either editing activity only or both RNA binding and editing capabilities, demonstrating that ADARs could regulate circRNAs processing through their editing-dependent and/or independent functions [78]. In the same study, the authors revealed that A-to-I editing could influence circRNA production by stabilizing or destabilizing dsRNA formed between the reverse complementary matches (RCMs).
8. Antireaders of RNA Epitranscriptomic Modifications
Most epitranscriptomic studies focus on RBPs that recognize and bind directly to the modified residue or the motif that becomes available after modification. These reader proteins are critical for facilitating the phenotypic effect of a modification. One can, however, envision an alternative mechanism in which RNA modifications would affect biological function by abolishing interactions between RBP and its target transcript. This phenomenon could be encountered when an epitranscriptomic modification invokes an incompatible structure that is either not recognizable or not accessible for RBP association. This effect would be expected to occur less frequently since RNA epitranscriptomic signatures constitute only a small fraction of the transcriptome compared to the canonical ribonucleotides.
Edupuganti et al. used a mass-spectrometry-based proteomics screen of m6A interactors in various mammalian cells to identify two stress granule proteins, namely G3BP1 and G3BP2, whose binding is repelled by m6A [79]. This phenomenon seemed to be sequence context-dependent, as m6A in a GGACU context strongly repelled G3BP1 binding, but not when the modification was present in a GAACU context. The authors also discovered that the G3BP1 association with RNAs can stabilize transcripts. They proposed that this can occur either by G3BP1 outcompeting the binding of the reader that predisposes RNAs to degradation in an m6A-dependent manner or by G3BP1-directed sequestration of transcripts into stress granules that could prevent access to the RNA degradation machinery. In any case, these findings reveal the intricate interplay between RNA-binding proteins and the regulation of RNA homeostasis. Another example of m6A preventing protein binding comes from C. elegans, where an m6A mark on the 3’ splice site of the SAM synthetase pre-mRNA inhibits its proper splicing and protein production [80]. This process is triggered by a rich diet and acts as an m6A-mediated switch to stop SAM production and regulate its homeostasis. Although the mammalian SAM synthetase pre-mRNA is not regulated via this mechanism, the splicing inhibition by 3′ splice site m6A is conserved in mammals.
Moreover, NSUN2-mediated m5C modification of cytosine 69 in vault RNA 1.1 was shown to prevent the association with serine/arginine-rich splicing factor 2 (SRSF2), resulting in different splicing outcomes [81]. It was concluded that both NSUN2 and SRSF2 orchestrate the maturation of the vault RNAs and produce distinct small-vault RNAs, which is crucial for efficient cellular differentiation. Interestingly, pseudouridine synthase 7 (PUS7) bound methylated vault RNA 1.1. with higher affinity, which hints at the dynamic interplay between different epitranscriptomic modifications, their associated cellular machinery, and specific outcomes for RNA biology. Further, m5C modification in XIST repeat 8 prevents the binding of polycomb repressive complex 2 [82].
Much like base methylation, RNA pseudouridylation can disrupt the RBP binding. For example, pseudouridine has been shown to inhibit Muscleblind-like 1 (MBNL1) binding to the toxic RNA CCUG repeats and minimally structured RNAs [83]. Melting studies and molecular dynamic simulations of unmodified and pseudouridylated minimally structured transcripts suggested that the reduced RNA flexibility and increased base-stacking interactions are responsible for the diminished binding by MBNL1. Additionally, both m6A and pseudouridine have been shown to weaken the binding of the human single-stranded RNA-binding protein Pumilio 2 to its consensus motif. These studies indicated that changing single uridines to pseudouridine or adenines to N6-methyladenines weakens RBP binding by two- to three-fold while replacing additional residues resulted in a further decrease in affinity, in agreement with simple energetic additivity [55]. Considering that many RNA modifications appear to be widely prevalent in the binding sites of many different RBPs, we expect that more examples of associated readers and antireaders will emerge.
9. Conclusions and Future Directions
RNA structure and function are intimately tied to RNA-binding protein recognition and regulation. Epitranscriptomic modifications further expand the intricacy of this relationship, as in different cell types, stages of development, and pathogenic states, specific combinations of RNA chemical signatures, and associated RBPs establish and modulate complex gene expression networks. Despite extensive efforts invested in understanding the molecular mechanisms underlying the role of epitranscriptomic modifications in the context of RNA–protein interactions, many questions remain unanswered.
The characterization of RNA–RBP interactions has been focused almost entirely on identifying RNA–protein interfaces rather than answering the fundamental question of how the RNAs and RBPs are regulated to control these interactions. One can envisage that RNAs may be enriched or depleted of specific RBPs in certain biological states, affecting the transcript’s biological fate. What regulates these states is currently unknown. Moreover, if ternary complexes with more than one RNA bound by a given epitranscriptomic reader are formed, relative levels between the target transcripts, the number of binding sites, the respective protein–RNA binding affinities, and the stability of the interactions could be decisive in the phenotypic outcome. Certainly, some epitranscriptomic modifications must exist in proximity with each other to coregulate the fate of the transcript and its interactions with RBPs. For example, specific readers such as YTHDF proteins are known to bind both m6A and m1A, further convoluting the epitranscriptomic crosstalk [57]. What factors guide these interactions and whether they work in concert or are antagonistic in cellular biological processes remains to be determined. Moreover, much work should be done to fully comprehend how RBPs select their RNA targets, how molecular networks can control RNA–RBP complex formation, how RBPs are regulated, and how such regulation alters RNA expression.
Although several studies exposed the prevalence of epitranscriptomic alternations in human diseases related to either up- or downregulation of their associated RBPs or RNA signatures, their mechanistic link to disease pathology remains obscure. Whether these alternations serve as disease drivers or simply disease manifestations has to be defined. This would require outlining the landscape of RNA modifications specific to healthy and diseased states across tissues, and developmental stages, which certainly would be a tremendous undertaking.
In addition to the above-discussed modifications, the functions of several less abundant modifications, e.g., N4-acetylcytosine (ac4C), N6,2′-O-dimethyladenosine (m6Am), 2′-O-methylation (Nm), and N7-methylguanosine (m7G) and their associated RBPs are currently being unfolded. A major challenge in studying these modifications is the lack of specific methodologies that would allow us to identify and quantify their frequency in modified targets. Future studies should implement holistic approaches to understand the interplay resulting in synergy or competition between different modifications and their associated effectors.
Author Contributions
Conceptualization, J.S.-S. and E.W.; writing—original draft preparation, E.W., A.M. and J.S.-S.; writing—review and editing, E.W., A.M. and J.S.-S.; visualization, E.W. and A.M.; supervision, J.S.-S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pertea, M. The Human Transcriptome: An Unfinished Story. Genes 2012, 3, 344–360. [Google Scholar] [CrossRef] [PubMed]
- Palazzo, A.F.; Lee, E.S. Non-coding RNA: What is functional and what is junk? Front. Genet. 2015, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- Wiener, D.; Schwartz, S. The epitranscriptome beyond m6A. Nat. Rev. Genet. 2020, 22, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, W.; Pandey, V.; Pokharel, Y.R. Membrane linked RNA glycosylation as new trend to envision epi-transcriptome epoch. Cancer Gene Ther. 2022, 2022, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Han, M.; Meng, L.; Chen, X. Transcriptome-wide discovery of coding and noncoding RNA-binding proteins. Proc. Natl. Acad. Sci. USA 2018, 115, E3879–E3887. [Google Scholar] [CrossRef]
- Qi, T.F.; Miao, W.; Wang, Y. Targeted Profiling of Epitranscriptomic Reader, Writer, and Eraser Proteins Accompanied with Radioresistance in Breast Cancer Cells. Anal. Chem. 2022, 94, 1525–1530. [Google Scholar] [CrossRef]
- Desrosiers, R.; Friderici, K.; Rottman, F. Identification of Methylated Nucleosides in Messenger RNA from Novikoff Hepatoma Cells. Proc. Natl. Acad. Sci. USA 1974, 71, 3971–3975. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.I.; Shi, H.; Lyu, R.; Wylder, A.C.; Matuszek, Ż.; Pan, J.N.; He, C.; Parisien, M.; Pan, T. Regulation of Co-transcriptional Pre-mRNA Splicing by m6A through the Low-Complexity Protein hnRNPG. Mol. Cell 2019, 76, 70–81.e9. [Google Scholar] [CrossRef] [PubMed]
- Chelmicki, T.; Roger, E.; Teissandier, A.; Dura, M.; Bonneville, L.; Rucli, S.; Dossin, F.; Fouassier, C.; Lameiras, S.; Bourc’His, D. m6A RNA methylation regulates the fate of endogenous retroviruses. Nature 2021, 591, 312–316. [Google Scholar] [CrossRef]
- Lesbirel, S.; Viphakone, N.; Parker, M.; Parker, J.; Heath, C.; Sudbery, I.; Wilson, S.A. The m6A-methylase complex recruits TREX and regulates mRNA export. Sci. Rep. 2018, 8, 13827. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.; Dong, L.; Liu, X.-M.; Guo, J.; Ma, H.; Shen, B.; Qian, S.-B. m6A in mRNA coding regions promotes translation via the RNA helicase-containing YTHDC2. Nat. Commun. 2019, 10, 5332. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Dai, Q.; Zheng, G.; He, C.; Parisien, M.; Pan, T. N6-methyladenosine-dependent RNA structural switches regulate RNA–protein interactions. Nature 2015, 518, 560–564. [Google Scholar] [CrossRef] [PubMed]
- Yue, Y.; Liu, J.; Cui, X.; Cao, J.; Luo, G.; Zhang, Z.; Cheng, T.; Gao, M.; Shu, X.; Ma, H.; et al. VIRMA mediates preferential m6A mRNA methylation in 3′UTR and near stop codon and associates with alternative polyadenylation. Cell Discov. 2018, 4, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Lv, R.; Ma, H.; Shen, H.; He, C.; Wang, J.; Jiao, F.; Liu, H.; Yang, P.; Tan, L.; et al. Zc3h13 Regulates Nuclear RNA m6A Methylation and Mouse Embryonic Stem Cell Self-Renewal. Mol. Cell 2018, 69, 1028–1038.e6. [Google Scholar] [CrossRef] [PubMed]
- Bayoumi, M.; Munir, M. Evolutionary conservation of the DRACH signatures of potential N6-methyladenosine (m6A) sites among influenza A viruses. Sci. Rep. 2021, 11, 4548. [Google Scholar] [CrossRef] [PubMed]
- Su, R.; Dong, L.; Li, Y.; Gao, M.; He, P.C.; Liu, W.; Wei, J.; Zhao, Z.; Gao, L.; Han, L.; et al. METTL16 exerts an m6A-independent function to facilitate translation and tumorigenesis. Nature 2022, 24, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Ignatova, V.V.; Stolz, P.; Kaiser, S.; Gustafsson, T.H.; Lastres, P.R.; Sanz-Moreno, A.; Cho, Y.-L.; Amarie, O.V.; Aguilar-Pimentel, A.; Klein-Rodewald, T.; et al. The rRNA m6A methyltransferase METTL5 is involved in pluripotency and developmental programs. Genes Dev. 2020, 34, 715–729. [Google Scholar] [CrossRef] [PubMed]
- Mendel, M.; Chen, K.-M.; Homolka, D.; Gos, P.; Pandey, R.R.; McCarthy, A.A.; Pillai, R.S. Methylation of Structured RNA by the m6A Writer METTL16 Is Essential for Mouse Embryonic Development. Mol. Cell 2018, 71, 986–1000.e11. [Google Scholar] [CrossRef] [PubMed]
- Van Tran, N.; Ernst, F.G.M.; Hawley, B.R.; Zorbas, C.; Ulryck, N.; Hackert, P.; Bohnsack, K.E.; Bohnsack, M.T.; Jaffrey, S.R.; Graille, M.; et al. The human 18S rRNA m6A methyltransferase METTL5 is stabilized by TRMT112. Nucleic Acids Res. 2019, 47, 7719–7733. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Liu, B.; Nie, Z.; Duan, L.; Xiong, Q.; Jin, Z.; Yang, C.; Chen, Y. The role of m6A modification in the biological functions and diseases. Signal Transduct. Target. Ther. 2021, 6, 1285–1297. [Google Scholar] [CrossRef]
- Shen, D.; Wang, B.; Gao, Y.; Zhao, L.; Bi, Y.; Zhang, J.; Wang, N.; Kang, H.; Pang, J.; Liu, Y.; et al. Detailed resume of RNA m6A demethylases. Acta Pharm. Sin. B 2022, 12, 2193–2205. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Yin, P. Structural Insights into N 6 -methyladenosine (m 6 A) Modification in the Transcriptome. Genom. Proteom. Bioinform. 2018, 16, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Chen, K.; Dong, X.; Xu, Y.; Sun, Q.; Wang, H.; Chen, Z.; Liu, C.; Liu, R.; Yang, Z.; et al. YTHDF1 promotes mRNA degradation via YTHDF1-AGO2 interaction and phase separation. Cell Prolif. 2021, 55, e13157. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Zhao, Y.; He, J.; Zhang, Y.; Xi, H.; Liu, M.; Ma, J.H.J.; Wu, H.D.Y.Z.H.X.M.L.L. YTHDF2 destabilizes m6A-containing RNA through direct recruitment of the CCR4–NOT deadenylase complex. Nat. Commun. 2016, 7, 12626. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Choe, J.; Park, O.H.; Kim, Y.K. Molecular Mechanisms Driving mRNA Degradation by m6A Modification. Trends Genet. 2020, 36, 177–188. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Chen, Y.-S.; Ping, X.-L.; Yang, X.; Xiao, W.; Yang, Y.; Sun, H.-Y.; Zhu, Q.; Baidya, P.; Wang, X.; et al. Cytoplasmic m6A reader YTHDF3 promotes mRNA translation. Cell Res. 2017, 27, 444–447. [Google Scholar] [CrossRef] [PubMed]
- Macveigh-Fierro, D.; Cicerchia, A.; Cadorette, A.; Sharma, V.; Muller, M. The m6A reader YTHDC2 is essential for escape from KSHV SOX-induced RNA decay. Proc. Natl. Acad. Sci. USA 2022, 119, e2116662119. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.-T.; Li, J.-H.; Zhu, X.-X.; Huang, C.-S.; Gao, Z.-X.; Xu, Q.-C.; Zhao, W.; Yin, X.-Y. HNRNPC impedes m6A-dependent anti-metastatic alternative splicing events in pancreatic ductal adenocarcinoma. Cancer Lett. 2021, 518, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Weng, H.; Sun, W.; Qin, X.; Shi, H.; Wu, H.; Zhao, B.S.; Mesquita, A.; Liu, C.; Yuan, C.L.; et al. Recognition of RNA N6-methyladenosine by IGF2BP proteins enhances mRNA stability and translation. Nat. Cell Biol. 2018, 20, 285–295. [Google Scholar] [CrossRef]
- Wang, J.; Chen, L.; Qiang, P. The role of IGF2BP2, an m6A reader gene, in human metabolic diseases and cancers. Cancer Cell Int. 2021, 21, 99. [Google Scholar] [CrossRef]
- Lin, Y.; Li, F.; Huang, L.; Polte, C.; Duan, H.; Fang, J.; Sun, L.; Xing, X.; Tian, G.; Cheng, Y.; et al. eIF3 Associates with 80S Ribosomes to Promote Translation Elongation, Mitochondrial Homeostasis, and Muscle Health. Mol. Cell 2020, 79, 575–587.e7. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Li, A.; Sun, B.; Sun, J.-G.; Zhang, J.; Zhang, T.; Chen, Y.; Xiao, Y.; Gao, Y.; Zhang, Q.; et al. A novel m6A reader Prrc2a controls oligodendroglial specification and myelination. Cell Res. 2018, 29, 23–41. [Google Scholar] [CrossRef]
- Baquero-Perez, B.; Antanaviciute, A.; Yonchev, I.D.; Carr, I.M.; Wilson, S.A.; Whitehouse, A. The Tudor SND1 protein is an m6A RNA reader essential for replication of Kaposi’s sarcoma-associated herpesvirus. eLife 2019, 8, 47261. [Google Scholar] [CrossRef]
- Wu, Y.; Yang, X.; Chen, Z.; Tian, L.; Jiang, G.; Chen, F.; Li, J.; An, P.; Lu, L.; Luo, N.; et al. m6A-induced lncRNA RP11 triggers the dissemination of colorectal cancer cells via upregulation of Zeb1. Mol. Cancer 2019, 18, 87. [Google Scholar] [CrossRef] [PubMed]
- Jin, D.; Guo, J.; Wu, Y.; Du, J.; Yang, L.; Wang, X.; Di, W.; Hu, B.; An, J.; Kong, L.; et al. m6A mRNA methylation initiated by METTL3 directly promotes YAP translation and increases YAP activity by regulating the MALAT1-miR-1914-3p-YAP axis to induce NSCLC drug resistance and metastasis. J. Hematol. Oncol. 2019, 12, 135. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Zhou, K.I.; Parisien, M.; Dai, Q.; Diatchenko, L.; Pan, T. N 6-methyladenosine alters RNA structure to regulate binding of a low-complexity protein. Nucleic Acids Res. 2017, 45, 6051–6063. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.-X.; Zheng, Z.-Q.; Yang, P.-Y.; Lin, L.; Zhou, G.-Q.; Lv, J.-W.; Zhang, L.-L.; Chen, F.; Li, Y.-Q.; Wu, C.-F.; et al. WTAP-mediated m6A modification of lncRNA DIAPH1-AS1 enhances its stability to facilitate nasopharyngeal carcinoma growth and metastasis. Cell Death Differ. 2022, 29, 1137–1151. [Google Scholar] [CrossRef]
- Yoneda, R.; Ueda, N.; Uranishi, K.; Hirasaki, M.; Kurokawa, R. Long noncoding RNA pncRNA-D reduces cyclin D1 gene expression and arrests cell cycle through RNA m6A modification. J. Biol. Chem. 2020, 295, 5626–5639. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Molinie, B.; Daneshvar, K.; Pondick, J.V.; Wang, J.; van Wittenberghe, N.; Xing, Y.; Giallourakis, C.C.; Mullen, A.C. Genome-Wide Maps of m6A circRNAs Identify Widespread and Cell-Type-Specific Methylation Patterns that Are Distinct from mRNAs. Cell Rep. 2017, 20, 2262–2276. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.-X.; Chen, X.; Xia, L.-P.; Zhang, J.-X.; Pan, Z.-Z.; Ma, X.-D.; Han, K.; Chen, J.-W.; Judde, J.-G.; Deas, O.; et al. N6-methyladenosine modification of circNSUN2 facilitates cytoplasmic export and stabilizes HMGA2 to promote colorectal liver metastasis. Nat. Commun. 2019, 10, 4695. [Google Scholar] [CrossRef]
- Rong, D.; Wu, F.; Lu, C.; Sun, G.; Shi, X.; Chen, X.; Dai, Y.; Zhong, W.; Hao, X.; Zhou, J.; et al. m6A modification of circHPS5 and hepatocellular carcinoma progression through HMGA2 expression. Mol. Ther. Nucleic Acids 2021, 26, 637–648. [Google Scholar] [CrossRef]
- Ji, F.; Lu, Y.; Chen, S.; Yu, Y.; Lin, X.; Zhu, Y.; Luo, X. IGF2BP2-modified circular RNA circARHGAP12 promotes cervical cancer progression by interacting m6A/FOXM1 manner. Cell Death Discov. 2021, 7, 215. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-Y.; Lin, Y.-C.; Cui, S.; Huang, Y.; Tang, Y.; Xu, J.; Bao, J.; Li, Y.; Wen, J.; Zuo, H.; et al. miRTarBase update 2022: An informative resource for experimentally validated miRNA–target interactions. Nucleic Acids Res. 2021, 50, D222–D230. [Google Scholar] [CrossRef]
- Han, X.; Guo, J.; Fan, Z. Interactions between m6A modification and miRNAs in malignant tumors. Cell Death Dis. 2021, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Alarcón, C.R.; Lee, H.; Goodarzi, H.; Halberg, N.; Tavazoie, S.F. N6-methyladenosine marks primary microRNAs for processing. Nature 2015, 519, 482–485. [Google Scholar] [CrossRef] [PubMed]
- Bhat, S.S.; Bielewicz, D.; Gulanicz, T.; Bodi, Z.; Yu, X.; Anderson, S.J.; Szewc, L.; Bajczyk, M.; Dolata, J.; Grzelak, N.; et al. mRNA adenosine methylase (MTA) deposits m6A on pri-miRNAs to modulate miRNA biogenesis in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2020, 117, 21785–21795. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Zhang, J.; Bin Liu, B.; Xu, J.; Cai, B.; Yang, H.; Straube, J.; Yu, X.; Ma, T. Biological roles of RNA m5C modification and its implications in Cancer immunotherapy. Biomark. Res. 2022, 10, 15. [Google Scholar] [CrossRef] [PubMed]
- Bohnsack, K.E.; Höbartner, C.; Bohnsack, M.T. Eukaryotic 5-methylcytosine (m5C) RNA Methyltransferases: Mechanisms, Cellular Functions, and Links to Disease. Genes 2019, 10, 102. [Google Scholar] [CrossRef] [PubMed]
- Basanta-Sanchez, M.; Wang, R.; Liu, Z.; Ye, X.; Li, M.; Shi, X.; Agris, P.F.; Zhou, Y.; Huang, Y.; Sheng, J. TET1-Mediated Oxidation of 5-Formylcytosine (5fC) to 5-Carboxycytosine (5caC) in RNA. ChemBioChem 2016, 18, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Auxilien, S.; Guérineau, V.; Szweykowska-Kulińska, Z.; Golinelli-Pimpaneau, B. The human tRNA m5C methyltransferase Misu is multisite-specific. RNA Biol. 2012, 9, 1331–1338. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yang, Y.; Sun, B.-F.; Chen, Y.-S.; Xu, J.-W.; Lai, W.-Y.; Li, A.; Wang, X.; Bhattarai, D.P.; Xiao, W.; et al. 5-methylcytosine promotes mRNA export—NSUN2 as the methyltransferase and ALYREF as an m5C reader. Cell Res. 2017, 27, 606–625. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Dunker, W.; Yu, Y.-T.; Karijolich, J. The Role of Noncoding RNA Pseudouridylation in Nuclear Gene Expression Events. Front. Bioeng. Biotechnol. 2018, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Levi, O.; Arava, Y.S. Pseudouridine-mediated translation control of mRNA by methionine aminoacyl tRNA synthetase. Nucleic Acids Res. 2020, 49, 432–443. [Google Scholar] [CrossRef]
- Rajan, K.S.; Adler, K.; Madmoni, H.; Peleg-Chen, D.; Cohen-Chalamish, S.; Doniger, T.; Galili, B.; Gerber, D.; Unger, R.; Tschudi, C.; et al. Pseudouridines on Trypanosoma brucei mRNAs are developmentally regulated: Implications to mRNA stability and protein binding. Mol. Microbiol. 2021, 116, 808–826. [Google Scholar] [CrossRef]
- Vaidyanathan, P.P.; AlSadhan, I.; Merriman, D.K.; Al-Hashimi, H.M.; Herschlag, D. Pseudouridine and N6-methyladenosine modifications weaken PUF protein/RNA interactions. RNA 2017, 23, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Guzzi, N.; Muthukumar, S.; Cieśla, M.; Todisco, G.; Ngoc, P.C.T.; Madej, M.; Munita, R.; Fazio, S.; Ekström, S.; Mortera-Blanco, T.; et al. Pseudouridine-modified tRNA fragments repress aberrant protein synthesis and predict leukaemic progression in myelodysplastic syndrome. Nature 2022, 24, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Seo, K.W.; Kleiner, R.E. YTHDF2 Recognition of N1-Methyladenosine (m1A)-Modified RNA Is Associated with Transcript Destabilization. ACS Chem. Biol. 2019, 15, 132–139. [Google Scholar] [CrossRef]
- Dominissini, D.; Nachtergaele, S.; Moshitch-Moshkovitz, S.; Peer, E.; Kol, N.; Ben-Haim, M.S.; Dai, Q.; Di Segni, A.; Salmon-Divon, M.; Clark, W.C.; et al. The dynamic N1-methyladenosine methylome in eukaryotic messenger RNA. Nature 2016, 530, 441–446. [Google Scholar] [CrossRef]
- Liu, F.; Clark, W.; Luo, G.; Wang, X.; Fu, Y.; Wei, J.; Wang, X.; Hao, Z.; Dai, Q.; Zheng, G.; et al. ALKBH1-Mediated tRNA Demethylation Regulates Translation. Cell 2016, 167, 816–828.e16. [Google Scholar] [CrossRef]
- Chujo, T.; Suzuki, T. Trmt61B is a methyltransferase responsible for 1-methyladenosine at position 58 of human mitochondrial tRNAs. RNA 2012, 18, 2269–2276. [Google Scholar] [CrossRef]
- Fu, L.; Guerrero, C.R.; Zhong, N.; Amato, N.J.; Liu, Y.; Liu, S.; Cai, Q.; Ji, D.; Jin, S.-G.; Niedernhofer, L.J.; et al. Tet-Mediated Formation of 5-Hydroxymethylcytosine in RNA. J. Am. Chem. Soc. 2014, 136, 11582–11585. [Google Scholar] [CrossRef]
- Rácz, I.; Király, I.; Lásztily, D. Effect of light on the nucleotide composition of rRNA of wheat seedlings. Planta 1978, 142, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Huber, S.M.; van Delft, P.; Mendil, L.; Bachman, M.; Smollett, K.; Werner, F.; Miska, E.A.; Balasubramanian, S. Formation and Abundance of 5-Hydroxymethylcytosine in RNA. ChemBioChem 2015, 16, 752–755. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Chen, D.; Su, R.; Chen, W.; Wei, L. iRNA5hmC: The First Predictor to Identify RNA 5-Hydroxymethylcytosine Modifications Using Machine Learning. Front. Bioeng. Biotechnol. 2020, 8, 227. [Google Scholar] [CrossRef] [PubMed]
- Delatte, B.; Wang, F.; Ngoc, L.V.; Collignon, E.; Bonvin, E.; Deplus, R.; Calonne, E.; Hassabi, B.; Putmans, P.; Awe, S.; et al. RNA biochemistry. Transcriptome-wide distribution and function of RNA hydroxymethylcytosine. Science 2016, 351, 282–285. [Google Scholar] [CrossRef]
- Miao, Z.; Xin, N.; Wei, B.; Hua, X.; Zhang, G.; Leng, C.; Zhao, C.; Wu, D.; Li, J.; Ge, W.; et al. 5-hydroxymethylcytosine is detected in RNA from mouse brain tissues. Brain Res. 2016, 1642, 546–552. [Google Scholar] [CrossRef]
- Ahmed, S.; Hossain, Z.; Uddin, M.; Taherzadeh, G.; Sharma, A.; Shatabda, S.; Dehzangi, A. Accurate prediction of RNA 5-hydroxymethylcytosine modification by utilizing novel position-specific gapped k-mer descriptors. Comput. Struct. Biotechnol. J. 2020, 18, 3528–3538. [Google Scholar] [CrossRef]
- Rebagliati, M.; Melton, D. Antisense RNA injections in fertilized frog eggs reveal an RNA duplex unwinding activity. Cell 1987, 48, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Quin, J.; Sedmík, J.; Vukić, D.; Khan, A.; Keegan, L.P.; O’Connell, M.A. ADAR RNA Modifications, the Epitranscriptome and Innate Immunity. Trends Biochem. Sci. 2021, 46, 758–771. [Google Scholar] [CrossRef]
- Kukharsky, M.S.; Ninkina, N.N.; An, H.; Telezhkin, V.; Wei, W.; de Meritens, C.R.; Cooper-Knock, J.; Nakagawa, S.; Hirose, T.; Buchman, V.L.; et al. Long non-coding RNA Neat1 regulates adaptive behavioural response to stress in mice. Transl. Psychiatry 2020, 10, 1–19. [Google Scholar] [CrossRef]
- Zhang, Y.; Lun, L.; Li, H.; Wang, Q.; Lin, J.; Tian, R.; Pan, H.; Zhang, H.; Chen, X. The Value of lncRNA NEAT1 as a Prognostic Factor for Survival of Cancer Outcome: A Meta-Analysis. Sci. Rep. 2017, 7, 13080. [Google Scholar] [CrossRef]
- Ning, L.; Li, Z.; Wei, D.; Chen, H.; Yang, C. LncRNA, NEAT1 is a prognosis biomarker and regulates cancer progression via epithelial-mesenchymal transition in clear cell renal cell carcinoma. Cancer Biomarkers 2017, 19, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Vlachogiannis, N.I.; Sachse, M.; Georgiopoulos, G.; Zormpas, E.; Bampatsias, D.; Delialis, D.; Bonini, F.; Galyfos, G.; Sigala, F.; Stamatelopoulos, K.; et al. Adenosine-to-inosine Alu RNA editing controls the stability of the pro-inflammatory long noncoding RNA NEAT1 in atherosclerotic cardiovascular disease. J. Mol. Cell. Cardiol. 2021, 160, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Loeb, S.; Partin, A.W. Review of the literature: PCA3 for prostate cancer risk assessment and prognostication. Rev. Urol. 2011, 13, e191–e195. [Google Scholar]
- Salameh, A.; Lee, A.K.; Cardó-Vila, M.; Nunes, D.N.; Efstathiou, E.; Staquicini, F.I.; Dobroff, A.S.; Marchiò, S.; Navone, N.M.; Hosoya, H.; et al. PRUNE2 is a human prostate cancer suppressor regulated by the intronic long noncoding RNA PCA3. Proc. Natl. Acad. Sci. USA 2015, 112, 8403–8408. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Chendrimada, T.P.; Wang, Q.; Higuchi, M.; Seeburg, P.H.; Shiekhattar, R.; Nishikura, K. Modulation of microRNA processing and expression through RNA editing by ADAR deaminases. Nat. Struct. Mol. Biol. 2006, 13, 13–21. [Google Scholar] [CrossRef]
- Paul, D.; Sinha, A.N.; Ray, A.; Lal, M.; Nayak, S.; Sharma, A.; Mehani, B.; Mukherjee, D.; Laddha, S.V.; Suri, A.; et al. A-to-I editing in human miRNAs is enriched in seed sequence, influenced by sequence contexts and significantly hypoedited in glioblastoma multiforme. Sci. Rep. 2017, 7, 2466. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; An, O.; Ren, X.; Song, Y.; Tang, S.J.; Ke, X.-Y.; Han, J.; Tay, D.J.T.; Ng, V.H.E.; Molias, F.B.; et al. ADARs act as potent regulators of circular transcriptome in cancer. Nat. Commun. 2022, 13, 1508. [Google Scholar] [CrossRef] [PubMed]
- Edupuganti, R.R.; Geiger, S.; Lu, Z.; Wang, S.-Y.; Baltissen, M.P.A.; Jansen, P.W.T.C.; Rossa, M.; Müller, M.; Stunnenberg, H.G.; He, C.; et al. N6-methyladenosine (m6A) recruits and repels proteins to regulate mRNA homeostasis. Nat. Struct. Mol. Biol. 2017, 24, 870–878. [Google Scholar] [CrossRef] [PubMed]
- Mendel, M.; Delaney, K.; Pandey, R.R.; Chen, K.-M.; Wenda, J.M.; Vågbø, C.B.; Steiner, F.A.; Homolka, D.; Pillai, R.S. Splice site m6A methylation prevents binding of U2AF35 to inhibit RNA splicing. Cell 2021, 184, 3125–3142.e25. [Google Scholar] [CrossRef] [PubMed]
- Sajini, A.A.; Choudhury, N.R.; Wagner, R.E.; Bornelöv, S.; Selmi, T.; Spanos, C.; Dietmann, S.; Rappsilber, J.; Michlewski, G.; Frye, M. Loss of 5-methylcytosine alters the biogenesis of vault-derived small RNAs to coordinate epidermal differentiation. Nat. Commun. 2019, 10, 2550. [Google Scholar] [CrossRef] [PubMed]
- Amort, T.; Soulière, M.F.; Wille, A.; Jia, X.-Y.; Fiegl, H.; Wörle, H.; Micura, R.; Lusser, A. Long non-coding RNAs as targets for cytosine methylation. RNA Biol. 2013, 10, 1002–1008. [Google Scholar] [CrossRef] [PubMed]
- Delorimier, E.; Hinman, M.N.; Copperman, J.; Datta, K.; Guenza, M.; Berglund, J.A. Pseudouridine Modification Inhibits Muscleblind-like 1 (MBNL1) Binding to CCUG Repeats and Minimally Structured RNA through Reduced RNA Flexibility. J. Biol. Chem. 2017, 292, 4350–4357. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).