Abstract
In this contribution, we provide an overview of gold compound applications against viruses or parasites during recent years. The special properties of gold have been the subject of intense investigation in recent years, which has led to the development of its chemistry with the synthesis of new compounds and the study of its applicability in various areas such as catalysis, materials, nanotechnology and medicine. Herein, thirteen gold articles with applications in several viruses, such as hepatitis C virus (HCV), influenza A virus (H1N1), vesicular stomatitis virus (VSV), coronavirus (SARS-CoV and SARS-CoV-2), Dengue virus, and several parasites such as Plasmodium sp., Leishmania sp., Tripanossoma sp., Brugia sp., Schistosoma sp., Onchocerca sp., Acanthamoeba sp., and Trichomonas sp. are described. Gold compounds with anti-viral activity include gold nanoparticles with the ligands mercaptoundecanosulfonate, 1-octanethiol and aldoses and gold complexes with phosphine and carbene ligands. All of the gold compounds with anti-parasitic activity reported are gold complexes of the carbene type. Auranofin is a gold drug already used against rheumatoid arthritis, and it has also been tested against virus and parasites.
1. Introduction
Through the ages in most major civilizations, gold has been the chemical element that has attracted the most attention and desire, due to its unique characteristics such as its bright yellow color, corrosion resistance and extraordinary physical properties. In the form of amulets and medallions, it was used to ward off disease and evil spirits. In many cases, potions containing gold powders were administered to ill patients. After the alchemists learned to use aqua regia to dissolve gold, compounds as well as elemental gold were used in medicinal treatments [1].
Nowadays, the properties of gold are interpreted with the application of the theory of relativity, hence providing an explanation for a series of properties for the gold complexes that differ from those of its congeners, such as a preference for linear coordination, a higher stability of higher oxidation states, the formation of gold compounds and the establishment of gold–gold interactions with a similar strength to hydrogen bonds [2,3]. All these facts promoted the research in gold chemistry in different areas from nanotechnology and material chemistry [4,5,6] to catalysis [7,8,9,10,11,12,13,14,15] and medicine [16,17,18,19].
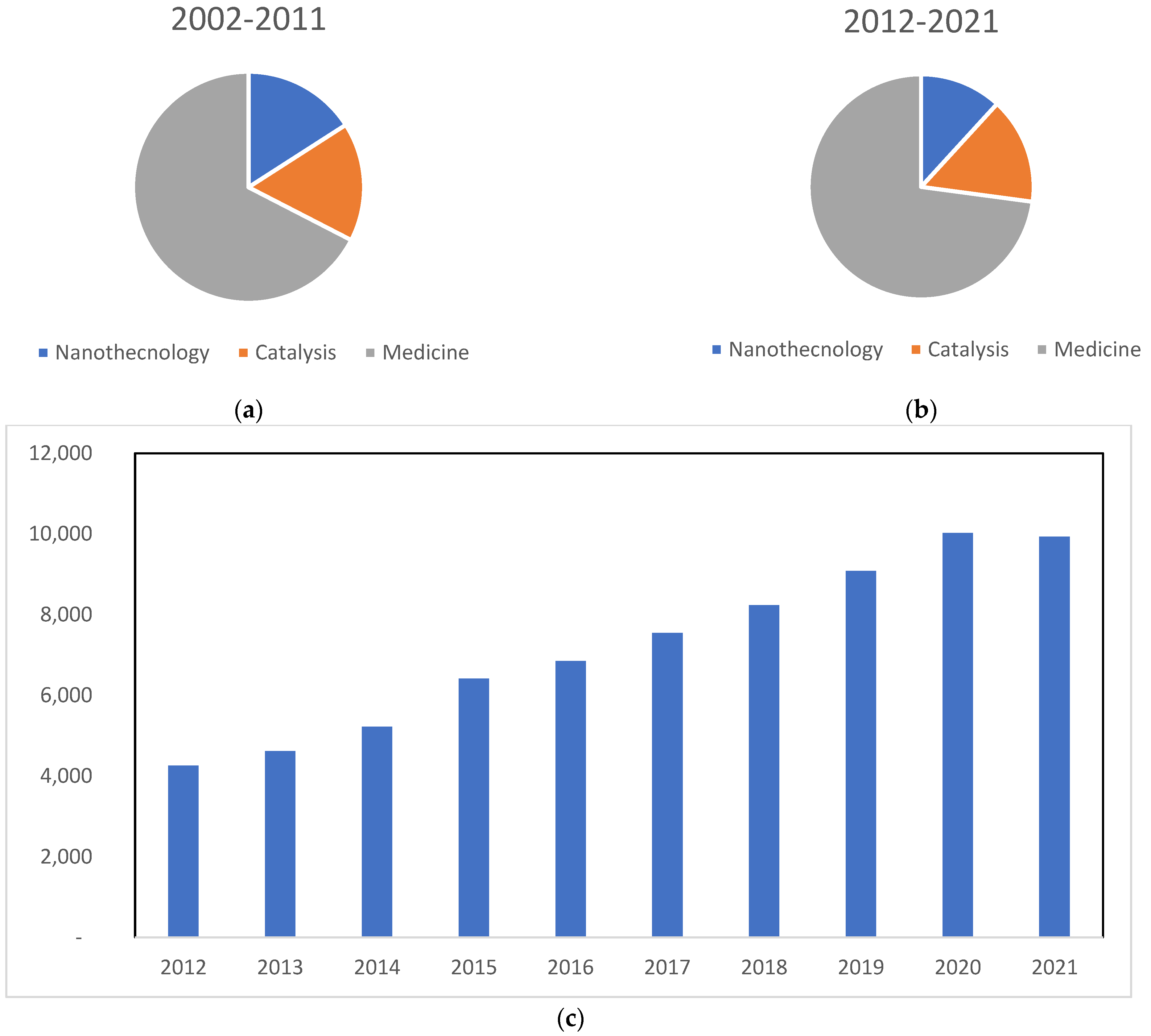
Gold compounds are well-known for their biological and medicinal applications, for instance, in rheumatoid arthritis, antibacterial, antivirus, and anti-parasite activity, as well as in Alzheimer’s disease [18,20,21,22,23,24,25,26]. The mechanisms of action of gold compounds are mostly unknown; however, several proteins have been studied as possible targets, such as tyrosine phosphatases, aquaporins and P-ATPases, among others [17,26,27,28,29]. However, the putative pharmacological targets of gold compounds are not the objective of this review, and the reader should look elsewhere for this topic [17,18,22,27,29]. Herein, we aim to reviewed recent applications of gold compounds against virus and parasites. It was observed that, after a search carried out in the Web of Science with the keyword “gold compound”, 99,014 articles were published in the last 10 years (2012–2021), more than triple the total in the previous period (2002–2011). Another fact to bear in mind is the steady increase over time, especially in the field of medicine. Although there are many articles and studies that overlap between the field of medicine and nanotechnology, specific papers on gold compounds in these categories, plus catalysis, can be found (Figure 1).
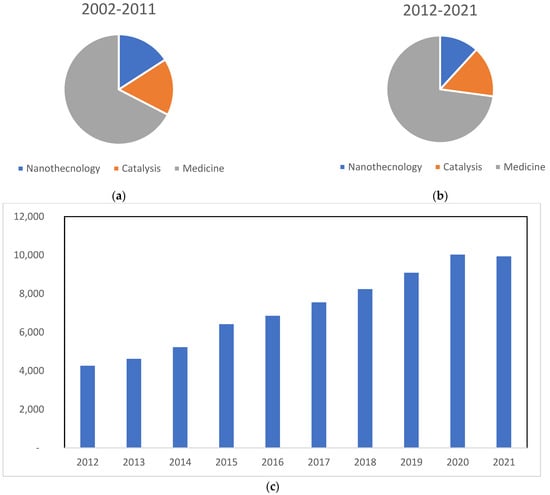
Figure 1.
Number of articles indexed on the Web of Science obtained using the keywords “gold compounds” and “nanotechnology”, “gold compounds” and “catalysis” and “gold compounds” and “medicine”. (a) Graph referring to the decade of 2002–2011; (b) graph referring to the decade of 2012–2021; (c) graph referring to articles published using gold compounds in medicine from 2012 until now. Review articles were excluded.
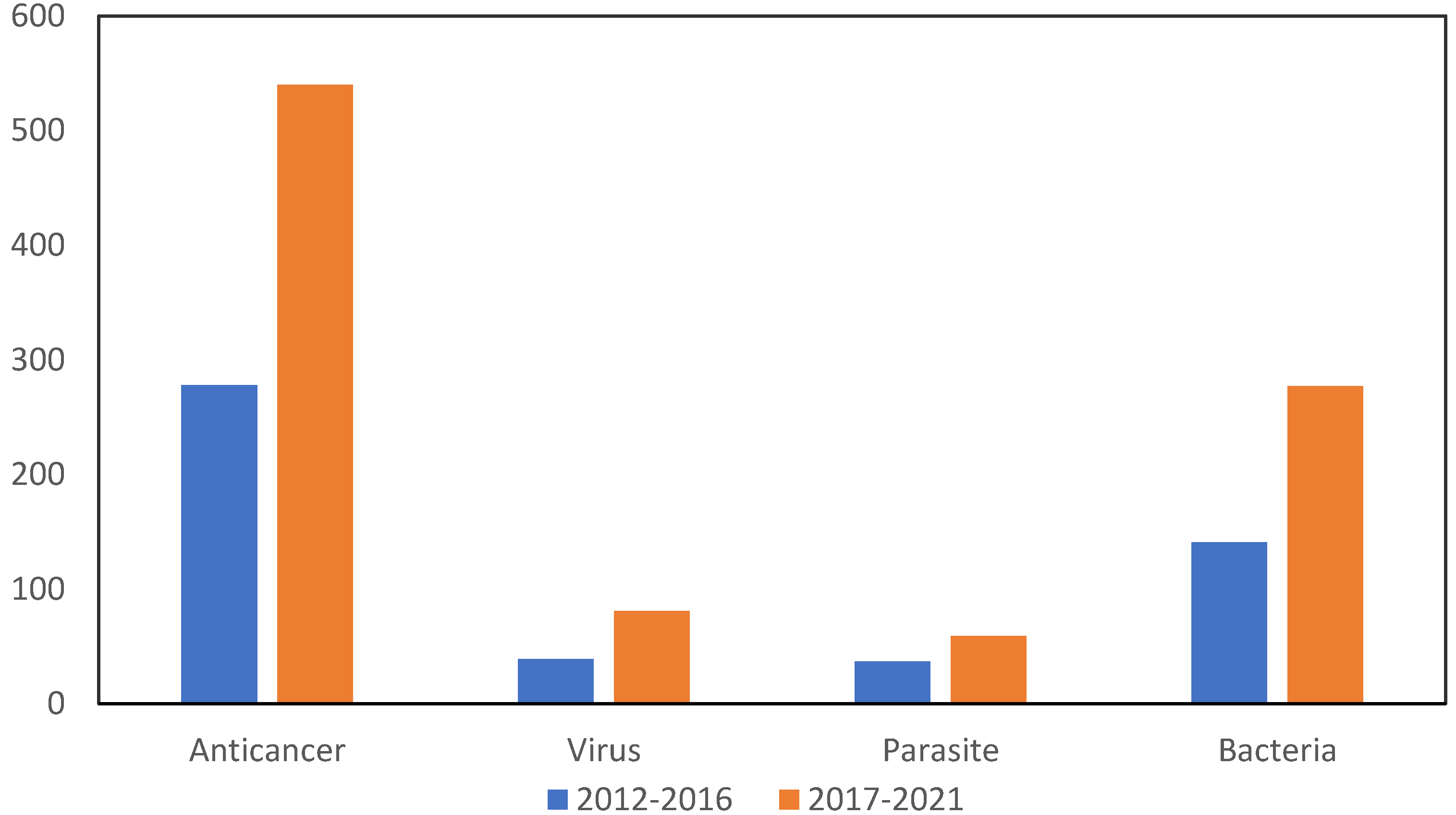
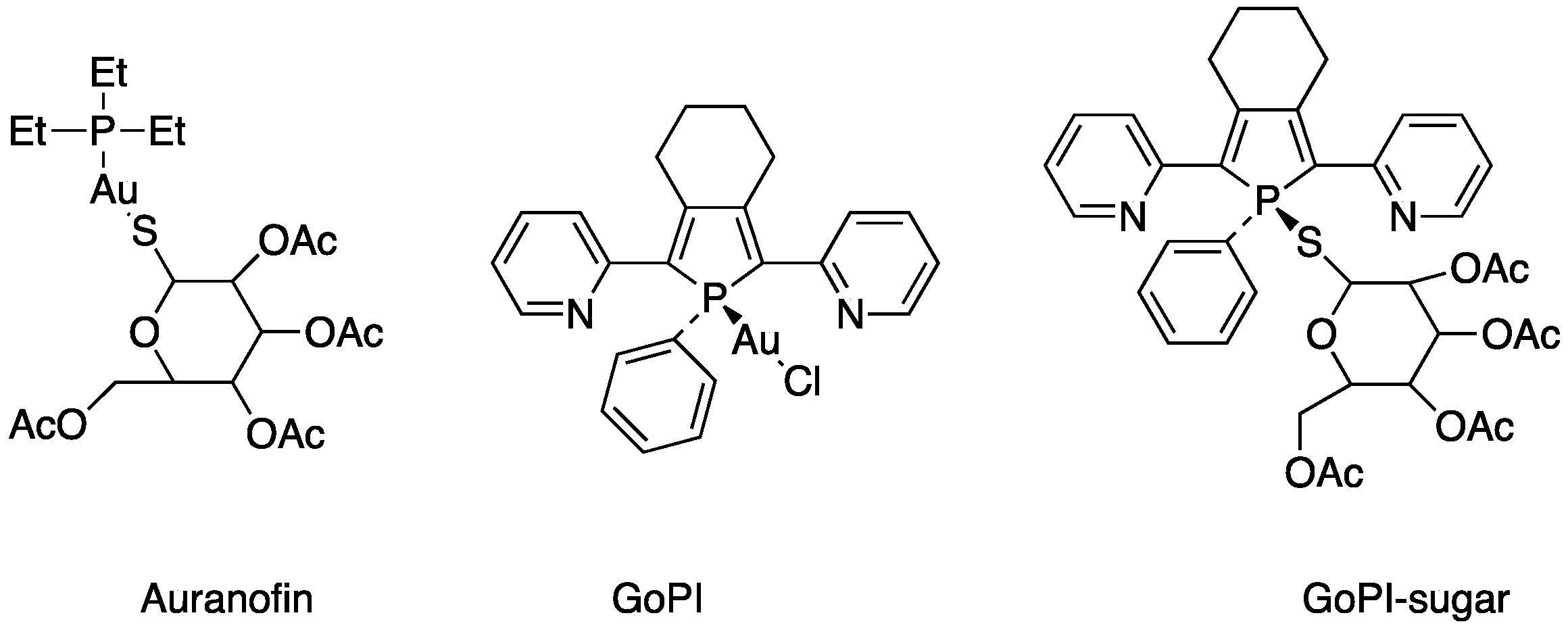
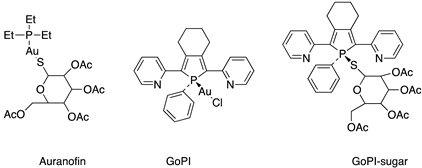
Although the first widespread application of gold compounds was against pulmonary tuberculosis, for which they do not provide an effective therapy, it is in the treatment of rheumatoid arthritis that gold compounds have proved effective [30]. Auranofin, triethylphosphine (2,3,4,6-tetra-O-acetyl-β-1-D-thiopyranosato-S) gold(I) is the example of this, as it continues to be used in the clinic today and is the subject of research [31,32]. Other gold compounds are also under investigation in the treatment of diseases such as cancer, viruses such as human HIV and SARS-CoV-2 and parasitosis such as malaria and amoebiasis (Figure 2).
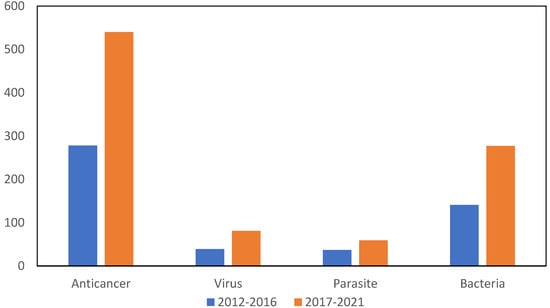
Figure 2.
Number of articles indexed to Web of Science where gold compounds are investigated for biological activity against cancer, parasitosis and viruses in last decade (2012–2021).
This paper will present a systematic review on the topic of “biological activity of gold compounds against virus and parasites”. The information obtained from the database is grouped and analyzed to examine the applications of gold molecules in diseases caused by those two types of organisms.
2. Methodology
In this systematic review, the Web of Science was chosen as a database. The search terms were “gold compound” and the target “virus” or “parasite”. The literature search was carried out for the last two years (2020–2021).
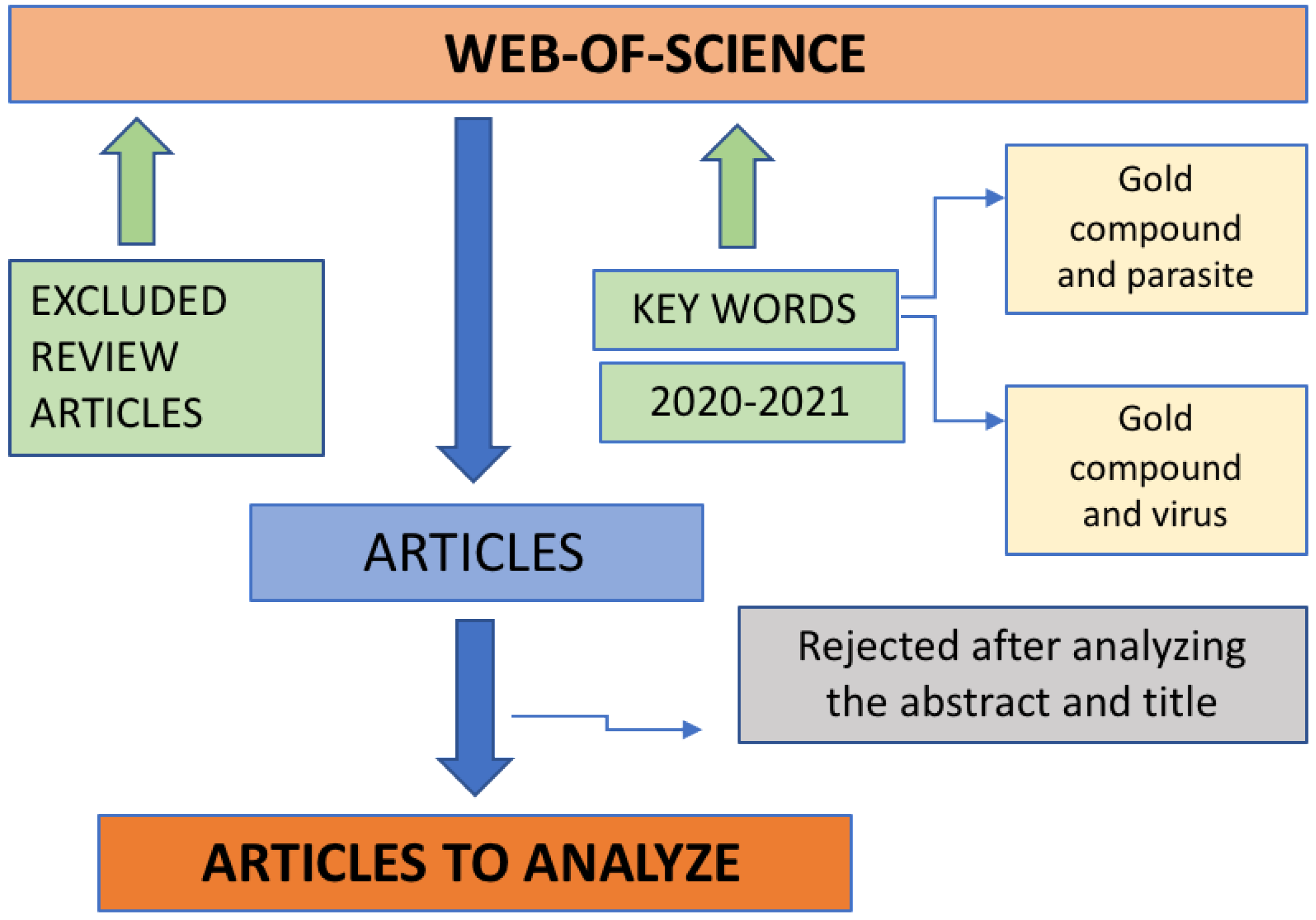
The initial research was carried out by introducing the keywords in the database, Web of Science, and the range date was 1 January 2020 to 30 December 2021. From the set of articles obtained, review articles were excluded and then the titles and abstracts were read in order to check if the study reported in the articles was within the theme of this review article, then it could be included or excluded (Figure 3).
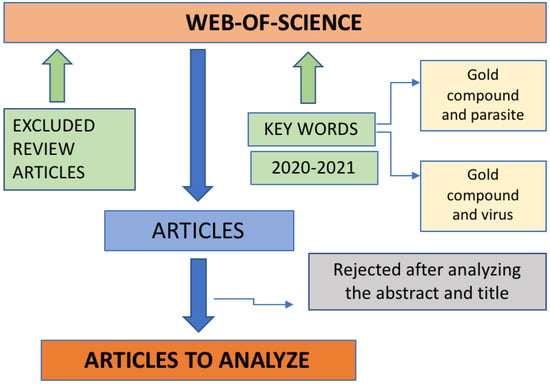
Figure 3.
Diagram of the method used to select the articles to be analyzed.
The analysis of the articles considered the type of compound studied and where the biological activity was determined.
3. Gold Compounds with Biological Activity against Virus and Parasites
3.1. Gold Compounds Used in Anti-Virus Studies
From the research carried out in Web of Science with the words “gold compound” and “virus” for the years 2020–2021, 43 articles were obtained, and review articles were excluded, leaving 30. These articles were analyzed by reading the title and abstract, leaving six articles under analysis, which form the basis of this review.
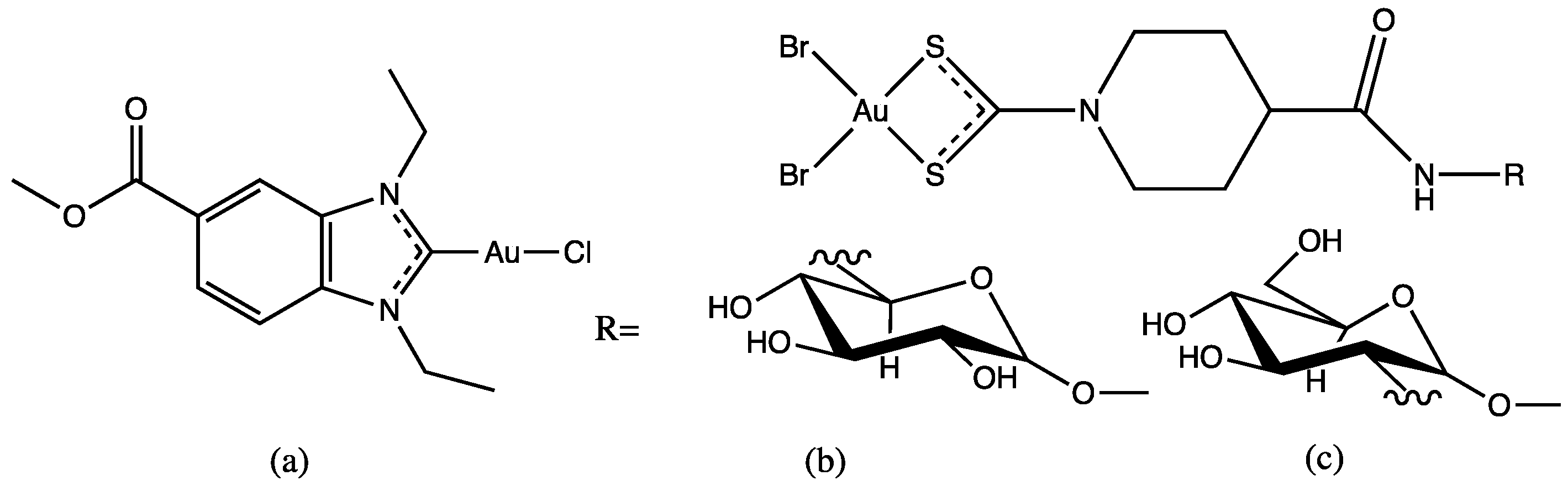
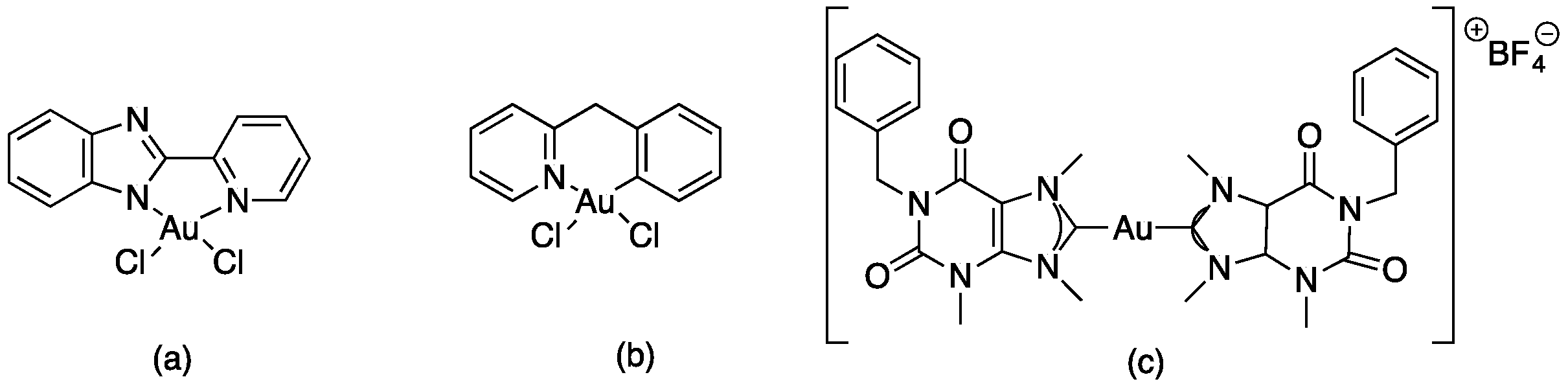
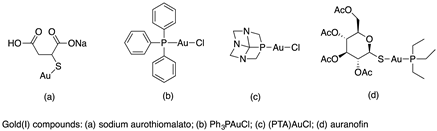
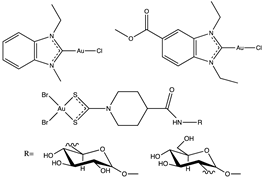
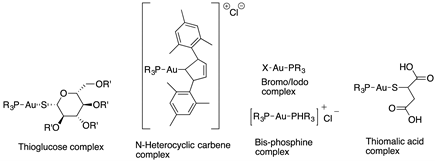
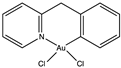
Molecular gold(I) species were examined for the inhibition pf liver fibrosis and the hepatitis C virus (HCV). Four gold(I) complexes, auranofin, sodium aurothiomalate, Ph3PAuCl and (PTA)AuCl (Table 1), were tested against HSC line Lx2, human hepatoma cells and Huh—K2040 cells. Two of these molecules, Ph3PAuCl [24,33] and (PTA)AuCl [34], were synthetized according to the references.

Table 1.
Gold compounds with structures and biological effects against viruses and parasitosis.
As a preliminary screen for anti-fibrotic activity, we used the Lx-2 cell morphology. Auranofin and Ph3PAuCl with 2 µM and 5 µM concentrations were the only active compounds, as they revert back to a quiescent state of the cells.
After being treated with auranofin (2 µM and 5 µM) and Ph3PAuCl (2 µM and 5 µM), the HSC were lysed and type I collagen expression and the proteins STAT3 and SMAD2 were determined. As compared to DMSO-treated human hepatic stellate cells (HSCs), auronafin treatment with concentrations of 2 µM and 5 µM resulted in 50% and 40% decreases in type I collagen expression, respectively. Ph3PAuCl also reduced type I collagen expression, but less dramatically, with only a 10% and 20% reduction at 2 µM and 5 µM, respectively. Next, two proteins, STAT3 and SMAD2, which play roles in type I collagen were determined. At both 2 µM and 5 µM concentrations, auranofin showed a complete abrogation of STAT3 phosphorylation, and also a dramatic increase in SMAD phosphorylation. Ph3PAuCl showed a 60% and 90% inhibition of STAT3 phosphorylation at concentrations of 2 µM and 5 µM, respectively, and also resulted in an increase in SMAD2 phosphorylation, albeit to a lesser extent than auranofin. None of the gold compounds appeared to have a significant impact on α-SMA protein levels. The auranofin 2 µM and 5 µM concentrations show an 80% and 98% decrease, respectively, in type I collagen mRNA, while the α-SMA mRNA levels decreased 80% and 96%, respectively. Meanwhile, for Ph3PAuCl, the decrease in collagen-I mRNA expression is not noticeable, while α-SMA expression is nearly 70% and 50% lower, respectively. A decrease in NS3 and NS5a expression in Huh7 K2040 cells was observed after being treated with auranofin and Ph3PAuCl. Auranofin at 5 µM inhibits 60% of HCV-NS5a, while it causes 50% inhibition of HCV-NS3 and a complete abrogation of STAT3 phosphorylation. In conclusion, these results indicate the inhibitory potential of these drugs against HCV protein expression [24].
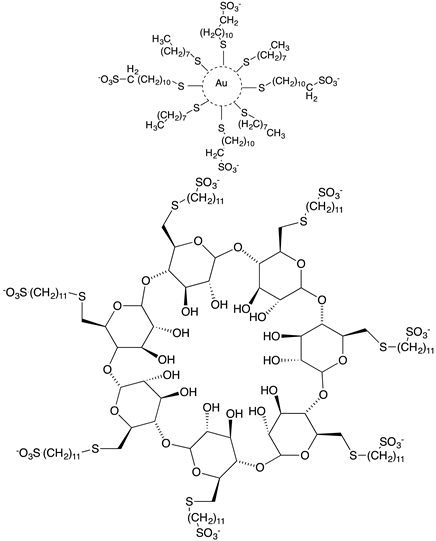
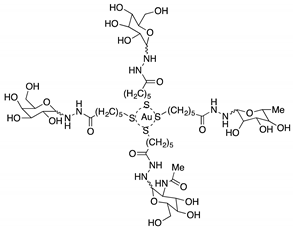
Cagno, V. et al. designed two compounds: in the first compound named MUST: OT NP (Table 1), the moiety was fixed on gold nanoparticles (4 nm gold nanoparticles (NPs) coated with a mixture of octane thiol (OT) and mercapto-undecane sulfonic acid (MUST) [35,48,49]); in the second compound, named CD1, the moiety was linked to the primary face of β-cyclodextrins (CD) [50]. In previous study, it was demonstrated that gold nanoparticles coated with mercapto-undecane sulfonic acid (MUS) inhibit heparan sulfate proteoglycan (HSPG)-dependent viruses irreversibly while retaining the low-toxicity profile [51]. These compounds display inhibitory activity in the absence of toxicity, with 50% effective concentrations, (EC50), values between 1.38 and 12.0 µM/m and for H1N1 stains. For vesicular stomatitis virus (VSV), the EC50 value is 0.053 µM/mL relative to compound MUS:OT-NP; for CD1, the EC50 determined was higher (for VSV, 45 µM, and for H1N1, between 6.28 and 53.2 µM).
The antiviral action is irreversible for influenza A virus (H1N1), while for VSV, the inhibition is reversible. These results further broaden the spectrum of activity of MUS-coated gold nanoparticles [35] (Table 1).
Synthesized gold glyconanoparticles [37] (Table 1) were tested against the Influenza virus H1N1 (A/Puerto Rico/8/34) via a standard protocol [36]. The nanoparticles revealed low cytotoxicity towards the MDCK cells (higher than 100 µg/mL) and high antiviral activity at concentrations of 3 and 6 μg/mL. The strongest antiviral activity was observed for the sample with a glycoligand composition GlcNAc:Man:Fuc = 20:75:5% mol.
These preliminary data on the activity against the A/Puerto Rico/8/34 (H1N1) demonstrated the prospects of their further investigation for the search for efficient drugs for the prevention and treatment of acute respiratory viral infections [36] (Table 1).
Among more than 100 structurally diverse metal complexes, 36 gold(I) and (III) complexes with different types of co-ligands, including N-heterocyclic carbenes (NHCs), alkynyls, dithiocarbamates, phosphines and chlorides, were selected for profiling as inhibitors of SARS-CoV-2. The inhibition can occur by means of one of two replication mechanisms, namely the interaction of the spike (S) protein with the ACE2 receptor and the papain-like protease PLpro. The chloroauric acid (HAuCl4; oxidation state + 3) has moderate inhibition (about 47% inhibitory activity, the other gold compounds were poorly active or inactive) [40].
Based on previous results, the chosen inhibitor concentration was 10 µM for assays with SARS-CoV PLpro, while 1.0 µM was used for SARS-CoV-2 PLpro. Ten gold complexes were tested against PLpro of SARS-CoV, and the values determined range from 0.3 to 1.2 μM, and against SARS-CoV-2 PLpro, the values determined range from 0.1 to 1.5 μM. These results confirm the high potential of gold compounds as protease-inhibiting antiviral drugs. For gold(III) dithiocarbamates, compounds 3 and 4 (Figure 4) had an IC50 of 0.21 µM and 0.09 µM, respectively. These molecules were the most active SARS-CoV-2 PLpro inhibitors identified in this study and show strong preference for the enzyme of SARS-CoV-2. Compounds 1 and 2 (Figure 4), with an IC50 of 0.35 µM and 0.33 µM, were stronger inhibitors for the SARS-CoV enzyme.
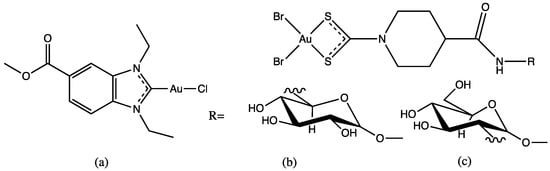
Figure 4.
Gold complexes tested against SARS-CoV-2: (a) Gold(I) NHC complex; (b) and (c) are Gold(III)-dithiocarbamato complexes.
The evaluation of structure–activity relationships indicated a preference for complexes with good leaving groups (e.g. chloride) over compounds with firmly coordinated ligands such as dicarbene gold complexes of the type (NHC)2Au+, which were inactive. Exceptionally strong and selective activity against SARS-CoV-2 PLpro was obtained with the gold(III)-dithiocarbamato glycoconjugates, compounds (b) and (c) in Figure 4. Most of the gold complexes tested showed too strong toxicity; however, for four compounds, very low or missing toxicity against the Caco-2 cell line was noted, and these four complexes were selected for the SARS-CoV-2 antiviral assays. Among these, the gold(I) NHC complex (a) showed very promising activity at low micromolar concentration, and two gold(III)-dithiocarbamato complexes (b) and (c), Figure 4,were strongly active at the highest applied nontoxic dosage [40].
Auranofin inhibits SARS-CoV-2 replication in human cells (Huh7 cells) at a low micromolar concentration (EC50 1.4 μM). The treatment of cells with auranofin resulted in a 95% reduction in the viral RNA at 48 h after infection. Auranofin treatment dramatically reduced the expression of SARS-CoV-2-induced cytokines in human cells. These data indicate that auranofin could be a useful drug to limit SARS-CoV-2 infection and associated lung injury due to its antiviral, anti-inflammatory and anti-reactive oxygen species (ROS) properties. Further animal studies are warranted to evaluate the safety and efficacy of auranofin for the management of SARS-CoV-2 associated disease [38].
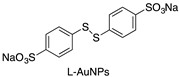
Different gold nanoparticles (AuNP) coated with ligands ω-terminated with sugars bearing multiple sulfonate groups (L-AuNPs) (Table 1) were tested against Dengue virus (DENV). The ligands varied in length, in number of sulfonated groups, as well as their spatial orientation induced by the sugar head groups. Two candidates were identified, a glucose- and a lactose-based ligand showing a low EC50 (effective concentration that inhibits 50% of the viral activity) for DENV-2 inhibition, moderate toxicity and a virucidal effect in hepatocytes with a titer reduction in the median tissue culture infectious dose log10TCI D50 2.5 and 3.1. Molecular docking simulations complemented the experimental findings, suggesting a molecular rationale behind the binding between sulfonated head groups and DENV-2 envelope protein [39].
3.2. Gold Compounds Used in Anti-Parasite Studies
From the research carried out in Web of Science with the words “gold compound” and “parasite” for the years 2020–2021, 27 articles were obtained, and review articles were excluded, leaving 23. These articles were analyzed by reading the title and abstract, leaving six articles under analysis, which form the basis of this review.
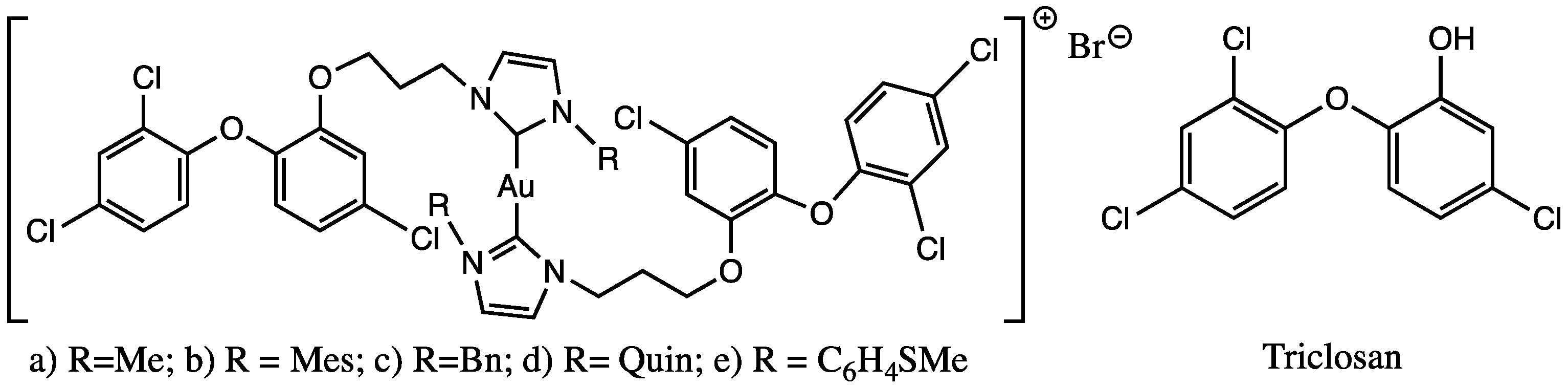
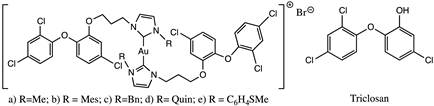
Ouji et al. synthesized and characterized five hybrid molecules combining triclosan and gold(I) complexes, gold(I)-NHC-TC (Table 1), and evaluated them, in vitro, to determine the activities on Plasmodium falciparum and Leishmania infantum parasites [41,42].
The antimalarial activity of the hybrid molecules was tested using P. falciparum strain F32-TEM and the IC50 determined range from 120 nM to 1465 nM. These values were determined in comparison with the corresponding proligand, triclosan, and artemether, the antiplasmodial control drug. Globally, the presence of gold(I) can have opposite effects, with an improvement in efficacy compared to the corresponding proligand; triclosan showed weak antiplasmodial activity with an IC50 value of 6000 nM, and the artemether had an IC50 of 6.1 nM.
All the hybrid compounds and their carbene precursors were also screened in vitro on both the promastigote and axenic amastigote stages of L. infantum, and the IC50 values compare to reference drugs for Leishmaniosis, as well as triclosan and auranofin. The results obtained against promastigotes of L. infantum show moderate or no activity with an IC50 range from 5.51 µM to 63.87 µM. Looking at the activities against promastigotes of the proligand/complex couples, there was just one clear enhanced addition of gold(I) in the compound a (Figure 5). In the case of axenic amastigotes L. infantum, the IC50 was 0.21–1.54 µM. The data observed could not be totally established in relation to the amastigote activity. These Au(I)bis(NHC-TC) series had similar activity to previously reported cationic Au(I)bis(NHC) and neutral Au(I)(NHC)Cl complexes including aliphatic and aromatic substituents [52,53], which suggests that no enhanced activity was found by the addition of the triclosan moiety. For compound a (Figure 5), a selectivity index (S.I.) value of 7.5 was determined, which was comparable to 6.0 for triclosan; these low S.I. values mean that compound a has the same range of activity against normal mammalian cells and P. falciparum. Concerning Leishmania, the S.I. value of amastigote/promastigote ranged from 4.99 to 14.81 for the proligands, and from 5.50 to 26.65 for the complexes, with increased selectivity in most cases by the coordination of the gold(I) on the NHCs. Auranofin exhibited higher activity and selectivity (IC50(promastigotes) 4.32 µM; S.I. 2.67 and IC50 (amastigotes) 0.07 µM; S.I. 38.14) and on two stages of L infantum than all complexes. The results obtained demonstrate that the incorporation of the triclosan derivative in complexes could be related to a loss in selectivity, without increasing the activity.
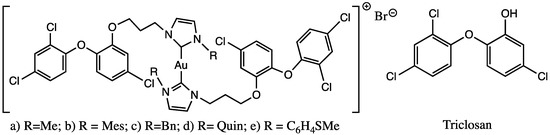
Figure 5.
Gold(I)-NHC-TC hybrid molecule and triclosan compound screening against Leishmania sp. and Plasmodium sp.
However, the cytotoxicity assays suggest a structure modulation to improve the selectivity and the IC50 values too.
The synthesized molecules were also tested in the context of Plasmodium falciparum artemisinin-resistance (strain F32-ART), and the results show no cross-resistance induced between one gold(I) complex and artemisinin [41].
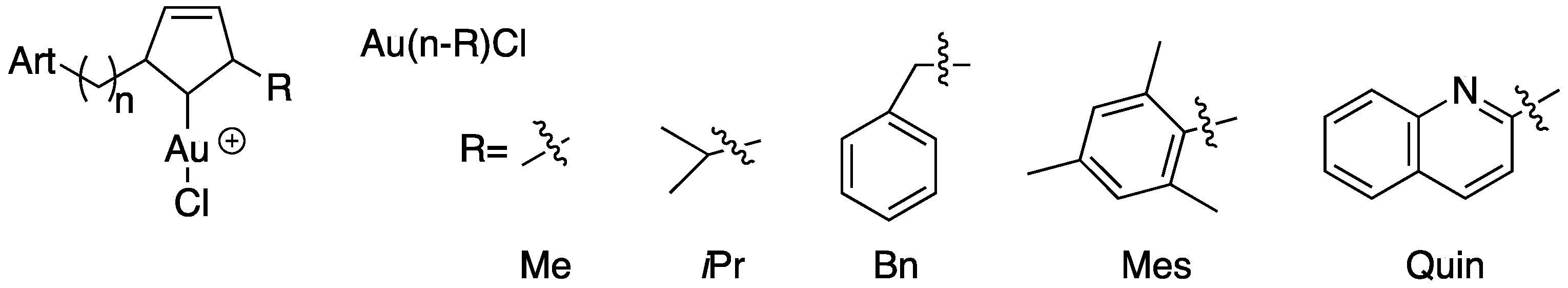
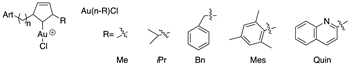
The same author as previously reported synthesized and fully characterized three families of gold(I) NHC complexes which incorporated a covalently attached dihydroartemisinin (DHA) derivative (Figure 6). The proligands (Ln-R) and the complexes (Au-bis(n-R) and Au(n-R)Cl), as well as reference molecules (artemisinin, artemether, auranofin and the proligand precursor DHA-C3), were screened in vitro against the P. falciparum, and the cytotoxicity on Vero cells was evaluated to determine the selectivity of the most active compounds. Among the 29 complexes compounds tested, an IC50 value range from 9 to 104 nM was obtained, and ten of them showed high antiplasmodial activities, with IC50 values less than 50 nM. Auranofin, used as a gold reference molecule, was not active against P. falciparum parasites, with a higher IC50 value of 1.5 µM. It was possible to establish a structure–activity relationship, and thus the potency of the complexes containing methyl or benzyl groups on the NHCs increased with the length of the spacer, whereas no correlation was spotted for the mesityl and the quinoline series. The selectivity indexes for the gold(I) complexes were between 8 and 178, with molecules with aliphatic R groups (Me, iPr) having the best selectivity with S.I. values of 143 and 178.
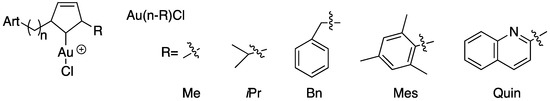
Figure 6.
Dihydroartemisinin derivatives and gold(I)-N-heterocyclic carbene (NHC) complexes.
Three hybrid molecules with the best selectivity indexes Au-bis(3-Me), Au-bis(4-Me) and Au-bis(5-Me) were evaluated in vitro for their efficacy in the context of resistance to artemisinins. The results obtained confirm the existence of a cross-resistance between artemisinin and the hybrid molecules, which can be explained by the DHA part of the hybrid, responsible for the quiescence entrance of the parasites and the lack of activity of the NHC part at the mitochondrial level due to limited access or pharmacodynamic properties. These data are in accordance with previously obtained results which highlighted the risks of parasite cross-resistance between artemisinins and endoperoxide-based compounds [42,52,54].
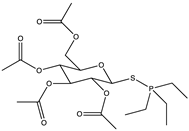
The screening of various parasites—protozoans, trematodes, and nematodes—was undertaken to determine the in vitro killing activity of a new gold(I) complexes, GoPI-sugar (Figure 7). GoPI-sugar is a novel 1-thio-β-D -glucopyranose 2,3,4,6-tetraacetato-S -derivative that is a chimera of the structures of GoPI and auranofin, designed to improve the stability and bioavailability of GoPI (Figure 7). GoPI-sugar was found to efficiently kill intra-macrophagic Leishmania donovani amastigotes and adult filarial and trematode worms. Among new gold(I) complexes, the phosphole-containing gold(I)complex {1-phenyl-2,5-di(2-pyridyl)phosphole}AuCl (abbreviated as GoPI) is an irreversible inhibitor of both purified human glutathione and thioredoxin reductases. Table 2 includes the results of biological activity of the GoPI-sugar compound against different parasites, expressed in killing % activity, motibility % inhibition and 50% inhibition concentration (IC50) [43].
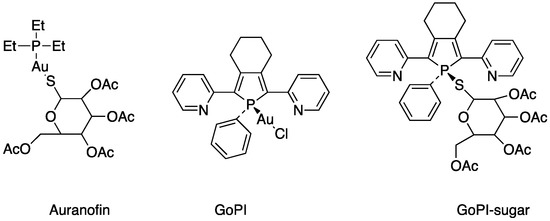
Figure 7.
Structures of gold(I) complexes screening against various parasites.

Table 2.
Biological activity (worm killing activity; motility inhibition; in vitro activity) of a gold complex, GoPI-sugar, in different species [43].
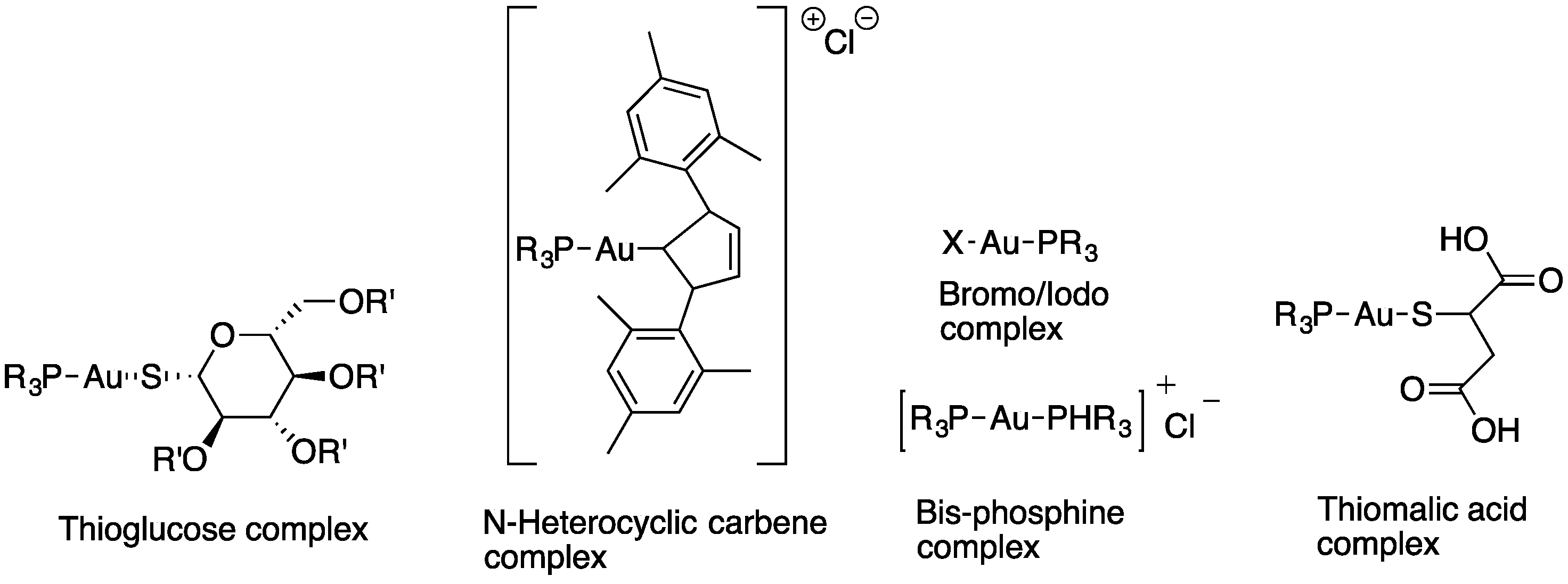
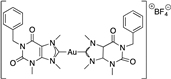
Miyamoto et al. synthesized several gold(I) phosphine complexes: nonacetylated and paracetylated thioglucose-Au-PR3 complexes; thiomalic acid-Au-PR3 complexes; NHC-Au-PR3 complexes; PR3-Au-Cl complexes; [PR3-Au-PR3]Cl; and PR3-Au(I)complexes (Figure 8) to study their activity against Trichomonas vaginalis. A systematic structure–activity relationship demonstrates that the diversification of gold(I) complexes, particularly as halides with simple trialkyl phosphines of the C1−C3 chain length or as bis(trialkyl phosphine) complexes, can markedly improve potency against T. vaginalis and selectivity. All gold(I) complexes effectively inhibited the two most abundant isoforms of the presumed target enzyme, TrxR, but a subset of compounds were markedly more active against live T. vaginalis than TrxR, suggesting that alternative targets exist. All of the tested gold(I) complexes inhibited TrxR in T. vaginalis, with the most potent compounds exhibiting IC50 values of <50 nM, little difference in the inhibition of two of the most abundantly expressed TrxR isoforms, and good correlations between the inhibition of individual isoforms and the inhibition of total cellular TrxR activity. Two of the compounds with increased activity against whole cells compared to TrxR were gold(I)-bis-phosphines [45].
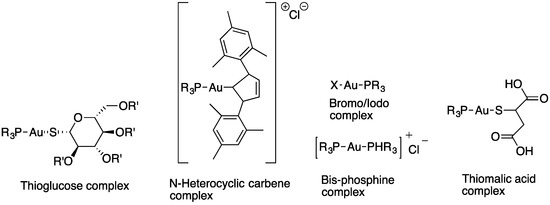
Figure 8.
Gold(I) phosphine complexes.
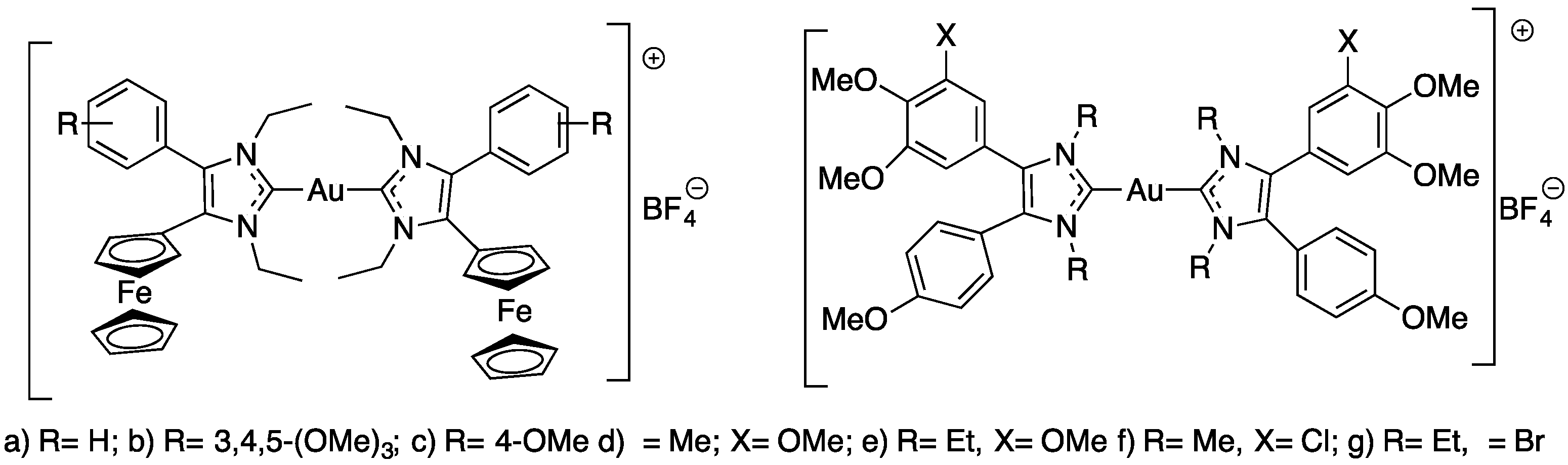
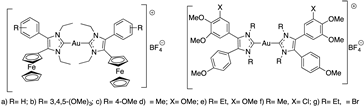
Koko et al. synthesized a series of cationic gold(I) carbenes with various 4,5-diarylimidazolylidene as ligands (Figure 9) to test their activity against Leishmania major, Toxoplasma gondii and Tripanossoma brucei parasites [44].
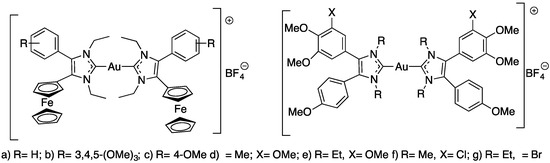
Figure 9.
Structures of the N-heterocyclic carbene-gold(I) complexes.
The results of this work are presented in Table 3. Ferrocene compound a–c (Figure 9) showed the highest activities against L. major amastigotes and T. gondii and distinct selectivity for T. gondii cells when compared with the activity against nonmalignant Vero cells. The ferrocene derivatives (a–c) (Figure 9) are generally more active against the L. major amastigotes and the T. gondii tachyzoites than the other tested anisyl gold complexes and the approved drugs atovaquone and amphotericin B. Compound f (Figure 9) showed the highest selectivity for L. major promastigotes; thus, it was the most active compound against L. major promastigotes of this series of compounds. The 3,4,5-trimethoxyphenyl analog 1b also exhibited a much greater selectivity for T. b. brucei cells when compared with its activity against human HeLa cells [44].

Table 3.
Biological activity of cationic gold(I) carbene against Leishmania major, Toxoplasma gondii and Tripanossoma brucei parasites [44].
Minori, K. et al. synthesized a series of gold(I) and gold(III) complexes in order to test them against Leishmania amazonensis and Leishmania braziliensis. One of the cationic Au(I) bis-N-heterocyclic carbenes [{c), Figure 10] has low EC50 values (ca. 4 μM) in promastigotes cells and no toxicity in host macrophages. Together with two other Au(III) complexes [(a) and (b), Figure 10], the compound is also extremely effective in intracellular amastigotes from L. amazonensis. Initial mechanistic studies include an evaluation of the gold complexes’ effect on L. amazonensis’ plasma membrane integrity [47].
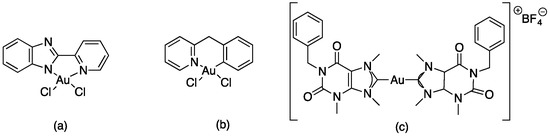
Figure 10.
Structure of Au(I) and Au(III) complexes tested in biological activity against Leishaminia sp; (a) Au(III) complex; (b)Au(III) complex; (c) Cationic Au(I) bis-N-heterocyclic carbenes.
4. Conclusions
From research works presented here, most of the compounds used are Au complexes, as opposed to nanoparticles of the same metal (see Table 1).
Gold nanoparticles have been used in many applications including drug delivery, cancer diagnostics and therapy, medical imaging and non-optical biosensor [56]. The nanoparticles used in the studies presented are similar to but also different from each other; most of them have sulfur as a binding element to gold and then have aliphatic hydrocarbon chains or cyclic monomers. Gold complexes have N-heterocylic ligands, carbenes or phosphines, which are the most common Au complexes. Auranofin, the gold drug already used against rheumatoid arthritis, was also tested against viruses and parasites.
The mechanism of action of these compounds is almost entirely unknown, since the number of compounds used for these studies is small, not allowing the establishment of a structure–activity relationship.
Finally, this review demonstrates the diversity of structures and application of gold compounds in viral and parasites diseases. Additionally, in order for the use of gold compounds in the treatment of viruses or parasitosis to be considered, further studies are needed, since different mechanisms of action might be involved. Putting it all together, taking into consideration its role in biomedical sciences, the future of gold compounds against virus and parasites could be bright.
Author Contributions
Conceptualization, C.F. and M.A.; formal analysis C.F.; funding acquisition, M.A.; investigation, C.F. and M.A.; methodology, C.F. and M.A.; supervision, M.A.; writing—original draft, C.F.; writing—review and editing, C.F. and M.A. All authors have read and agreed to the published version of the manuscript.
Funding
This study received Portuguese national funds from FCT—Foundation for Science and Technology through the projects UIDB/04326/2020 and LA/P/0101/2020(M.A. and C.F.).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| HCV | hepatitis C virus |
| HSC | (human) hepatic stellate cells |
| MUST | mercapto-undecane sulfonic acid |
| OT | octane thiol |
| CD | β-cyclodextrins |
| ACE2 | Angiotensin converting enzyme 2 |
| S | spike |
| PLpro | papain-like protease |
| ROS | reactive oxygen species |
| AuNPs | gold nanoparticles |
| EC50 | Effective concentration that inhibits 50% of the viral/cell activity |
| DENV | Dengue virus |
| DHA | dihydroartemisinin |
| S.I. | selectivity index |
References
- Higby, G.J. Gold in Medicine: A Review of Its Use in the West before 1900. Gold Bull. 1982, 15, 130–140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mirzadeh, N.; Privér, S.H.; Blake, A.J.; Schmidbaur, H.; Bhargava, S.K. Innovative Molecular Design Strategies in Materials Science Following the Aurophilicity Concept. Chem. Rev. 2020, 120, 7551–7591. [Google Scholar] [CrossRef] [PubMed]
- Schmidbaur, H.; Schier, A. Aurophilic Interactions as a Subject of Current Research: An up-Date. Chem. Soc. Rev. 2012, 41, 370–412. [Google Scholar] [CrossRef] [PubMed]
- Stratakis, M.; Garcia, H. Catalysis by Supported Gold Nanoparticles: Beyond Aerobic Oxidative Processes. Chem. Rev. 2012, 112, 4469–4506. [Google Scholar] [CrossRef]
- Yeh, Y.-C.; Creran, B.; Rotello, V.M. Gold Nanoparticles: Preparation, Properties, and Applications in Bionanotechnology. Nanoscale 2012, 4, 1871–1880. [Google Scholar] [CrossRef]
- Chakraborty, I.; Pradeep, T. Atomically Precise Clusters of Noble Metals: Emerging Link between Atoms and Nanoparticles. Chem. Rev. 2017, 117, 8208–8271. [Google Scholar] [CrossRef]
- Hashmi, A.S.K. Dual Gold Catalysis. Acc. Chem. Res. 2014, 47, 864–876. [Google Scholar] [CrossRef]
- Hashmi, A.S.K.; Hutchings, G.J. Gold Catalysis. Angew. Chem. Int. Ed. 2006, 45, 7896–7936. [Google Scholar] [CrossRef]
- Li, Z.; Brouwer, C.; He, C. Gold-Catalyzed Organic Transformations. Chem. Rev. 2008, 108, 3239–3265. [Google Scholar] [CrossRef]
- Arcadi, A. Alternative Synthetic Methods through New Developments in Catalysis by Gold. Chem. Rev. 2008, 108, 3266–3325. [Google Scholar] [CrossRef]
- Corma, A.; Leyva-Pérez, A.; Sabater, M.J. Gold-Catalyzed Carbon−Heteroatom Bond-Forming Reactions. Chem. Rev. 2011, 111, 1657–1712. [Google Scholar] [CrossRef] [PubMed]
- Gorin, D.J.; Toste, F.D. Relativistic Effects in Homogeneous Gold Catalysis. Nature 2007, 446, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Gorin, D.J.; Sherry, B.D.; Toste, F.D. Ligand Effects in Homogeneous Au Catalysis. Chem. Rev. 2008, 108, 3351–3378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Obradors, C.; Echavarren, A.M. Gold-Catalyzed Rearrangements and Beyond. Acc. Chem. Res. 2014, 47, 902–912. [Google Scholar] [CrossRef]
- Gaillard, S.; Cazin, C.S.J.; Nolan, S.P. N-Heterocyclic Carbene Gold(I) and Copper(I) Complexes in C–H Bond Activation. Acc. Chem. Res. 2012, 45, 778–787. [Google Scholar] [CrossRef]
- Ott, I. On the Medicinal Chemistry of Gold Complexes as Anticancer Drugs. Coord. Chem. Rev. 2009, 253, 1670–1681. [Google Scholar] [CrossRef]
- Zou, T.; Lum, C.T.; Lok, C.-N.; Zhang, J.-J.; Che, C.-M. Chemical Biology of Anticancer Gold(iii) and Gold(i) Complexes. Chem. Soc. Rev. 2015, 44, 8786–8801. [Google Scholar] [CrossRef]
- Mora, M.; Gimeno, M.C.; Visbal, R. Recent Advances in Gold–NHC Complexes with Biological Properties. Chem. Soc. Rev. 2019, 48, 447–462. [Google Scholar] [CrossRef]
- Herrera, R.P.; Gimeno, M.C. Main Avenues in Gold Coordination Chemistry. Chem. Rev. 2021, 121, 8311–8363. [Google Scholar] [CrossRef]
- Sadler, P.J.; Sue, R.E. The Chemistry of Gold Drugs. Metal-Based Drugs 1994, 1, 107–144. [Google Scholar] [CrossRef] [Green Version]
- Azharuddin, M.; Zhu, G.H.; Das, D.; Ozgur, E.; Uzun, L.; Turner, A.P.F.; Patra, H.K. A Repertoire of Biomedical Applications of Noble Metal Nanoparticles. Chem. Commun. 2019, 55, 6964–6996. [Google Scholar] [CrossRef] [PubMed]
- Pettenuzzo, N.; Brustolin, L.; Coltri, E.; Gambalunga, A.; Chiara, F.; Trevisan, A.; Biondi, B.; Nardon, C.; Fregona, D. CuII and AuIII Complexes with Glycoconjugated Dithiocarbamato Ligands for Potential Applications in Targeted Chemotherapy. ChemMedChem 2019, 14, 1162–1172. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Gust, R. Metal N-Heterocyclic Carbene Complexes as Potential Antitumor Metallodrugs. Chem. Soc. Rev. 2013, 42, 755–773. [Google Scholar] [CrossRef] [PubMed]
- Stratton, M.; Ramachandran, A.; Camacho, E.J.M.; Patil, S.; Waris, G.; Grice, K.A. Anti-Fibrotic Activity of Gold and Platinum Complexes—Au(I) Compounds as a New Class of Anti-Fibrotic Agents. J. Inorg. Biochem. 2020, 206, 111023. [Google Scholar] [CrossRef]
- Azria, D.; Blanquer, S.; Verdier, J.-M.; Belamie, E. Nanoparticles as Contrast Agents for Brain Nuclear Magnetic Resonance Imaging in Alzheimer’s Disease Diagnosis. J. Mater. Chem. B 2017, 5, 7216–7237. [Google Scholar] [CrossRef]
- Bondžić, A.M.; Janjić, G.V.; Dramićanin, M.D.; Messori, L.; Massai, L.; Parac Vogt, T.N.; Vasić, V.M. Na/K-ATPase as a Target for Anticancer Metal Based Drugs: Insights into Molecular Interactions with Selected Gold(III) Complexes. Metallomics 2017, 9, 292–300. [Google Scholar] [CrossRef]
- Fonseca, C.; Fraqueza, G.; Carabineiro, S.A.C.; Aureliano, M. The Ca2+-ATPase Inhibition Potential of Gold(I, III) Compounds. Inorganics 2020, 8, 49. [Google Scholar] [CrossRef]
- Aikman, B.; Wenzel, M.; Mósca, A.; de Almeida, A.; Klooster, W.; Coles, S.; Soveral, G.; Casini, A. Gold(III) Pyridine-Benzimidazole Complexes as Aquaglyceroporin Inhibitors and Antiproliferative Agents. Inorganics 2018, 6, 123. [Google Scholar] [CrossRef] [Green Version]
- Berrocal, M.; Cordoba-Granados, J.J.; Carabineiro, S.A.C.; Gutierrez-Merino, C.; Aureliano, M.; Mata, A.M. Gold Compounds Inhibit the Ca2+-ATPase Activity of Brain PMCA and Human Neuroblastoma SH-SY5Y Cells and Decrease Cell Viability. Metals 2021, 11, 1934. [Google Scholar] [CrossRef]
- Forestier, J. Rheumatoid arthritis and its treatment by gold salts: The results of six years’ experience. J. Lab. Clin. Med. 1935, 20, 827–840. [Google Scholar]
- Abdalbari, F.H.; Telleria, C.M. The Gold Complex Auranofin: New Perspectives for Cancer Therapy. Discov. Oncol. 2021, 12, 42. [Google Scholar] [CrossRef] [PubMed]
- Gamberi, T.; Chiappetta, G.; Fiaschi, T.; Modesti, A.; Sorbi, F.; Magherini, F. Upgrade of an Old Drug: Auranofin in Innovative Cancer Therapies to Overcome Drug Resistance and to Increase Drug Effectiveness. Med. Res. Rev. 2021, 42, 1111–1146. [Google Scholar] [CrossRef] [PubMed]
- Vollenbroek, F.A.; Van den Berg, J.P.; Van der Velden, J.W.A.; Bour, J.J. Phosphorus-31 [Proton] Nuclear Magnetic Resonance Investigation of Gold Cluster Compounds. Inorg. Chem. 1980, 19, 2685–2688. [Google Scholar] [CrossRef]
- Assefa, Z.; McBurnett, B.G.; Staples, R.J.; Fackler, J.P.; Assmann, B.; Angermaier, K.; Schmidbaur, H. Syntheses, Structures, and Spectroscopic Properties of Gold(I) Complexes of 1,3,5-Triaza-7-Phosphaadamantane (TPA). Correlation of the Supramolecular Au.Cntdot. .Cntdot. .Cntdot.Au Interaction and Photoluminescence for the Species (TPA)AuCl and [(TPA-HCl)AuCl]. Inorg. Chem. 1995, 34, 75–83. [Google Scholar]
- Cagno, V.; Gasbarri, M.; Medaglia, C.; Gomes, D.; Clement, S.; Stellacci, F.; Tapparel, C. Sulfonated Nanomaterials with Broad-Spectrum Antiviral Activity Extending beyond Heparan Sulfate-Dependent Viruses. Antimicrob. Agents Chemother. 2020, 64, e02001-20. [Google Scholar] [CrossRef]
- Ershov, A.Y.; Martynenkov, A.A.; Lagoda, I.V.; Kopanitsa, M.A.; Zarubaev, V.V.; Slita, A.V.; Buchkov, E.V.; Panarin, E.F.; Yakimansky, A.V. Gold Glyconanoparticles Based on Aldoses 6-Mercaptohexanoyl Hydrazones and Their Anti-Influenza Activity. Russ. J. Gen. Chem. 2021, 91, 1735–1739. [Google Scholar] [CrossRef]
- Ershov, A.Y.; Martynenkov, A.A.; Lagoda, I.V.; Yakimansky, A.V. Synthesis of 6-Mercaptohexanoylhydrazones of Mono- and Disaccharides as a Potential Glycoligands of Noble Metal Glyconanoparticles. Russ. J. Gen. Chem. 2020, 90, 1863–1868. [Google Scholar] [CrossRef]
- Rothan, H.A.; Stone, S.; Natekar, J.; Kumari, P.; Arora, K.; Kumar, M. The FDA-Approved Gold Drug Auranofin Inhibits Novel Coronavirus (SARS-COV-2) Replication and Attenuates Inflammation in Human Cells. Virology 2020, 547, 7–11. [Google Scholar] [CrossRef]
- Zacheo, A.; Hodek, J.; Witt, D.; Mangiatordi, G.F.; Ong, Q.K.; Kocabiyik, O.; Depalo, N.; Fanizza, E.; Laquintana, V.; Denora, N.; et al. Multi-Sulfonated Ligands on Gold Nanoparticles as Virucidal Antiviral for Dengue Virus. Sci. Rep. 2020, 10, 9052. [Google Scholar] [CrossRef]
- Gil-Moles, M.; Türck, S.; Basu, U.; Pettenuzzo, A.; Bhattacharya, S.; Rajan, A.; Ma, X.; Büssing, R.; Wölker, J.; Burmeister, H.; et al. Metallodrug Profiling against SARS-CoV-2 Target Proteins Identifies Highly Potent Inhibitors of the S/ACE2 Interaction and the Papain-like Protease PLpro. Chem. A Eur. J. 2021, 27, 17928–17940. [Google Scholar] [CrossRef]
- Ouji, M.; Bourgeade-Delmas, S.; Álvarez, F.; Augereau, J.; Valentin, A.; Hemmert, C.; Gornitzka, H.; Benoit-Vical, F. Design, Synthesis and Efficacy of Hybrid Triclosan-Gold Based Molecules on Artemisinin-Resistant Plasmodium Falciparum and Leishmania Infantum Parasites. ChemistrySelect 2020, 5, 619–625. [Google Scholar] [CrossRef]
- Ouji, M.; Barnoin, G.; Fernández Álvarez, Á.; Augereau, J.-M.; Hemmert, C.; Benoit-Vical, F.; Gornitzka, H. Hybrid Gold(I) NHC-Artemether Complexes to Target Falciparum Malaria Parasites. Molecules 2020, 25, 2817. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Pomel, S.; Latre de Late, P.; Taravaud, A.; Loiseau, P.M.; Maes, L.; Cho-Ngwa, F.; Bulman, C.A.; Fischer, C.; Sakanari, J.A.; et al. Repurposing Auranofin and Evaluation of a New Gold(I) Compound for the Search of Treatment of Human and Cattle Parasitic Diseases: From Protozoa to Helminth Infections. Molecules 2020, 25, 5075. [Google Scholar] [CrossRef]
- Koko, W.S.; Jentzsch, J.; Kalie, H.; Schobert, R.; Ersfeld, K.; Al Nasr, I.S.; Khan, T.A.; Biersack, B. Evaluation of the Antiparasitic Activities of Imidazol-2-ylidene–Gold(I) Complexes. Arch. Pharm. 2020, 353, e1900363. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, Y.; Aggarwal, S.; Celaje, J.J.A.; Ihara, S.; Ang, J.; Eremin, D.B.; Land, K.M.; Wrischnik, L.A.; Zhang, L.; Fokin, V.V.; et al. Gold(I) Phosphine Derivatives with Improved Selectivity as Topically Active Drug Leads to Overcome 5-Nitroheterocyclic Drug Resistance in Trichomonas vaginalis. J. Med. Chem. 2021, 64, 6608–6620. [Google Scholar] [CrossRef]
- Tu, X.; Tan, X.; Qi, X.; Huang, A.; Ling, F.; Wang, G. Proteome Interrogation Using Gold Nanoprobes to Identify Targets of Arctigenin in Fish Parasites. J. Nanobiotechnol. 2020, 18, 32. [Google Scholar] [CrossRef] [Green Version]
- Minori, K.; Rosa, L.B.; Bonsignore, R.; Casini, A.; Miguel, D.C. Comparing the Antileishmanial Activity of Gold(I) and Gold(III) Compounds in L. amazonensis and L. braziliensis In Vitro. ChemMedChem 2020, 15, 2146–2150. [Google Scholar] [CrossRef]
- Cagno, V.; Andreozzi, M. Broad-Spectrum Non-Toxic Antiviral Nanoparticles with a Virucidal Inhibition Mechanism. Nat. Mater. 2018, 17, 195–203. [Google Scholar] [CrossRef]
- Cagno, V.; Tseligka, E.D.; Jones, S.T.; Tapparel, C. Heparan Sulfate Proteoglycans and Viral Attachment: True Receptors or Adaptation Bias? Viruses 2019, 11, 596. [Google Scholar] [CrossRef] [Green Version]
- Jones, S.T.; Cagno, V.; Janeček, M.; Ortiz, D.; Gasilova, N.; Piret, J.; Gasbarri, M.; Constant, D.A.; Han, Y.; Vuković, L.; et al. Modified Cyclodextrins as Broad-Spectrum Antivirals. Sci. Adv. 2020, 6, eaax9318. [Google Scholar] [CrossRef] [Green Version]
- Guven, Z.P.; Silva, P.H.J.; Luo, Z.; Cendrowska, U.B.; Gasbarri, M.; Jones, S.T.; Stellacci, F. Synthesis and Characterization of Amphiphilic Gold Nanoparticles. J. Vis. Exp. 2019, 149, 58872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paloque, L.; Hemmert, C.; Valentin, A.; Gornitzka, H. Synthesis, Characterization, and Antileishmanial Activities of Gold(I) Complexes Involving Quinoline Functionalized N-Heterocyclic Carbenes. Eur. J. Med. Chem. 2015, 94, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Bourgeade Delmas, S.; Fernández Álvarez, Á.; Valentin, A.; Hemmert, C.; Gornitzka, H. Synthesis, Characterization, and Antileishmanial Activity of Neutral N-Heterocyclic Carbenes Gold(I) Complexes. Eur. J. Med. Chem. 2018, 143, 1635–1643. [Google Scholar] [CrossRef] [PubMed]
- Straimer, J.; Gnädig, N.F.; Stokes, B.H.; Ehrenberger, M.; Crane, A.A.; Fidock, D.A. Plasmodium Falciparum K13 Mutations Differentially Impact Ozonide Susceptibility and Parasite Fitness In Vitro. mBio 2017, 8, e00172-17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jortzik, E.; Farhadi, M.; Ahmadi, R.; Tóth, K.; Lohr, J.; Helmke, B.M.; Kehr, S.; Unterberg, A.; Ott, I.; Gust, R.; et al. Antiglioma Activity of GoPI-Sugar, a Novel Gold(I)–Phosphole Inhibitor: Chemical Synthesis, Mechanistic Studies, and Effectiveness In Vivo. Biochim. Biophys. Acta (BBA)-Proteins Proteom. 2014, 1844, 1415–1426. [Google Scholar] [CrossRef] [PubMed]
- Ielo, I.; Rando, G.; Giacobello, F.; Sfameni, S.; Castellano, A.; Galletta, M.; Drommi, D.; Rosace, G.; Plutino, M.R. Synthesis, Chemical–Physical Characterization, and Biomedical Applications of Functional Gold Nanoparticles: A Review. Molecules 2021, 26, 5823. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).