Future Developments in the Treatment of AL Amyloidosis
Abstract
1. Introduction
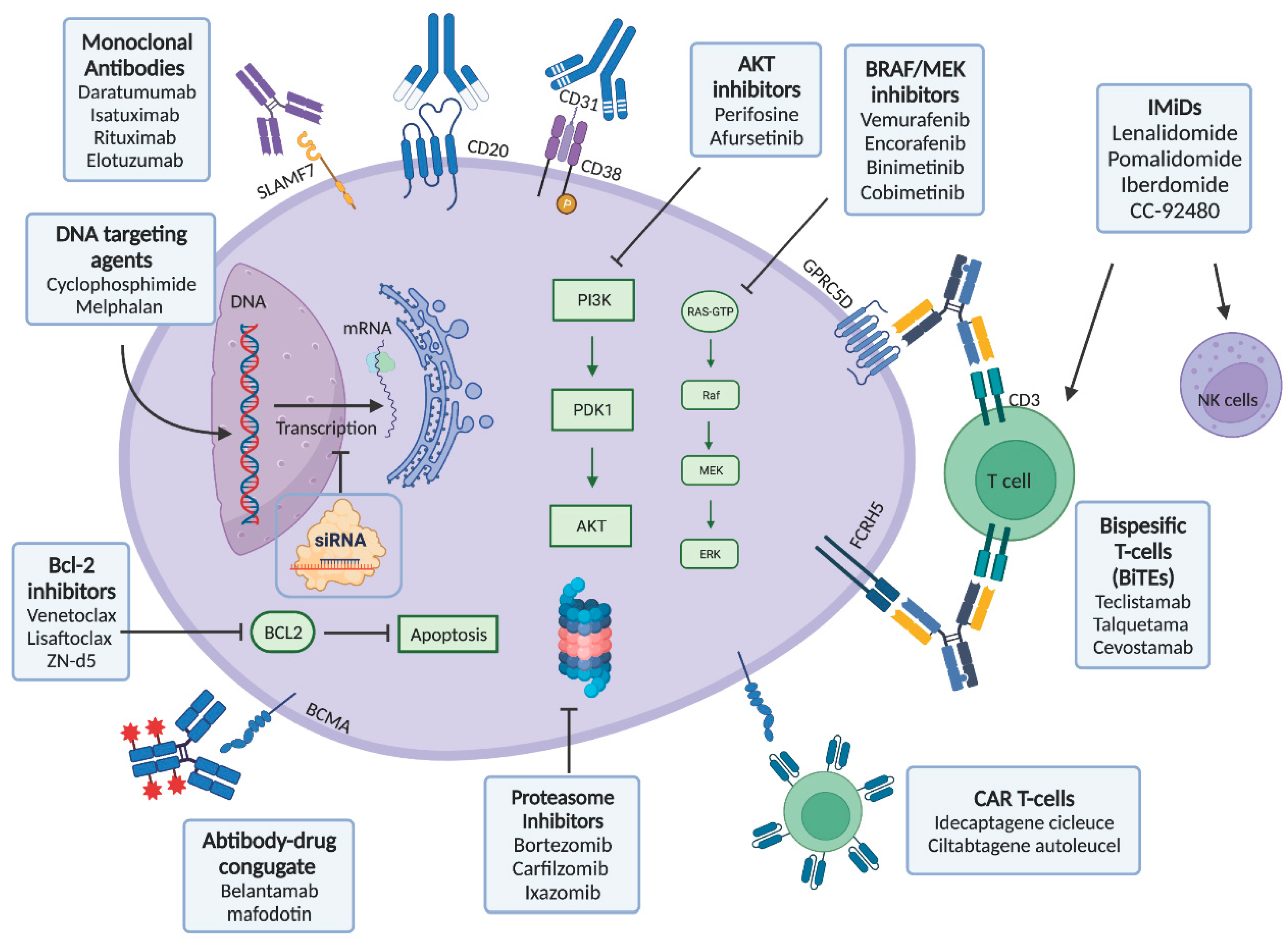
2. Therapies Targeting the Plasma Cell Clone
2.1. Current Treatment Algorithm
2.2. IgM-Amyloidosis
2.3. Clonal Characteristics of Plasma Cells and BCL2 Inhibition
2.4. Antibody Drug Conjugates
Belantamab Mafodotin
2.5. Bispecifics Active in MM also Relevant in AL Amyloidosis
2.6. Chimeric Antigen Receptor (CAR) T-Cells
2.7. Pathway Directed Therapy
2.7.1. BRAF Inhibitors and MEK Inhibitors
2.7.2. PI3K/AKT Pathway Directed Therapies
2.7.3. Transcription Factor Directed Therapies
2.8. New Immunomodulatory Agents (IMiDs)
2.9. TLX66
3. Special Considerations; Toxicity Associated with Anti-Clonal Therapies
4. Treatment Strategies Targeting Immunoglobulin mRNA
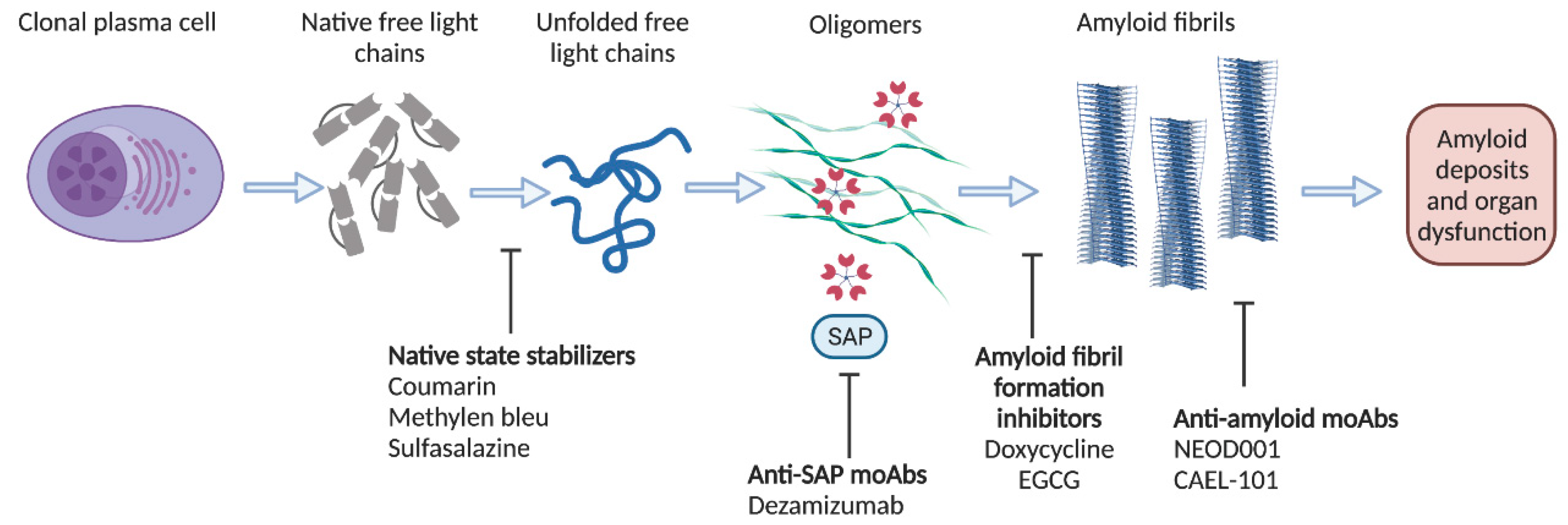
5. Anti-Amyloid Targeting Treatment Options
5.1. Targeting Amyloid Formation and Dissolution
5.2. Targeting Amyloid Deposits
6. Future Combinations and Treatment Algorithms
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Merlini, G.; Bellotti, V. Molecular mechanisms of amyloidosis. N. Engl. J. Med. 2003, 349, 583–596. [Google Scholar] [CrossRef]
- Muchtar, E.; Gertz, M.A.; Kumar, S.K.; Lacy, M.Q.; Dingli, D.; Buadi, F.K.; Grogan, M.; Hayman, S.R.; Kapoor, P.; Leung, N.; et al. Improved outcomes for newly diagnosed AL amyloidosis between 2000 and 2014: Cracking the glass ceiling of early death. Blood 2017, 129, 2111–2119. [Google Scholar] [CrossRef]
- Palladini, G.; Sachchithanantham, S.; Milani, P.; Gillmore, J.; Foli, A.; Lachmann, H.; Basset, M.; Hawkins, P.; Merlini, G.; Wechalekar, A.D. A European collaborative study of cyclophosphamide, bortezomib, and dexamethasone in upfront treatment of systemic AL amyloidosis. Blood 2015, 126, 612–615. [Google Scholar] [CrossRef]
- Kastritis, E.; Palladini, G.; Minnema, M.C.; Wechalekar, A.D.; Jaccard, A.; Lee, H.C.; Sanchorawala, V.; Gibbs, S.; Mollee, P.; Venner, C.P.; et al. Daratumumab-Based Treatment for Immunoglobulin Light-Chain Amyloidosis. N. Engl. J. Med. 2021, 385, 46–58. [Google Scholar] [CrossRef]
- Kastritis, E.; Sanchorawala, V.; Merlini, G.; the ANDROMEDA Study Group. Subcutaneous daratumumab + bortezomib, cyclophosphamide, and dexamethasone (VCd) in patients with newly diagnosed light chain (AL) amyloidosis: Updated results from the phase 3 ANDROMEDA study. J. Clin. Oncol. 2021, 39, 8003. [Google Scholar] [CrossRef]
- Kastritis, E.; Leleu, X.; Arnulf, B.; Zamagni, E.; Cibeira, M.T.; Kwok, F.; Mollee, P.; Hajek, R.; Moreau, P.; Jaccard, A.; et al. Bortezomib, Melphalan, and Dexamethasone for Light-Chain Amyloidosis. J. Clin. Oncol 2020, 38, 3252–3260. [Google Scholar] [CrossRef]
- Reece, D.E.; Hegenbart, U.; Sanchorawala, V.; Merlini, G.; Palladini, G.; Bladé, J.; Fermand, J.P.; Hassoun, H.; Heffner, L.; Vescio, R.A.; et al. Efficacy and safety of once-weekly and twice-weekly bortezomib in patients with relapsed systemic AL amyloidosis: Results of a phase 1/2 study. Blood 2011, 118, 865–873. [Google Scholar] [CrossRef]
- Palladini, G.; Milani, P.; Foli, A.; Obici, L.; Lavatelli, F.; Nuvolone, M.; Caccialanza, R.; Perlini, S.; Merlini, G. Oral melphalan and dexamethasone grants extended survival with minimal toxicity in AL amyloidosis: Long-term results of a risk-adapted approach. Haematologica 2014, 99, 743–750. [Google Scholar] [CrossRef]
- Palladini, G.; Merlini, G. When should treatment of AL amyloidosis start at relapse? Early, to prevent organ progression. Blood Adv. 2019, 3, 212–215. [Google Scholar] [CrossRef]
- Sanchorawala, V. Delay treatment of AL amyloidosis at relapse until symptomatic: Devil is in the details. Blood Adv. 2019, 3, 216–218. [Google Scholar] [CrossRef]
- Abeykoon, J.P.; Zanwar, S.; Dispenzieri, A.; Gertz, M.A.; Leung, N.; Kourelis, T.; Gonsalves, W.; Muchtar, E.; Dingli, D.; Lacy, M.Q.; et al. Daratumumab-based therapy in patients with heavily-pretreated AL amyloidosis. Leukemia 2019, 33, 531–536. [Google Scholar] [CrossRef]
- Sanchorawala, V.; Sarosiek, S.; Schulman, A.; Mistark, M.; Migre, M.E.; Cruz, R.; Sloan, J.M.; Brauneis, D.; Shelton, A.C. Safety, Tolerability, and Response Rates of Daratumumab in Relapsed AL Amyloidosis: Results of a Phase II Study. Blood 2020, 135, 1541–1547. [Google Scholar] [CrossRef]
- Warsame, R.; LaPlant, B.; Kumar, S.K.; Laumann, K.; Perez Burbano, G.; Buadi, F.K.; Gertz, M.A.; Kyle, R.A.; Lacy, M.Q.; Dingli, D.; et al. Long-term outcomes of IMiD-based trials in patients with immunoglobulin light-chain amyloidosis: A pooled analysis. Blood Cancer J. 2020, 10, 4. [Google Scholar] [CrossRef]
- Basset, M.; Kimmich, C.R.; Schreck, N.; Krzykalla, J.; Dittrich, T.; Veelken, K.; Goldschmidt, H.; Seckinger, A.; Hose, D.; Jauch, A.; et al. Lenalidomide and dexamethasone in relapsed/refractory immunoglobulin light chain (AL) amyloidosis: Results from a large cohort of patients with long follow-up. Br. J. Haematol. 2021, 195, 230–243. [Google Scholar] [CrossRef]
- Kastritis, E.; Gavriatopoulou, M.; Roussou, M.; Bagratuni, T.; Migkou, M.; Fotiou, D.; Ziogas, D.C.; Kanellias, N.; Eleutherakis-Papaiakovou, E.; Dialoupi, I.; et al. Efficacy of lenalidomide as salvage therapy for patients with AL amyloidosis. Amyloid 2018, 25, 234–241. [Google Scholar] [CrossRef]
- Mahmood, S.; Venner, C.P.; Sachchithanantham, S.; Lane, T.; Rannigan, L.; Foard, D.; Pinney, J.H.; Gibbs, S.D.; Whelan, C.J.; Lachmann, H.J.; et al. Lenalidomide and dexamethasone for systemic AL amyloidosis following prior treatment with thalidomide or bortezomib regimens. Br. J. Haematol. 2014, 166, 842–848. [Google Scholar] [CrossRef]
- Palladini, G.; Russo, P.; Foli, A.; Milani, P.; Lavatelli, F.; Obici, L.; Nuvolone, M.; Brugnatelli, S.; Invernizzi, R.; Merlini, G. Salvage therapy with lenalidomide and dexamethasone in patients with advanced AL amyloidosis refractory to melphalan, bortezomib, and thalidomide. Ann. Hematol. 2012, 91, 89–92. [Google Scholar] [CrossRef]
- Sanchorawala, V.; Wright, D.G.; Rosenzweig, M.; Finn, K.T.; Fennessey, S.; Zeldis, J.B.; Skinner, M.; Seldin, D.C. Lenalidomide and dexamethasone in the treatment of AL amyloidosis: Results of a phase 2 trial. Blood 2007, 109, 492–496. [Google Scholar] [CrossRef]
- Dispenzieri, A.; Buadi, F.; Laumann, K.; LaPlant, B.; Hayman, S.R.; Kumar, S.K.; Dingli, D.; Zeldenrust, S.R.; Mikhael, J.R.; Hall, R.; et al. Activity of pomalidomide in patients with immunoglobulin light-chain amyloidosis. Blood 2012, 119, 5397–5404. [Google Scholar] [CrossRef]
- Palladini, G.; Milani, P.; Foli, A.; Basset, M.; Russo, F.; Perlini, S.; Merlini, G. A phase 2 trial of pomalidomide and dexamethasone rescue treatment in patients with AL amyloidosis. Blood 2017, 129, 2120–2123. [Google Scholar] [CrossRef]
- Sanchorawala, V.; Shelton, A.C.; Lo, S.; Varga, C.; Sloan, J.M.; Seldin, D.C. Pomalidomide and dexamethasone in the treatment of AL amyloidosis: Results of a phase 1 and 2 trial. Blood 2016, 128, 1059–1062. [Google Scholar] [CrossRef] [PubMed]
- Kastritis, E.; Dispenzieri, A.; Wechalekar, A.D.; Schönland, S.O.; Kim, K.; Sanchorawala, V.; Landau, H.J.; Kwok, F.; Suzuki, K.; Comenzo, R.; et al. Ixazomib-dexamethasone (Ixa-Dex) vs physician’s choice (PC) in relapsed/refractory (RR) primary systemic AL amyloidosis (AL) patients (pts) by prior proteasome inhibitor (PI) exposure in the phase III TOURMALINE-AL1 trial. J. Clin. Oncol. 2020, 38 (Suppl. S15), 8546. [Google Scholar] [CrossRef]
- Cohen, O.C.; Sharpley, F.; Gillmore, J.D.; Lachmann, H.J.; Sachchithanantham, S.; Mahmood, S.; Fontana, M.; Whelan, C.J.; Martinez-Naharro, A.; Kyriakou, C.; et al. Use of ixazomib, lenalidomide and dexamethasone in patients with relapsed amyloid light-chain amyloidosis. Br. J. Haematol. 2020, 189, 643–649. [Google Scholar] [CrossRef]
- Sachchithanantham, S.; Roussel, M.; Palladini, G.; Klersy, C.; Mahmood, S.; Venner, C.P.; Gibbs, S.; Gillmore, J.; Lachmann, H.; Hawkins, P.N. European collaborative study defining clinical profile outcomes and novel prognostic criteria in monoclonal immunoglobulin M-related light chain amyloidosis. J. Clin. Oncol. 2016, 34, 2037–2045. [Google Scholar] [CrossRef]
- Sidiqi, M.H.; Buadi, F.K.; Dispenzieri, A.; Warsame, R.; Lacy, M.Q.; Dingli, D.; Leung, N.; Gonsalves, W.I.; Kapoor, P.; Kourelis, T.V.; et al. Autologous Stem Cell Transplant for IgM-Associated Amyloid Light-Chain Amyloidosis. Biol. Blood Marrow Transplant. 2019, 25, e108–e111. [Google Scholar] [CrossRef]
- Chakraborty, R.; Novak, A.J.; Ansell, S.M.; Muchtar, E.; Kapoor, P.; Hayman, S.R.; Dispenzieri, A.; Buadi, F.K.; Lacy, M.Q.; King, R.L. First report of MYD88 L265P somatic mutation in IgM-associated light-chain amyloidosis. Blood J. Am. Soc. Hematol. 2016, 127, 2936–2938. [Google Scholar]
- Pika, T.; Hegenbart, U.; Flodrova, P.; Maier, B.; Kimmich, C.; Schönland, S.O. First report of ibrutinib in IgM-related amyloidosis: Few responses, poor tolerability, and short survival. Blood J. Am. Soc. Hematol. 2018, 131, 368–371. [Google Scholar] [CrossRef]
- Ozga, M.; Zhao, Q.; Benson, D., Jr.; Elder, P.; Williams, N.; Bumma, N.; Rosko, A.; Chaudhry, M.; Khan, A.; Devarakonda, S.; et al. AL amyloidosis: The effect of fluorescent in situ hybridization abnormalities on organ involvement and survival. Cancer Med. 2021, 10, 965–973. [Google Scholar] [CrossRef]
- Dumas, B.; Yameen, H.; Sarosiek, S.; Sloan, J.M.; Sanchorawala, V. Presence of t(11;14) in AL amyloidosis as a marker of response when treated with a bortezomib-based regimen. Amyloid 2020, 27, 244–249. [Google Scholar] [CrossRef]
- Bochtler, T.; Hegenbart, U.; Kunz, C.; Benner, A.; Seckinger, A.; Dietrich, S.; Granzow, M.; Neben, K.; Goldschmidt, H.; Ho, A.D.; et al. Gain of chromosome 1q21 is an independent adverse prognostic factor in light chain amyloidosis patients treated with melphalan/dexamethasone. Amyloid 2014, 21, 9–17. [Google Scholar] [CrossRef]
- Bochtler, T.; Hegenbart, U.; Kunz, C.; Granzow, M.; Benner, A.; Seckinger, A.; Kimmich, C.; Goldschmidt, H.; Ho, A.D.; Hose, D.; et al. Translocation t(11;14) is associated with adverse outcome in patients with newly diagnosed AL amyloidosis when treated with bortezomib-based regimens. J. Clin. Oncol. 2015, 33, 1371–1378. [Google Scholar] [CrossRef] [PubMed]
- Leung, N.; Thome, S.D.; Dispenzieri, A. Venetoclax induced a complete response in a patient with immunoglobulin light chain amyloidosis plateaued on cyclophosphamide, bortezomib and dexamethasone. Haematologica 2018, 103, e135–e137. [Google Scholar] [CrossRef] [PubMed]
- Sidiqi, M.H.; Al Saleh, A.S.; Leung, N.; Jevremovic, D.; Aljama, M.A.; Gonsalves, W.I.; Buadi, F.K.; Kourelis, T.V.; Warsame, R.; Muchtar, E.; et al. Venetoclax for the treatment of translocation (11;14) AL amyloidosis. Blood Cancer J. 2020, 10, 55. [Google Scholar] [CrossRef] [PubMed]
- Premkumar, V.J.; Lentzsch, S.; Pan, S.; Bhutani, D.; Richter, J.; Jagannath, S.; Liedtke, M.; Jaccard, A.; Wechalekar, A.D.; Comenzo, R.; et al. Venetoclax induces deep hematologic remissions in t(11;14) relapsed/refractory AL amyloidosis. Blood Cancer J. 2021, 11, 10. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.K.; Harrison, S.J.; Cavo, M.; de la Rubia, J.; Popat, R.; Gasparetto, C.; Hungria, V.; Salwender, H.; Suzuki, K.; Kim, I.; et al. Venetoclax or placebo in combination with bortezomib and dexamethasone in patients with relapsed or refractory multiple myeloma (BELLINI): A randomised, double-blind, multicentre, phase 3 trial. Lancet Oncol. 2020, 21, 1630–1642. [Google Scholar] [CrossRef]
- Bahlis, N.J.; Baz, R.; Harrison, S.J.; Quach, H.; Ho, S.J.; Vangsted, A.J.; Plesner, T.; Moreau, P.; Gibbs, S.D.; Coppola, S.; et al. Phase I Study of Venetoclax Plus Daratumumab and Dexamethasone, With or Without Bortezomib, in Patients With Relapsed or Refractory Multiple Myeloma With and Without t(11;14). J. Clin. Oncol. 2021, 10, 3602–3612. [Google Scholar] [CrossRef]
- Roberts, A.W.; Wei, A.H.; Huang, D.C.S. BCL2 and MCL1 inhibitors for hematologic malignancies. Blood 2021, 138, 1120–1136. [Google Scholar] [CrossRef]
- Ailawadhi, S.; Chanan-Khan, A.A.A.; Chen, Z.; Huang, B.; Konopleva, M.; Brander, D.M.; Rizzieri, D.; Lasica, M.; Tam, C.S.L.; Yannakou, C.K.; et al. First-in-human study of lisaftoclax (APG-2575), a novel BCL-2 inhibitor (BCL-2i), in patients (pts) with relapsed/refractory (R/R) CLL and other hematologic malignancies (HMs). J. Clin. Oncol. 2021, 39 (Suppl. S15), 7502. [Google Scholar] [CrossRef]
- Wuilleme-Toumi, S.; Robillard, N.; Gomez, P.; Moreau, P.; Le Gouill, S.; Avet-Loiseau, H.; Harousseau, J.L.; Amiot, M.; Bataille, R. Mcl-1 is overexpressed in multiple myeloma and associated with relapse and shorter survival. Leukemia 2005, 19, 1248–1252. [Google Scholar] [CrossRef]
- Puthier, D.; Pellat-Deceunynck, C.; Barille, S.; Robillard, N.; Rapp, M.J.; Juge-Morineau, N.; Harousseau, J.L.; Bataille, R.; Amiot, M. Differential expression of Bcl-2 in human plasma cell disorders according to proliferation status and malignancy. Leukemia 1999, 13, 289–294. [Google Scholar] [CrossRef][Green Version]
- Tu, Y.; Renner, S.; Xu, F.; Fleishman, A.; Taylor, J.; Weisz, J.; Vescio, R.; Rettig, M.; Berenson, J.; Krajewski, S.; et al. BCL-X expression in multiple myeloma: Possible indicator of chemoresistance. Cancer Res. 1998, 58, 256–262. [Google Scholar] [PubMed]
- Peperzak, V.; Vikstrom, I.; Walker, J.; Glaser, S.P.; LePage, M.; Coquery, C.M.; Erickson, L.D.; Fairfax, K.; Mackay, F.; Strasser, A.; et al. Mcl-1 is essential for the survival of plasma cells. Nat. Immunol. 2013, 14, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Derenne, S.; Monia, B.; Dean, N.M.; Taylor, J.K.; Rapp, M.J.; Harousseau, J.L.; Bataille, R.; Amiot, M. Antisense strategy shows that Mcl-1 rather than Bcl-2 or Bcl-x(L) is an essential survival protein of human myeloma cells. Blood 2002, 100, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Caenepeel, S.; Brown, S.P.; Belmontes, B.; Moody, G.; Keegan, K.S.; Chui, D.; Whittington, D.A.; Huang, X.; Poppe, L.; Cheng, A.C.; et al. AMG 176, a Selective MCL1 Inhibitor, Is Effective in Hematologic Cancer Models Alone and in Combination with Established Therapies. Cancer Discov. 2018, 8, 1582–1597. [Google Scholar] [CrossRef]
- Tron, A.E.; Belmonte, M.A.; Adam, A.; Aquila, B.M.; Boise, L.H.; Chiarparin, E.; Cidado, J.; Embrey, K.J.; Gangl, E.; Gibbons, F.D.; et al. Discovery of Mcl-1-specific inhibitor AZD5991 and preclinical activity in multiple myeloma and acute myeloid leukemia. Nat. Commun. 2018, 9, 5341. [Google Scholar] [CrossRef]
- Kotschy, A.; Szlavik, Z.; Murray, J.; Davidson, J.; Maragno, A.L.; Le Toumelin-Braizat, G.; Chanrion, M.; Kelly, G.L.; Gong, J.N.; Moujalled, D.M.; et al. The MCL1 inhibitor S63845 is tolerable and effective in diverse cancer models. Nature 2016, 538, 477–482. [Google Scholar] [CrossRef]
- Balachander, S.B.; Criscione, S.W.; Byth, K.F.; Cidado, J.; Adam, A.; Lewis, P.; Macintyre, T.; Wen, S.; Lawson, D.; Burke, K.; et al. AZD4320, A Dual Inhibitor of Bcl-2 and Bcl-xL, Induces Tumor Regression in Hematologic Cancer Models without Dose-limiting Thrombocytopenia. Clin. Cancer Res. 2020, 26, 6535–6549. [Google Scholar] [CrossRef]
- Yu, B.; Liu, D. Antibody-drug conjugates in clinical trials for lymphoid malignancies and multiple myeloma. J. Hematol Oncol 2019, 12, 94. [Google Scholar] [CrossRef]
- Lonial, S.; Lee, H.C.; Badros, A.; Trudel, S.; Nooka, A.K.; Chari, A.; Abdallah, A.O.; Callander, N.; Lendvai, N.; Sborov, D.; et al. Belantamab mafodotin for relapsed or refractory multiple myeloma (DREAMM-2): A two-arm, randomised, open-label, phase 2 study. Lancet Oncol. 2020, 21, 207–221. [Google Scholar] [CrossRef]
- Bal, S.; Sigler, A.; Chan, A.; Chung, D.J.; Dogan, A.; Giralt, S.A.; Hassoun, H.; Landau, H.J. First Description of B Cell Maturation Antigen Expression in Light Chain Amyloidosis; American Society of Hematology: Washington, DC, USA, 2019. [Google Scholar]
- Godara, A.; Zhou, P.; Rosenthal, B.; Kugelmass, A.; Toskic, D.; Fogaren, T.; Varga, C.; Comenzo, R.L. B-Cell Maturation Antigen (BCMA) in Systemic Light-Chain Amyloidosis (AL): Association with Disease Activity and Its Modulation with Gamma-Secretase Inhibition. Blood 2019, 134 (Suppl. S1), 4409. [Google Scholar] [CrossRef]
- Godara, A.; Zhou, P.; Kugelmass, A.; Ma, X.; Rosenthal, B.; Toskic, D.; Fogaren, T.; Varga, C.; Comenzo, R.L. Presence of soluble and cell-surface B-cell maturation antigen in systemic light-chain amyloidosis and its modulation by gamma-secretase inhibition. Am. J. Hematol. 2020, 95, E110–E113. [Google Scholar] [CrossRef]
- Ailawadhi, S.; Kelly, K.R.; Vescio, R.A.; Jagannath, S.; Wolf, J.; Gharibo, M.; Sher, T.; Bojanini, L.; Kirby, M.; Chanan-Khan, A. A Phase I Study to Assess the Safety and Pharmacokinetics of Single-agent Lorvotuzumab Mertansine (IMGN901) in Patients with Relapsed and/or Refractory CD-56-positive Multiple Myeloma. Clin. Lymphoma Myeloma Leuk. 2019, 19, 29–34. [Google Scholar] [CrossRef]
- Cho, S.; Lin, L.; Xing, L.; Liu, J.; Yu, T.; Wen, K.; Hsieh, P.; Munshi, N.; Anderson, K.; Tai, Y.-T. Anti-BCMA BiTE® AMG 701 potently induces specific T cell lysis of human multiple myeloma (MM) cells and immunomodulation in the bone marrow microenvi- ronment. Blood 2018, 132, 592. [Google Scholar] [CrossRef]
- Lesokhin, A.M.; Raje, N.; Gasparetto, C.; Gasparetto, C.J.; Walker, J.; Krupka, P.H.I.; Joh, T.; Taylor, M.C.T.; Jakubowiak, A. Phase I, open-label study to evaluate the safety, pharmacokinetic, pharmacodynamic, and clinical activity of PF-06863135, a B-cell maturation anti- gen/CD3 bispecific antibody, in patients with relapsed/refractory advanced multiple myeloma. Blood 2016, 132, 3229. [Google Scholar] [CrossRef]
- Hipp, S.; Tai, Y.T.; Blanset, D.; Deegen, P.; Wahl, J.; Thomas, O.; Rattel, B.; Adam, P.J.; Anderson, K.C.; Friedrich, M. A novel BCMA/CD3 bispecific T-cell engager for the treatment of multiple myeloma induces selective lysis in vitro and in vivo. Leukemia 2017, 31, 2278. [Google Scholar] [CrossRef]
- Zuch de Zafra, C.L.; Fajardo, F.; Zhong, W.; Bernett, M.J.; Muchhal, U.S.; Moore, G.L.; Stevens, J.; Case, R.; Pearson, J.T.; Liu, S.; et al. Targeting Multiple Myeloma with AMG 424, a Novel Anti-CD38/CD3 Bispecific T-cell-recruiting Antibody Optimized for Cytotoxicity and Cytokine Release. Clin. Cancer Res. 2019, 25, 3921–3933. [Google Scholar] [CrossRef]
- Topp, M.S.; Duell, J.; Zugmaier, G.; Attal, M.; Moreau, P.; Langer, C.; Kronke, J.; Facon, T.; Salnikov, A.V.; Lesley, R.; et al. Anti-B-Cell Maturation Antigen BiTE Molecule AMG 420 Induces Responses in Multiple Myeloma. J. Clin. Oncol. 2020, 38, 775–783. [Google Scholar] [CrossRef]
- Berdeja, J.G.; Madduri, D.; Usmani, S.Z.; Jakubowiak, A.; Agha, M.; Cohen, A.D.; Stewart, A.K.; Hari, P.; Htut, M.; Lesokhin, A.; et al. Ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in patients with relapsed or refractory multiple myeloma (CARTITUDE-1): A phase 1b/2 open-label study. Lancet 2021, 398, 314–324. [Google Scholar] [CrossRef]
- Costa, L.; Wong, S.; Bermudez, A.; de la Rubia, J.; Mateos, M.-V.; Ocio, E.M.; Rodríguez-Otero, P.; San-Miguel, J.; Li, S.; Sarmiento, R.; et al. First Clinical Study of the B-Cell Maturation Antigen (BCMA) 2+1 T Cell Engager (TCE) CC-93269 in Pati ents (Pts) with Relapsed/Refractory Multiple Myeloma (RRMM): Interim Results of a Phase 1 Multicenter Trial. Blood 2019, 134, 143. [Google Scholar] [CrossRef]
- Chari, A.; Berdeja, J.; Oriol, A.; Van De Donk, N.W.C.J.; Rodriguez, P.; Askari, E.; Mateos, M.-V.; Minnema, M.C.; Verona, R.; Girgis, S.; et al. A phase 1, first-in-human study of talquetamad, a G protein-coupled receptor family C group 5 member D (GPRC5D) x CD3 bispecific antibody, in patients with relapsed and/or refractory multiple myeloma. In Proceedings of the 2020 ASH Annual Meeting and Exposition, Atlanta, GA, USA, 5 December 2020. [Google Scholar]
- Cohen, A.D.; Harrison, S.J.; Krishnan, A.; Fonseca, R.; Forsberg, P.A.; Spencer, A.; Berdeja, J.G.; Laubach, J.P.; Li, M.; Choeurng, V.; et al. Initial clinical activity and safety of BFCR4350A, a FcRH5/CD3 T-cell-engaging bispecific antibody, in relapsed/refractory multiple myeloma. In Proceedings of the 2020 ASH Annual Meeting and Exposition, Atlanta, GA, USA, 5 December 2020. [Google Scholar]
- June, C.H.; Sadelain, M. Chimeric Antigen Receptor Therapy. N. Engl. J. Med. 2018, 379, 64–73. [Google Scholar] [CrossRef]
- Maude, S.L.; Laetsch, T.W.; Buechner, J.; Rives, S.; Boyer, M.; Bittencourt, H.; Bader, P.; Verneris, M.R.; Stefanski, H.E.; Myers, G.D.; et al. Tisagenlecleucel in Children and Young Adults with B-Cell Lymphoblastic Leukemia. N. Engl. J. Med. 2018, 378, 439–448. [Google Scholar] [CrossRef]
- Schuster, S.J.; Svoboda, J.; Chong, E.A.; Nasta, S.D.; Mato, A.R.; Anak, O.; Brogdon, J.L.; Pruteanu-Malinici, I.; Bhoj, V.; Landsburg, D.; et al. Chimeric Antigen Receptor T Cells in Refractory B-Cell Lymphomas. N. Engl. J. Med. 2017, 377, 2545–2554. [Google Scholar] [CrossRef]
- Neelapu, S.S.; Locke, F.L.; Bartlett, N.L.; Lekakis, L.J.; Miklos, D.B.; Jacobson, C.A.; Braunschweig, I.; Oluwole, O.O.; Siddiqi, T.; Lin, Y.; et al. Axicabtagene Ciloleucel CAR T-Cell Therapy in Refractory Large B-Cell Lymphoma. N. Engl. J. Med. 2017, 377, 2531–2544. [Google Scholar] [CrossRef]
- Munshi, N.C.; Anderson, L.D., Jr.; Shah, N.; Madduri, D.; Berdeja, J.; Lonial, S.; Raje, N.; Lin, Y.; Siegel, D.; Oriol, A.; et al. Idecabtagene Vicleucel in Relapsed and Refractory Multiple Myeloma. N. Engl. J. Med. 2021, 384, 705–716. [Google Scholar] [CrossRef]
- Raje, N.; Berdeja, J.; Lin, Y.; Siegel, D.; Jagannath, S.; Madduri, D.; Liedtke, M.; Rosenblatt, J.; Maus, M.V.; Turka, A.; et al. Anti-BCMA CAR T-Cell Therapy bb2121 in Relapsed or Refractory Multiple Myeloma. N. Engl. J. Med. 2019, 380, 1726–1737. [Google Scholar] [CrossRef]
- Xu, J.; Chen, L.J.; Yang, S.S.; Sun, Y.; Wu, W.; Liu, Y.F.; Xu, J.; Zhuang, Y.; Zhang, W.; Weng, X.Q.; et al. Exploratory trial of a biepitopic CAR T-targeting B cell maturation antigen in relapsed/refractory multiple myeloma. Proc. Natl. Acad. Sci. USA 2019, 116, 9543–9551. [Google Scholar] [CrossRef]
- Alsina, M.; Shah, N.; Raje, N.S.; Jagannath, S.; Madduri, D.; Kaufman, J.L.; Siegel, D.S.; Munshi, N.C.; Rosenblatt, J.; Lin, Y.; et al. Updated Results from the Phase I CRB-402 Study of Anti-Bcma CAR-T Cell Therapy bb21217 in Patients with Relapsed and Refractory Multiple Myeloma: Correlation of Expansion and Duration of Response with T Cell Phenotypes. Blood 2020, 136 (Suppl. S1), 25–26. [Google Scholar] [CrossRef]
- Kumar, S.K.; Baz, R.C.; Orlowski, R.Z.; Anderson, L.D., Jr.; Ma, H.; Shrewsbury, A.; Croghan, K.A.; Bilgi, M.; Kansagra, A.; Kapoor, P.; et al. Results from Lummicar-2: A Phase 1b/2 Study of Fully Human B-Cell Maturation Antigen-Specific CAR T Cells (CT053) in Patients with Relapsed and/or Refractory Multiple Myeloma. Blood 2020, 136 (Suppl. S1), 28–29. [Google Scholar] [CrossRef]
- Costello, C.L.; Cohen, A.D.; Patel, K.K.; Ali, S.S.; Berdeja, J.G.; Shah, N.; Ganguly, S.; Kocoglu, M.H.; Abedi, M.; Ostertag, E.M.; et al. Phase 1/2 Study of the Safety and Response of P-BCMA-101 CAR-T Cells in Patients with Relapsed/Refractory (r/r) Multiple Myeloma (MM) (PRIME) with Novel Therapeutic Strategies. Blood 2020, 136 (Suppl. S1), 29–30. [Google Scholar] [CrossRef]
- Jiang, H.; Dong, B.; Gao, L.; Liu, L.; Ge, J.; He, A.; Du, J., Jr.; Li, L.; Lu, J.; Chen, X.; et al. Clinical Results of a Multicenter Study of the First-in-Human Dual BCMA and CD19 Targeted Novel Platform Fast CAR-T Cell Therapy for Patients with Relapsed/Refractory Multiple Myeloma. Blood 2020, 136 (Suppl. S1), 25–26. [Google Scholar] [CrossRef]
- Mailankody, S.; Matous, J.V.; Liedtke, M.; Sidana, S.; Malik, S.; Nath, R.; Oluwole, O.O.; Karski, E.E.; Lovelace, W.; Zhou, X.; et al. Universal: An Allogeneic First-in-Human Study of the Anti-Bcma ALLO-715 and the Anti-CD52 ALLO-647 in Relapsed/Refractory Multiple Myeloma. Blood 2020, 136 (Suppl. S1), 24–25. [Google Scholar] [CrossRef]
- Rosenzweig, M.; Urak, R.; Walter, M.; Lim, L.; Sanchez, J.F.; Krishnan, A.; Forman, S.; Wang, X. Preclinical data support leveraging CS1 chimeric antigen receptor T-cell therapy for systemic light chain amyloidosis. Cytotherapy 2017, 19, 861–866. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Pfarr, N.; Endris, V.; Mai, E.K.; Md Hanafiah, N.H.; Lehners, N.; Penzel, R.; Weichert, W.; Ho, A.D.; Schirmacher, P.; et al. Molecular signaling in multiple myeloma: Association of RAS/RAF mutations and MEK/ERK pathway activation. Oncogenesis 2017, 6, e337. [Google Scholar] [CrossRef]
- Chng, W.J.; Gonzalez-Paz, N.; Price-Troska, T.; Jacobus, S.; Rajkumar, S.V.; Oken, M.M.; Kyle, R.A.; Henderson, K.J.; Van Wier, S.; Greipp, P.; et al. Clinical and biological significance of RAS mutations in multiple myeloma. Leukemia 2008, 22, 2280–2284. [Google Scholar] [CrossRef]
- Walker, B.A.; Mavrommatis, K.; Wardell, C.P.; Ashby, T.C.; Bauer, M.; Davies, F.E.; Rosenthal, A.; Wang, H.; Qu, P.; Hoering, A.; et al. Identification of novel mutational drivers reveals oncogene dependencies in multiple myeloma. Blood 2018, 132, 587–597. [Google Scholar] [CrossRef]
- Chapman, M.A.; Lawrence, M.S.; Keats, J.J.; Cibulskis, K.; Sougnez, C.; Schinzel, A.C.; Harview, C.L.; Brunet, J.P.; Ahmann, G.J.; Adli, M.; et al. Initial genome sequencing and analysis of multiple myeloma. Nature 2011, 471, 467–472. [Google Scholar] [CrossRef]
- Raab, M.S.; Giesen, P.L.; Scheid, C.; Besemer, B.; Miah, K.; Benner, A.; Metzler, I.; Khandanpour, P.C.; Seidel-Glaetzer, A.; Trautmann-Grill, K.; et al. Safety and Preliminary Efficacy Results from a Phase II Study Evaluating Combined BRAF and MEK Inhibition in Relapsed/Refractory Multiple Myeloma (rrMM) Patients with Activating BRAF V600E Mutations: The GMMG-Birma Trial. Blood 2020, 136, 44–45. [Google Scholar] [CrossRef]
- Schjesvold, F.H.; Ribrag, V.; Rodriguez-Otero, P.; Robak, P.J.; Hansson, M.; Hajek, R.; Amor, A.A.; Martinez-López, J.; Onishi, M.; Gallo, J.D.; et al. Safety and Preliminary Efficacy Results from a Phase Ib/II Study of Cobimetinib As a Single Agent and in Combination with Venetoclax with or without Atezolizumab in Patients with Relapsed/Refractory Multiple Myeloma. Blood 2020, 136, 45–46. [Google Scholar] [CrossRef]
- Guo, C.; Chenard-Poirier, M.; Roda, D.; de Miguel, M.; Harris, S.J.; Candilejo, I.M.; Sriskandarajah, P.; Xu, W.; Scaranti, M.; Constantinidou, A.; et al. Intermittent schedules of the oral RAF-MEK inhibitor CH5126766/VS-6766 in patients with RAS/RAF-mutant solid tumours and multiple myeloma: A single-centre, open-label, phase 1 dose-escalation and basket dose-expansion study. Lancet Oncol. 2020, 21, 1478–1488. [Google Scholar] [CrossRef]
- Ismail, S.I.; Mahmoud, I.S.; Msallam, M.M.; Sughayer, M.A. Hotspot mutations of PIK3CA and AKT1 genes are absent in multiple myeloma. Leuk. Res. 2010, 34, 824–826. [Google Scholar] [CrossRef]
- Jakubowiak, A.J.; Richardson, P.G.; Zimmerman, T.; Alsina, M.; Kaufman, J.L.; Kandarpa, M.; Kraftson, S.; Ross, C.W.; Harvey, C.; Hideshima, T.; et al. Perifosine plus lenalidomide and dexamethasone in relapsed and relapsed/refractory multiple myeloma: A Phase I Multiple Myeloma Research Consortium study. Br. J. Haematol. 2012, 158, 472–480. [Google Scholar] [CrossRef] [PubMed]
- Richardson, P.G.; Wolf, J.; Jakubowiak, A.; Zonder, J.; Lonial, S.; Irwin, D.; Densmore, J.; Krishnan, A.; Raje, N.; Bar, M.; et al. Perifosine plus bortezomib and dexamethasone in patients with relapsed/refractory multiple myeloma previously treated with bortezomib: Results of a multicenter phase I/II trial. J. Clin. Oncol. 2011, 29, 4243–4249. [Google Scholar] [CrossRef] [PubMed]
- Richardson, P.G.; Nagler, A.; Ben-Yehuda, D.; Badros, A.; Hari, P.N.; Hajek, R.; Spicka, I.; Kaya, H.; Leblanc, R.; Yoon, S.; et al. Randomized, placebo-controlled, phase 3 study of perifosine combined with bortezomib and dexamethasone in patients with relapsed, refractory multiple myeloma previously treated with bortezomib. Eur. J. Haematol. 2020, 1, 94–102. [Google Scholar] [CrossRef]
- Spencer, A.; Yoon, S.S.; Harrison, S.J.; Morris, S.R.; Smith, D.A.; Brigandi, R.A.; Gauvin, J.; Kumar, R.; Opalinska, J.B.; Chen, C. The novel AKT inhibitor afuresertib shows favorable safety, pharmacokinetics, and clinical activity in multiple myeloma. Blood 2014, 124, 2190–2195. [Google Scholar] [CrossRef]
- Voorhees, J.R.; Spencer, A.; Sutherland, H.J.; O’Dwyer, M.; Huang, S.-Y.; Stewart, K.; Chari, A.; Rosenzwieg, M.; Nooka, A.K.; Rosenbaum, C.A.; et al. Novel AKT Inhibitor Afuresertib in Combination with Bortezomib and Dexamethasone Demonstrates Favorable Safety Profile and Significant Clinical Activity in Patients with Relapsed Refractory Multiple Myeloma. Blood 2013, 122, 283. [Google Scholar]
- Vassilev, L.T.; Vu, B.T.; Graves, B.; Carvajal, D.; Podlaski, F.; Filipovic, Z.; Kong, N.; Kammlott, U.; Lukacs, C.; Klein, C.; et al. In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science 2004, 303, 844–848. [Google Scholar] [CrossRef]
- Saha, M.N.; Jiang, H.; Jayakar, J.; Reece, D.; Branch, D.R.; Chang, H. MDM2 antagonist nutlin plus proteasome inhibitor velcade combination displays a synergistic anti-myeloma activity. Cancer Biol. Ther. 2010, 9, 936–944. [Google Scholar] [CrossRef]
- Issaeva, N.; Bozko, P.; Enge, M.; Protopopova, M.; Verhoef, L.G.; Masucci, M.; Pramanik, A.; Selivanova, G. Small molecule RITA binds to p53, blocks p53-HDM-2 interaction and activates p53 function in tumors. Nat. Med. 2004, 10, 1321–1328. [Google Scholar] [CrossRef]
- Surget, S.; Descamps, G.; Brosseau, C.; Normant, V.; Maiga, S.; Gomez-Bougie, P.; Gouy-Colin, N.; Godon, C.; Bene, M.C.; Moreau, P.; et al. RITA (Reactivating p53 and Inducing Tumor Apoptosis) is efficient against TP53abnormal myeloma cells independently of the p53 pathway. BMC Cancer 2014, 14, 437. [Google Scholar] [CrossRef]
- Matyskiela, M.E.; Zhang, W.; Man, H.W.; Muller, G.; Khambatta, G.; Baculi, F.; Hickman, M.; LeBrun, L.; Pagarigan, B.; Carmel, G.; et al. A Cereblon Modulator (CC-220) with Improved Degradation of Ikaros and Aiolos. J. Med. Chem. 2018, 61, 535–542. [Google Scholar] [CrossRef]
- Hansen, J.D.; Correa, M.; Nagy, M.A.; Alexander, M.; Plantevin, V.; Grant, V.; Whitefield, B.; Huang, D.; Kercher, T.; Harris, R.; et al. Discovery of CRBN E3 Ligase Modulator CC-92480 for the Treatment of Relapsed and Refractory Multiple Myeloma. J. Med. Chem. 2020, 63, 6648–6676. [Google Scholar] [CrossRef] [PubMed]
- Lonial, S.; van de Donk, N.; Ropat, R.; Zonder, J.; Minnema, M.C.; Laesen, J.; Nguyen, N.L.; Chen, M.S.; Bensmaine, A. First clinical (phase 1b/2a) study of iberdomide (CC-220; IBER), a CELMoD, in combination with dexamethasone (DEX) in patients (pts) with relapsed/refractory multiple myeloma (RRMM). J. Clin. Oncol. 2019, 37, 8006. [Google Scholar] [CrossRef]
- Orchard, K.; Cooper, M.; Lewington, V.; Tristam, M.; Zivanovic, M.; Thom, J.; Quadri, S.; Richardson, D.; Causer, L.; Johnson, P. Targeted radiotherapy in the conditioning prior to haematopoietic stem cell transplantation: Results of a phase I trial using an yttrium-90-labelled anti-CD66 murine monoclonal antibody demonstrating consistently high BM uptake. Biol. Blood Marrow Transplant. 2006, 12, 10–11. [Google Scholar] [CrossRef][Green Version]
- Fasslrinner, F.; Stolzel, F.; Kramer, M.; Teipel, R.; Brogsitter, C.; Morgner, A.; Arndt, C.; Bachmann, M.; Hanel, M.; Rollig, C.; et al. Radioimmunotherapy in Combination with Reduced-Intensity Conditioning for Allogeneic Hematopoietic Cell Transplantation in Patients with Advanced Multiple Myeloma. Biol. Blood Marrow Transplant. 2020, 26, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Hossein Taghizadeh, M.; Thomas, R.; Franz, D.; Christina, B.; Kornauth, M.; Christoph, T.; Agis, H. Daratumumab–a safe first-line treatment of cardiac AL amyloidosis in heavily compromised patients. In Proceedings of the XVIth International Symposium on Amyloidosis, Kumamoto, Japan, 26–29 March 2018. [Google Scholar]
- Van de Wyngaert, Z.; Carpentier, B.; Pascal, L.; Lionne-Huyghe, P.; Leduc, I.; Srour, M.; Vasseur, M.; Demarquette, H.; Terriou, L.; Herbaux, C.; et al. Daratumumab is effective in the relapsed or refractory systemic light-chain amyloidosis but associated with high infection burden in a frail real-life population. Br. J. Haematol. 2020, 188, e24–e27. [Google Scholar] [CrossRef]
- Kastritis, E.; Dialoupi, I.; Gavriatopoulou, M.; Roussou, M.; Kanellias, N.; Fotiou, D.; Ntanasis-Stathopoulos, I.; Papadopoulou, E.; Ziogas, D.C.; Stamatelopoulos, K.; et al. Primary treatment of light-chain amyloidosis with bortezomib, lenalidomide, and dexamethasone. Blood Adv. 2019, 3, 3002–3009. [Google Scholar] [CrossRef]
- Zonder, J.A.; Sanchorawala, V.; Kukreti, V.; Tuchman, S.A.; Kang, Y.; Bhutani, D.; Burt, S.; Houde, C.; Reichel, K.A.; Pregja, S.; et al. Phase I Trial of Pomalidomide, Bortezomib, and Dexamethasone As Frontline Treatment of AL Amyloidosis. Blood 2017, 130 (Suppl. S1), 3158. [Google Scholar]
- Teachey, D.T.; Lacey, S.F.; Shaw, P.A.; Melenhorst, J.J.; Maude, S.L.; Frey, N.; Pequignot, E.; Gonzalez, V.E.; Chen, F.; Finklestein, J.; et al. Identification of Predictive Biomarkers for Cytokine Release Syndrome after Chimeric Antigen Receptor T-cell Therapy for Acute Lymphoblastic Leukemia. Cancer Discov. 2016, 6, 664–679. [Google Scholar] [CrossRef]
- Adams, D.; Gonzalez-Duarte, A.; O’Riordan, W.D.; Yang, C.-C.; Ueda, M.; Kristen, A.V.; Tournev, I.; Schmidt, H.H.; Coelho, T.; Berk, J.L.; et al. Patisiran, an RNAi Therapeutic, for Hereditary Transthyretin Amyloidosis. N. Engl. J. Med. 2018, 379, 11–21. [Google Scholar] [CrossRef]
- Benson, M.D.; Waddington-Cruz, M.; Berk, J.L.; Polydefkis, M.; Dyck, P.J.; Wang, A.K.; Planté-Bordeneuve, V.; Barroso, F.A.; Merlini, G.; Obici, L.; et al. Inotersen Treatment for Patients with Hereditary Transthyretin Amyloidosis. N. Engl. J. Med. 2018, 379, 22–31. [Google Scholar] [CrossRef]
- Kluve-Beckerman, B.; Hardwick, J.; Du, L.; Benson, M.D.; Monia, B.P.; Watt, A.; Crooke, R.M.; Mullick, A. Antisense oligonucleotide suppression of serum amyloid A reduces amyloid deposition in mice with AA amyloidosis. Amyloid 2011, 18, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Ohno, S.; Yoshimoto, M.; Honda, S.; Miyachi, S.; Ishida, T.; Itoh, F.; Endo, T.; Chiba, S.; Imai, K. The Antisense Approach in Amyloid Light Chain Amyloidosis: Identification of Monoclonal Ig and Inhibition of Its Production by Antisense Oligonucleotides in In Vitro and In Vivo Models. J. Immunol. 2002, 169, 4039–4045. [Google Scholar] [CrossRef] [PubMed]
- Phipps, J.E.; Kestler, D.P.; Foster, J.S.; Kennel, S.J.; Donnell, R.; Weiss, D.T.; Solomon, A.; Wall, J.S. Inhibition of pathologic immunoglobulin-free light chain production by small interfering RNA molecules. Exp. Hematol. 2010, 38, 1006–1013. [Google Scholar] [CrossRef] [PubMed]
- Hovey, B.M.; Ward, J.E.; Soo Hoo, P.; O’Hara, C.J.; Connors, L.H.; Seldin, D.C. Preclinical development of siRNA therapeutics for AL amyloidosis. Gene Ther. 2011, 18, 1150–1156. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Ma, X.; Iyer, L.; Chaulagain, C.; Comenzo, R.L. One siRNA pool targeting the λ constant region stops λ light-chain production and causes terminal endoplasmic reticulum stress. Blood 2014, 123, 3440–3451. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Zhou, P.; Wong, S.W.; Warner, M.; Chaulagain, C.; Comenzo, R.L. siRNA targeting the κ light chain constant region: Preclinical testing of an approach to nonfibrillar and fibrillar light chain deposition diseases. Gene Ther. 2016, 23, 727–733. [Google Scholar] [CrossRef]
- Bertrand, J.R.; Pottier, M.; Vekris, A.; Opolon, P.; Maksimenko, A.; Malvy, C. Comparison of antisense oligonucleotides and siRNAs in cell culture and in vivo. Biochem. Biophys. Res. Commun. 2002, 296, 1000–1004. [Google Scholar] [CrossRef]
- Klimtchuk, E.S.; Gursky, O.; Patel, R.S.; Laporte, K.L.; Connors, L.H.; Skinner, M.; Seldin, D.C. The Critical Role of the Constant Region in Thermal Stability and Aggregation of Amyloidogenic Immunoglobulin Light Chain. Biochemistry 2010, 49, 9848–9857. [Google Scholar] [CrossRef]
- Ma, X.; Zhou, P.; Kugelmass, A.; Toskic, D.; Warner, M.; Lee, L.; Fogaren, T.; Godara, A.; Wang, M.; Li, Y.; et al. A novel xenograft mouse model for testing approaches targeting human kappa light-chain diseases. Gene Ther. 2019, 26, 187–197. [Google Scholar] [CrossRef]
- Maurer, M.S.; Schwartz, J.H.; Gundapaneni, B.; Elliott, P.M.; Merlini, G.; Waddington-Cruz, M.; Kristen, A.V.; Grogan, M.; Witteles, R.; Damy, T.; et al. Tafamidis Treatment for Patients with Transthyretin Amyloid Cardiomyopathy. N. Engl. J. Med. 2018, 379, 1007–1016. [Google Scholar] [CrossRef]
- Morgan, G.J.; Yan, N.L.; Mortenson, D.E.; Rennella, E.; Blundon, J.M.; Gwin, R.M.; Lin, C.-Y.; Stanfield, R.L.; Brown, S.J.; Rosen, H.; et al. Stabilization of amyloidogenic immunoglobulin light chains by small molecules. Proc. Natl. Acad. Sci. USA 2019, 116, 8360–8369. [Google Scholar] [CrossRef] [PubMed]
- Brumshtein, B.; Esswein, S.R.; Salwinski, L.; Phillips, M.L.; Ly, A.T.; Cascio, D.; Sawaya, M.R.; Eisenberg, D.S. Inhibition by small-molecule ligands of formation of amyloid fibrils of an immunoglobulin light chain variable domain. Elife 2015, 4, e10935. [Google Scholar] [CrossRef] [PubMed]
- Morgan, G.J. Barriers to Small Molecule Drug Discovery for Systemic Amyloidosis. Molecules 2021, 26, 3571. [Google Scholar] [CrossRef] [PubMed]
- Wechalekar, A.D.; Whelan, C. Encouraging impact of doxycycline on early mortality in cardiac light chain (AL) amyloidosis. Blood Cancer J. 2017, 7, e546. [Google Scholar] [CrossRef]
- Shen, K.N.; Fu, W.J.; Wu, Y.; Dong, Y.J.; Huang, Z.X.; Wei, Y.Q.; Li, C.R.; Sun, C.Y.; Chen, Y.; Miao, H.L.; et al. Doxycycline Combined With Bortezomib-Cyclophosphamide-Dexamethasone Chemotherapy for Newly Diagnosed Cardiac Light-Chain Amyloidosis: A Multicenter Randomized Controlled Trial. Circulation 2021, 145, 8–17. [Google Scholar] [CrossRef]
- Edwards, C.V.; Gould, J.; Langer, A.L.; Mapara, M.Y.; Radhakrishnan, J.; Maurer, M.S.; Raza, S.; Mears, J.G.; Leng, S.; Wall, J.S.; et al. Final Analysis of the Phase 1a/b Study of Chimeric Fibril-Reactive Monoclonal Antibody 11-1F4 in Patients with Relapsed or Refractory AL Amyloidosis. Blood 2017, 130 (Suppl. S1), 509. [Google Scholar] [CrossRef]
- Bhutani, D.; Leng, S.; Eisenberger, A.; Maurer, M.S.; Shames, S.; Goldsmith, J.; Lentzsch, S. Improvement in Global Longitudinal Strain (GLS) Correlates with NT-Probnp Response in Patients with Cardiac Amyloidosis Treated on a Phase 1b Study of Anti-Amyloid Mab Cael-101. Blood 2018, 132 (Suppl. S1), 958. [Google Scholar] [CrossRef]
- Gertz, M.A.; Landau, H.; Comenzo, R.L.; Seldin, D.; Weiss, B.; Zonder, J.; Merlini, G.; Schonland, S.; Walling, J.; Kinney, G.G.; et al. First-in-Human Phase I/II Study of NEOD001 in Patients With Light Chain Amyloidosis and Persistent Organ Dysfunction. J. Clin. Oncol. 2016, 34, 1097–1103. [Google Scholar] [CrossRef]
| Name | Mechanism of Action | Study Phase | Results in Patients with AL Amyloidosis | Comments |
|---|---|---|---|---|
| Venetoclax | BCL-2 inhibitor | Phase III (in MM) | Yes in retrospective studies ORR: 81%, CR/VGPR: 78%, Organ response rate: 83% | Only in patients with t(11;14) |
| Liftasoclax | BCL-2 inhibitor | Phase I/II (in MM and AL) | No, only in patients with myeloma | Only in patients with t(11;14) |
| Belantamab mafodotin | Anti-BCMA ADC | Phase II (in AL) | Study is recruiting | Ocular toxicity of concern |
| Milatuzumab doxorubicin | Anti-CD56 | Phase I (in MM) | No, only in patients with myeloma | |
| Indatuximab ravtansine | Anti-CD138 | Phase I/IIa (in MM) | No, only in patients with myeloma | |
| Talquetamab | BiTEs (GP3C5D/ CD3 on T cells) | Phase I | No, only in patients with myeloma | |
| Cevostamab | BiTEs (FcRH5/CD3) | Phase I | No, only in patients with myeloma | |
| Idecaptagene cicleucel | BCMA targeting CAR-T cells | Phase II | No, only in patients with myeloma | Toxicity is a concern–selected patients only |
| Ciltacabtagene autoleucel | BCMA targeting CAR-T cells | Phase I/IIa | No, only in patients with myeloma | Toxicity is a concern–selected patients only |
| Tisagenlecleucel | CD19 CAR T-cell | Approved for RR B-cell precursor acute Lymphoblastic leukemia and large B-cell lymphoma | Ongoing phase II studies in B-cell lymphomas to include also indolent diseases | Future option for IgM amyloidosis |
| Axicabtagene ciloleucel | CD19 CAR-T-cell | Approved for RR large B-cell lymphoma | ||
| Brexucabtagene autoleucel, | CD19-CAR-T-cell | Approved for RR mantle cell Lymphoma and B-cell precursor acute lymphoblastic leukemia | ||
| Lisocabtagene maraleucel | CD19 CAR-T cell | Approved for RR Large B-cell Lymphomas | ||
| Iberdomide (CC-220) | IMiDs | Phase II | No, only in patients with myeloma | |
| CC-92480 | IMiDs | Phase I/II | No, only in patients with myeloma | |
| Encorafenib and binimetinib | Dual BRAF and MEK inhibitor | Phase II | No, only in patients with myeloma | In patients with BRAF V600E mutation |
| TLX66 | Bone marrow conditioning agent | Phase I/II (in AL) | Yes, hemResponse in 7/9 patients | Potential as a conditioning regimen for patients otherwise ineligible for high dose therapy |
| Cael-101 | Amyloid targeting | Phase 3 | Yes, cardiac response: 67%, renal response: 50% | Phase 2 data available, ongoing phase 3 study in newly diagnosed patients with stage 3 disease |
| Birtamimab | Amyloid targeting | Phase 3 | Yes, 50% relative risk reduction for all-cause mortality for Mayo stage IV patients | Initial phase 2 data positive, not confirmed in randomized phase 2 and phase 3. Under evaluation in stage IV (Mayo 2012) based on positive results in post hoc analysis |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Theodorakakou, F.; Fotiou, D.; Dimopoulos, M.A.; Kastritis, E. Future Developments in the Treatment of AL Amyloidosis. Hemato 2022, 3, 131-152. https://doi.org/10.3390/hemato3010012
Theodorakakou F, Fotiou D, Dimopoulos MA, Kastritis E. Future Developments in the Treatment of AL Amyloidosis. Hemato. 2022; 3(1):131-152. https://doi.org/10.3390/hemato3010012
Chicago/Turabian StyleTheodorakakou, Foteini, Despina Fotiou, Meletios A. Dimopoulos, and Efstathios Kastritis. 2022. "Future Developments in the Treatment of AL Amyloidosis" Hemato 3, no. 1: 131-152. https://doi.org/10.3390/hemato3010012
APA StyleTheodorakakou, F., Fotiou, D., Dimopoulos, M. A., & Kastritis, E. (2022). Future Developments in the Treatment of AL Amyloidosis. Hemato, 3(1), 131-152. https://doi.org/10.3390/hemato3010012







