Effects of Nearby Construction Work on the Behavior of Asiatic Lions (Panthera leo persica)
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Subjects and Housing
2.2. Behavior Data Collection
- Pre-construction: 40 days of data collected prior to the start of construction (14 September 2017 to 28 November 2017).
- During construction: 40 days of data collected while construction work was ongoing (30 November 2017 to 1 March 2018).
- Post-construction: 40 days of data collected a year after the pre-construction data (13 September 2018 to 12 December 2018).
2.3. Statistical Analysis
3. Results
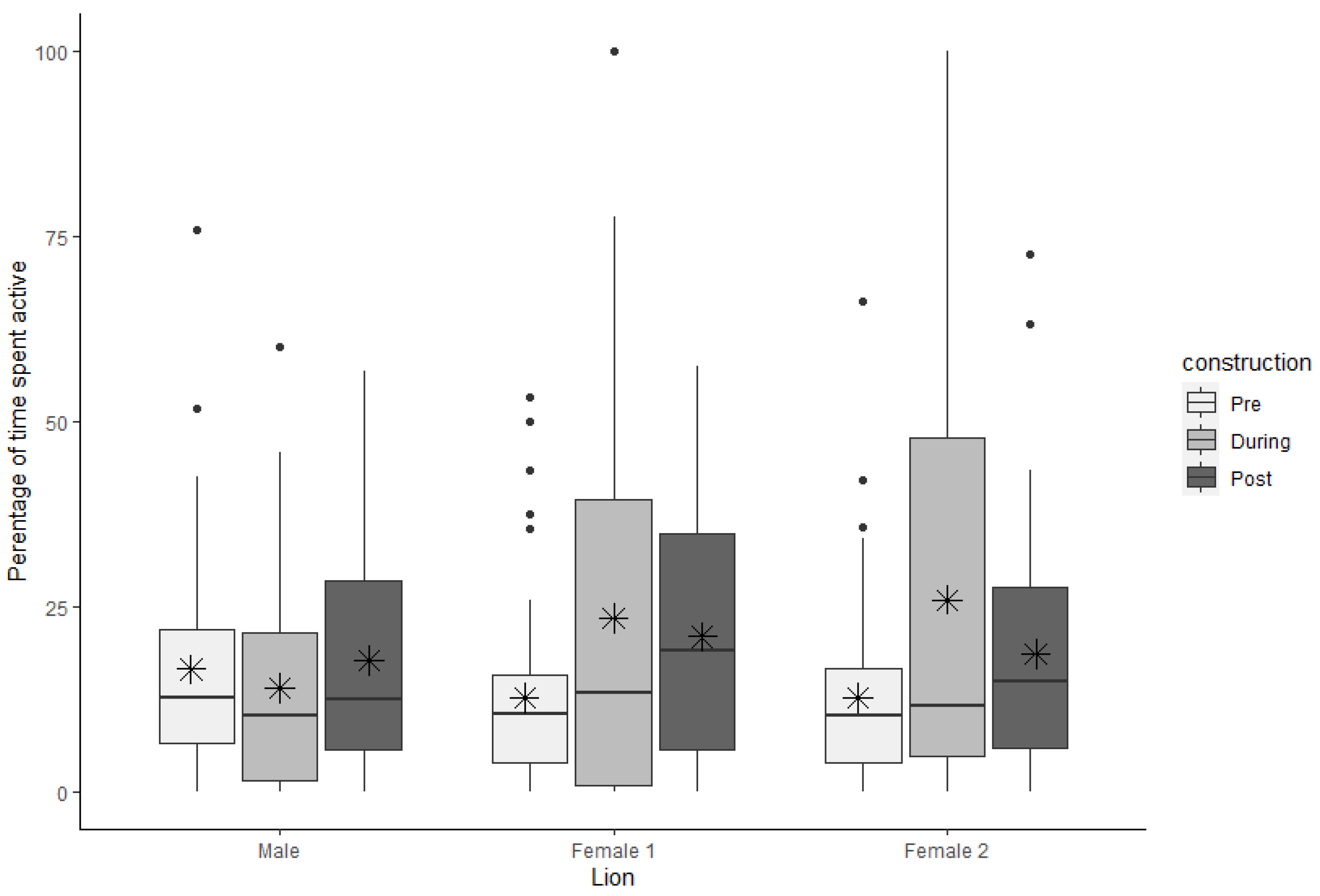
3.1. Natural Active Behaviors
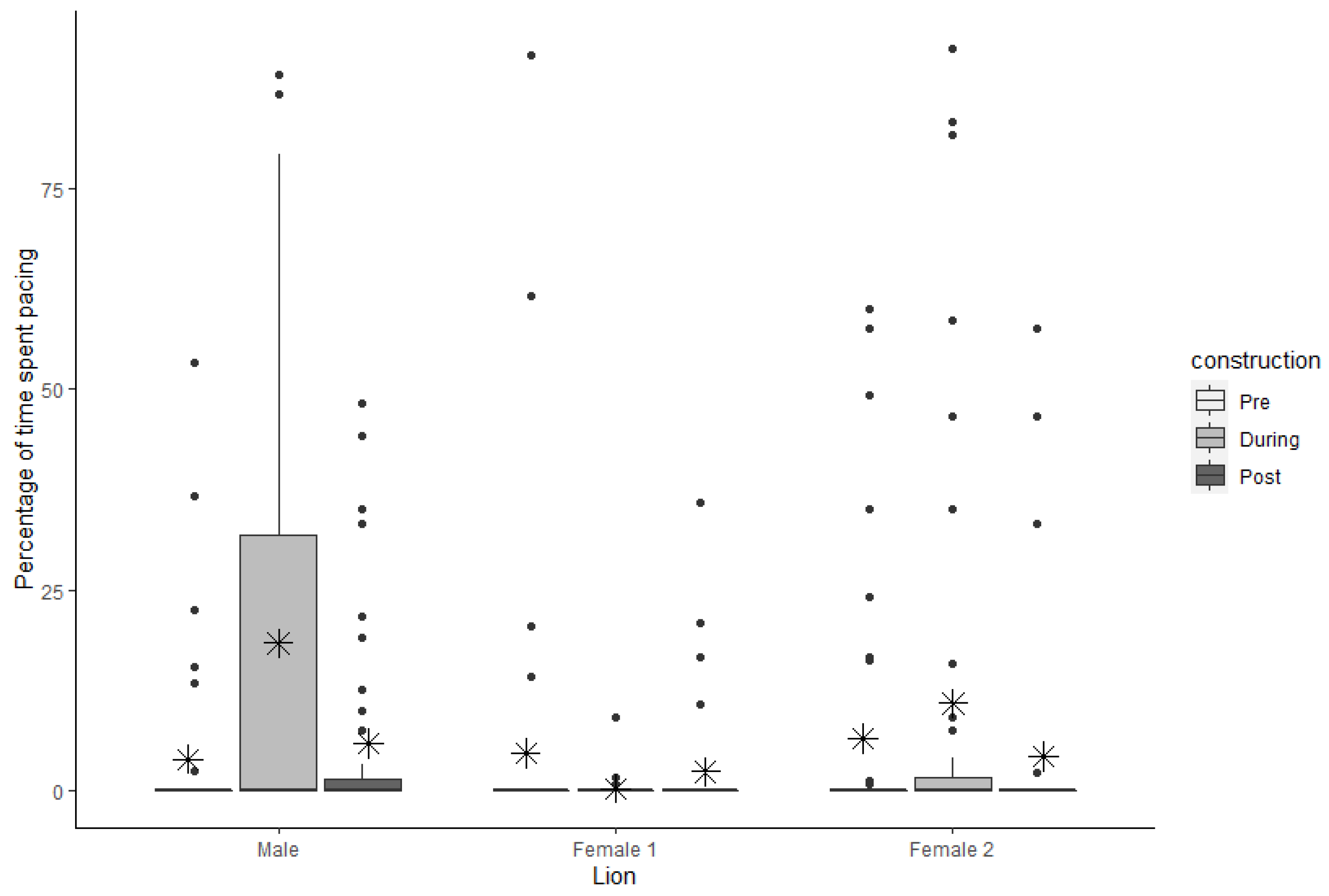
3.2. Pacing
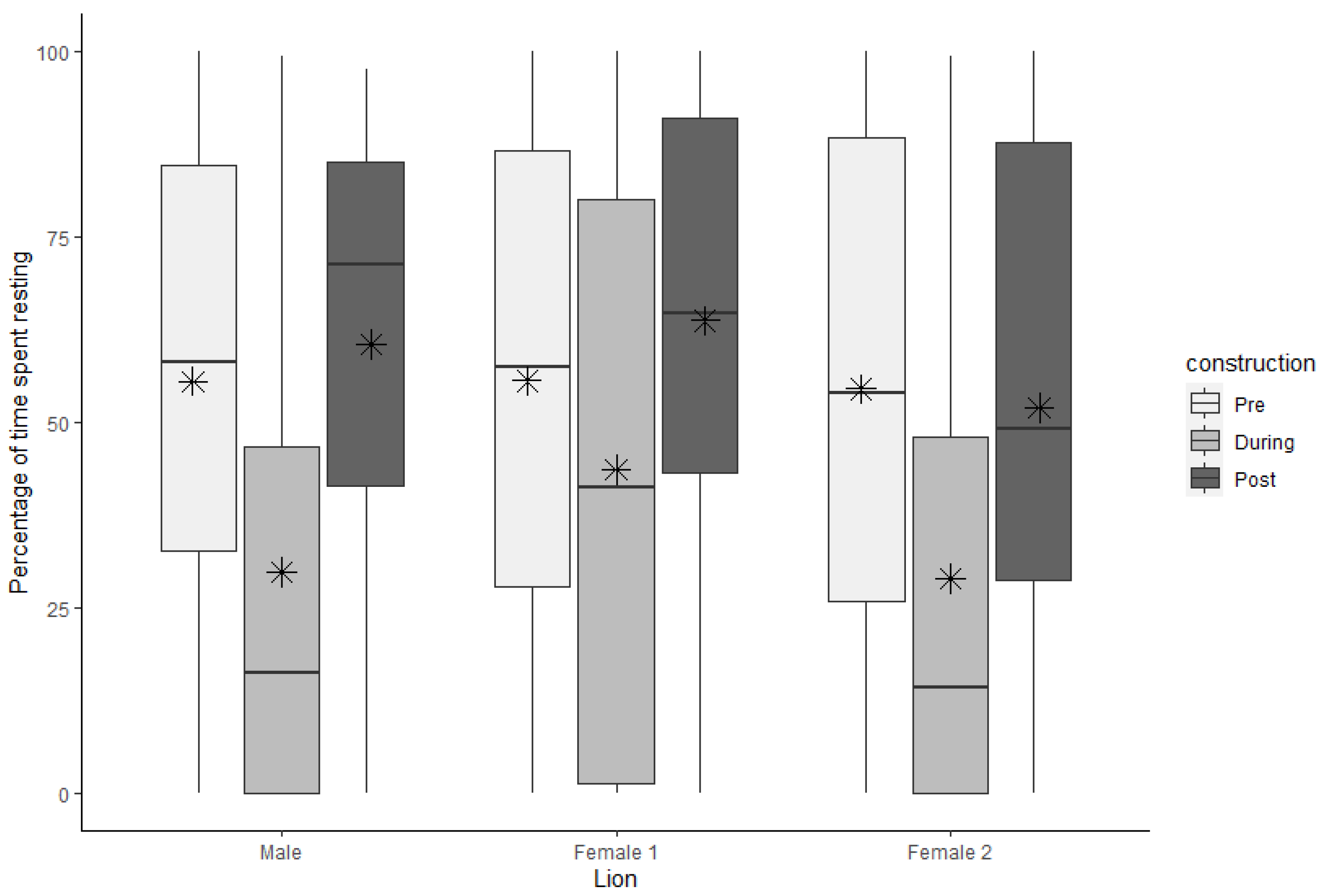
3.3. Resting
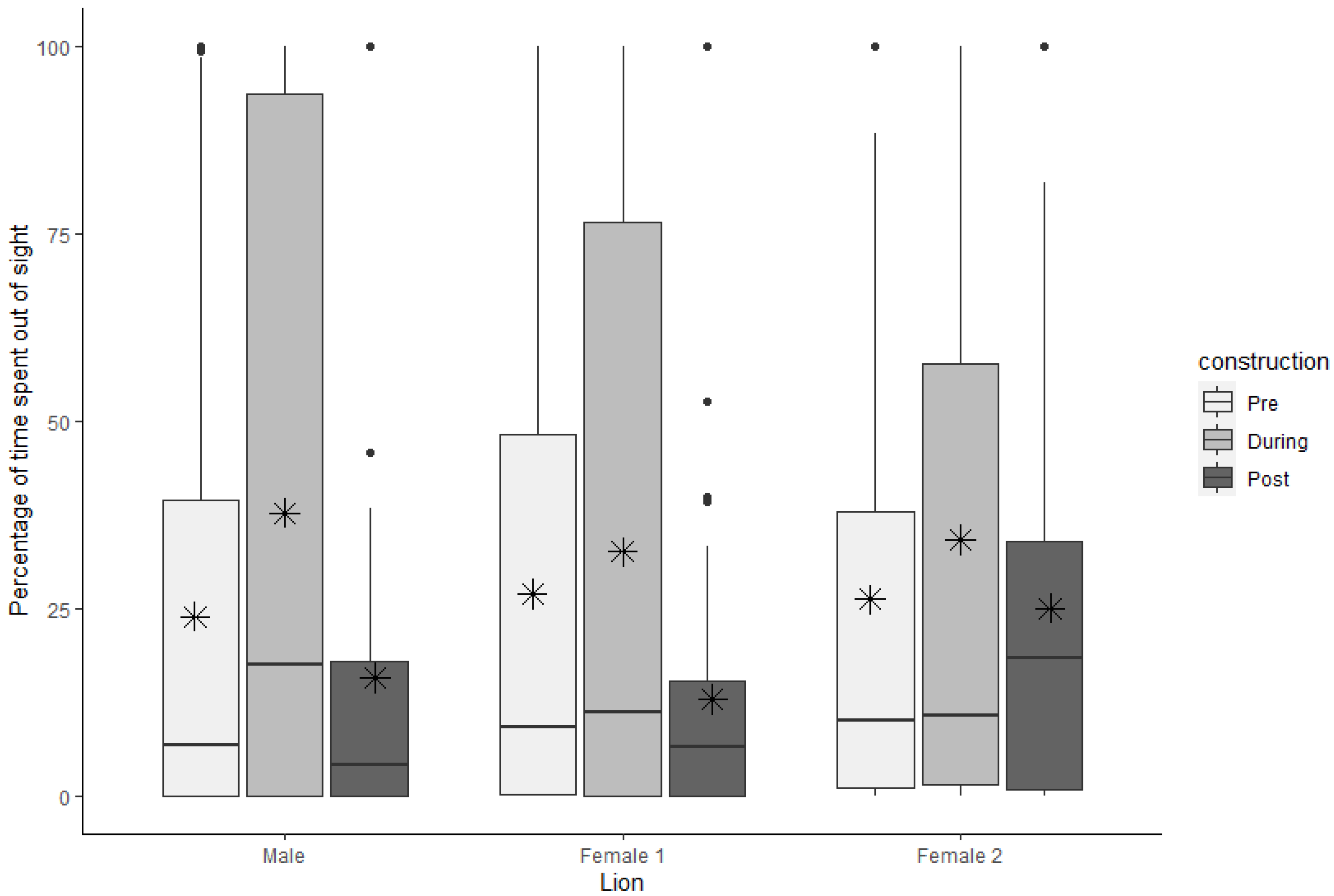
3.4. Visibility
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hosey, G.; Melfi, V.; Pankhurst, S. Zoo Animals: Behavior, Management, and Welfare, 2nd ed.; Oxford University Press: Oxford, UK, 2013. [Google Scholar]
- Melfi, V.A.; McCormick, W.; Gibbs, A. A preliminary assessment of how zoo visitors evaluate animal welfare according to enclosure style and the expression of behavior. Anthrozoös 2004, 17, 98–108. [Google Scholar] [CrossRef]
- Davey, G. Relationships between exhibit naturalism, animal visibility and visitor interest in a Chinese Zoo. Appl. Anim. Behav. Sci. 2006, 96, 93–102. [Google Scholar] [CrossRef]
- Powell, D.M.; Carlstead, K.; Tarou, L.R.; Brown, J.L.; Monfort, S.L. Effects of construction noise on behavior and cortisol levels in a pair of captive giant pandas (Ailuropoda melanoleuca). Zoo Biol. 2006, 25, 391–408. [Google Scholar] [CrossRef]
- Hosey, G. Zoo animals and their human audiences: What is the visitor effect? Anim. Welf. 2000, 9, 242–257. [Google Scholar]
- Carlstead, K.; Shepherdson, D. Effects of environmental enrichment on reproduction. Zoo Biol. 1994, 13, 447–458. [Google Scholar] [CrossRef]
- Morgan, K.N.; Tromborg, C.T. Sources of stress in captivity. Appl. Anim. Behav. Sci. 2007, 102, 262–302. [Google Scholar] [CrossRef]
- Sulser, C.E.; Steck, B.L.; Baur, B. Effects of construction noise on behavior of and exhibit use by Snow leopards Uncia uncia at Basel zoo. Int. Zoo. Yearb. 2008, 42, 199–205. [Google Scholar] [CrossRef]
- Jakob-Hoff, R.; Kingan, M.; Fenemore, C.; Schmid, G.; Cockrem, J.F.; Crackle, A.; Van Bemmel, E.; Connor, R.; Descovich, K. Potential impact of construction noise on selected zoo animals. Animals 2019, 9, 504. [Google Scholar] [CrossRef] [PubMed]
- Orban, D.A.; Soltis, J.; Perkins, L. Sound at the zoo: Using animal monitoring, sound measurement, and noise reduction in zoo animal management. Zoo Biol. 2017, 36, 231–236. [Google Scholar] [CrossRef]
- Chosey, J.; Wilson, M.; Santymire, R. Behavioral and Physiological Responses in Felids to Exhibit Construction. Zoo Biol. 2014, 33, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Ijichi, C.L.; Collins, L.M.; Elwood, R.W. Evidence for the role of personality in stereotypy predisposition. Anim. Behav. 2013, 85, 1145–1151. [Google Scholar] [CrossRef]
- Mason, G.J.; Clubb, R.; Latham, N.; Vickery, S. Why and how should we use environmental enrichment to tackle stereotypic behavior. Appl. Anim. Behav. Sci. 2007, 102, 163–188. [Google Scholar] [CrossRef]
- Clubb, R.; Mason, G.J. Natural behavioral biology as a risk factor in carnivore welfare: How analyzing species differences could help zoos improve enclosures. Appl. Anim. Behav. Sci. 2007, 102, 302–328. [Google Scholar] [CrossRef]
- Holmes, L. Chester Zoo Behavior Report, 2017 Impact of Lanterns on Asiatic Lion Behavior. Chester Zoo, North of England Zoological Society, Upton by Chester, UK. Unpublished work, 2017.
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Martin, P.; Bateson, P. Measuring Behavior: An Introductory Guide, 3rd ed.; Cambridge University Press: Cambridge, UK, 1993. [Google Scholar]
- Fanning, L.; Larsen, H.; Taylor, P.S. A Preliminary Study Investigating the Impact of Musical Concerts on the Behavior of Captive Fiordland Penguins (Eudyptes pachyrhynchus) and Collared Peccaries (Pecari tajacu). Animals 2020, 10, 2035. [Google Scholar] [CrossRef]
- Bastian, M.L.; Glendinning, D.R.; Brown, J.L.; Boisseau, N.P.; Edwards, K.L. Effects of a recurring late-night event on the behavior and welfare of a population of zoo-housed gorillas. Zoo Biol. 2020, 39, 217–229. [Google Scholar] [CrossRef] [PubMed]
- Barber, J.C.E. Programmatic approaches to assessing and improving animal welfare in zoos and aquariums. Zoo Biol. 2009, 28, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Dunston, E.J.; Abell, J.; Doyle, R.E.; Evershed, M.; Freire, R. Exploring African lion (Panthera leo) behavioral phenotypes: Individual differences and correlations between sociality, boldness and behavior. J. Ethol. 2016, 34, 277–290. [Google Scholar] [CrossRef]
- Pastorino, G.Q.; Viau, A.; Curone, G.; Pearce-Kelly, P.; Faustini, M.; Vigo, D.; Mazzola, S.M.; Preziosi, R. Role of Personality in Behavioral Responses to New Environments in Captive Asiatic Lions (Panthera leo persica). Vet. Med. Int. 2017, 2017, 6585380. [Google Scholar]
- Goswami, S.; Patel, S.K.; Pandit, S.; Kadivar, R.; Tyagi, P.C.; Malik, P.K.; Mondol, S. Effects of a combined enrichment intervention on the behavioral and physiological welfare of captive Asiatic lions (Panthera leo persica). PeerJ 2020, 8, e8425. [Google Scholar] [CrossRef] [PubMed]
- Clubb, R.; Vickery, S. Locomotory stereotypies in carnivores: Does pacing stem from hunting, ranging, or frustrated escape. In Stereotypic Animal Behaviour: Fundamentals and Applications to Welfare, 2nd ed.; Mason, G.J., Rushen, J., Eds.; Cabi: Wallingford, UK, 2008; pp. 58–79. [Google Scholar]
- Breton, G.; Barrot, S. Influence of enclosure size on the distances covered and paced by captive tigers (Panthera tigris). Appl. Anim. Behav. Sci. 2014, 154, 66–75. [Google Scholar] [CrossRef]
- Bashaw, M.J.; Kelling, A.S.; Bloomsmith, M.A.; Maple, T.L. Environmental Effects on the Behavior of Zoo-housed Lions and Tigers, with a Case Study on the Effects of a Visual Barrier on Pacing. J. Appl. Anim. Welf. Sci. 2007, 10, 95–109. [Google Scholar] [CrossRef] [PubMed]
- Finch, K.; Williams, L.J.; Holmes, L. Using longitudinal data to evaluate the behavioral impact of a switch to carcass feeding on an Asiatic lion Panthera leo persica. JZAR 2020, 8, 283–287. [Google Scholar]
- Rees, P.A. Zoo and exhibit design. In An Introduction to Zoo Biology and Management; Wiley Blackwell: Oxford, UK, 2011; pp. 189–199. [Google Scholar]
- Schaller, G.B. The Serengeti Lion: A Study of Predator-prey Relations; University of Chicago Press: Chicago, IL, USA, 1972; pp. 119–125. [Google Scholar]
- Bitgood, S.; Patterson, D.; Benefield, A. Exhibit design and visitor behavior: Empirical relationships. Environ. Behav. 1988, 20, 474–491. [Google Scholar] [CrossRef]
- Kohari, D.; Sunada, A.; Matsui, Y.; Ootaki, A.; Hori, H. Behavioral restriction effects on activity motivation of a captive lion (Panthera leo persica). J. Vet. Behav. 2017, 17, 14–18. [Google Scholar] [CrossRef]
| Behavioral Category | Behavior | Description |
|---|---|---|
| Natural active | Alert | Highly responsive to stimuli. Looking around or focused in a specific direction. |
| Natural active | Digging | Using paws to displace substrate. |
| Natural active | Feeding | Ingestion of food item or liquid. |
| Natural active | Excretion | Elimination of urine and feces from body. |
| Natural active | Grooming | Self-maintenance, including rolling, stretching, licking, scratching, and biting of own body. |
| Natural active | Licking | Rubbing tongue against object. |
| Natural active | Locomotion | Walking or running from one place to another. Does not include pacing. |
| Natural active | Rubbing | Pressing and moving body against an object. |
| Natural active | Scent marking | Depositing an odorous substance on an object. |
| Natural active | Scratching | Using claws to scratch an object. |
| Natural active | Sniffing | Brief inhalation of object, ground, or air during olfactory investigation. |
| Natural active | Social interaction | Any form of interaction with a conspecific. Includes aggression, rubbing heads, courtship, mating, grooming, submissive behavior, etc. |
| Natural active | Vocalization | Opening mouth and producing sound. May occur while solitary, at a conspecific, or at human(s). |
| Natural active | Yawning | Opening mouth in a yawn. |
| Pacing | Pacing | Walking back and forth in a repetitive, unvarying, sustained pattern. The same complete pattern must be travelled at least three times. |
| Rest | Resting | Lying down, body motionless, note if eyes open or closed. |
| Out of sight | Out of sight | The subject is not visible to the observer. |
| Behavior | Lion | Pre-Construction to During Construction | During Construction to Post-Construction | Pre-Construction to Post-Construction | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SE | Median | IQR | Mean | SE | Median | IQR | Mean | SE | Median | IQR | ||
| Natural active | Male | 16.59 | 2.42 | 12.70 | 15.30 | 14.01 | 2.22 | 10.40 | 19.79 | 17.78 | 2.52 | 12.50 | 22.90 |
| Female 1 | 12.70 | 2.16 | 10.63 | 11.88 | 23.45 | 4.45 | 13.33 | 38.54 | 20.96 | 2.67 | 19.10 | 29.12 | |
| Female 2 | 12.73 | 2.10 | 10.40 | 12.70 | 25.93 | 4.65 | 11.66 | 42.91 | 18.69 | 2.67 | 14.89 | 21.64 | |
| Pacing | Male | 3.93 | 1.73 | 0.00 | 0.00 | 18.39 | 4.32 | 0.00 | 31.87 | 5.90 | 2.02 | 0.00 | 1.40 |
| Female 1 | 4.70 | 2.77 | 0.00 | 0.00 | 0.29 | 0.23 | 0.00 | 0.00 | 2.38 | 1.13 | 0.00 | 0.00 | |
| Female 2 | 6.52 | 2.53 | 0.00 | 0.00 | 10.89 | 3.98 | 0.00 | 1.67 | 4.33 | 2.11 | 0.00 | 0.00 | |
| Rest | Male | 55.52 | 5.09 | 58.12 | 51.87 | 29.84 | 5.50 | 16.10 | 46.00 | 60.50 | 4.93 | 71.25 | 43.62 |
| Female 1 | 55.59 | 5.45 | 57.50 | 58.64 | 43.66 | 5.82 | 41.25 | 78.75 | 63.70 | 4.57 | 64.64 | 47.87 | |
| Female 2 | 54.49 | 5.43 | 53.95 | 62.39 | 28.95 | 5.34 | 14.17 | 47.91 | 51.92 | 5.33 | 49.17 | 59.04 | |
| Out of sight | Male | 23.96 | 5.10 | 6.88 | 39.38 | 37.76 | 6.73 | 17.50 | 93.50 | 15.83 | 4.32 | 4.16 | 17.90 |
| Female 1 | 27.01 | 5.10 | 9.16 | 47.81 | 32.60 | 6.40 | 11.29 | 76.46 | 12.97 | 3.06 | 6.67 | 15.37 | |
| Female 2 | 26.26 | 5.30 | 10.00 | 36.77 | 34.24 | 6.15 | 10.83 | 56.00 | 25.07 | 4.65 | 18.33 | 33.12 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Williams, L.J.; Finch, K.; Agnew, R.; Holmes, L. Effects of Nearby Construction Work on the Behavior of Asiatic Lions (Panthera leo persica). J. Zool. Bot. Gard. 2021, 2, 66-74. https://doi.org/10.3390/jzbg2010005
Williams LJ, Finch K, Agnew R, Holmes L. Effects of Nearby Construction Work on the Behavior of Asiatic Lions (Panthera leo persica). Journal of Zoological and Botanical Gardens. 2021; 2(1):66-74. https://doi.org/10.3390/jzbg2010005
Chicago/Turabian StyleWilliams, Leah J., Katherine Finch, Rose Agnew, and Lisa Holmes. 2021. "Effects of Nearby Construction Work on the Behavior of Asiatic Lions (Panthera leo persica)" Journal of Zoological and Botanical Gardens 2, no. 1: 66-74. https://doi.org/10.3390/jzbg2010005
APA StyleWilliams, L. J., Finch, K., Agnew, R., & Holmes, L. (2021). Effects of Nearby Construction Work on the Behavior of Asiatic Lions (Panthera leo persica). Journal of Zoological and Botanical Gardens, 2(1), 66-74. https://doi.org/10.3390/jzbg2010005








