The Effect of Enrichment Filling and Engagement Time on Regurgitation and Reingestion Behaviour in Three Zoo-Housed Orangutans
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Animals and Site
2.2. Eenrichment Program and Experimental Protocol
2.3. Behavioural Coding
2.4. Data Analysis
2.4.1. Analysis 1
2.4.2. Analysis 2
2.4.3. Analysis 3
3. Results
3.1. Descriptive Statistics for Regurgitation/Reingestion (R/R) Behaviour, Enrichment Engagement, and Eating Time
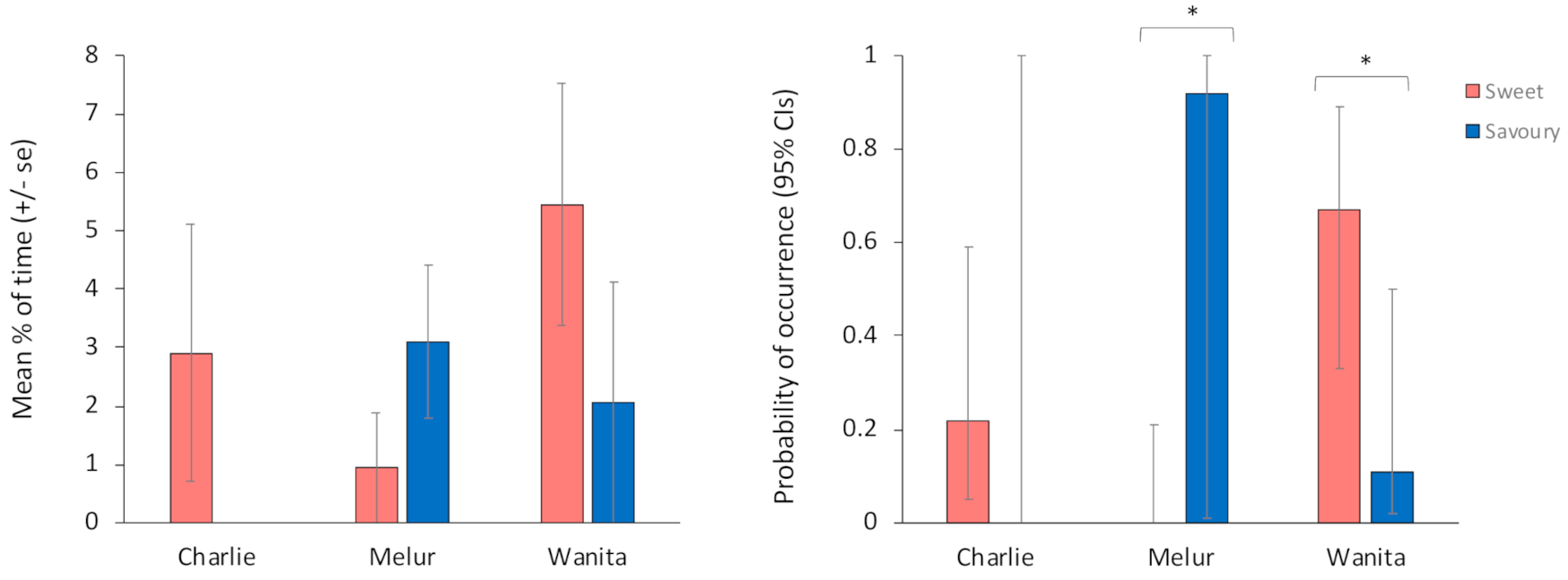
3.2. The Influence of Filling Type, Eating and Engagement Time on the Amount of R/R Behaviour Performed
3.3. The Influence of Filling Type on the Probability of R/R Occurrence
3.4. The Influence of Filling Type on the Amount of Engagement Time
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hill, S.P. Regurgitation and reingestion’ (R/R) in great apes: A review of current knowledge. Int. Zoo Yearbook 2018, 52, 62–78. [Google Scholar] [CrossRef]
- Melfi, V.A. There are big gaps in our knowledge, and thus approach, to zoo animal welfare: A case for evidence-based zoo animal management. Zoo Biol. 2009, 28, 574–588. [Google Scholar] [CrossRef] [PubMed]
- Lukas, K.E. A review of nutritional and motivational factors contributing to the performance of regurgitation and reingestion in captive lowland gorillas (Gorilla gorilla gorilla). Appl. Anim. Behav. Sci. 1999, 63, 237–249. [Google Scholar] [CrossRef]
- Strombeck, D.R. Small Animal Gastroenterology; Stonegate Publishing: Davis, CA, USA, 1979. [Google Scholar]
- Hill, S.P. Do gorillas regurgitate potentially-injurious stomach acid during “regurgitation and reingestion?”. Anim. Welf. 2009, 18, 123–127. [Google Scholar]
- Cooper, J. Gorilla Pathology and Health with a Catalogue of Preserved Materials; Cooper, J.E., Hull, G., Eds.; Academic Press: London, UK, 2017. [Google Scholar]
- Butcher, F.; (Melbourne Zoo, Melbourne, Australia). Personal communication, 2020.
- Baker, K.C.; Easley, S.P. An analysis of regurgitation and reingestion in captive chimpanzees. Appl. Anim. Behav. Sci. 1996, 49, 403–415. [Google Scholar] [CrossRef]
- Baker, K.C. Straw and forage material ameliorate abnormal behaviors in adult chimpanzees. Zoo Biol. 1997, 16, 225–236. [Google Scholar] [CrossRef]
- Herrelko, E.S.; Buchanan-Smith, H.M.; Vick, S.J. Perception of available space during chimpanzee introductions: Number of accessible areas is more important than enclosure size. Zoo Biol. 2015, 34, 397–405. [Google Scholar] [CrossRef]
- Cassella, C.M.; Mills, A.; Lukas, K.E. Prevalence of regurgitation and reingestion in orangutans housed in North American zoos and an examination of factors influencing its occurrence in a single group of bornean orangutans. Zoo Biol. 2012, 31, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.J.; Tobey, J.R. Regurgitation and reingestion in bonobos (Pan paniscus): Relationships between abnormal and social behavior. Appl. Anim. Behav. Sci. 2012, 141, 65–70. [Google Scholar] [CrossRef]
- Lukas, K.E.; Hamor, G.; Bloomsmith, M.A.; Horton, C.L.; Maple, T.L. Removing milk from captive gorilla diets: The impact on regurgitation and reingestion (R/R) and other behaviors. Zoo Biol. 1999, 18, 515–528. [Google Scholar] [CrossRef]
- Fuller, G.; Murray, A.; Thueme, M.; McGuire, M.; Vonk, J.; Allard, S. Behavioral and hormonal responses to the availability of forage material in Western lowland gorillas (Gorilla gorilla gorilla). Zoo Biol. 2018, 37, 23–34. [Google Scholar] [CrossRef]
- Ting, C.Y. Orangutan Behaviour in Captivity: Activity Budgets, Enclosure Use and the Visitor Effect. Master’s Thesis, National University of Singapore, Singapore, 2011. [Google Scholar]
- Birkett, L.P.; Newton-Fisher, N.E. How abnormal is the behaviour of captive, zoo-living chimpanzees? PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Gould, E.; Bres, M. Regurgitation in gorillas: Possible model for human eating disorders (Rumination/bulimia). Dev. Behav. Pediatrics 1986, 7, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Pizzutto, C.S.; Sgai, M.G.F.G.; Lopes, D.A.; Pessutti, C.; Nunes, A.; Furtado, P.V.; de Oliveira, C.A.; Guimarães, M.A. Relation between the level of self-mutilation and the concentration of fecal metabolites of glucocorticoids in captive chimpanzees (Pan troglodytes). Wildl. Med. 2015, 35, 62–66. [Google Scholar]
- Baker, K.C. Benefits of positive human interaction for socially housed chimpanzees. Anim. Welf. 2004, 13, 239–245. [Google Scholar] [PubMed]
- Monagas, J.; Ritwik, P.; Kolomensky, A.; Acosta, J.; Kay, D.; Clendaniel, L.; Hyman, P.E. Rumination syndrome and dental erosions in children. J. Pediatric Gastroenterol. Nutr. 2017, 64, 930–932. [Google Scholar] [CrossRef] [PubMed]
- Masi, S.; Cipolletta, C.; Robbins, M.M. Western lowland gorillas (Gorilla gorilla gorilla) change their activity patterns in response to frugivory. Am. J. Primatol. 2009, 71, 91–100. [Google Scholar] [CrossRef]
- Wich, S.A.; Atmoko, U.S.U.; Setia, T.M.; van Schaik, C.P. Orangutans: Geographic Variation in Behavioral Ecology and Conservation; Oxford University Press: Oxford, UK, 2009. [Google Scholar]
- Struck, K.; Videan, E.N.; Fritz, J.; Murphy, J. Attempting to reduce regurgitation and reingestion in a captive chimpanzee through increased feeding opportunities: A case study. Lab. Anim. 2007, 36, 35–38. [Google Scholar] [CrossRef]
- Hill, S.P. Behavioural and Physiological Investigations of Welfare in Captive Western Lowland Gorillas (Gorilla gorilla gorilla). Ph.D. Thesis, Cambridge University, Cambridge, UK, 2004. [Google Scholar]
- Morrogh-Bernard, H.C.; Husson, S.J.; Knott, C.D.; Wich, S.A.; van Schaik, C.P.; van Noordwijk, M.A.; Lackman-Ancrenaz, I.; Marshall, A.J.; Kanamori, T.; Kuze, N.; et al. Orangutan activity budgets and diet: A comparison between species, populations and habitats. In Orangutans: Geographic Variation in Behavioral Ecology and Conservation; Wich, S.A., Utami Atmoko, S.S., Setia, T.M., van Schaik, C.P., Eds.; Oxford University Press: Oxford, UK, 2008. [Google Scholar]
- Jaeggi, A.V.; Dunkel, L.P.; van Noordwijk, M.A.; Wich, S.A.; Sura, A.A.L.; van Schaik, C.P. Social learning of diet and foraging skills by wild immature Bornean orangutans: Implications for culture. Am. J. Primatol. 2010, 72, 62–71. [Google Scholar] [CrossRef]
- Cabana, F.; Jasmi, R.; Maguire, R. Great ape nutrition: Low-sugar and high-fibre diets can lead to increased natural behaviours, decreased regurgitation and reingestion, and reversal of prediabetes. Int. Zoo Yearb. 2018, 52, 48–61. [Google Scholar] [CrossRef]
- Birke, L. Effects of browse, human visitors and noise on the behaviour of captive orang utans. Anim Welf. 2002, 11, 189–202. [Google Scholar]
- Less, E.H.; Lukas, K.E.; Bergl, R.; Ball, R.; Kuhar, C.W.; Lavin, S.R.; Raghanti, M.A.; Wensvoort, J.; Willis, M.A.; Dennis, P.M. Implementing a low-starch biscuit-free diet in zoo gorillas: The impact on health. Zoo Biol. 2014, 33, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Seiffert, S. Ten years of browse provision at ZSL London Zoo and ZSL Whipsnade Zoo: What have we learned and where are we going? In Proceedings of the 10th European Zoo Nutrition Conference, Marwell Zoo, Winchester, UK, 17–20 January 2019. [Google Scholar]
- Kim-McCormack, N.N.E.; Smith, C.L.; Behie, A.M. Is interactive technology a relevant and effective enrichment for captive great apes? Appl. Anim. Behav. Sci. 2016, 185, 1–8. [Google Scholar] [CrossRef]
- Mellen, J.; Macphee, M.S. Philosophy of Environmental Enrichment: Past, Present, and Future. Zoo Biol. 2001, 226, 211–226. [Google Scholar] [CrossRef]
- Maple, T.; Perdue, B. Environmental enrichment. In Zoo Animal Welfare; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Robbins, A.; (Auckland Zoo, Auckland, New Zealand). Personal communication, 2016.
- Pastell, P. Cowlog—Cross-platform application for coding behaviours from video. J. Open Res. Softw. 2016, 4. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Lenth, R. Emmeans: Estimated Marginal Means, aka Least-Squares Means. Available online: https://CRAN.R-project.org/package=emmeans (accessed on 15 October 2020).
- Clark, F.E.; Gray, S.I.; Bennett, P.; Mason, L.J.; Burgess, K.V. High-tech and tactile: Cognitive enrichment for zoo-housed gorillas. Front. Psychol. 2019, 10, 1–15. [Google Scholar] [CrossRef]
- Egelkamp, C.L.; Ross, S.R. A review of zoo-based cognitive research using touchscreen interfaces. Zoo Biol. 2019, 38, 220–235. [Google Scholar] [CrossRef]
- Perdue, B.M.; Clay, A.W.; Gaalema, D.E.; Maple, T.L.; Stoinski, T.S. Technology at the zoo: The influence of a touchscreen computer on orangutans and zoo visitors. Zoo Biol. 2012, 31, 27–39. [Google Scholar] [CrossRef]
- Yamanashi, Y.; Hayashi, M. Assessing the effects of cognitive experiments on the welfare of captive chimpanzees (Pan troglodytes) by direct comparison of activity budget between wild and captive chimpanzees. Am. J. Primatol. 2011, 73, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Clark, F. Cognitive enrichment and welfare: Current approaches and future directions. Anim. Behav. Cogn. 2017, 4, 52–71. [Google Scholar] [CrossRef] [Green Version]
- Chapman, C.A.; Lambert, J.E.; Rothman, J.M. Primate foraging strategies and nutrition: Behavioural and evolutionary implications. In The Evolution of Primate Societies; University of Chicago Press: Chicago, IL, USA, 2012; pp. 145–167. [Google Scholar]
- Campbell, C.L.; Wagoner, T.B.; Foegeding, E.A. Designing foods for satiety: The roles of food structure and oral processing in satiation and satiety. Food Struct. 2017, 13, 1–12. [Google Scholar] [CrossRef]
- Andrew, G.; Large, M.; Adams, N.; Robbins, A. Dietary browse reduces frequency of regurgitation and reingestion events in captive bornean orang-utans (Pongo pygmaeus). Unpublished work. 2020. [Google Scholar]
- Foster, K.R.; Kokko, H. The evolution of superstitious and superstition-like behaviour. Proc. R. Soc. B Biol. Sci. 2008, 31–37. [Google Scholar] [CrossRef]
- Fernandez, E.J.; Timberlake, W. Superstition revisited: Sex, species, and adventitious reinforcement. Behav. Process. 2020, 170. [Google Scholar] [CrossRef]
- Verspeek, J.; Stevens, J.M.G. Food preference and nutrient composition in captive bonobos (Pan paniscus). Primates 2020, 61. [Google Scholar] [CrossRef]
- Mason, G.; Farland, D.M.C.; Garner, J. A demanding task: Using economic techniques to assess animal priorities. Anim. Behav. 1998, 55, 1071–1075. [Google Scholar] [CrossRef] [Green Version]
- Hill, S.P.; Broom, D.M. Measuring zoo animal welfare: Theory and practice. Zoo Biol. 2009, 28, 531–544. [Google Scholar] [CrossRef]
- Many Primates; Altschul, D.M.; Beran, M.J.; Bohn, M.; Call, J.; Detroy, S.; Duguid, S.J.; Egelkamp, C.L.; Fichtel, C.; Fischer, J.; et al. Establishing an infrastructure for collaboration in primate cognition research. PLoS ONE 2019, 14, e0223675. [Google Scholar]
| Enrichment Item | Treatment | Treatment Filling |
|---|---|---|
| Large boxes | SW | Popcorn, raisins, caraway seeds, and ginger |
| SA | Popcorn, peanuts, Chinese five spice, and caraway seeds | |
| Firehose boxes | SW | Popcorn, prunes, sesame seeds and mixed spice |
| SA | Popcorn, peanuts, poppy seeds, and lemon pepper | |
| Firehose with holes | SW | Jam and sesame seeds |
| SA | Cooked lentils and fennel seeds | |
| Clothing shapes and hose pipes | SW | Popcorn, chopped dates, cinnamon, rosemary, jam, and fennel seeds |
| SA | Popcorn, peanuts, cinnamon, cooked lentils, and sesame seeds | |
| Sonar tubes | SW | Popcorn, chopped apricot, fennel seeds, and peppermint essence |
| SA | Popcorn, peanuts, mixed herbs, and poppy seeds | |
| Large iceys | SW | Diluted juice and frozen strawberries |
| SA | Choysa brown tea, lemons, and ground cloves | |
| Sacks | SW | Popcorn, mixed spice, dates, and mixed seeds |
| SA | Popcorn, peanuts, sesame seeds, and curry powder | |
| Logs | SW | Cooked rice, stewed quince |
| SA | Cooked rice, mixed vegetables, and soy sauce | |
| Noodle pinecones | SW | Cooked vermicelli, and jam |
| SA | Cooked vermicelli, and fresh chives |
| Behaviour | Description |
|---|---|
| Regurgitation and reingestion | While sitting, the individual’s shoulders are hunched and heaving until vomitus material has been brought up which is then reingested. This behaviour may be very subtle, and for Melur this was often accompanied or preceded by shoulder slapping or face rubbing. |
| Enrichment interaction/engagement | Individual is engaged with or touching the enrichment item but is not eating/foraging |
| Eating/foraging enrichment interaction | Individual is eating food obtained from the enrichment item. |
| Out of view | Individual is not in view of the camera or the behaviour being performed is not clear. |
| Other | Individual is performing any other behaviour that is not included in the catalogue. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nash, R.; Johnston, H.; Robbins, A.; Descovich, K. The Effect of Enrichment Filling and Engagement Time on Regurgitation and Reingestion Behaviour in Three Zoo-Housed Orangutans. J. Zool. Bot. Gard. 2021, 2, 10-20. https://doi.org/10.3390/jzbg2010002
Nash R, Johnston H, Robbins A, Descovich K. The Effect of Enrichment Filling and Engagement Time on Regurgitation and Reingestion Behaviour in Three Zoo-Housed Orangutans. Journal of Zoological and Botanical Gardens. 2021; 2(1):10-20. https://doi.org/10.3390/jzbg2010002
Chicago/Turabian StyleNash, Rebecca, Helen Johnston, Amy Robbins, and Kris Descovich. 2021. "The Effect of Enrichment Filling and Engagement Time on Regurgitation and Reingestion Behaviour in Three Zoo-Housed Orangutans" Journal of Zoological and Botanical Gardens 2, no. 1: 10-20. https://doi.org/10.3390/jzbg2010002
APA StyleNash, R., Johnston, H., Robbins, A., & Descovich, K. (2021). The Effect of Enrichment Filling and Engagement Time on Regurgitation and Reingestion Behaviour in Three Zoo-Housed Orangutans. Journal of Zoological and Botanical Gardens, 2(1), 10-20. https://doi.org/10.3390/jzbg2010002







