Nanografting of Polymer Brushes on Gold Substrate by RAFT-RIGP †
Abstract
:1. Introduction
2. Materials and Methods
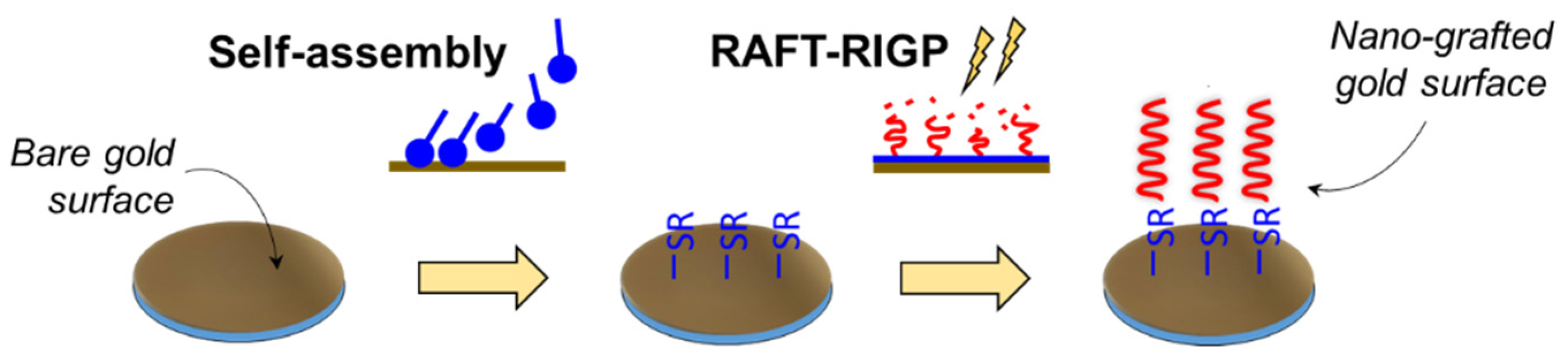
2.1. Formation of Self-Assembled Monolayer (SAM)
2.2. RAFT-RIGP Adiation Grafting of Abaca Fibers
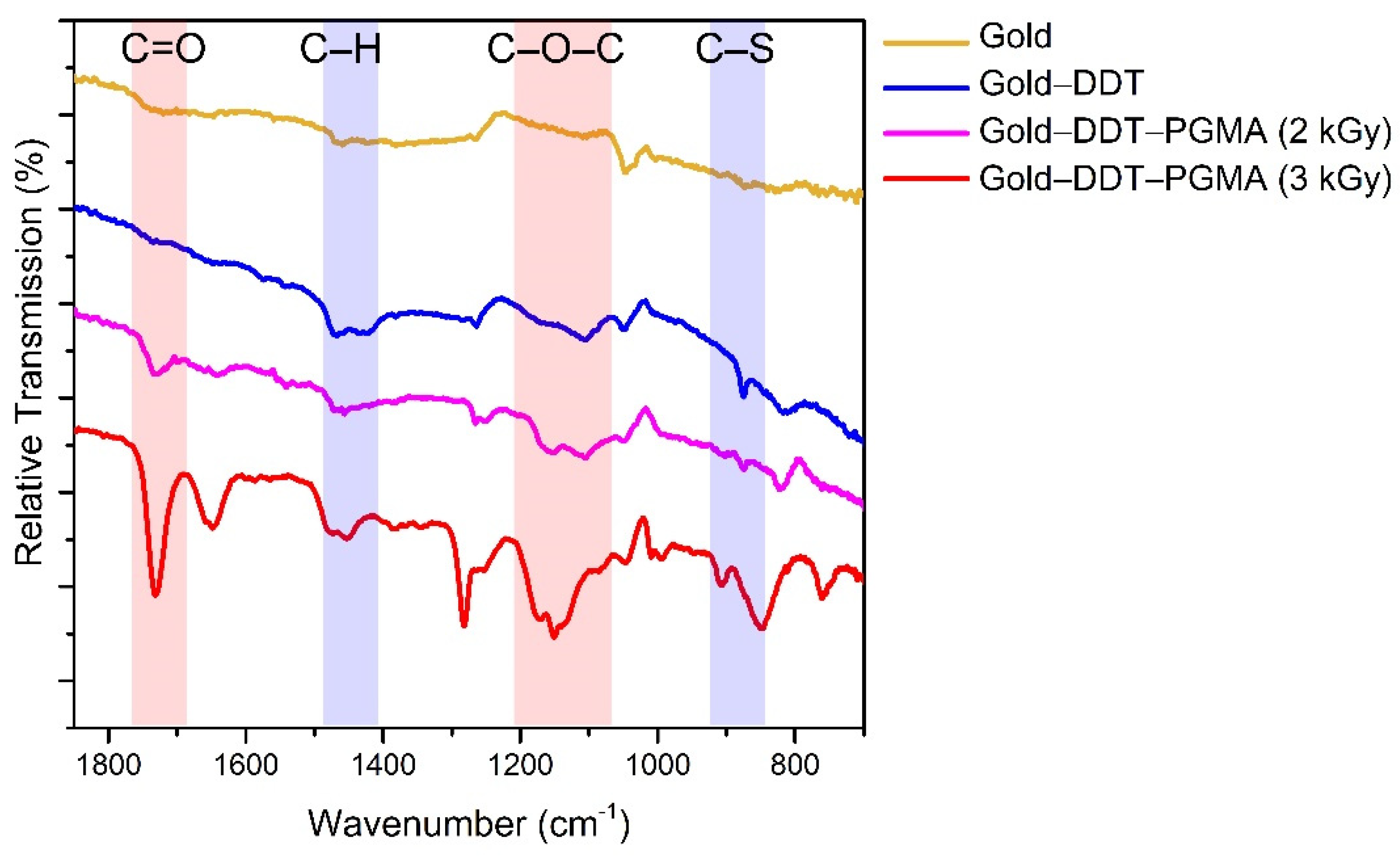
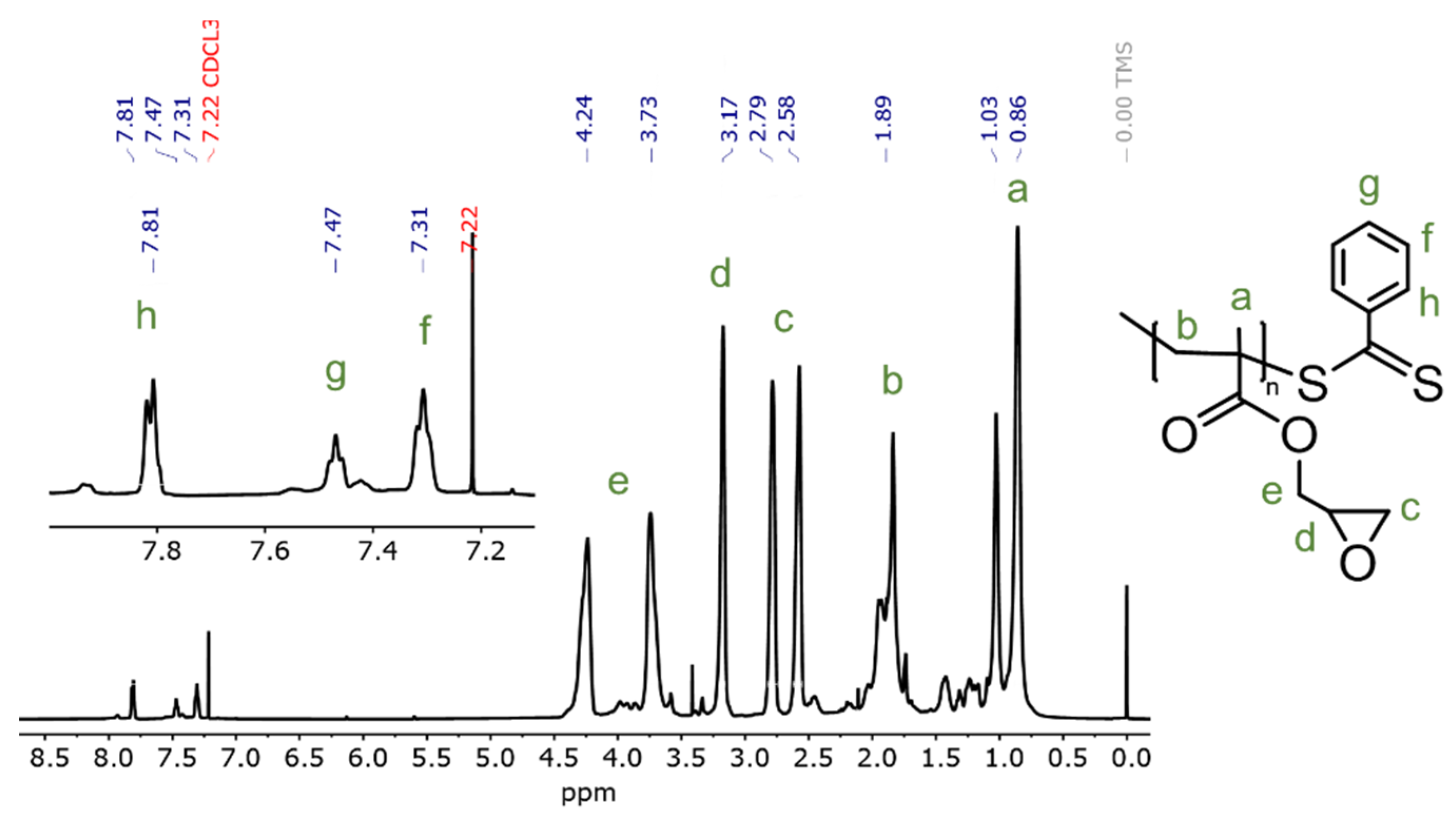
2.3. Characterization
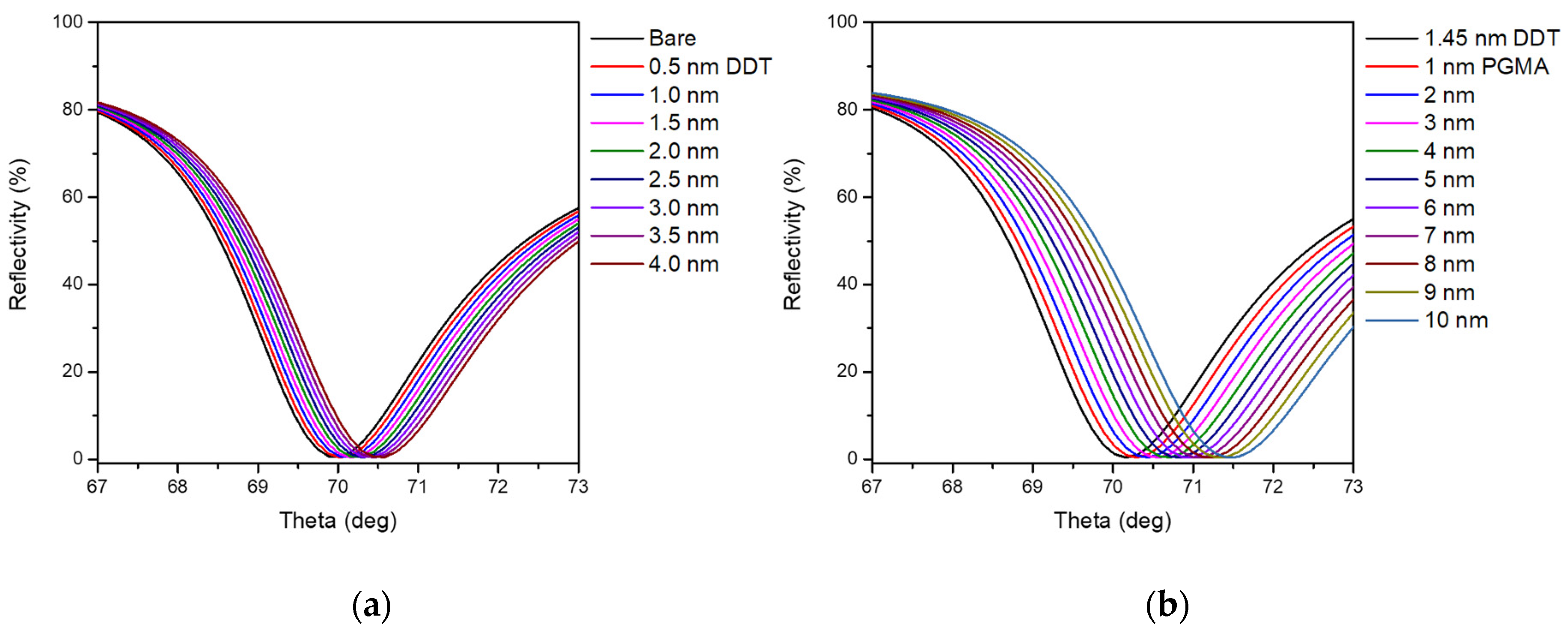
2.4. Simulation and Fitting of SPR Data
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Homola, J.; Yee, S.S.; Gauglitz, G. Surface plasmon resonance sensors: Review. Sens. Actuators B Chem. 1999, 54, 3–15. [Google Scholar] [CrossRef]
- Wijaya, E.; Lenaerts, C.; Maricot, S.; Hastanin, J.; Habraken, S.; Vilcot, J.P.; Boukherroub, R.; Szunerits, S. Surface plasmon resonance-based biosensors: From the development of different SPR structures to novel surface functionalization strategies. Curr. Opin. Solid State Mater. Sci. 2011, 15, 208–224. [Google Scholar] [CrossRef]
- Vosloo, J.J.; De Wet-Roos, D.; Tonge, M.P.; Sanderson, R.D. Controlled free radical polymerization in water-borne dispersion using reversible addition-fragmentation chain transfer. Macromolecules 2002, 35, 4894–4902. [Google Scholar] [CrossRef]
- Grasselli, M.; Betz, N. Electron-beam induced RAFT-graft polymerization of poly (acrylic acid) onto PVDF. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms 2005, 236, 201–207. [Google Scholar] [CrossRef]
- Matyjaszewski, K.; Spanswick, J. Controlled/living radical polymerization. Mater. Today 2005, 8, 26–33. [Google Scholar] [CrossRef]
- Barsbay, M.; Güven, O. RAFT mediated grafting of poly (acrylic acid) (PAA) from polyethylene/polypropylene (PE/PP) nonwoven fabric via preirradiation. Polymer 2013, 54, 4838–4848. [Google Scholar] [CrossRef]
- Barsbay, M.; Güven, O. A short review of radiation-induced raft-mediated graft copolymerization: A powerful combination for modifying the surface properties of polymers in a controlled manner. Radiat. Phys. Chem. 2009, 78, 1054–1059. [Google Scholar] [CrossRef]
- Barsbay, M.; Kodama, Y.; Güven, O. Functionalization of cellulose with epoxy groups via γ-initiated RAFT-mediated grafting of glycidyl methacrylate. Cellulose 2014, 21, 4067–4079. [Google Scholar] [CrossRef]
- Barsbay, M.; Güven, O.; Stenzel, M.H.; Davis, T.P.; Barner-Kowollik, C.; Barner, L. Verification of controlled grafting of styrene from cellulose via radiation-induced RAFT polymerization. Macromolecules 2007, 40, 7140–7147. [Google Scholar] [CrossRef] [Green Version]
- Madrid, J.F.; Ueki, Y.; Abad, L.V.; Yamanobe, T.; Seko, N. RAFT-mediated graft polymerization of glycidyl methacrylate in emulsion from polyethylene/polypropylene initiated with γ-radiation. J. Appl. Polym. Sci. 2017, 134, 45270. [Google Scholar] [CrossRef]
- Mitsushio, M.; Masunaga, T.; Higo, M. Theoretical considerations on the responses of gold-deposited surface plasmon resonance-based glass rod sensors using a three-layer Fresnel equation. Plasmonics 2014, 9, 451–459. [Google Scholar] [CrossRef]
- Res-Tec Surface Plasmon Resonance—The RES-TEC Quick Datasheet. Available online: http://res-tec.de/pdf/res-tec-optical-constants-database.pdf (accessed on 10 September 2021).
- Johnson, P.B.; Christy, R.W. Optical Constants of the Noble Metals. Phys. Rev. B 1972, 6, 4370. [Google Scholar] [CrossRef]
- Sigma-Aldrich 1-Dodecanethiol. Available online: https://www.sigmaaldrich.com/PH/en/product/aldrich/471364 (accessed on 10 September 2021).
- Sigma-Aldrich Glycidyl Methacrylate. Available online: https://www.sigmaaldrich.com/PH/en/product/aldrich/779342 (accessed on 10 September 2021).
- Guo, Q.; Li, F. Self-assembled alkanethiol monolayers on gold surfaces: Resolving the complex structure at the interface by STM. Phys. Chem. Chem. Phys. 2014, 16, 19074–19090. [Google Scholar] [CrossRef]
- Sharma, A.; Singh, B.P.; Gathania, A.K. Synthesis and characterization of dodecanethiol-stabilized gold nanoparticles. Indian J. Pure Appl. Phys. 2014, 52, 93–100. [Google Scholar]
- Chiefari, J.; Chong, Y.K.; Ercole, F.; Krstina, J.; Jeffery, J.; Le, T.P.T.; Mayadunne, R.T.A.; Meijs, G.F.; Moad, C.L.; Moad, G. Living free-radical polymerization by reversible addition−fragmentation chain transfer: The RAFT process. Macromolecules 1998, 31, 5559–5562. [Google Scholar] [CrossRef]
- Sharif, J.; Mohamad, S.F.; Fatimah Othman, N.A.; Bakaruddin, N.A.; Osman, H.N.; Güven, O. Graft copolymerization of glycidyl methacrylate onto delignified kenaf fibers through pre-irradiation technique. Radiat. Phys. Chem. 2013, 91, 125–131. [Google Scholar] [CrossRef]
- Perrier, S. 50th Anniversary Perspective: RAFT Polymerization—A User Guide. Macromolecules 2017, 50, 7433–7447. [Google Scholar] [CrossRef]
- Knoll, W. Interfaces and thin films as seen by bound electromagnetic waves. Annu. Rev. Phys. Chem. 1998, 49, 569–638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, G.; Deng, S.; Baba, A.; Huang, C.; Advincula, R.C. On the Monolayer Adsorption of Thiol-Terminated Dendritic Oligothiophenes onto Gold Surfaces. Macromol. Chem. Phys. 2010, 211, 2562–2572. [Google Scholar] [CrossRef]
- Godin, M.; Williams, P.J.; Tabard-Cossa, V.; Laroche, O.; Beaulieu, L.Y.; Lennox, R.B.; Grütter, P. Surface stress, kinetics, and structure of alkanethiol self-assembled monolayers. Langmuir 2004, 20, 7090–7096. [Google Scholar] [CrossRef] [Green Version]
- Reed, K.M.; Borovicka, J.; Horozov, T.S.; Paunov, V.N.; Thompson, K.L.; Walsh, A.; Armes, S.P. Adsorption of sterically stabilized latex particles at liquid surfaces: Effects of steric stabilizer surface coverage, particle size, and chain length on particle wettability. Langmuir 2012, 28, 7291–7298. [Google Scholar] [CrossRef] [PubMed]
| Layer | Thickness (nm) | Refractive Index |
|---|---|---|
| BK-7 prism | 0 | 1.5151 [12] |
| Gold | 50 | 0.1377 [13] |
| DDT | 0–4.0 | 1.4590 [14] |
| PGMA | 0–10 | 1.4490 [15] |
| Water | 0 | 1.3334 [12] |
| Dose, kGy | Conversion, % | Molecular Weight, kDa | |
|---|---|---|---|
| Calculated * | NMR | ||
| 2 | 62 | 2.3 | 2.8 |
| 3 | 92 | 3.3 | 4.3 |
| Layer | Relative Resonance Angle (°) | Calculated Thickness (nm) | Theoretical Thickness (nm) |
|---|---|---|---|
| Gold | 1.93 ± 0.33 | n.a. | n.a. |
| DDT | 2.13 ± 0.33 | 1.45 ± 0.18 | 1.7 a |
| PGMA (2 kGy) | 2.63 ± 0.30 | 4.25 ± 0.69 | 4.6 b |
| PGMA (3 kGy) | 3.18 ± 0.39 | 8.10 ± 0.43 | 6.9 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barba, B.J.D.; Heruela, P.N.L.; Cabalar, P.J.E.; Luna, J.A.A.; Yago, A.C.C.; Madrid, J.F. Nanografting of Polymer Brushes on Gold Substrate by RAFT-RIGP. Mater. Proc. 2021, 7, 5. https://doi.org/10.3390/IOCPS2021-11587
Barba BJD, Heruela PNL, Cabalar PJE, Luna JAA, Yago ACC, Madrid JF. Nanografting of Polymer Brushes on Gold Substrate by RAFT-RIGP. Materials Proceedings. 2021; 7(1):5. https://doi.org/10.3390/IOCPS2021-11587
Chicago/Turabian StyleBarba, Bin Jeremiah D., Patricia Nyn L. Heruela, Patrick Jay E. Cabalar, John Andrew A. Luna, Allan Christopher C. Yago, and Jordan F. Madrid. 2021. "Nanografting of Polymer Brushes on Gold Substrate by RAFT-RIGP" Materials Proceedings 7, no. 1: 5. https://doi.org/10.3390/IOCPS2021-11587
APA StyleBarba, B. J. D., Heruela, P. N. L., Cabalar, P. J. E., Luna, J. A. A., Yago, A. C. C., & Madrid, J. F. (2021). Nanografting of Polymer Brushes on Gold Substrate by RAFT-RIGP. Materials Proceedings, 7(1), 5. https://doi.org/10.3390/IOCPS2021-11587






