1. Introduction
At present, the main mineral source for MgO and Mg(OH)2 products is MgCO3. The evaluation of molar masses shows that the production of one ton of MgO from MgCO3 directly results in the release of 1.09 tons of CO2 into the atmosphere, not counting the CO2 released due to process heating. If MgO is produced from silicate sources, the liberation of carbonate-fixed CO2 can be avoided.
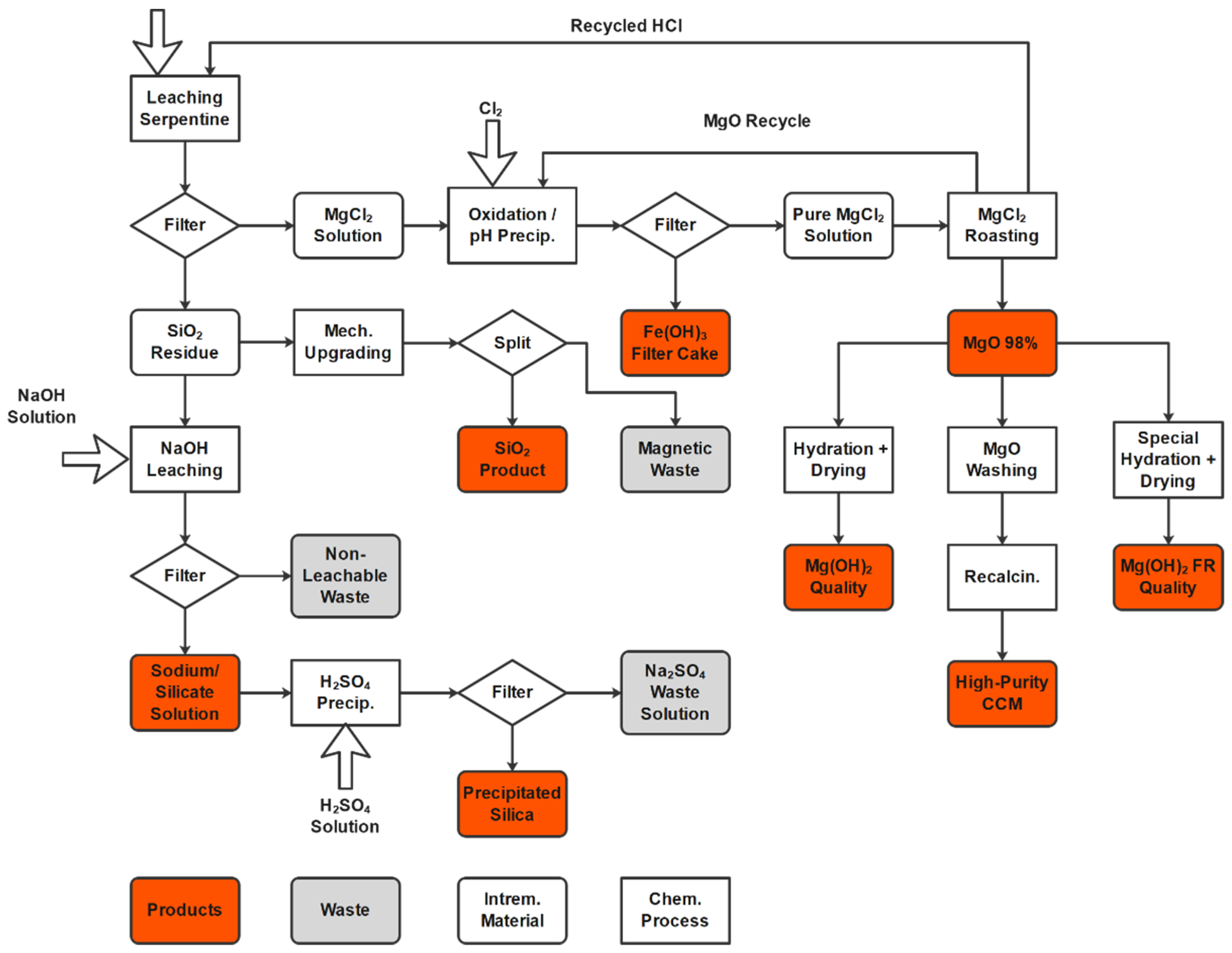
This paper presents a HCl-based zero-waste process route for serpentine ores that will produce SiO2, water glass, precipitated silica, MgO, high-purity MgO, Mg(OH)2, and an iron hydroxide fraction under carbon-neutral conditions.
The process starts with a HCl leaching stage, where the dissolvable components, such as Mg and Fe, are separated from the non-dissolvable silica fraction.
The silica generated can be mechanically upgraded to a basic-grade silica fraction. Alternatively, a caustic leaching procedure can be employed, using NaOH to dissolve the amorphous SiO2 component and generate water glass with a high ratio of silica to sodium. An additional acid treatment will convert the water glass to precipitated silica.
The liquid phase that results after the HCl leaching process is purified by the adjustment of the pH. Iron and other elements precipitate as hydroxides, leaving a pure MgCl2 solution. The filter cake, which mainly consists of iron hydroxides, can be used as a source of iron for a steel mill.
The pure MgCl
2 solution undergoes a spray roasting process that produces technical-grade MgO as well as HCl (which can be reused in the leaching process), as per the following equation:
Highly purified caustic calcined magnesia (CCM) can be produced if the technical-grade MgO is further washed and calcined. Alternatively, a hydration procedure can be applied to produce Mg(OH)2 at different, market-specific quality levels.
Looking at the entire process as a whole, it can be stated that spray roasting is the second-largest producer of CO2. If new, non-carbon-emitting energy sources can be used in this stage, such as direct electrical heating or hydrogen burner systems, this would represent a significant step toward the CO2-free production of MgO.
Figure 1 shows a block flow diagram of the described MgO production process, where serpentine is used as a feedstock.
2. Materials and Methods
2.1. Industrial Style Process Flow Diagram
A commercial application of the conceptual carbon-neutral process is shown in the process flow diagram (PFD) in
Figure 2. The process comprises unit operations currently offered by KON Chemical Solutions and Tenova and incorporates public-domain knowledge gained from decades of process development [
1,
2,
3,
4]. It includes the commercially established unit operation for producing high-quality synthetic magnesium oxide (MgO). If the process includes a carbon-neutral heating system, it will constitute the first industrial-scale carbon-neutral process for the production of MgO. There is also the potential for the minimization of waste by commercializing by-products such as silica [
5]. The process for this is described below.
2.1.1. Leaching
In the leaching section, the raw material (serpentine) is brought into contact with regenerated acid at a temperature of about 80 °C and a HCl strength of about 18%m/m. The leaching operation has two stages, with the first stage controlled to a specific amount of excess HCl to maximize the leaching efficiency of the raw material. The undissolved fraction, mainly silica, is separated by a filtration step and the liquid part, containing MgCl2 and other dissolvable elements, is transferred to the second leaching stage. In this step, an additional amount of fine raw material is added to consume the free HCl. The slurry is now brought to a second filter stage, where the solid fraction is separated and put back to the first leaching stage to finalize the leaching procedure. The liquid phase is transferred to the next unit of the operation for oxidation and precipitation.
2.1.2. Oxidation and Precipitation
The impure MgCl
2 solution gained from leaching is purified by oxidation and precipitation. Firstly, Cl
2 gas is used to oxidize the Fe
2+ in the solution to Fe
3+ according to Equation (2).
Pure MgO originating from further down the process is then used to raise the pH value of the solution and precipitate Fe, Ni, and Al as hydroxides. The hydroxides are removed via filtration, while MgCl2 and CaCl2 remain as chlorides in dissolved form.
2.1.3. Tank Farm
The tank farm is a buffer unit that creates separation between single process steps at an industrial scale. Buffer volumes are important to the maintenance of continuous operation and ensure that a small interruption at a single unit will not stop the whole plant.
2.1.4. MgO Roasting and HCl Recovery
Roasting is the heart of the process. Purified MgCl2 solution is added to a venturi circuit where the preconcentration of the solution and the cooling of the hot roast gases take place. The concentrated and purified solution is then sprayed into the reactor, where it is transformed into solid MgO and gaseous HCl. The MgO falls to the bottom of the reactor, where it is extracted as a primary product. The hot gaseous HCl leaves the reactor out of the top. It is cleaned, quenched, and finally absorbed in water to generate an aqueous acid that is recycled back to the leaching unit operation.
The groundbreaking new approach within this conceptual process is the use of a carbon-neutral heating source to achieve carbon-neutral production. A flame temperature of more than 1300 °C provides the energy input required to run the system at reasonable volume flows. Natural gas heating is depicted in
Figure 2. This can be replaced by a carbon-neutral system, such as hydrogen burners or plasma. In the mass and energy balance model considered later in this article, plasma is used to heat air to 1300 °C to facilitate the roasting process.
2.1.5. Silica Treatment
As serpentine has a high silica content, the commercialization of this component is important in the development of a sustainable commercial approach to the production of magnesia. The filter cake generated at the leaching stage can be treated with sodium hydroxide to produce a water glass solution. Depending on the specific raw material used, it could be sufficient to use mild leaching conditions below 100 °C and an ambient pressure. The non-dissolved solid fraction can be separated from the pure water glass, which can then be sold as a product. Alternatively, a second processing step can be introduced, using H2SO4 or CO2 to convert the water glass to precipitated silica. The production of a salable silica by-product would make MgO production a near-zero waste process.
2.2. Mass and Energy Balance
A mass and energy balance model of the process from raw material leaching to MgO roasting was built using the Outotec HSC software. The output from the model is summarized in
Figure 3.
The assumed 8.75 t/h of raw material input leads to an annual plant capacity of 70,000 t material input and a theoretical MgO production of 28,000 t MgO (MgO roasted—pH MgO) for 8000 operation hours.
3. Results
West High Yield (WHY) Resources Ltd., a company in Calgary, Alberta, Canada, that engages in the acquisition, exploration, and development of mineral resource properties is currently developing a magnesia project using serpentine material extracted from their Record Ridge mine. The chemical composition of the serpentine is summarized in
Table 1. This composition was also considered in the mass and energy balance model shown in
Figure 3.
Preliminary tests were carried out at Kingston Process Metallurgy Inc. (KPM), located in Kingston, Ontario, Canada, to evaluate the basic material behavior at the kilogram scale. The test program was engineered to generate fundamental chemical data for the proposed process concept.
The HCl leaching was carried out at moderate temperatures at an ambient pressure using 18%m/m of HCl Solution. Repeat testing led to an average Mg recovery rate of 91.7%m/m.
The oxidation step was executed with Cl2 gas and the Fe2+ concentration was reduced from a starting point of 4.8 g/L down to 0.1 g/L.
Using MgO for the pH adjustment and subsequent filtration of the precipitated hydroxides, it was possible to generate a MgCl2 solution with a purity of 98.9%m/m, with the main contaminant being Ca.
The purified solution was then thermally treated to generate a solid particulate MgO of technical-grade purity (i.e., >98%m/m).
The technical-grade MgO was then washed and recalcined to produce a high-purity (99.87%m/m) MgO, equivalent to high-purity caustic calcined MgO (CCM).
Preliminary NaOH leaching and H2SO4 precipitation tests were also conducted on the silica leach residue. A reasonable leach efficiency of 82%m/m was achieved under mild leaching conditions and the precipitated silica product had a purity of 99.5%m/m SiO2.
4. Discussion
This paper presents a process route using non-carbonate magnesium ores for the production of synthetic MgO. The process includes treatment options that commercialize the silica component of the ore. With the use of carbon-neutral heating, the process opens the door to a novel means of MgO production with a lower ecological footprint.
Building on the successful test work conducted at KPM, a pilot-scale demonstration is the logical next step in the development of the process ahead of commercialization. Key parts of the process have already been demonstrated at a commercial scale. In 2012, Tenova built an industrial-scale pilot plant for the MgCl
2 roasting operation. This was the first MgO roaster system built and operated in the West in approximately thirty years.
Figure 4a shows a 3D model developed during the design of the plant and
Figure 4b shows the completed unit.
5. Conclusions/Outlook
The technological concepts for the carbon-neutral production of MgO are ready for industrial implementation. The preliminary testing of aspects of the concept using ore from the WHY serpentine deposit gave very promising results. The continued development of the concept with WHY in Canada, the evaluation of other non-carbonate magnesia sources, and the further rollout of this technology should lead to the more sustainable production of MgO in the future.
Author Contributions
Conceptualization, D.K. and G.K.; methodology, D.K.; validation, D.K.; investigation, D.K.; writing—original draft preparation, D.K.; writing—review and editing, G.K.; visualization, D.K. and G.K.; supervision, G.K. All authors have read and agreed to the published version of the manuscript.
Acknowledgments
We are very grateful for the assistance given by West High Yield Resources Ltd. (P.O. Box 68121, Calgary, Alberta, Canada T3G 3N8) in allowing us to use the specific data and results of testing of materials taken from the property under development at Record Ridge. We also wish to acknowledge the help given by the team at Kingston Process Metallurgy Inc. (759 Progress Avenue Kingston, Ontario, Canada K7M 6N6) in executing the practical bench-scale lab tests for WHY, in cooperation with the remote supervision of KON Chem.
Conflicts of Interest
The authors advise that their company offers and distributes the described technology on the world market. The figures and information given have been compiled to the best of their knowledge and belief, but assumptions have also been made based on existing experience. No guarantees or obligations are given by the authors relating to this technological summary.
References
- Metallgesellschaft, A. Gewinnung von Magnesiumoxyd aus Wasserhaltigen Magnesiumchloridlösungen Oder Schmelzen; Reichspatentamt: Berlin, Germany, 1935. [Google Scholar]
- Grohmann, H.; Grill, M. Method and Apparatus for Large Scale Manufacture of Metal Oxides, Especially Magnesium Oxide; Veitscher Magnesitwerke A.-G.: Vienna, Austria, 1979. [Google Scholar]
- Grill, M. Die Fällung von Eisenhydroxid aus Magnesiumchloridlösungen; Institut für Physikalische Chemie; Montanuniversität Leoben: Leoben, Austria, 1983. [Google Scholar]
- Grill, M.; Graf, G. Manufacture of Pure Magnesium Oxide, Especially for the Manufacture of Refractory Products; Veitscher Magnesitwerke A.-G.: Vienna, Austria, 1988. [Google Scholar]
- Meier, A. Charakterisierung der Oberfläche Einer Neuen Amorphen Kieselsäure; Institut für Physikalische Chemie; Montanuniversität Leoben: Leoben, Austria, 1989. [Google Scholar]
| Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).