Cytotoxic Effects of Zinc Oxide Nanoparticles on Human Glial Cells †
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Nanoparticle Characterization and Cellular Uptake
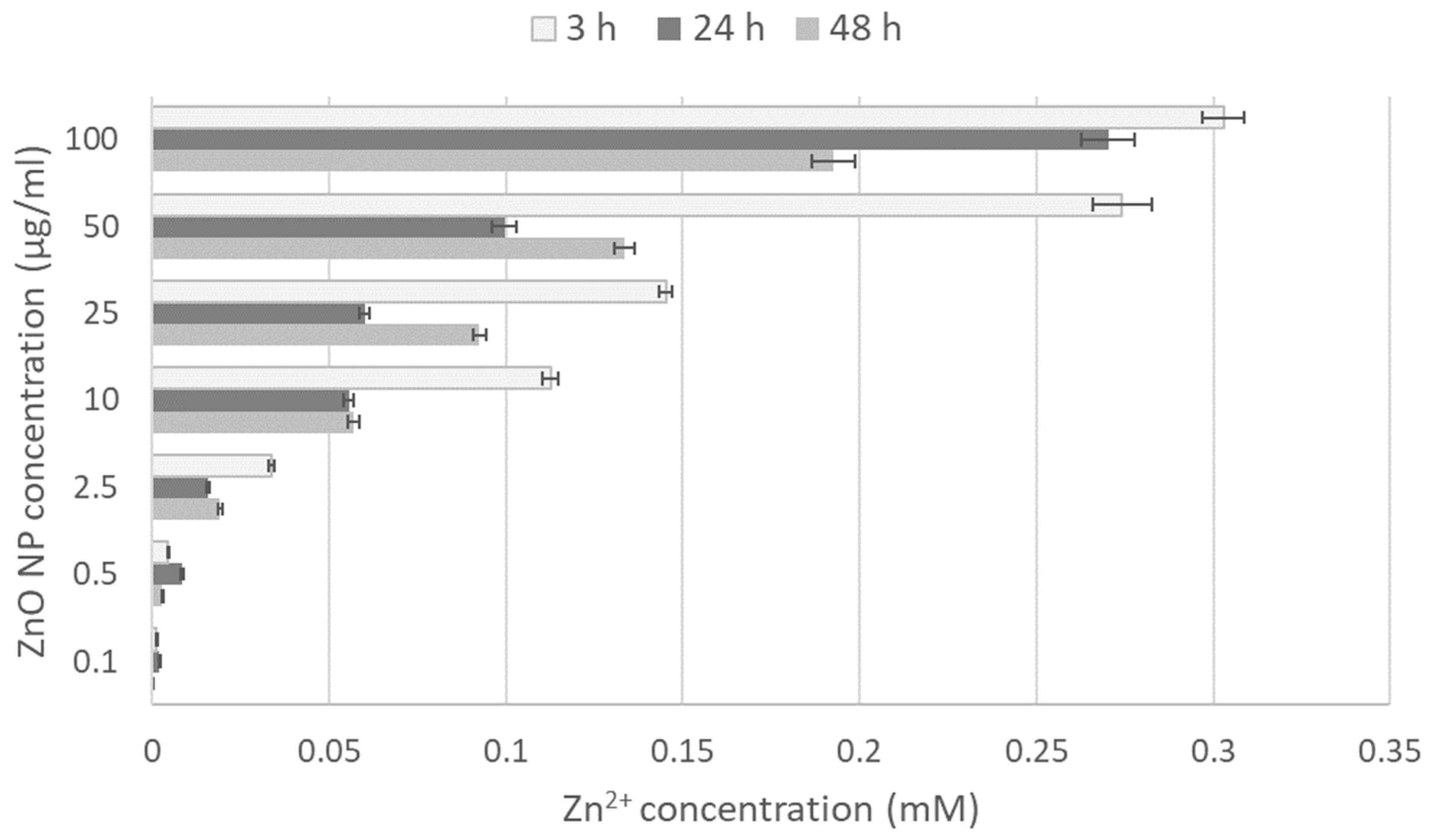
3.2. Zn2+ Ion Release
3.3. ZnO NP Effects on Glial Cells
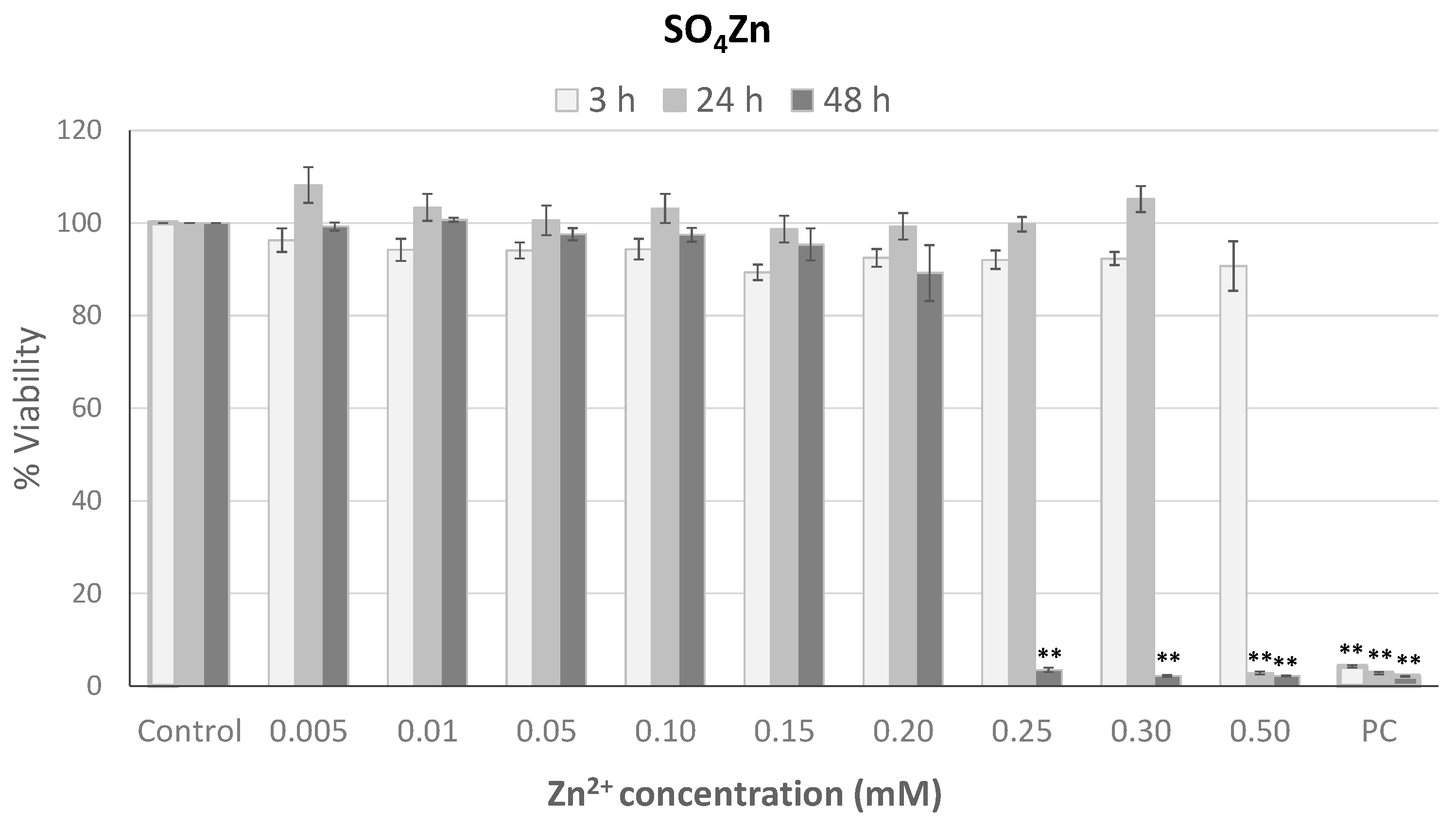
3.4. Role of Zn2+ Ions Released from ZnO NP
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Du, J.; Tang, J.; Xu, S.; Ge, J.; Dong, Y.; Li, H.; Jin, M. ZnO nanoparticles: Recent advances in ecotoxicity and risk assessment. Drug Chem. Toxicol. 2020, 43, 322–333. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.E.; Li, Z.H.; Zheng, W.; Zhao, Y.F.; Jin, Y.F.; Tang, Z.X. Food Additives & Contaminants: Part A Synthesis, antibacterial activity, antibacterial mechanism and food applications of ZnO nanoparticles: A review. Food Addit. Contam. Part A 2014, 31, 173–186. [Google Scholar]
- Singh, S. Zinc oxide nanoparticles impacts: Cytotoxicity, genotoxicity, developmental toxicity, and neurotoxicity. Toxicol. Mech. Methods 2019, 29, 300–311. [Google Scholar] [CrossRef] [PubMed]
- Smaoui, S.; Chérif, I.; Hlima, H.B.; Khan, M.U.; Rebezov, M.; Thiruvengadam, M.; Sarkar, T.; Shariati, M.A.; Lorenzo, J.M. Zinc oxide nanoparticles in meat packaging: A systematic review of recent literature. Food Packag. Shelf Life 2023, 36, 101045. [Google Scholar] [CrossRef]
- Guo, Z.; Zhang, P.; Chakraborty, S.; Chetwynd, A.J.; Monikh, F.A.; Stark, C.; Ali-Boucetta, H.; Wilson, S.; Lynch, I.; Valsami-Jones, E. Biotransformation modulates the penetration of metallic nanomaterials across an artificial blood–brain barrier model. Proc. Natl. Acad. Sci. USA 2021, 118, e2105245118. [Google Scholar] [CrossRef]
- Valdiglesias, V.; Costa, C.; Kiliç, G.; Costa, S.; Pásaro, E.; Laffon, B.; Teixeira, J.P.; Paulo, J. Neuronal cytotoxicity and genotoxicity induced by zinc oxide nanoparticles. Environ. Int. 2013, 55, 92–100. [Google Scholar] [CrossRef]
- Fernández-Bertólez, N.; Costa, C.; Brandão, F.; Duarte, J.A.; Teixeira, J.P.; Pásaro, E.; Valdiglesias, V.; Laffon, B. Evaluation of cytotoxicity and genotoxicity induced by oleic acid-coated iron oxide nanoparticles in human astrocytes. Environ. Mol. Mutagen 2019, 60, 816–829. [Google Scholar] [CrossRef]
- Valdiglesias, V.; Costa, C.; Sharma, V.; Kiliç, G.; Pásaro, E.; Teixeira, J.P.; Dhawan, A.; Laffon, B. Comparative study on effects of two different types of titanium dioxide nanoparticles on human neuronal cells. Food Chem. Toxicol. 2013, 57, 352–361. [Google Scholar] [CrossRef]
- Sharma, A.K.; Singh, V.; Gera, R.; Purohit, M.P.; Ghosh, D. Zinc oxide nanoparticle induces microglial death by NADPH-oxidase-independent reactive oxygen species as well as energy depletion. Mol. Neurobiol. 2017, 54, 6273–6286. [Google Scholar] [CrossRef]
- Sruthi, S.; Mohanan, P.V. Investigation on cellular interactions of astrocytes with zinc oxide nanoparticles using rat C6 cell lines. Colloids Surf. B Biointerfaces 2015, 133, 1–11. [Google Scholar] [CrossRef]
- Heim, J.; Felder, E.; Tahir, M.N.; Kaltbeitzel, A.; Heinrich, U.R.; Brochhausen, C.; Mailänder, V.; Tremel, W.; Brieger, J. Genotoxic effects of zinc oxide nanoparticles. Nanoscale 2015, 7, 8931–8938. [Google Scholar] [CrossRef]
- Zhang, J.; Qin, X.; Wang, B.; Xu, G.; Qin, Z.; Wang, J.; Wu, L.; Ju, X.; Bose, D.D.; Qiu, F.; et al. Zinc oxide nanoparticles harness autophagy to induce cell death in lung epithelial cells. Cell Death Dis. 2017, 8, e2954. [Google Scholar] [CrossRef] [PubMed]
- Mittag, A.; Hoera, C.; Kämpfe, A.; Westermann, M.; Kuckelkorn, J.; Schneider, T.; Glei, M. Cellular uptake and toxicological effects of differently sized zinc oxide nanoparticles in intestinal cells. Toxics 2021, 9, 96. [Google Scholar] [CrossRef] [PubMed]
- Eixenberger, J.E.; Anders, C.B.; Hermann, R.J.; Brown, R.J.; Reddy, K.M.; Punnoose, A.; Wingett, D.G. Rapid dissolution of ZnO nanoparticles induced by biological buffers significantly impacts cytotoxicity. Chem. Res. Toxicol. 2017, 30, 1641–1651. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Zhang, F.; Zhang, H. In Vitro response of immune cells on metal oxide nanoparticles with different solubility. J. Nanosci. Nanotechnol. 2016, 16, 5546–5552. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Anderson, D.; Dhawan, A. oxide nanoparticles induce oxidative DNA damage and ROS-triggered mitochondria mediated apoptosis in human liver cells (HepG2). Apoptosis Zinc 2012, 17, 852–870. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Wang, G.; Huang, J.; Zhang, Y.; Cheng, X.; Chuai, M.; Brand-Saberi, B.; Chen, G.; Jiang, X.; Yang, X. Zinc oxide nanoparticles exposure-induced oxidative stress restricts cranial neural crest development during chicken embryogenesis. Ecotoxicol. Environ. Saf. 2020, 194, 110415. [Google Scholar] [CrossRef]
- Pandurangan, M.; Kim, D.H. In vitro toxicity of zinc oxide nanoparticles: A review. J. Nanoparticle Res. 2015, 17, 158. [Google Scholar] [CrossRef]
- Bozym, R.A.; Chimienti, F.; Giblin, L.J.; Gross, G.W.; Korichneva, I.; Li, Y.; Libert, S.; Maret, W.; Parviz, M.; Frederickson, C.J.; et al. Free zinc ions outside a narrow concentration range are toxic to a variety of cells in vitro. Exp. Biol. Med. 2010, 235, 741–750. [Google Scholar] [CrossRef]
- Holmes, A.M.; Mackenzie, L.; Roberts, M.S. Disposition and measured toxicity of zinc oxide nanoparticles and zinc ions against keratinocytes in cell culture and viable human epidermis. Nanotoxicology 2020, 14, 263–274. [Google Scholar] [CrossRef]
- Nair, S.; Sasidharan, A.; Divya Rani, V.V.; Menon, D.; Nair, S.; Manzoor, K.; Raina, S. Role of size scale of ZnO nanoparticles and microparticles on toxicity toward bacteria and osteoblast cancer cells. J. Mater. Sci. Mater. Med. 2009, 20, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Patel, P.; Kansara, K.; Senapati, V.A.; Shanker, R.; Dhawan, A.; Kumar, A. Cell cycle dependent cellular uptake of zinc oxide nanoparticles in human epidermal cells. Mutagenesis 2016, 31, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.W.Y.; Leung, P.T.Y.; Djurišić, A.B.; Leung, K.M.Y. Toxicities of nano zinc oxide to five marine organisms: Influences of aggregate size and ion solubility, in: Analytical and Bioanalytical Chemistry. Anal. Bioanal. Chem. 2010, 396, 609–618. [Google Scholar] [CrossRef] [PubMed]

| Hydrodynamic Diameter (nm) a | Zeta Potential (mV) a | ||
|---|---|---|---|
| Time (h) | 0 | 302.09 ± 0.84 | −1.73 ± 4.05 |
| 3 | 315.01 ± 3.16 | 2.79 ± 3.01 | |
| 24 | 269.16 ± 1.36 | −20.25 ± 2.03 | |
| 48 | 273.97 ± 5.91 | −13.46 ± 2.36 | |
| ZnO NP Concentration (µg/mL) | Exposure Time | ||
|---|---|---|---|
| 3 h | 24 h | 48 h | |
| Control | 1.06 ± 0.18 | 1.13 ± 0.16 | 0.76 ± 0.06 |
| 0.1 | 1.12 ± 1.16 | 2.04 ± 0.24 ** | 1.98 ± 0.29 ** |
| 0.5 | 1.21 ± 0.18 | 4.41 ± 1.20 ** | 3.18 ± 0.47 ** |
| 2.5 | 1.21 ± 0.12 | 4.99 ± 1.28 ** | 2.15 ± 0.39 ** |
| 10 | 1.03 ± 0.13 | 7.06 ± 1.80 ** | 4.11 ± 0.61 ** |
| 25 | 1.05 ± 0.21 | 6.79 ± 1.53 ** | 6.42 ± 0.68 ** |
| 50 | 1.71 ± 0.31 | 9.34 ± 1.06 ** | 11.75 ± 0.88 ** |
| 100 | 5.28 ± 0.55 ** | 7.92 ± 0.69 ** | 28.06 ± 2.41 ** |
| Positive control | 42.42 ± 4.06 ** | 64.11 ± 2.25 ** | 71.88 ± 1.21 ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valdiglesias, V.; Touzani, A.; Ramos-Pan, L.; Alba-González, A.; Folgueira, M.; Moreda-Piñeiro, J.; Méndez, J.; Pásaro, E.; Fernández-Bertólez, N.; Laffon, B. Cytotoxic Effects of Zinc Oxide Nanoparticles on Human Glial Cells. Mater. Proc. 2023, 14, 23. https://doi.org/10.3390/IOCN2023-14509
Valdiglesias V, Touzani A, Ramos-Pan L, Alba-González A, Folgueira M, Moreda-Piñeiro J, Méndez J, Pásaro E, Fernández-Bertólez N, Laffon B. Cytotoxic Effects of Zinc Oxide Nanoparticles on Human Glial Cells. Materials Proceedings. 2023; 14(1):23. https://doi.org/10.3390/IOCN2023-14509
Chicago/Turabian StyleValdiglesias, Vanessa, Assia Touzani, Lucía Ramos-Pan, Anabel Alba-González, Mónica Folgueira, Jorge Moreda-Piñeiro, Josefina Méndez, Eduardo Pásaro, Natalia Fernández-Bertólez, and Blanca Laffon. 2023. "Cytotoxic Effects of Zinc Oxide Nanoparticles on Human Glial Cells" Materials Proceedings 14, no. 1: 23. https://doi.org/10.3390/IOCN2023-14509
APA StyleValdiglesias, V., Touzani, A., Ramos-Pan, L., Alba-González, A., Folgueira, M., Moreda-Piñeiro, J., Méndez, J., Pásaro, E., Fernández-Bertólez, N., & Laffon, B. (2023). Cytotoxic Effects of Zinc Oxide Nanoparticles on Human Glial Cells. Materials Proceedings, 14(1), 23. https://doi.org/10.3390/IOCN2023-14509







