N,N-Dimethyl-4-amino-2,1,3-benzothiadiazole: Synthesis and Luminescent Solvatochromism †
Abstract
:1. Introduction
2. Materials and Methods
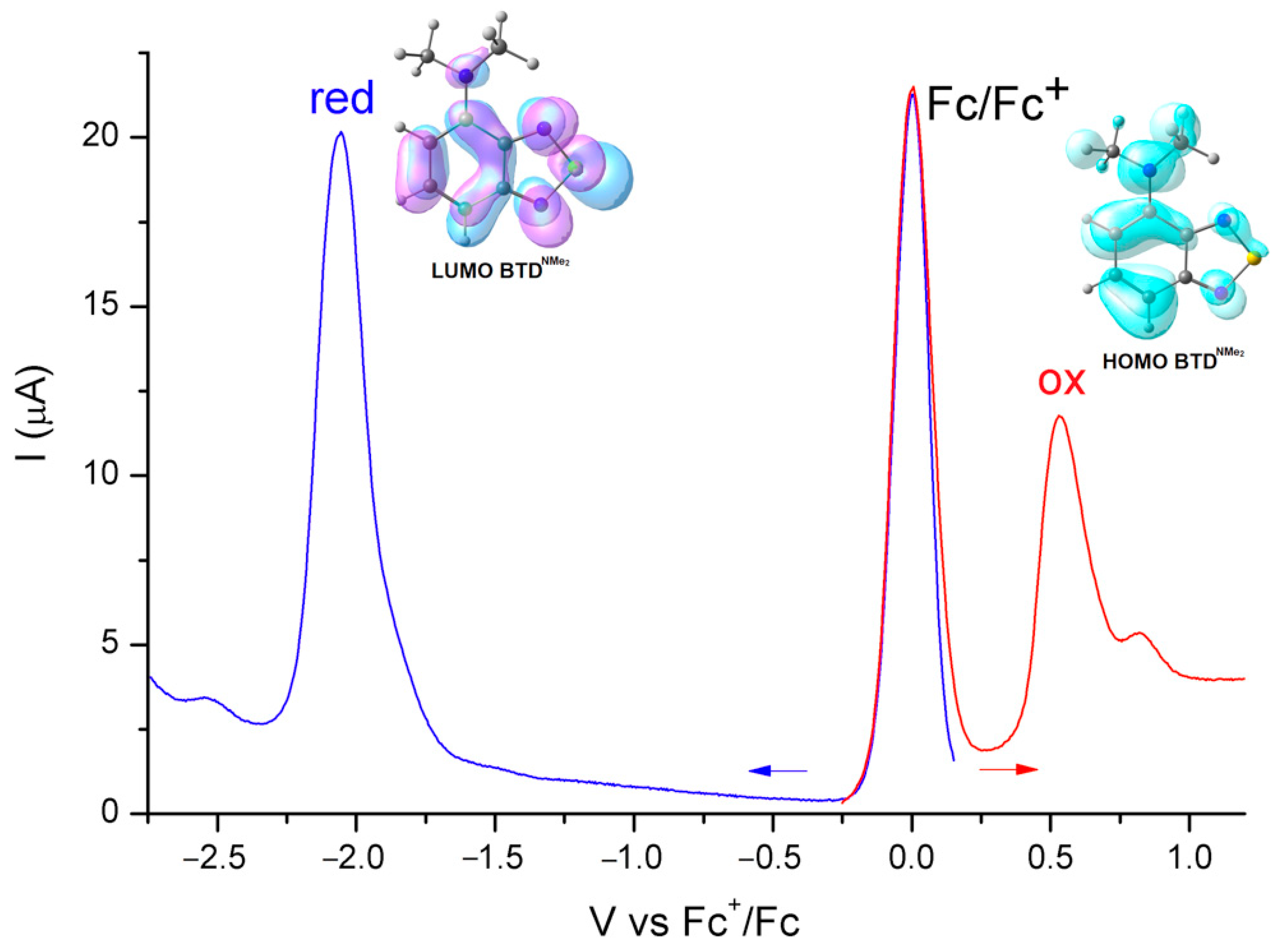
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Neto, B.A.D.; Lapis, A.A.M.; da Silva Júnior, E.N.; Dupont, J. 2,1,3-benzothiadiazole and derivatives: Synthesis, properties, reactions, and applications in light technology of small molecules. Eur. J. Org. Chem. 2013, 2013, 228–255. [Google Scholar] [CrossRef]
- Sukhikh, T.S.; Ogienko, D.S.; Bashirov, D.A.; Konchenkoa, S.N. Luminescent complexes of 2,1,3-benzothiadiazole derivatives. Russ. Chem. Bull. 2019, 68, 651–661. [Google Scholar] [CrossRef]
- Langis-Barsetti, S.; Maris, T.; Wuest, J.D. Molecular organization of 2,1,3-benzothiadiazoles in the solid state. J. Org. Chem. 2017, 82, 5034–5045. [Google Scholar] [CrossRef]
- Nakabayashi, K.; Takahashi, T.; Sugawara, R.; Lo, C.-T.; Mori, H. Benzothiadiazole-based donor–acceptor nanoparticles with solvatochromic and thermoresponsive properties. React. Funct. Polym. 2018, 131, 350–360. [Google Scholar] [CrossRef]
- Benevides, T.O.; Regis, E.; Nicoleti, C.S.; Bechtold, I.H.; Vieira, A.A. Phase-dependent photoluminescence of non-symmetric 2,1,3-benzothiadiazole liquid crystals. Dye. Pigment. 2019, 163, 300–307. [Google Scholar] [CrossRef]
- Zhao, X.; Chaudhry, S.T.; Mei, J. Heterocyclic building blocks for organic semiconductors. In Advances in Heterocyclic Chemistry; Elsevier: Amsterdam, The Netherlands, 2017; Volume 121, pp. 133–171. [Google Scholar] [CrossRef]
- Volz, D.; Wallesch, M.; Fléchon, C.; Danz, M.; Verma, A.; Navarro, J.M.; Zink, D.M.; Bräse, S.; Baumann, T. From iridium and platinum to copper and carbon: New avenues for more sustainability in organic light-emitting diodes. Green Chem. 2017, 17, 1988–2011. [Google Scholar] [CrossRef]
- Zhang, Y.; Song, J.; Qu, J.; Qian, P.-C.; Wong, W.-Y. Recent progress of electronic materials based on 2,1,3-benzothiadiazole and its derivatives: Synthesis and their application in organic light-emitting diodes. Sci. China Chem. 2021, 64, 341–357. [Google Scholar] [CrossRef]
- Pazini, A.; Maqueira, L.; Stieler, R.; Aucélio, R.Q.; Limberger, J. Synthesis, characterization and photophysical properties of luminescent non-symmetric 4-pyridyl benzothiadiazole derivatives. J. Mol. Struct. 2017, 1131, 181–189. [Google Scholar] [CrossRef]
- Bardi, B.; Dall’Agnese, C.; Moineau-Chane Ching, K.I.; Painelli, A.; Terenziani, F. Spectroscopic investigation and theoretical modeling of benzothiadiazole-based charge-transfer chromophores: From solution to nanoaggregates. J. Phys. Chem. C 2017, 121, 17466–17478. [Google Scholar] [CrossRef]
- Pazini, A.; Maqueira, L.; Avila, H.C.; Valente, F.M.; Aderne, R.E.; Back, D.; Aucélio, R.Q.; Cremona, M.; Limberger, J. Phenoxy-benzothiadiazole dyes: Synthesis, photophysical properties and preliminary application in OLEDs. Tetrahedron Lett. 2018, 59, 2994–2999. [Google Scholar] [CrossRef]
- Paczkowski, I.M.; Coelho, F.L.; Campo, L.F. 2,1,3-Benzothiadiazole dyes conjugated with benzothiazole and benzoxazole: Synthesis, solvatochromism and solid-state properties. J. Mol. Liq. 2020, 319, 114277. [Google Scholar] [CrossRef]
- Paisley, N.R.; Tonge, C.M.; Mayder, D.M.; Thompson, K.A.; Hudson, Z.M. Tunable benzothiadiazole-based donor–acceptor materials for two-photon excited fluorescence. Mater. Chem. Front. 2020, 4, 555–566. [Google Scholar] [CrossRef]
- Gao, S.; Balan, B.; Yoosaf, K.; Monti, F.; Bandini, E.; Barbieri, A.; Armaroli, N. Highly efficient luminescent solar concentrators based on benzoheterodiazole dyes with large stokes shifts. Chem. Eur. J. 2020, 26, 11013–11023. [Google Scholar] [CrossRef]
- Wu, Y.; Zhu, W. Organic sensitizers from D–π–A to D–A–π–A: Effect of the internal electron-withdrawing units on molecular absorption, energy levels and photovoltaic performances. Chem. Soc. Rev. 2013, 42, 2039–2058. [Google Scholar] [CrossRef]
- Holliday, S.; Li, Y.; Luscombe, C.K. Recent advances in high performance donor-acceptor polymers for organic photovoltaics. Prog. Polym. Sci. 2017, 70, 34–51. [Google Scholar] [CrossRef]
- Zheng, P.; Xu, J.; Peng, F.; Peng, S.; Liao, J.; Zhao, H.; Li, L.; Zeng, X.; Yu, H. Novel dual acceptor (D–D′–A′–π–A) dye-sensitized solar cells based on the triarylamine structure and benzothiadiazole double electron withdrawing unit. New J. Chem. 2021, 45, 4443–4452. [Google Scholar] [CrossRef]
- Wang, C.; Liu, F.; Chen, Q.-M.; Xiao, C.-Y.; Wu, Y.-G.; Li, W.-W. Benzothiadiazole-based conjugated polymers for organic solar cells. Chin. J. Polym. Sci. 2021, 39, 525–536. [Google Scholar] [CrossRef]
- Raitz, I.; de Souza Filho, R.Y.; de Andrade, L.P.; Correa, J.R.; Neto, B.A.D.; Pilli, R.A. Preferential mitochondrial localization of a goniothalamin fluorescent derivative. ACS Omega 2017, 2, 3774–3784. [Google Scholar] [CrossRef] [Green Version]
- Appelqvist, H.; Stranius, K.; Börjesson, K.; Nilsson, K.P.R.; Dyrager, C. Specific imaging of intracellular lipid droplets using a benzothiadiazole derivative with solvatochromic properties. Bioconjug. Chem. 2017, 28, 1363–1370. [Google Scholar] [CrossRef] [Green Version]
- Souza, V.S.; Corrêa, J.R.; Carvalho, P.H.; Zanotto, G.M.; Matiello, G.I.; Guido, B.C.; Gatto, C.C.; Ebeling, G.; Gonçalves, P.F.B.; Dupont, J.; et al. Appending ionic liquids to fluorescent benzothiadiazole derivatives: Light up and selective lysosome staining. Sens. Actuators B Chem. 2020, 321, 128530. [Google Scholar] [CrossRef]
- Uchiyama, S.; Tsuji, T.; Ikado, K.; Yoshida, A.; Kawamoto, K.; Hayashic, T.; Inadac, N. A cationic fluorescent polymeric thermometer for the ratiometric sensing of intracellular temperature. Analyst 2015, 140, 4498–4506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uchiyama, S.; Kimura, K.; Gota, C.; Okabe, K.; Kawamoto, K.; Inada, N.; Yoshihara, T.; Tobita, S. Environment-sensitive fluorophores with benzothiadiazole and benzoselenadiazole structures as candidate components of a fluorescent polymeric thermometer. Chem. Eur. J. 2012, 18, 9552–9563. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Byun, J.; Huang, W.; Ayed, C.; Wang, L.; Zhang, K.A.I. Poly(benzothiadiazoles) and their derivatives as heterogeneous photocatalysts for visible-light-driven chemical transformations. ACS Catal. 2018, 8, 4735–4750. [Google Scholar] [CrossRef]
- Wang, G.-B.; Li, S.; Yan, C.-X.; Lin, Q.-Q.; Zhu, F.-C.; Geng, Y.; Dong, Y.-B. A benzothiadiazole-based covalent organic framework for highly efficient visible-light driven hydrogen evolution. Chem. Commun. 2020, 56, 12612–12615. [Google Scholar] [CrossRef] [PubMed]
- Broncová, G.; Shishkanova, T.V.; Dendisová, M.; Člupek, M.; Kubáč, D.; Matějka, P. Poly(4-amino-2,1,3-benzothiadiazole) films: Preparation, characterization and applications. Chem. Pap. 2017, 71, 359–366. [Google Scholar] [CrossRef]
- Munakata, M.; He, H.; Kuroda-Sowa, T.; Maekawa, M.; Suenaga, Y. Dicopper complexes derived from 4-amino-2,1,3-benzothiadiazole with versatile co-ordination number and geometry. J. Chem. Soc. Dalton Trans. 1998, 1499–1502. [Google Scholar] [CrossRef]
- Bashirov, D.A.; Sukhikh, T.S.; Kuratieva, N.V.; Chulanova, E.A.; Yushina, I.V.; Gritsan, N.P.; Konchenko, S.N.; Zibarev, A.V. Novel applications of functionalized 2,1,3-benzothiadiazoles for coordination chemistry and crystal engineering. RSC Adv. 2014, 4, 28309–28316. [Google Scholar] [CrossRef]
- Sukhikh, T.S.; Ogienko, D.S.; Bashirov, D.A.; Kuratieva, N.V.; Komarov, V.Y.; Rakhmanova, M.I.; Konchenko, S.N. New red-luminescent cadmium coordination polymers with 4-amino-2,1,3-benzothiadiazole. J. Coord. Chem. 2016, 69, 3284–3293. [Google Scholar] [CrossRef]
- Sukhikh, T.S.; Bashirov, D.A.; Ogienko, D.S.; Kuratieva, N.V.; Sherin, P.S.; Rakhmanova, M.I.; Chulanova, E.A.; Gritsan, N.P.; Konchenko, S.N.; Zibarev, A.V. Novel luminescent β-ketoimine derivative of 2,1,3-benzothiadiazole: Synthesis, complexation with Zn(ii) and photophysical properties in comparison with related compounds. RSC Adv. 2016, 6, 43901–43910. [Google Scholar] [CrossRef]
- Sukhikh, T.S.; Komarov, V.Y.; Konchenko, S.N.; Benassi, E. The hows and whys of peculiar coordination of 4-amino-2,1,3-benzothiadiazole. Polyhedron 2018, 139, 33–43. [Google Scholar] [CrossRef]
- Sukhikh, T.S.; Bashirov, D.A.; Shuvaev, S.; Komarov, V.Y.; Kuratieva, N.V.; Konchenko, S.N.; Benassi, E. Noncovalent interactions and photophysical properties of new Ag(I) complexes with 4-amino-2,1,3-benzothiadiazole. Polyhedron 2018, 141, 77–86. [Google Scholar] [CrossRef]
- Sukhikh, T.S.; Khisamov, R.M.; Bashirov, D.A.; Komarov, V.Y.; Molokeev, M.S.; Ryadun, A.A.; Benassi, E.; Konchenko, S.N. Tuning of the coordination and emission properties of 4-amino-2,1,3-benzothiadiazole by introduction of diphenylphosphine group. Cryst. Growth Des. 2020, 20, 5796–5807. [Google Scholar] [CrossRef]
- Slavachevskaya, N.M.; Belen’kaya, I.A.; Tsepova, N.S.; Levocheskaya, E.I.; Krasil’nikov, I.I. Synthesis of certain quaternary derivatives in the aminophenol and benzo-2,1,3-thiadiazole series as potential radiation-protecting materials. Pharm. Chem. J. 1976, 10, 327–331. [Google Scholar] [CrossRef]
- Armarego, W.L.F.; Perrin, D.D. Purification of Laboratory Chemicals, 4th ed.; Butterworth-Heinemann: Oxford, UK, 1996. [Google Scholar]
- Komin, A.P.; Carmack, M. The chemistry of 1,2,5-thiadiazoles, IV. Benzo [1,2-c:3,4-c′:5,6-c″]tris [1,2,5]thiadiazole. J. Heterocycl. Chem. 1975, 12, 829–833. [Google Scholar] [CrossRef]
- Da Silva Miranda, F.; Signori, A.M.; Vicente, J.; de Souza, B.; Priebe, J.P.; Szpoganicz, B.; Sanches Gonçalves, N.; Neves, A. Synthesis of substituted dipyrido[3,2-a:2′,3′-c]phenazines and a new heterocyclic dipyrido[3,2-f:2′,3′-h]quinoxalino[2,3-b]quinoxaline. Tetrahedron 2008, 64, 5410–5415. [Google Scholar] [CrossRef]
- Liu, Y.; Lu, Y.; Prashad, M.; Repič, O.; Blacklock, T.J. A practical and chemoselective reduction of nitroarenes to anilines using activated iron. Adv. Synth. Catal. 2005, 347, 217–219. [Google Scholar] [CrossRef]
- Fery-Forgues, S.; Lavabre, D. Are fluorescence quantum yields so tricky to measure? a demonstration using familiar stationery products. J. Chem. Educ. 1999, 76, 1260–1264. [Google Scholar] [CrossRef]
- Gerber, I.C.; Ángyán, J.G. Hybrid functional with separated range. Chem. Phys. Lett. 2005, 415, 100–105. [Google Scholar] [CrossRef]
- Chai, J.D.; Head-Gordon, M. Long-range corrected hybrid density functionals with damped atom–atom dispersion corrections. Phys. Chem. Chem. Phys. 2008, 10, 6615–6620. [Google Scholar] [CrossRef] [Green Version]
- Minenkov, Y.; Singstad, Å.; Occhipinti, G.; Jensen, V.R. The accuracy of DFT-optimized geometries of functional transition metal compounds: A validation study of catalysts for olefin metathesis and other reactions in the homogeneous phase. Dalton Trans. 2012, 41, 5526–5541. [Google Scholar] [CrossRef]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305. [Google Scholar] [CrossRef] [PubMed]
- Cossi, M.; Rega, N.; Scalmani, G.; Barone, V. Energies, structures, and electronic properties of molecules in solution with the CPCM solvation model. J. Comput. Chem. 2003, 24, 669–681. [Google Scholar] [CrossRef] [PubMed]
- Barone, V.; Cossi, M. Quantum calculation of molecular energies and energy gradients in solution by a conductor solven-model. J. Phys. Chem. A 1998, 102, 1995–2001. [Google Scholar] [CrossRef]
- Ullrich, C.A. Time-Dependent Density Functional Theory; Oxford University Press: Oxford, UK, 2012. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16, Revision C.01; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Reddy, M.D.; Fronczek, F.R.; Watkins, E.B. Rh-catalyzed, regioselective, C–H bond functionalization: Access to quinoline-branched amines and dimers. Org. Lett. 2016, 18, 5620–5623. [Google Scholar] [CrossRef] [PubMed]
- Eaton, D.F. Reference materials for fluorescence measurement. Pure Appl. Chem. 1988, 60, 1107–1114. [Google Scholar] [CrossRef]
- Valeur, B. Molecular Fluorescence: Principles and Applications; WILEY-VCH Verlag GmbH: Weinheim, Germany, 2002. [Google Scholar]
- Mataga, N.; Kaifu, Y.; Koizumi, M. Solvent effects upon fluorescence spectra and the dipole moments of excited molecules. Bull. Chem. Soc. Jpn. 1956, 29, 465–470. [Google Scholar] [CrossRef] [Green Version]
- Lakowicz, J.R. Principles of Fluorescence Spectroscopy, 3rd ed.; Springer: Singapore; Japan, 2006. [Google Scholar]
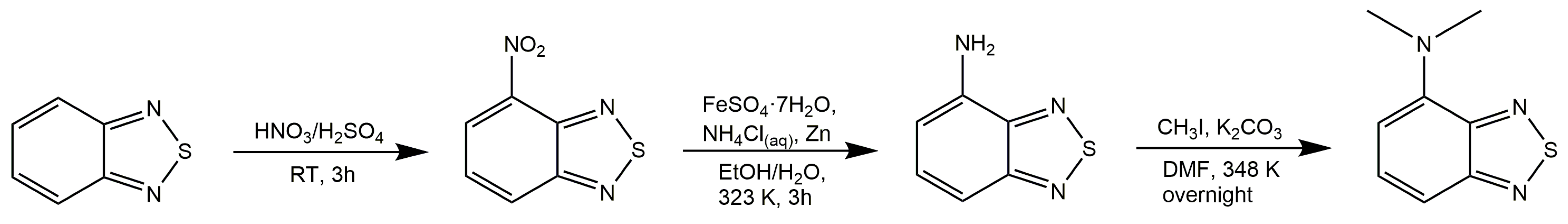
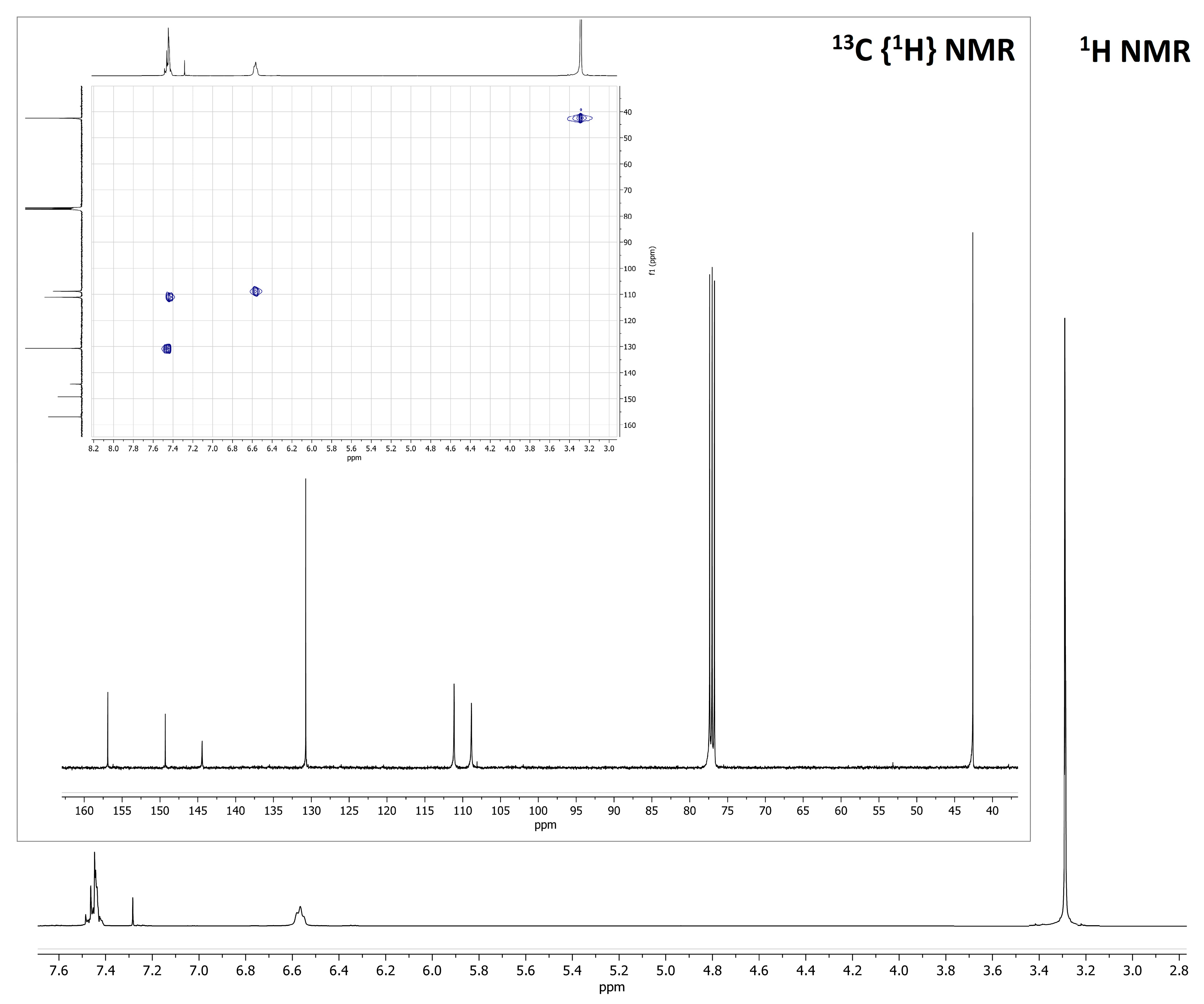
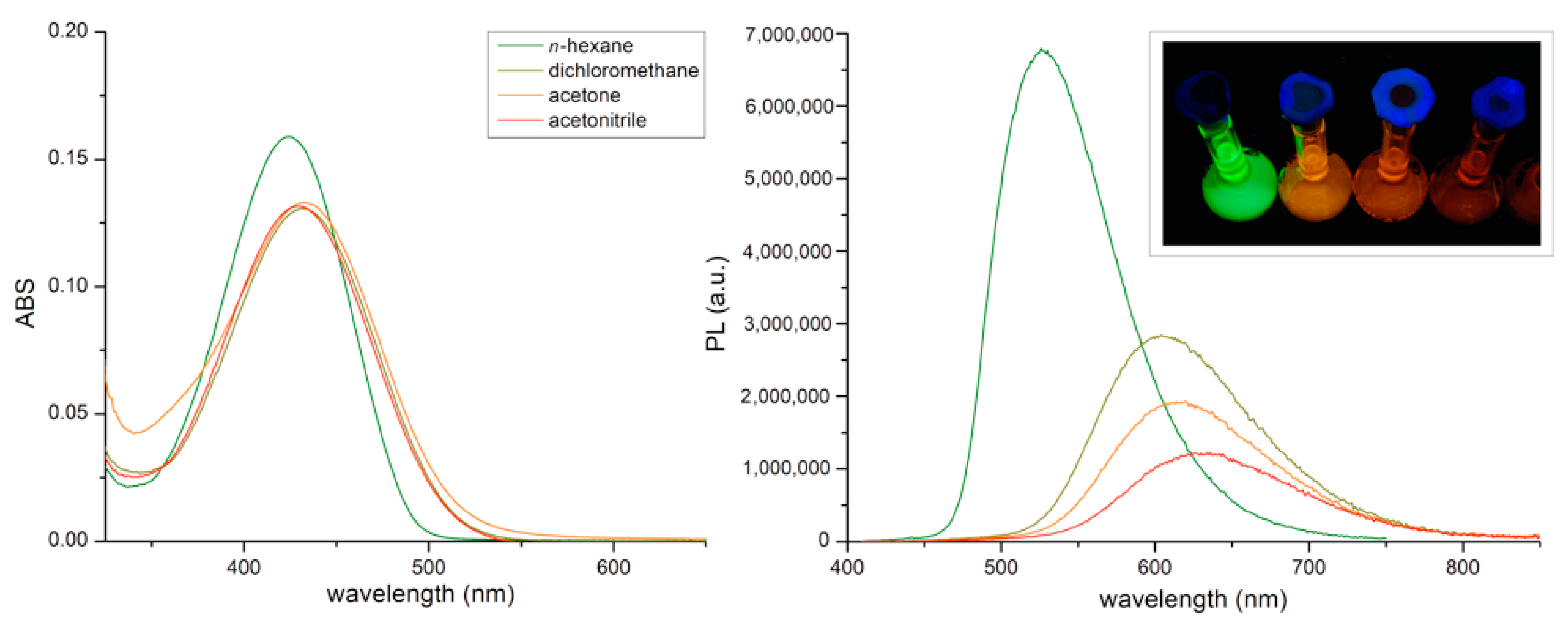

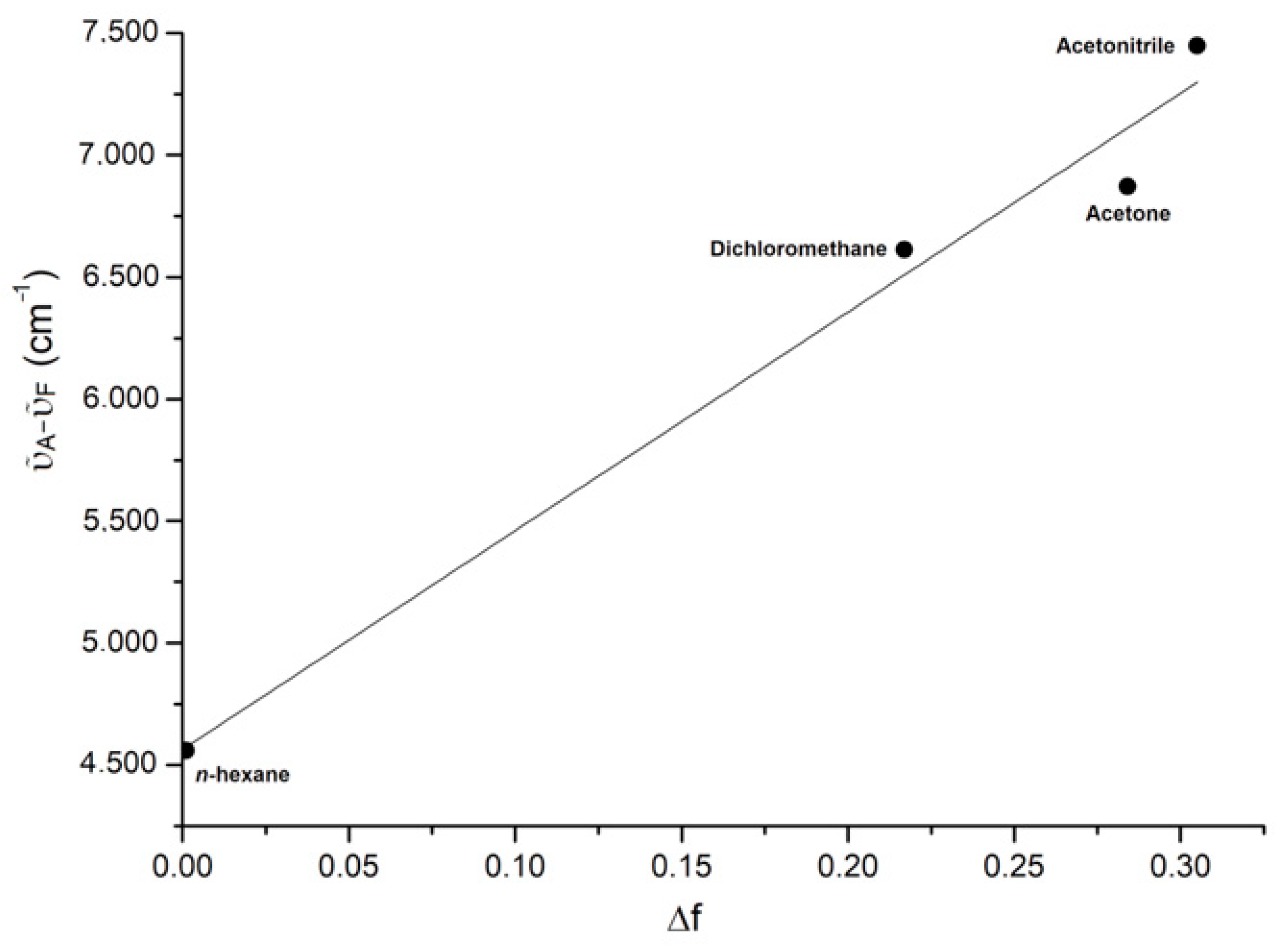
| Solvent | ε | n | ABS Max (nm) a | PL Max (nm) b | ῦA−ῦF (cm−1) | ΦF (%) c | Δf |
|---|---|---|---|---|---|---|---|
| n-hexane | 1.9 | 1.375 | 424 | 526 | 4559 | 52 | 0.001 |
| Dichloromethane | 8.9 | 1.424 | 432 | 604 | 6613 | 41 | 0.217 |
| Acetone | 20.7 | 1.359 | 433 | 616 | 6872 | 23 | 0.284 |
| Acetonitrile | 37.5 | 1.479 | 430 | 630 | 7448 | 16 | 0.305 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferraro, V.; Girotto, M.; Bortoluzzi, M. N,N-Dimethyl-4-amino-2,1,3-benzothiadiazole: Synthesis and Luminescent Solvatochromism. Chem. Proc. 2022, 8, 87. https://doi.org/10.3390/ecsoc-25-11658
Ferraro V, Girotto M, Bortoluzzi M. N,N-Dimethyl-4-amino-2,1,3-benzothiadiazole: Synthesis and Luminescent Solvatochromism. Chemistry Proceedings. 2022; 8(1):87. https://doi.org/10.3390/ecsoc-25-11658
Chicago/Turabian StyleFerraro, Valentina, Matteo Girotto, and Marco Bortoluzzi. 2022. "N,N-Dimethyl-4-amino-2,1,3-benzothiadiazole: Synthesis and Luminescent Solvatochromism" Chemistry Proceedings 8, no. 1: 87. https://doi.org/10.3390/ecsoc-25-11658
APA StyleFerraro, V., Girotto, M., & Bortoluzzi, M. (2022). N,N-Dimethyl-4-amino-2,1,3-benzothiadiazole: Synthesis and Luminescent Solvatochromism. Chemistry Proceedings, 8(1), 87. https://doi.org/10.3390/ecsoc-25-11658






