1. Introduction
The study of heterocyclic compounds is of great significance owing to their wide-range applications, particularly in the pharmaceutical industry, where most active pharmaceutical ingredients contain heterocyclic cores. Among them, benzodiazepines represent one of the most successful families, widely employed since the 1960s in the treatment of central nervous system disorders. 1,4-benzodiazepin-2-ones have been the focus of intensive research due to their anxiolytic, anticonvulsant, and muscle relaxant activities [
1,
2,
3].
In this context, N′-substituted 2-amino-N-phenylacetamides are of interest as key intermediates in the synthesis of novel benzodiazepines through cyclisation reactions, such as Pictet–Spengler or Bischler–Napieralski. The investigation of their structure is relevant, since the partial rigidity of the amide C(O)–N bond can give rise to E/Z rotamers, which influence the course of annulation reactions.
Amides constitute a fundamental model for understanding the conformation and dynamic behaviour of peptides and proteins [
4]. The rotational restriction of this bond generates
E/
Z isomerism [
5], which can be often observed by means of NMR spectroscopy [
6]. This technique enables the analysis of both static aspects (signal assignment and determination of equilibrium constants) and dynamic aspects (estimation of rotational barriers, ΔG). To complement experimental analysis, computational methods—particularly DFT—are employed to optimise geometries, calculate relative energies, and predict chemical shifts [
7,
8].
Finally, the DP4+ statistical method provides a robust tool for validating the assignment of
E/
Z isomers by correlating experimental data with theoretical values, thereby strengthening the reliability of structural elucidation of
N-substituted amides and their heterocyclic derivatives [
9].
2. Materials and Methods
2.1. Synthesis
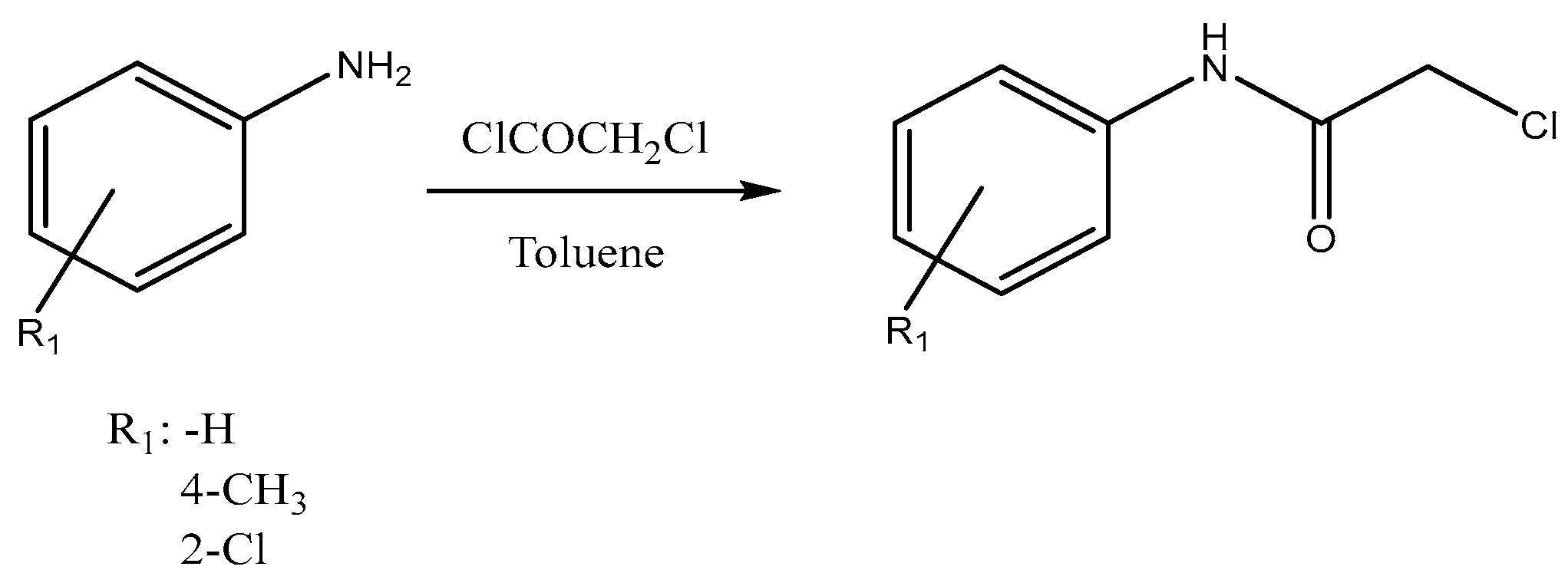
The synthesis of
N′-substituted 2-aminoacetanilides was conducted in two consecutive stages. In the first, a series of substituted anilines reacted with chloroacetyl chloride, affording the corresponding 2-chloroacetanilides, which were generally isolated as crystalline solids in good yields (
Figure 1).
Experimental procedure: In a 100 mL round-bottom flask, the corresponding substituted aniline (25 mmol) and triethylamine (36 mmol) were dissolved in 30 mL of toluene. To this solution, a solution of chloroacetyl chloride (25 mmol) in 20 mL of toluene was added dropwise. Once the addition was complete, the reaction mixture was heated under reflux for 1 h, and the reaction progress was monitored by thin-layer chromatography. Upon completion, the mixture was cooled and extracted with 10% aqueous HCl solution (3 × 10 mL). The organic layer was dried over anhydrous sodium sulphate, filtered, and the solvent was removed under reduced pressure using a rotary evaporator. The resulting solid was recrystallised from ethanol, affording typical yields of 65–75%.
In the second stage, these intermediates underwent nucleophilic substitution with methylamine, thereby yielding the desired 2-aminoacetanilides (
Figure 2).
Experimental procedure: In a 100 mL round-bottom flask, the corresponding 2-chloroacetanilide (2 mmol) was combined with 10 mL of a 33% w/w methylamine solution. The reaction mixture was stirred at room temperature for 1 h, with the progress monitored by thin-layer chromatography. Upon completion, the excess methylamine was removed under reduced pressure using a rotary evaporator, and the resulting residue was treated with 10% aqueous NaOH solution, followed by extraction with dichloromethane (3 × 10 mL). The organic phase was dried over anhydrous sodium sulphate, filtered, and the solvent was removed under reduced pressure. The resulting solid was recrystallised from ethanol, affording typical yields of 90–95%.
The compounds initially synthesised in this work were 2-chloro-N-phenylacetamide, 2-chloro-N-(p-tolyl)acetamide, and 2-chloro-N-(o-chlorophenyl)acetamide, which, upon reaction with methylamine, furnished 2-(methylamino)-N-phenylacetamide, 2-(methylamino)-N-(p-tolyl)acetamide, and 2-(methylamino)-N-(o-chlorophenyl)acetamide, respectively.
2.2. Characterisation
The compounds were characterised by 1H and 13C NMR spectroscopy, including two-dimensional techniques (COSY, HSQC). Melting points were determined, and purity was assessed by thin-layer chromatography.
2.3. Theoretical Calculations
Theoretical calculations were performed on a system equipped with an Intel® Core™ i5-9400 2.9 GHz processor (Intel Corporation, Santa Clara, CA, USA) and a 64-bit Windows 10 Pro operating system (Microsoft Corporation, Redmond, WA, USA). Both the relative stabilities of the possible species involved, and the NMR calculations were conducted using Gaussian software.
Relative stabilities were determined by DFT calculations [
10] employing the 6-31G(d,p) and 6-311G(d,p) basis sets. Geometries were optimised at the B3LYP level in vacuum. To identify the most stable conformer, a scan was performed by rotating the dihedral angle between the C–N–C(O) bonds, with the energy calculated at each rotation step. Two minima were found for each structure, corresponding to the
E and
Z rotamers.
These structures were subsequently optimised using the DFT method with the B3LYP functional and the 6-311++G(d,p) basis set [
11,
12]. Once the optimised structures were obtained, the chemical shifts (δcalc) relative to tetramethylsilane were computed for
1H and
13C nuclei in both conformers. Isotropic magnetic shielding tensors were evaluated in DMSO by GIAO NMR calculations at the B3LYP/6-31+G(d,p) level of theory, employing the self-consistent reaction field polarizable continuum model (SCRF-PCM). Each isotropic shielding value (σ
i) was converted into a chemical shift by subtracting it from the corresponding σ value of
1H or
13C in TMS, which were calculated using the same method and basis set.
3. Results and Discussion
N′-substituted 2-aminoacetanilides were obtained in yields ranging from 56% to 98%. The NMR spectra of all compounds were carefully analysed. The compound 2-(methylamino)-
N-(p-tolyl)acetamide was selected as a representative model for analysis.
Figure 3 shows the proposed numbering of the carbon atoms to facilitate the discussion.
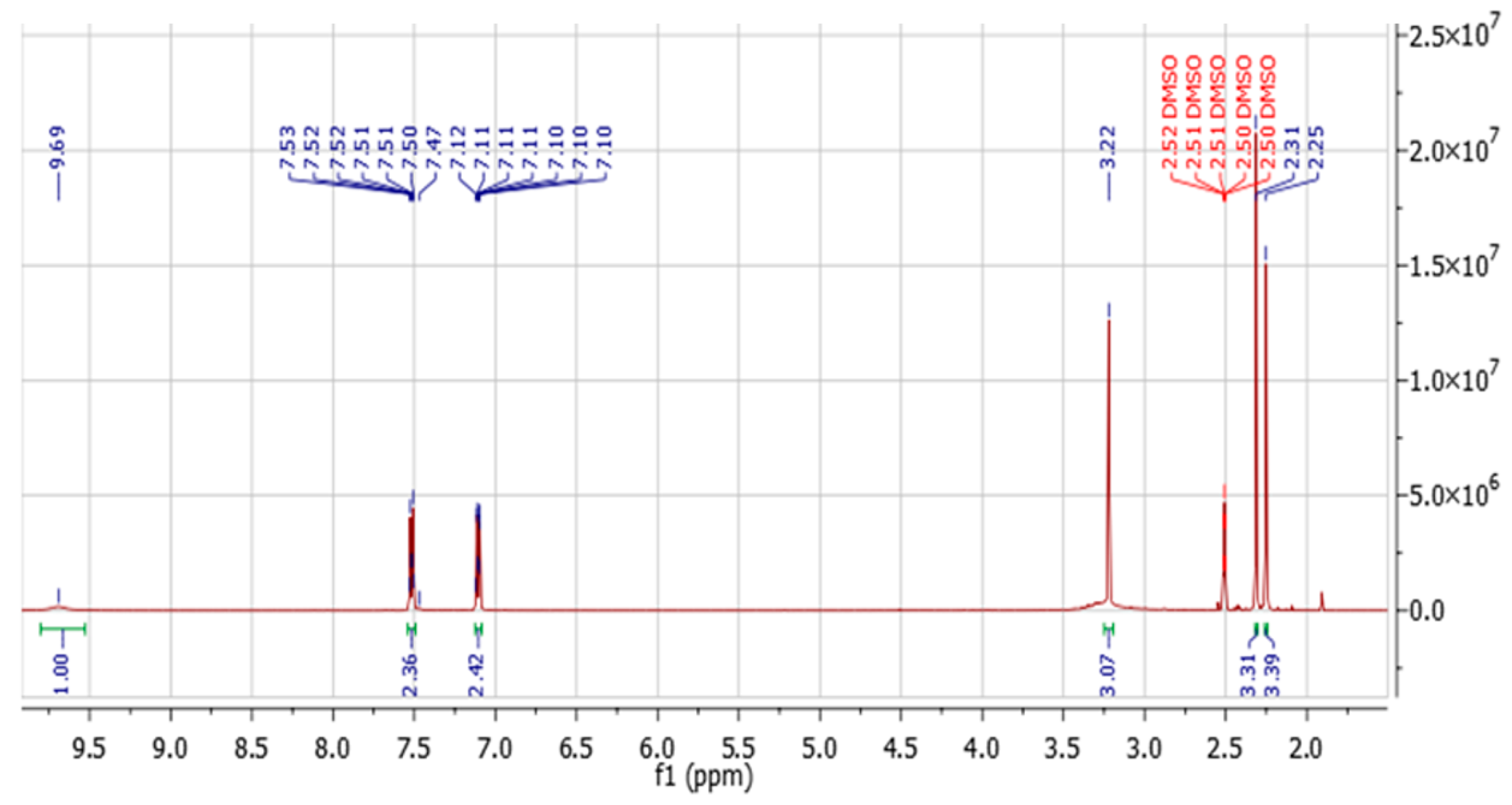
The
1H NMR spectrum of this compound is shown in
Figure 4 below:
Seven well-defined signals were observed: the amide proton at δ 9.69, a characteristic singlet of the –NHCO– group;, aromatic protons appearing as double doublets at δ 7.52 and 7.11; and aliphatic signals corresponding to the methylene group (δ 3.22), the methyl groups C9 and C10 (δ 2.31 and 2.25, respectively), and the amine proton, which overlapped with the methylene resonance at δ 3.22 ppm.
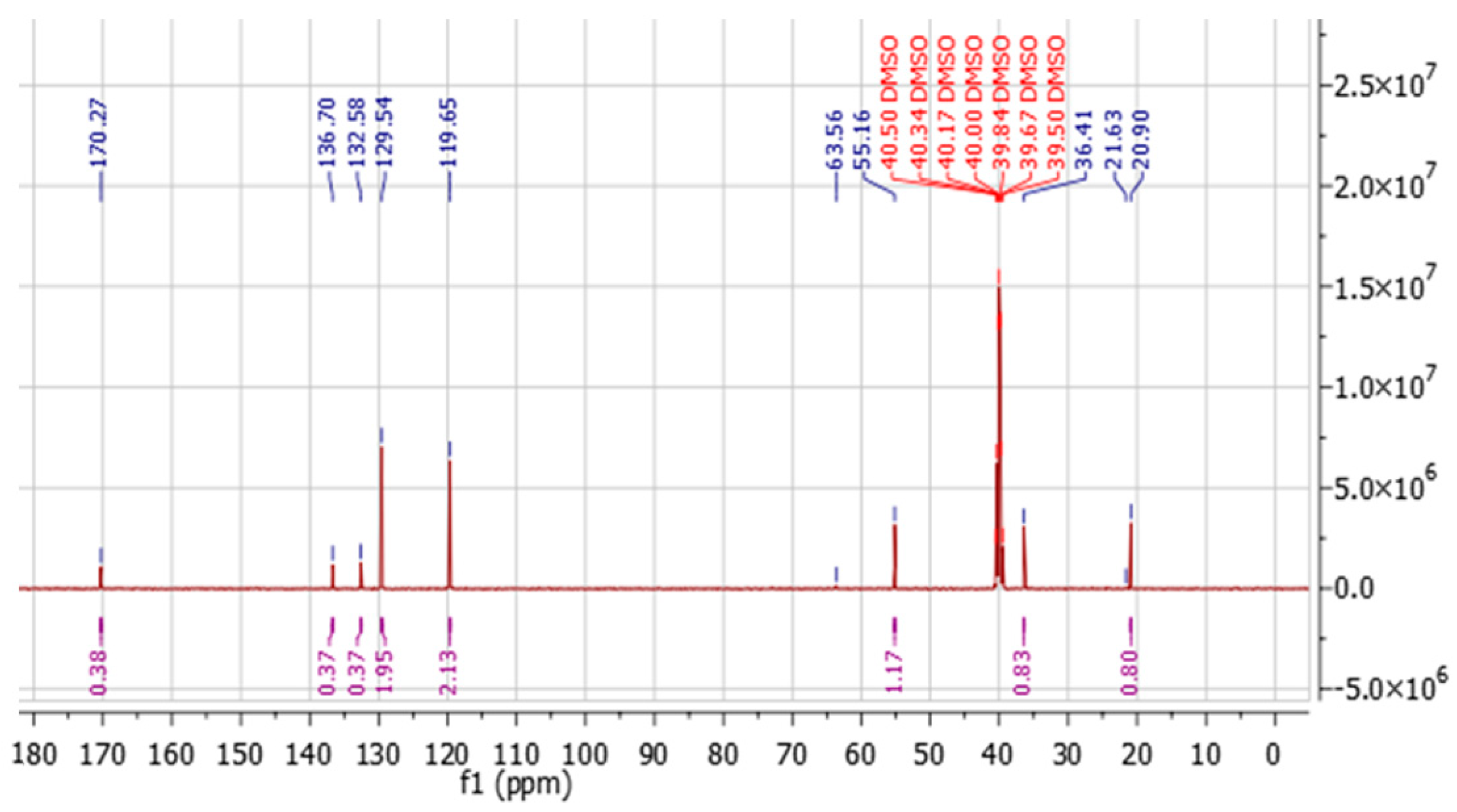
Figure 5 shows the
13C NMR spectrum, where eight signals were identified: a carbonyl carbon at δ 170.27, two quaternary aromatic carbons (C1 and C6), two aromatic signals corresponding to four CH atoms (C2, C3, C4 and C5), and three aliphatic signals, assigned through HSQC and COSY correlations.
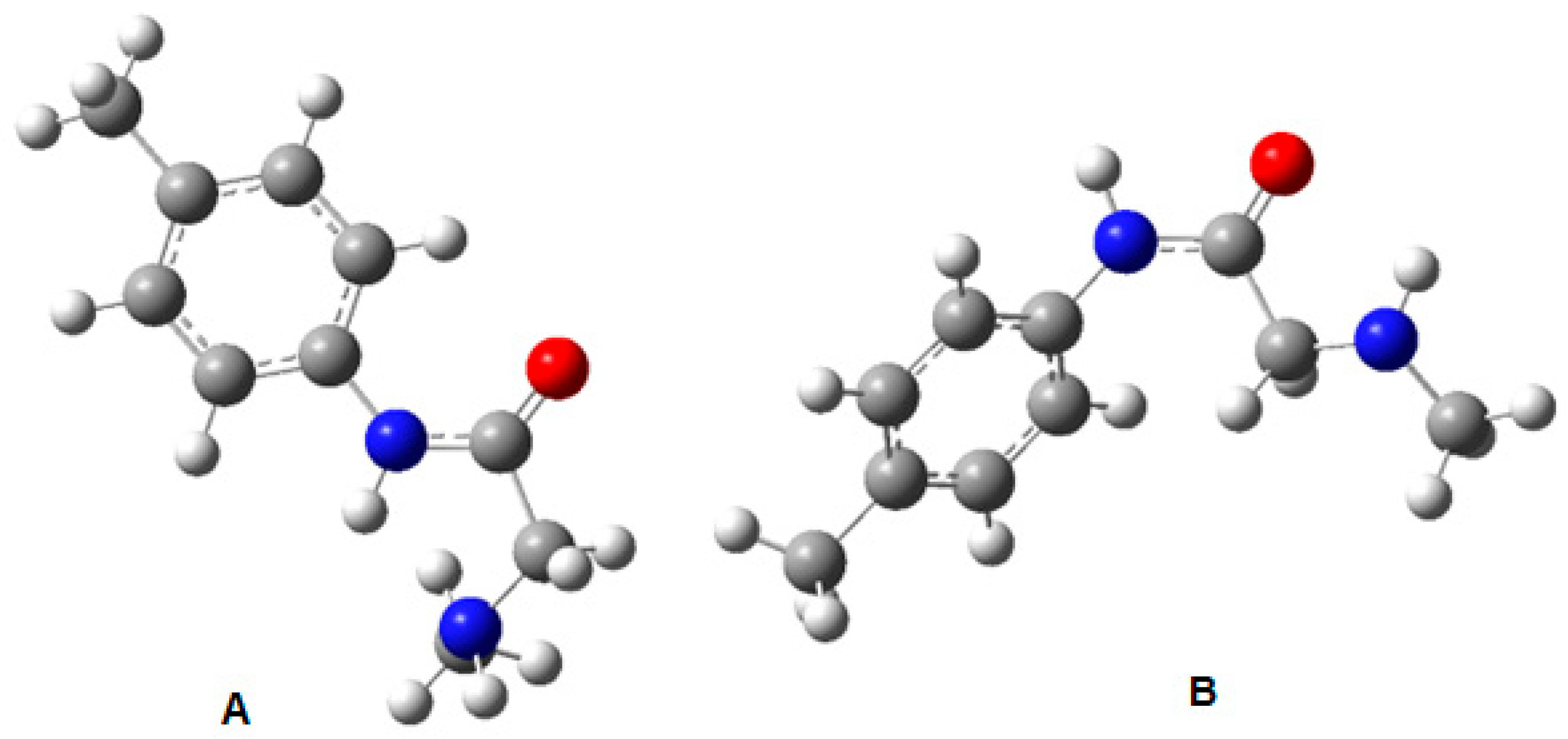
The Gaussian-optimised structures of the rotamer pair are shown in
Figure 6:
DFT calculations (B3LYP/6-311++G(d,p)) indicated that the Z rotamer is more stable than the E isomer, with a ΔG of 1.17 kcal/mol. This prediction was corroborated by comparing theoretical and experimental chemical shifts, which showed greater agreement for the Z conformation. Furthermore, the DP4+ statistical analysis assigned this rotamer a probability of 98.67%, thus validating its predominance in solution.
An analogous analysis was performed for the remaining synthesised derivatives. In all cases, the 1H and 13C NMR spectra, complemented by HSQC and COSY, displayed patterns consistent with the presence of a single main set of signals, suggesting the predominance of one rotamer.
Theoretical calculations and DP4+ analysis consistently indicated that the Z rotamer is the most stable and predominant in solution, except in the particular case of 2-chloro-N-(o-chlorophenyl)acetamide, where the probabilities were distributed between both isomers. These results confirm that the E/Z isomerism of N-substituted amides is strongly influenced by steric and electronic factors of the substituents, and that the Z geometry is generally favoured.
The combined strategy of NMR spectroscopy and DFT calculations enabled the robust assignment of the predominant rotamers in solution. The use of 2-(methylamino)-N-(p-tolyl)acetamide as a model compound allowed a detailed illustration of the correlation between experimental and theoretical data, and this behaviour was reproduced in most of the systems studied.
4. Conclusions
The successful synthesis of a series of 2-aminoacetanilides and their corresponding N′-substituted 2-aminoacetanilides was achieved by means of acylation and nucleophilic substitution procedures.
In all cases, the spectra showed the presence of a single set of signals, indicating the predominance of a single rotamer under the measurement conditions.
DFT calculations enabled the optimisation of molecular geometries and the comparison of relative energies of the possible rotamers, confirming that one of them exhibits marked stability.
Application of the DP4+ method provided a statistically reliable assignment of the observed signals, supporting the correspondence with both experimental results and theoretical simulations.
The coherence between experimental and theoretical data consolidated the validity of the conformational analysis and highlighted the relevance of rotamer predominance for future cyclisation towards benzodiazepines.
Author Contributions
Conceptualization, S.L. and L.S.; methodology, M.C., E.P. and G.P.; software, E.P. and G.P.; validation, M.C., E.P. and G.P.; formal analysis, S.L., L.S. and M.C.; investigation, E.P. and G.P.; resources, S.L. and L.S.; data curation, M.C.; writing—original draft preparation, M.C.; writing—review and editing, S.L., L.S. and M.C.; visualization, S.L., L.S. and M.C.; supervision, S.L. and L.S.; project administration, M.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Mombereau, C.; Kaupmann, K.; van der Putten, H.; Cryan, J.F. Altered response to benzodiazepine anxiolytics in mice lacking GABAB(1) receptors. Eur. J. Pharmacol. 2004, 497, 119–120. [Google Scholar] [CrossRef] [PubMed]
- Basile, A.S.; Lippa, A.S.; Skolnick, P. Anxioselective anxiolytics: Can less be more? Eur. J. Pharmacol. 2004, 500, 441–451. [Google Scholar] [CrossRef] [PubMed]
- Berezhnoy, D.; Nyfeler, Y.; Gonthier, A.; Schwob, H.; Goeldner, M.; Sigel, E. On the benzodiazepine binding pocket in GABAA receptors. J. Biol. Chem. 2004, 279, 3160–3168. [Google Scholar] [CrossRef] [PubMed]
- Olszewska, A.; Pohl, R.; Brázdová, M.; Fojta, M.; Hocek, M. Chloroacetamide-linked nucleotides and DNA for cross-linking with peptides and proteins. Bioconjugate Chem. 2016, 27, 2089–2094. [Google Scholar] [CrossRef] [PubMed]
- Wuts, P.G.M.; Greene, T.W. Greene’s Protective Groups in Organic Synthesis, 4th ed.; Wiley-Interscience: Hoboken, NJ, USA, 2007. [Google Scholar]
- Jacobs, W.A.; Heidelberger, M.; Rolf, I.P. On Certain Aromatic Amines and Chloroacetyl Derivatives. J. Am. Chem. Soc. 1919, 41, 458–474. [Google Scholar] [CrossRef]
- Speziale, A.J.; Hamm, P.C. Preparation of Some New 2-Chloroacetamides. J. Am. Chem. Soc. 1956, 78, 2556–2559. [Google Scholar] [CrossRef]
- Surrey, A.R.; Rukwid, M.K. New Amebacides. II. The Preparation of Some N-Alkyl-N-benzylhaloacetamides. J. Am. Chem. Soc. 1955, 77, 3798–3800. [Google Scholar] [CrossRef]
- Shafer, D.E.; Inman, J.K.; Lees, A. Reaction of Tris(2-Carboxyethyl)Phosphine (TCEP) with Maleimide and α-Haloacyl Groups: Anomalous Elution of TCEP by Gel Filtration. Anal. Biochem. 2000, 282, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Parr, R.G.; Yang, W. Density-Functional Theory of Atoms and Molecules; Oxford University Press: New York, NY, USA, 1989. [Google Scholar]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef] [PubMed]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).