Synthesis, Self-Assembling and Photophysical Property Exploration of Water Self-Dispersible, Grafted Poly(p-Phenylene Vinylene)s with Nonionic, Hydrophilic and Biocompatible Side Chains †
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Syntheses
2.3. Methods
3. Results and Discussion
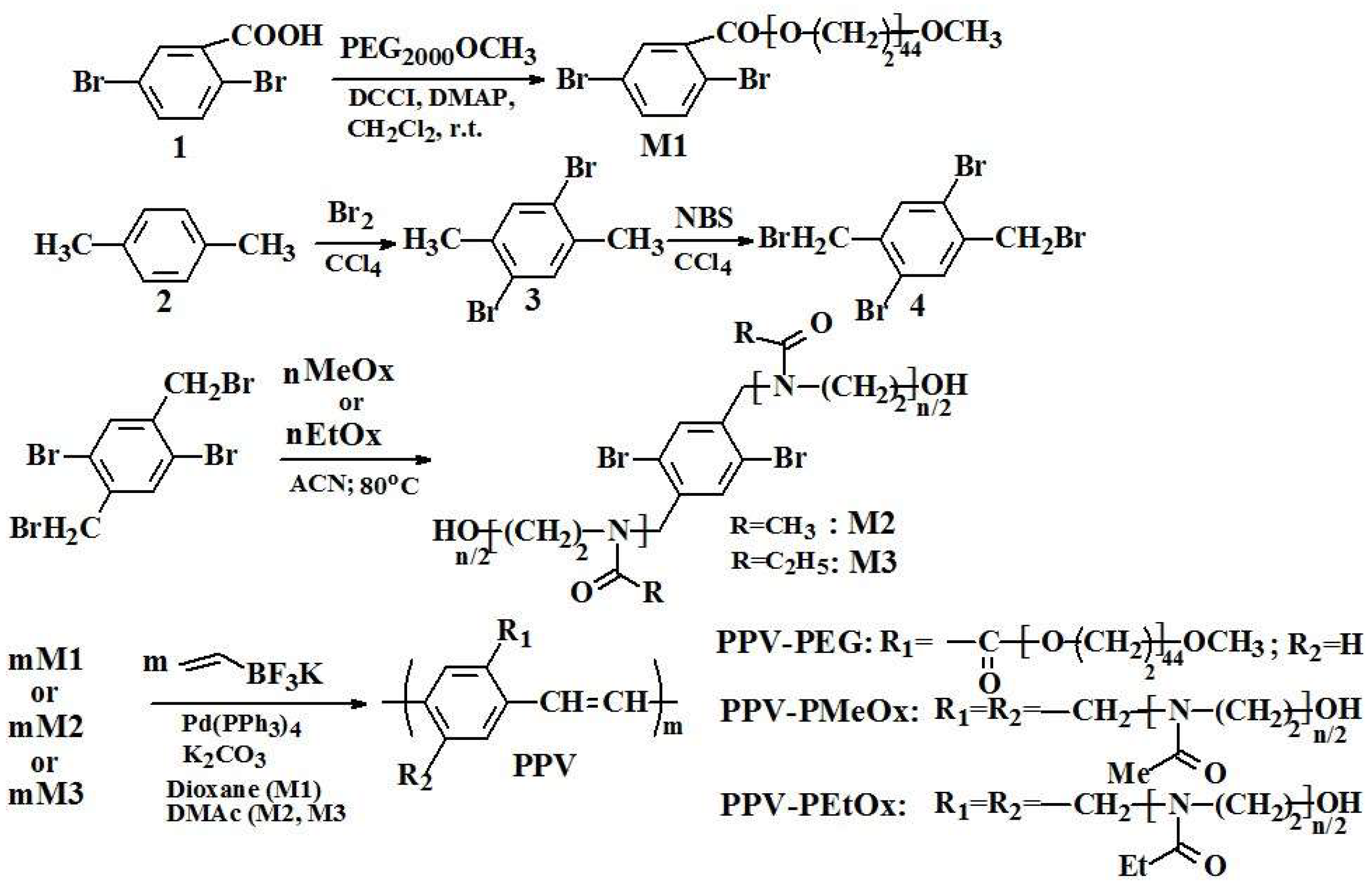
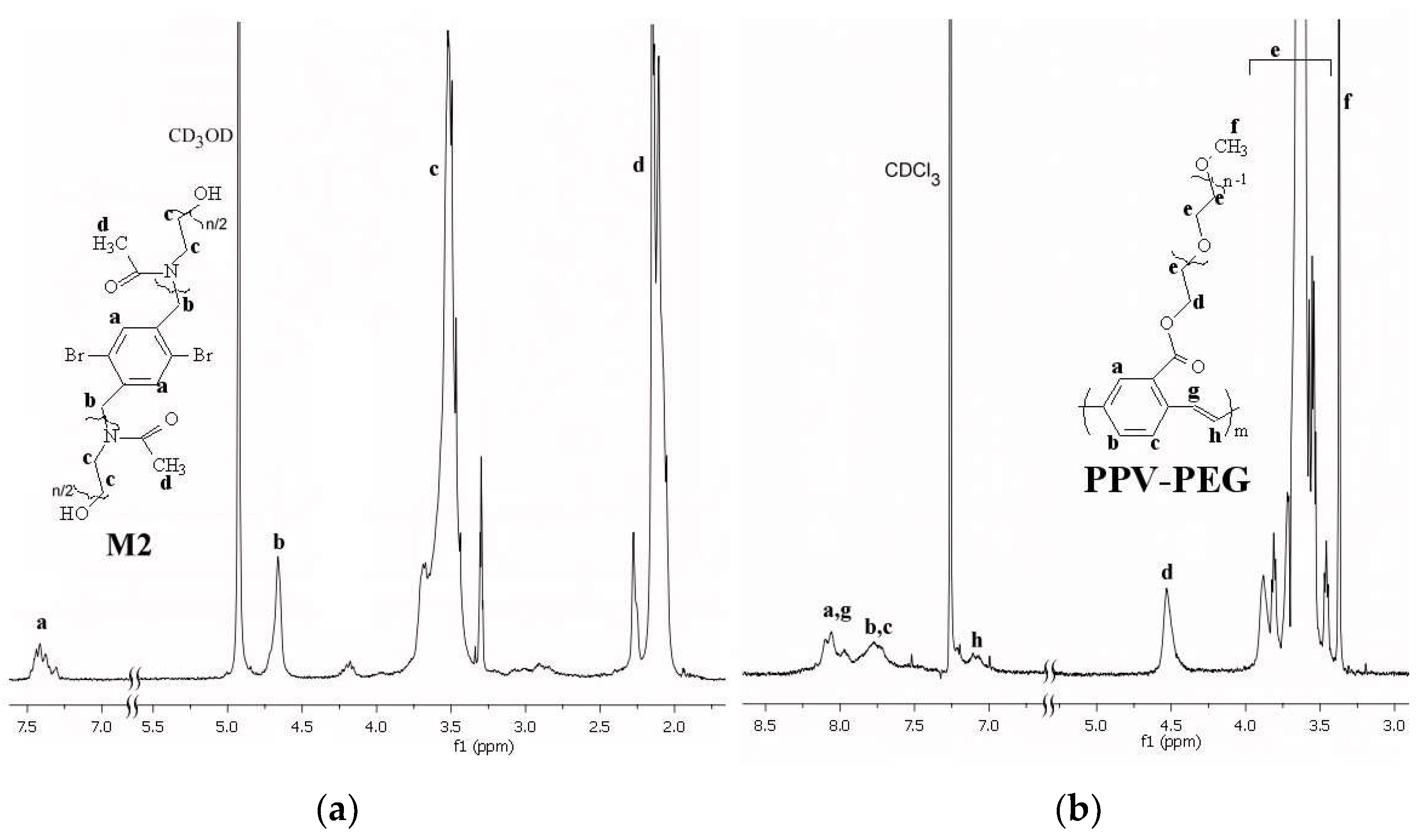
3.1. Syntheses of Macromonomers and of g-PPVs Based on Them-Structural Characterization
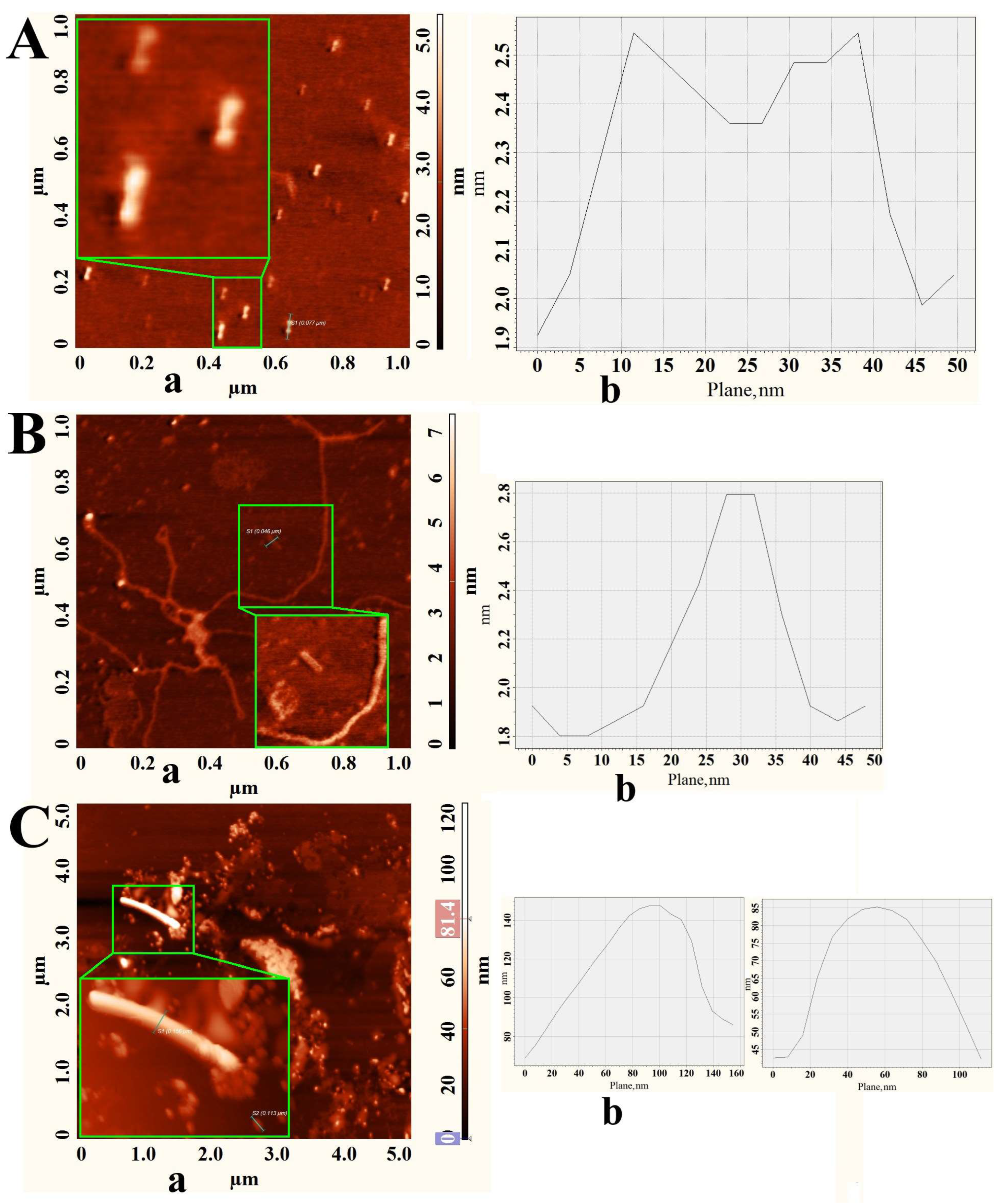
3.2. Self-Assembling of PPVs in Water by Direct Dissolution
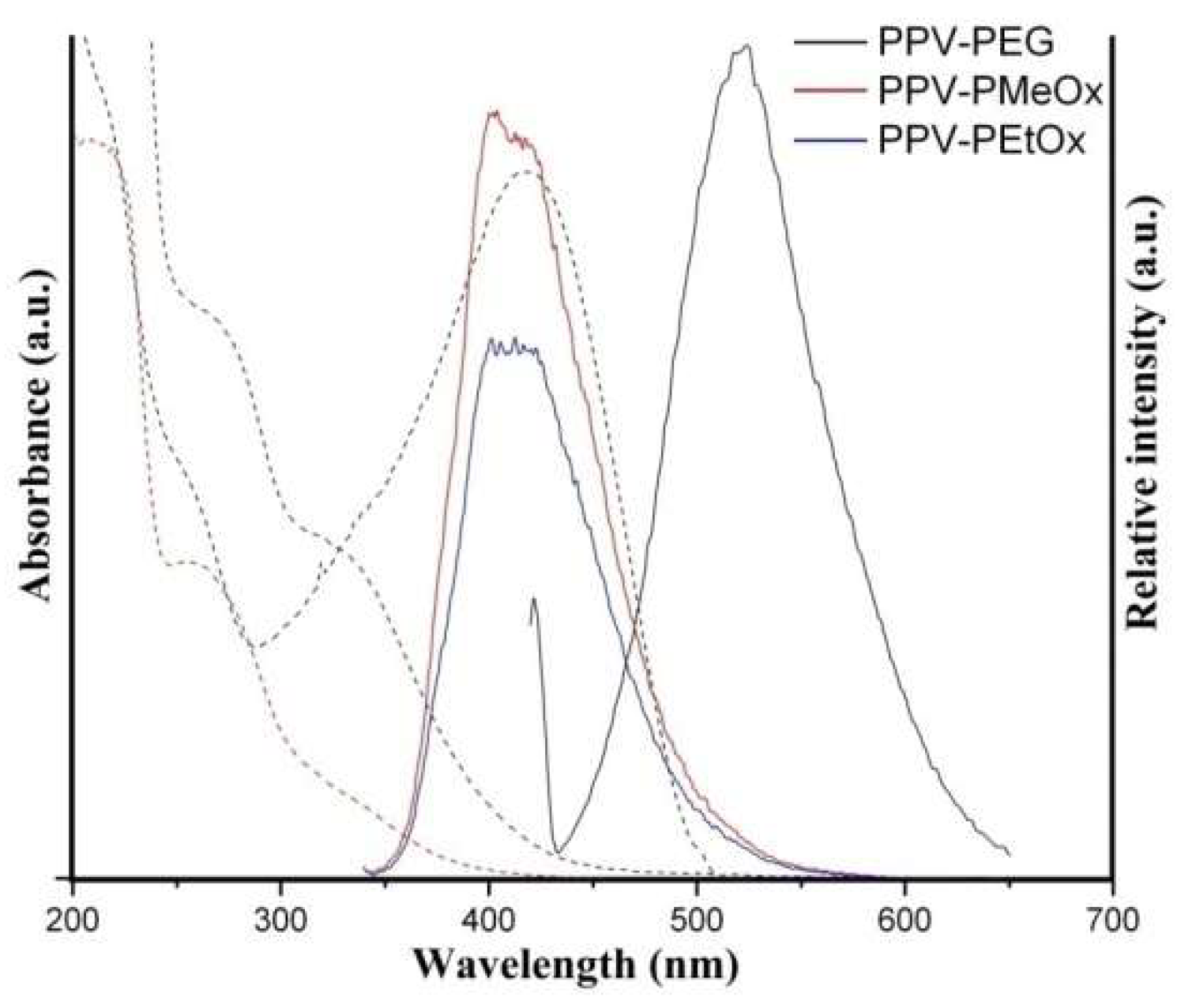
3.3. Optical Properties of the PPVs’ Self-Assembled Micellar Nanoparticles
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shan, D.; Gerhard, E.; Zhang, C.; Tierney, J.W.; Xie, D.; Liu, Z.; Yang, J. Polymeric biomaterials for biophotonic applications. Bioact. Mater. 2018, 3, 434–445. [Google Scholar] [CrossRef] [PubMed]
- Rebelo, R.; Caldas, M.; Neves, M.A.D.; Kundu, S.C.; Reis, R.L.; Correlo, V. Natural Polymers for Biophotonic Use. In Polysaccharides of Microbial Origin; Oliveira, J.M., Radhouani, H., Reis, R.L., Eds.; Springer: Cham, Switzerland, 2022. [Google Scholar] [CrossRef]
- Jiang, Y.; Pu, K. Multimodal biophotonics of semiconducting polymer nanoparticles. Acc. Chem. Res. 2018, 51, 1840–1849. [Google Scholar] [CrossRef] [PubMed]
- Burroughes, J.H.; Bradley, D.D.C.; Brown, A.R.; Marks, R.N.; Mackay, K.; Friend, R.H.; Bums, P.L.; Holmes, A.B. Light-emitting diodes based on conjugated polymers. Nature 1990, 347, 539–541. [Google Scholar] [CrossRef]
- Kim, S.; Lim, C.K.; Na, J.; Lee, Y.D.; Kim, K.; Choi, K.; Leary, J.F.; Kwon, I.C. Conjugated polymer nanoparticles for biomedical in vivo imaging. Chem. Commun. 2010, 46, 1617–1619. [Google Scholar] [CrossRef]
- Doshi, M.; Copik, A.; Gesquiere, A.J. Development and characterization of conducting polymer nanoparticles for photodynamic therapy in vitro. Photodiagn. Photodyn. Ther. 2015, 12, 476–489. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Yuan, H.; Chen, H.; Wang, X.; Zhang, P.; Lv, F.; Liu, L.; Wang, S. Cationic Poly(p-phenylene vinylene) Materials as a Multifunctional Platform for Light-Enhanced siRNA Delivery. Chem. Asian J. 2016, 11, 2686–2689. [Google Scholar] [CrossRef]
- Yang, Z.; Shen, Q.; Xing, L.; Fu, X.; Qiu, Z.; Xiang, H.; Huang, Y.; Lv, F.; Bai, H.; Huo, Y.; et al. A biophotonic device based on a conjugated polymer and a macrophage-laden hydrogel for triggering immunotherapy. Mater. Horiz. 2023, 10, 2226–2236. [Google Scholar] [CrossRef] [PubMed]
- Senthilkumar, T.; Zhou, L.; Gu, Q.; Liu, L.; Lv, F.; Wang, S. Conjugated Polymer Nanoparticles with Appended Photo-Responsive Units for Controlled Drug Delivery Release, and Imaging. Angew. Chem. Int. Ed. 2018, 57, 13114–13119. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.; Zhen, X.; Miao, Q.; Lyu, Y.; Pu, K. Self-Assembled Semiconducting Polymer Nanoparticles for Ultrasensitive Near-Infrared Afterglow Imaging of Metastatic Tumors. Adv. Mater. 2018, 30, 1801331. [Google Scholar] [CrossRef] [PubMed]
- Zhen, X.; Xie, C.; Pu, K. Temperature-Correlated Afterglow of a Semiconducting Polymer Nanococktail for Imaging-Guided Photothermal Therapy. Angew. Chem. Int. Ed. 2018, 57, 3938–3942. [Google Scholar] [CrossRef] [PubMed]
- Colak, D.G.; Cianga, I.; Demirkol, D.O.; Kozgus, O.; Medine, E.I.; Sakarya, S.; Unak, P.; Timur, S.; Yagci, Y. The synthesis and targeting of PPP-type copolymersto breast cancer cells: Multifunctional platforms for imaging and diagnosis. J. Mater. Chem. 2012, 22, 9293–9300. [Google Scholar] [CrossRef]
- Xie, C.; Zhen, X.; Lei, Q.; Ni, R.; Pu, K. Self-Assembly of Semiconducting Polymer Amphiphiles for In Vivo Photoacoustic Imaging. Adv. Funct. Mater. 2017, 27, 1605397. [Google Scholar] [CrossRef]
- Bendrea, A.-D.; Cianga, L.; Ailiesei, G.-L.; Colak, D.G.; Popescu, I.; Cianga, I. Thiophene α-Chain-End-Functionalized Oligo(2-methyl-2-oxazoline) as Precursor Amphiphilic Macromonomer for Grafted Conjugated Oligomers/Polymers and as a Multifunctional Material with Relevant Properties for Biomedical Applications. Int. J. Mol. Sci. 2022, 23, 7495. [Google Scholar] [CrossRef]
- Grisorio, R.; Mastrorilli, P.; Nobile, C.F.; Romanazzi, G.; Suranna, G.P. A novel synthetic protocol for poly(fluorenylenevinylene)s: A cascade Suzuki–Heck reaction. Tetrahedron Lett. 2005, 46, 2555–2558. [Google Scholar] [CrossRef]
- Damavandi, M.; Pilkington, L.I.; Whitehead, K.A.; Wilson-Nieuwenhuis, J.; McBrearty, J.; Dempsey-Hibbert, N.; Travis-Sejdic, J.; Barker, D. Poly(para-phenyleneethynylene) (PPE)- and poly(para-phenylene vinylene) (PPV)-poly[(2-(methacryloyloxy)ethyl) trimethylammonium chloride] (PMETAC) graft copolymers exhibit selective antimicrobial activity. Eur. Polym. J. 2018, 98, 368–374. [Google Scholar] [CrossRef]
- Cianga, I.; Yagci, Y. Synthesis and characterization of comb-like polyphenylenes viaSuzuki coupling of polystyrene macromonomers prepared by atom transfer radical polymerization. Eur. Polym. J. 2002, 38, 695–703. [Google Scholar] [CrossRef]
- Wong, W.W.H.; Banal, J.L. Poly(arylene-vinylene)s. In Encyclopedia of Polymeric Nanomaterials; Kobayashi, S., Müllen, K., Eds.; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar] [CrossRef]
- Colak, D.G.; Egbe, D.A.M.; Birckner, E.; Yurteri, S.; Cianga, I.; Tekin, E.; Schubert, U.S.; Yagci, Y. Photophysical properties of PPP and PPV derivatives bearing polystyrene or polycaprolactone as side groups. Eur. Polym. J. 2009, 45, 940–945. [Google Scholar] [CrossRef]
- Colak, D.G.; Cianga, I.; Yagci, Y.; Cirpan, A.; Karasz, F.E. Novel Poly(phenyle vinylenes) with Well-Defined Poly(e-caprolactone) or Polystyrene as Lateral Substituents: Synthesis and Characterization. Macromolecules 2007, 40, 5301–5310. [Google Scholar] [CrossRef]
- Kerr-Phillips, T.; Damavandi, M.; Pilkington, L.I.; Whitehead, K.A.; Travas-Sejdic, J.; Barker, D. Effects of Neutral, Anionic and Cationic Polymer Brushes Grafted from Poly(para-phenylene vinylene) and Poly(para-phenylene ethynylene) on the Polymer’s Photoluminescent Properties. Polymers 2022, 14, 2767. [Google Scholar] [CrossRef] [PubMed]
| Sample | Mn,GPC (IPD) a | Mn,GPC (IPD) b |
|---|---|---|
| PPV-PEG | 3825 (1.13) | 44,580 (1.13) |
| PPV-PMeOx PPV-PEtOx | 276 * (1.02) 705 * (1.07) | 8834 (1.07) 10,950 (1.1) |
| Sample | Concentration (mg/mL) | Size (nm) | λabs/λem (nm) a |
|---|---|---|---|
| PPV-PEG | 0.001 | - * | nd |
| 0.01 | 6.1; 806 | 418/524 | |
| 0.1 | - * | nd | |
| PPV-PMeOx | 0.001 | 600; 4830 | nd |
| 0.01 0.1 1 | - * 28 373; 5290 | nd 331/403 nd | |
| PPV-PEtOx | 0.001 | 72.5; 4960 | nd |
| 0.01 0.1 1 | 16.5; 4430 1.58; 134 383; 4030 | nd 267/403; 420 nd |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bendrea, A.-D.; Göen-Colak, D.; Cianga, L.; Hitruc, E.-G.; Cianga, I.; Pinteala, M. Synthesis, Self-Assembling and Photophysical Property Exploration of Water Self-Dispersible, Grafted Poly(p-Phenylene Vinylene)s with Nonionic, Hydrophilic and Biocompatible Side Chains. Chem. Proc. 2024, 16, 73. https://doi.org/10.3390/ecsoc-28-20198
Bendrea A-D, Göen-Colak D, Cianga L, Hitruc E-G, Cianga I, Pinteala M. Synthesis, Self-Assembling and Photophysical Property Exploration of Water Self-Dispersible, Grafted Poly(p-Phenylene Vinylene)s with Nonionic, Hydrophilic and Biocompatible Side Chains. Chemistry Proceedings. 2024; 16(1):73. https://doi.org/10.3390/ecsoc-28-20198
Chicago/Turabian StyleBendrea, Anca-Dana, Demet Göen-Colak, Luminita Cianga, Elena-Gabriela Hitruc, Ioan Cianga, and Mariana Pinteala. 2024. "Synthesis, Self-Assembling and Photophysical Property Exploration of Water Self-Dispersible, Grafted Poly(p-Phenylene Vinylene)s with Nonionic, Hydrophilic and Biocompatible Side Chains" Chemistry Proceedings 16, no. 1: 73. https://doi.org/10.3390/ecsoc-28-20198
APA StyleBendrea, A.-D., Göen-Colak, D., Cianga, L., Hitruc, E.-G., Cianga, I., & Pinteala, M. (2024). Synthesis, Self-Assembling and Photophysical Property Exploration of Water Self-Dispersible, Grafted Poly(p-Phenylene Vinylene)s with Nonionic, Hydrophilic and Biocompatible Side Chains. Chemistry Proceedings, 16(1), 73. https://doi.org/10.3390/ecsoc-28-20198







