A One Pot Synthesis of Diketopiperazines via Multicomponent Reactions Based on Isocyanides †
Abstract
1. Introduction
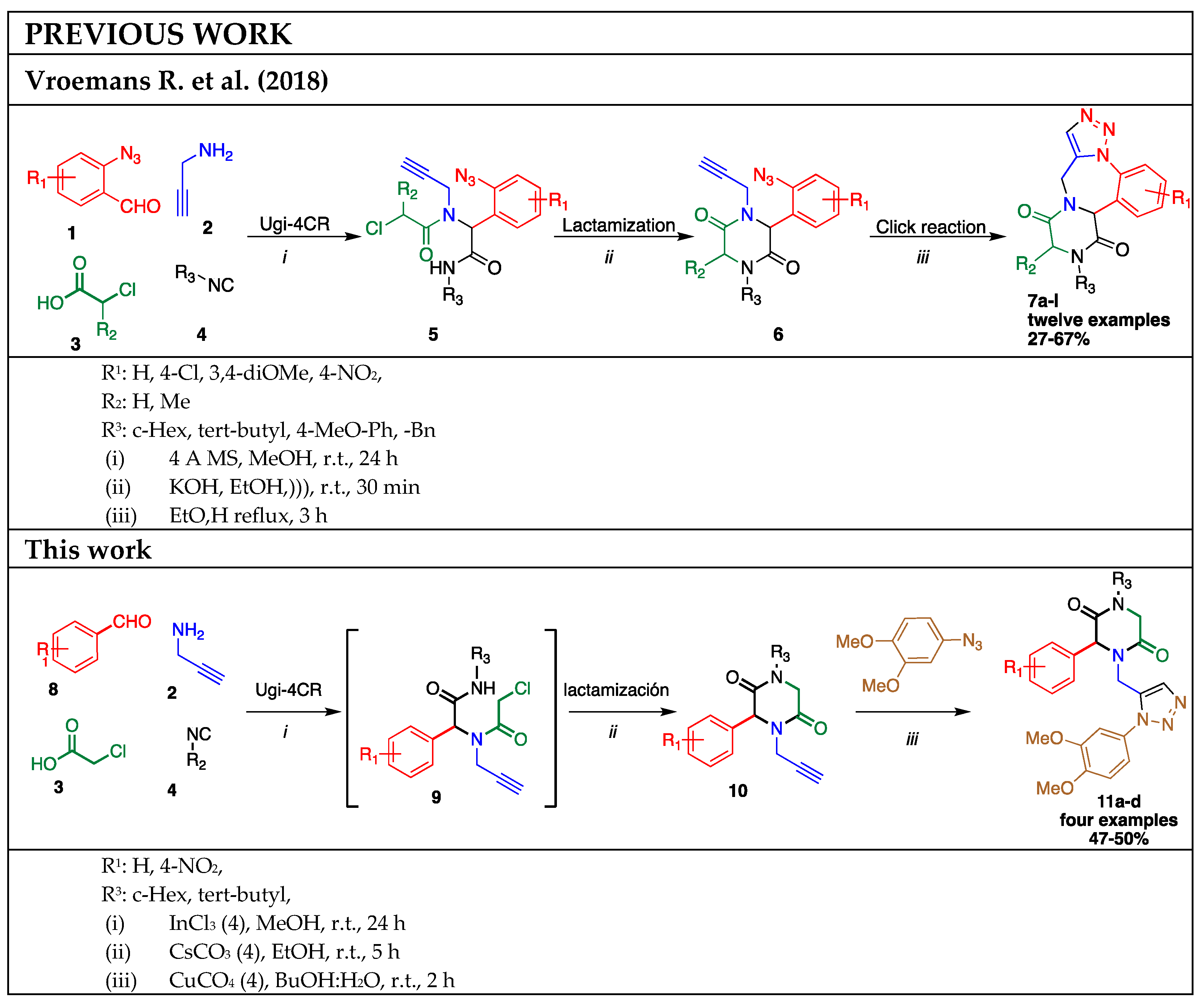
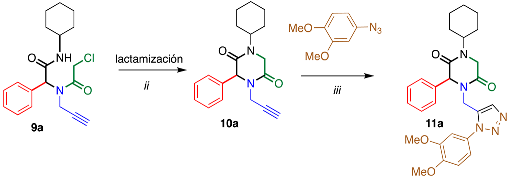
2. Results and Discussion
3. Experimental Section
3.1. General Information, Instrumentation and Chemicals
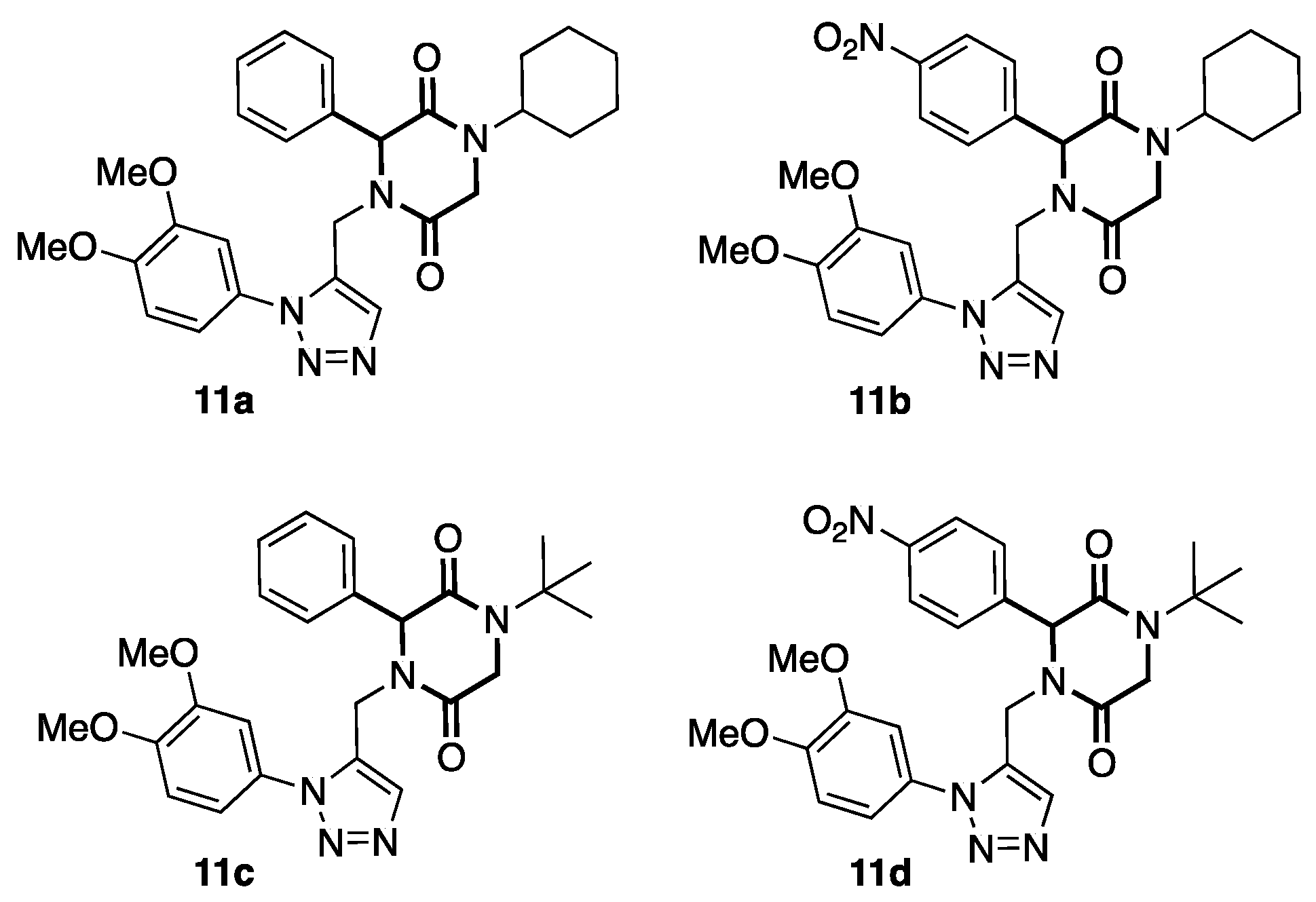
3.2. General Procedure (11a–d)
3.3. Spectral Data
3.3.1. 1-cyclohexyl-4-((1-(3,4-dimethoxyphenyl)-1H-1,2,3-triazol-4-yl) methyl)-3-phenylpiperazine-2,5-dione (11a)

3.3.2. 1-cyclohexyl-4-((1-(3,4-dimethoxyphenyl)-1H-1,2,3-triazol-4-yl) methyl)-3-(4-nitrophenyl) piperazine-2,5-dione (11b)

3.3.3. 1-(tert-butyl)-4-((1-(3,4-dimethoxyphenyl)-1H-1,2,3-triazol-4-yl) methyl)-3-phenylpiperazine-2,5-dione (11c)
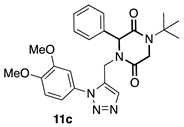
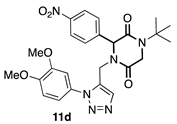
3.3.4. 1-(tert-butyl)-4-((1-(3,4-dimethoxyphenyl)-1H-1,2,3-triazol-4-yl) methyl)-3-(4-nitrophenyl) piperazine-2,5-dione (11d)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mendes, L.L.; Varejão, J.O.S.; de Souza, J.A.; de Carneiro, J.W.M.; Valdo, A.K.S.M.; Martins, F.T.; Ferreira, B.W.; Barreto, R.W.; da Silva, T.I.; Kohlhoff, M.; et al. 2,5-Diketopiperazines via Intramolecular N-Alkylation of Ugi Adducts: A Contribution to the Synthesis, Density Functional Theory Study, X-ray Characterization, and Potential Herbicide Application. J. Agric. Food Chem. 2022, 70, 1799–1809. [Google Scholar] [CrossRef] [PubMed]
- Bojarska, J.; Mieczkowski, A.; Ziora, Z.M.; Skwarczynski, M.; Toth, I.; Shalash, A.O.; Parang, K.; El-Mowafi, S.A.; Mohammed, E.H.M.; Elnagdy, S.; et al. Cyclic Dipeptides: The Biological and Structural Landscape with Special Focus on the Anti-Cancer Proline-Based Scaffold. Biomolecules 2021, 11, 1515. [Google Scholar] [CrossRef] [PubMed]
- Pharande, S.G.; Rentería-Gómez, M.A.; Gámez-Montaño, R. Mechanochemical IMCR and IMCR-Post Transformation Domino Strategies: Towards the Sustainable DOS of Dipeptide-like and Heterocyclic Peptidomimetics. New J. Chem. 2022, 46, 9298–9303. [Google Scholar] [CrossRef]
- Unnamatla, M.V.B.; Islas-Jácome, A.; Quezada-Soto, A.; Ramírez-López, S.C.; Flores-Álamo, M.; Gámez-Montaño, R. Multicomponent One-Pot Synthesis of 3-Tetrazolyl and 3-Imidazo [1,2-a]Pyridin Tetrazolo [1,5-a]Quinolines. J. Org. Chem. 2016, 81, 10576–10583. [Google Scholar] [CrossRef] [PubMed]
- Rentería-Gómez, M.A.; Islas-Jácome, A.; Pharande, S.G.; Vosburg, D.A.; Gámez-Montaño, R. Synthesis of Tris-Heterocycles via a Cascade IMCR/Aza Diels-Alder + CuAAC Strategy. Front. Chem. 2019, 7, 1. [Google Scholar] [CrossRef] [PubMed]
- Pharande, S.G.; Corrales Escobosa, A.R.; Gámez-Montaño, R. Endogenous Water-Triggered and Ultrasound Accelerated Synthesis of 1,5-Disubstituted Tetrazoles via a Solvent and Catalyst-Free Ugi-Azide Reaction. Green Chem. 2017, 19, 1259–1262. [Google Scholar] [CrossRef]
- Vroemans, R.; Bamba, F.; Winters, J.; Thomas, J.; Jacobs, J.; Van Meervelt, L.; John, J.; Dehaen, W. Sequential Ugi Reaction/Base-Induced Ring Closing/IAAC Protocol toward Triazolobenzodiazepine-Fused Diketopiperazines and Hydantoins. Beilstein J. Org. Chem. 2018, 14, 626–633. [Google Scholar] [CrossRef] [PubMed]
- Kaveti, B.; Ramírez-López, S.C.; Gámez Montaño, R. Ultrasound-Assisted Green One-Pot Synthesis of Linked Bis-Heterocycle Peptidomimetics via IMCR/Post-Transformation/Tandem Strategy. Tetrahedron Lett. 2018, 59, 4355–4358. [Google Scholar] [CrossRef]
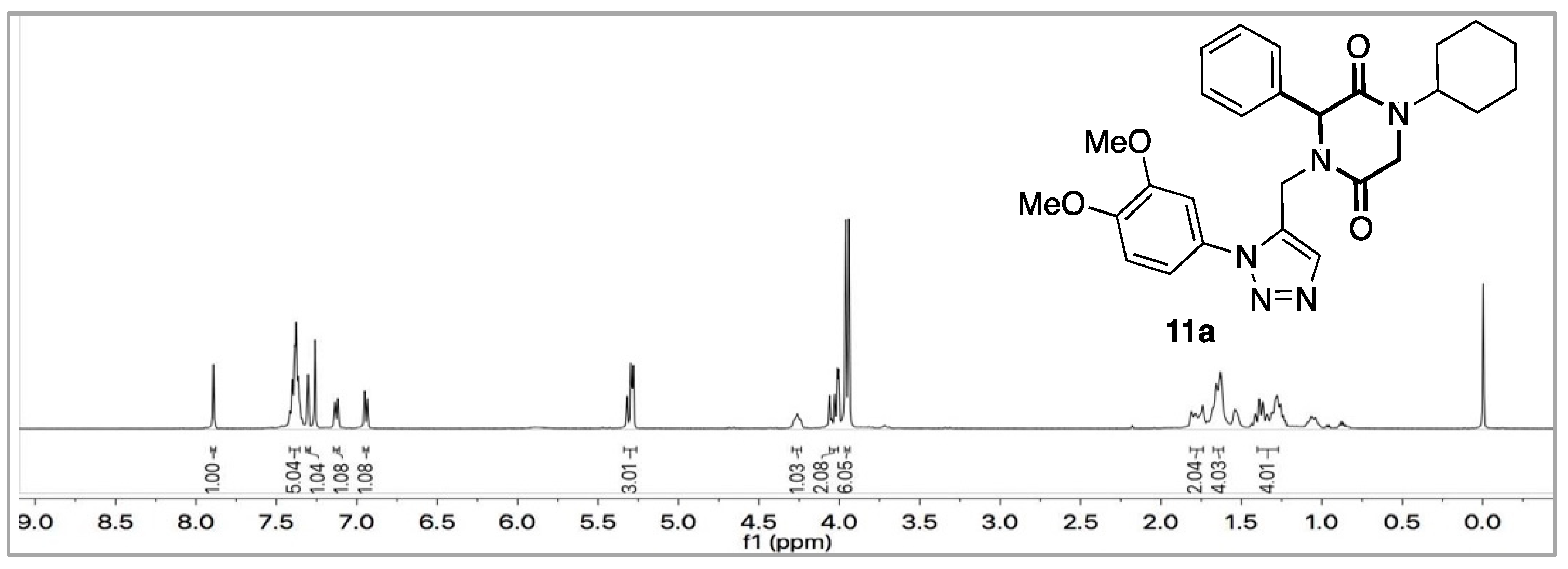
 | ||||
| (i)—Lactamization (entries 1–2) | ||||
| Entry | Solvent | Base | Time | Conversion |
| 1 | EtOH | KOH a | 1 h | Decomp |
| 2 | EtOH | Cs2CO3 a | 5 h | C |
| (ii)—Click reaction (entries 3–4) | ||||
| Entry | Solvent | Catalyst | Time | Yield 11a (%) |
| 3 | tBuOH:H2O | Cu(OAc)2 a,b | 4 h | 40 |
| 4 | tBuOH:H2O | CuSO4 a,b | 2 h | 50 |
| a = stirring, b = with sodium ascorbate, C = conversion, decomp = decomposition. | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corona-Díaz, A.; García García, D.G.; Pharande, S.G.; Rentería-Gómez, M.A.; Gámez-Montaño, R. A One Pot Synthesis of Diketopiperazines via Multicomponent Reactions Based on Isocyanides. Chem. Proc. 2022, 12, 79. https://doi.org/10.3390/ecsoc-26-13648
Corona-Díaz A, García García DG, Pharande SG, Rentería-Gómez MA, Gámez-Montaño R. A One Pot Synthesis of Diketopiperazines via Multicomponent Reactions Based on Isocyanides. Chemistry Proceedings. 2022; 12(1):79. https://doi.org/10.3390/ecsoc-26-13648
Chicago/Turabian StyleCorona-Díaz, Alejandro, Diana G. García García, Shirikant G. Pharande, Manuel A. Rentería-Gómez, and Rocío Gámez-Montaño. 2022. "A One Pot Synthesis of Diketopiperazines via Multicomponent Reactions Based on Isocyanides" Chemistry Proceedings 12, no. 1: 79. https://doi.org/10.3390/ecsoc-26-13648
APA StyleCorona-Díaz, A., García García, D. G., Pharande, S. G., Rentería-Gómez, M. A., & Gámez-Montaño, R. (2022). A One Pot Synthesis of Diketopiperazines via Multicomponent Reactions Based on Isocyanides. Chemistry Proceedings, 12(1), 79. https://doi.org/10.3390/ecsoc-26-13648






