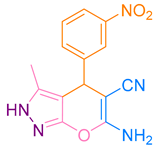
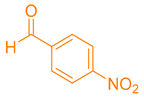
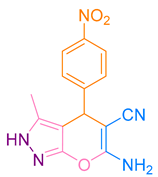
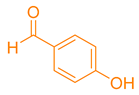
Abstract
Graphitic carbon nitride-supported L-arginine (g-C3N4@L-arginine) has been prepared as a heterogeneous catalyst for synthesizing heterocyclic compounds such as pyranopyrazole and acridinedione derivatives. High efficiency, short reaction time, and easy separation are significant features that are reasons for using g-C3N4@L-arginine as a catalyst in one-pot multicomponent reactions. Synthesized nanocatalyst was detected by numerous analyses, such as FE-SEM (Field Emission Scanning Electron Microscopy), EDX (Energy Dispersive X-ray spectroscopy), XRD (X-Ray Diffraction analysis), TGA (Thermo Gravimetric Analysis), and FT-IR (Fourier Transform Infrared Spectroscopy). G-C3N4@L-arginine nanocatalyst was reused 5 times in the reaction with no apparent decrease in reaction yield, which shows acceptable recyclability.
1. Introduction
In the last decades, heterogeneous catalysts have been noticed because of large-scale production and selective product formation [1,2]. G-C3N4 is a widely used support for catalytic entities due to high physical and thermal stability, low density, versatile performance, and recyclability. Moreover, the preparation of g-C3N4 is mostly performed by Cyanamid, urea, dicyanamide, melamine, and thiourea as the precursor [3]. To increase the efficiency of the catalytic performance of g-C3N4 in organic reactions, it is suggested to modify it with organic compounds [4,5,6,7,8].
Significantly, L-arginine is a semi-essential amino acid in living organisms [9], while the guanidine group in L-arginine is the precursor for synthesizing nitrogen derivatives. Using L-arginine with g-C3N4 as a catalyst support can decrease the cost and toxicity. Among other benefits of composite productions with L-arginine, it should be mentioned that making composite with this amino acid can increase thermal stability and molar heat capacities. Although, on the other hand, it can reduce the thermal expansion coefficient. Moreover, the utilization of composites is one of the best ways for synthesizing heterocyclic compounds [10,11,12,13], while heterocyclic compounds have been considered essential groups of organic materials. In addition, they have biological activities which could be effective in the treatment of different diseases. What makes these compounds more important than others is their application in various fields such as medicines, veterinary products, disinfectants, and antioxidants. There are several ways of synthesizing heterocyclic compounds including the multi-stages and one-pot multicomponent reactions. Lately, projects indicate that multicomponent reactions could be the best way for preparing heterocyclic compounds.
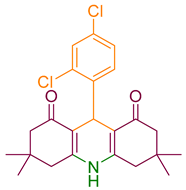
Multicomponent reactions have been mostly used for producing heterocyclic compounds because of their advantages including step efficiency, atom economy, and reducing the waste production [14,15,16,17,18]. Pyranopyrazoles are nitrogen-containing heterocyclic compounds with various properties such as anti-cancer, anti-inflammatory, anti-bacterial, antioxidant, and antihypertensive. Knoevenagel condensation, Micheal addition, and cyclization are the main procedures for making pyranopyrazoles derivatives. Various catalysts can be utilized to prepare pyranopyrazole and its derivatives by multicomponent reactions such as cetyltrimethylammonium chloride (CTACl), montmorillonite K10, agave leaf ash, cytosine@MCM-41, Et3N, and PTSA [19,20,21,22,23,24].
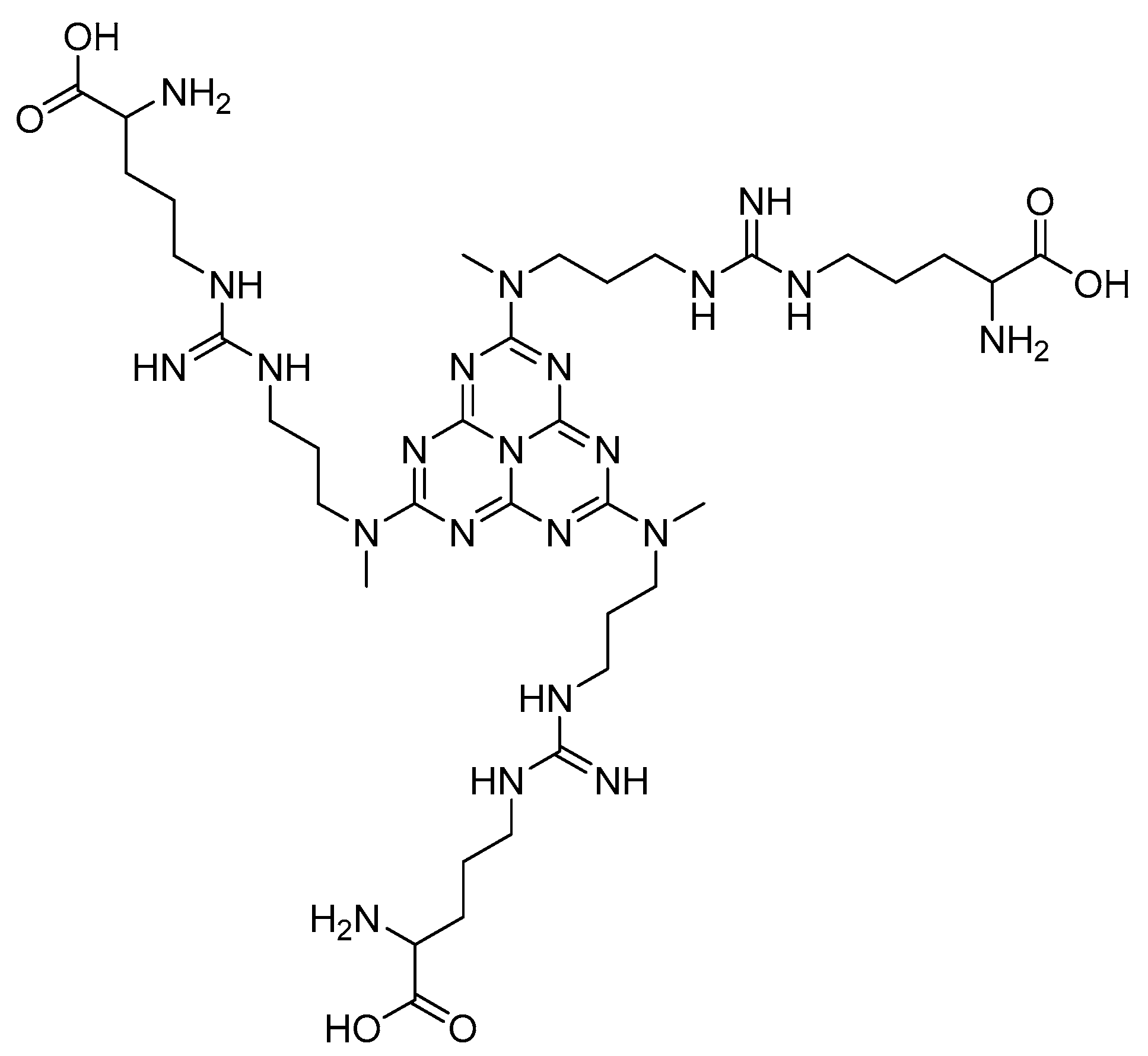
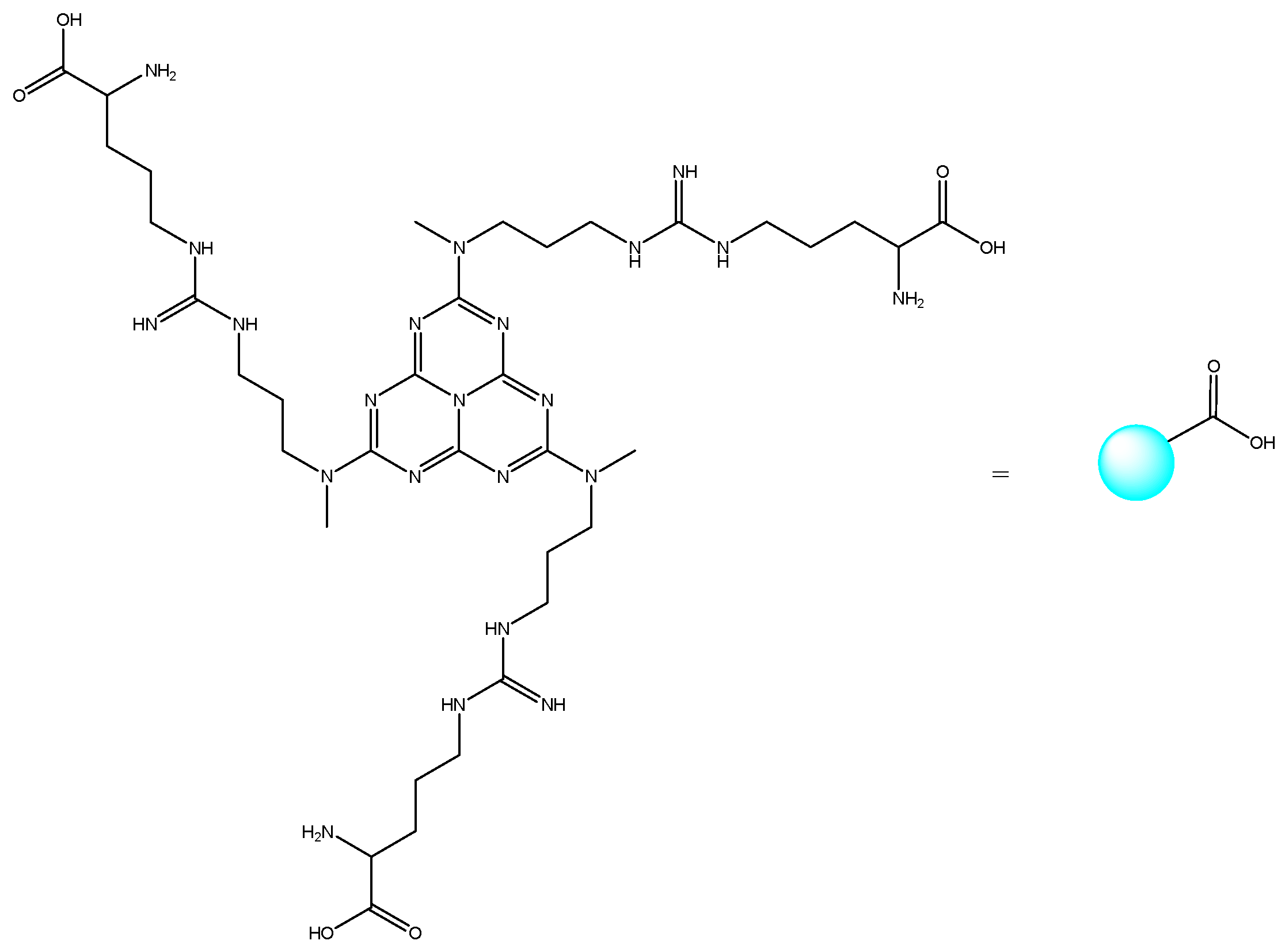
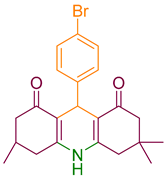
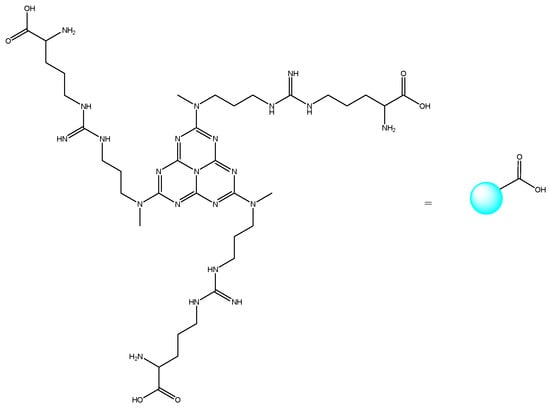
Other heterocyclic compounds with biological activities that can be produced with multicomponent reactions are Acridinedione derivatives [25]. They are nitrogen-mediated heterocyclic compounds with a vast spectrum of pharmaceutical and biological activities, namely anti-tumor, SIRT1 inhibitors, anticancer, and antimicrobial agents [26,27,28,29]. There are different precursors such as heterogeneous catalysts for preparing acridinedione, including f-MWCNT, Amberlyst -15, CTAB, and Proline [30,31,32,33]. Usually, recent methods can cover problems of the latest projects such as harsh conditions, long reaction time, and using toxic solvents. Therefore, new methods for synthesizing pyranopyrazole and acridinedione derivatives are a critical challenge in chemistry society. Consequently, in this research, we have synthesized g-C3N4@L-arginine nanocomposite and applied it as a catalyst in the synthesizing pyranopyrazole and acridinedione derivatives in a high yield. The schematic of g-C3N4@L-arginine is shown in Scheme 1.
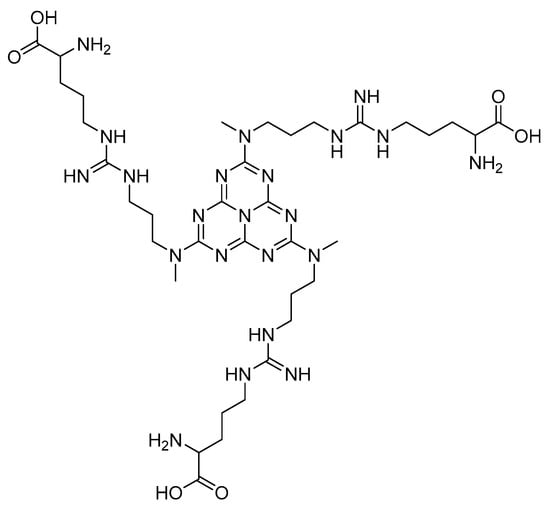
Scheme 1.
Schematic of g-C3N4@L-arginine.
2. Experimental
2.1. Materials
All chemicals were obtained from Sigma–Aldrich and Merck companies. Many analyses have been performed, including Fourier Transform Infrared Spectroscopy (FT-IR), which was recorded by Tensor27 for detecting functional groups of products; Thermal Gravimetric Analysis (TGA) under argon atmosphere was taken by STA 504, which displayed the thermal stability of nanocatalyst; Nuclear Magnetic Resonance (NMR) with Varian-Inova 500 MHz, X-Ray Powder Diffraction (XRD) was performed by Dron-8; Energy-Dispersive X-ray (EDX) Numerix DXP–X10P was employed for indicating the existence of elements of synthesized nanocatalyst; and Field Emission Scanning Electron Microscopy (FE-SEM) with TESCAN-MIRA lll was used for displaying the morphology of synthesized nanocatalyst.
2.2. Preparation of Bulk C3N4 and g-C3N4
Melamine is precursor for preparing bulk carbon nitride, which was heated to 550 °C temperature by the ramp of 2.5 °C.min−1 in a furnace for 4 h. Eventually, a yellow powder was formed. Then, for preparing g-C3N4, 1.0 g bulk C3N4 was stirred with 20 mL H2SO4 at 90 °C for 5 h. Afterward, the mixture was diluted with 200 mL ethanol and stirred at room temperature for 2 h. Then, the mixture was dispersed in 100 mL water/isopropanol (1:1), sonicated for 6 h, and centrifuged to obtain g-C3N4.
2.3. Preparation of g-C3N4@L-Arginine
A total of (1.0 g) g-C3N4 with (20.0 mL) dry toluene was dispersed. Then, (2.0 mL) 1,3-dibromopropane was poured into the final mixture and refluxed for 24 h under an N2 atmosphere. After filtration and washing with ethyl acetate, the product was dried at room temperature. The final product was dissolved in a mixture of water and methanol (1:1). Then, each of the following ingredients was added, respectively: L-arginine (1 mmol), K2CO3 (1.0 mmol), and NaI (1.0 mmol)). Afterward, it was stirred for 24 h at room temperature, washed with water and methanol, then dried at 80 °C.
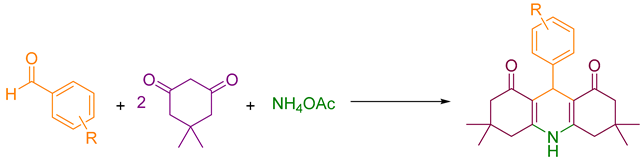
2.4. Synthesizing Acridinedione Derivatives
A mixture of dimedone (2 mmol), ammonium acetate (1 mmol), aromatic aldehyde (1 mmol), ethanol (5 mL), and catalyst (0.18 mol %) was poured into a flask and refluxed for the appropriate time. The reaction progress was monitored by TLC. After completion of the reaction, the mixture was cooled to room temperature, the catalyst was filtered, and the intended product was obtained by crystallization.
2.5. Synthesizing Pyranopyrazole Derivatives
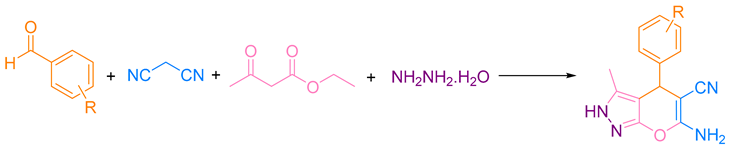
A mixture of aldehyde (1.0 mmol), ethyl acetoacetate (1.0 mmol), hydrazine hydrate (1.0 mmol), malononitrile (1.0 mmol), catalyst (0.18 mol %), and ethanol (2.0 mL) was poured into a 25 mL round bottom flask and refluxed for the appropriate time. The reaction progress was monitored by TLC. After completion of the reaction, the mixture was cooled to room temperature, the catalyst was filtered, and the intended product was obtained by crystallization.
3. Results and Discussion
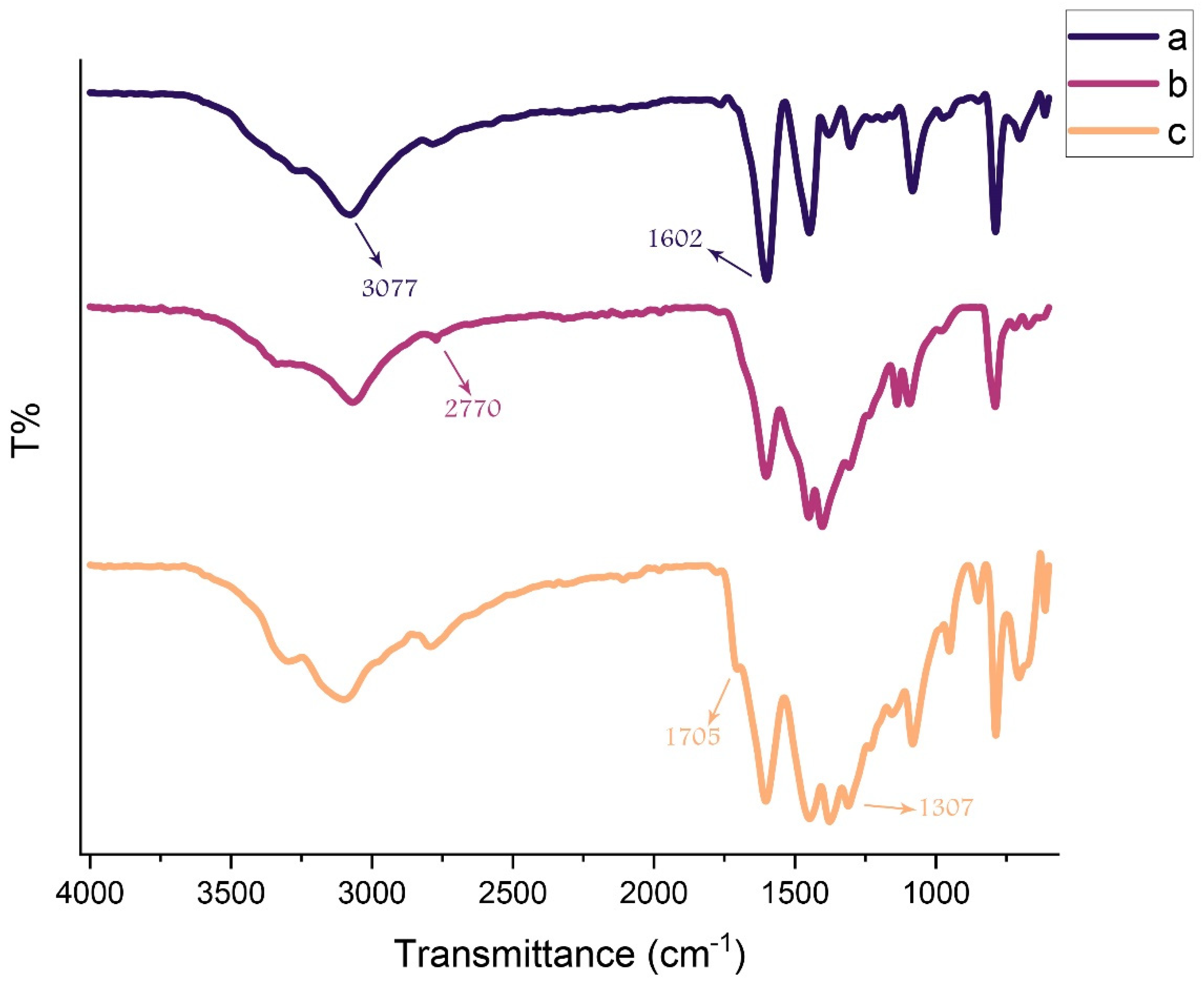
FT-IR spectra of a) g-C3N4, b) modified g-C3N4, and c) g-C3N4@L-arginine are shown in Figure 1. In Figure 1a, there is a broad peak around 3000–3300 cm−1 for N-H group stretching vibrations which is related to H- bonding or actually the existence of the OH group of water adsorption by g-C3N4 nanosheets. Figure 1b demonstrates the modified g-C3N4 nanosheets around 3000–2800 cm−1 which is related to C-H stretching vibrations. In Figure 1c, stretching vibrations of C=O and C-O were shown at (1705 cm−1) and (1320–1210 cm−1), respectively. A peak around 1602 cm−1 indicates carbon double bond nitrogen and its stretching vibrations. Values of 1303 and 1082 cm−1 are related to the C-N bond stretching vibrations which are formed from triazine and N-H groups. The C-N stretching vibrations in the ring are significantly revealed at 1448 and 1379 cm−1. A value of 786 cm−1 was shown because of tri-s-triazine vibrations. According to the mentioned peaks, g-C3N4@L-arginine was synthesized [3,4].
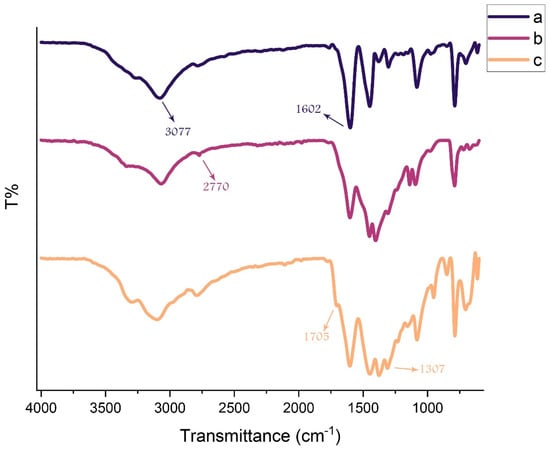
Figure 1.
FT-IR spectra of (a) g-C3N4, (b) modified g-C3N4, and (c) g-C3N4@L-arginine.
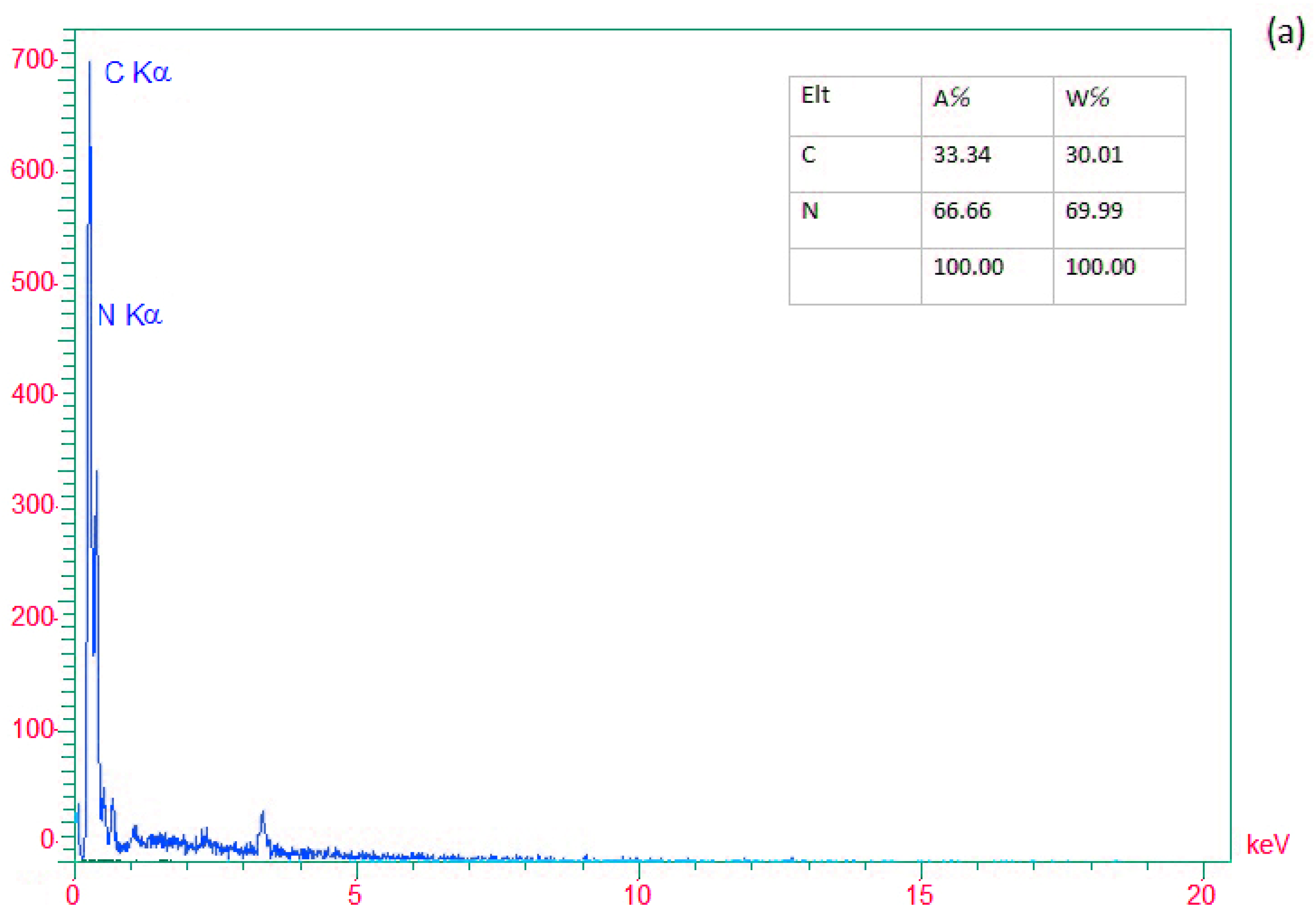
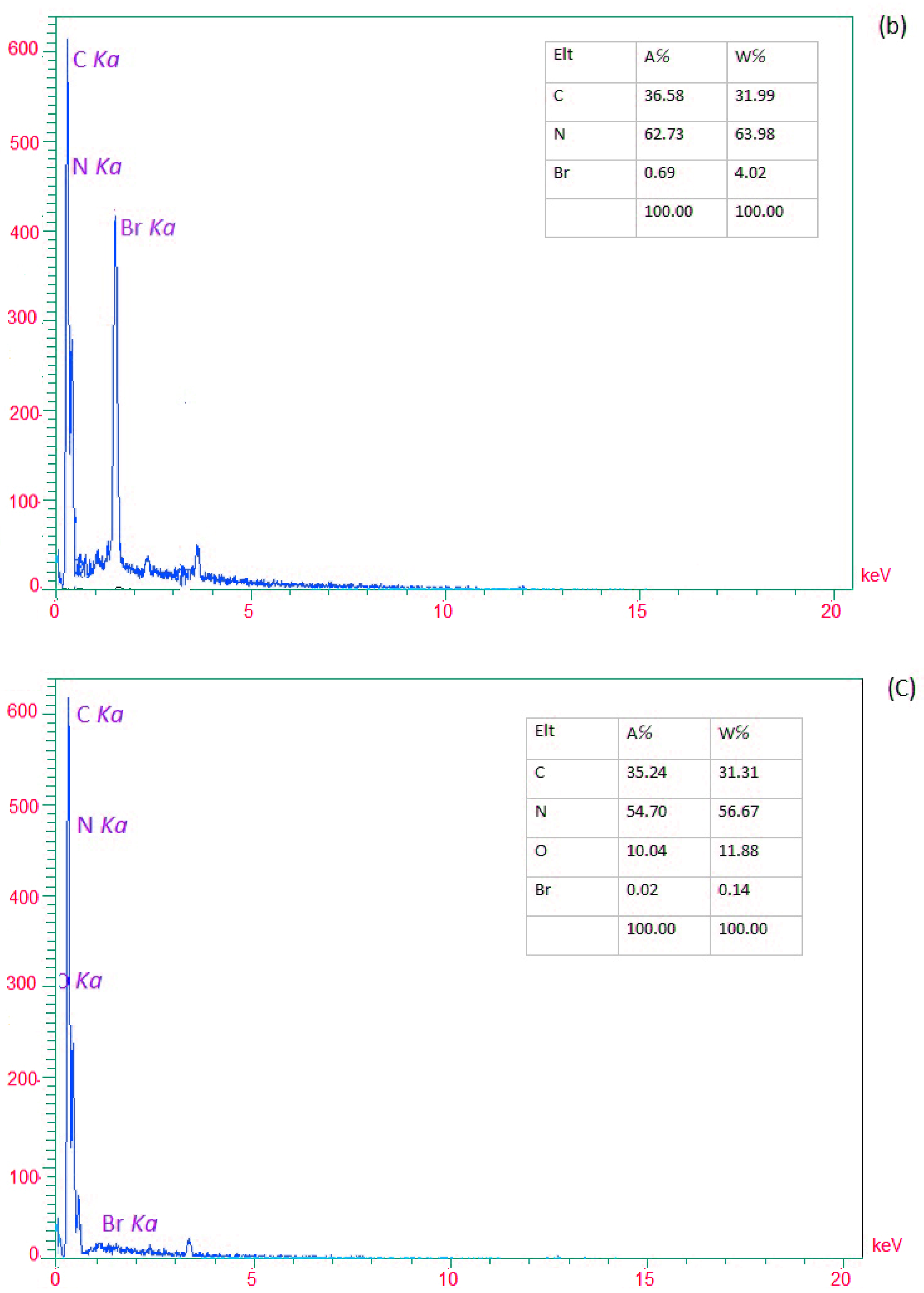
EDX analysis determined the presence of elements in (a) g-C3N4 nanosheets, (b) modified g-C3N4, and (c) g-C3N4@L-arginine. Nitrogen and Carbon elements in nanosheet g-C3N4 are visible in Figure 2a. In Figure 2b, the existence of the Br element would confirm the modification of g-C3N4 nanosheets. Moreover, Figure 2c revealed the presence of carbon, nitrogen, and oxygen, which confirm the synthesizing of g-C3N4@L-arginine.
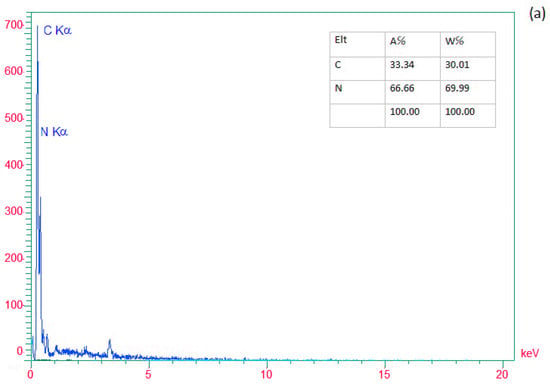
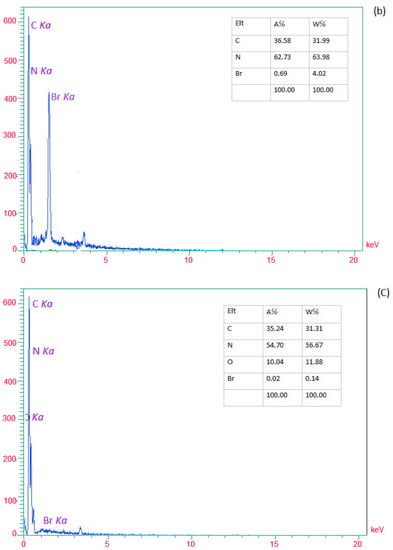
Figure 2.
EDX spectra of (a) g-C3N4 nanosheets, (b) modified g-C3N4, and (c) g-C3N4@L-arginine.
The morphology of g-C3N4@L-arginine nanocatalyst was studied by FE-SEM analysis in two scales (200 nm and 1 m). Graphitic and nanosheet properties of C3N4 are apparent based on Figure 3. It can be concluded that the g-C3N4@L-arginine nanocatalyst synthesizing has been successfully performed by observing the g-C3N4 surface roughness.

Figure 3.
FE-SEM images of g-C3N4@L-arginine.
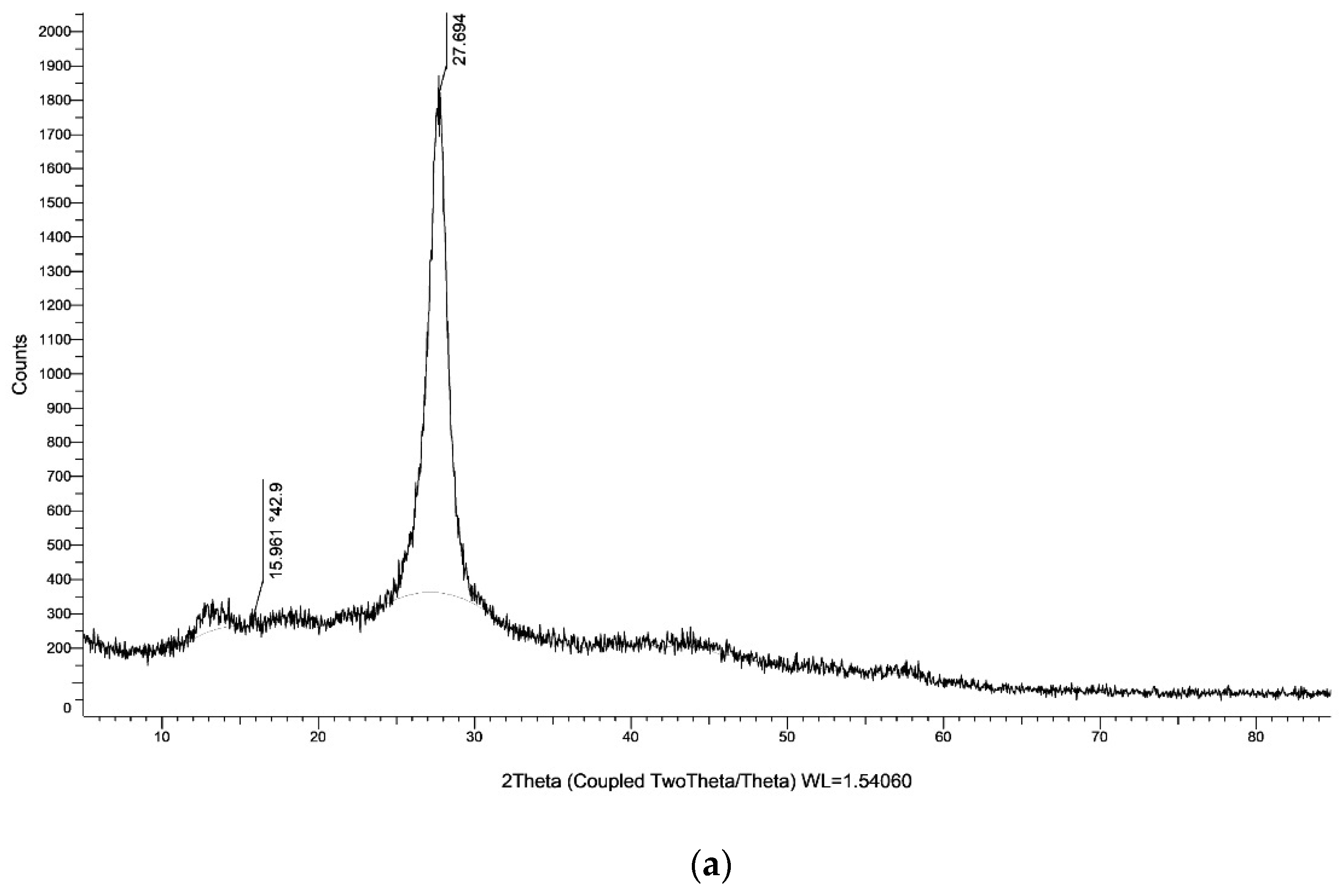
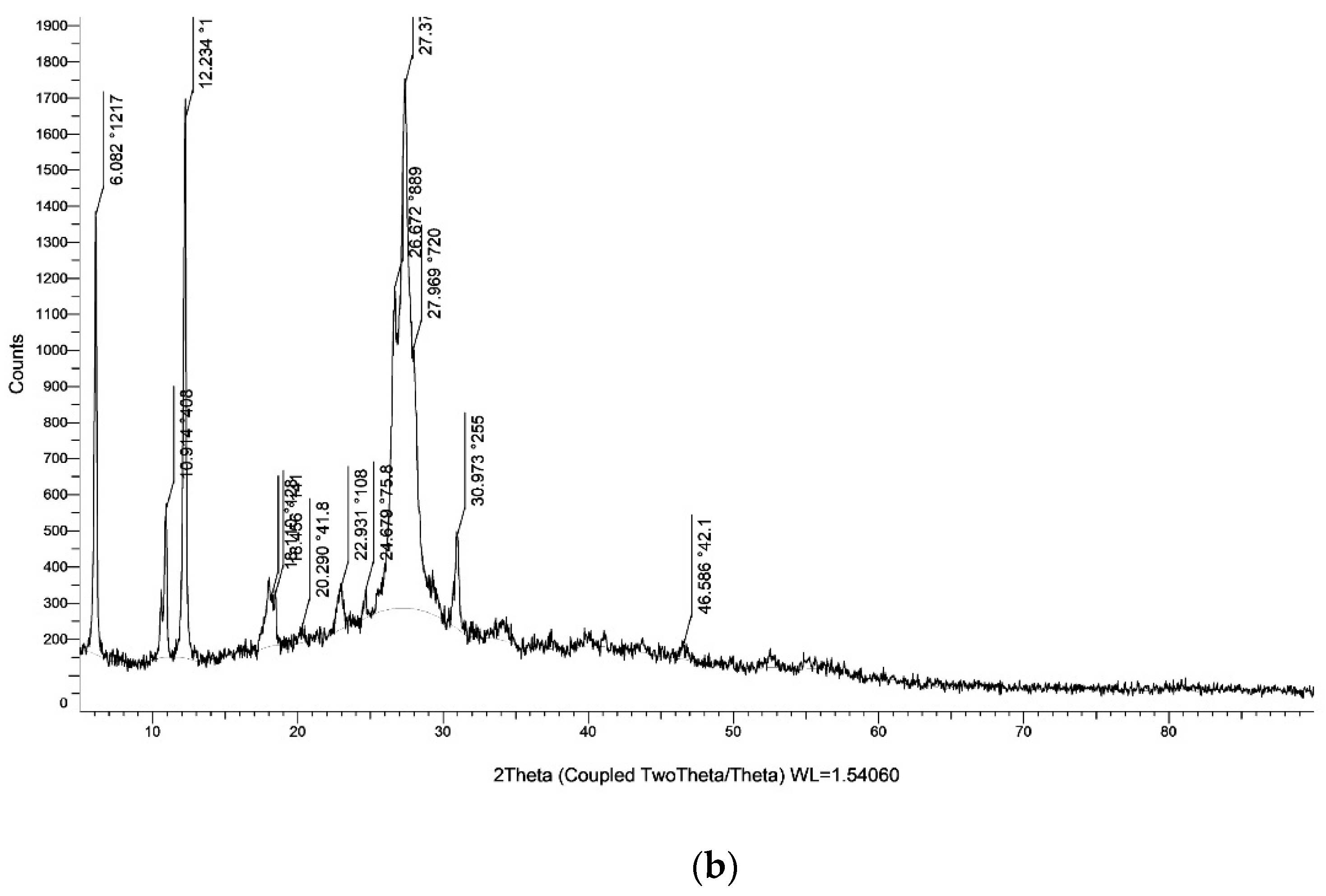
The XRD of g-C3N4 nanosheets and g-C3N4@L-arginine have been shown in Figure 4a,b. XRD pattern of nanosheet g-C3N4 in part (a) indicates the diffraction angles of 2θ = 15.96° and 2θ = 27.69°, which approve the synthesizing of g-C3N4 [34]. Diffraction angles of 2θ = 30.97°, 23.60°, 12.21°, 10.85°, 6.07° in XRD pasttern part (b) indicate the L-arginine on the surface of g-C3N4@L-arginine (JCPDS card no. 00–004-0180).
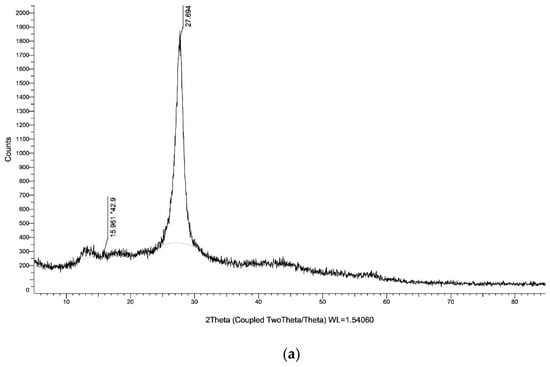
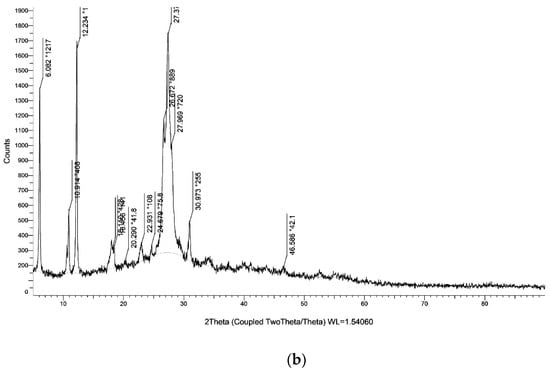
Figure 4.
XRD spectra of (a) g-C3N4 nanosheets and (b) g-C3N4@L-arginine.
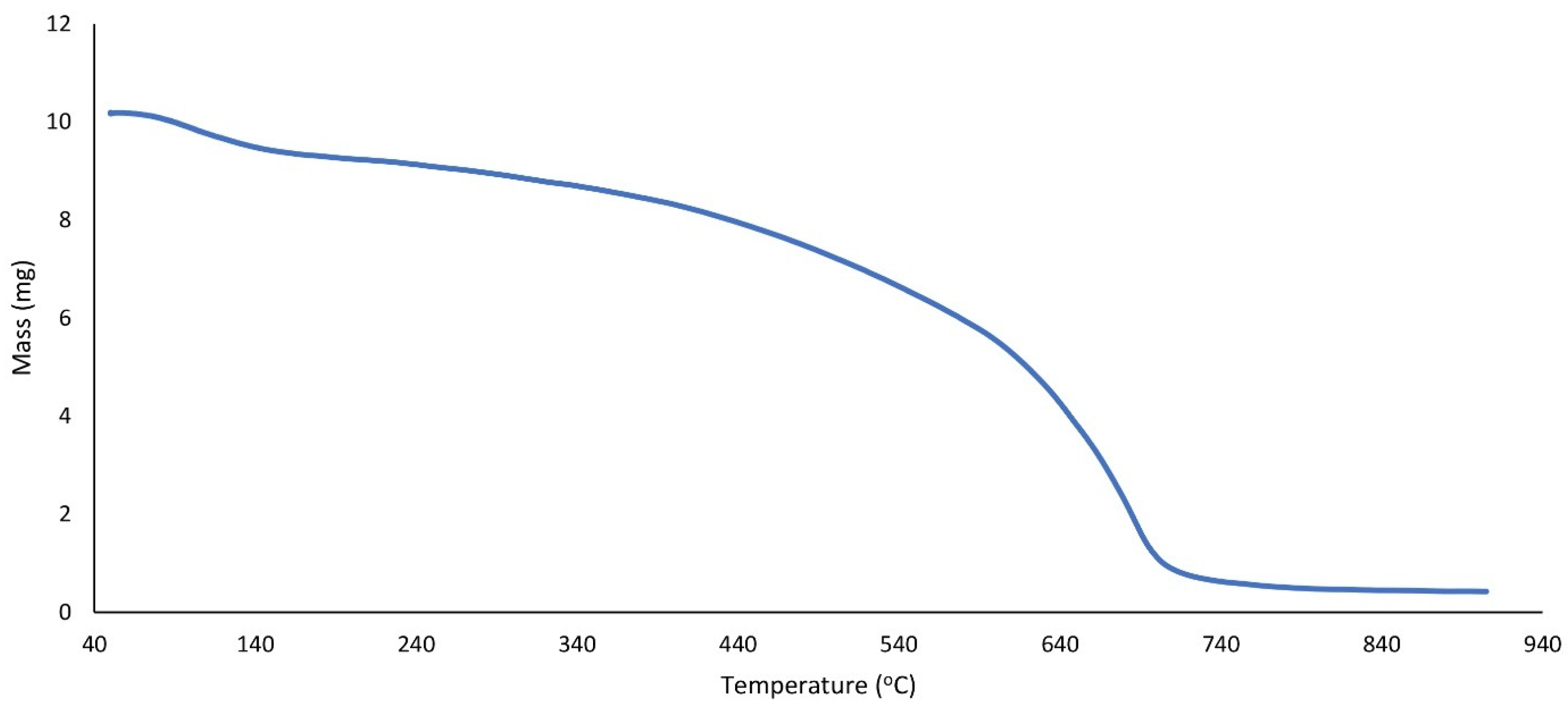
In Figure 5, g-C3N4@L-arginine thermal stability was shown at the range from 50 to 800 °C. The weight ratio has decreased gradually from 100 to 200 °C because of the removal of absorbed water from g-C3N4@L-arginine. L-arginine’s separation was observed from 200 to 400 °C. There is a dramatic decrease from 400 to 700 °C which is related to g-C3N4 nanosheet decomposition.
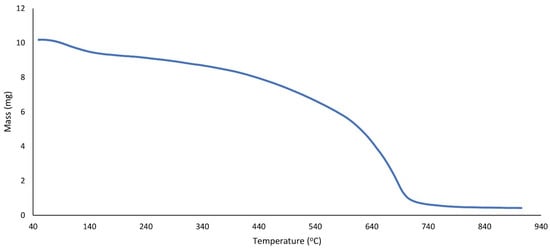
Figure 5.
TGA spectrum of g-C3N4@L-arginine.
3.1. Application
The catalytic activity of produced heterogeneous nanocatalyst g-C3N4@L-arginine was studied for multi-component reactions. The optimum reaction conditions for synthesizing acridinedione and pyranopyrazole derivatives were evaluated. The synthesis of acridinedione derivatives was performed by using dimedone (2 mmol), 4-chloro benzaldehyde (1 mmol), ammonium acetate (1 mmol), ethanol (5 mL), and catalyst (0.18 mol %) (model reaction 1). In addition, pyranopyrazole derivatives were produced by malononitrile (1.0 mmol), 4-chloro benzaldehyde (1.0 mmol), hydrazine hydrate (1.0 mmol), ethyl acetoacetate (1.0 mmol), ethanol (2.0 mL), and catalyst (0.18 mol %) (model reaction 2). The possibility of aldol reaction in aliphatic aldehydes would be the significant reason for using aromatic aldehydes compared to aliphatic aldehydes. Moreover, the reaction has been monitored by thin-layer chromatography (TLC). The model reactions have been investigated under different and convertible conditions. Initially, the reaction was performed with no catalyst at two different temperatures and the same reaction time (20 min). There was no acceptable efficiency as expected for both reactions (Table 1, entries 1–2). After using the catalyst (Table 1, entries 3–4), the desired products were produced in very small quantities at two different temperatures with the same environmental solvent. By using the catalyst at 80 °C for 20 min, there was a significant yield and efficiency of up to 92% for the first reaction and 91% for the second one (Table 1, entry 5). Moreover, despite increasing the reaction time up to 30 min, no noteworthy changes in the efficiency were observed (Table 1, entry 6).

Table 1.
Optimization of g-C3N4@L-arginine for reaction 1 and 2.
In addition, changing the used solvent to water with the same condition as Table 1- entry 5 can decrease the efficiency of reactions 1 and 2 to 65% and 68%, respectively (Table 1, entry 7). If the solvent of the reactions changed to methanol and acetonitrile (Table 1, entries 8 and 9), the reaction yield, in comparison with entry 5, will be increased and decreased, respectively. Likewise, the model reactions were performed by g-C3N4 (0.18 mol %) and L-arginine (0.18 mol %) with the same conditions, while the yield of the final products was decreased.
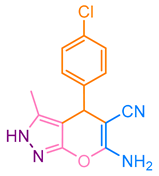
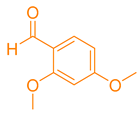
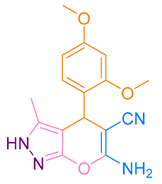
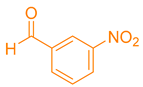
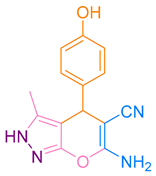
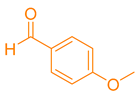
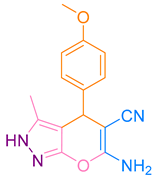
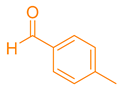
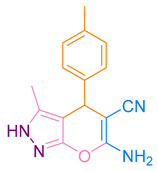
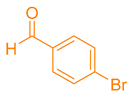
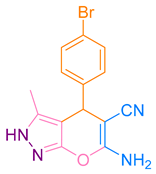
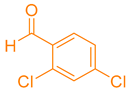
After optimization, different aromatic aldehydes were used to show the merits of g-C3N4@L-arginine catalytic activity and different pyranopyrazole and acridinedione derivatives were synthesized (Table 2 and Table 3).

Table 2.
Synthesis acridinedione derivatives by g-C3N4@L-arginine (a,b).

Table 3.
Synthesis pyranopyrazole derivatives by g-C3N4@L-arginine (a,b).
3.2. Mechanism of Using Nanocatalyst for Synthesizing Pyranopyrazole and Acridinedione Derivatives
3.2.1. Pyranopyrazoles
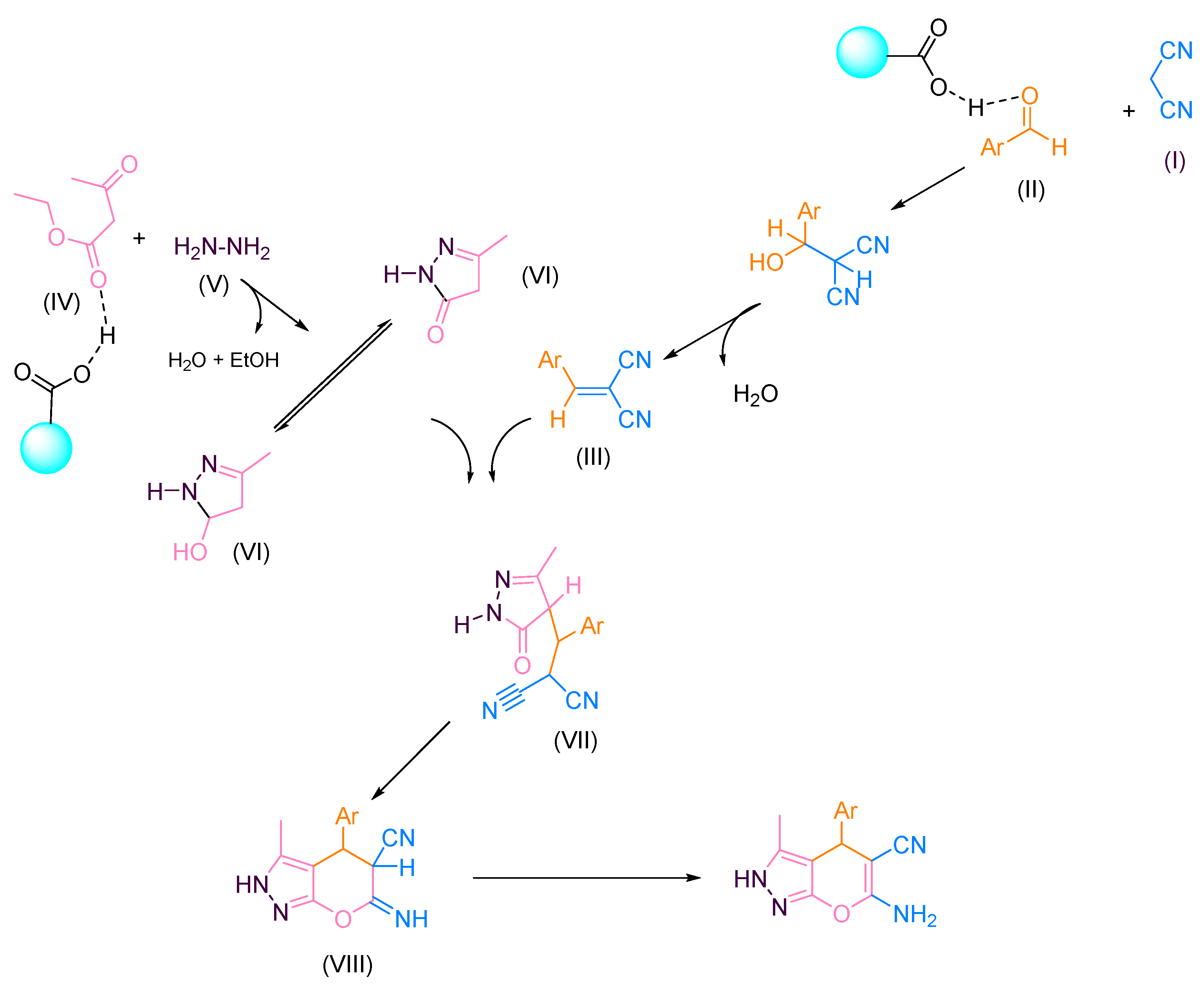
The study of the mechanism for pyranopyrazole derivatives and the proposed mechanism is shown in Scheme 2. In addition, g-C3N4@L-arginine is needed for activating different intermediates and reactants. Malononitrile (I) and aromatic aldehyde (II) would react with each other by the carbon as a nucleophile. Then, it would reacts with the carbonyl group by releasing water and produce intermediate (III). Simultaneously, ethyl acetoacetate (IV) and hydrazine hydrate (V) react with each other and form the intermediate (VI). Afterward, the amine group’s non-bonding electron pair reacts with the ethyl acetoacetate’s carbonyl group. In the following step, the 5-member ring was closed by removing the water molecule. In the last step, two produced intermediates, ((III) and (VI)), would react with each other, and the pryranopyrazole derivative was synthesized.
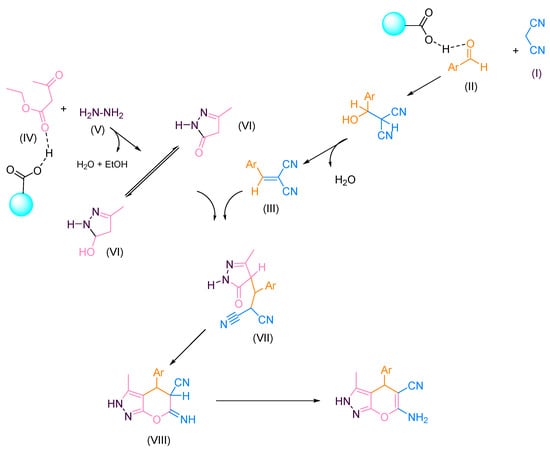
Scheme 2.
Proposed mechanism for synthesizing pyranopyrazole derivatives.
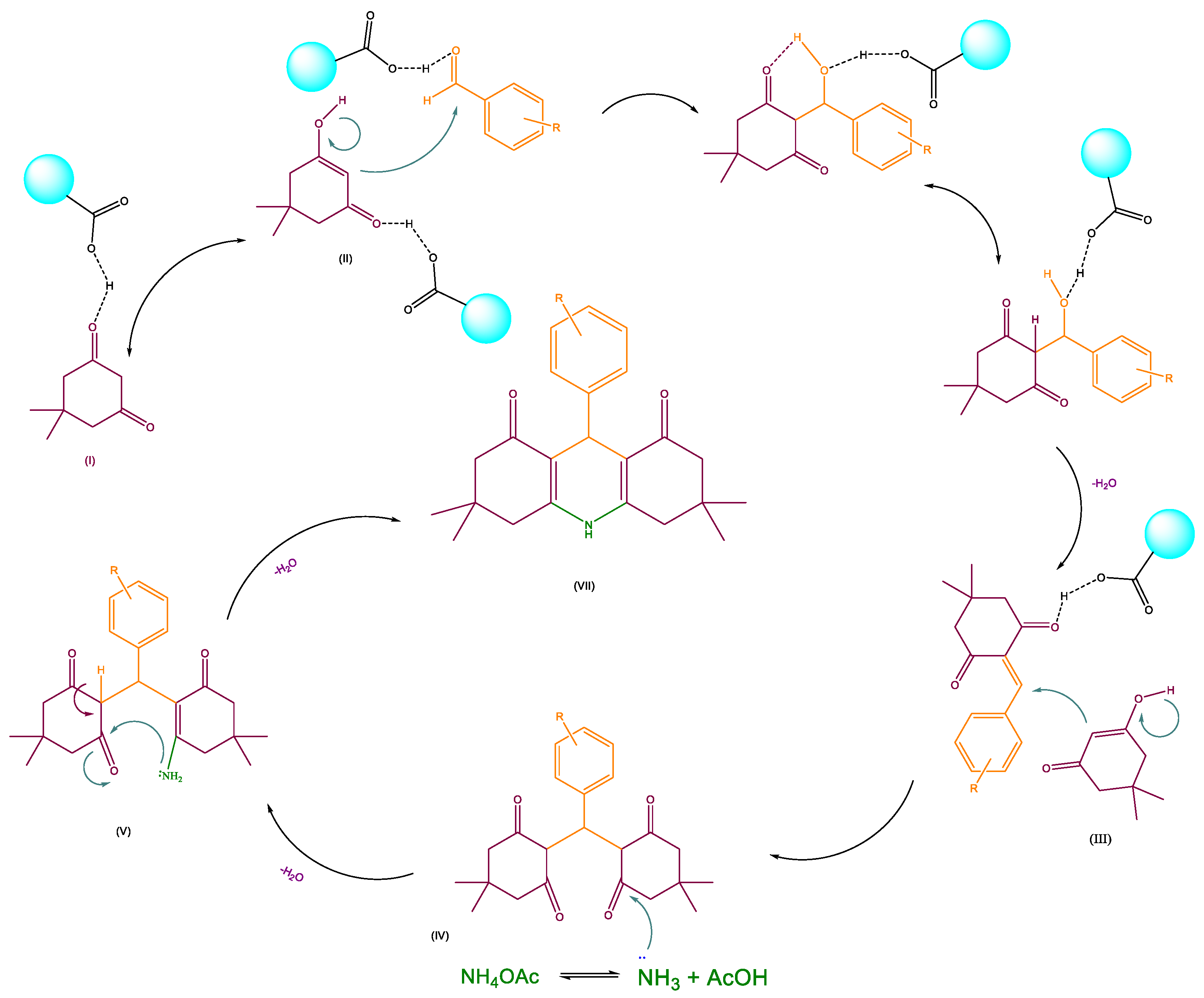
3.2.2. Acridinediones
The study of the mechanism for acridinedione derivative synthesis and the proposed mechanism is exhibited in Scheme 3. For activating the carbonyl group of aldehydes, the presence of g-C3N4 @L-arginine is essential. After activating the carbonyl group with nanocatalyst (I) and producing the hydroxyl group on dimedone (II), the carbon nucleophile would react with activated aromatic aldehyde. Then, the other dimedone reacts with the double bond for donating electrons (III), and after a water molecule removal, the ring is closed by an intramolecular reaction (IV, V and VI). Eventually, the intended product is obtained (VII).
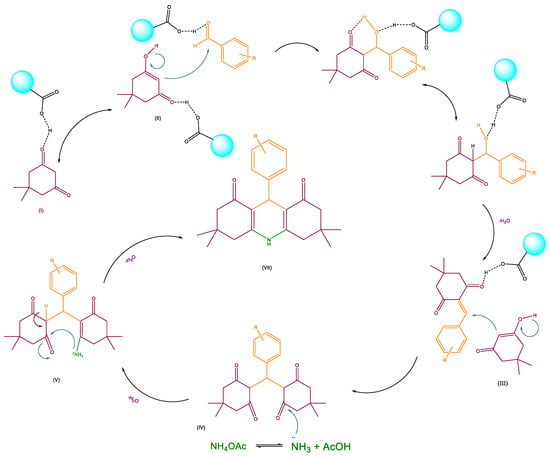
Scheme 3.
Proposed mechanism for synthesizing acridinedione derivatives.
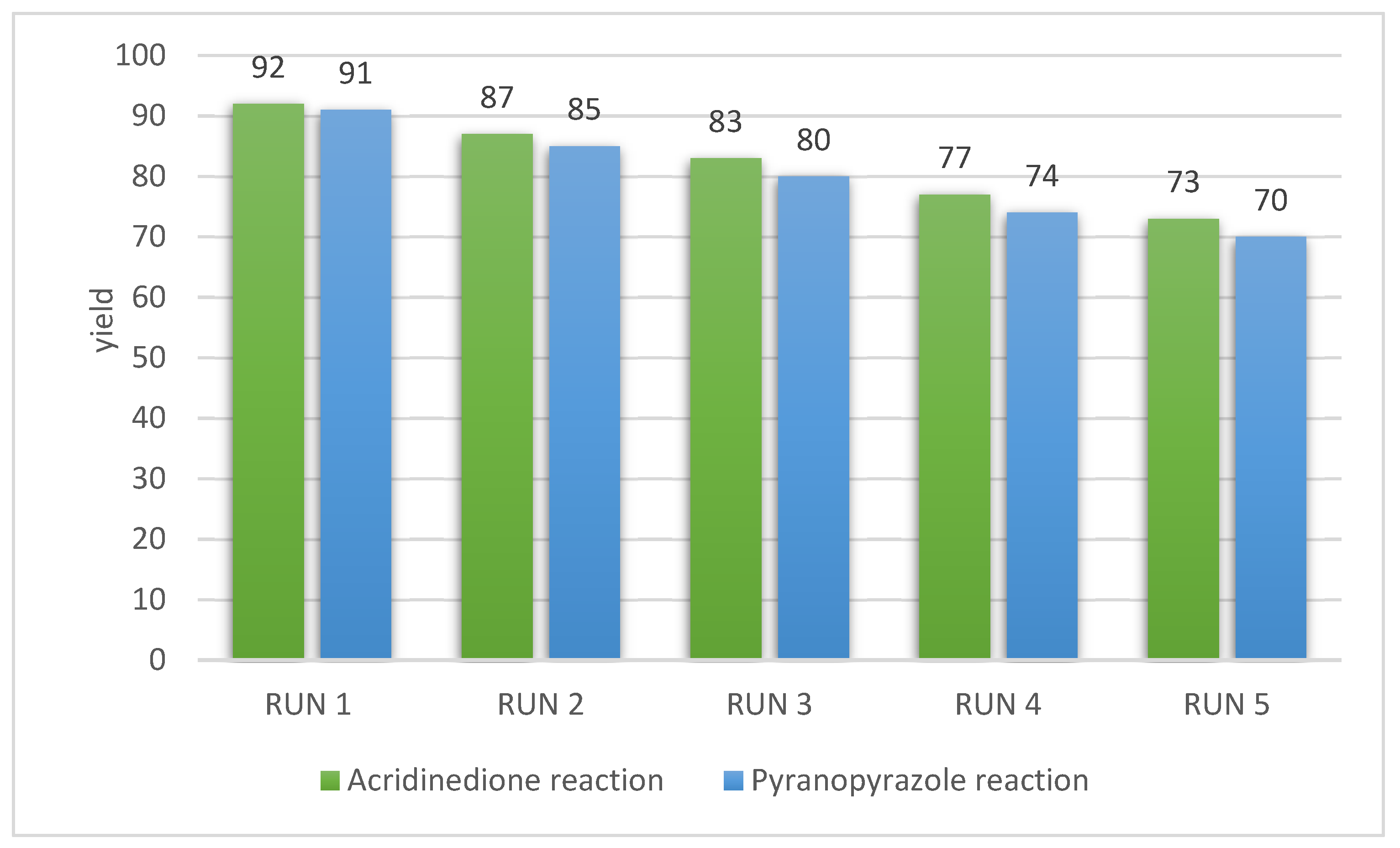
3.3. Reusability
The recovery and recyclability of the catalyst are the essential principles of green chemistry. Therefore, the reusability of g-C3N4@L-arginine was studied for synthesizing pyranopyrazole and acridinedione derivatives. G-C3N4@L-arginine was extracted from the reaction, washed with water and ethanol, then dried at 70 °C. It was repeated 5 times in the same conditions. After each reaction, the yield decreased gradually, but it was acceptable (Figure 6).
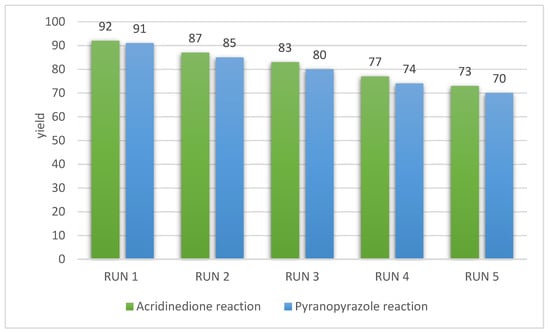
Figure 6.
Reusability of g-C3N4@L-arginine in acridinedione and pyranopyrazole derivatives.
4. Conclusions
In conclusion, in this project, we utilized an easy and convenient method for preparing g-C3N4@L-arginine nanocatalyst and applied it for producing pyranopyrazole and acridinedione derivatives. G-C3N4@L-arginine nanocatalyst has remarkable advantages such as reusability, easy separation, high efficiency, and short reaction time. According to the results, produced nanocatalyst is the superior compared to other reported catalysts.
Author Contributions
Conceptualization, supervision, validation, H.G.; investigation, Z.T.; methodology, P.H., M.T. and Z.T.; writing—review and editing, writing—original draft preparation, H.G., P.H. and F.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Swain, S.; Altaee, A.; Saxena, M.; Samal, A.K. A comprehensive study on heterogeneous single atom catalysis: Current progress, and challenges. Coord. Chem. Rev. 2022, 470, 214710. [Google Scholar] [CrossRef]
- Zhi, Y.; Wang, Z.; Zhang, H.; Zhang, Q. Recent progress in metal-free covalent organic frameworks as heterogeneous catalysts. Small 2020, 16, 2001070. [Google Scholar] [CrossRef] [PubMed]
- Ghafuri, H.; Tajik, Z.; Ghanbari, N.; Hanifehnejad, P. Preparation and characterization of graphitic carbon nitride-supported l-arginine as a highly efficient and recyclable catalyst for the one-pot synthesis of condensation reactions. Sci. Rep. 2021, 11, 19792. [Google Scholar] [CrossRef] [PubMed]
- Ghafuri, H.; Rashidizadeh, A. Facile preparation of CuS-g-C3N4/Ag nanocomposite with improved photocatalytic activity for the degradation of rhodamine B. Polyhedron 2020, 179, 114368. [Google Scholar] [CrossRef]
- Akhtar, B.; Ghafuri, H.; Rashidizadeh, A. Synergistic effect of iodine doped TiO2 nanoparticle/g-C3N4 nanosheets with upgraded visible-light-sensitive performance toward highly efficient and selective photocatalytic oxidation of aromatic alcohols under blue LED irradiation. Mol. Catal. 2021, 506, 111527. [Google Scholar] [CrossRef]
- Rahmati, M.; Ghafuri, H. Catalytic Strecker reaction: G-C3N4-anchored sulfonic acid organocatalyst for the synthesis of α-aminonitriles. Res. Chem. Intermed. 2021, 47, 1489–1502. [Google Scholar] [CrossRef]
- Rashidizadeh, A.; Ghafuri, H.; Rezazadeh, Z. Improved visible-light photocatalytic activity of g-C3N4/CuWO4 nanocomposite for degradation of methylene blue. Multidiscip. Digit. Publ. Inst. Proc. 2020, 41, 43. [Google Scholar]
- Rashidizadeh, A.; Ghafuri, H. g-C3N4/Ni nanocomposite: An efficient and eco-friendly recyclable catalyst for the synthesis of quinoxalines. Multidiscip. Digit. Publ. Inst. Proc. 2019, 9, 49. [Google Scholar]
- Singh, L.; Shrivastav, A.; Verma, N. Effect of L-arginine amino acid on liver regeneration after hepatocyte damage in rats: An experimental study. J. Drug Deliv. Ther. 2019, 9, 470–476. [Google Scholar]
- Hamzavi, S.F.F.; Jamili, S.; Yousefzadi, M.; Moradi, A.M.; Biuki, N.A. Silver nanoparticles supported on chitosan as a green and robust heterogeneous catalyst for direct synthesis of nitrogen heterocyclic compounds under green conditions. Bull. Chem. React. Eng. Catal. 2019, 14, 51–59. [Google Scholar] [CrossRef]
- Kamalzare, M.; Ahghari, M.R.; Bayat, M.; Maleki, A. Fe3O4@chitosan-tannic acid bionanocomposite as a novel nanocatalyst for the synthesis of pyranopyrazoles. Sci. Rep. 2021, 11, 20021. [Google Scholar] [CrossRef]
- Lin, Y.Y.; Hung, K.Y.; Liu, F.Y.; Dai, Y.M.; Lin, J.H.; Chen, C.C. Photocatalysts of quaternary composite, bismuth oxyfluoride/bismuth oxyiodide/graphitic carbon nitride: Synthesis, characterization, and photocatalytic activity. Mol. Catal. 2022, 528, 112463. [Google Scholar] [CrossRef]
- Edrisi, M.; Azizi, N. Sulfonic acid-functionalized graphitic carbon nitride composite: A novel and reusable catalyst for the one-pot synthesis of polysubstituted pyridine in water under sonication. J. Iran. Chem. Soc. 2020, 17, 901–910. [Google Scholar] [CrossRef]
- Nasiriani, T.; Javanbakht, S.; Nazeri, M.T.; Farhid, H.; Khodkari, V.; Shaabani, A. Isocyanide-Based Multicomponent Reactions in Water: Advanced Green Tools for the Synthesis of Heterocyclic Compounds. Top. Curr. Chem. 2022, 380, 50. [Google Scholar] [CrossRef]
- Nandi, S.; Jamatia, R.; Sarkar, R.; Sarkar, F.K.; Alam, S.; Pal, A.K. One-Pot Multicomponent Reaction: A Highly Versatile Strategy for the Construction of Valuable Nitrogen-Containing Heterocycles. ChemistrySelect 2022, 7, e202201901. [Google Scholar] [CrossRef]
- Farhid, H.; Khodkari, V.; Nazeri, M.T.; Javanbakht, S.; Shaabani, A. Multicomponent reactions as a potent tool for the synthesis of benzodiazepines. Org. Biomol. Chem. 2021, 19, 3318–3358. [Google Scholar] [CrossRef]
- Goddard, J.-P.; Malacria, M.; Ollivier, C. Multi-component Reactions in Molecular Diversity; John Wiley & Sons: Hoboken, NJ, USA, 2020. [Google Scholar]
- Becerra, D.; Abonia, R.; Castillo, J.-C. Recent Applications of the Multicomponent Synthesis for Bioactive Pyrazole Derivatives. Molecules 2022, 27, 4723. [Google Scholar] [CrossRef]
- Wu, M.; Feng, Q.; Wan, D.; Ma, J. CTACl as catalyst for four-component, one-pot synthesis of pyranopyrazole derivatives in aqueous medium. Synth. Commun. 2013, 43, 1721–1726. [Google Scholar] [CrossRef]
- Reddy, G.; Raul, J. Garcia Synthesis of Pyranopyrazoles under Eco-friendly Approach by Using Acid Catalysis. J. Heterocycl. Chem. 2017, 54, 89–94. [Google Scholar] [CrossRef]
- Patil, U.; Patil, R.; Patil, S. An Eco-friendly Catalytic System for One-pot Multicomponent Synthesis of Diverse and Densely Functionalized Pyranopyrazole and Benzochromene Derivatives. J. Heterocycl. Chem. 2019, 56, 1898–1913. [Google Scholar] [CrossRef]
- Nikoorazm, M.; Tahmasbi, B.; Gholami, S.; Moradi, P. Copper and nickel immobilized on cytosine@ MCM-41: As highly efficient, reusable and organic–inorganic hybrid nanocatalysts for the homoselective synthesis of tetrazoles and pyranopyrazoles. Appl. Organomet. Chem. 2020, 34, e5919. [Google Scholar] [CrossRef]
- Mukherjee, P.; Das, A. Spirocyclopropanes from Intramolecular Cyclopropanation of Pyranopyrazoles and Pyranopyrimidine-diones and Lewis Acid Mediated (3 + 2) Cycloadditions of Spirocyclopropylpyrazolones. J. Org. Chem. 2017, 82, 2794–2802. [Google Scholar] [CrossRef] [PubMed]
- Khandare, P.M.; Ingale, R.D.; Taware, A.S.; Shisodia, S.U.; Pawar, S.S.; Kotai, L.; Pawar, R.P. One pot synthesis and biological evaluation of pyranopyrazoles in aqueous medium. Eur. Chem. Bull. 2017, 6, 410–414. [Google Scholar] [CrossRef]
- Madar, J.M.; Samundeeswari, S.; Holiyachi, M.; Naik, N.S.; Pawar, V.; Gudimani, P.; Shastri, L.A.; Kumbar, V.M.; Sunagar, V.A. Solvent-Free Synthesis, Characterization, and In Vitro Biological Activity Study of Xanthenediones and Acridinediones. Russ. J. Bioorg. Chem. 2021, 47, 535–542. [Google Scholar] [CrossRef]
- Jamalian, A.; Miri, R.; Firuzi, O.; Amini, M.; Moosavi-Movahedi, A.A.; Shafieea, A. Synthesis, cytotoxicity and calcium antagonist activity of novel imidazolyl derivatives of 1,8-acridinediones. J. Iran. Chem. Soc. 2011, 8, 983–991. [Google Scholar] [CrossRef]
- Alvala, M.; Bhatnagar, S.; Ravi, A.; Jeankumar, V.U.; Manjashetty, T.H.; Yogeeswari, P.; Sriram, D. Novel acridinedione derivatives: Design, synthesis, SIRT1 enzyme and tumor cell growth inhibition studies. Bioorg. Med. Chem. Lett. 2012, 22, 3256–3260. [Google Scholar] [CrossRef]
- Behbahani, F.S.; Tabeshpour, J.; Mirzaei, S.; Golmakaniyoon, S.; Tayarani-Najaran, Z.; Ghasemi, A.; Ghodsi, R. Synthesis and biological evaluation of novel benzo[c]acridine-diones as potential anticancer agents and tubulin polymerization inhibitors. Arch. der Pharm. 2019, 352, 1800307. [Google Scholar] [CrossRef]
- Aday, B.; Yıldız, Y.; Ulus, R.; Eris, S.; Sen, F.; Kaya, M. One-pot, efficient and green synthesis of acridinedione derivatives using highly monodisperse platinum nanoparticles supported with reduced graphene oxide. New J. Chem. 2016, 40, 748–754. [Google Scholar] [CrossRef]
- Ulus, R.; Yıldız, Y.; ERiŞ, S.; Aday, B.; Sen, F.; Kaya, M. Functionalized multi-walled carbon nanotubes (f-MWCNT) as highly efficient and reusable heterogeneous catalysts for the synthesis of acridinedione derivatives. ChemistrySelect 2016, 1, 3861–3865. [Google Scholar] [CrossRef]
- Xia, J.-J.; Zhang, K.-H. Synthesis of N-substituted acridinediones and polyhydroquinoline derivatives in refluxing water. Molecules 2012, 17, 5339–5345. [Google Scholar] [CrossRef]
- Zhu, A.; Liu, R.; Du, C.; Li, L. Betainium-based ionic liquids catalyzed multicomponent Hantzsch reactions for the efficient synthesis of acridinediones. RSC Adv. 2017, 7, 6679–6684. [Google Scholar] [CrossRef]
- Mansoor, S.S.; Aswin, K.; Logaiya, K.; Sudhan, S. Aqua-mediated synthesis of acridinediones with reusable silica-supported sulfuric acid as an efficient catalyst. J. Taibah Univ. Sci. 2014, 8, 265–275. [Google Scholar] [CrossRef]
- Qiu, P.; Chen, H.; Xu, C.; Zhou, N.; Jiang, F.; Wang, X.; Fu, Y. Fabrication of an exfoliated graphitic carbon nitride as a highly active visible light photocatalyst. J. Mater. Chem. A 2015, 3, 24237–24244. [Google Scholar] [CrossRef]
- Taheri-Ledari, R.; Esmaeili, M.S.; Varzi, Z.; Eivazzadeh-Keihan, R.; Maleki, A.; Shalan, A.E. Facile route to synthesize Fe3O4@ acacia–SO3 H nanocomposite as a heterogeneous magnetic system for catalytic applications. RSC Adv. 2020, 10, 40055–40067. [Google Scholar] [CrossRef]
- Zolfigol, M.A.; Karimi, F.; Yarie, M.; Torabi, M. Catalytic application of sulfonic acid-functionalized titana-coated magnetic nanoparticles for the preparation of 1,8-dioxodecahydroacridines and 2,4,6-triarylpyridines via anomeric-based oxidation. Appl. Organomet. Chem. 2018, 32, e4063. [Google Scholar] [CrossRef]
- Mahesh, P.; Guruswamy, K.; Diwakar, B.S.; Devi, B.R.; Murthy, Y.L.N.; Kollu, P.; Pammi, S.V.N. Magnetically separable recyclable nano-ferrite catalyst for the synthesis of acridinediones and their derivatives under solvent-free conditions. Chem. Lett. 2015, 44, 1386–1388. [Google Scholar] [CrossRef]
- Kiani, M.; Mohammadipour, M. Fe3O4@SiO2–MoO3H nanoparticles: A magnetically recyclable nanocatalyst system for the synthesis of 1,8-dioxo-decahydroacridine derivatives. RSC Adv. 2017, 7, 997–1007. [Google Scholar] [CrossRef]
- Aher, D.; Khillare, K.; Shankarwar, S. Incorporation of Keggin-based H3PW7Mo5O40 into bentonite: Synthesis, characterization and catalytic applications. RSC Adv. 2021, 11, 11244–11254. [Google Scholar] [CrossRef]
- Bazdid-Vahdaty, N.; Mamaghani, M.; Khalili, B.; Tavakoli, F. Ag/CuO/MCM-48 AS A potential CATALYST for the synthesis of symmetrical and unsymmetrical polyhydroquinolines. J. Chil. Chem. Soc. 2021, 66, 5136–5141. [Google Scholar] [CrossRef]
- Alponti, L.H.; Picinini, M.; Urquieta-Gonzalez, E.A.; Corrêa, A.G. USY-zeolite catalyzed synthesis of 1,4-dihydropyridines under microwave irradiation: Structure and recycling of the catalyst. J. Mol. Struct. 2021, 1227, 129430. [Google Scholar] [CrossRef]
- Hasannezhad, N.; Shadjou, N. KCC-1-nPr-NH-Arg as an efficient organo-nanocatalyst for the green synthesis of 1,8-dioxo decahydroacridine derivatives. J. Mol. Recognit. 2022, 35, e2956. [Google Scholar] [CrossRef] [PubMed]
- Khojastehnezhad, A.; Rahimizadeh, M.; Eshghi, H.; Moeinpour, F.; Bakavoli, M. Ferric hydrogen sulfate supported on silica-coated nickel ferrite nanoparticles as new and green magnetically separable catalyst for 1,8 dioxodecahydroacridine synthesis. Chin. J. Catal. 2014, 35, 376–382. [Google Scholar] [CrossRef]
- Hojati, S.F.; Amiri, A.; MoeiniEghbali, N.; Mohamadi, S. Polypyrrole/Fe3O4/CNT as a recyclable and highly efficient catalyst for one-pot three-component synthesis of pyran derivatives. Appl. Organomet. Chem. 2018, 32, e4235. [Google Scholar] [CrossRef]
- Heravi, M.M.; Malakooti, R.; Kafshdarzadeh, K.; Amiri, Z.; Zadsirjan, V.; Atashin, H. Supported palladium oxide nanoparticles in Al-SBA-15 as an efficient and reusable catalyst for the synthesis of pyranopyrazole and benzylpyrazolyl coumarin derivatives via multicomponent reactions. Res. Chem. Intermed. 2022, 48, 203–234. [Google Scholar] [CrossRef]
- Maleki, A.; Eskandarpour, V. Design and development of a new functionalized cellulose-based magnetic nanocomposite: Preparation, characterization, and catalytic application in the synthesis of diverse pyrano [2,3-c] pyrazole derivatives. J. Iran. Chem. Soc. 2019, 16, 1459–1472. [Google Scholar] [CrossRef]
- Abdolahi, S.; Hajjami, M.; Gholamian, F. An approach to the synthesis and characterization of HMS/Pr-Rh-Zr as efficient catalyst for synthesis of tetrahydrobenzo[b]pyran and 1,4-dihydropyrano[2,3-c]pyrazole derivatives. Res. Chem. Intermed. 2021, 47, 1883–1904. [Google Scholar] [CrossRef]
- Kumari, R.; Varghese, A.; George, L.; Akshaya, K.B. Photophysical study of 6-amino-3-methyl-4-(4-nitrophenyl)-1,4-dihydropyrano[2,3-c]pyrazole-5-carbonitrile and estimation of ground-state and singlet excited-state dipole moments by solvatochromic approaches. J. Mol. Liq. 2016, 222, 828–835. [Google Scholar] [CrossRef]
- Shaterian, H.; Azizi, K. Mild, four-component synthesis of 6-amino-4-aryl-3-methyl-1,4-dihydropyrano[2, 3-c]pyrazole-5-carbonitriles catalyzed by titanium dioxide nano-sized particles. Res. Chem. Intermed. 2014, 40, 661–667. [Google Scholar] [CrossRef]
- Hassanzadeh-Afruzi, F.; Dogari, H.; Esmailzadeh, F.; Maleki, A. Magnetized melamine-modified polyacrylonitrile (PAN@ melamine/Fe3O4) organometallic nanomaterial: Preparation, characterization, and application as a multifunctional catalyst in the synthesis of bioactive dihydropyrano[2,3-c]pyrazole and 2-amino-3-cyano 4H-pyran derivatives. Appl. Organomet. Chem. 2021, 35, e6363. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).