Abstract
The accurate assessment of vitamin A in animal feed and tissues is vital for safeguarding animal well-being and ensuring high-quality nutritional feed. However, challenges in achieving precise results persist, necessitating a comprehensive understanding of the influencing factors. This review delves into the historical progression of analytical techniques, from colorimetric assays and spectrophotometry to advanced chromatographic methods and non-destructive spectroscopic approaches. Factors influencing analytical precision are scrutinized, encompassing sample preparation, storage conditions, interfering substances, and human errors. The crucial role of quality control and standardized protocols in ensuring the reproducibility and reliability of results is emphasized. Moreover, this review highlights the need for tailored analytical approaches to account for the complexities of sample matrices and the significance of cutting-edge technologies, including on-site analysis and data science integration, in enhancing analytical precision. By acknowledging the challenges and prospects in vitamin A analysis, this paper provides insights for optimizing analytical methodologies and promoting animal welfare and product safety.
1. Introduction
Ensuring that animals receive a well-balanced diet and appropriate feed formulation can significantly enhance animal productivity, improve the quality of their products, and promote better animal welfare [1]. Vitamin A, also known as retinol, is a vital micronutrient crucial for maintaining optimal health and well-being in livestock [2,3]. It plays a central role in various physiological functions, including immune response regulation, vision maintenance, and cellular differentiation [4]. Consequently, the accurate determination of vitamin A levels in animal feed and premixes is essential for ensuring animal health and production. Beyond preventing deficiencies and associated health issues, it forms the foundation for their overall growth and performance [5,6,7].
In the realm of animal nutrition, ensuring the precise measurement of vitamin A content is of utmost importance. It is not merely a matter of scientific accuracy, but a critical factor in guaranteeing the safety and quality of the end products consumed by livestock [8]. With the increasing emphasis on food safety standards, the precise determination of vitamin A underscores the commitment to providing nutritious and safe feed for animals, safeguarding their well-being, and subsequently ensuring the quality of animal-derived products [9]. Likewise, evaluating the retinol levels in animal tissues is crucial for assessing nutritional status, preventing illnesses, and improving the general health and productivity of livestock [10].
However, the pursuit of accurate results in vitamin A determination faces several challenges. Researchers and analysts encounter various obstacles that impede the attainment of reliable outcomes, presenting a significant predicament in the analysis of animal feed and other samples [9,11]. Challenges range from intricate sample preparation and extraction procedures to the complexities associated with analytical methodologies and the absence of standardized regulatory guidelines [12]. Achieving precision in vitamin A determination necessitates a comprehensive understanding of these challenges and their implications.
Therefore, this review paper aims to explore the multifaceted challenges confronted by researchers and analysts in accurately determining vitamin A in premixes, feed, and biological tissues. The focus of the synthesis will be specifically on the following key aspects:
- Historical progress in vitamin A analysis techniques.
- Quantitative vitamin A analysis: diverse analytical approaches.
- Deciphering the factors: unveiling the complexities of analytical precision.
- The crucial role of quality control: navigating the path to reliable results.
- Reflection and future prospects: charting the course for enhanced analytical precision.
Through comprehensive analysis of these five subjects, this review underscores the imperative need for standardized protocols and advanced scientific methodologies. These measures are crucial in guaranteeing the precision and dependability of vitamin A determination, thereby fostering the overall improvement of animal health and productivity.
2. Historical Progress in Vitamin A Analysis Techniques
Vitamin A has been a focal point of significant interest within the realm of analytical chemistry. From its initial detection endeavors to the current sophisticated methodologies, the progression of analytical methods for vitamin A determination has been an evolution marked by challenges, breakthroughs, and paradigm shifts [8,13,14,15]. This section offers an overview of the historical development of these techniques, from their inception to the contemporary state-of-the-art methodologies.
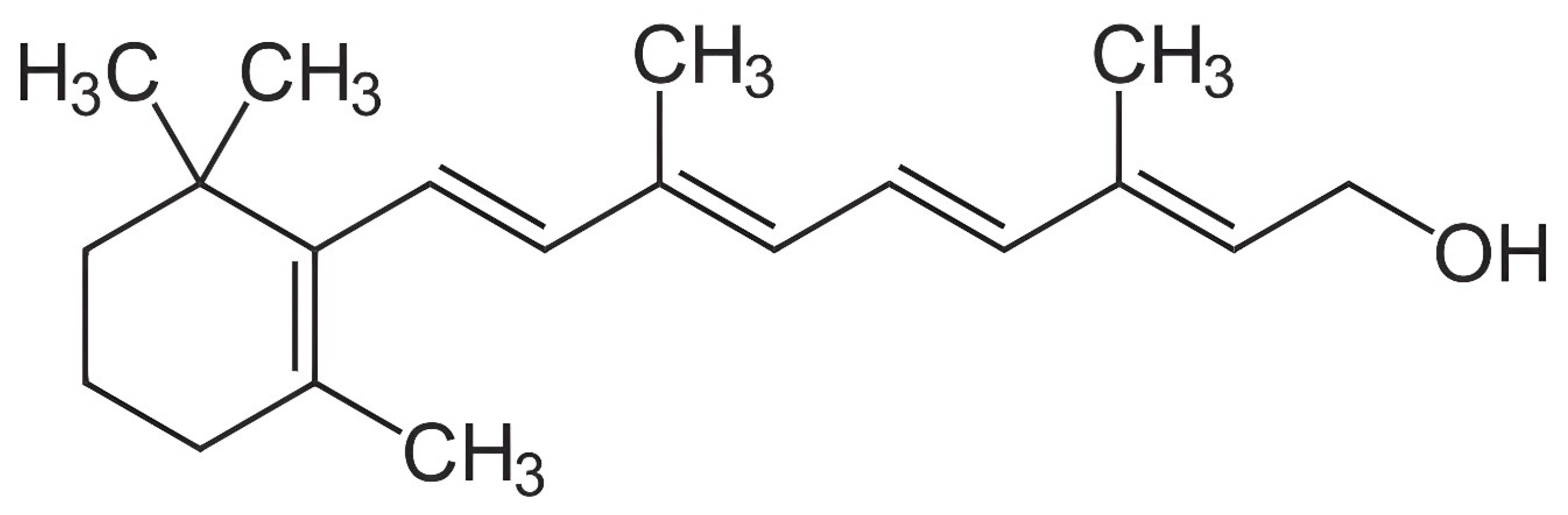
The journey commenced in the early 20th century, coinciding with the identification of vitamin A’s chemical structure by Paul Karrer in 1932 [16] (Figure 1). Pivotal studies during this period focused on the isolation and purification of vitamin A from various natural sources, laying the foundation for subsequent analytical pursuits [17,18]. Elmer McCollum and Marguerite Davis’ groundbreaking work in the early 1910s, emphasizing the vital role of vitamin A in human health, catalyzed the interest in analytical methodologies [16].
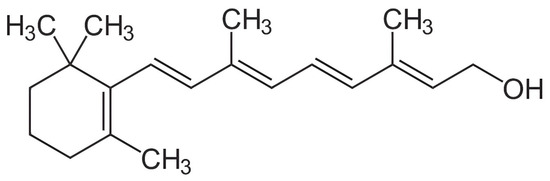
Figure 1.
Structural formula for all-trans-retinol.
The development of analytical methods faced challenges, primarily due to the intricate nature of vitamin A’s chemical structure and the existence of multiple isomers [19,20,21]. These isomers encompass various forms, such as all-trans-, 13-cis-, 11-cis-, 9-cis-, 9,13-dicis-, and 11,13-dicis-retinol [22]. Initial techniques, including colorimetric assays and spectrophotometric analyses, provided a starting point, but were restricted by issues related to specificity and sensitivity [23,24]. The interference posed by other dietary components further complicated the accurate quantification of vitamin A in complex matrices, such as food products, feed, and biological samples [25].
Overcoming these challenges required a collective effort from the scientific community. One critical hurdle was the development of robust extraction methods capable of efficiently isolating vitamin A from complex matrices, minimizing loss and cross-contamination [26]. The introduction of chromatographic techniques in the 1960s improved the precision and specificity of vitamin A determination [27]. However, the analytical landscape continued to face obstacles, particularly in quantifying trace amounts of vitamin A in diverse samples [28].
The integration of advanced spectroscopic methods, such as high-performance liquid chromatography (HPLC) and gas chromatography (GC), in the late 20th century addressed the need for heightened sensitivity [29,30,31,32,33]. These innovations facilitated the reliable detection and quantification of vitamin A at previously unattainable levels, ushering in a paradigm shift in the analytical approach to vitamin A analysis. Additionally, high-performance liquid chromatography has made it feasible to separate and determine the six cis–trans isomers of vitamin A, even in the form of commercially available esters like acetate, palmitate, and propionate [22].
The modern era has witnessed a renaissance in the field, characterized by the integration of mass spectrometry with chromatographic separations, providing unparalleled resolution and specificity in the determination of various vitamin A isomers [34,35,36]. Tandem mass spectrometry (MS/MS) and high-resolution mass spectrometry (HRMS) have further enhanced analytical capabilities, enabling the precise identification and quantification of vitamin A and its metabolites in intricate biological systems [37].
Furthermore, non-destructive analytical methods, such as nuclear magnetic resonance (NMR) spectroscopy and near-infrared spectroscopy (NIRS), have expanded the horizons of retinol analysis, allowing for the non-invasive monitoring of vitamin A dynamics within living systems as well as in premixes and feed [38,39]. These advancements have not only fostered a deeper comprehension of the physiological roles of vitamin A, but have also paved the way for tailored nutritional interventions and personalized healthcare strategies [40].
In conclusion, the journey through analytical techniques for vitamin A analysis highlights the resilience of the scientific community in unraveling the complexities of this essential micronutrient. The historical evolution, marked by challenges and refinements, has led to a sophisticated analytical landscape where modern methodologies have transcended the limitations of the past.
3. Quantitative Vitamin A Analysis: Diverse Analytical Approaches
In the field of nutritional science, the precise evaluation of vitamin A plays a pivotal role in understanding dietary sufficiency and identifying potential deficiencies. Similarly, the assessment of retinol levels in animal tissues is indispensable for gauging nutritional well-being, preventing diseases, and enhancing overall animal health and productivity. This chapter explores the intricate landscape of analytical techniques employed in the quantification of vitamin A, offering a succinct explanation of the fundamental principles that underlie these methodologies.
3.1. Colorimetric Assays
Colorimetric assays have long been employed as a dependable means of quantifying vitamin A or retinol within various samples [41]. The methodology involves introducing a chromogenic reagent into a soluble fortified food or feed sample, resulting in a reaction with retinol and the development of a distinctive color complex [42]. The intensity of the resultant color is directly proportional to the concentration of vitamin A in the sample. The fundamental principles of colorimetric assays used for measuring vitamin A are represented by the following methods:
- The Carr and Price assay: This method involves the quantitative evaluation of retinol utilizing antimony trichloride (SbCl3) as a crucial component [43].
- The Sobel and Werbin assay: This assay employs activated 1,3-dichloro-2-propanol to react with vitamin A, as initially proposed by Sobel and Werbin [44] and later expounded upon by Blake and Moran [23].
- Trifluoroacetic acid-based colorimetric determination: This technique relies on the interaction of a vitamin A solution in food or feed materials with several Lewis acids, resulting in the transient manifestation of a blue color [41].
Colorimetric assays offer several advantages. They are a cost-effective and straightforward alternative to complex methodologies such as high-performance liquid chromatography (HPLC) [45]. Additionally, their versatility enables the measurement of vitamin A in diverse samples, ranging from foods and feeds to biological fluids [46,47]. Nevertheless, utilizing colorimetric assays for measuring vitamin A or retinol does have its drawbacks. They tend to be less sensitive when compared to more sophisticated techniques like HPLC [48]. Moreover, the presence of interfering substances within the sample can significantly impact the accuracy of the results [49]. Lastly, the lack of specificity for vitamin A or retinol means that these assays can inadvertently identify other compounds forming stable color complexes with the reagent, leading to potential inaccuracies [50].
3.2. Spectrophotometric Analyses
Spectrophotometric analysis represents a widely utilized approach for the determination of retinol levels in food and feed samples [43,51]. This technique operates on the fundamental principle that all-trans-retinol in isopropanol exhibits maximal absorption at 325 nm [43,51]. The analytical methods for quantifying vitamin A rely on the effective dispersion of the fortified food, premix, or feed matrix to liberate vitamin A, including its various esters [42]. To facilitate this process, a UV-light irradiation system is employed, which serves to degrade the retinol. The setup for this system may include a simple configuration, such as employing a UV lamp along with a protective curtain to safeguard operators from potential exposure to light [52]. Key to the success of this approach is the meticulous determination of the optimal duration of irradiation and the precise positioning of the UV light in relation to the solutions under examination [42].
Spectrophotometric analysis for assessing retinol offers both advantages and challenges. It is lauded for its simplicity, cost-effectiveness, and high sensitivity to trace amounts of retinol [43]. Additionally, its non-destructive nature allows for sample reusability, contributing to resource conservation [45]. Extensive validation across various food and feed matrices underscores its reliability, yet the method is susceptible to yielding inaccurate results in the presence of interfering substances [43]. Ensuring accurate analysis requires meticulous sample preparation and the effective extraction of vitamin A from complex food/feed matrices [40]. The lack of specificity in differentiating retinol from other compounds with similar light absorption characteristics presents a challenge [40]. Notably, it is less suitable for the simultaneous determination of retinol and its esters, necessitating alternative analytical approaches in such cases [53].
3.3. Chromatographic Techniques
Chromatographic techniques for the analysis of vitamin A are grounded in the fundamental principle of differentiating components based on their distinct interactions with a stationary phase and a mobile phase [54]. These methodologies capitalize on the varying affinities exhibited by the different forms of vitamin A towards the stationary phase, facilitating their separation and eventual quantification [55,56]. Several types of chromatography have been employed for the purpose of vitamin A analysis.
- a.
- High-performance liquid chromatography (HPLC)
HPLC is a widely used analytical technique that employs high-pressure pumps to propel the mobile phase, comprising the sample, through a column containing the stationary phase [57]. The methodology for determining retinol in food, premix, feed, and biological samples using HPLC commonly involves an isocratic, aqueous solvent system consisting of a mixture of methanol and water as the mobile phase, enabling the isolation of retinol from the extracted sample [58,59]. The column is a crucial element in the HPLC setup, serving as the site for the separation of sample components [60]. The fundamental principle behind HPLC separation relies on the distribution of the analyte (sample) between the mobile phase (eluent) and the stationary phase (packing material) [61]. In the case of retinol, a reverse-phase column is commonly employed as the stationary phase, facilitating the segregation of retinol from other sample constituents based on variations in their hydrophobicity [62]. Prior to HPLC analysis, the retinol must be extracted from the sample and prepared for injection into the HPLC system. This typically involves a lipid extraction step aimed at eliminating interfering lipids and other components [63]. Subsequent to the separation of retinol by the HPLC system, identification and quantification become imperative. This task is typically accomplished using a UV detector, which gauges the absorbance of retinol at a specific wavelength [64].
- b.
- Gas–liquid chromatography (GLC)
GLC is an established analytical technique employed for the separation and analysis of volatile compounds within a given sample [65]. Prior to the chromatographic process, the sample undergoes an extraction and purification procedure to eliminate any potentially confounding substances [65]. The utilization of a glass column, packed with meticulously chosen glass beads, is imperative to enable the effective separation of retinol from other compounds within the sample [66].
A crucial aspect of GLC revolves around the selection of an inert carrier gas that does not interact with the sample constituents. Helium, known for its inert nature, has emerged as a preferred choice for the carrier gas in GLC [67]. Furthermore, precise regulation of the column temperature is paramount to ensure optimal separation of the compounds within the sample. For instance, the introduction of elevated column temperatures and extended retention times can facilitate the generation of anhydro-retinol during gas chromatography, thereby serving as an effective assay for retinol analysis [66].
To accurately detect the effluent from the column, the implementation of an appropriate detector, such as a mass spectrometer, is fundamental in the GLC process. This detector aids in the identification and quantification of the separated compounds, thereby facilitating comprehensive analysis and interpretation [65].
- c.
- Liquid–liquid chromatography (LLC)
LLC is a separation technique in which the stationary phase comprises a liquid supported on a solid, while the mobile phase is also a liquid. LLC amalgamates the principles of liquid–liquid extraction and chromatography [68]. Similar to chromatography, one of the phases involved remains stationary during the separation process. However, several significant distinctions differentiate LLC from traditional chromatography utilizing solid stationary phases, such as HPLC [68].
In the extraction of retinol from the sample, a suitable solvent is employed. The selection of the solvent is contingent upon the characteristics of the sample and the specific type of liquid chromatography utilized. Within this method, the compounds are segregated based on their distribution between the two immiscible liquid phases [62,69].
- d.
- Waters UltraPerformance Convergence Chromatography (UPC)
UPC represents a modern separation technique that harnesses compressed carbon dioxide as the primary mobile phase [70]. This method capitalizes on the utilization of sub-2 μm particle chromatography columns, taking advantage of the low-viscosity properties of CO2 and a sophisticated chromatography system. This approach distinguishes itself from conventional HPLC and notably enhances the sensitivity of the assay. Moreover, UPC demonstrates a significant reduction in solvent waste generation in comparison to traditional liquid chromatography [70].
- e.
- Ultra-high-performance liquid chromatography–tandem triple quadrupole mass spectrometry (UHPLC-MS/MS)
UHPLC, an advanced form of HPLC known for its enhanced capabilities in terms of resolution, analysis speed, and sensitivity [71], has paved the way for more sophisticated analytical methodologies. One such prominent application is the utilization of UHPLC-MS/MS for the precise determination of vitamin A levels in biological tissues, particularly in blood samples [72]. The approach demonstrates rapidity, accuracy, and heightened sensitivity, coupled with straightforward preprocessing procedures. Mass spectrometric analysis is carried out in the positive ion mode using the multiple reaction monitoring mode, while quantification is facilitated through the utilization of the internal standard method [72].
Generally, chromatographic techniques offer a multitude of benefits in the assessment of vitamin A, including superior sensitivity and specificity, adaptability across various sample types, and reduced sample preparation requirements [73,74]. Despite these advantages, chromatography methodologies also exhibit certain drawbacks, such as prolonged analysis time, significant financial investment, and susceptibility to matrix effects [45,73].
3.4. Nuclear Magnetic Resonance (NMR) Spectroscopy
NMR spectroscopy is a powerful analytical technique employed in the determination of retinol, leveraging the magnetic properties of hydrogen nuclei [75]. Grounded in the principle of resonance, it involves the matching of electromagnetic radiation frequencies to specific energy transitions of the nuclei, thereby facilitating the comprehensive analysis of the chemical structure and environment of retinol [76]. The phenomenon of chemical shift, denoting the displacement of the resonance frequency relative to a standard reference, plays a critical role in discerning the functional groups and molecular constituents of retinol [75]. Additionally, the integration of NMR signals allows for quantitative insights into the relative abundance of distinct types of hydrogen atoms within various retinol molecules [77].
NMR spectroscopy offers several advantages for the precise quantification of vitamin A in samples, owing to its high sensitivity, versatility, and dependable analytical capabilities [78]. However, it comes with certain drawbacks, including significant financial costs and the requirement for specialized equipment and expertise [79].
3.5. Near-Infrared Spectroscopy (NIRS)
NIRS is a non-invasive analytical technique widely used for studying the molecular structure, composition, and concentration of various substances [80]. It functions on the basis of the unique light absorption properties of different molecules at specific wavelengths, enabling the identification and quantification of compounds of interest. Near-infrared light, falling within the 800- to 2500-nanometer wavelength range, is particularly favored for its ability to penetrate biological samples effectively, making it a suitable tool for analyzing complex biological materials [81].
In practical applications, NIRS involves the transmission of near-infrared light through a sample, followed by the measurement of the absorbed or reflected light [82]. Through the analysis of absorption and reflection patterns, researchers can discern the presence and concentration of specific compounds or functional groups within the sample. Notably, NIR spectroscopy has also found utility in the examination of retinol within vitamin compositions [38].
NIRS is recognized as a non-destructive, expedient, and cost-effective technique for determining retinol in diverse samples [83,84]. Nevertheless, its sensitivity and accuracy are relatively restricted compared to standard reference methods such as HPLC, necessitating calibration with these methods [85]. It is imperative to note that NIRS might not be universally applicable across all sample types, as certain samples could potentially introduce interference during the analysis process [83].
3.6. Enzyme-Linked Immunosorbent Assays (ELISAs) for Biological Tissues
ELISA kits are used to measure the amount of a specific substance, such as vitamin A, in a sample. The kits use antibodies to detect and measure the substance, and there are different types of ELISA kits available depending on the specific needs of the experiment [86]. For example, the competitive EIA ELISA kit uses a competitive inhibition method to measure the amount of vitamin A in a sample [87,88], while the sandwich ELISA kit uses a sandwich method [89]. ELISA kits can be used to measure vitamin A in a variety of biological tissues, including plasma, serum, tissue homogenates, cell lysates, and cell culture supernatants [90].
ELISA kits for measuring vitamin A in biological tissues offer the advantages of high sensitivity and user-friendliness for processing multiple samples simultaneously, but they can be time-consuming and may lack sensitivity for detecting low levels of vitamin A [91]. Cross-reactivity with other substances in the sample can also lead to occasional false positives.
4. Deciphering the Factors: Unveiling the Complexities of Analytical Precision
Historically, the analysis of vitamin A has been recognized for its challenging reproducibility and repeatability [8]. For instance, AAFCO [92] states that there is an accepted analytical fluctuation of 30% in the measurement of vitamin A within feed samples. This deviation is typically encountered when analyzing a sample on two separate occasions, corresponding to twice the coefficient of variation or relative standard deviation [1]. Additionally, VDLUFA [93] provides a detailed reference, highlighting the allowable range for different concentrations of vitamin A within a sample (Table 1). Despite this acknowledged permissible fluctuation, the precise measurement of vitamin A in biological tissues, feed, and premixes remains essential for upholding the optimal nourishment and overall health of livestock and pets [4]. Numerous factors intricately influence the accuracy of this determination, rendering the analytical process complex [11]. In this section, we explore the various elements that significantly impact the accurate determination of vitamin A, shedding light on the difficulties encountered by analysts in maintaining analytical precision within this domain.

Table 1.
Vitamin A analysis leeway for premix and feed investigations [93].
The determination of vitamin A activity is not a straightforward process, and its accuracy is influenced by numerous complex factors:
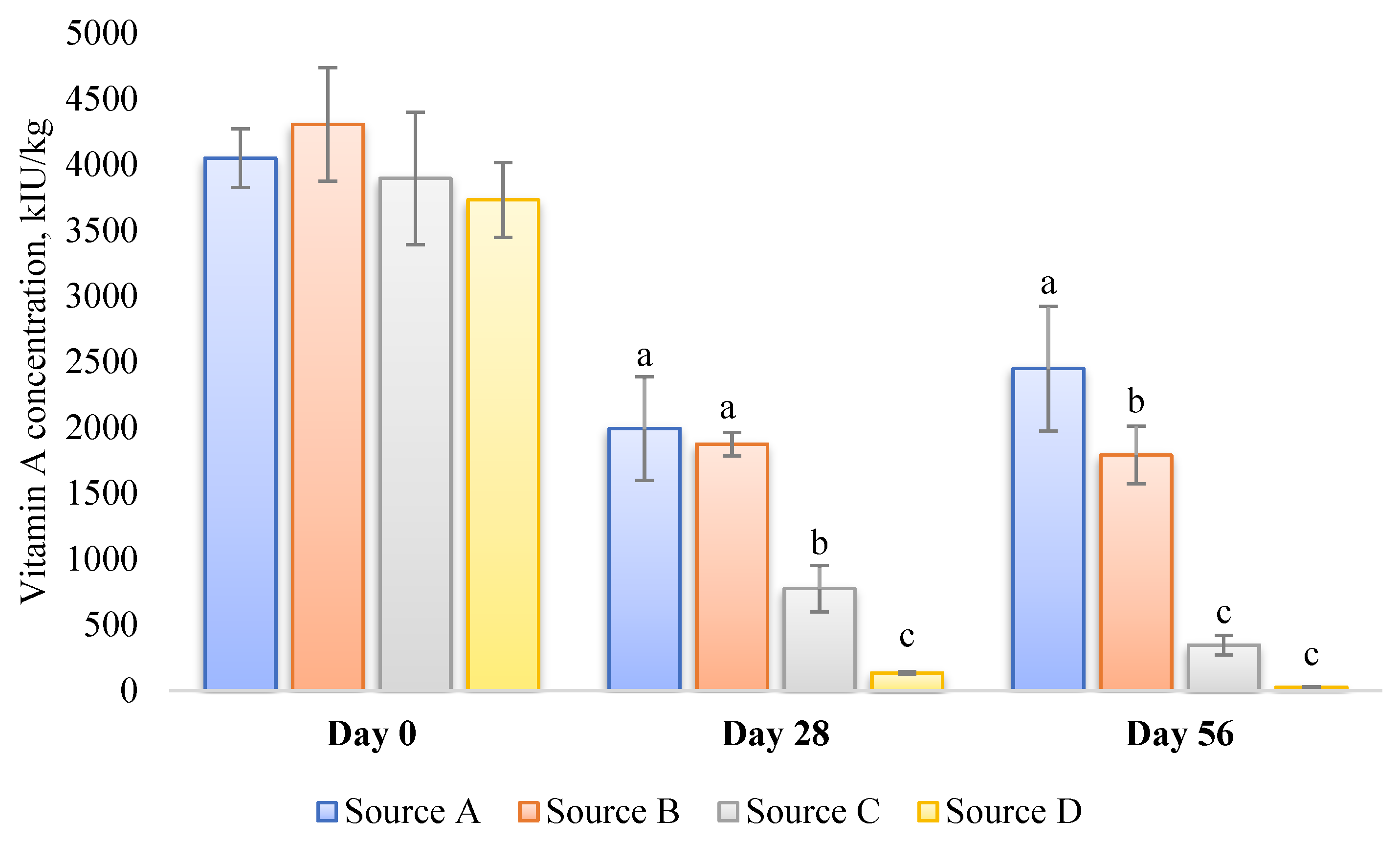
- Source of vitamin A: The susceptibility of various sources or commercial products of vitamin A to degradation can vary significantly due to differences in their formulation [94] (Figure 2). Factors such as light, oxygen, temperature, and moisture play crucial roles in the degradation process. Consequently, these variations can potentially influence the analytical outcomes, even if the initial activity of multiple vitamin A sources in an identical premix composition is similar. Furthermore, repeatability in retinol analysis is influenced by the physical properties of the vitamin A source (beadlet) utilized during the production of the premix or feed [8]. It is inversely correlated with the concentration of vitamin A present in the sample.
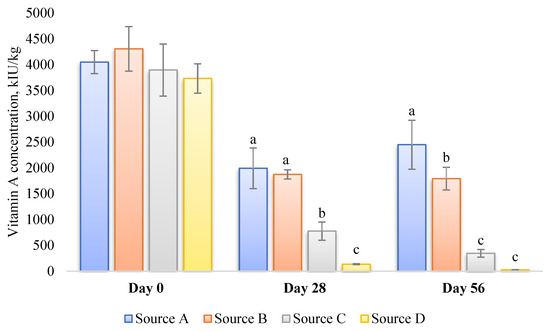 Figure 2. Stability of four different commercial vitamin A sources stored for up to 56 days in a broiler vitamin–mineral premix (incl., choline chloride) at 35 °C and 60–70% r.h. [94]. Values are presented as mean ± SD (n = 3). The premixes were prepared to contain 4.4 Mio IU vitamin A per kg. a–c within a time frame of 0, 28, or 56 d. Values not sharing a common superscript letter are significantly different (p < 0.05).
Figure 2. Stability of four different commercial vitamin A sources stored for up to 56 days in a broiler vitamin–mineral premix (incl., choline chloride) at 35 °C and 60–70% r.h. [94]. Values are presented as mean ± SD (n = 3). The premixes were prepared to contain 4.4 Mio IU vitamin A per kg. a–c within a time frame of 0, 28, or 56 d. Values not sharing a common superscript letter are significantly different (p < 0.05).
- 2.
- Type of sample: Different types of samples, such as premix, feed, blood, or other tissues, may necessitate distinct analytical methodologies [43].
- 3.
- Quality of the sample: The accuracy of analytical procedures can be significantly impacted by the quality of the sample. Contaminants or interfering substances within the sample can exert substantial influence on the physicochemical processes utilized during analysis [23].
- 4.
- Representativeness of the sample: Ensuring a representative sample is imperative. Ideally, the laboratory should only determine the amount of vitamin A present in the sample. If the sample does not accurately reflect the entire batch, the precision achieved is rendered ineffective.
- 5.
- Method of analysis: The precision of the outcomes can be influenced by the analytical approach employed [95]. Various methodologies may exhibit varied sensitivities to distinct configurations of vitamin A [23]. Furthermore, variations in the adherence of analysts to established and sanctioned protocols within a specific methodology may also exert an influence [96].
- 6.
- Laboratory: An empirical analysis reveals that the discrepancy in the precision of vitamin A analysis among different laboratories surpasses the variation attributed to differences in analytical methods [96]. Certain techniques or procedures can significantly contribute to substantial interlaboratory variation [96]. Examples of such techniques include the inconsistent reporting or calculation of results, particularly when comparing retinol palmitate with retinyl acetate. Furthermore, modifications made to the vitamin A analysis procedure, which lack validation through rigorous interlaboratory collaborative studies or statistically sound within-laboratory comparisons with validated test methods, can also be a source of significant variability. Additionally, within-laboratory sampling techniques may further compound this issue.
- 7.
- Storage conditions: The stability of vitamin A is known to be influenced by various storage conditions, including temperature, light exposure, and oxygen levels [97]. The improper storage of laboratory samples under such conditions can significantly impact the precision and reliability of the analysis.
- 8.
- Target tissue cellularity, integrity, and function (for biological tissues): Vitamin A status is characterized by the cellular structure, integrity, and functional capabilities of the target tissues. Unlike some biochemical indicators, any compromise in these aspects may require several weeks of restoration following vitamin A repletion or depletion [98].
- 9.
- Sample preparation approaches: It is crucial to emphasize the importance of obtaining an adequately sized initial sample for the evaluation. Moreover, it is essential to refrain from presuming uniform dispersion of vitamin A throughout the sample during the analysis [11]. Following the grinding process, it is imperative to ensure comprehensive remixing of the ground sample and repeat this process before proceeding with the weighing of a test portion [11].
- 10.
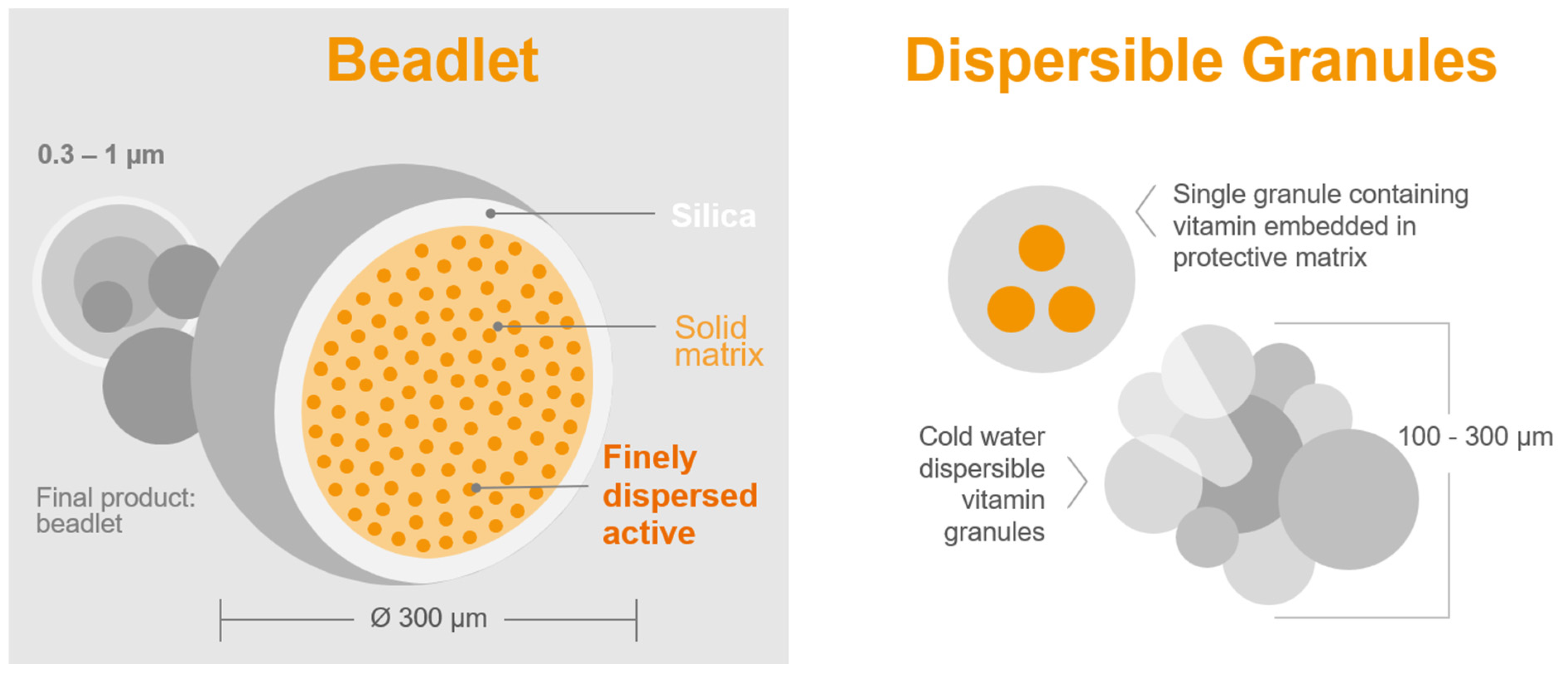
- Sample quantity for analysis: The precision of the chemical analysis of vitamin A in feed or premix samples is significantly affected by the weight of the sample. Dry vitamin A supplements are composed of beadlets (Figure 3 and Table 2) that contain multiple units of retinyl acetate [99]. When assessing a small sample of the feed, there might be a limited number of particles per sample [100]. A recent study by Inerowicz et al. [8] indicated that the relative standard deviations for vitamin A determinations in feed varied between 10.5–24.7% and 2.26–10.7% for sample sizes of 10 g and 100 g, respectively (Table 3). The findings of the study suggest that the mass of the sample can considerably influence the accuracy of vitamin A testing in animal feed materials.
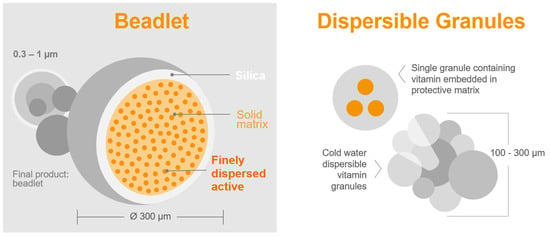 Figure 3. Microencapsulation: delivering a diverse range of products tailored to target applications [101].
Figure 3. Microencapsulation: delivering a diverse range of products tailored to target applications [101]. Table 2. Results of microscopic particle size measurements obtained through microscopic examination [8].
Table 2. Results of microscopic particle size measurements obtained through microscopic examination [8]. Table 3. Vitamin A content in 10 g and 100 g test portions with various samples [8].
Table 3. Vitamin A content in 10 g and 100 g test portions with various samples [8].
- 11.
- Analytical standards as benchmarks for the identification and quantification of retinol: The variability in the purity of these standards is a critical factor contributing to the observed inconsistencies among laboratories engaged in vitamin A analysis. In the comparison with the recognized US Pharmacopeia (USP) standard retinyl acetate, varying standards often display significant disparities in measurements, with values fluctuating between 50% and 140% of the officially stated value [11]. Regrettably, certain laboratories fail to validate the vitamin A content of the reference materials and exhibit insufficient quality control protocols for their analytical methods [96]. Laboratories exhibiting exemplary accuracy and precision continuously validate reference materials and incorporate in-house quality control samples, employing robust statistical methodologies to ensure and confirm the reliability of their results [96].
- 12.
- Extraction during analytical procedure: In certain instances, the presence of significant quantities of carotenoids following hydrolysis in the solution, coupled with a low concentration of vitamin A, might necessitate the implementation of multiple extraction procedures [102]. In the context of high-fat samples, the formation of extra soaps during the saponification process has the potential to influence the partition coefficient, thereby favoring the aqueous alcohol phase. Consequently, in such scenarios, it becomes imperative to conduct multiple extractions to ensure the efficient separation of retinol into the solvent [11]. According to Moore et al. [96], the primary cause of variation in retinol analysis in feed among various laboratories is, in fact, the vitamin extraction procedure.
- 13.
- Evaporation in the analytical phase: During the process of solvent evaporation, the thermal degradation of retinol solutions can occur, particularly at temperatures exceeding 40 degrees Celsius [11]. Thus, it is vital to control and maintain the temperature below this threshold to prevent the degradation of retinol. Additionally, it is essential to minimize the exposure of the retinol residues to ambient air, as this could potentially compromise the stability of the solution [11].
- 14.
- Other factors: During the analytical process, other factors, such as the isomerization of all-trans retinol, quality control protocols, precise equipment calibration, potential human errors, systematic and bias errors, and various other influences, could potentially impact the final analytical results [11].
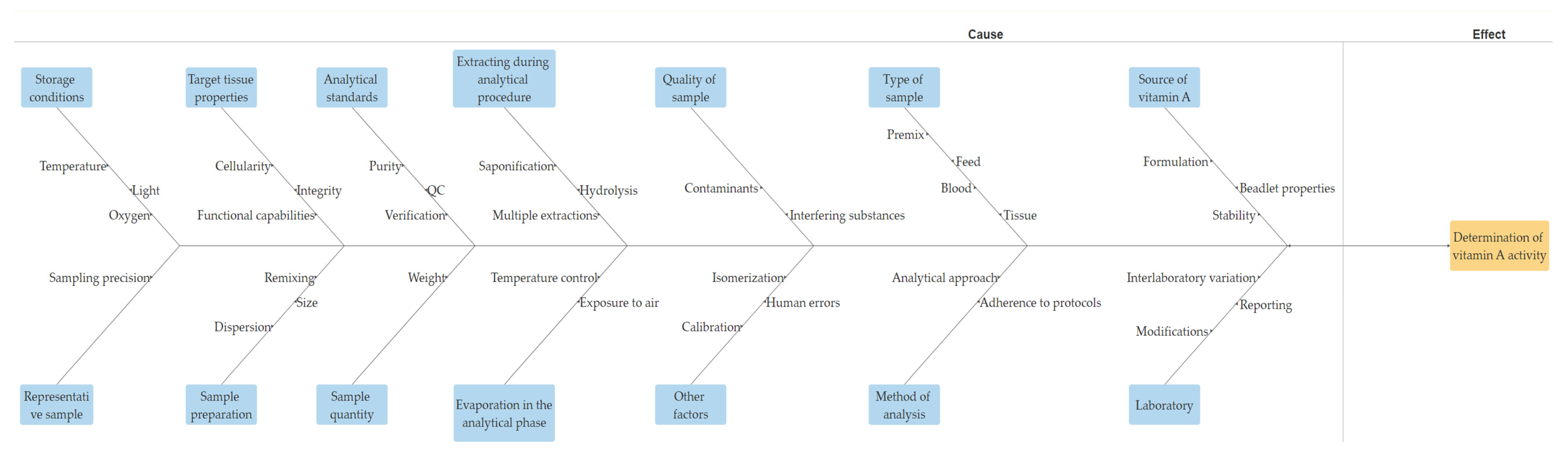
In summary, analyzing vitamin A in different samples is a complex process that requires careful consideration of various factors. Factors such as sample type, quantity, storage conditions, analytical determinants, and a range of other parameters pose challenges for maintaining accurate measurements of retinol. Understanding and managing these factors are crucial for producing high-quality animal feed and premixes that meet nutritional requirements. Figure 4 illustrates an Ishikawa diagram detailing the factors influencing the determination of vitamin A.
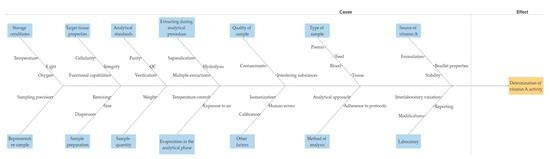
Figure 4.
Factors affecting the determination of vitamin A.
6. Reflection and Future Prospects: Charting the Course for Enhanced Analytical Precision
One of the key findings highlighted in this overview emphasizes the crucial importance of recognizing the complex interplay between the sampling of test materials, sample preparation, and the selected analytical approach in the analysis of retinol. Variations in sample matrices, including diverse feeds like total mixed ration and complimentary feed, as well as various biological samples, consistently underscore the need for tailored methodologies. It is apparent that a standardized approach often falls short in capturing the intricacies within these matrices [120,121,122]. Furthermore, the need for robust preservation protocols to account for the stability and potential degradation of vitamin A during storage and analysis has been highlighted in recent research [94].
Delving deeper into the range of influencing factors, the role of method validation has emerged as a crucial factor in ensuring the reliability and accuracy of vitamin A determination [123,124]. A meticulous validation process, including considerations for specificity, linearity, and precision, serves as a safeguard against erroneous results [122]. Moreover, the recognition that calibration strategies must be adapted to the characteristics of the analytical technique employed underscores the necessity for a nuanced and adaptable approach [125].
A significant aspect gleaned from the review of historical progress in vitamin A analysis techniques is the continual evolution of analytical technologies. Traditional HPLC has evolved into more advanced methodologies such as chromatography on-line coupled to mass spectrometry, offering greater precision and sensitivity [59,76,78,126]. The integration of novel separation techniques, alongside advanced detection methodologies, shows promise in mitigating the challenges posed by complex matrices and low concentrations [127,128]. Embracing these cutting-edge technologies is crucial to achieving enhanced analytical precision.
Envisioning the future, interdisciplinary collaboration and technological advancements will serve as the foundation for achieving unparalleled precision in vitamin A determination [109,129,130]. Recognizing the multifaceted nature of the challenges at hand, interdisciplinary collaboration, including experts from nutrition science, analytical chemistry, and bioinformatics, presents an opportunity to develop a holistic approach to understanding vitamin A analysis [131,132,133]. By creating a collaborative ecosystem that facilitates the exchange of knowledge and methodologies, comprehensive frameworks for accurate determination can be synthesized.
Simultaneously, the rapid advancement of miniaturized and portable analytical devices offers the potential for on-site analysis and real-time monitoring, minimizing the risk of sample degradation and allowing for immediate intervention [48,134]. Additionally, the integration of data science and analytical chemistry can leverage advanced algorithms and machine learning techniques to interpret complex datasets, unravel intricate patterns, and enhance the predictive capabilities of retinol determination [135,136].
Moving forward, it is crucial to prioritize research and development initiatives that bring together the strengths of various disciplines and harness the potential of cutting-edge technologies. By fostering an ecosystem that promotes collaborative innovation and embraces technological advancements, the goal of achieving unparalleled analytical precision in vitamin A determination can be realized. Pursuing this vision not only promises to revolutionize the field of nutritional analysis, but also has the potential to drive advancements in analytical methodologies across various domains, paving the way for a future characterized by unprecedented precision and reliability.
7. Conclusions
Based on the information provided in this scientific review paper, the following conclusions can be drawn:
- The accurate determination of vitamin A is crucial for animal health and product quality.
- Historical advancements in analysis techniques have evolved from basic methods to sophisticated chromatographic and spectroscopic approaches, improving precision and sensitivity.
- Various factors, including sample quality, the method of analysis, and storage conditions, significantly impact analytical precision in retinol determination, necessitating a comprehensive understanding and careful consideration.
- Emphasizing the critical role of quality control through stringent protocols and regular proficiency testing is essential for ensuring consistent and reliable results.
- Future progress in analytical precision lies in the integration of advanced technologies, such as miniaturized devices and data-driven approaches, promising to overcome current challenges and enhance accuracy in vitamin A analysis.
In light of these insights, a concerted focus on addressing the complexities of vitamin A analysis through continued advancements in technology and stringent quality control measures is imperative to ensuring precise and reliable determination, thereby bolstering animal welfare and feed safety standards.
Author Contributions
Conceptualization, Y.S.; methodology, Y.S. and W.P.; software, Y.S.; validation, W.P. and A.K.; formal analysis, W.P.; investigation, Y.S.; resources, Y.S. and W.P.; data curation, Y.S. and A.K.; writing—original draft preparation, Y.S.; writing—review and editing, W.P. and A.K.; visualization, Y.S.; supervision, W.P.; project administration, Y.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
No new data were created or analyzed in this study.
Conflicts of Interest
All three authors of this work are affiliated with BASF, a manufacturer of vitamins and carotenoids, including vitamin A. Nevertheless, it is crucial to underscore that the content of this manuscript has been sourced exclusively from scientific peer-reviewed data. Our unwavering commitment lies in upholding transparency and adhering to ethical research principles.
References
- FAO (Food and Agriculture Organization of the United Nation). Quality assurance for animal feed analysis laboratories. In FAO Animal Production and Health Manual No. 14; FAO: Rome, Italy, 2011. [Google Scholar]
- Khan, R.U.; Khan, A.; Naz, S.; Ullah, Q.; Puvača, N.; Laudadio, V.; Mazzei, D.; Seidavi, A.; Ayasan, T.; Tufarelli, V. Pros and Cons of Dietary Vitamin A and Its Precursors in Poultry Health and Production: A Comprehensive Review. Antioxidants 2023, 12, 1131. [Google Scholar] [CrossRef]
- Shastak, Y.; Gordillo, A.; Pelletier, W. The relationship between vitamin A status and oxidative stress in animal production. J. Appl. Anim. Res. 2023, 51, 546–553. [Google Scholar] [CrossRef]
- Shastak, Y.; Pelletier, W. The role of vitamin A in non-ruminant immunology. Front. Anim. Sci. 2023, 4, 1197802. [Google Scholar] [CrossRef]
- Shastak, Y.; Pelletier, W. Delving into Vitamin A Supplementation in Poultry Nutrition: Current Knowledge, Functional Effects, and Practical Implications. Worlds Poult. Sci. J. 2023, 79. [Google Scholar] [CrossRef]
- Shastak, Y.; Pelletier, W. Vitamin A supply in swine production: Current science and practical considerations. Appl. Anim. Sci. 2023, 39, 289–305. [Google Scholar] [CrossRef]
- Shastak, Y.; Pelletier, W. Balancing Vitamin A Supply for Cattle: A Review of the Current Knowledge. In Advances in Animal Science and Zoology 21; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2023; Chapter 2; Available online: https://novapublishers.com/wp-content/uploads/2023/11/Advances-in-Animal-Science-and-Zoology.-Volume-21-Chapter-2.pdf (accessed on 14 December 2023).
- Inerowicz, H.D.; Novotny, L.; Ramsey, C.A.; Riter, K.L.; Swarbrick, M.; Thiex, N. Effect of Test Portion Mass on Vitamin A Testing in Animal Feed Materials. J. AOAC Int. 2022, 105, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Stroka, J.; Dehouck, P.; Bouten, K.; Serano, F.; Stroh, A.; von Holst, C. Determination of the Content of Vitamin A in Compound Feed; Proficiency Testing Report FAC-22/01; European Commission: Brussels, Belgium, 2023. [CrossRef]
- Czuba, L.C.; Zhong, G.; Yabut, K.C.; Isoherranen, N. Analysis of vitamin A and retinoids in biological matrices. Methods Enzymol. 2020, 637, 309–340. [Google Scholar] [CrossRef] [PubMed]
- Thiex, N.; Smallidge, R.; Beine, R. Sources of Error in Vitamin A Analysis. J. AOAC Int. 1996, 79, 1269–1276. [Google Scholar] [CrossRef][Green Version]
- AAFCO (The Association of American Feed Control Officials). Determination of Vitamin A in Animal Feed by HPLC/UV. In Proceedings of the AAFCO Annual Meeting, Bellevue, WC, USA, 10–12 August 2017; Available online: https://www.aafco.org/wp-content/uploads/2023/01/201708_Determination_of_Vitamin_A_in_Animal_Feed.pdf (accessed on 22 October 2023).
- Munsell, H.E. Vitamin A: Methods of assay and sources in food. JAMA 1938, 111, 245–252. [Google Scholar] [CrossRef]
- Parrish, D.B. Determination of vitamin A in foods-a review. CRC Crit. Rev. Food Sci. Nutr. 1977, 9, 375–394. [Google Scholar] [CrossRef]
- Kaur, H.; Kewalramani, N.; Garg, M.R.; Kumar, P. Methodology for simultaneous estimation of vitamin A and E in animal feeds using high performance liquid chromatography. Indian J. Anim. Sci. 2004, 74, 1236–1238. [Google Scholar]
- Semba, R.D. On the ‘Discovery’ of Vitamin, A. Ann. Nutr. Metab. 2012, 61, 192–198. Available online: https://www.jstor.org/stable/48508230 (accessed on 20 October 2023). [CrossRef]
- Anonymous. Isolation of Vitamin A. Nature 1932, 129, 88. [Google Scholar] [CrossRef]
- Holmes, H.N.; Corbet, R.E. The Isolation of Crystalline Vitamin A1. J. Am. Chem. Soc. 1937, 59, 2042–2047. [Google Scholar] [CrossRef]
- Brown, P.; Blum, W.; Stern, M. Isomers of vitamin A in fish liver oils. Nature 1959, 184, 1377–1379. [Google Scholar] [CrossRef]
- Braekkan, O.; Myklestad, H.; Njaa, L.; Utne, F. Vitamin A Isomers in the Liver of Rats and Chicks. Nature 1960, 186, 312. [Google Scholar] [CrossRef]
- Pitt, G. Chemical structure and vitamin A activity. Proc. Nutr. Soc. 1965, 24, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Steuerle, H. Untersuchungen zur Bildung von cis-Isomeren aus all-trans-Vitamin-A-Acetat während des alkalischen Aufschlusses bei der Bestimmung von Vitamin A in Futtermitteln. Z. Lebensm. Unters. Forsch. 1985, 181, 400–403. [Google Scholar] [CrossRef] [PubMed]
- Blake, J.A.; Moran, J.J. An improved colorimetric procedure for the analysis of vitamin A. Can. J. Chem. 1976, 54, 1757–1764. [Google Scholar] [CrossRef]
- Rutkowski, M.; Grzegorczyk, K. Modifications of spectrophotometric methods for antioxidative vitamins determination convenient in analytic practice. Acta Sci. Pol. Technol. Aliment. 2007, 6, 17–28. [Google Scholar]
- Md Noh, M.F.; Gunasegavan, R.D.; Mustafa Khalid, N.; Balasubramaniam, V.; Mustar, S.; Abd Rashed, A. Recent Techniques in Nutrient Analysis for Food Composition Database. Molecules 2020, 25, 4567. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, W.E.; Yan, J.Q.; Liu, M.; Zhou, Y.; Shen, X.; Ma, Y.L.; Feng, X.S.; Yang, J.; Li, G.H. A Review of the Extraction and Determination Methods of Thirteen Essential Vitamins to the Human Body: An Update from 2010. Molecules 2018, 23, 1484. [Google Scholar] [CrossRef]
- Bilek, M.; Namieśnik, J. Chromatographic techniques in pharmaceutical analysis in poiand: History and the presence on the basis of papers published in selected polish pharmaceutical journals in xx century. Acta Pol. Pharm. 2016, 73, 605–612. [Google Scholar] [PubMed]
- Köseoğlu, K.; Ulusoy, H.I.; Yilmaz, E.; Soylak, M. Simple and sensitive determination of vitamin A and E in the milk and egg yolk samples by using dispersive solid phase extraction with newly synthesized polymeric material. J. Food Compos. Anal. 2020, 90, 103482. [Google Scholar] [CrossRef]
- Sheppard, A.J.; Prosser, A.R.; Hubbard, W.D. Gas chromatography of the fat-soluble vitamins: A review. J. Am. Oil Chem. Soc. 1972, 49, 619–633. [Google Scholar] [CrossRef] [PubMed]
- Bohman, O.; Engdahl, K.-A.; Johnsson, H. High performance liquid chromatography of vitamin A. J. Chem. Educ. 1982, 59, 251. [Google Scholar] [CrossRef]
- Smidt, C.R.; Jones, A.D.; Clifford, A.J. Gas chromatography of retinol and alpha-tocopherol without derivatization. J. Chromatogr. 1988, 434, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Ryndakova, I.A.; Grigor’eva, M.P.; Veshchikov, V.V.; Zolotareva, S.I.; Dracheva, O.V. Use of high performance liquid chromatography for analysis of vitamin A in food products. Vopr. Pitan. 1991, 1991, 64–68. [Google Scholar]
- Lee, B.L.; Chua, S.C.; Ong, H.Y.; Ong, C.N. High-performance liquid chromatographic method for routine determination of vitamins A and E and beta-carotene in plasma. J. Chromatogr. 1992, 581, 41–47. [Google Scholar] [CrossRef]
- Van Breemen, R.B.; Huang, C.R. High-performance liquid chromatography-electrospray mass spectrometry of retinoids. FASEB J. 1996, 10, 1098–1101. [Google Scholar] [CrossRef]
- Goetz, H.J.; Kopec, R.E.; Riedl, K.M.; Cooperstone, J.L.; Narayanasamy, S.; Curley, R.W., Jr.; Schwartz, S.J. An HPLC-MS/MS method for the separation of α-retinyl esters from retinyl esters. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2016, 6, 1029–1030. [Google Scholar] [CrossRef]
- Ertugrul, S.; Yucel, C.; Sertoglu, E.; Ozkan, Y.; Ozgurtas, T. Development and optimization of simultaneous determination of fat soluble vitamins by liquid chromatography tandem mass spectrometry. Chem. Phys. Lipids 2020, 230, 104932. [Google Scholar] [CrossRef]
- Dunning, M.W. Quantification and Profiling of Hepatic Retinoids in Freshwater Fishes by Liquid Chromatography Tandem Mass Spectrometry. Master’s Thesis, University of Waterloo, Waterloo, ON, Canada, 2018. Available online: https://uwspace.uwaterloo.ca/bitstream/handle/10012/13059/Dunning_Michael.pdf?sequence=3 (accessed on 23 October 2023).
- Bodin, J.C.; Dussert, L.; D’alfonso, T. Near Infrared Spectroscopic Analysis of Vitamins. World Intellectual Property Organization Patent WO2000039562A1, 22 December 1999. Application PCT/EP1999/010502. [Google Scholar]
- Eiff, J.; Monakhova, Y.B.; Diehl, B.W. Multicomponent analysis of fat- and water-soluble vitamins and auxiliary substances in multivitamin preparations by qNMR. J. Agric. Food Chem. 2015, 63, 3135–3143. [Google Scholar] [CrossRef]
- Carazo, A.; Macákova, K.; Matoušová, K.; Krčmová, K.; Protti, M.; Mladěnka, P. Vitamin A update: Forms, sources, kinetics, detection, function, deficiency, therapeutic use and toxicity. Nutrients 2021, 13, 1703. [Google Scholar] [CrossRef]
- Dugan, R.E.; Frigerio, N.A.; Siebert, J.M. Colorimetric Determination of Vitamin A and Its Derivatives with Trifluoroacetic Acid. Anal. Chem. 1964, 36, 114–117. [Google Scholar] [CrossRef]
- Dary, O.; Arroyave, G. Manual for Sugar Fortification with Vitamin A Part 3: Analytical Methods for the Control and Evaluation of Sugar Fortification with Vitamin A; INCAP, USAID/OMNI: Arlington, VA, USA, 1996.
- Egbuonu, R. Evaluation of Colorimetric Determination of Vitamin A in Foods. Master’s Thesis, Texas Tech University, Lubbock, TX, USA, 1986. [Google Scholar]
- Sobel, A.E.; Werbin, H. Spectrophotometric study of a new colorimetrie reaction of vitamin A. J. Biol. Chem. 1945, 159, 681–691. [Google Scholar] [CrossRef]
- Cortés-Herrera, C.; Artavia, G.; Leiva, A.; Granados-Chinchilla, F. Liquid Chromatography Analysis of Common Nutritional Components, in Feed and Food. Foods. 2018, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Mueller, V.; Lengerken, V.; Wetterau, H. Vitamin A determination in industrially produced feeds. Part 1. Determination of vitamin A in pre-mixes and mixes of active ingredients. Nahrung 1974, 18, 425–437. [Google Scholar] [CrossRef]
- Alqahtani, N.S. Vitamin A Quantification using Photometric Methods. J. Nutr. Weight. Loss. 2021, 6, 140. [Google Scholar]
- Huey, S.L.; Krisher, J.T.; Morgan, D.; Mkambula, P.; Srinivasan, B.; Gannon, B.M.; Mbuya, M.N.N.; Mehta, S. Portable Devices for Measurement of Vitamin A Concentrations in Edible Oil: Field Readiness of Available Options. ACS Omega 2022, 7, 17502–17518. [Google Scholar] [CrossRef] [PubMed]
- Koehn, C.J.; Sherman, W.C. The determination of vitamin a and carotene with the photoelectric colorimeter. J. Biol. Chem. 1940, 132, 527–538. [Google Scholar] [CrossRef]
- Bayfield, R.; Cole, E. Colorimetric estimation of vitamin A with trichloroacetic acid. Methods Enzymol. 1980, 67, 189–195. [Google Scholar] [CrossRef]
- U.S.P. The Pharmacopeia of the United States of America; 18th Revision; Mack Printing Co.: Easton, PA, USA, 1970; pp. 775, 914. [Google Scholar]
- Arroyave, G.; de Funes, C. Enriquecimiento de azUcar con vitamina A. Metodo para la determinacion cuantitativa de retinol en azUcar blanca de mesa. Arch. Latinoam. Nutr. 1974, 24, 147–153. [Google Scholar]
- Rathi, D.N.G.; Rashed, A.A.; Noh, M.F.M. Determination of retinol and carotenoids in selected Malaysian food products using high-performance liquid chromatography (HPLC). SN Appl. Sci. 2022, 4, 93. [Google Scholar] [CrossRef]
- Coskun, O. Separation techniques: Chromatography. North Clin. Istanb. 2016, 3, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Braithwaite, A.; Smith, F.J. Theoretical Considerations in Chromatographic Methods; Springer: Dordrecht, The Netherlands, 1985; pp. 24–84. [Google Scholar] [CrossRef]
- Rodriguez, E.L.; Poddar, S.; Iftekhar, S.; Suh, K.; Woolfork, A.G.; Ovbude, S.; Pekarek, A.; Walters, M.; Lott, S.; Hage, D.S. Affinity chromatography: A review of trends and developments over the past 50 years. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2020, 1157, 122332. [Google Scholar] [CrossRef]
- Bower, P. High-Performance Liquid Chromatography. J. Vis. Exp. 2023. Available online: https://app.jove.com/cn/v/10156/operation-of-high-performance-liquid-chromatography-hplc (accessed on 29 October 2023).
- Al-Abdulaly, A.B. Determination of Retinol by Aqueous Reverse Phase Open Column System. Ph.D. Thesis, University of Rhode Island, Kingston, RI, USA, 1986. Available online: https://digitalcommons.uri.edu/oa_diss/543 (accessed on 5 December 2023).
- Hosain, M.Z.; Islam, S.M.S.; Kamal, M.M.; Rahman, M.M. Quantitative Analysis of Fat-Soluble Vitamins in Feed Additives Using an In-House Developed and Validated HPLC Method. Austin. J. Anal. Phar. M Chem. 2022, 9, 1143. [Google Scholar]
- GMI. Exploring the Key Components of an HPLC System: A Comprehensive Guide. HPLC, Chromatography. 2023. Available online: https://www.gmi-inc.com/exploring-the-key-components-of-an-hplc-system-a-comprehensive-guide/#:~:text=The%20column%20is%20where%20the,for%20achieving%20the%20desired%20separation (accessed on 29 October 2023).
- Boettcher, J.; Margraf, M.; Monks, K. HPLC Basics—Principles and parameters. VSP0019, KNAUER Wissenschaftliche Geräte GmbH, Hegauer Weg 38, 14163 Berlin. 2023. Available online: https://www.knauer.net/Application/application_notes/VSP0019_HPLC%20Basics%20-%20principles%20and%20parameters_final%20-web-.pdf (accessed on 29 October 2023).
- Kim, Y.K.; Quadro, L. Reverse-phase high-performance liquid chromatography (HPLC) analysis of retinol and retinyl esters in mouse serum and tissues. Methods Mol. Biol. 2010, 652, 263–275. [Google Scholar] [CrossRef]
- Howells, D.W.; Brown, I.R.; Brooke, O.G.; Newey, V. A simple automated injection technique for the high-pressure liquid chromatographic determination of plasma retinol. Ann. Clin. Biochem. 1983, 20, 308–311. [Google Scholar] [CrossRef]
- Kane, M.A.; Folias, A.E.; Napoli, J.L. HPLC/UV quantitation of retinal, retinol, and retinyl esters in serum and tissues. Anal. Biochem. 2008, 378, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Falaki, F. Sample Preparation Techniques for Gas Chromatography; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Ryhage, R. Use of a mass spectrometer as a detector and analyzer for effluents emerging from high temperature gas liquid chromatography columns. Anal. Chem. 1964, 36, 759–764. [Google Scholar] [CrossRef]
- Pitt, J.J. Principles and applications of liquid chromatography-mass spectrometry in clinical biochemistry. Clin. Biochem. Rev. 2009, 30, 19–34. [Google Scholar]
- Minceva, M. Liquid-Liquid Chromatography. Wiley Analytical Science. 2016. Available online: https://analyticalscience.wiley.com/content/article-do/liquid-liquid-chromatography (accessed on 29 October 2023).
- Maryutina, T.A.; Savonina, E.Y.; Fedotov, P.S.; Smith, R.M.; Siren, H.; Hibbert, D.B. Terminology of separation methods (IUPAC Recommendations 2017). Pure Appl. Chem. 2018, 90, 181–231. [Google Scholar] [CrossRef]
- Gu, G.; Brosig, K.; Kennedy, L.; Oglobline, A.; Richardson, G.; Walker, B. Simultaneous Analysis of Vitamin A and D3 in Vitamin Premixes and Concentrates by Convergence Chromatography/PDA Detection; Dairy Technical Services Ltd.: Waters Corporation, MA, USA, 2019; Available online: https://www.waters.com/content/dam/waters/en/app-notes/2014/720005220/720005220-en.pdf (accessed on 30 October 2023).
- Turner, J.E.; Jenkins, K.M. The Evolution of Ultra High-Performance Liquid Chromatography: Expanding the Future of Separation Technologies. Chromatography Today May/June 2018. 2018. Available online: https://www.chromatographytoday.com/article/hplc-uhplc/31/waters-corporation/pthe-evolution-of-ultra-high-performance-liquid-chromatography-expanding-the-future-of-separation-technologiesp/2376 (accessed on 30 October 2023).
- Chen, X.; Gong, Z.; Shen, S. Determination of vitamin A and vitamin E in human serum by ultra-high performance liquid chromatography-tandem triple quadrupole mass spectrometry. Wei Sheng Yan Jiu 2021, 50, 301–307. [Google Scholar] [CrossRef]
- Eitenmiller, R.R. Strengths and weaknesses of assessing vitamin content of foods. J. Food Qual. 1990, 13, 1–69. [Google Scholar] [CrossRef]
- Jornet-Martínez, N.; Moliner-Martínez, Y.; Molins-Legua, C.; Campíns-Falcó, P. Trends for the Development of In Situ Analysis Devices. In Encyclopedia of Analytical Chemistry; Meyers, R.A., Ed.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2017. [Google Scholar] [CrossRef]
- Choi, Y.H.; Kim, H.K.; Wilson, E.G.; Erkelens, C.; Trijzelaar, B.; Verpoorte, R. Quantitative analysis of retinol and retinol palmitate in vitamin tablets using 1H-nuclear magnetic resonance spectroscopy. Anal. Chim. Acta 2004, 512, 141–147. [Google Scholar] [CrossRef]
- Koshani, R.; Jafari, S.M.; van de Ven, T.G.M. Going deep inside bioactive-loaded nanocarriers through Nuclear Magnetic Resonance (NMR) spectroscopy. Trends Food Sci. Technol. 2020, 101, 198–212. [Google Scholar] [CrossRef]
- Bharti, S.K.; Roy, R. Quantitative 1H NMR spectroscopy. TrAC Trends Anal. Chem. 2012, 35, 5–26. [Google Scholar] [CrossRef]
- Williamson, K.; Hatzakis, E. NMR Spectroscopy as a Robust Tool for the Rapid Evaluation of the Lipid Profile of Fish Oil Supplements. J. Vis. Exp. 2017, 123, 55547. [Google Scholar] [CrossRef]
- Balthazar, C.F.; Guimarães, J.T.; Rocha, R.S.; Pimentel, T.C.; Neto, R.P.C.; Tavares, M.I.B.; Graça, J.S.; Filho, E.G.A.; Freitas, M.Q.; Esmerino, E.A.; et al. Nuclear magnetic resonance as an analytical tool for monitoring the quality and authenticity of dairy foods. Trends Food Sci. Technol. 2021, 108, 84–91. [Google Scholar] [CrossRef]
- Bec, K.B.; Grabska, J.; Huck, C.W. Near-Infrared Spectroscopy in Bio-Applications. Molecules 2020, 25, 2948. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, Y.; Genkawa, T.; Futami, Y. Near-infrared spectroscopy. In Encyclopedia of Spectroscopy and Spectrometry; Lindon, J.C., Tranter, G.E., Koppenaal, D.W., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 40–49. [Google Scholar] [CrossRef]
- Jaren, C.; Lopez, A.; Arazuri, S. Advanced analytical techniques for quality evaluation of potato and its products. In Advances in Potato Chemistry and Technology Book; Jaspreet, S., Lovedeep, K., Eds.; Academic Press: Cambridge, MA, USA, 2016; Chapter 19; pp. 563–602. [Google Scholar]
- Pires, F.F.; Lemos, M.C.; Petersen, J.C.; Kessler, A.M. Use of Near-Infrared Reflectance Spectroscopy to Analyze Vitamin Content. J. Appl. Poult. Res. 2001, 10, 412–418. [Google Scholar] [CrossRef]
- Jia, L.P.; Tian, S.L.; Zheng, X.C.; Jiao, P.; Jiang, X.P. Application of near-infrared spectroscopy in the detection of fat-soluble vitamins in premix feed. In Proceedings of the Fourth Seminar on Novel Optoelectronic Detection Technology and Application, Nanjing, China, 24–26 October 2017; Volume 10697, p. 106971U. [Google Scholar] [CrossRef]
- Tamura, Y.; Inoue, H.; Takemoto, S.; Hirano, K.; Miyaura, K. A Rapid Method to Measure Serum Retinol Concentrations in Japanese Black Cattle Using Multidimensional Fluorescence. J. Fluoresc. 2021, 31, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Alhajj, M.; Zubair, M.; Farhana, A. Enzyme Linked Immunosorbent Assay; StatPearls Publishing LLC: Tampa, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK555922/ (accessed on 3 November 2023).
- Lequin, R.M. Enzyme Immunoassay (EIA)/Enzyme-Linked Immunosorbent Assay (ELISA). Clin. Chem. 2005, 51, 2415–2418. [Google Scholar] [CrossRef] [PubMed]
- Aydin, S. A short history, principles, and types of ELISA, and our laboratory experience with peptide/protein analyses using ELISA. Peptides 2015, 72, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Osmekhina, E.; Neubauer, A.; Klinzing, K.; Myllyharju, J.; Neubauer, P. Sandwich ELISA for quantitative detection of human collagen prolyl 4-hydroxylase. Microb. Cell Fact. 2010, 9, 48. [Google Scholar] [CrossRef] [PubMed]
- Innovative Research. 2023. Available online: https://www.innov-research.com/products/vitamin-a-elisa-kit (accessed on 27 November 2023).
- Hosseini, S.; Vázquez-Villegas, P.; Rito-Palomares, M.; Martinez-Chapa, S.O. Advantages, Disadvantages and Modifications of Conventional ELISA. In Enzyme-Linked Immunosorbent Assay (ELISA); Springer Briefs in Applied Sciences and Technology; Springer: Singapore, 2018. [Google Scholar] [CrossRef]
- AAFCO (The Association of American Feed Control Officials). Official Publication. 2011, pp. 298–299. Available online: https://www.aafco.org/resources/official-publication/ (accessed on 2 December 2023).
- VDLUFA (Association of German Agricultural Analytic and Research Institutes e. V.). Analysis Leeways (ASR) Version 13 (2022); VDLUFA-Verlag: Darmstadt, Germany, 2022; p. 13. [Google Scholar]
- Hirai, R.A.; De Leon, D.; Randig-Biar, M.; Silva, A.; Sanchez, E.; McElroy, A.P.; Bailey, C.A.; Martinez, N.; Sokale, A.; Music, L. Evaluation of the stability of vitamin A acetate concentrates mixed in a vitamin-trace mineral premix over a 56-day high temperature and humidity storage stress. In Proceedings of the 2023 the International Poultry Scientific Forum, Atlanta, GA, USA, 23–24 January 2023; p. 112. [Google Scholar]
- Parrish, D.B. Study of the Method for Vitamin A in Mixed Feeds. J. AOAC Int. 1960, 43, 30–34. [Google Scholar] [CrossRef]
- Moore, W.R.; DeVries, J.; MacDonald, J.; Hare, L.; Carson, J.; Chaudhari, P.; DeVries, J.; Fontana, J.; Golz, P.; King, J.; et al. Assessing analytical variability of measurement of vitamin A in corn-soy blend. J. AOAC Int. 2010, 93, 638–649. [Google Scholar] [CrossRef]
- Duarte Fávaro, R.M.; Iha, M.H.; Mazzi, T.C.; Fávaro, R.; de Lourdes Pires Bianchi, M. Stability of vitamin A during storage of enteral feeding formulas. Food Chem. 2011, 126, 827–830. [Google Scholar] [CrossRef]
- NASEM (The National Academies of Sciences, Engineering, and Medicine). Dietary Reference Intakes for Vitamin, A.; Vitamin, K.; Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. Institute of Medicine (US) Panel on Micronutrients. 2001. Available online: https://www.ncbi.nlm.nih.gov/books/NBK222318/ (accessed on 31 October 2023).
- Lehman, R.W. A Statistical Procedure for Estimating Vitamin A Assay Variation Caused by Particulate Distribution of Dry Vitamin A in Feed Samples. J. Assoc. Off. Agric. Chem. 1960, 43, 15–20. [Google Scholar] [CrossRef]
- Tinkler, F.H.; Hanley, J.B.; Lehman, R.W. The Use of Large Samples in the Determination of Vitamin A in Mixed Feeds Fortified with Dry Vitamin A Supplements. J. AOAC Int. 1960, 43, 25–28. [Google Scholar] [CrossRef]
- Shastak, Y.; Pelletier, W. Nutritional Balance Matters: Assessing the Ramifications of Vitamin A Deficiency on Poultry Health and Productivity. Poultry 2023, 2, 493–515. [Google Scholar] [CrossRef]
- Parrish, D.B. Vitamin A in Mixed Feeds, Premixes, and Foods: Development of Method. J. AOAC Int. 1974, 57, 897–902. [Google Scholar] [CrossRef]
- Taverniers, I.; Van Bockstaele, E.; De Loose, M. Trends in quality in the analytical laboratory. I. Traceability and measurement uncertainty of analytical results. Trends Anal. Chem. 2004, 23, 480–490. [Google Scholar] [CrossRef]
- IYTE (Izmir Yüksek Teknoloji Enstitüsü). Chapter 5: Errors in Chemical Analyses. 2023. Available online: https://web.iyte.edu.tr/~serifeyalcin/lectures/chem201/cn_5.pdf (accessed on 31 October 2023).
- Yadav, V. Food Analysis and Quality Control. Government Polytechnic, Mandi Adampur, Hisar, Haryana, India. 2023. Available online: https://gpadampur.files.wordpress.com/2011/11/6-2-faqc-class-notes-08022014.pdf (accessed on 31 October 2023).
- Galyean, M.L. Laboratory Procedures in Animal Nutrition Research; Department of Animal and Food Sciences Texas Tech University: Lubbock, TX, USA, 2010; Available online: https://www.depts.ttu.edu/agriculturalsciences/vetSciences/mgalyean/lab_man.pdf (accessed on 1 November 2023).
- Krishna, G. Livestock Nutrition: Analytical Techniques; New India Pub Agency: New Delhi, India, 2012; p. 763. [Google Scholar]
- de Jonge, L.H.; Jackson, F.S. The feed analysis laboratory: Establishment and quality control. Setting up a feed analytical laboratory, and implementing a quality assurance system compliant with ISO/IEC 17025:2005. In Animal Production and Health Guidelines No. 15; Makkar, H.P.S., Ed.; FAO: Rome, Italy, 2005. [Google Scholar]
- Holland, I.; Davies, J.A. Automation in the Life Science Research Laboratory. Front. Bioeng. Biotechnol. 2020, 8, 571777. [Google Scholar] [CrossRef]
- Hurst, W.J. Automation in Food and Agricultural Laboratories. JALA J. Assoc. Lab. Automat. 2009, 14, A9–A15. [Google Scholar] [CrossRef]
- von Holst, C.; Boix, A.; Vincent, U. Addressing a multidisciplinary challenge from animal nutrition to analytical science. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2017, 34, 1271. [Google Scholar] [CrossRef]
- Makkar, H.P.S.; Strnad, I.; Mittendorfer, J. Proficiency Testing of Feed Constituents: A Comparative Evaluation of European and Developing Country Laboratories and Its Implications for Animal Production. J. Agric. Food Chem. 2016, 64, 7679–7687. [Google Scholar] [CrossRef] [PubMed]
- Dragacci, S.; Grosso, F.; Pfauwathel-Marchond, N.; Fremy, J.M.; Venant, A.; Lombard, B. Proficiency testing for the evaluation of the ability of European Union-National Reference laboratories to determine aflatoxin M1 in milk at levels corresponding to the new European Union legislation. Food Addit. Contam. 2001, 18, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Thiex, N.J.; Ramsey, C.A. Piloting a Proficiency Testing Program for Laboratory Sampling of Animal Feed Materials. J. AOAC Int. 2023, 106, 472–483. [Google Scholar] [CrossRef] [PubMed]
- Earnshaw, A.; Smith, R.A.; Owen, L. How proficiency testing can improve the quality of analytical data using vitamin analysis as an example. Food Chem. 2009, 113, 781–783. [Google Scholar] [CrossRef]
- Sykes, M.; Croucher, J.; Smith, R.A. Proficiency testing has improved the quality of data of total vitamin B2 analysis in liquid dietary supplement. Anal. Bioanal. Chem. 2011, 400, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Nemser, S.; Lindemann, S.; Chen, Y.; Lopez, S.; Pickens, S.; Ulaszek, J.; Kmet, M.; Powers, C.; Ensley, S.; Schrunk, D.; et al. A review of proficiency exercises offered by the Veterinary Laboratory Investigation and Response Network (Vet-LIRN) and Moffett Proficiency Testing Laboratory from 2012 to 2018. Accredit. Qual. Assur. 2021, 26, 143–156. [Google Scholar] [CrossRef]
- Waugh, C.; Clark, G. Factors affecting test reproducibility among laboratories. Rev. Sci. Tech. 2021, 40, 131–143. [Google Scholar] [CrossRef]
- Malomo, G.A.; Ihegwuagu, N.E. Some Aspects of Animal Feed Sampling and Analysis [Internet]. In Ideas and Applications Toward Sample Preparation for Food and Beverage Analysis; InTech: London, UK, 2017. [Google Scholar] [CrossRef]
- Greaves, R.; Jolly, L.; Woollard, G.; Hoad, K. Serum vitamin A and E analysis: Comparison of methods between laboratories enrolled in an external quality assurance programme. Ann. Clin. Biochem. 2010, 47 Pt 1, 78–80. [Google Scholar] [CrossRef]
- Binkley, N.; Sempos, C.T. Vitamin D Standardization Program (VDSP). Standardizing vitamin D assays: The way forward. J. Bone Min. Res. 2014, 29, 1709–1714. [Google Scholar] [CrossRef]
- Greaves, R.F.; Woollard, G.A.; Hoad, K.E.; Walmsley, T.A.; Johnson, L.A.; Briscoe, S.; Koetsier, S.; Harrower, T.; Gill, J.P. Laboratory medicine best practice guideline: Vitamins a, e and the carotenoids in blood. Clin. Biochem. Rev. 2014, 35, 81–113. [Google Scholar]
- AOAC (the Association of Official Agricultural Chemists). AOAC Guidelines for Single Laboratory Validation of Chemical Methods for Dietary Supplements and Botanicals. 2002. Available online: https://s27415.pcdn.co/wp-content/uploads/2020/01/64ER20-7/Validation_Methods/d-AOAC_Guidelines_For_Single_Laboratory_Validation_Dietary_Supplements_and_Botanicals.pdf (accessed on 2 November 2023).
- Thompson, L.B.; Schimpf, K.J.; Stiner, L.A.; Schmitz, D.J. Determination of vitamin A (retinol) in infant and medical nutritional formulas with AOAC method 992.06 using a modified extraction procedure: Single-laboratory validation. J. AOAC Int. 2010, 93, 1523–1529. [Google Scholar] [CrossRef]
- Cuadros-Rodriguez, L.; Gamiz-Gracia, L.; Almansa-Lopez, E.; Bosque-Sendra, J.M. Calibration in chemical measurement processes. II. A methodological approach. TrAC Trends Anal. Chem. 2001, 20, 620–636. [Google Scholar] [CrossRef]
- Donno, D.; Mellano, M.G.; Gamba, G.; Riondato, I.; Beccaro, G.L. Analytical Strategies for Fingerprinting of Antioxidants, Nutritional Substances, and Bioactive Compounds in Foodstuffs Based on High Performance Liquid Chromatography-Mass Spectrometry: An Overview. Foods 2020, 9, 1734. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.N.; Patel, J.K.; Patel, M.P.; Rajput, G.C.; Patel, H.A. Introduction to hyphenated techniques and their applications in pharmacy. Pharm. Methods 2010, 1, 2–13. [Google Scholar] [CrossRef]
- Corradini, D.; Nicoletti, I. High performance separation techniques for identification, characterization and quantification of plant secondary metabolites with health-promoting properties. In Proceedings of the 30th International Symposium on the Chemistry of Natural Products, Athens, Greece, 25–29 November 2018. [Google Scholar]
- Betz, J.M.; Rimmer, C.A.; Saldanha, L.G.; Phillips, M.M.; Andrews, K.W.; Wise, S.A.; Wood, L.J.; Kuszak, A.J.; Gusev, P.A.; Pehrsson, P.R. Challenges in Developing Analytically Validated Laboratory-Derived Dietary Supplement Databases. J. Nutr. 2018, 148 (Suppl. 2), 1406S–1412S. [Google Scholar] [CrossRef]
- Alonso, N.; Zelzer, S.; Eibinger, G.; Herrmann, M. Vitamin D Metabolites: Analytical Challenges and Clinical Relevance. Calcif Tissue Int. 2023, 112, 158–177. [Google Scholar] [CrossRef]
- Ferranti, P. The future of analytical chemistry in foodomics. Curr. Opin. Food Sci. 2018, 22, 102–108. [Google Scholar] [CrossRef]
- Valdés, A.; Álvarez-Rivera, G.; Socas-Rodríguez, B.; Herrero, M.; Ibáñez, E.; Cifuentes, A. Foodomics: Analytical Opportunities and Challenges. Anal. Chem. 2022, 94, 366–381. [Google Scholar] [CrossRef] [PubMed]
- Słowik-Borowiec, M.; Głąb, N.; Stach, S.; Szpyrka, E. A Miniaturized Sample Preparation Method for the Determination of Vitamins A and E in Food Products. Molecules 2023, 28, 3449. [Google Scholar] [CrossRef]
- Ahmadi, M.; Amouzegar, Z.; Khalili, S.; Asadi, S.; Aghajani, S.; Aryanrad, P.; Afkhami, A.; Madrakian, T.; Thomas, S.; Nguyen, T.A. Chapter 1—Miniaturization—An introduction to miniaturized analytical devices. In Micro and Nano Technologies, Micro- and Nanotechnology Enabled Applications for Portable Miniaturized Analytical Systems; Thomas, S., Ahmadi, M., Nguyen, T.A., Afkhami, A., Madrakian, T., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 3–16. [Google Scholar] [CrossRef]
- Szymańska, E. Modern data science for analytical chemical data—A comprehensive review. Anal. Chim. Acta 2018, 1028, 1–10. [Google Scholar] [CrossRef]
- Dotzert, M. The Power of Algorithms in Analytical Chemistry. Creating a Positive Lab Culture 15(6). 2020. Available online: https://www.labmanager.com/the-power-of-algorithms-in-analytical-chemistry-23167 (accessed on 2 November 2023).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).