Input Selection Drives Molecular Logic Gate Design
Abstract
:1. Introduction
2. Analytes
2.1. Potential Toxic Metals
2.2. Alkaline and Alkaline Earth Metals
2.3. Anionic Species
2.4. Aminoacids
3. Optical Detection Devices
- Internal (or intramolecular) charge transfer (ICT): An electron-rich donor moiety transfers charge to an electron-deficient acceptor moiety; therefore, the entire process occurs within a single molecule [96].
- Metal–ligand charge transfer (MLCT): Involves the transfer of electronic charge between a metal ion and a ligand, as the d orbitals of the metal atom and the ligand orbitals (p, d, or f, depending on the type of ligand) can overlap. It predominantly occurs in metal complexes [97].
4. Molecular Logic Gates
4.1. General Strategies for Building MLGs
4.2. Strategies for Building MLGs Based on Molecular Architecture
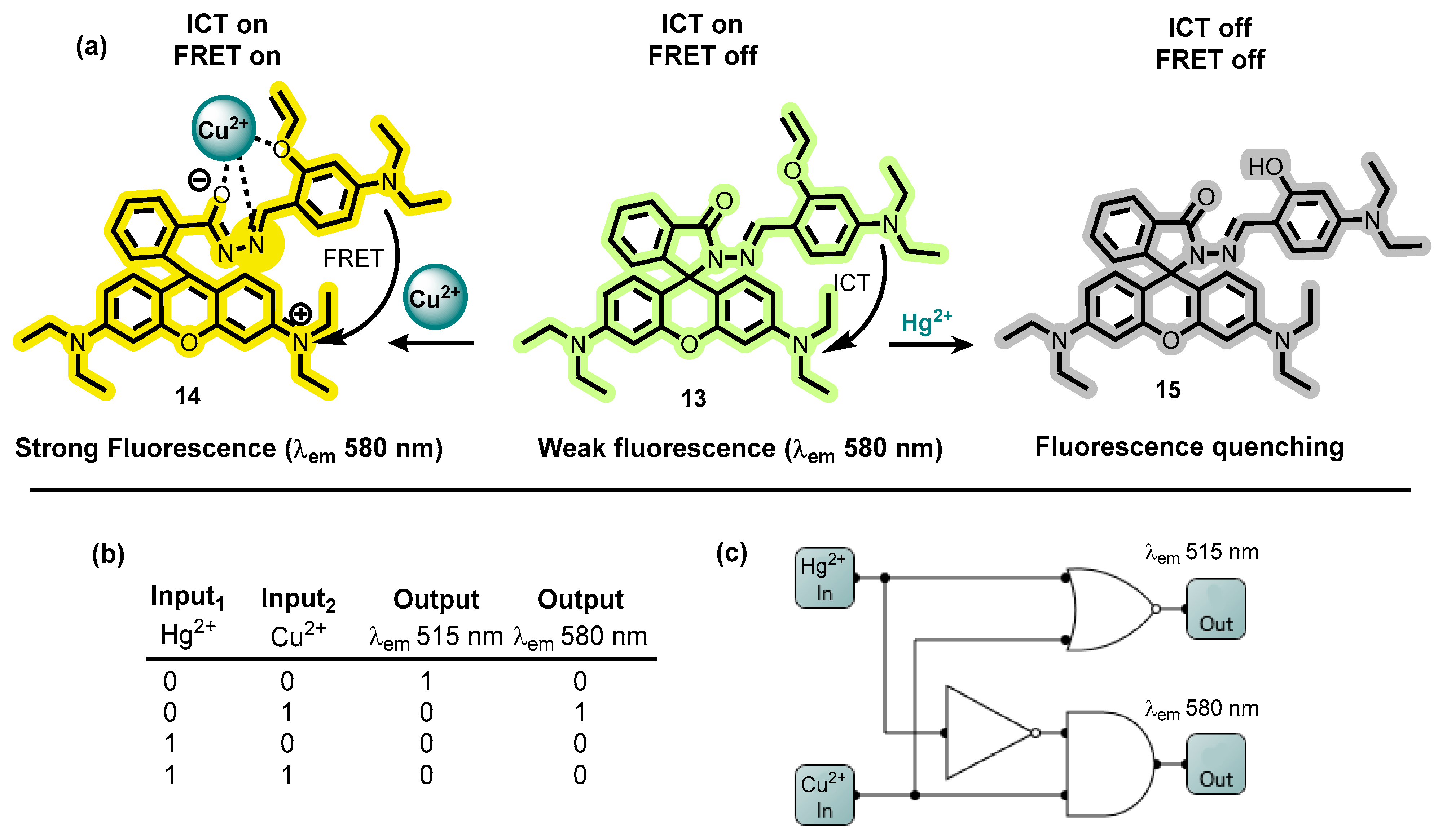
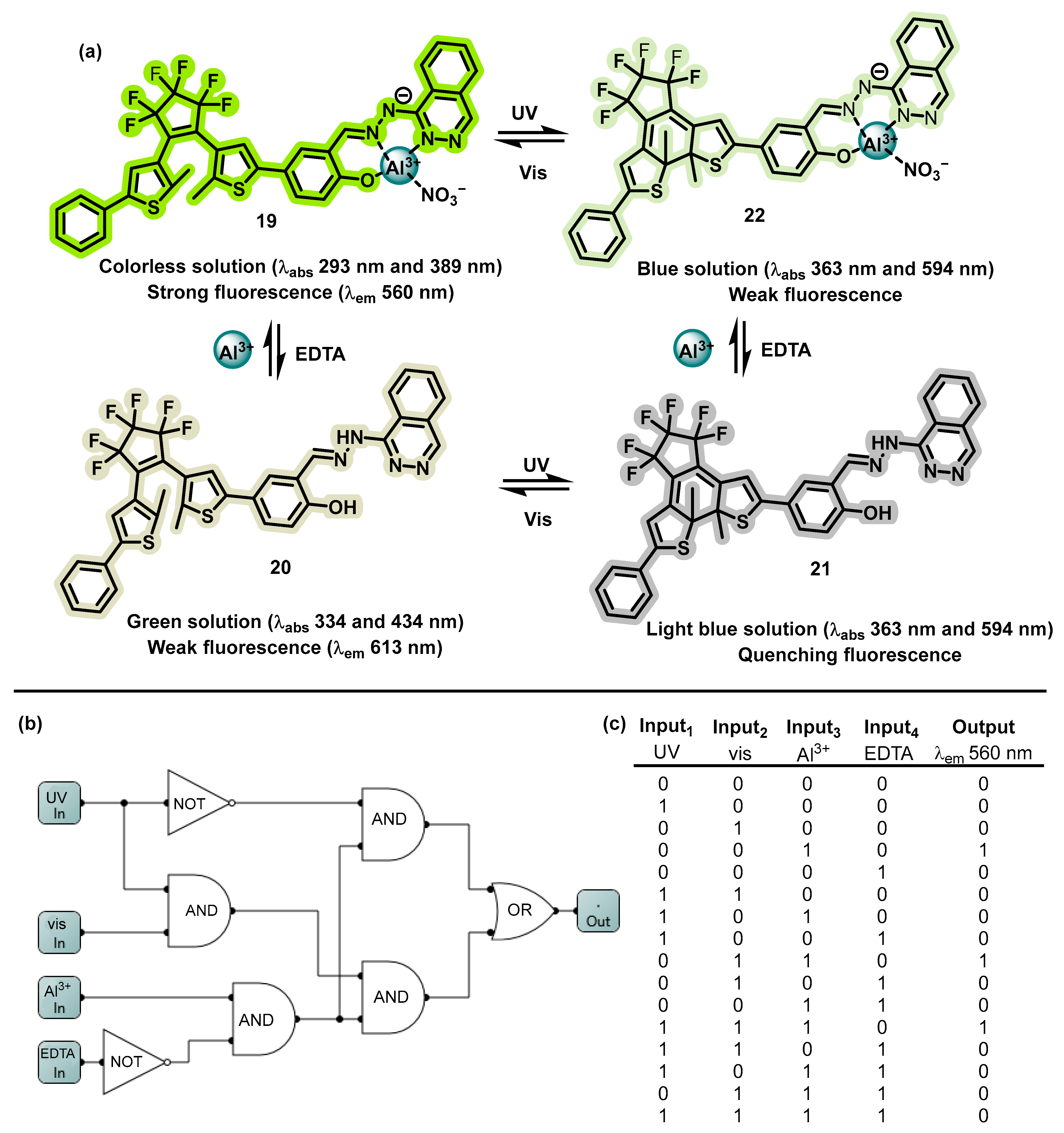
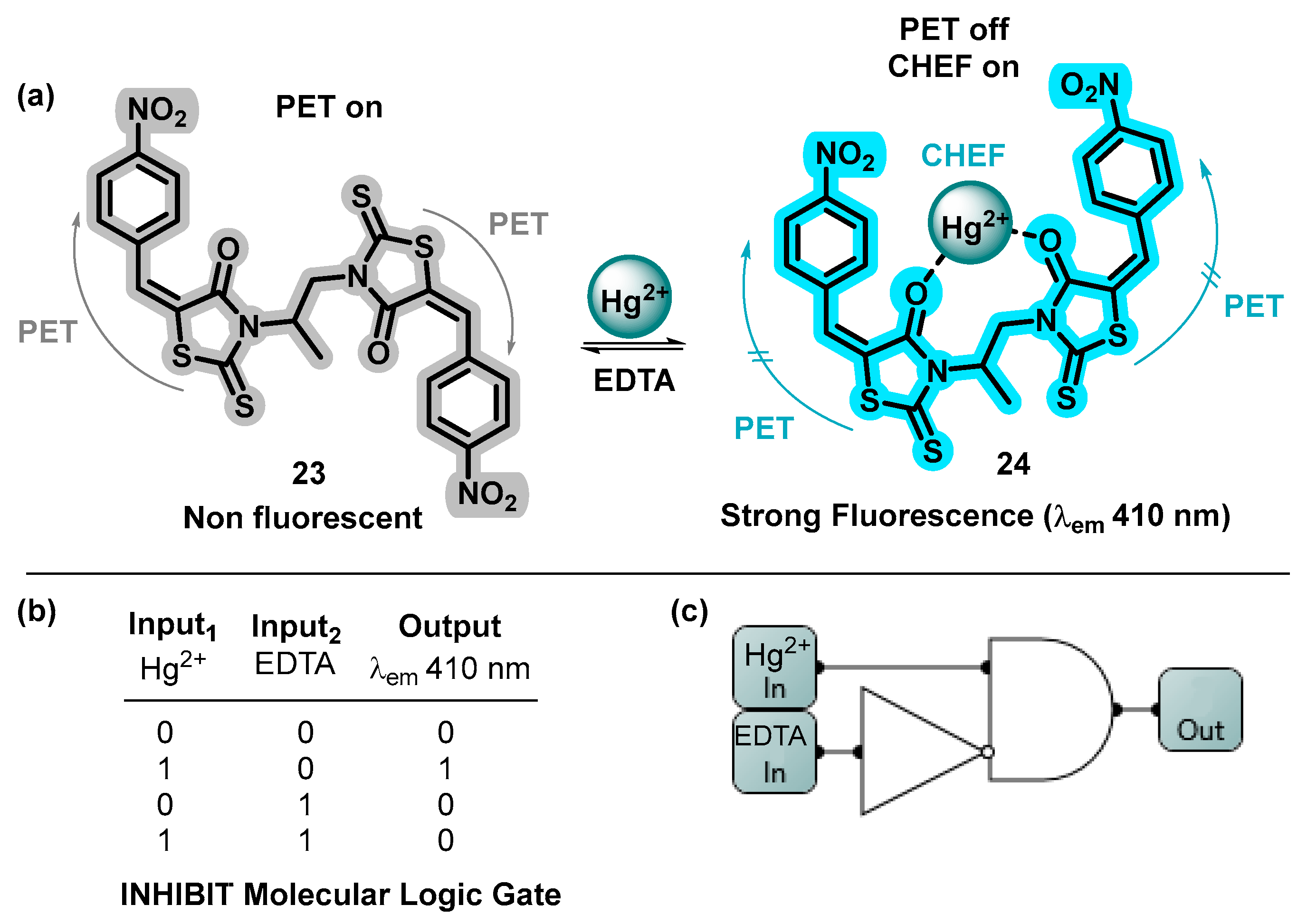
4.3. MLGs Based on EDTA as Input2
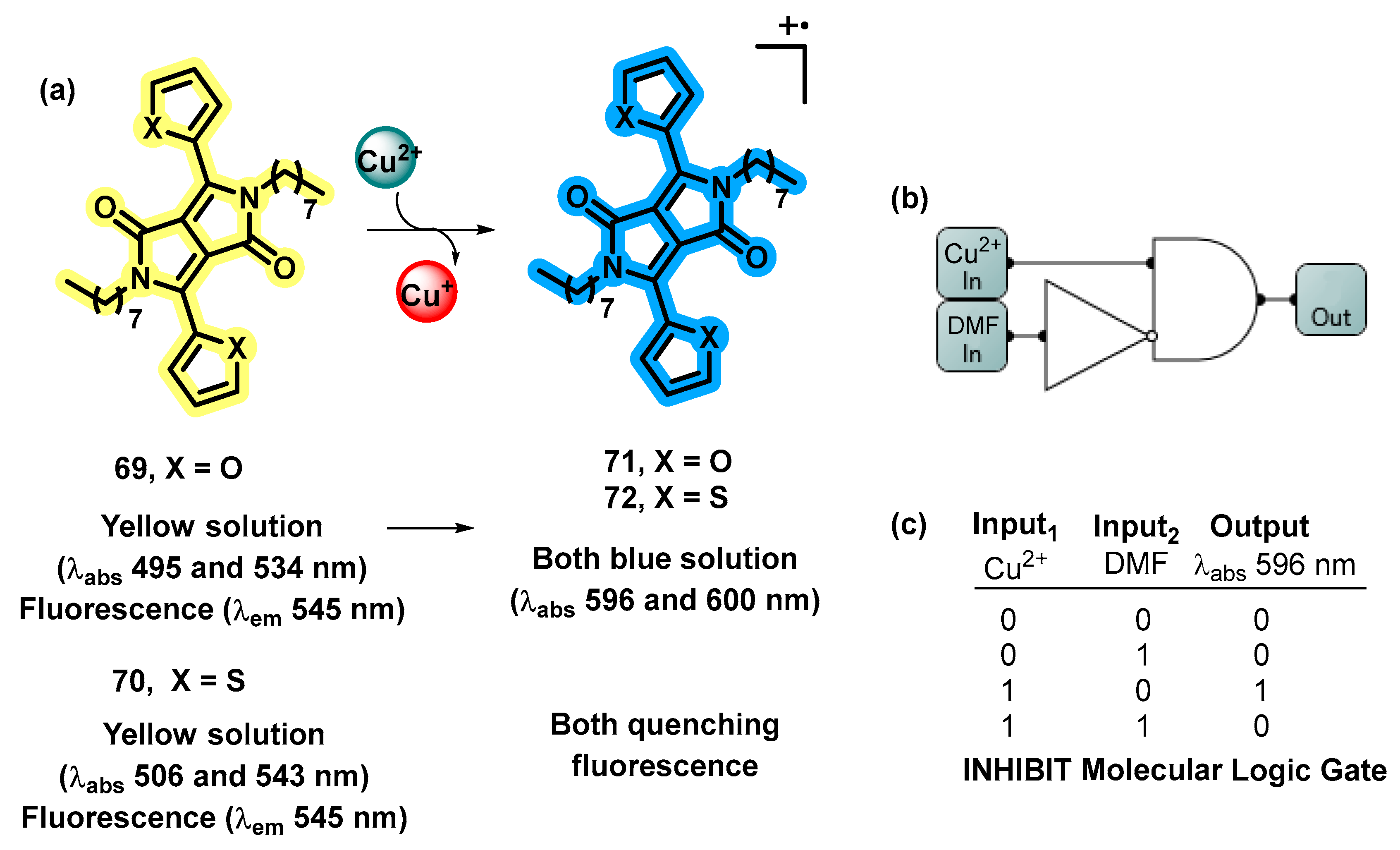
4.4. MLGs Based on Sulfides, Halides, Carbonate, and Cyanides as Input2
4.5. MLGs Based on Phosphate and Derivatives as Input2
4.6. MLGs Based on Other Anions as Input2
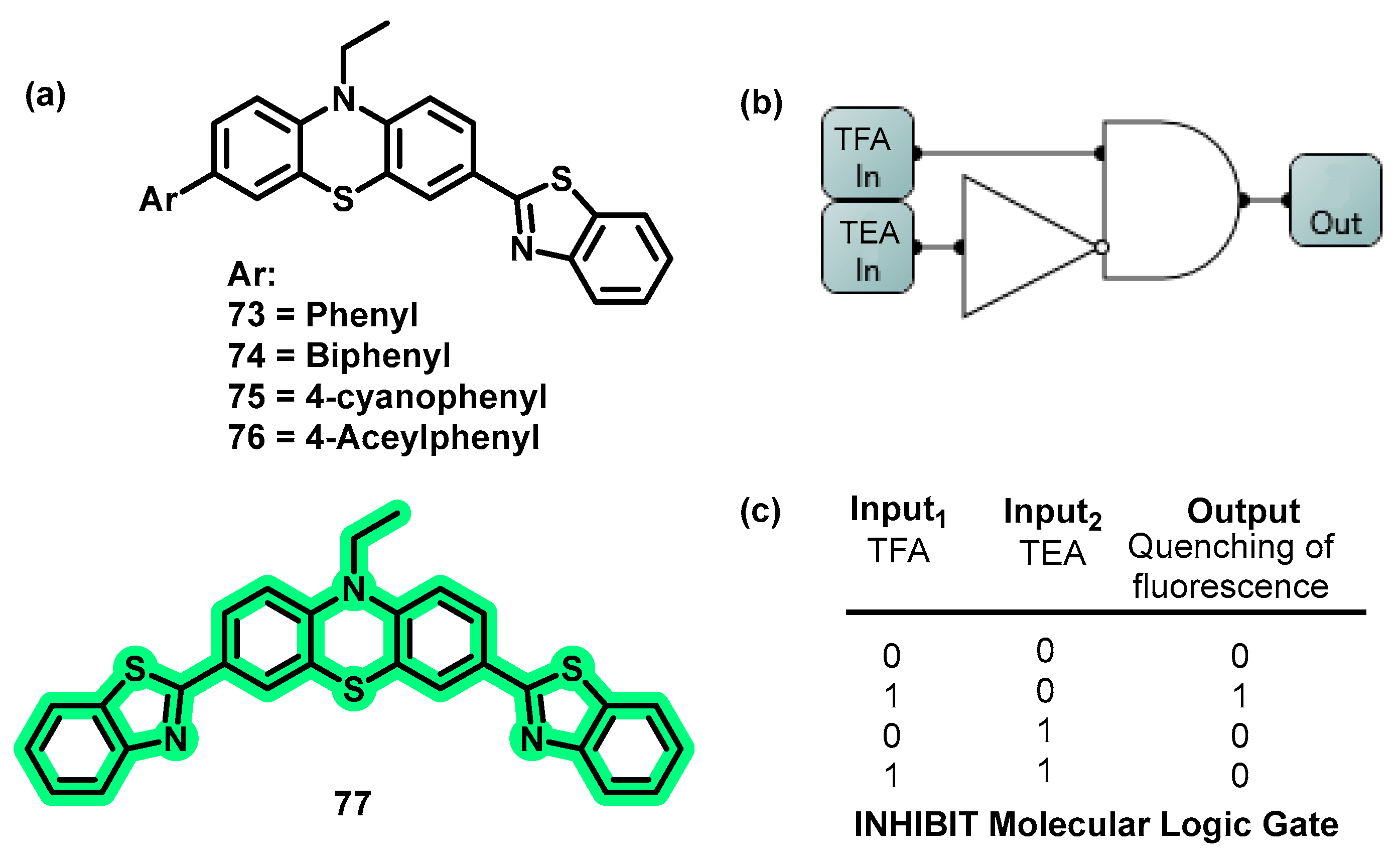
4.7. MLGs Based on Neutral Molecules as Input2
5. Strategies for MLGs Based on Solvent Effects
Acid-Based Effect in Reversibility
6. Advantages, Limitations, and Challenges of MLGs
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| AcO− | Acetate |
| ADP | Adenosine Diphosphate |
| Arg | Arginine |
| ASSURED | Affordable, Sensitive, Specific, User-friendly, Rapid/Robust, Equipment free, and Deliverable |
| A–D–A | Acceptor–Donor–Acceptor |
| ATP | Adenosine Triphosphate |
| CHEF | Chelation Enhanced Fluorescence |
| cit3− | Citrate |
| Cys | Cysteine |
| D–A | Donor–Acceptor |
| D–A–D | Donor–Acceptor–Donor |
| DFT | Density Functional Theory |
| DMSO | Dimethyl Sulfoxide |
| DNA | Deoxyribonucleic Acid |
| EDTA | Ethylenediaminetetraacetic Acid |
| EPR | Electron Spin Resonance |
| ESIPT | Excited State Internal Proton Transfer |
| EtOH | Ethanol |
| FTIR | Fourier-Transform Infrared Spectroscopy |
| GSH | Glutathione |
| HCy | Homocysteine |
| His | Histidine |
| ICT | Internal (or Intramolecular) Charge Transfer |
| INH | INHIBIT Molecular Logic Gates |
| Ka | Binding Constant |
| LG | Logic Gate |
| LOD | Limit of detection |
| LOQ | Limit of Quantification |
| Lys | Lysine |
| MeOH | Methanol |
| MLCT | Ligand Charge Transfer |
| MLG | Molecular Logic Gates |
| MS | Mass Spectra |
| NMR | Nuclear Magnetic Resonance |
| nPrOH | Propan-1-ol |
| PET | Photoinduced Electron Transfer |
| PA | Picric Acid |
| Ppb | Part Per Billion |
| PPi | Pyrophosphate |
| Ppm | Part Per Million |
| QY | Quantum Yield |
| RNA | Ribonucleic acid |
| TEA | Triethylamine |
| TFA | Trifluoroacetic Acid |
| THF | Tetrahydrofuran |
| Trp | Tryptophan |
| λabs | Maximum absorption wavelength |
| λem | Maximum emission wavelength |
| λmax | Maximum wavelength |
References
- Nepovimova, E.; Kuca, K. The History of Poisoning: From Ancient Times until Modern ERA. Arch. Toxicol. 2019, 93, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Michaleas, S.N.; Laios, K.; Tsoucalas, G.; Androutsos, G. Theophrastus Bombastus Von Hohenheim (Paracelsus) (1493–1541): The Eminent Physician and Pioneer of Toxicology. Toxicol. Rep. 2021, 8, 411–414. [Google Scholar] [CrossRef] [PubMed]
- Valeur, B. Analytical Atomic Spectrometry with Flames and Plasmas Handbook of Analytical Techniques Single-Molecule Detection in Solution. In Methods and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2001; Volume 8, ISBN 352729919X. [Google Scholar]
- Stephan, H.; Kubeil, M.; Gloe, K.; Gloe, K. Extraction Methods. In Analytical Methods in Supramolecular Chemistry, Volume 1 & 2, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2012; Volume 1, pp. 105–127. [Google Scholar] [CrossRef]
- Westbroek, P. Electrochemical Methods. In Analytical Electrochemistry in Textiles; Elsevier: Amsterdam, The Netherlands, 2005; pp. 37–69. [Google Scholar] [CrossRef]
- Schalley, C. Analytical Methods in Supramolecular Chemistry; John Wiley & Sons: Hoboken, NJ, USA, 2007; ISBN 9783527315055. [Google Scholar]
- Umapathi, R.; Park, B.; Sonwal, S.; Rani, G.M.; Cho, Y.; Huh, Y.S. Advances in Optical-Sensing Strategies for the on-Site Detection of Pesticides in Agricultural Foods. Trends Food Sci. Technol. 2022, 119, 69–89. [Google Scholar] [CrossRef]
- Jiang, N.; Tansukawat, N.D.; Gonzalez-Macia, L.; Ates, H.C.; Dincer, C.; Güder, F.; Tasoglu, S.; Yetisen, A.K. Low-Cost Optical Assays for Point-of-Care Diagnosis in Resource-Limited Settings. ACS Sens. 2021, 6, 2108–2124. [Google Scholar] [CrossRef]
- Kim, H.N.; Ren, W.X.; Kim, J.S.; Yoon, J. Fluorescent and Colorimetric Sensors for Detection of Lead, Cadmium, and Mercury Ions. Chem. Soc. Rev. 2012, 41, 3210–3244. [Google Scholar] [CrossRef]
- Goyer, R.A.; Cherian, M.G. Toxicology of Metals: Biochemical Aspects; Springer Science & Business Media: Berlin, Germany, 1995; Volume 115, ISBN 9783642791642. [Google Scholar]
- Clevenger, W.L.; Smith, B.W.; Winefordner, J.D. Trace Determination of Mercury: A Review. Crit. Rev. Anal. Chem. 1997, 27, 1–26. [Google Scholar] [CrossRef]
- Shuai, H.; Xiang, C.; Qian, L.; Bin, F.; Xiaohui, L.; Jipeng, D.; Chang, Z.; Jiahui, L.; Wenbin, Z. Fluorescent Sensors for Detection of Mercury: From Small Molecules to Nanoprobes. Dye. Pigment. 2021, 187, 109125. [Google Scholar] [CrossRef]
- Chen, G.; Guo, Z.; Zeng, G.; Tang, L. Fluorescent and Colorimetric Sensors for Environmental Mercury Detection. Analyst 2015, 140, 5400–5443. [Google Scholar] [CrossRef]
- Kallithrakas-kontos, N.; Foteinis, S. Recent Advances in the Analysis of Mercury in Water-Review. Curr. Anal. Chem. 2016, 12, 22–36. [Google Scholar] [CrossRef]
- Bernhoft, R.A. Mercury Toxicity and Treatment: A Review of the Literature. J. Environ. Public Health 2012, 2012, 460508. [Google Scholar] [CrossRef]
- Tchounwou, P.B.; Ayensu, W.K.; Ninashvili, N.; Sutton, D. Environmental Exposure to Mercury and Its Toxicopathologic Implications for Public Health. Environ. Toxicol. 2003, 18, 149–175. [Google Scholar] [CrossRef] [PubMed]
- Micaroni, R.C.d.C.M.; Bueno, M.I.M.S.; Jardim, W.d.F. Compostos de Mercúrio. Revisão de Métodos de Determinação, Tratamento e Descarte. Quim. Nova 2000, 23, 487–495. [Google Scholar] [CrossRef]
- Gidlow, D.A. Lead Toxicity. Occup. Med. (Chic. Ill) 2015, 65, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Godwin, H.A. The Biological Chemistry of Lead. Curr. Opin. Chem. Biol. 2001, 5, 223–227. [Google Scholar] [CrossRef]
- Hildebrand, M.P. Lead Toxicity in a Newborn. J. Pediatr. Health Care 2011, 25, 328–331. [Google Scholar] [CrossRef]
- Mason, A.; Sheridan, A.; Sutherland, I.O.; Vincent, A. Selective Chromogenic Reagents for Lead. J. Chem. Soc. Chem. Commun. 1994, 22, 2627–2628. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Gobe, G.C. Current Health Risk Assessment Practice for Dietary Cadmium: Data from Different Countries. Food Chem. Toxicol. 2017, 106, 430–445. [Google Scholar] [CrossRef]
- Horiguchi, H.; Oguma, E.; Sasaki, S.; Miyamoto, K.; Ikeda, Y.; MacHida, M.; Kayama, F. Dietary Exposure to Cadmium at Close to the Current Provisional Tolerable Weekly Intake Does Not Affect Renal Function among Female Japanese Farmers. Environ. Res. 2004, 95, 20–31. [Google Scholar] [CrossRef]
- Satarug, S.; Moore, M.R. Adverse Health Effects of Chronic Exposure to Low-Level Cadmium in Foodstuffs and Cigarette Smoke. Environ. Health Perspect. 2004, 112, 1099–1103. [Google Scholar] [CrossRef]
- Yokel, R.A.; Hicks, C.L.; Florence, R.L. Aluminum Bioavailability from Basic Sodium Aluminum Phosphate, an Approved Food Additive Emulsifying Agent, Incorporated in Cheese. Food Chem. Toxicol. 2008, 46, 2261–2266. [Google Scholar] [CrossRef]
- Greger, J.L.; Sutherland, J.E. Aluminum Exposure and Metabolism. Crit. Rev. Clin. Lab. Sci. 1997, 34, 439–474. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wan, X.; Li, Y. Advanced Aluminium Products and Manufacturing Technologies Applied on Vehicles Presented at the EuroCarBody Conference. In Proceedings of the Materials Today: Proceedings; Elsevier Ltd.: Amsterdam, The Netherlands, 2015; Volume 2, pp. 5015–5022. [Google Scholar]
- Darbre, P.D. Aluminium, Antiperspirants and Breast Cancer. J. Inorg. Biochem. 2005, 99, 1912–1919. [Google Scholar] [CrossRef] [PubMed]
- Campbell, A. The Potential Role of Aluminium in Alzheimer’s Disease. Nephrol. Dial. Transplant. 2002, 17, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Leal, M.F.C.; Catarino, R.I.L.; Pimenta, A.M.; Souto, M.R.S. Roles of Metal Microelements in Neurodegenerative Diseases. Neurophysiology 2020, 52, 80–88. [Google Scholar] [CrossRef]
- Sahu, M.; Manna, A.K.; Chowdhury, S.; Patra, G.K. A Novel Dihydro Phenylquinazolinone-Based Two-in-One Colourimetric Chemosensor for Nickel(II), Copper(II) and Its Copper Complex for the Fluorescent Colourimetric Nanomolar Detection of the Cyanide Anion. RSC Adv. 2020, 10, 44860–44875. [Google Scholar] [CrossRef]
- Ejaz, H.W.; Wang, W.; Lang, M. Copper Toxicity Links to Pathogenesis of Alzheimer’s Disease and Therapeutics Approaches. Int. J. Mol. Sci. 2020, 21, 7660. [Google Scholar] [CrossRef]
- Trevors, J.T.; Cotter, C.M. Copper Toxicity and Uptake in Microorganisms. J. Ind. Microbiol. 1990, 6, 77–84. [Google Scholar] [CrossRef]
- Chopra, T.; Sasan, S.; Devi, L.; Parkesh, R.; Kapoor, K.K. A Comprehensive Review on Recent Advances in Copper Sensors. Coord. Chem. Rev. 2022, 470, 214704. [Google Scholar] [CrossRef]
- Hessels, A.M.; Merkx, M. Genetically-Encoded FRET-Based Sensors for Monitoring Zn2+ in Living Cells. Metallomics 2015, 7, 258–266. [Google Scholar] [CrossRef]
- Andrews, G.K. Cellular Zinc Sensors: MTF-1 Regulation of Gene Expression. BioMetals 2001, 14, 223–237. [Google Scholar] [CrossRef]
- Truong-Tran, A.Q.; Carter, J.; Ruffin, R.E.; Zalewski, P.D. The Role of Zinc in Caspase Activation and Apoptotic Cell Death. BioMetals 2001, 14, 315–330. [Google Scholar] [CrossRef] [PubMed]
- Bush, A.I. The Metallobiology of Alzheimer’s Disease. Trends Neurosci. 2003, 26, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Chausmer, A.B. Zinc, Insulin and Diabetes. J. Am. Coll. Nutr. 1998, 17, 109–115. [Google Scholar] [CrossRef]
- Wessling-Resnick, M. Biochemistry of Iron Uptake. Crit. Rev. Biochem. Mol. Biol. 1999, 34, 285–314. [Google Scholar] [CrossRef]
- Bordini, J.; Calandreli, I.; Silva, G.O.; Ferreira, K.Q.; Leitão-Mazzi, D.P.S.; Espreafico, E.M.; Tfouni, E. A Rhodamine-B-Based Turn-on Fluorescent Sensor for Biological Iron(III). Inorg. Chem. Commun. 2013, 35, 255–259. [Google Scholar] [CrossRef]
- Yu, Y.; Bogliotti, N.; Maisonneuve, S.; Tang, J.; Xie, J. Fluorescent Dyad for Cooperative Recognition of Copper Cation and Halogen Anion. Tetrahedron Lett. 2013, 54, 1877–1883. [Google Scholar] [CrossRef]
- Padan, E.; Landau, M. Sodium-Proton (Na+/H+) Antiporters: Properties and Roles in Health and Disease. In Metal Ions in Life Sciences; Sigel, A., Sigel, H., Sigel, R.K.O., Eds.; Springer International Publishing AG: Cham, Switzerland, 2016; Volume 16, pp. 391–458. ISBN 1559-0836. [Google Scholar]
- Pohl, H.R.; Wheeler, J.S.; Murray, H.E. Sodium and Potassium in Health and Disease. Met. Ions Life Sci. 2013, 13, 29–47. [Google Scholar] [CrossRef]
- Crossley, R.; Goolamali, Z.; Gosper, J.J.; Sammes, P.G. Synthesis and Spectral Properties of New Fluorescent Probes for Potassium. Chem. Soc. Perkin Trans. 1994, 2, 513–520. [Google Scholar] [CrossRef]
- Gao, G.; Cao, Y.; Liu, W.; Li, D.; Zhou, W.; Liu, J. Fluorescent Sensors for Sodium Ions. Anal. Methods 2017, 9, 5570–5579. [Google Scholar] [CrossRef]
- Jaitovich, A.; Bertorello, A.M. Intracellular Sodium Sensing: SIK1 Network, Hormone Action and High Blood Pressure. Biochim. Biophys. Acta 2010, 1802, 1140–1149. [Google Scholar] [CrossRef]
- Liu, M.; Yu, X.; Li, M.; Liao, N.; Bi, A.; Jiang, Y.; Liu, S.; Gong, Z.; Zeng, W. Fluorescent Probes for the Detection of Magnesium Ions (Mg2+): From Design to Application. RSC Adv. 2018, 8, 12573–12587. [Google Scholar] [CrossRef] [PubMed]
- Lazarou, T.S.; Buccella, D. Advances in Imaging of Understudied Ions in Signaling: A Focus on Magnesium. Curr. Opin. Chem. Biol. 2020, 57, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Wolf, F.I.; Cittadini, A. Chemistry and Biochemistry of Magnesium. Mol. Asp. Med. 2003, 24, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Way, J.L. Cyanide Intoxication and Its Mechanism of Antagonism. Annu. Rev. Pharmacol. Toxicol. 1984, 24, 451–481. [Google Scholar] [CrossRef]
- Simeonova, F.P.; Fishbein, L.; World HealthOrganization & International Programme on Chemical Safety; Inter-Organization Programme for the Sound Management of Chemicals; World Health Organization. Hydrogen Cyanide and Cyanides: Human Health Aspects; World Health Organization: Geneva, Switzerland, 2004; ISBN 9241530618. [Google Scholar]
- Ballantyne, B. Acute Systemic Toxicity of Cyanides by Topical Application to the Eye. Cutan. Ocul. Toxicol. 1983, 2, 119–129. [Google Scholar] [CrossRef]
- Xu, Z.; Chen, X.; Kim, H.N.; Yoon, J. Sensors for the Optical Detection of Cyanide Ion. Chem. Soc. Rev. 2010, 39, 127–137. [Google Scholar] [CrossRef]
- Chakraborty, S.; Paul, S.; Roy, P.; Rayalu, S. Detection of Cyanide Ion by Chemosensing and Fluorosensing Technology. Inorg. Chem. Commun. 2021, 128, 108562. [Google Scholar] [CrossRef]
- Gai, L.; Mack, J.; Lu, H.; Nyokong, T.; Li, Z.; Kobayashi, N.; Shen, Z. Organosilicon Compounds as Fluorescent Chemosensors for Fluoride Anion Recognition. Coord. Chem. Rev. 2015, 285, 24–51. [Google Scholar] [CrossRef]
- Udhayakumari, D. Detection of Toxic Fluoride Ion via Chromogenic and Fluorogenic Sensing. A Comprehensive Review of the Year 2015–2019. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2020, 228, 117817. [Google Scholar] [CrossRef]
- Cheng, J.; Zhou, X.; Xiang, H. Fluorescent Metal Ion Chemosensors via Cation Exchange Reactions of Complexes, Quantum Dots, and Metal-Organic Frameworks. Analyst 2015, 140, 7082–7115. [Google Scholar] [CrossRef]
- Zhang, Y.M.; Lin, Q.; Wei, T.B.; Wang, D.D.; Yao, H.; Wang, Y.L. Simple Colorimetric Sensors with High Selectivity for Acetate and Chloride in Aqueous Solution. Sens. Actuators B Chem. 2009, 137, 447–455. [Google Scholar] [CrossRef]
- Ho, T.Y.; Scranton, M.I.; Taylor, G.T.; Varela, R.; Thunell, R.C.; Muller-Karger, F. Acetate Cycling in the Water Column of the Cariaco Basin: Seasonal and Vertical Variability and Implication for Carbon Cycling. Limnol. Oceanogr. 2002, 47, 1119–1128. [Google Scholar] [CrossRef]
- Gupta, V.K.; Singh, A.K.; Gupta, N. Colorimetric Sensor for Cyanide and Acetate Ion Using Novel Biologically Active Hydrazones. Sens. Actuators B Chem. 2014, 204, 125–135. [Google Scholar] [CrossRef]
- Manju, S.; Jose, L.; Gopal, T.K.S.; Ravishankar, C.N.; Lalitha, K.V. Effects of Sodium Acetate Dip Treatment and Vacuum-Packaging on Chemical, Microbiological, Textural and Sensory Changes of Pearlspot (Etroplus Suratensis) during Chill Storage. Food Chem. 2007, 102, 27–35. [Google Scholar] [CrossRef]
- Ghosh, K.; Ranjan Sarkar, A. Pyridinium-Based Symmetrical Diamides as Chemosensors in Visual Sensing of Citrate through Indicator Displacement Assay (IDA) and Gel Formation. Org. Biomol. Chem. 2011, 9, 6551–6558. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Fu, J.; Yao, K.; Li, B.; Xu, K.; Pang, X. Novel Fluorescent Probes for Sequential Detection of Cu2+ and Citrate Anion and Application in Living Cell Imaging. Dye. Pigment. 2019, 161, 331–340. [Google Scholar] [CrossRef]
- Takeda, E.; Taketani, Y.; Sawada, N.; Sato, T.; Yamamoto, H. The Regulation and Function of Phosphate in the Human Body. BioFactors 2004, 21, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Hruska, K.A.; Mathew, S.; Lund, R.; Qiu, P.; Pratt, R. Hyperphosphatemia of Chronic Kidney Disease. Kidney Int. 2008, 74, 148–157. [Google Scholar] [CrossRef]
- Kuhlmann, M.K. Management of Hyperphosphatemia. Hemodial. Int. 2006, 10, 338–345. [Google Scholar] [CrossRef]
- Zhou, C.; Shi, Z.; Ouyang, N.; Ruan, X. Hyperphosphatemia and Cardiovascular Disease. Front. Cell Dev. Biol. 2021, 9, 644363. [Google Scholar] [CrossRef]
- Koumakis, E.; Cormier, C.; Roux, C.; Briot, K. The Causes of Hypo- and Hyperphosphatemia in Humans. Calcif. Tissue Int. 2021, 108, 41–73. [Google Scholar] [CrossRef]
- Orriss, I.R.; Arnett, T.R.; Russell, R.G.G. Pyrophosphate: A Key Inhibitor of Mineralisation. Curr. Opin. Pharmacol. 2016, 28, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, A.K.; Ryan, L.M. Calcium Pyrophosphate Deposition Disease. N. Engl. J. Med. 2016, 374, 2575–2584. [Google Scholar] [CrossRef] [PubMed]
- Lohmann, K. The Pyrophosphate Fraction in Muscle. In Source Book in Chemistry, 1900–1950; Harvard University Press: Cambridge, MA, USA, 1968; pp. 367–369. ISBN 9780674366701. [Google Scholar]
- Kim, S.K.; Lee, D.H.; Hong, J.-I.; Yoon, J. Chemosensors for Pyrophosphate. Acc. Chem. Res. 2009, 42, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Mutihac, L.; Lee, J.H.; Kim, J.S.; Vicens, J. Chromogenic and Fluorogenic Chemosensors and Reagents for Anions. A Comprehensive Review of the Year 2009. Chem. Soc. Rev. 2011, 40, 2593–2643. [Google Scholar] [CrossRef]
- Figueroa, L.E.S.; Moragues, M.E.; Climent, E.; Agostini, A.; Martínez-Máñez, R.; Sancenón, F. Chromogenic and Fluorogenic Chemosensors and Reagents for Anions. A Comprehensive Review of the Years 2010–2011. Chem. Soc. Rev. 2013, 42, 3489–3613. [Google Scholar] [CrossRef]
- Schmidtchen, F.P.; Berger, M. Artificial Organic Host Molecules for Anions. Chem. Rev. 1997, 97, 1609–1646. [Google Scholar] [CrossRef]
- Mohr, G.J. New Chromogenic and Fluorogenic Reagents and Sensors for Neutral and Ionic Analytes Based on Covalent Bond Formation-a Review of Recent Developments. Anal. Bioanal. Chem. 2006, 386, 1201–1214. [Google Scholar] [CrossRef]
- Gräfe, A.; Haupt, K.; Mohr, G.J. Optical Sensor Materials for the Detection of Amines in Organic Solvents. Anal. Chim. Acta 2006, 565, 42–47. [Google Scholar] [CrossRef]
- Kumar, V.; Kim, H.; Pandey, B.; James, T.D.; Yoon, J.; Anslyn, E.V. Recent Advances in Fluorescent and Colorimetric Chemosensors for the Detection of Chemical Warfare Agents: A Legacy of the 21st Century. Chem. Soc. Rev. 2023, 52, 663–704. [Google Scholar] [CrossRef]
- Meng, W.-Q.; Sedgwick, A.C.; Kwon, N.; Sun, M.; Xiao, K.; He, X.-P.; Anslyn, E.V.; James, T.D.; Yoon, J. Fluorescent Probes for the Detection of Chemical Warfare Agents. Chem. Soc. Rev. 2023, 52, 601–662. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Li, Z.; Lu, Z.; Chang, R.; Sun, Z.; You, J. Recent Progress in the Development of Chemodosimeters for Fluorescence Visualization of Phosgene. Dye. Pigm. 2021, 193, 109540. [Google Scholar] [CrossRef]
- Kumar, N.M.; Picchetti, P.; Hu, C.; Grimm, L.M.; Biedermann, F. Chemiluminescent Cucurbit[n]Uril-Based Chemosensor for the Detection of Drugs in Biofluids. ACS Sens. 2022, 7, 2312–2319. [Google Scholar] [CrossRef]
- Yan, J.; Lee, S.; Zhang, A.; Yoon, J. Self-Immolative Colorimetric, Fluorescent and Chemiluminescent Chemosensors. Chem. Soc. Rev. 2018, 47, 6900–6916. [Google Scholar] [CrossRef] [PubMed]
- Abu-Taweel, G.M.; Ibrahim, M.M.; Khan, S.; Al-Saidi, H.M.; Alshamrani, M.; Alhumaydhi, F.A.; Alharthi, S.S. Medicinal Importance and Chemosensing Applications of Pyridine Derivatives: A Review. Crit. Rev. Anal. Chem. 2022, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, F.; Minhaz, A.; Shah, M.R.; Ain, N.; Khan, A.A.; Shah, K.; Ullah, S.; Ishaq, M. Highly Selective and Sensitive Chemosensor for Detection of Pefloxacin in Tap Water Based on Click Generated Triazole. Microchem. J. 2019, 146, 332–338. [Google Scholar] [CrossRef]
- Rajendran, S.; Vipinachandran, V.U.; Haroon, K.H.B.; Ashokan, I.; Bhunia, S.K. A Comprehensive Review on Multi-Colored Emissive Carbon Dots as Fluorescent Probes for the Detection of Pharmaceutical Drugs in Water. Anal. Methods 2022, 14, 4263–4291. [Google Scholar] [CrossRef]
- Yaqoob, S.; Rahim, S.; Bhayo, A.M.; Shah, M.R.; Hameed, A.; Malik, M.I. A Novel and Efficient Colorimetric Assay for Quantitative Determination of Amlodipine in Environmental, Biological and Pharmaceutical Samples. ChemistrySelect 2019, 4, 10046–10053. [Google Scholar] [CrossRef]
- Kumar, A.; Chae, P.S. Fluorescence Tunable Thiophene-Bis(Benzimidazole)-Based Probes for a Cascade Trace Detection of Hg2+ and Lysine: A Molecular Switch Mimic. Sens. Actuators B Chem. 2019, 281, 933–944. [Google Scholar] [CrossRef]
- Tavallali, H.; Espergham, O.; Deilamy-Rad, G.; Karimi, M.A.; Rostami, S.; Rouhani-Savestani, A.-R. Dye/Metal Ion-Based Chemosensing Ensemble towards L-Histidine and L-Lysine Determination in Water via Different Optical Responses. Anal. Biochem. 2020, 604, 113811. [Google Scholar] [CrossRef]
- Guo-Qiang, Z.; Gong-Xuan, L. A NOR Fluorescent Logic Gate Based on N-(9-Anthracylmethyl)-L-Histidine. Acta Phys.-Chim. Sin. 2008, 24, 1902–1906. [Google Scholar] [CrossRef]
- Gu, Z.; Cao, Z. Molecular Switch-Modulated Fluorescent Copper Nanoclusters for Selective and Sensitive Detection of Histidine and Cysteine. Anal. Bioanal. Chem. 2018, 410, 4991–4999. [Google Scholar] [CrossRef]
- Coucouvanis, D.; Rosa, D.; Pike, J. Recognition and Transport of Amphiphilic Molecules by a New Class of Inorganic Ditopic Receptors. The Synthesis of M-tBu4-Salphen-3n-Cr-n Complexes and Their Use (M = Mn,Fe, n = 6) in the Transport of Tryptophan and Serotonin across Bulk Liquid Membranes. Comptes Rendus Chim. 2003, 6, 317–327. [Google Scholar] [CrossRef]
- Lipton, S.A.; Choi, Y.-B.; Takahashi, H.; Zhang, D.; Li, W.; Godzik, A.; Bankston, L.A. Cysteine Regulation of Protein Function-as Exemplified by NMDA-Receptor Modulation. Trends Neurosci. 2002, 25, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Zhang, J.; Qu, W.; Zhong, X.; Liu, H.; Ren, J.; He, H.; Zhang, X.; Wang, S. Development of a Novel Benzothiadiazole-Based Fluorescent Turn-on Probe for Highly Selective Detection of Glutathione over Cysteine/Homocysteine. Sens. Actuators B Chem. 2018, 266, 528–533. [Google Scholar] [CrossRef]
- Chen, X.; Zhou, Y.; Peng, X.; Yoon, J. Fluorescent and Colorimetric Probes for Detection of Thiols. Chem. Soc. Rev. 2010, 39, 2120–2135. [Google Scholar] [CrossRef]
- Misra, R.; Bhattacharyya, S.P. Intramolecular Charge Transfer: Theory and Applications, 1st ed.; Wiley: Hoboken, NJ, USA, 2018; ISBN 978-3-527-34156-6. [Google Scholar]
- Ghosh, S.; Singharoy, D.; Naskar, J.P.; Bhattacharya, S.C. Deciphering of Ligand-to-Metal Charge-Transfer Process: Synthesis, Spectroscopic and Theoretical Study. ChemistrySelect 2019, 4, 458–464. [Google Scholar] [CrossRef]
- Hanaoka, K.; Iwaki, S.; Yagi, K.; Myochin, T.; Ikeno, T.; Ohno, H.; Sasaki, E.; Komatsu, T.; Ueno, T.; Uchigashima, M.; et al. General Design Strategy to Precisely Control the Emission of Fluorophores via a Twisted Intramolecular Charge Transfer (TICT) Process. J. Am. Chem. Soc. 2022, 144, 19778–19790. [Google Scholar] [CrossRef]
- Wang, C.; Chi, W.; Qiao, Q.; Tan, D.; Xu, Z.; Liu, X. Twisted Intramolecular Charge Transfer (TICT) and Twists beyond TICT: From Mechanisms to Rational Designs of Bright and Sensitive Fluorophores. Chem. Soc. Rev. 2021, 50, 12656–12678. [Google Scholar] [CrossRef]
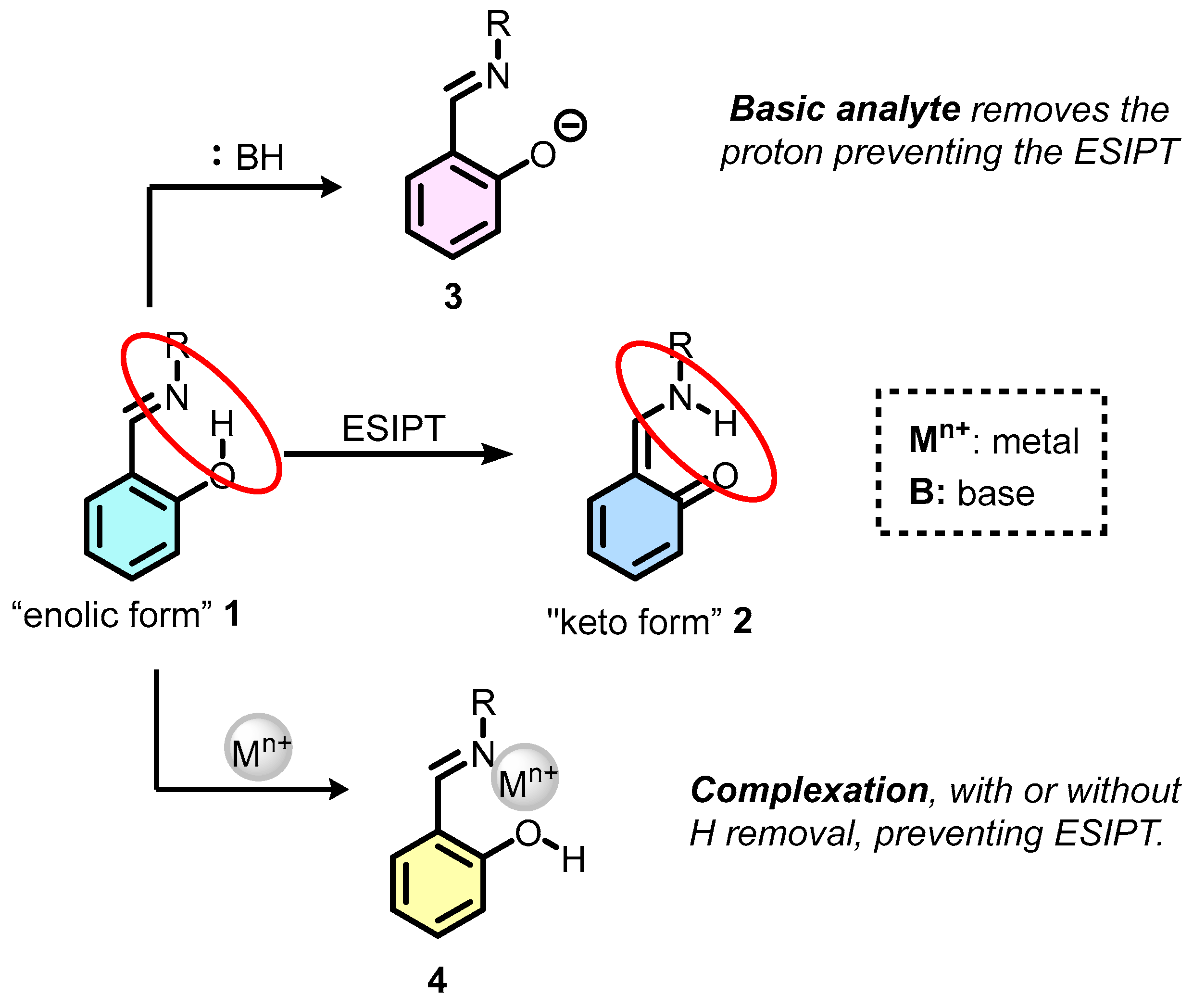
- Sedgwick, A.C.; Wu, L.; Han, H.-H.; Bull, S.D.; He, X.-P.; James, T.D.; Sessler, J.L.; Tang, B.Z.; Tian, H.; Yoon, J. Excited-State Intramolecular Proton-Transfer (ESIPT) Based Fluorescence Sensors and Imaging Agents. Chem. Soc. Rev. 2018, 47, 8842–8880. [Google Scholar] [CrossRef]
- Liu, Z.; He, W.; Guo, Z. Metal Coordination in Photoluminescent Sensing. Chem. Soc. Rev. 2013, 42, 1568–1600. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.E.; Park, S.Y. Advanced Organic Optoelectronic Materials: Harnessing Excited-State Intramolecular Proton Transfer (ESIPT) Process. Adv. Mater. 2011, 23, 3615–3642. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Han, K. ESIPT-Based AIE Luminogens: Design Strategies, Applications, and Mechanisms. Aggregate 2022, 3, e160. [Google Scholar] [CrossRef]
- Jung, H.S.; Verwilst, P.; Kim, W.Y.; Kim, J.S. Fluorescent and Colorimetric Sensors for the Detection of Humidity or Water Content. Chem. Soc. Rev. 2016, 45, 1242–1256. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, E.; Lam, J.W.Y.; Tang, B.Z. AIE Luminogens: Emission Brightened by Aggregation. Mater. Today 2015, 18, 365–377. [Google Scholar] [CrossRef]
- Mei, J.; Leung, N.L.C.; Kwok, R.T.K.; Lam, J.W.Y.; Tang, B.Z. Aggregation-Induced Emission: Together We Shine, United We Soar! Chem. Rev. 2015, 115, 11718–11940. [Google Scholar] [CrossRef]
- Gao, M.; Tang, B.Z. Fluorescent Sensors Based on Aggregation-Induced Emission: Recent Advances and Perspectives. ACS Sens. 2017, 2, 1382–1399. [Google Scholar] [CrossRef]
- Yuan, L.; Lin, W.; Zheng, K.; Zhu, S. FRET-Based Small-Molecule Fluorescent Probes: Rational Design and Bioimaging Applications. Acc. Chem. Res. 2013, 46, 1462–1473. [Google Scholar] [CrossRef]
- Ustimova, M.A.; Lebedeva, A.Y.; Fedorov, Y.V.; Berdnikova, D.V.; Fedorova, O.A. FRET-Based Metal Ion Sensing by a Crown-Containing Bisstyryl Dye. New J. Chem. 2018, 42, 7908–7913. [Google Scholar] [CrossRef]
- Kim, D.; Ryu, H.G.; Ahn, K.H. Recent Development of Two-Photon Fluorescent Probes for Bioimaging. Org. Biomol. Chem. 2014, 12, 4550–4566. [Google Scholar] [CrossRef]
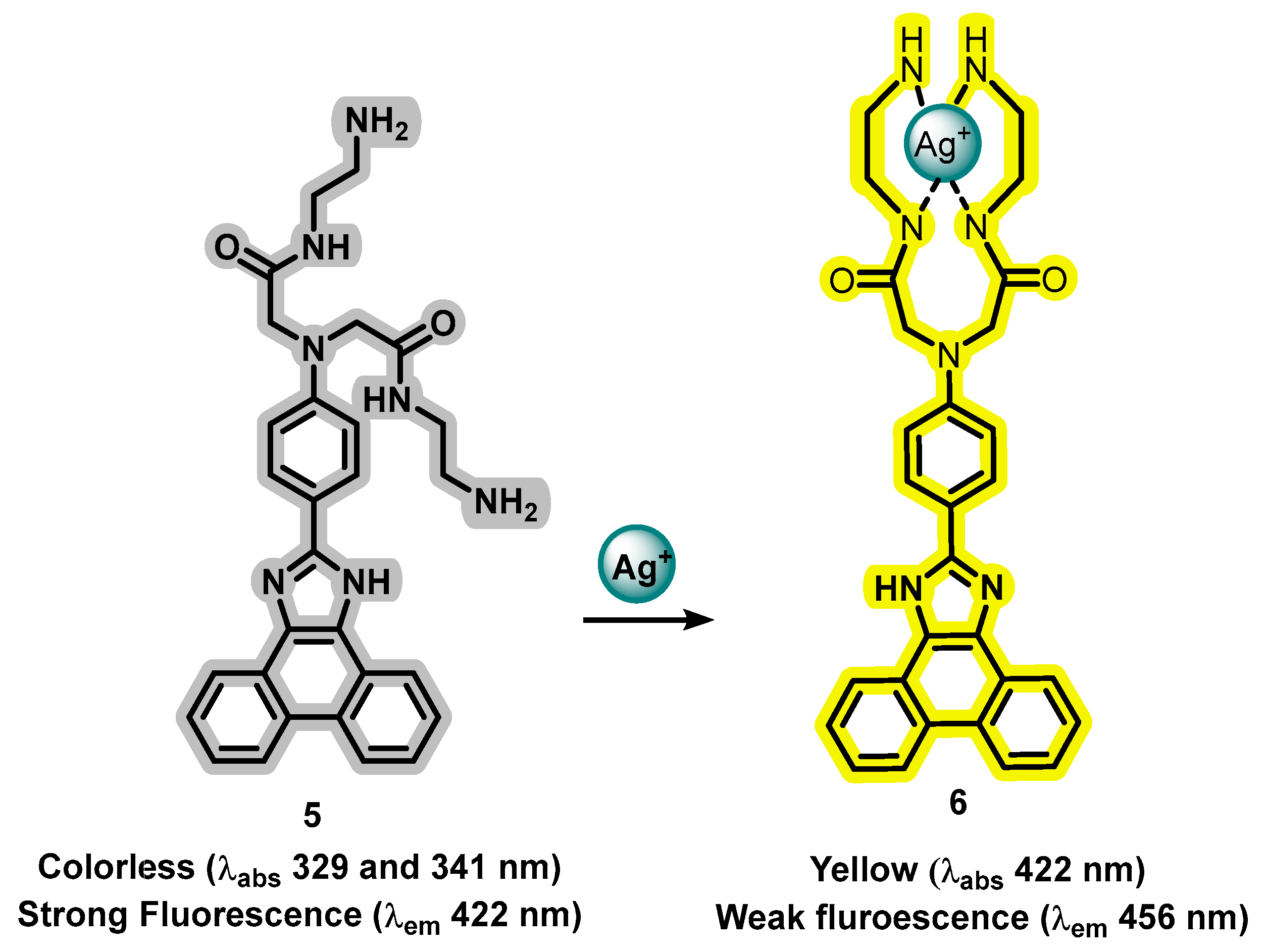
- Zhao, B.; Xu, Y.; Fang, Y.; Wang, L.; Deng, Q. Synthesis and Fluorescence Properties of Phenanthro[9,10-d]Imidazole Derivative for Ag+ in Aqueous Media. Tetrahedron Lett. 2015, 56, 2460–2465. [Google Scholar] [CrossRef]
- Chen, Y.; Shi, X.; Lu, Z.; Wang, X.; Wang, Z. A Fluorescent Probe for Hydrogen Peroxide in Vivo Based on the Modulation of Intramolecular Charge Transfer. Anal. Chem. 2017, 89, 5278–5284. [Google Scholar] [CrossRef]
- Halmos, P.; Givant, S. Introduction to Boolean Algebras, 1st ed.; Springer: New York, NY, USA, 2009; ISBN 978-0-387-40293-2. [Google Scholar]
- Zhao, S.; Yu, L.; Yang, S.; Tang, X.; Chang, K.; Chen, M. Boolean Logic Gate Based on DNA Strand Displacement for Biosensing: Current and Emerging Strategies. Nanoscale Horiz. 2021, 6, 298–310. [Google Scholar] [CrossRef] [PubMed]
- De Silva, A.P.; Gunaratne, N.H.Q.; McCoy, C.P. A Molecular Photoionic and Gate Based on Fluorescent Signalling. Nature 1993, 364, 42–44. [Google Scholar] [CrossRef]
- De Silva, A.P. Molecular Logic Gate Arrays. Chem. Asian J. 2011, 6, 750–766. [Google Scholar] [CrossRef] [PubMed]
- Erbas-Cakmak, S.; Kolemen, S.; Sedgwick, A.C.; Gunnlaugsson, T.; James, T.D.; Yoon, J.; Akkaya, E.U. Molecular Logic Gates: The Past, Present and Future. Chem. Soc. Rev. 2018, 47, 2228–2248. [Google Scholar] [CrossRef]
- Magri, D.C.; Vance, T.P.; de Silva, A.P. From Complexation to Computation: Recent Progress in Molecular Logic. Inorg. Chim. Acta 2007, 360, 751–764. [Google Scholar] [CrossRef]
- Yao, C.-Y.; Lin, H.-Y.; Crory, H.S.N.; de Silva, A.P. Supra-Molecular Agents Running Tasks Intelligently (SMARTI): Recent Developments in Molecular Logic-Based Computation. Mol. Syst. Des. Eng. 2020, 5, 1325–1353. [Google Scholar] [CrossRef]
- Andréasson, J.; Pischel, U. Light-Stimulated Molecular and Supramolecular Systems for Information Processing and Beyond. Coord. Chem. Rev. 2021, 429, 213695. [Google Scholar] [CrossRef]
- Liu, L.; Liu, P.; Ga, L.; Ai, J. Advances in Applications of Molecular Logic Gates. ACS Omega 2021, 6, 30189–30204. [Google Scholar] [CrossRef]
- Liu, L.; Ga, L.; Ai, J. Ratiometric Fluorescence Sensing with Logical Operation: Theory, Design and Applications. Biosens. Bioelectron. 2022, 213, 114456. [Google Scholar] [CrossRef] [PubMed]
- Andréasson, J.; Pischel, U. Molecules for Security Measures: From Keypad Locks to Advanced Communication Protocols. Chem. Soc. Rev. 2018, 47, 2266–2279. [Google Scholar] [CrossRef] [PubMed]
- Georgiev, N.I.; Bakov, V.V.; Bojinov, V.B. A Tutorial Review on the Fluorescent Probes as a Molecular Logic Circuit—Digital Comparator. Molecules 2023, 28, 6327. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Wan, S.; Hou, W.; Zhang, L.; Xu, J.; Cui, C.; Wang, Y.; Hu, J.; Tan, W. A Survey of Advancements in Nucleic Acid-Based Logic Gates and Computing for Applications in Biotechnology and Biomedicine. Chem. Commun. 2015, 51, 3723–3734. [Google Scholar] [CrossRef]
- Zanella, S.; Hernández-Rodríguez, M.A.; Fu, L.; Carlos, L.D.; Ferreira, R.A.S.; Brites, C.D.S. Reprogrammable and Reconfigurable Photonic Molecular Logic Gates Based on Ln3+ Ions. Adv. Opt. Mater. 2022, 10, 2200138. [Google Scholar] [CrossRef]
- Magri, D.C. Logical Sensing with Fluorescent Molecular Logic Gates Based on Photoinduced Electron Transfer. Coord. Chem. Rev. 2021, 426, 213598. [Google Scholar] [CrossRef]
- Zuo, Y.; Gou, Z.; Lan, Y.; Yan, M. Design Strategies of Logic Gate Sensors Based on FRET Mechanism. TrAC Trends Anal. Chem. 2023, 167, 117271. [Google Scholar] [CrossRef]
- Nguyen, B.T.; Anslyn, E.V. Indicator-Displacement Assays. Coord. Chem. Rev. 2006, 250, 3118–3127. [Google Scholar] [CrossRef]
- Sedgwick, A.C.; Brewster, J.T.; Wu, T.; Feng, X.; Bull, S.D.; Qian, X.; Sessler, J.L.; James, T.D.; Anslyn, E.V.; Sun, X. Indicator Displacement Assays (IDAs): The Past, Present and Future. Chem. Soc. Rev. 2021, 50, 9–38. [Google Scholar] [CrossRef]
- Lou, X.; Ou, D.; Li, Q.; Li, Z. An Indirect Approach for Anion Detection: The Displacement Strategy and Its Application. Chem. Commun. 2012, 48, 8462–8477. [Google Scholar] [CrossRef]
- Gülle, S.; Çelik Erbaş, S. A Selective Fluorescence Sensor for Fe (III) Based on Phenanthroimidazole Imine Compound. J. Fluoresc. 2018, 28, 445–451. [Google Scholar] [CrossRef] [PubMed]
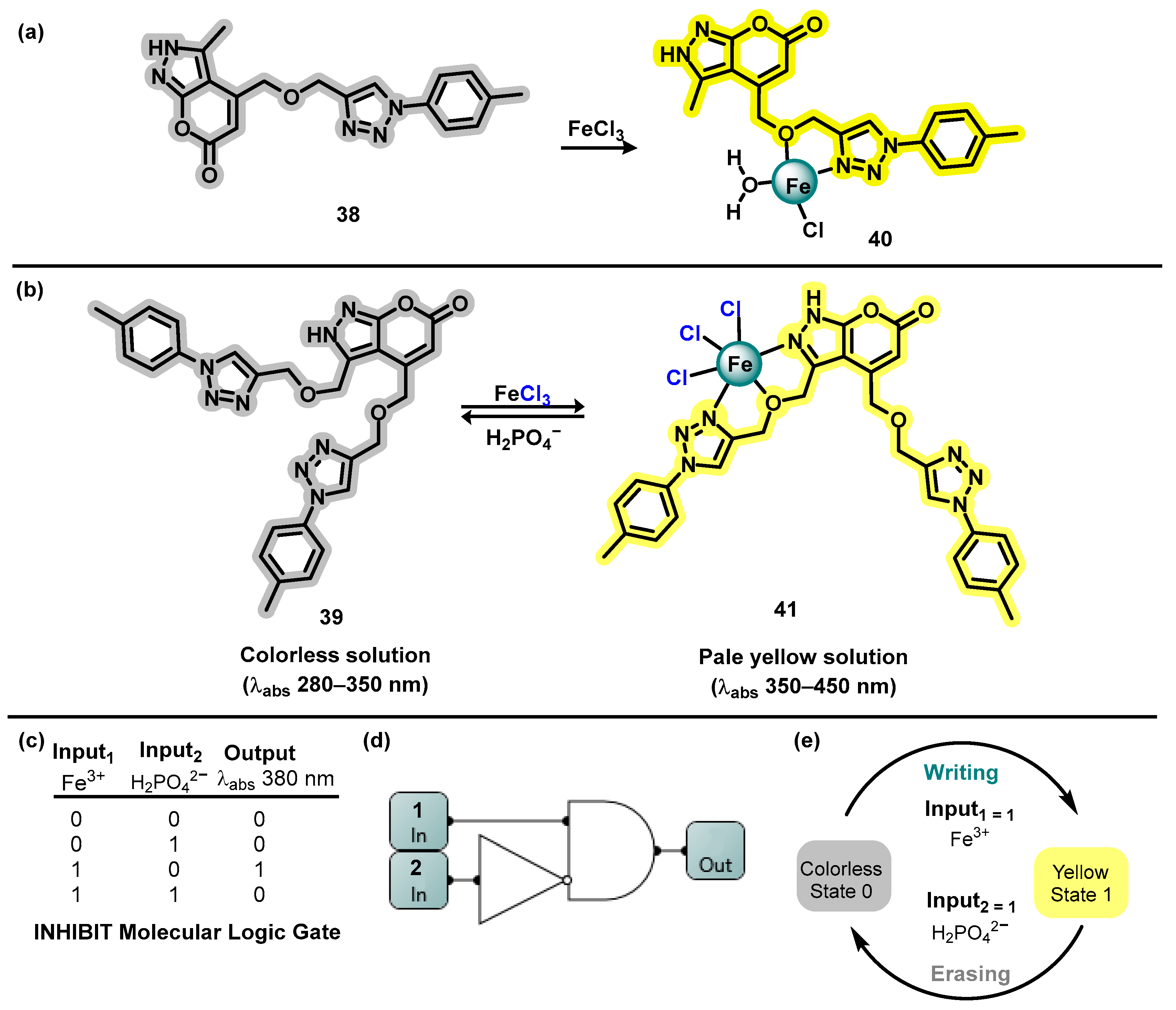
- Dias, G.G.; Rodrigues, M.O.; Paz, E.R.S.; Nunes, M.P.; Araujo, M.H.; Rodembusch, F.S.; Da Silva Júnior, E.N. Aryl-Phenanthro[9,10-d]Imidazole: A Versatile Scaffold for the Design of Optical-Based Sensors. ACS Sens. 2022, 7, 2865–2919. [Google Scholar] [CrossRef] [PubMed]
- Dias, G.G.; Paz, E.R.S.; Nunes, M.P.; Carvalho, R.L.; Rodrigues, M.O.; Rodembusch, F.S.; da Silva Júnior, E.N. Imidazoles and Oxazoles from Lapachones and Phenanthrene-9,10-Dione: A Journey through Their Synthesis, Biological Studies, and Optical Applications. Chem. Rec. 2021, 21, 2702–2738. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.; Liu, X.; Yang, H.; Zhang, T.; Han, A.; Zang, L. A Cu2+-Selective Probe Based on Phenanthro-Imidazole Derivative. Sensors 2017, 17, 35. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, F.S.; Dias, G.G.; de Freitas, R.P.; Santos, L.S.; de Lima, G.F.; Duarte, H.A.; de Simone, C.A.; Rezende, L.M.S.L.; Vianna, M.J.X.; Correa, J.R.; et al. Redox Center Modification of Lapachones towards the Synthesis of Nitrogen Heterocycles as Selective Fluorescent Mitochondrial Imaging Probes. Eur. J. Org. Chem. 2017, 2017, 3763–3773. [Google Scholar] [CrossRef]
- Dias, G.G.; Pinho, P.V.B.; Duarte, H.A.; Resende, J.M.; Rosa, A.B.B.; Correa, J.R.; Neto, B.A.D.; Da Silva Júnior, E.N. Fluorescent Oxazoles from Quinones for Bioimaging Applications. RSC Adv. 2016, 6, 76053–76063. [Google Scholar] [CrossRef]
- Jiao, Y.; Zhou, L.; He, H.; Yin, J.; Gao, Q.; Wei, J.; Duan, C.; Peng, X. A Novel Rhodamine B-Based “off-on’’ Fluorescent Sensor for Selective Recognition of Copper(II) Ions. Talanta 2018, 184, 143–148. [Google Scholar] [CrossRef]
- Majumdar, A.; Lim, C.S.; Kim, H.M.; Ghosh, K. New Six-Membered PH-Insensitive Rhodamine Spirocycle in Selective Sensing of Cu2+ through C-C Bond Cleavage and Its Application in Cell Imaging. ACS Omega 2017, 2, 8167–8176. [Google Scholar] [CrossRef]
- Arumugaperumal, R.; Srinivasadesikan, V.; Lin, M.C.; Shellaiah, M.; Shukla, T.; Lin, H.C. Facile Rhodamine-Based Colorimetric Sensors for Sequential Detections of Cu(II) Ions and Pyrophosphate (P2O74−) Anions. RSC Adv. 2016, 6, 106631–106640. [Google Scholar] [CrossRef]
- Cao, D.; Liu, Z.; Verwilst, P.; Koo, S.; Jangjili, P.; Kim, J.S.; Lin, W. Coumarin-Based Small-Molecule Fluorescent Chemosensors. Chem. Rev. 2019, 119, 10403–10519. [Google Scholar] [CrossRef]
- Devendhiran, T.; Kumarasamy, K.; Lin, M.C.; Yang, Y.X. Synthesis and Physical Studies of Coumarin-Based Chemosensor for Cyanide Ions. Inorg. Chem. Commun. 2021, 134, 108951. [Google Scholar] [CrossRef]
- Sun, X.Y.; Liu, T.; Sun, J.; Wang, X.J. Synthesis and Application of Coumarin Fluorescence Probes. RSC Adv. 2020, 10, 10826–10847. [Google Scholar] [CrossRef] [PubMed]
- Janeková, H.; Gašpar, J.; Gáplovský, A.; Stankovičová, H. Selective Fluoride Chemosensors Based on Coumarin Semicarbazones. J. Photochem. Photobiol. A Chem. 2021, 410, 113168. [Google Scholar] [CrossRef]
- Kaur, P.; Singh, K. Recent Advances in the Application of BODIPY in Bioimaging and Chemosensing. J. Mater. Chem. C Mater. 2019, 7, 11361–11405. [Google Scholar] [CrossRef]
- Wang, L.; Ding, H.; Ran, X.; Tang, H.; Cao, D. Recent Progress on Reaction-Based BODIPY Probes for Anion Detection. Dye. Pigment. 2020, 172, 107857. [Google Scholar] [CrossRef]
- Shi, W.J.; Huang, Y.; Liu, W.; Xu, D.; Chen, S.T.; Liu, F.; Hu, J.; Zheng, L.; Chen, K. A BODIPY-Based “OFF-ON” Fluorescent Probe for Fast and Selective Detection of Hypochlorite in Living Cells. Dye. Pigment. 2019, 170, 107566. [Google Scholar] [CrossRef]
- Rathod, R.V.; Bera, S.; Maity, P.; Mondal, D. Mechanochemical Synthesis of a Fluorescein-Based Sensor for the Selective Detection and Removal of Hg2+ Ions in Industrial Effluents. ACS Omega 2020, 5, 4982–4990. [Google Scholar] [CrossRef]
- Keerthana, S.; Sam, B.; George, L.; Sudhakar, Y.N.; Varghese, A. Fluorescein Based Fluorescence Sensors for the Selective Sensing of Various Analytes. J. Fluoresc. 2021, 31, 1251–1276. [Google Scholar] [CrossRef]
- Mikata, Y.; Nodomi, Y.; Kizu, A.; Konno, H. Quinoline-Attached Triazacyclononane (TACN) Derivatives as Fluorescent Zinc Sensors. Dalton Trans. 2014, 43, 1684–1690. [Google Scholar] [CrossRef]
- Loya, M.; Hazarika, S.I.; Pahari, P.; Atta, A.K. Fluorometric Detection of Cu2+ and Ni2+ by a Quinoline-Based Glucopyranose Derivative via the Excimer of Quinoline Subunit. J. Mol. Struct. 2021, 1241, 130634. [Google Scholar] [CrossRef]
- Ranee, S.J.; Sivaraman, G.; Pushpalatha, A.M.; Muthusubramanian, S. Quinoline Based Sensors for Bivalent Copper Ions in Living Cells. Sens. Actuators B Chem. 2018, 255, 630–637. [Google Scholar] [CrossRef]
- Ruan, Y.B.; Maisonneuve, S.; Li, C.; Tang, J.; Xie, J. Cooperative Recognition of Cu2+ Based on Amino Acids Tethered Benzothiadiazoyl-Bistriazoles. Front. Chem. China 2010, 5, 208–213. [Google Scholar] [CrossRef]
- Moro, A.V.; Ferreira, P.C.; Migowski, P.; Rodembusch, F.S.; Dupont, J.; Lüdtke, D.S. Synthesis and Photophysical Properties of Fluorescent 2,1,3- Benzothiadiazole-Triazole-Linked Glycoconjugates: Selective Chemosensors for Ni(II). Tetrahedron 2013, 69, 201–206. [Google Scholar] [CrossRef]
- Wang, Z.C.; Tan, Y.Z.; Yu, H.; Bao, W.H.; Tang, L.L.; Zeng, F. A Benzothiadiazole-Based Self-Assembled Cage for Cadmium Detection. Molecules 2023, 28, 1841. [Google Scholar] [CrossRef] [PubMed]
- Qiu, C.Q.; Li, L.Q.; Yao, S.L.; Liu, S.J.; Xu, H.; Zheng, T.F. Two Benzothiadiazole-Based Compounds as Multifunctional Fluorescent Sensors for Detection of Organic Amines and Anions. Polyhedron 2021, 199, 115100. [Google Scholar] [CrossRef]
- Attia, G.; Rahali, S.; Teka, S.; Fourati, N.; Zerrouki, C.; Seydou, M.; Chehimi, S.; Hayouni, S.; Mbakidi, J.P.; Bouquillon, S.; et al. Anthracene Based Surface Acoustic Wave Sensors for Picomolar Detection of Lead Ions. Correlation between Experimental Results and DFT Calculations. Sens. Actuators B Chem. 2018, 276, 349–355. [Google Scholar] [CrossRef]
- Guoqiang, Z.; Gongxuan, L. An Anthracene-Based Chemosensor for Multiple Logic Operations at the Molecular Level. J. Phys. Chem. C 2009, 113, 2541–2546. [Google Scholar] [CrossRef]
- Han, D.Y.; Kim, J.M.; Kim, J.; Jung, H.S.; Lee, Y.H.; Zhang, J.F.; Kim, J.S. ESIPT-Based Anthraquinonylcalix[4]Crown Chemosensor for In3+. Tetrahedron Lett. 2010, 51, 1947–1951. [Google Scholar] [CrossRef]
- Verma, M.; Kaur, N.; Singh, N. Naphthalimide-Based DNA-Coupled Hybrid Assembly for Sensing Dipicolinic Acid: A Biomarker for Bacillus Anthracis Spores. Langmuir 2018, 34, 6591–6600. [Google Scholar] [CrossRef]
- Lee, S.H.; Kim, J.Y.; Kim, S.K.; Lee, J.H.; Kim, J.S. Pyrene-Appended Calix[4]Crowned Logic Gates Involving Normal and Reverse PET: NOR, XNOR and INHIBIT. Tetrahedron 2004, 60, 5171–5176. [Google Scholar] [CrossRef]
- Guo, Y.; Wang, L.; Zhuo, J.; Xu, B.; Li, X.; Zhang, J.; Zhang, Z.; Chi, H.; Dong, Y.; Lu, G. A Pyrene-Based Dual Chemosensor for Colorimetric Detection of Cu2+ and Fluorescent Detection of Fe3+. Tetrahedron Lett. 2017, 58, 3951–3956. [Google Scholar] [CrossRef]
- Hangarge, R.V.; La, D.D.; Boguslavsky, M.; Jones, L.A.; Kim, Y.S.; Bhosale, S.V. An Aza-12-Crown-4 Ether-Substituted Naphthalene Diimide Chemosensor for the Detection of Lithium Ion. ChemistrySelect 2017, 2, 11487–11491. [Google Scholar] [CrossRef]
- Ushakov, E.N.; Alfimov, M.V.; Gromov, S.P. Crown Ether-Based Optical Molecular Sensors and Photocontrolled Ionophores. Macroheterocycles 2010, 3, 189–200. [Google Scholar] [CrossRef]
- Hsieh, Y.C.; Chir, J.L.; Wu, H.H.; Chang, P.S.; Wu, A.T. A Sugar-Aza-Crown Ether-Based Fluorescent Sensor for Hg2+ and Cu2+. Carbohydr. Res. 2009, 344, 2236–2239. [Google Scholar] [CrossRef]
- Gokel, G.W.; Leevy, W.M.; Weber, M.E. Crown Ethers: Sensors for Ions and Molecular Scaffolds for Materials and Biological Models. Chem. Rev. 2004, 104, 2723–2750. [Google Scholar] [CrossRef] [PubMed]
- Lamelas, R.; García, V.; Liñares, A.; Bastida, R.; Labisbal, E.; Fernández-Lodeiro, A.; Lodeiro, C.; Núñez, C.; Valencia, L. Novel Trans-Disubstituted Hexaaza-Macrocyclic Ligands Containing Pyridine Head Units: Synthesis, Disubstitution and Colorimetric Properties. Sens. Actuators B Chem. 2016, 225, 481–491. [Google Scholar] [CrossRef]
- Liu, J.B.; Yang, C.; Ko, C.N.; Vellaisamy, K.; Yang, B.; Lee, M.Y.; Leung, C.H.; Ma, D.L. A Long Lifetime Iridium(III) Complex as a Sensitive Luminescent Probe for Bisulfite Detection in Living Zebrafish. Sens. Actuators B Chem. 2017, 243, 971–976. [Google Scholar] [CrossRef]
- Wang, G.; Qi, H.; Yang, X.F. A Ratiometric Fluorescent Probe for Bisulphite Anion, Employing Intramolecular Charge Transfer. Luminescence 2013, 28, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Dong, M.; Tang, J.; Lv, Y.; Liu, Y.; Wang, J.; Wang, T.; Bian, J.; Li, C. A Dual–Function Fluorescent Probe for Hg(II) and Cu(II) Ions with Two Mutually Independent Sensing Pathways and Its Logic Gate Behavior. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2020, 226, 117645. [Google Scholar] [CrossRef]
- Madhuprasad; Bhat, M.P.; Jung, H.Y.; Losic, D.; Kurkuri, M.D. Anion Sensors as Logic Gates: A Close Encounter? Chem.-Eur. J. 2016, 22, 6148–6178. [Google Scholar] [CrossRef]
- Skoog, D.A.; West, D.M.; Holler, F.J.; Crouch, S.R. Fundamentals of Analytical Chemistry, 10th ed.; Cengage Learning: Boston, MA, USA, 2021; Volume 1, ISBN 978-0357450390. [Google Scholar]
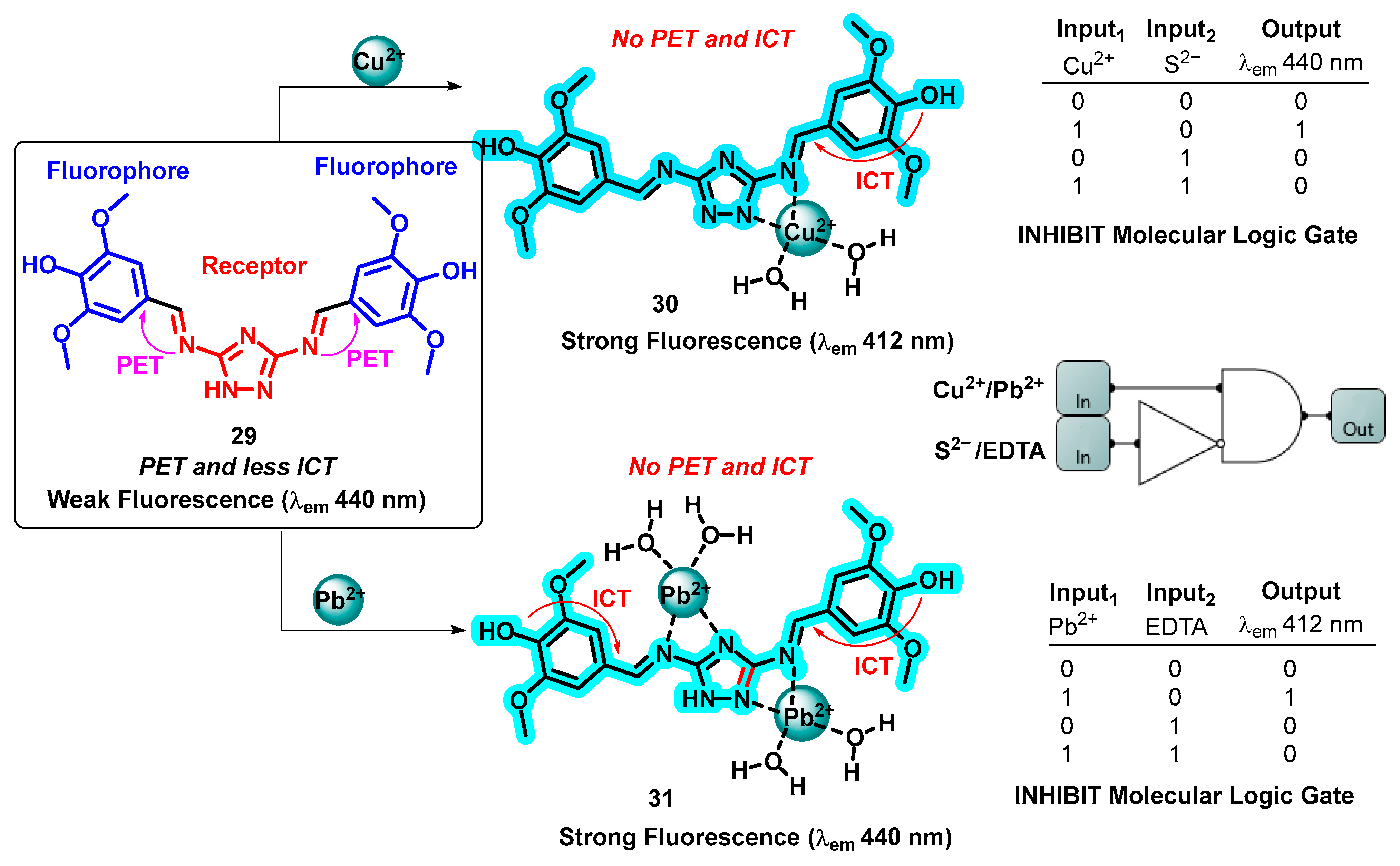
- Manna, A.K.; Chowdhury, S.; Patra, G.K. A Novel Hydrazide-Based Selective and Sensitive Optical Chemosensor for the Detection of Ni2+ Ions: Applications in Live Cell Imaging, Molecular Logic Gates and Smart Phone-Based Analysis. Dalton Trans. 2019, 48, 12336–12348. [Google Scholar] [CrossRef] [PubMed]
- Acharyya, S.; Gharami, S.; Sarkar, D.; Ghosh, P.; Murmu, N.; Mondal, T.K. A Thioether Containing Reversible Fluorescence “Turn-on” Chemosensor for Selective Detection of Zinc(II): Applications in Live Cell Imaging and Inhibit Logic Gate. J. Mol. Struct. 2021, 1224, 129179. [Google Scholar] [CrossRef]
- Qiu, S.; Cui, S.; Shi, F.; Pu, S. Novel Diarylethene-Based Fluorescent Switching for the Detection of Al3+ and Construction of Logic Circuit. ACS Omega 2019, 4, 14841–14848. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Ravi, S.; David, C.I.; Nandhakumar, R. A Photo-Induced Electron Transfer Based Reversible Fluorescent Chemosensor for Specific Detection of Mercury (II) Ions and Its Applications in Logic Gate, Keypad Lock and Real Samples. Arab. J. Chem. 2021, 14, 102911. [Google Scholar] [CrossRef]
- Marini, V.G.; Zimmermann, L.M.; Machado, V.G. A Simple and Efficient Anionic Chromogenic Chemosensor Based on 2,4-Dinitrodiphenylamine in Dimethyl Sulfoxide and in Dimethyl Sulfoxide-Water Mixtures. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2010, 75, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Nandi, L.G.; Facin, F.; Marini, V.G.; Zimmermann, L.M.; Giusti, L.A.; Da Silva, R.; Caramori, G.F.; Machado, V.G. Nitro-Substituted 4-[(Phenylmethylene)Imino]Phenolates: Solvatochromism and Their Use as Solvatochromic Switches and as Probes for the Investigation of Preferential Solvation in Solvent Mixtures. J. Org. Chem. 2012, 77, 10668–10679. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann-Dimer, L.M.; Machado, V.G. Chromogenic Anionic Chemosensors Based on Protonated Merocyanine Solvatochromic Dyes: Influence of the Medium on the Quantitative and Naked-Eye Selective Detection of Anionic Species. Dye. Pigment. 2009, 82, 187–195. [Google Scholar] [CrossRef]
- Souto, F.T.; Buske, J.L.D.O.; Nicoleti, C.R.; Dreyer, J.P.; Heying, R.D.S.; Bortoluzzi, A.J.; Machado, V.G. Chromogenic Chemodosimeter Based on a Silylated Azo Compound Detects Cyanide in Water and Cassava. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2021, 260, 119950. [Google Scholar] [CrossRef]
- Schramm, A.D.S.; Nicoleti, C.R.; Stock, R.I.; Heying, R.S.; Bortoluzzi, A.J.; Machado, V.G. Anionic Optical Devices Based on 4-(Nitrostyryl)Phenols for the Selective Detection of Fluoride in Acetonitrile and Cyanide in Water. Sens. Actuators B Chem. 2017, 240, 1036–1048. [Google Scholar] [CrossRef]
- Sarkar, D.; Ghosh, P.; Gharami, S.; Mondal, T.K.; Murmu, N. A Novel Coumarin Based Molecular Switch for the Sequential Detection of Al3+ and F−: Application in Lung Cancer Live Cell Imaging and Construction of Logic Gate. Sens. Actuators B Chem. 2017, 242, 338–346. [Google Scholar] [CrossRef]
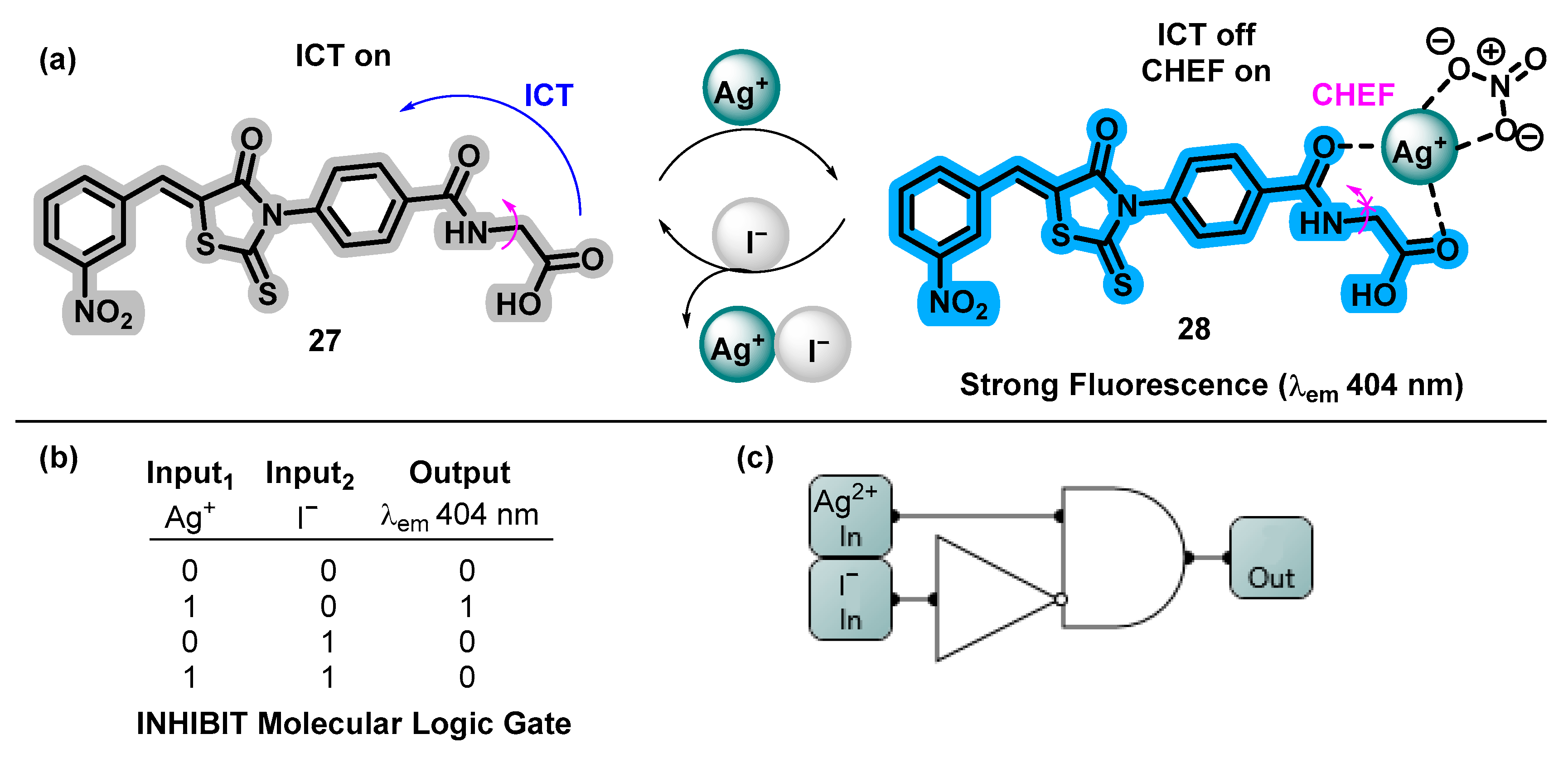
- David, C.I.; Prabakaran, G.; Sundaram, K.; Ravi, S.; devi, D.P.; Abiram, A.; Nandhakumar, R. Rhodanine-Based Fluorometric Sequential Monitoring of Silver (I) and Iodide Ions: Experiment, DFT Calculation and Multifarious Applications. J. Hazard. Mater. 2021, 419, 126449. [Google Scholar] [CrossRef] [PubMed]
- Rout, K.; Manna, A.K.; Sahu, M.; Mondal, J.; Singh, S.K.; Patra, G.K. Triazole-Based Novel Bis Schiff Base Colorimetric and Fluorescent Turn-on Dual Chemosensor for Cu2+ and Pb2+: Application to Living Cell Imaging and Molecular Logic Gates. RSC Adv. 2019, 9, 25919–25931. [Google Scholar] [CrossRef]
- Gupta, R.C.; Razi, S.S.; Ali, R.; Dwivedi, S.K.; Srivastava, P.; Singh, P.; Koch, B.; Mishra, H.; Misra, A. An Efficient Hg2+ Ensemble Based on a Triazole Bridged Anthracene and Quinoline System for Selective Detection of Cyanide through Fluorescence Turn-off–on Response in Solution and Live Cell. Sens. Actuators B Chem. 2017, 251, 729–738. [Google Scholar] [CrossRef]
- Liu, X.; Yao, H.; Li, N.; Xu, M.M.; Jiang, C.; Chen, Z.; Wang, Y.; Shi, H. Cascade Recognition for Fe3+ and CO32− Based on Asymmetric Squaraine Dye: An Application in Mimicking an INHIBIT Logic Gate. Tetrahedron 2020, 76, 131626. [Google Scholar] [CrossRef]
- Mahato, M.; Tohora, N.; Rahman, Z.; Sultana, T.; Ghanta, S.; Das, S.K. A Benzoxazole-Based Smart Molecule for Relay Detection of Zinc and Phosphate Ions and Its Implication towards Molecular Logic Gate Constructions. J. Photochem. Photobiol. A Chem. 2022, 432, 114113. [Google Scholar] [CrossRef]
- Jain, H.; Deswal, N.; Joshi, A.; Ramachandran, C.N.; Kumar, R. Triazole-Appended Pyrano[2,3-c] Pyrazolone Based Colorimetric Chemosensors for Recognition of Fe3+ Ions and Their Molecular Logic Gate Behavior. Anal. Methods 2019, 11, 3230–3243. [Google Scholar] [CrossRef]
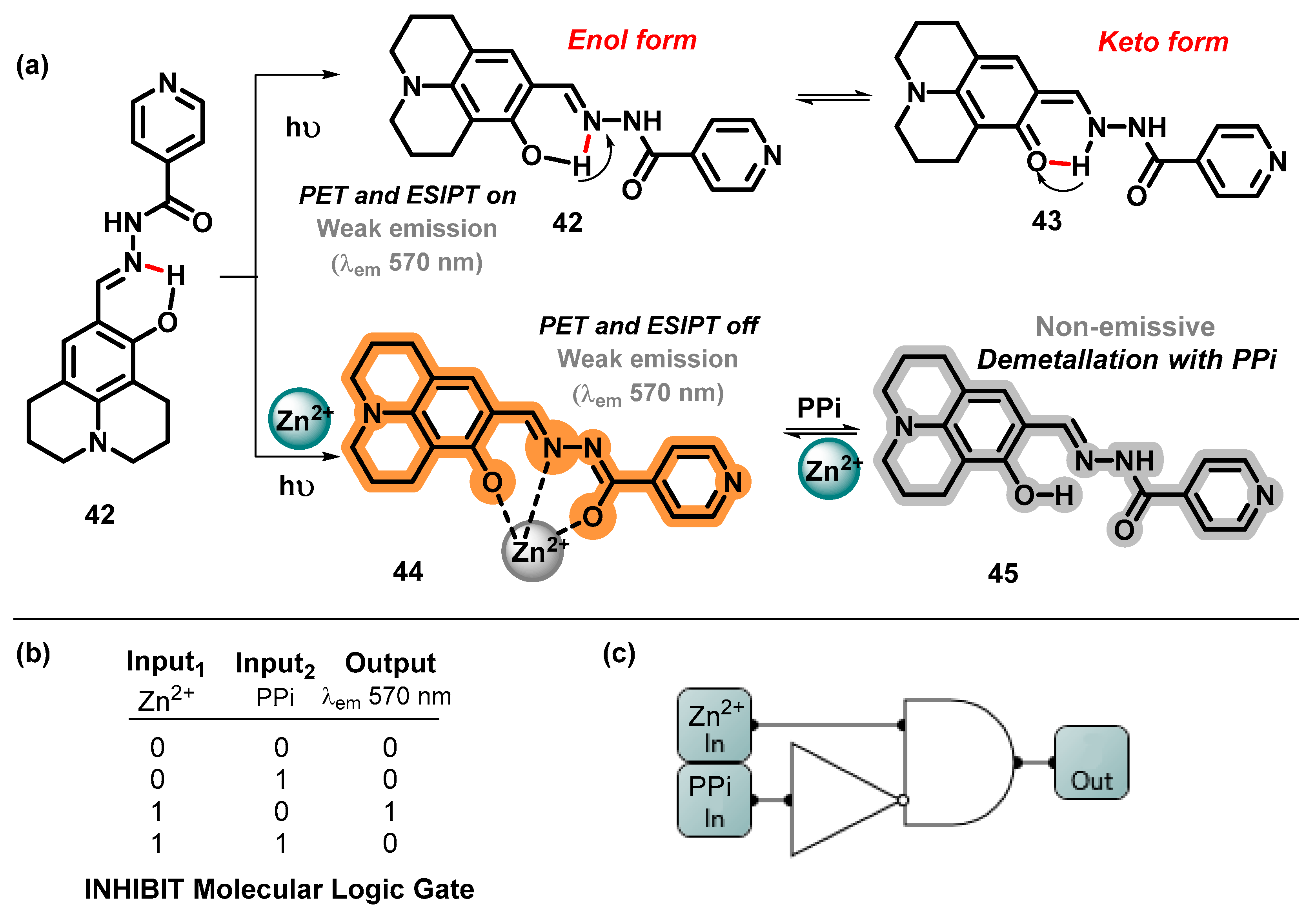
- Megha; Kumar, V.; Kaur, P.; Singh, K. Julolidine-Hydrazone Based Chemosensor for Detection of Zn2+: Fluorescent in-Situ Formed Zn2+ Complex Discriminates PPi from ADP and ATP. Anal. Chim. Acta 2023, 1240, 340758. [Google Scholar] [CrossRef]
- Zhang, J.; She, M.; Li, J.; Wang, C.; Wang, S.; Liu, P.; Zhang, S.; Li, J. Substituent Effect on Fluorescence Signaling of the Naphthalene Carbohydrazone Based Chemosensor: Its Implication in the Detection of Zn(II) Ions and Secondary Sensing PPi. Sens. Actuators B Chem. 2018, 270, 362–370. [Google Scholar] [CrossRef]
- Ravikumar, I.; Ghosh, P. Zinc(II) and PPi Selective Fluorescence OFF–ON–OFF Functionality of a Chemosensor in Physiological Conditions. Inorg. Chem. 2011, 50, 4229–4231. [Google Scholar] [CrossRef]
- Wang, J.; Liu, X.; Pang, Y. A Benzothiazole-Based Sensor for Pyrophosphate (PPi) and ATP: Mechanistic Insight for Anion-Induced ESIPT Turn-On. J. Mater. Chem. B 2014, 2, 6634–6638. [Google Scholar] [CrossRef]
- Naskar, B.; Modak, R.; Maiti, D.K.; Drew, M.G.B.; Bauzá, A.; Frontera, A.; Das Mukhopadhyay, C.; Mishra, S.; Das Saha, K.; Goswami, S. A Schiff Base Platform: Structures, Sensing of Zn(II) and PPi in Aqueous Medium and Anticancer Activity. Dalton Trans. 2017, 46, 9498–9510. [Google Scholar] [CrossRef] [PubMed]
- Tavallali, H.; Deilamy-Rad, G.; Parhami, A.; Hasanli, N. An Efficient and Ultrasensitive Rhodamine B-Based Reversible Colorimetric Chemosensor for Naked-Eye Recognition of Molybdenum and Citrate Ions in Aqueous Solution: Sensing Behavior and Logic Operation. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2015, 139, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Cruywagen, J.J.; Rohwer, E.A.; Wessels, G.F.S. Molybdenum(VI) Complex Formation—8. Equilibria and Thermodynamic Quantities for the Reactions with Citrate. Polyhedron 1995, 14, 3481–3493. [Google Scholar] [CrossRef]
- Manna, A.K.; Mondal, J.; Rout, K.; Patra, G.K. A Benzohydrazide Based Two-in-One Ni2+/Cu2+ Fluorescent Colorimetric Chemosensor and Its Applications in Real Sample Analysis and Molecular Logic Gate. Sens. Actuators B Chem. 2018, 275, 350–358. [Google Scholar] [CrossRef]
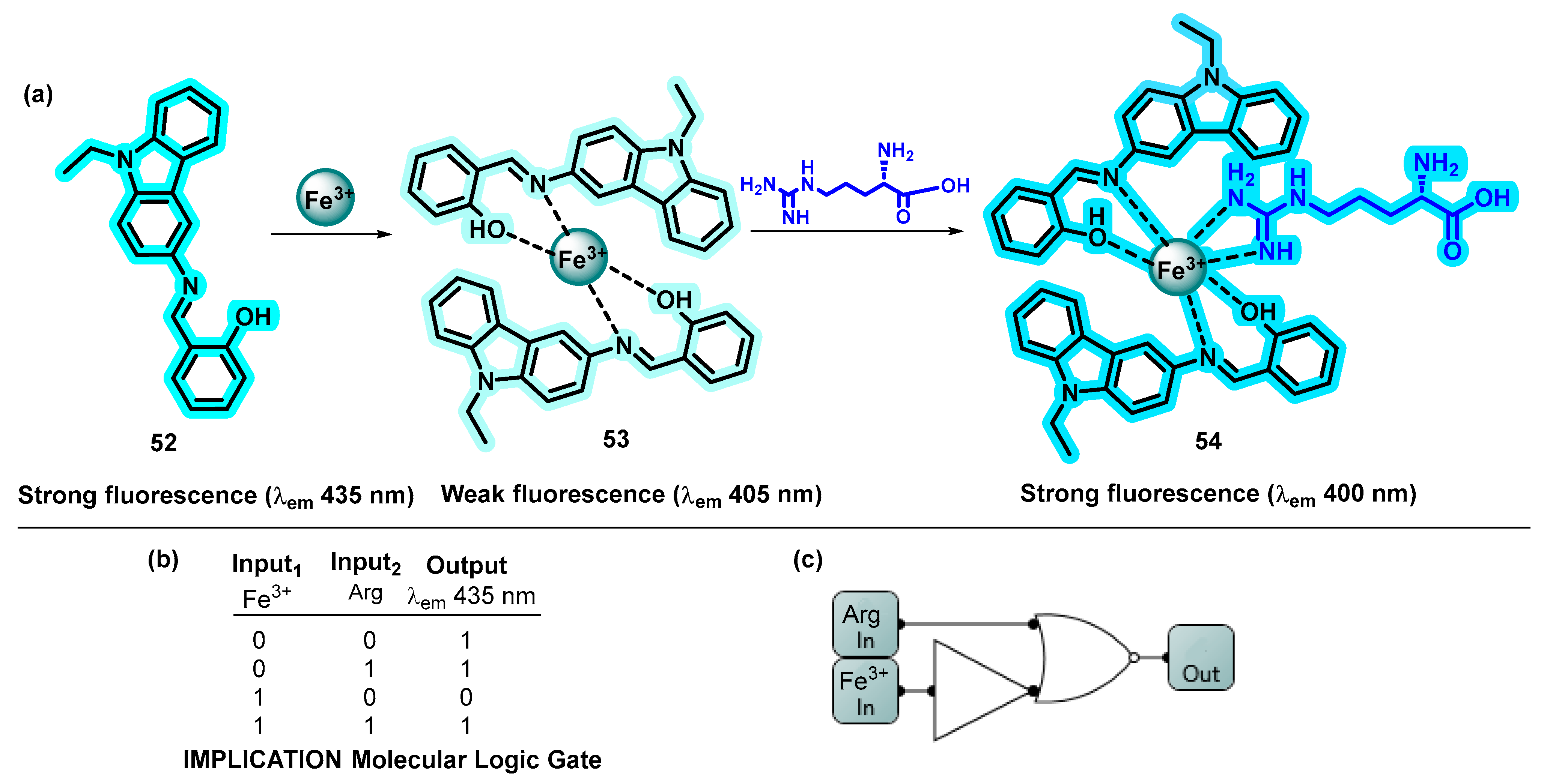
- David, C.I.; Prabakaran, G.; Karuppasamy, A.; Veetil, J.C.; Kumar, R.S.; Almansour, A.I.; Perumal, K.; Ramalingan, C.; Nandhakumar, R. A Single Carbazole Based Chemosensor for Multiple Targets: Sensing of Fe3+ and Arginine by Fluorimetry and Its Applications. J. Photochem. Photobiol. A Chem. 2022, 425, 113693. [Google Scholar] [CrossRef]
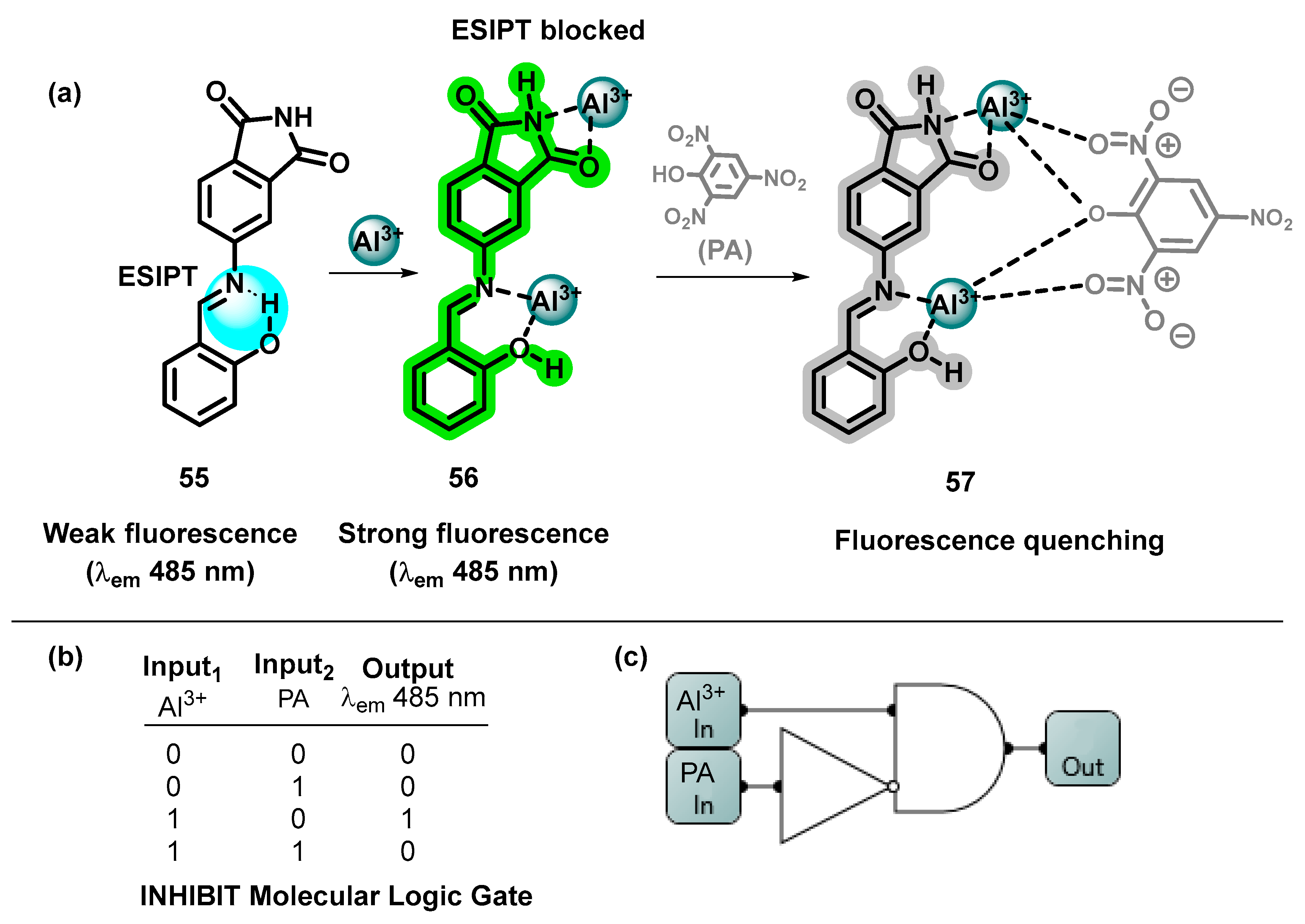
- Rahman, Z.; Tohora, N.; Mahato, M.; Sultana, T.; Pramanik, P.; Ghanta, S.; Das, A.; Datta, P.; Kumar Das, S. A 4-Aminophthalimide Derive Smart Molecule for Sequential Detection of Aluminum Ions and Picric Acid. J. Photochem. Photobiol. A Chem. 2023, 439, 114593. [Google Scholar] [CrossRef]
- Yang, Z.; Cao, J.; He, Y.; Yang, J.H.; Kim, T.; Peng, X.; Kim, J.S. Macro-/Micro-Environment-Sensitive Chemosensing and Biological Imaging. Chem. Soc. Rev. 2014, 43, 4563–4601. [Google Scholar] [CrossRef]
- Huang, X.; Miao, Q.; Wang, L.; Jiao, J.; He, X.; Cheng, Y. A Highly Sensitive and Selective Fluorescence Chemosensor for Cu2+ and Zn2+ Based on Solvent Effect. Chin. J Chem. 2013, 31, 195–199. [Google Scholar] [CrossRef]
- Das, B.; Dolai, M.; Dhara, A.; Ghosh, A.; Mabhai, S.; Misra, A.; Dey, S.; Jana, A. Solvent-Regulated Fluorimetric Differentiation of Al3+ and Zn2+ Using an AIE-Active Single Sensor. J. Phys. Chem. A 2021, 125, 1490–1504. [Google Scholar] [CrossRef]
- Cha, S.; Choi, M.G.; Jeon, H.R.; Chang, S.K. Negative Solvatochromism of Merocyanine Dyes: Application as Water Content Probes for Organic Solvents. Sens. Actuators B Chem. 2011, 157, 14–18. [Google Scholar] [CrossRef]
- Gui, X.-Q.; Guan, A.-L.; Niu, C.-G.; Zeng, G.-M.; Qin, P.-Z. Fluorescence Sensor for Water in Organic Solvents Prepared from Covalent Immobilization of 4-Morpholinyl-1, 8-Naphthalimide. Anal. Bioanal. Chem. 2006, 387, 1067–1074. [Google Scholar] [CrossRef]
- Gauci, G.; Magri, D.C. Solvent-Polarity Reconfigurable Fluorescent 4-Piperazino-N-Aryl-1,8-Naphthalimide Crown Ether Logic Gates. RSC Adv. 2022, 12, 35270–35278. [Google Scholar] [CrossRef]
- Han, X.; Hu, F.; Chi, W.; Ma, X.; Liu, S.H.; Liu, X.; Yin, J. Unusual Intermolecular Charge Transfer Enables Supramolecular Fluorescent Viscosity Sensors. Sens. Actuators B Chem. 2018, 277, 55–61. [Google Scholar] [CrossRef]
- Li, S.J.; Li, Y.F.; Liu, H.W.; Zhou, D.Y.; Jiang, W.L.; Ou-Yang, J.; Li, C.Y. A Dual-Response Fluorescent Probe for the Detection of Viscosity and H2S and Its Application in Studying Their Cross-Talk Influence in Mitochondria. Anal. Chem. 2018, 90, 9418–9425. [Google Scholar] [CrossRef]
- Wang, D.; Tang, B.Z. Aggregation-Induced Emission Luminogens for Activity-Based Sensing. Acc. Chem. Res. 2019, 52, 2559–2570. [Google Scholar] [CrossRef] [PubMed]
- Chua, M.H.; Zhou, H.; Zhu, Q.; Tang, B.Z.; Xu, J.W. Recent Advances in Cation Sensing Using Aggregation-Induced Emission. Mater. Chem. Front. 2021, 5, 659–708. [Google Scholar] [CrossRef]
- Massaad, J.; Micheau, J.C.; Coudret, C.; Sanchez, R.; Guirado, G.; Delbaere, S. Gated Photochromism and Acidity Photomodulation of a Diacid Dithienylethene Dye. Chem. Eur. J. 2012, 18, 6568–6575. [Google Scholar] [CrossRef]
- Reichardt, C. Solvents and Solvent Effects in Organic Chemistry; Wiley: Hoboken, NJ, USA, 2002; ISBN 9783527306183. [Google Scholar]
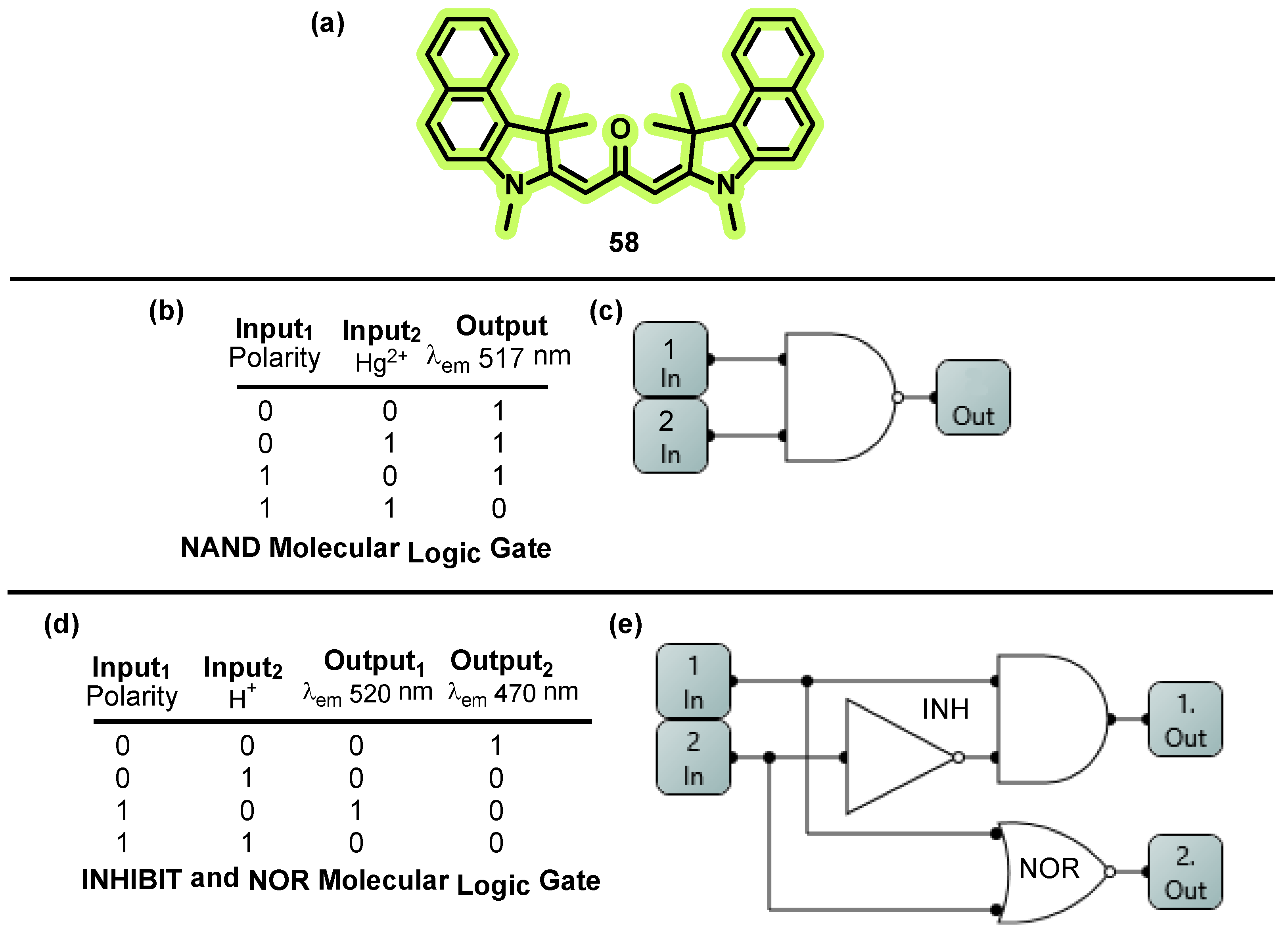
- Zhang, D.; Su, J.; Ma, X.; Tian, H. An Efficient Multiple-Mode Molecular Logic System for PH, Solvent Polarity, and Hg2+ Ions. Tetrahedron 2008, 64, 8515–8521. [Google Scholar] [CrossRef]
- Testoni, F.M.; Ribeiro, E.A.; Giusti, L.A.; Machado, V.G. Merocyanine Solvatochromic Dyes in the Study of Synergistic Effects in Mixtures of Chloroform with Hydrogen-Bond Accepting Solvents. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2009, 71, 1704–1711. [Google Scholar] [CrossRef]
- Da Silva, D.C.; Ricken, I.; Silva, M.A.D.R.; Machado, V.G. Solute-Solvent and Solvent-Solvent Interactions in the Preferential Solvation of Brooker’s Merocyanine in Binary Solvent Mixtures. J. Phys. Org. Chem. 2002, 15, 420–427. [Google Scholar] [CrossRef]
- Stock, R.I.; Dreyer, J.P.; Nunes, G.E.; Bechtold, I.H.; Machado, V.G. Optical Chemosensors and Chemodosimeters for Anion Detection Based on Merrifield Resin Functionalized with Brooker’s Merocyanine Derivatives. ACS Appl. Polym. Mater. 2019, 1, 1757–1768. [Google Scholar] [CrossRef]
- Nandi, L.G.; Nicoleti, C.R.; Bellettini, I.C.; Machado, V.G. Optical Chemosensor for the Detection of Cyanide in Water Based on Ethyl(Hydroxyethyl)Cellulose Functionalized with Brooker’s Merocyanine. Anal. Chem. 2014, 86, 4653–4656. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Halim, S.T.; Awad, M.K. Absorption, Fluorescence, and Semiempirical ASED-MO Studies on a Typical Brooker’s Merocyanine Dye. J. Mol. Struct. 2005, 754, 16–24. [Google Scholar] [CrossRef]
- Ribeiro, E.A.; Sidooski, T.; Nandi, L.G.; Machado, V.G. Interaction of Protonated Merocyanine Dyes with Amines in Organic Solvents. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2011, 81, 745–753. [Google Scholar] [CrossRef] [PubMed]
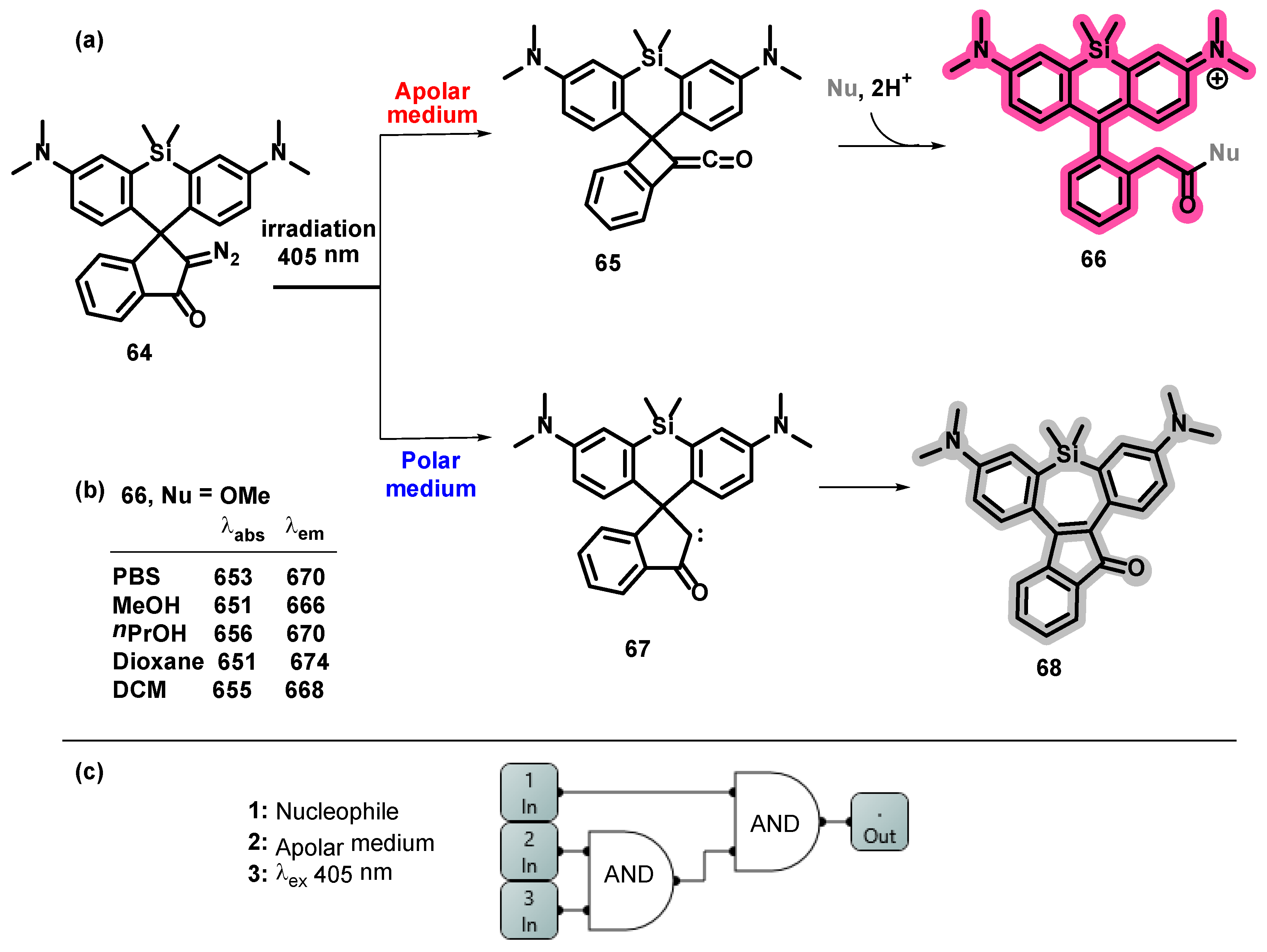
- Eördögh, Á.; Paganini, C.; Pinotsi, D.; Arosio, P.; Rivera-Fuentes, P. A Molecular Logic Gate Enables Single-Molecule Imaging and Tracking of Lipids in Intracellular Domains. ACS Chem. Biol. 2020, 15, 2597–2604. [Google Scholar] [CrossRef] [PubMed]
- Nie, K.; Dong, B.; Shi, H.; Chao, L.; Duan, X.; Jiang, X.F.; Liu, Z.; Liang, B. N-Alkylated Diketopyrrolopyrrole-Based Ratiometric/Fluorescent Probes for Cu2+ Detection via Radical Process. Dye. Pigment. 2019, 160, 814–822. [Google Scholar] [CrossRef]
- Kuder, J.E.; Wychick, D. Acid-Base Equilibria in the Ground and Excited Stated of a Solvatochromic Merocyanine Dye. Chem. Phys. Lett. 1974, 24, 69–72. [Google Scholar] [CrossRef]
- Berezin, M.Y.; Kao, J.; Achilefu, S. pH-Dependent Optical Properties of Synthetic Fluorescent Imidazoles. Chem. Eur. J. 2009, 15, 3560–3566. [Google Scholar] [CrossRef]
- Minkin, V.I.; Tsukanov, A.V.; Dubonosov, A.D.; Bren, V.A. Tautomeric Schiff Bases: Iono-, Solvato-, Thermo- and Photochromism. J. Mol. Struct. 2011, 998, 179–191. [Google Scholar] [CrossRef]
- Luo, Z.; Liu, B.; Si, S.; Lin, Y.; Luo, C.S.; Pan, C.; Zhao, C.; Wang, L. A Fluorescent Chemosensor Based on Nonplanar Donor-Acceptor Structure for Highly Sensitive and Selective Detection of Picric Acid in Water. Dye. Pigment. 2017, 143, 463–469. [Google Scholar] [CrossRef]
- Shiraishi, Y.; Tokitoh, Y.; Nishimura, G.; Hirai, T. Solvent-Driven Multiply Configurable on/off Fluorescent Indicator of the PH Window: A Diethylenetriamine Bearing Two End Pyrene Fragments. J. Phys. Chem. B 2007, 111, 5090–5100. [Google Scholar] [CrossRef] [PubMed]
- Mu, L.; Shi, W.; Zhang, T.; Zhang, H.; She, G. pH-Controlled Silicon Nanowires Fluorescence Switch. Appl. Surf. Sci. 2010, 256, 5935–5938. [Google Scholar] [CrossRef]
- Zhang, H.; Shao, X.; Chipot, C.; Cai, W. PH-Controlled Fluorescence Probes for Rotaxane Isomerization. J. Phys. Chem. C 2019, 123, 11304–11309. [Google Scholar] [CrossRef]
- Sachdeva, T.; Gupta, S.; Milton, M.D. Smart Organic Materials with Acidochromic Properties. Curr. Org. Chem. 2020, 24, 1976–1998. [Google Scholar] [CrossRef]
- Sachdeva, T.; Milton, M.D. Novel Push-Pull Based Phenothiazine-Benzothiazole Derivatives Integrated with Molecular Logic Gate Operation for Reversible Volatile Acid Detection. J. Mol. Struct. 2021, 1243, 130768. [Google Scholar] [CrossRef]
- Said, A.I.; Georgiev, N.I.; Bojinov, V.B. A Fluorescent Bichromophoric “off-on-off” PH Probe as a Molecular Logic Device (Half-Subtractor and Digital Comparator) Operating by Controlled PET and ICT Processes. Dye. Pigment. 2019, 162, 377–384. [Google Scholar] [CrossRef]
- Nicoli, F.; Paltrinieri, E.; Bakić, M.T.; Baroncini, M.; Silvi, S.; Credi, A. Binary Logic Operations with Artificial Molecular Machines. Coord. Chem. Rev. 2021, 428, 213589. [Google Scholar] [CrossRef]
- Leigh, D.A.; Morales, M.Á.F.; Pérez, E.M.; Wong, J.K.Y.; Saiz, C.G.; Slawin, A.M.Z.; Carmichael, A.J.; Haddleton, D.M.; Brouwer, A.M.; Buma, W.J.; et al. Patterning through Controlled Submolecular Motion: Rotaxane-Based Switches and Logic Gates That Function in Solution and Polymer Films. Angew. Chem.-Int. Ed. 2005, 44, 3062–3067. [Google Scholar] [CrossRef]
- Feinberg, M. Validation of Analytical Methods Based on Accuracy Profiles. J. Chromatogr. A 2007, 1158, 174–183. [Google Scholar] [CrossRef]
- Panuwet, P.; Hunter, R.E.; D’Souza, P.E.; Chen, X.; Radford, S.A.; Cohen, J.R.; Marder, M.E.; Kartavenka, K.; Ryan, P.B.; Barr, D.B. Biological Matrix Effects in Quantitative Tandem Mass Spectrometry-Based Analytical Methods: Advancing Biomonitoring. Crit. Rev. Anal. Chem. 2016, 46, 93–105. [Google Scholar] [CrossRef]
- Kim, S.H.; Lee, Y.H.; Jeong, M.J.; Gwon, D.Y.; Lee, J.H.; Shin, Y.; Choi, H. LC-MS/MS Method Minimizing Matrix Effect for the Analysis of Bifenthrin and Butachlor in Chinese Chives and Its Application for Residual Study. Foods 2023, 12, 1683. [Google Scholar] [CrossRef] [PubMed]

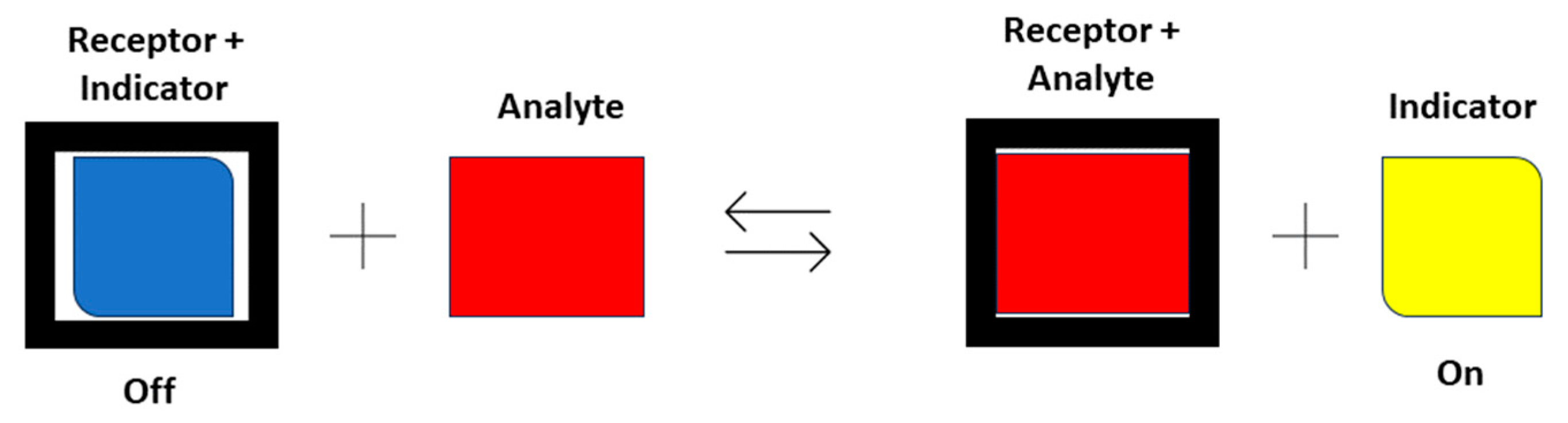











| Logic Gate Truth Table with Single Input | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Input | Output PASS 0 | Output NOT | Output YES | Output PASS 1 | |||||
| 0 | 0 | 1 | 0 | 1 | |||||
| 1 | 0 | 0 | 1 | 1 | |||||
| Logic Gate Truth Table with double input | |||||||||
 | |||||||||
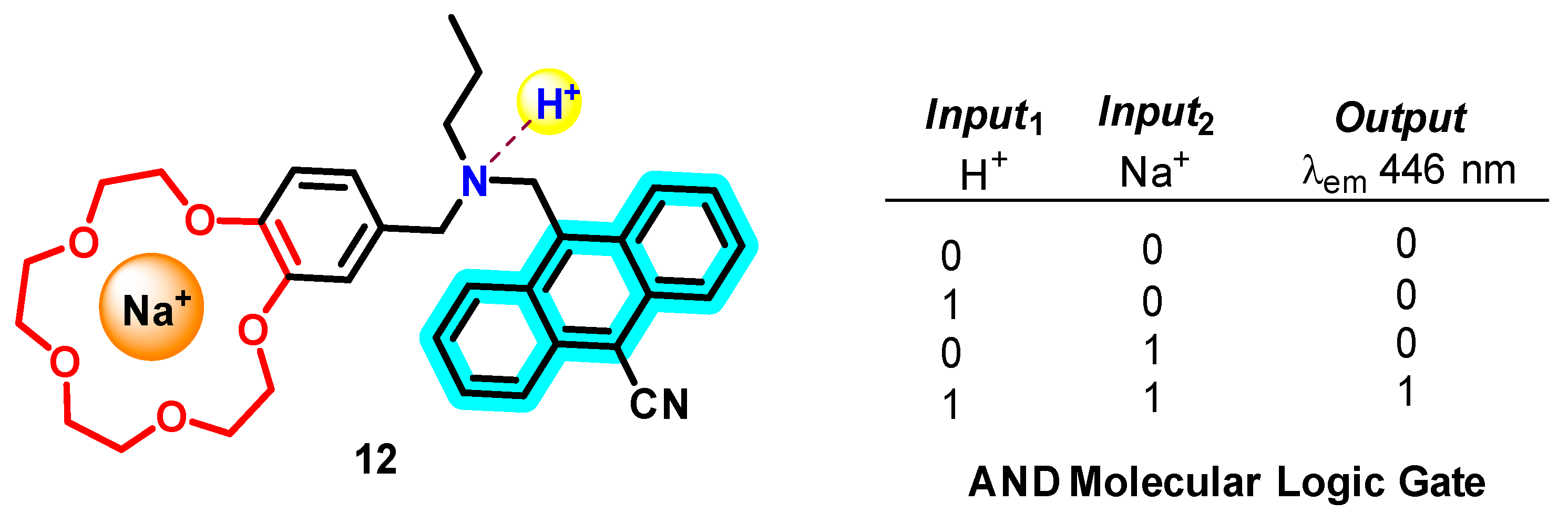
| Input1 | Input2 | AND | NAND | OR | XOR | NOR | XNOR | INHIBIT | IMPLICATION |
| 0 | 0 | 0 | 1 | 0 | 0 | 1 | 1 | 0 | 1 |
| 0 | 1 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 1 |
| 1 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 1 | 0 |
| 1 | 1 | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 1 |
| Cation | Ka | Log K |
|---|---|---|
| Mg2+ | 4.9 × 108 | 8.69 |
| Ni2+ | 4.2 × 1018 | 18.62 |
| Cu2+ | 6.3 × 1018 | 18.80 |
| Zn2+ | 3.2 × 1016 | 16.50 |
| Hg2+ | 6.3 × 1021 | 21.80 |
| Pb2+ | 1.1 × 1018 | 18.04 |
| Al3+ | 1.3 × 1016 | 16.13 |
| Fe3+ | 1.3 × 1025 | 25.10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Souto, F.T.; Dias, G.G. Input Selection Drives Molecular Logic Gate Design. Analytica 2023, 4, 456-499. https://doi.org/10.3390/analytica4040033
Souto FT, Dias GG. Input Selection Drives Molecular Logic Gate Design. Analytica. 2023; 4(4):456-499. https://doi.org/10.3390/analytica4040033
Chicago/Turabian StyleSouto, Francielly T., and Gleiston G. Dias. 2023. "Input Selection Drives Molecular Logic Gate Design" Analytica 4, no. 4: 456-499. https://doi.org/10.3390/analytica4040033
APA StyleSouto, F. T., & Dias, G. G. (2023). Input Selection Drives Molecular Logic Gate Design. Analytica, 4(4), 456-499. https://doi.org/10.3390/analytica4040033







