A Computational Fluid Dynamics Analysis of Hydrogen Leakage and Nitrogen Purging of a Solid Oxide Fuel Cell Stack
Abstract
:1. Introduction
- Simulate the purging of the hot box with nitrogen and assess the amount of nitrogen required and how long it takes until the maximum oxygen concentration reaches 5%, given that it is filled with air at atmospheric pressure and initial temperatures of 800 °C and 300 °C, respectively.
- Assess the cooling effect of the purge on the hot box.
- Assess how long it takes for the hydrogen concentration to exceed 0.4% at the outlet if a leak occurs from the fuel-to-fuel heat exchanger during the OCV test.
2. Mathematical Model
2.1. Main Assumptions
- The gases are assumed as ideal.
- The flow is assumed as incompressible.
- Buoyancy effects are neglected during the purge simulations.
- For the modelling of diffusive transport, both the laminar and turbulent Schmidt numbers are equal to one.
2.2. Governing Equations
2.3. Turbulence Models
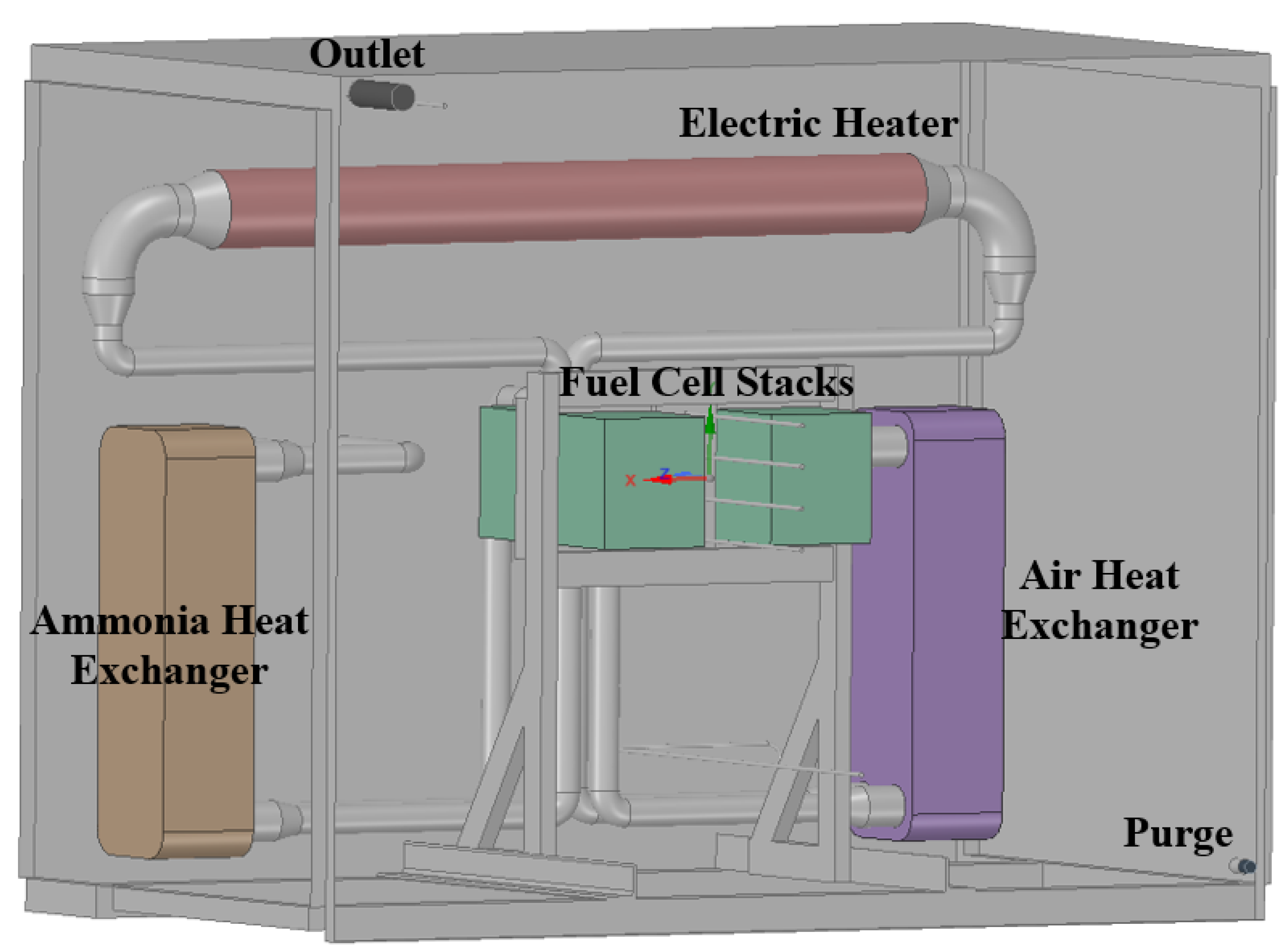
2.4. Boundary and Initial Conditions
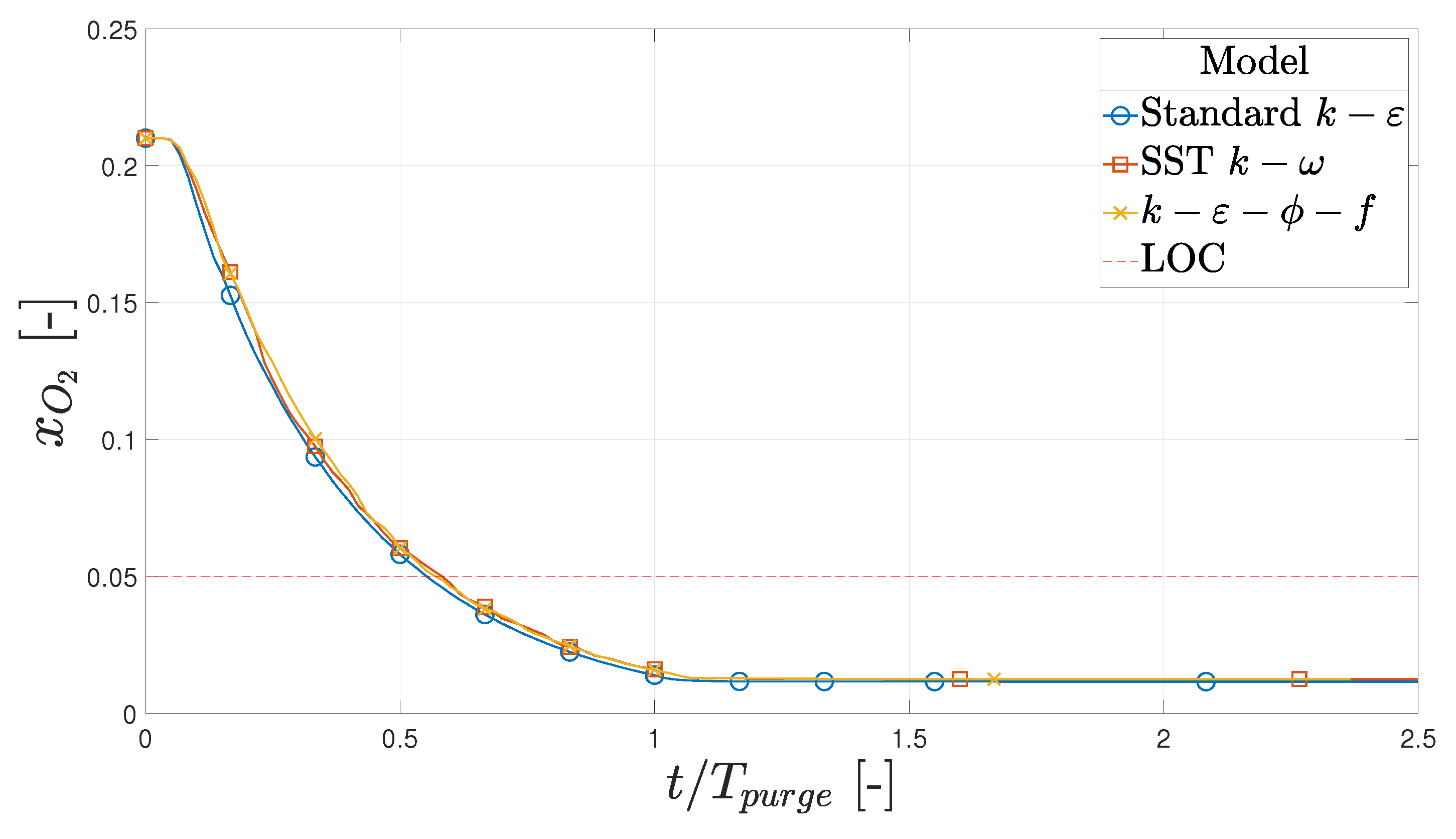
2.5. Numerical Verification
3. Results and Discussion
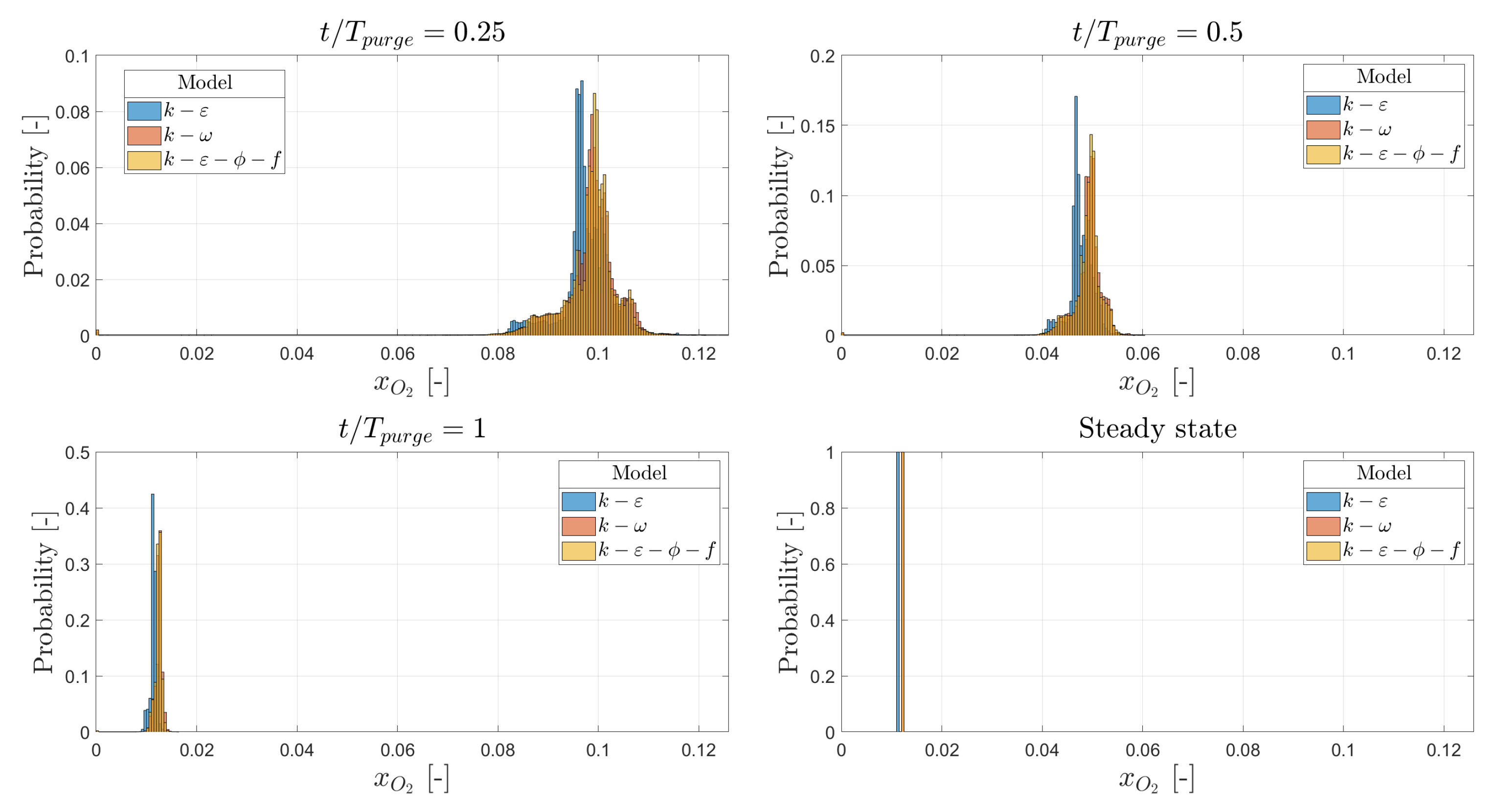
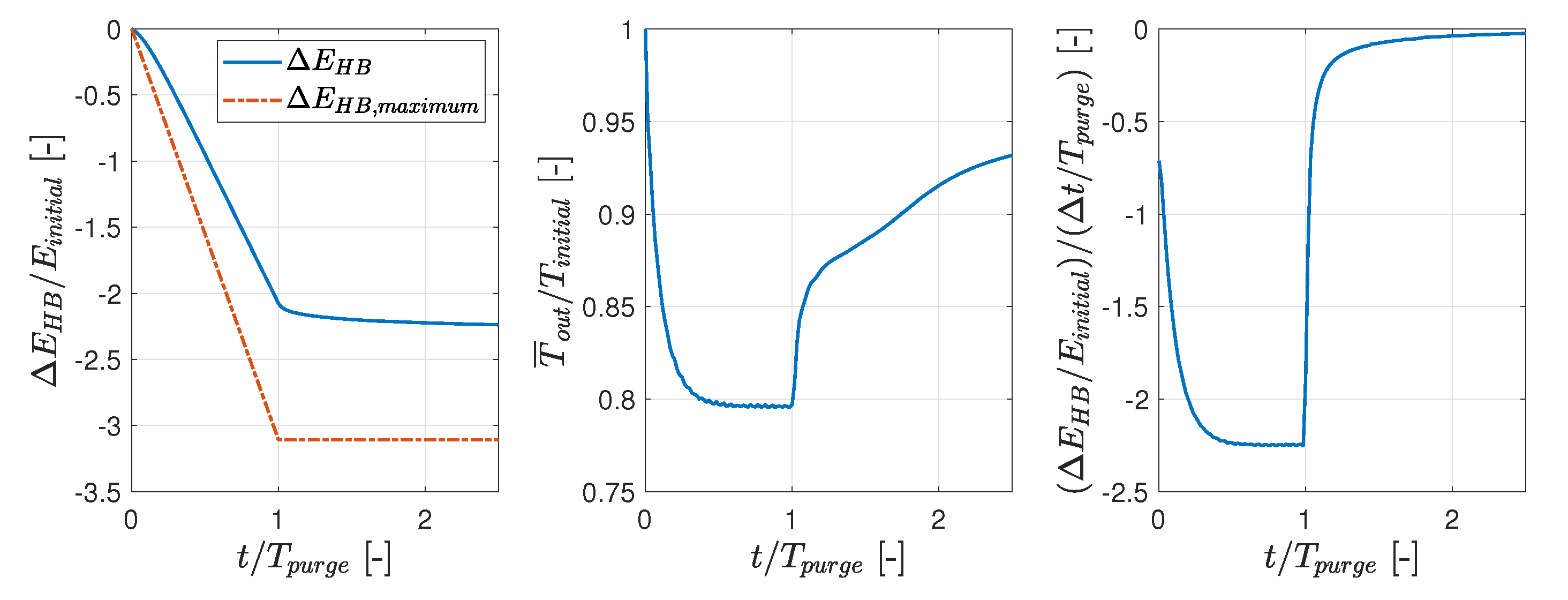
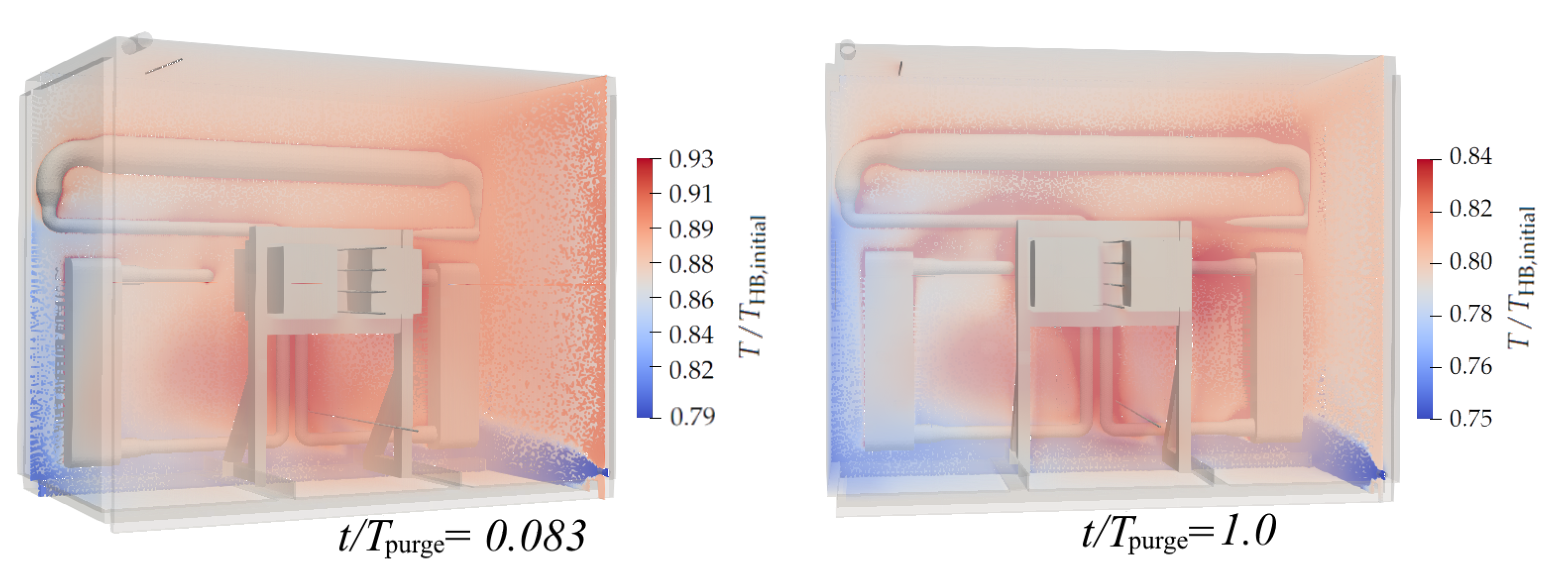
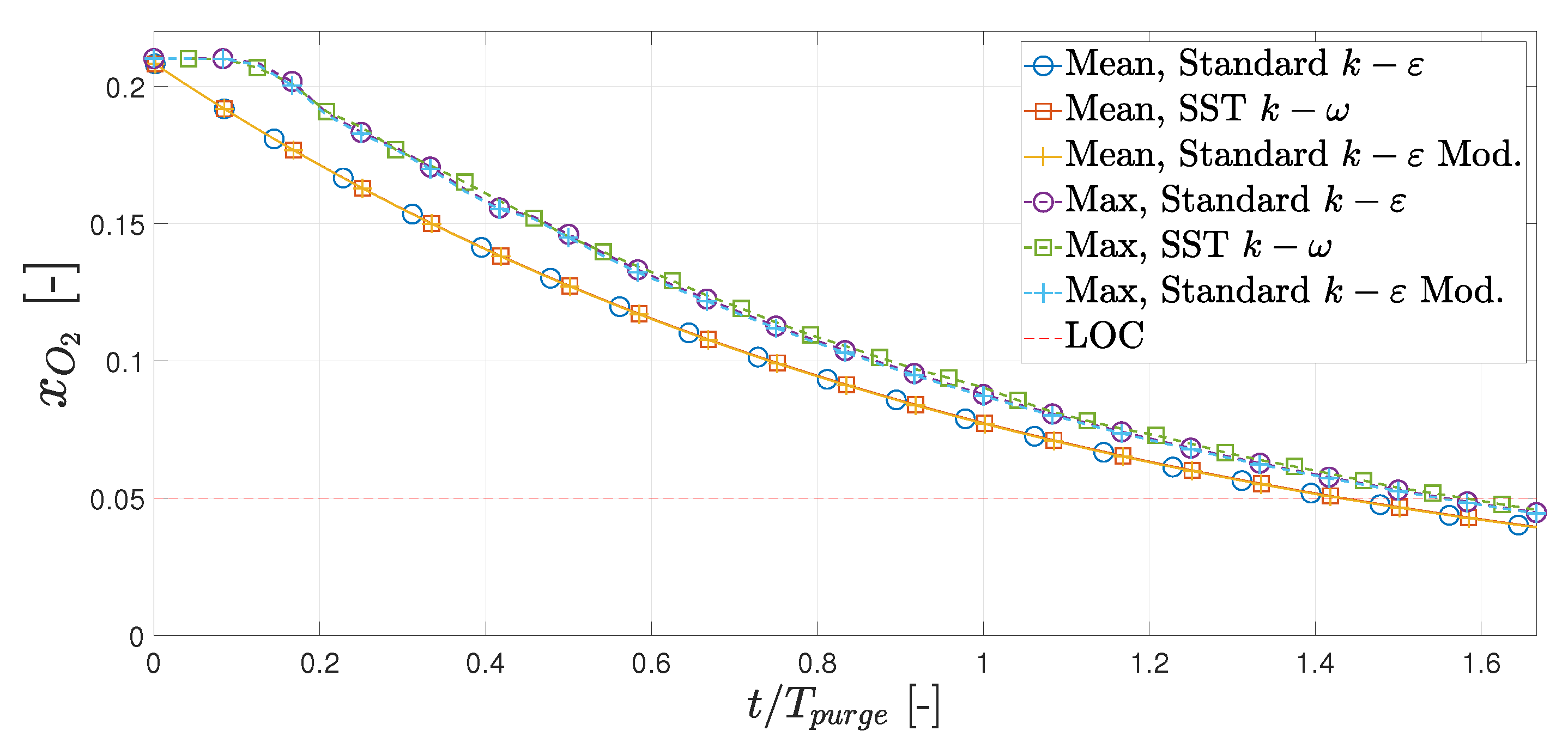
3.1. Hot Purge
3.2. Cold Purge
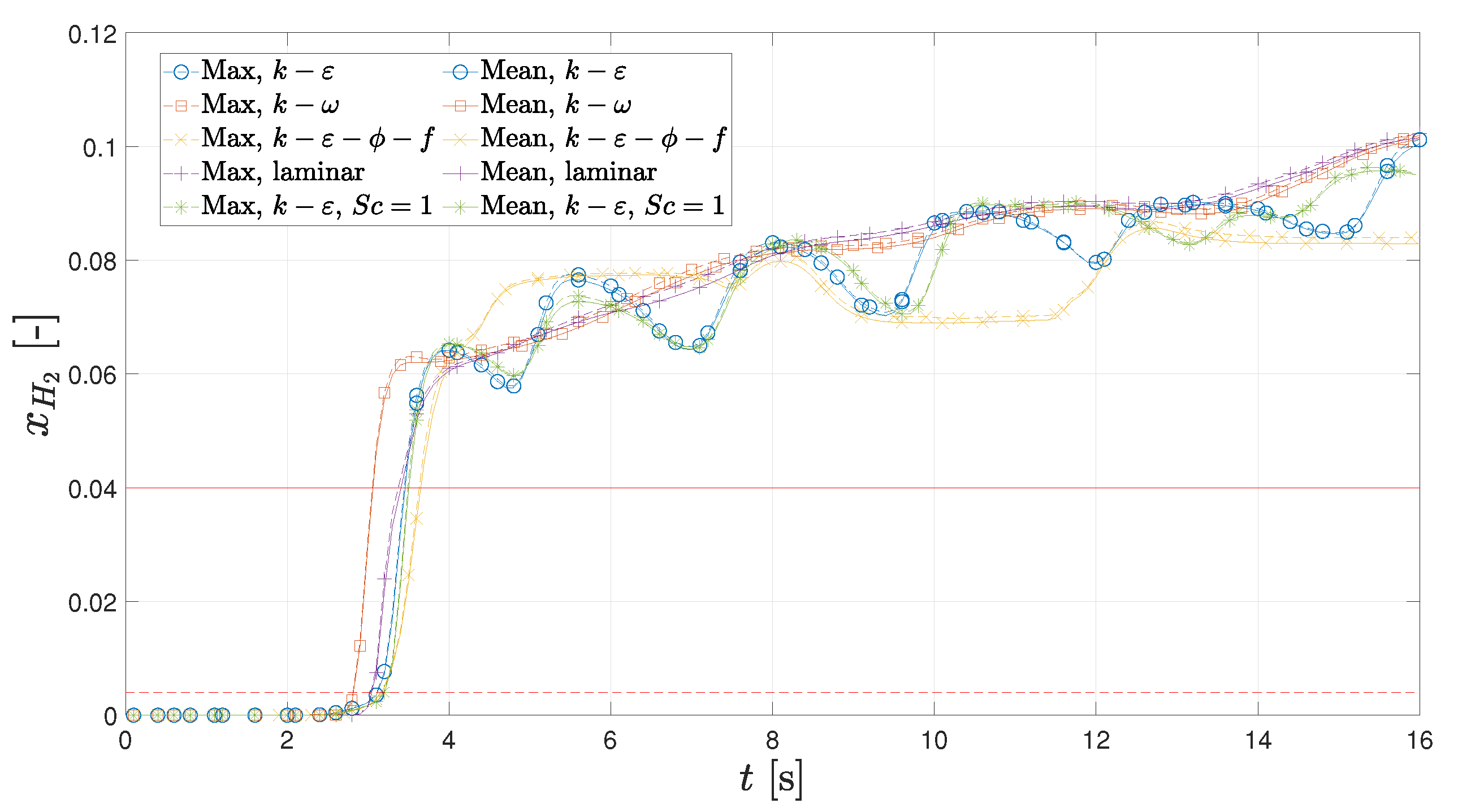
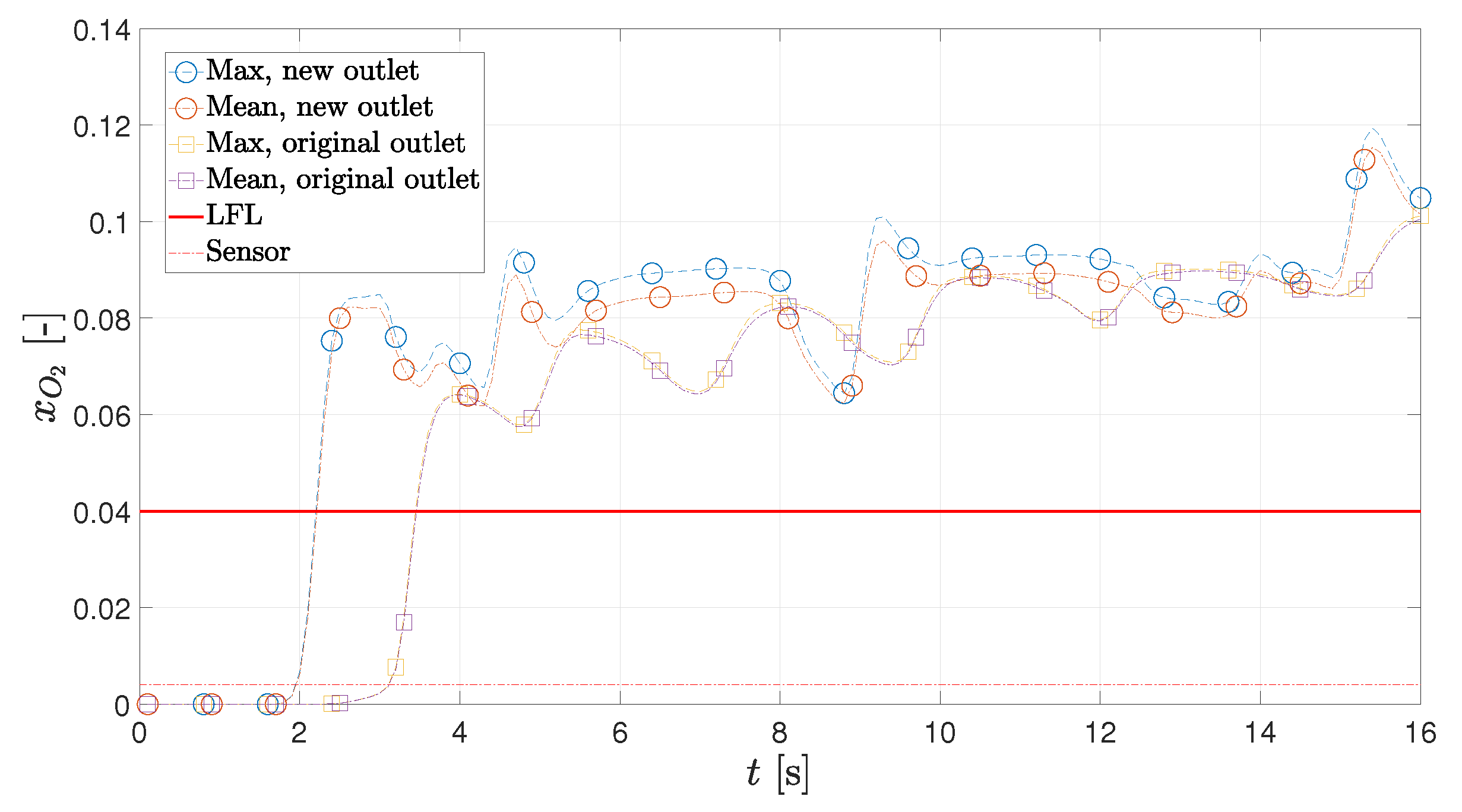
3.3. OCV Leak
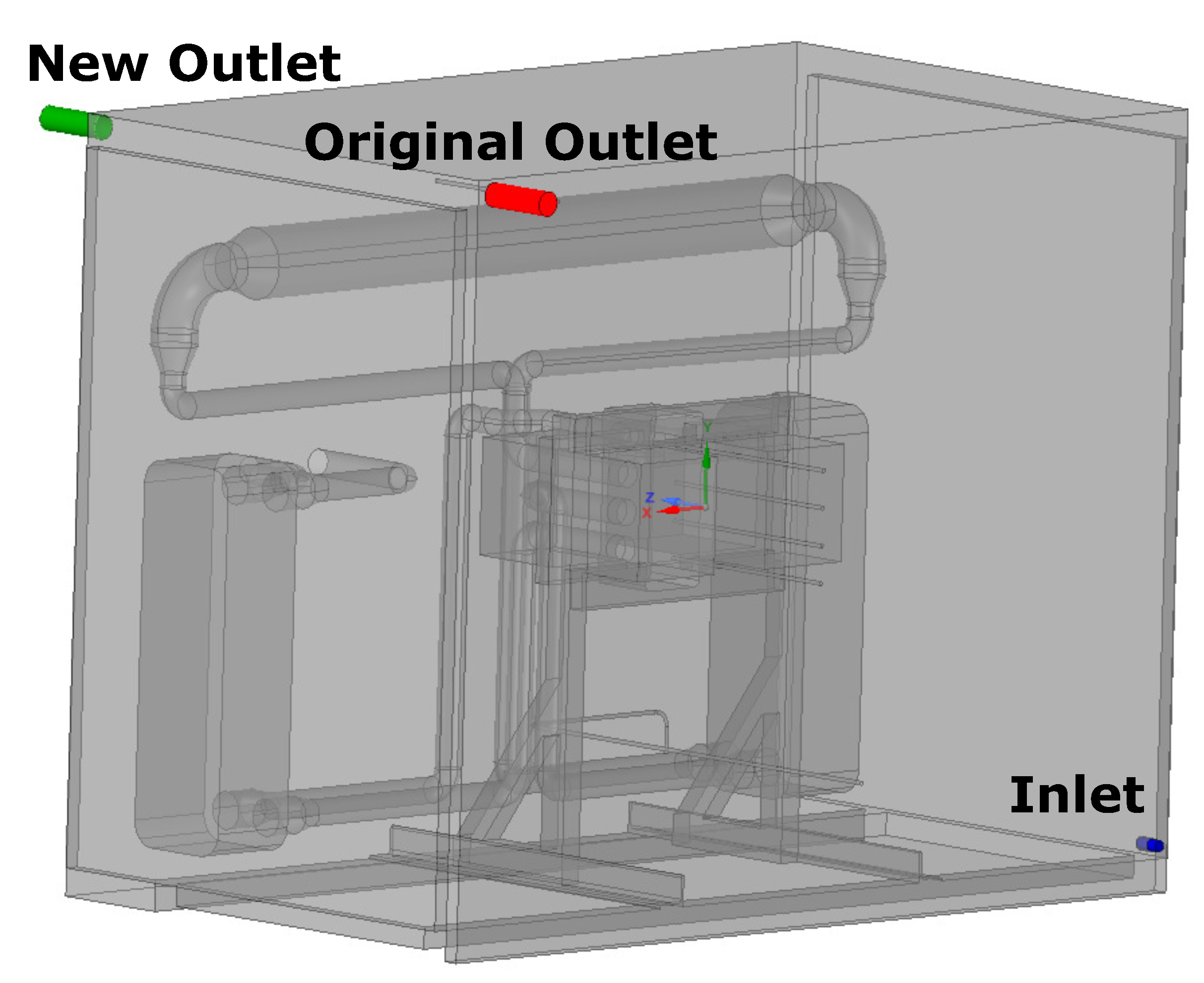
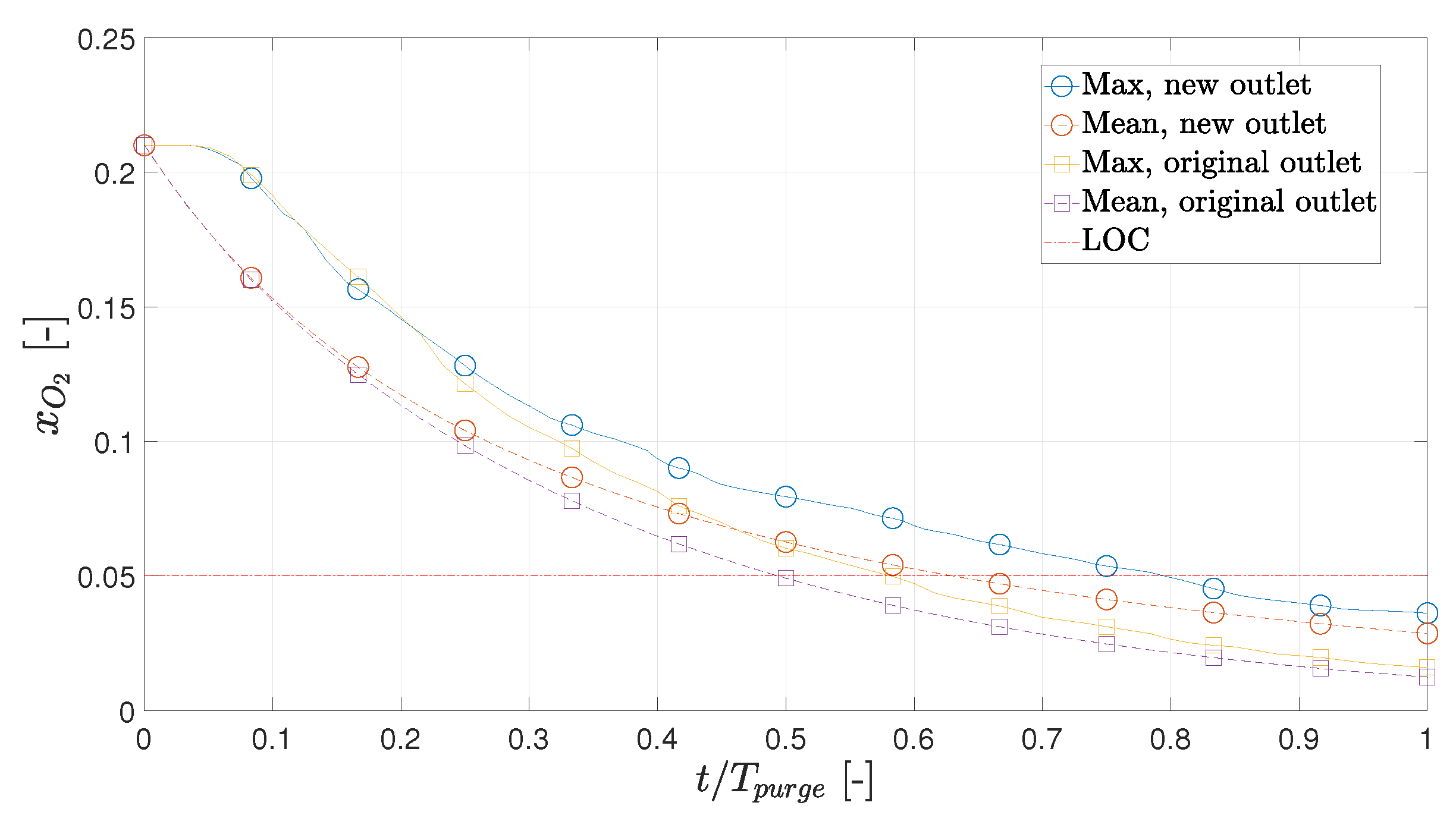
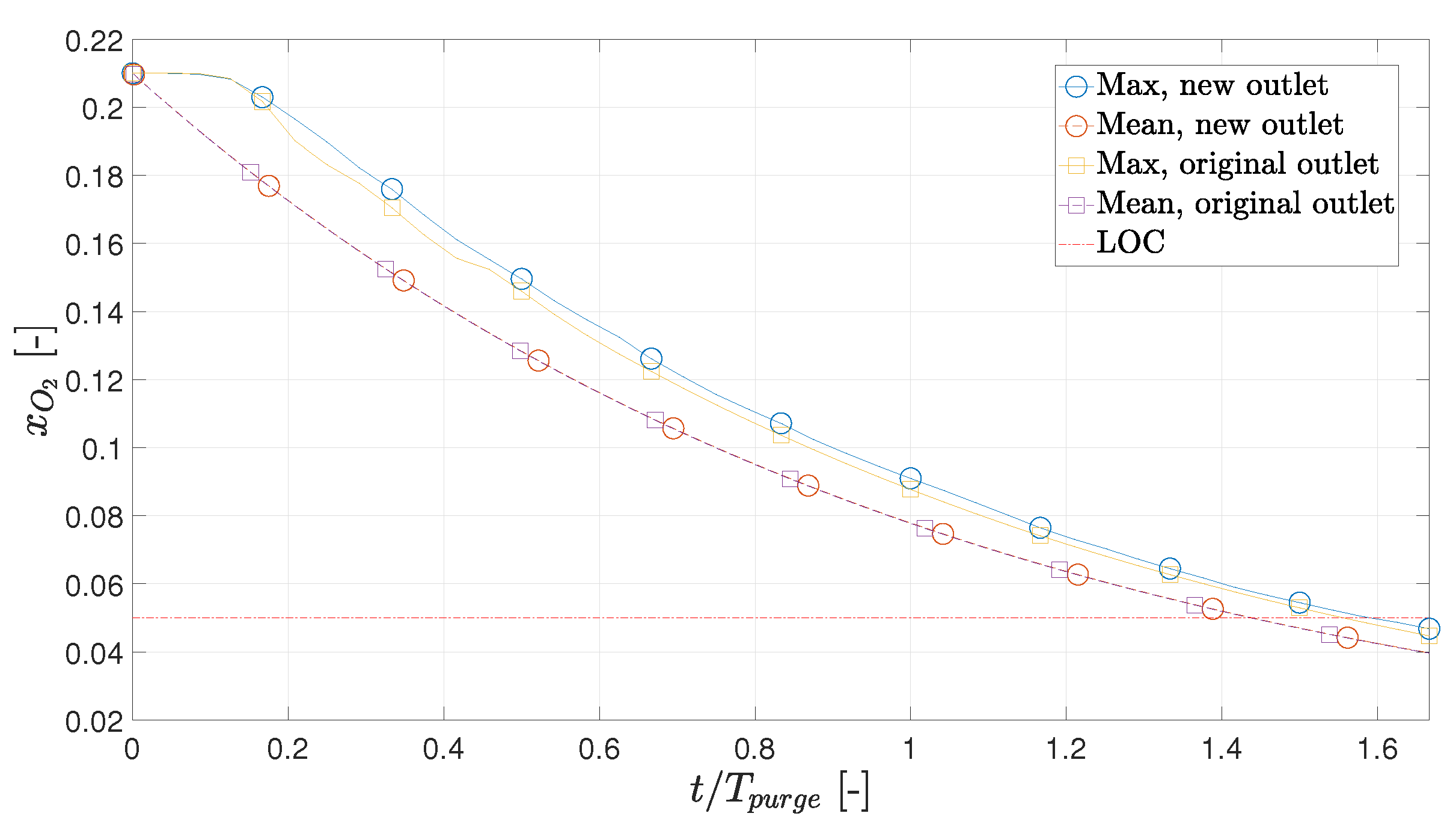
3.4. Alternate Outlet
4. Conclusions
- Based on this study, the initial purge of the hot box, when it is still cold, should last a minimum of (94.8 s), corresponding to 3.0 kg of nitrogen. If the outlet is moved to the opposite side, the minimum initial purge period is (95.5 s), meaning the effect is negligible.
- If the hot box is filled with air at K, the hot box should be purged for (35 s), corresponding to 1.1 kg of nitrogen. Moving the outlet to the opposite side would increase this period to 48 s. This means the original position is 37% more effective at purging compared to the new position.
- The leak at the original outlet would take 3.2 s to be detected if the leak was to occur during an OCV test. Moving the outlet to the opposite side would result in a reduction in the detection time by 1.2 s, meaning it is 39% faster.
- During periodic purges of the hot box, while it is operating, it can be expected that 72% of the purging nitrogen is heated from K to K. During the purge, the average heat loss is 17.9 kW.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CAD | Computer Aided Design |
| CFD | Computational Fluid Dynamics |
| FC | Fuel Cell |
| HB | Hot Box |
| IMO | International Maritime Organization |
| LEL | Lower Exlosive Level |
| LFL | Lower Flammable Level |
| LOC | Lower Oxygen Concentration |
| OCV | Open Circuit Voltage |
| RANS | Reynolds-averaged Navier-Stokes |
| SOFC | Solid Oxide Fuel Cell |
| SST | Shear Stress Transport |
References
- EU. Europe’s 2030 Climate and Energy Targets: Research & Innovation Actions; Technical Report; European Commission, Directorate-General for Research and Innovation: Brussels, Belgium, 2021. [Google Scholar]
- UNFCCC. Adoption of the Paris Agreement. In Proceedings of the 21st Conference of the Parties, United Nations/Framework Convention on Climate Change, Paris, France, 30 November–12 December 2015. [Google Scholar]
- IMO. IMO 2020—Cutting Sulphur Oxide Emissions; Technical Report; International Maritime Organization: London, UK, 2020. [Google Scholar]
- O’Hayre, R.; Cha, S.W.; Colella, W.; Prinz, F.B. Fuel Cell Fundamentals; John Wiley & Sons: Hoboken, NJ, USA, 2016. [Google Scholar] [CrossRef]
- Liso, V.; Xie, P.; Araya, S.S.; Guerrero, J.M. Modelling a modular configuration of PEM fuel cell system for vessels applications. In Proceedings of the 7th World Maritime Technology Conference, Copenhagen, Denmark, 26–28 April 2022; pp. 226–234. [Google Scholar]
- Araya, S.S.; Liso, V.; Cui, X.; Li, N.; Zhu, J.; Sahlin, S.L.; Jensen, S.H.; Nielsen, M.P.; Kær, S.K. A Review of The Methanol Economy: The Fuel Cell Route. Energies 2020, 13, 596. [Google Scholar] [CrossRef]
- Hagen, A.; Langnickel, H.; Sun, X. Operation of solid oxide fuel cells with alternative hydrogen carriers. Int. J. Hydrogen Energy 2019, 44, 18382–18392. [Google Scholar] [CrossRef]
- ABS; CE-DELFT; ARCSILEA. Potential of Ammonia as Fuel in Shipping; Technical report; European Maritime Safety Agency: Lisbon, Portugal, 2022. [Google Scholar]
- Heitsch, M.; Baraldi, D.; Moretto, P. Numerical analysis of accidental hydrogen release in a laboratory. Int. J. Hydrog. Energy 2010, 35, 4409–4419. [Google Scholar] [CrossRef]
- Dadashzadeh, M.; Ahmad, A.; Khan, F. Dispersion modelling and analysis of hydrogen fuel gas released in an enclosed area: A CFD-based approach. Fuel 2016, 184, 192–201. [Google Scholar] [CrossRef]
- Malakhov, A.A.; Avdeenkov, A.V.; du Toit, M.H.; Bessarabov, D.G. CFD simulation and experimental study of a hydrogen leak in a semi-closed space with the purpose of risk mitigation. Int. J. Hydrogen Energy 2020, 45, 9231–9240. [Google Scholar] [CrossRef]
- Cinti, G.; Barelli, L.; Bidini, G. The use of ammonia as a fuel for transport: Integration with solid oxide fuel cells. In Proceedings of the 74th ATI NATIONAL CONGRESS: Energy Conversion: Research, Innovation and Development for Industry and Territories, Modena, Italy, 11–13 September 2019. [Google Scholar] [CrossRef]
- Molkov, V. Fundamentals of Hydrogen Safety Engineering I; Bookboon: Copenhagen, Denmark, 2012. [Google Scholar]
- Kayfeci, M.; Kecebas, A.; Bayat, M. Solar Hydrogen Production, 1st ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 45–83. [Google Scholar]
- Kikukawa, S. Consequence analysis and safety verification of hydrogen fueling stations using CFD simulation. Int. J. Hydrogen Energy 2008, 33, 1425–1434. [Google Scholar] [CrossRef]
- Bédard-Tremblay, L.; Fang, L.; Melguizo-Gavilanes, J.; Bauwens, L.; Finstad, P.; Cheng, Z.; Tchouvelev, A. Simulation of detonation after an accidental hydrogen release in enclosed environments. Int. J. Hydrogen Energy 2009, 34, 5894–5901. [Google Scholar] [CrossRef]
- Mack, A.; Spruijt, M.P. Validation of OpenFoam for heavy gas dispersion applications. J. Hazard. Mater. 2013, 262, 504–516. [Google Scholar] [CrossRef] [PubMed]
- Fiates, J.; Vianna, S.S. Numerical modelling of gas dispersion using OpenFOAM. Process. Saf. Environ. Prot. 2016, 104, 277–293. [Google Scholar] [CrossRef]
- Yang, Q.; Zhao, P.; Ge, H. reactingFoam-SCI: An open source CFD platform for reacting flow simulation. Comput. Fluids 2019, 190, 114–127. [Google Scholar] [CrossRef]
- Versteeg, H.K.; Malalasekera, W. An Introduction to Computational Fluid Dynamics the Finite Volume Method, 2nd ed.; Pearson Education Limited: London, UK, 2007. [Google Scholar]
- Gualtieri, C.; Angeloudis, A.; Bombardelli, F.; Jha, S.; Stoesser, T. On the values for the turbulent schmidt number in environmental flows. Fluids 2017, 2, 17. [Google Scholar] [CrossRef]
- Greenshields, C. OpenFOAM v10 User Guide; The OpenFOAM Foundation: London, UK, 2022. [Google Scholar]
- Laurence, D.R.; Uribe, J.C.; Utyuzhnikov, S.V. A Robust Formulation of the v2-f Model. Flow Turbul. Combust. 2004, 73, 169–185. [Google Scholar] [CrossRef]
- Greenshields, C.; Weller, H. Notes on Computational Fluid Dynamics: General Principles; CFD Direct Ltd.: Reading, UK, 2022; p. 291. [Google Scholar]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sørensen, R.D.; Berning, T. A Computational Fluid Dynamics Analysis of Hydrogen Leakage and Nitrogen Purging of a Solid Oxide Fuel Cell Stack. Hydrogen 2023, 4, 917-931. https://doi.org/10.3390/hydrogen4040054
Sørensen RD, Berning T. A Computational Fluid Dynamics Analysis of Hydrogen Leakage and Nitrogen Purging of a Solid Oxide Fuel Cell Stack. Hydrogen. 2023; 4(4):917-931. https://doi.org/10.3390/hydrogen4040054
Chicago/Turabian StyleSørensen, Rasmus Dockweiler, and Torsten Berning. 2023. "A Computational Fluid Dynamics Analysis of Hydrogen Leakage and Nitrogen Purging of a Solid Oxide Fuel Cell Stack" Hydrogen 4, no. 4: 917-931. https://doi.org/10.3390/hydrogen4040054
APA StyleSørensen, R. D., & Berning, T. (2023). A Computational Fluid Dynamics Analysis of Hydrogen Leakage and Nitrogen Purging of a Solid Oxide Fuel Cell Stack. Hydrogen, 4(4), 917-931. https://doi.org/10.3390/hydrogen4040054





