Hydrogen Gas Inhalation Prevents Erythrocyte Aggregation and Promotes Leukocyte Phagocytosis Together with Increases in Serum Antioxidant Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. The Device for Hydrogen Gas Inhalation
2.2. The Participants in the Trial of Hydrogen gas Inhalation with Informed Consents
2.3. Antioxidant Capacity in Serum Determined by the ORAC Assay
2.4. Effects of Hydrogen gas Inhalation on Rheological/Morphological Statuses of Erythrocytes and Phagocytotic Activity of Leukocytes
2.5. Statistical Analysis
3. Results
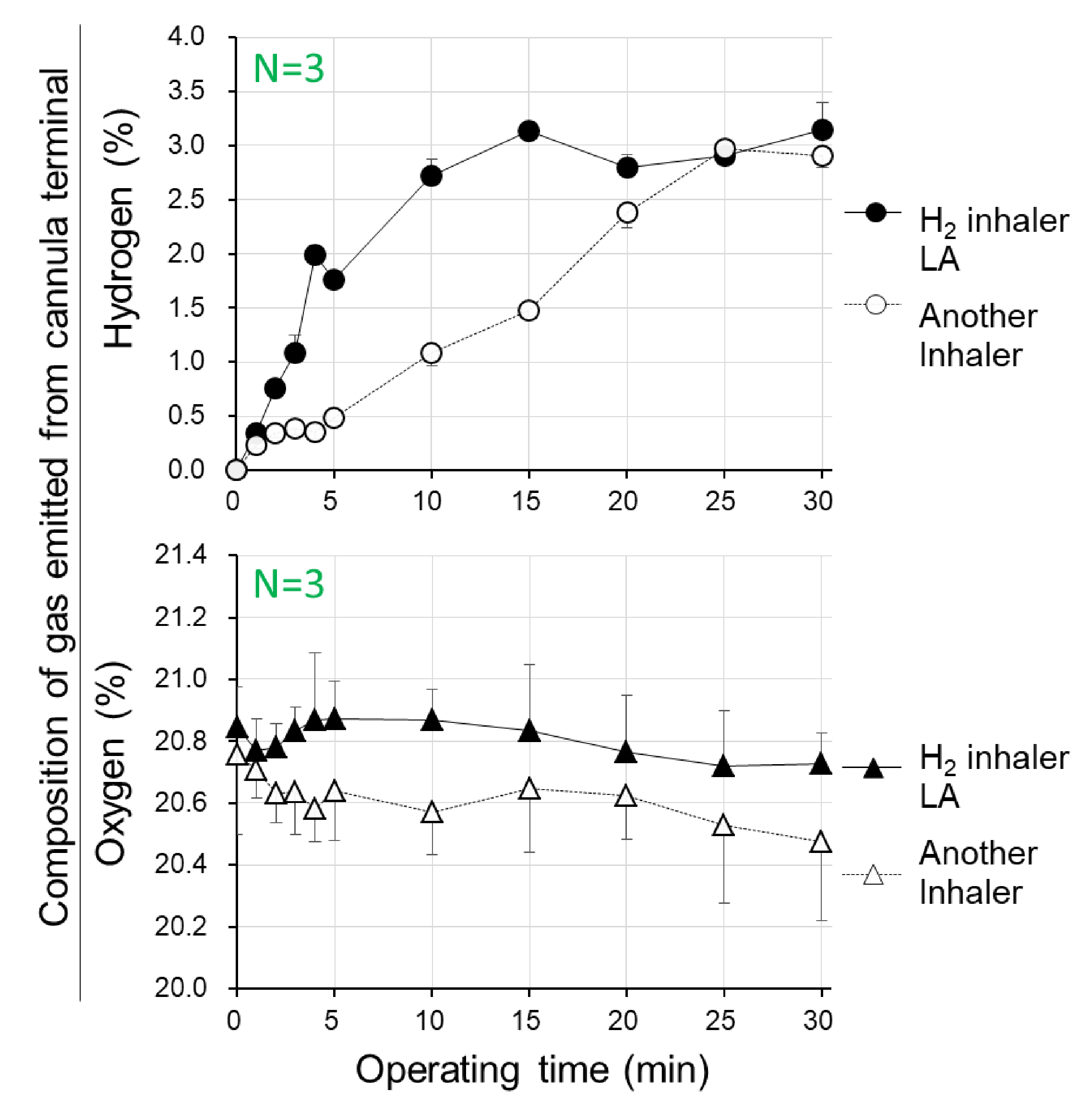
3.1. Concentrations of Hydrogen/Oxygen Gas being Emitted from Cannula Terminal of the Hydrogen Inhaler
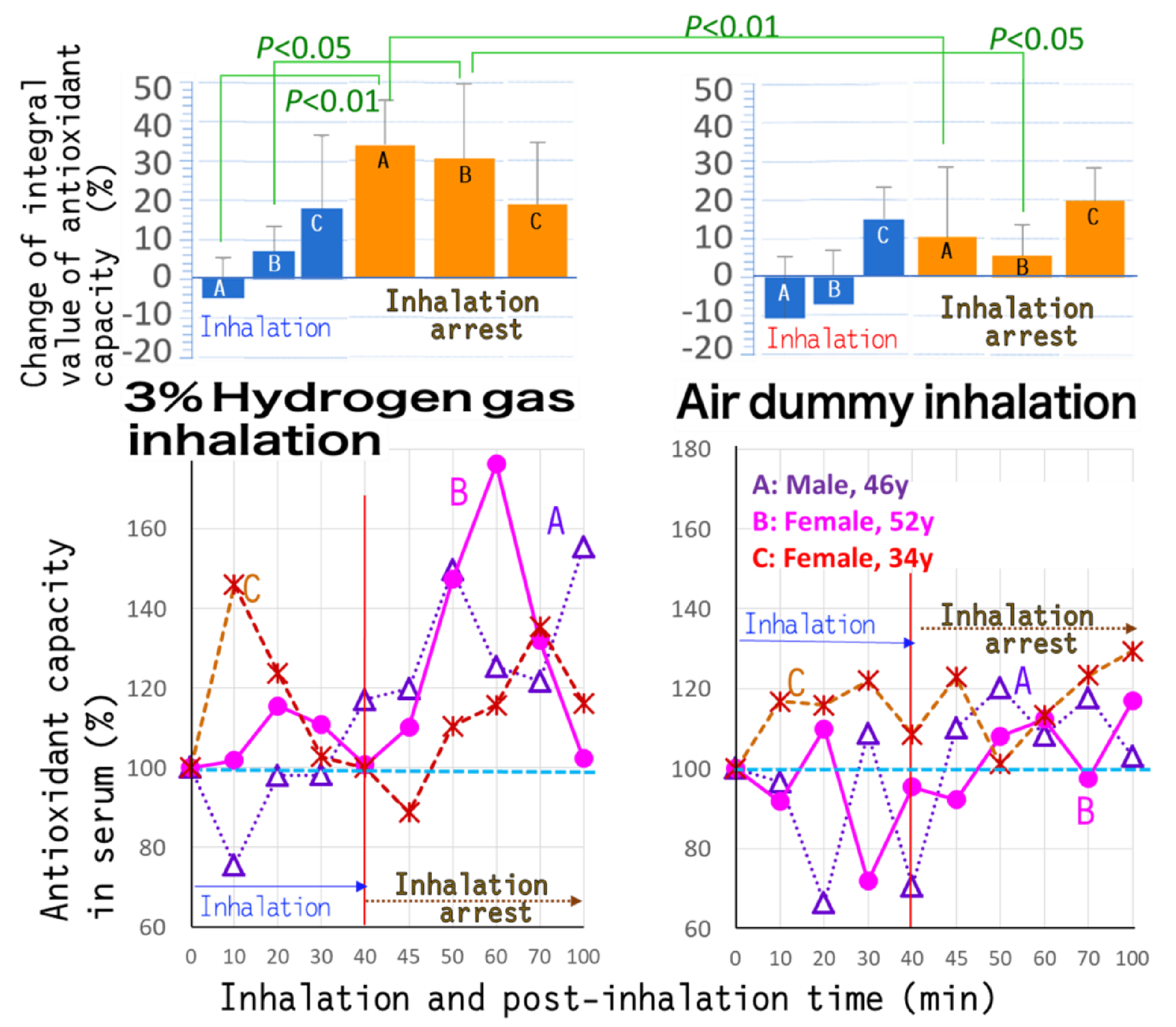
3.2. Antioxidant Capacity in Serum Determined by ORAC Assay
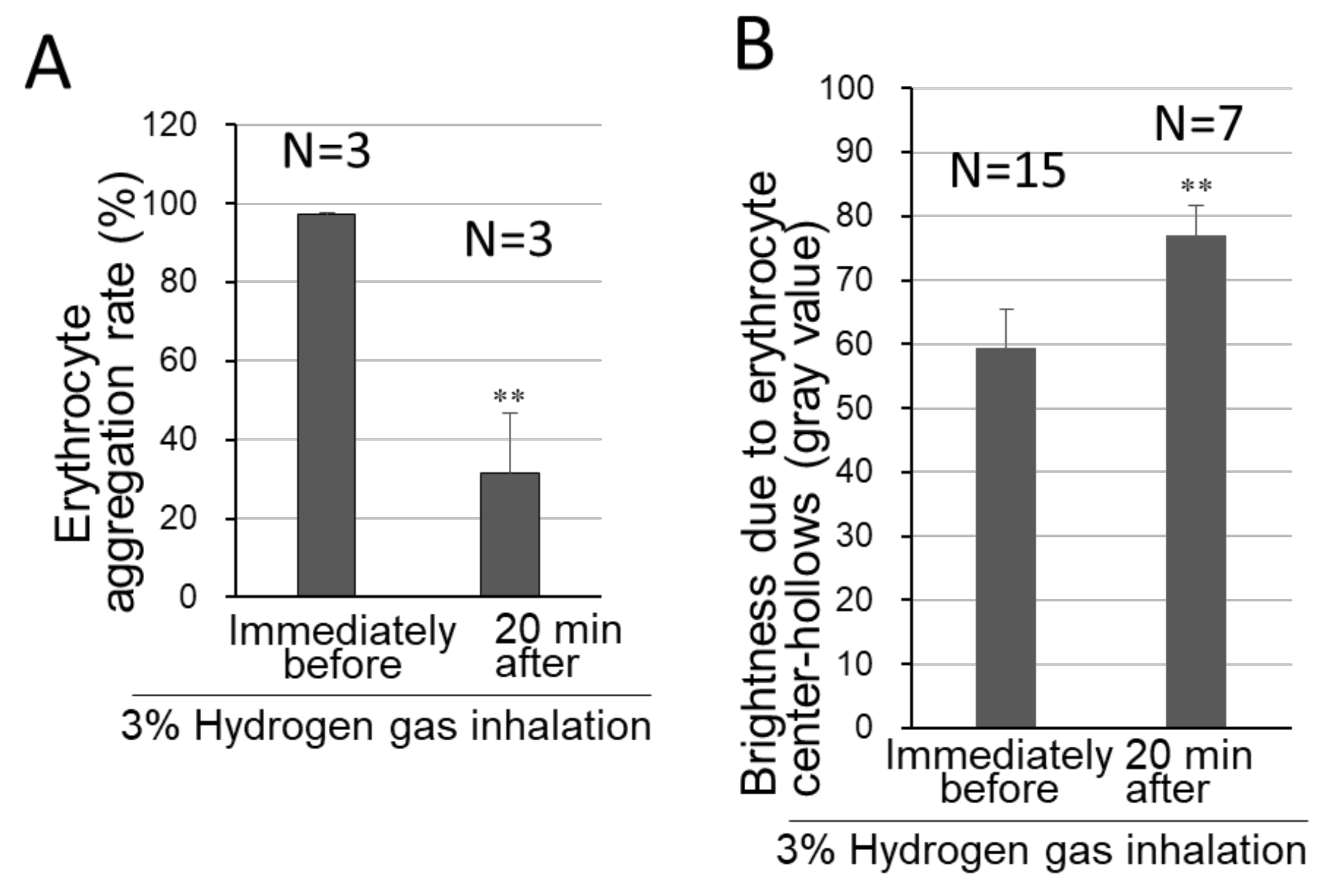
3.3. Preventive Effects of Hydrogen Inhalation against Erythrocyte Aggregation
3.4. Preservative Effects of Hydrogen Inhalation on the Cell-Center Hollow-Shaped Morphology of Erythrocytes
3.5. Activation to Phagocytosis of Leukocytes by Hydrogen Gas Inhalation
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, Q.; Zennadi, R. Oxidative Stress and Thrombosis during Aging: The Roles of Oxidative Stress in RBCs in Venous Thrombosis. Int. J. Mol. Sci. 2020, 21, 4259. [Google Scholar] [CrossRef] [PubMed]
- Dahlgren, C.; Karlsson, A.; Bylund, J. Measurement of respiratory burst products generated by professional phagocytes. Methods Mol. Biol. 2007, 412, 349–363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alvarado, C.; Alvarez, P.; Puerto, M.; Gausserès, N.; Jiménez, L.; De la Fuente, M. Dietary supplementation with antioxidants improves functions and decreases oxidative stress of leukocytes from prematurely aging mice. Nutrition 2006, 22, 767–777. [Google Scholar] [CrossRef]
- Baskurt, O.K.; Temiz, A.; Meiselman, H.J. Effect of superoxide anions on red blood cell rheologic properties. Free Radic. Biol. Med. 1998, 24, 102–110. [Google Scholar] [CrossRef]
- Kato, S.; Takada, Y.; Miwa, N. Heat-retention effects of hydrogen-rich water bath assessed by thermography for humans. J. Therm. Biol. 2021, 95, 102805. [Google Scholar] [CrossRef]
- Kato, S.; Hokama, R.; Okayasu, H.; Saitoh, Y.; Iwai, K.; Miwa, N. Colloidal platinum in hydrogen-rich water exhibits radical-scavenging activity and improves blood fluidity. J. Nanosci. Nanotechnol. 2012, 12, 4019–4027. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Kato, S.; Matsuoka, D.; Tanaka, H.; Miwa, N. Hydrogen water intake via tube-feeding for patients with pressure ulcer and its reconstructive effects on normal human skin cells in vitro. Med. Gas Res. 2013, 3, 20. [Google Scholar] [CrossRef] [Green Version]
- Iketani, M.; Ohsawa, I. Molecular Hydrogen as a Neuroprotective Agent. Curr Neuropharmacol. 2017, 15, 324–331. [Google Scholar] [CrossRef] [Green Version]
- Lazar, H.L. Molecular hydrogen: A novel therapy for the treatment of pulmonary hypertension. J. Thorac. Cardiovasc. Surg. 2015, 150, 654–655. [Google Scholar] [CrossRef]
- Kato, S.; Saitoh, Y.; Miwa, N. Inhibitions by hydrogen-occluding silica microcluster to melanogenesis in human pigment cells and tyrosinase reaction. J. Nanosci. Nanotechnol. 2013, 13, 52–59. [Google Scholar] [CrossRef]
- Kato, S.; Saitoh, Y.; Iwai, K.; Miwa, N. Hydrogen-rich electrolyzed warm water represses wrinkle formation against UVA ray together with type-I collagen production and oxidative-stress diminishment in fibroblasts and cell-injury prevention in keratinocytes. J. Photochem. Photobiol. B 2012, 106, 24–33. [Google Scholar] [CrossRef]
- Kato, S.; Saitoh, Y.; Miwa, N. Hydrogen-bubbled platinum-colloid suppresses human esophagus- or tongue-carcinoma cells with intracellular platinum-uptake and the diminished normal-cell mortality. Hum. Cell 2020, 33, 1294–1301. [Google Scholar] [CrossRef]
- Kumagai, K.; Toyooka, T.; Takeuchi, S.; Otani, N.; Wada, K.; Tomiyama, A.; Mori, K. Hydrogen gas inhalation improves delayed brain injury by alleviating early brain injury after experimental subarachnoid hemorrhage. Sci. Rep. 2020, 10, 12319. [Google Scholar] [CrossRef]
- Haam, S.; Lee, J.G.; Paik, H.C.; Park, M.S.; Lim, B.J. Hydrogen gas inhalation during ex vivo lung perfusion of donor lungs recovered after cardiac death. J. Heart Lung Transplant. 2018, 37, 1271–1278. [Google Scholar] [CrossRef]
- Chen, J.B.; Kong, X.F.; Qian, W.; Mu, F.; Lu, T.Y.; Lu, Y.Y.; Xu, K.C. Two weeks of hydrogen inhalation can significantly reverse adaptive and innate immune system senescence patients with advanced non-small cell lung cancer: A self-controlled study. Med. Gas Res. 2020, 10, 149–154. [Google Scholar] [CrossRef]
- Huang, P.; Wei, S.; Huang, W.; Wu, P.; Chen, S.; Tao, A.; Wang, H.; Liang, Z.; Chen, R.; Yan, J.; et al. Hydrogen gas inhalation enhances alveolar macrophage phagocytosis in an ovalbumin-induced asthma model. Int. Immunopharmacol. 2019, 74, 105646. [Google Scholar] [CrossRef]
- Matsumoto, N. Morphology of erythrocytes: Hematological examination for erythrocyte morphological abnormalities. Medicina 1989, 26, 1704–1705. [Google Scholar] [CrossRef]
- Christy, R.M.; Baskurt, O.K.; Gass, G.C.; Gray, A.B.; Marshall-Gradisnik, S.M. Erythrocyte aggregation and neutrophil function in an aging population. Gerontology 2010, 56, 175–180. [Google Scholar] [CrossRef]
- Simmonds, M.J.; Meiselman, H.J.; Baskurt, O.K. Blood rheology and aging. J. Geriatr. Cardiol. 2013, 10, 291–301. [Google Scholar] [CrossRef]
- Izumida, Y.; Seiyama, A.; Maeda, N. Erythrocyte aggregation: Bridging by macromolecules and electrostatic repulsion by sialic acid. Biochim. Biophys. Acta 1991, 1067, 221–226. [Google Scholar] [CrossRef]
- Baskurt, O.K.; Meiselman, H.J. Lessons from comparative hemorheology studies. Clin. Hemorheol. Microcirc. 2010, 45, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Pries, A.R.; Secomb, T.W.; Gaehtgens, P. Biophysical aspects of blood flow in the microvasculature. Cardiovasc. Res. 1996, 32, 654–667. [Google Scholar] [CrossRef]
- Machiedo, G.W.; Powell, R.J.; Rush, B.F., Jr.; Swislocki, N.I.; Dikdan, G. The incidence of decreased red blood cell deformability in sepsis and the association with oxygen free radical damage and multiple-system organ failure. Arch. Surg. 1989, 124, 1386–1389. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, M.J.; Meiselman, H.J.; Marshall-Gradisnik, S.M.; Pyne, M.; Kakanis, M.; Keane, J.; Brenu, E.; Christy, R.; Baskurt, O.K. Assessment of oxidant susceptibility of red blood cells in various species based on cell deformability. Biorheology 2011, 48, 293–304. [Google Scholar] [CrossRef]
- Brajovich, M.I.; Rucci, A.; Acosta, I.L.; Cotorruelo, C.; García Borrás, S.; Racca, L.; Biondi, C.; Racca, A. Effects of aging on antioxidant response and phagocytosis in senescent erythrocytes. Immunol. Investig. 2009, 38, 551–559. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takada, Y.; Miwa, N. Hydrogen Gas Inhalation Prevents Erythrocyte Aggregation and Promotes Leukocyte Phagocytosis Together with Increases in Serum Antioxidant Activity. Hydrogen 2022, 3, 72-82. https://doi.org/10.3390/hydrogen3010006
Takada Y, Miwa N. Hydrogen Gas Inhalation Prevents Erythrocyte Aggregation and Promotes Leukocyte Phagocytosis Together with Increases in Serum Antioxidant Activity. Hydrogen. 2022; 3(1):72-82. https://doi.org/10.3390/hydrogen3010006
Chicago/Turabian StyleTakada, Yuki, and Nobuhiko Miwa. 2022. "Hydrogen Gas Inhalation Prevents Erythrocyte Aggregation and Promotes Leukocyte Phagocytosis Together with Increases in Serum Antioxidant Activity" Hydrogen 3, no. 1: 72-82. https://doi.org/10.3390/hydrogen3010006
APA StyleTakada, Y., & Miwa, N. (2022). Hydrogen Gas Inhalation Prevents Erythrocyte Aggregation and Promotes Leukocyte Phagocytosis Together with Increases in Serum Antioxidant Activity. Hydrogen, 3(1), 72-82. https://doi.org/10.3390/hydrogen3010006





