Distribution of Five Aquatic Plants Native to South America and Invasive Elsewhere under Current Climate
Abstract
1. Introduction
2. Experimental Section
2.1. Study Species and Occurrence Data
2.2. Climate and Freshwater System Data
2.3. Background Domain
2.4. Species Distribution Models SDMs
3. Results
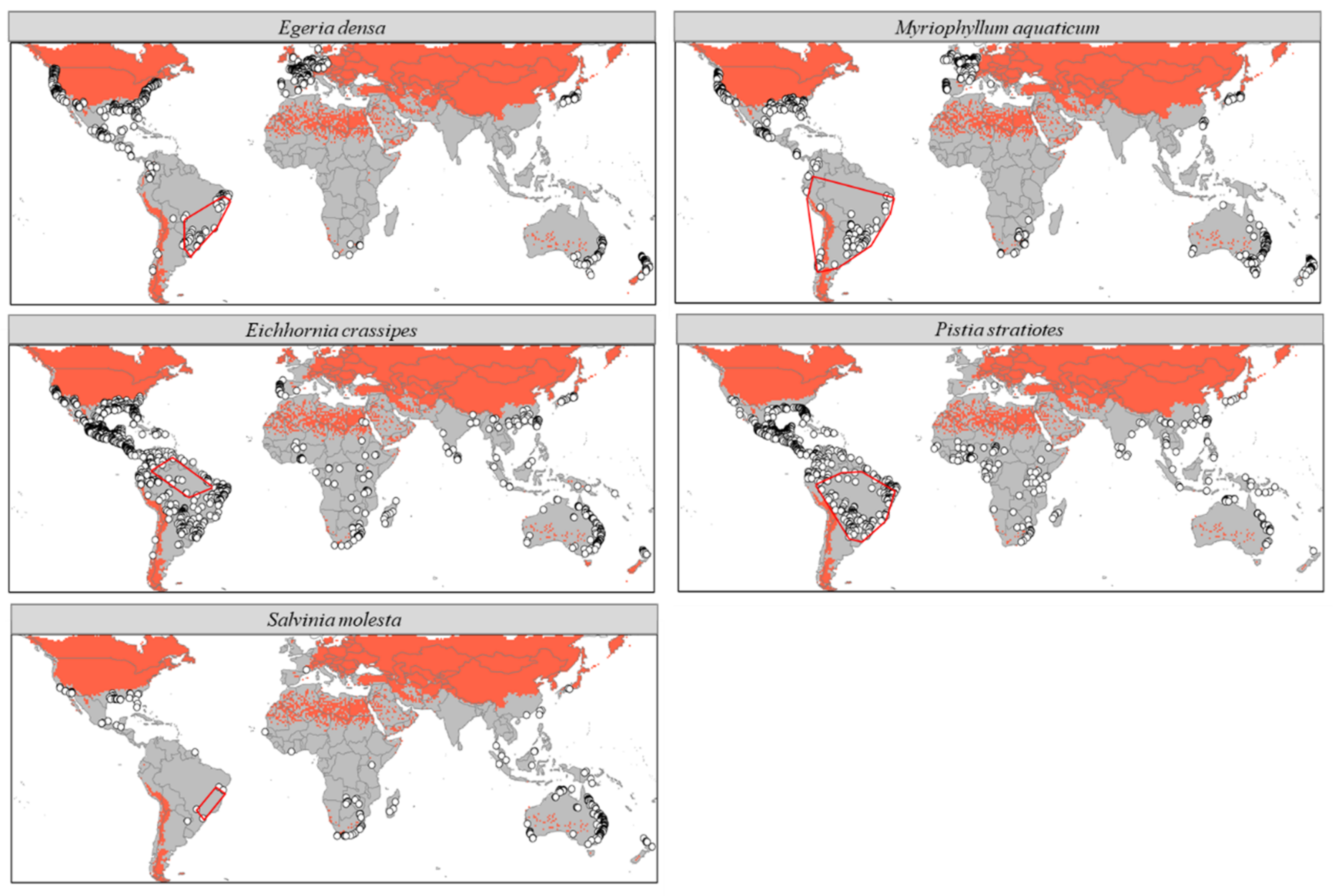
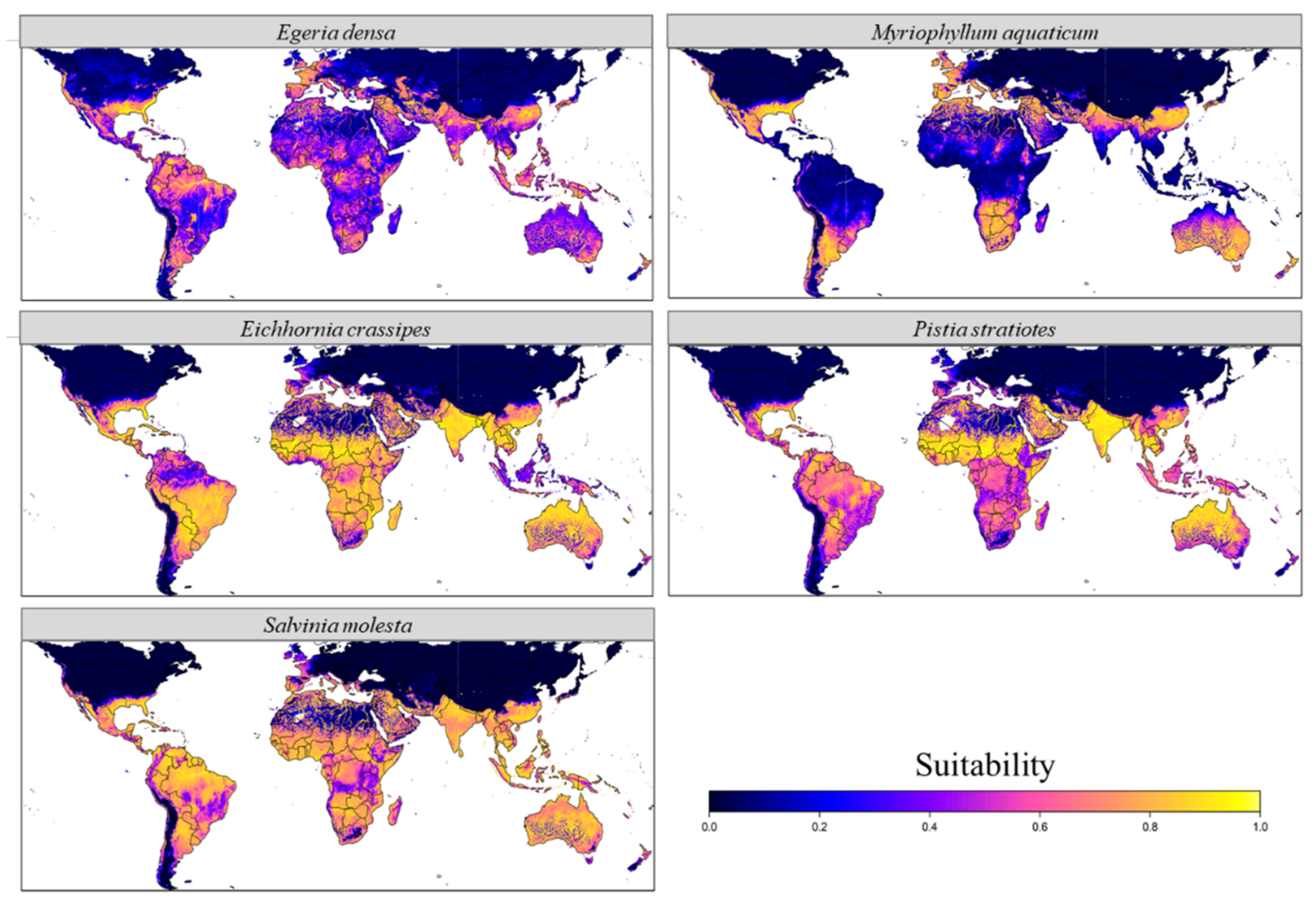
3.1. Current Potential Distribution
3.2. Model Evaluation
4. Discussion
4.1. Distribution in the Native Range
4.2. Distribution in the Non-Native Range
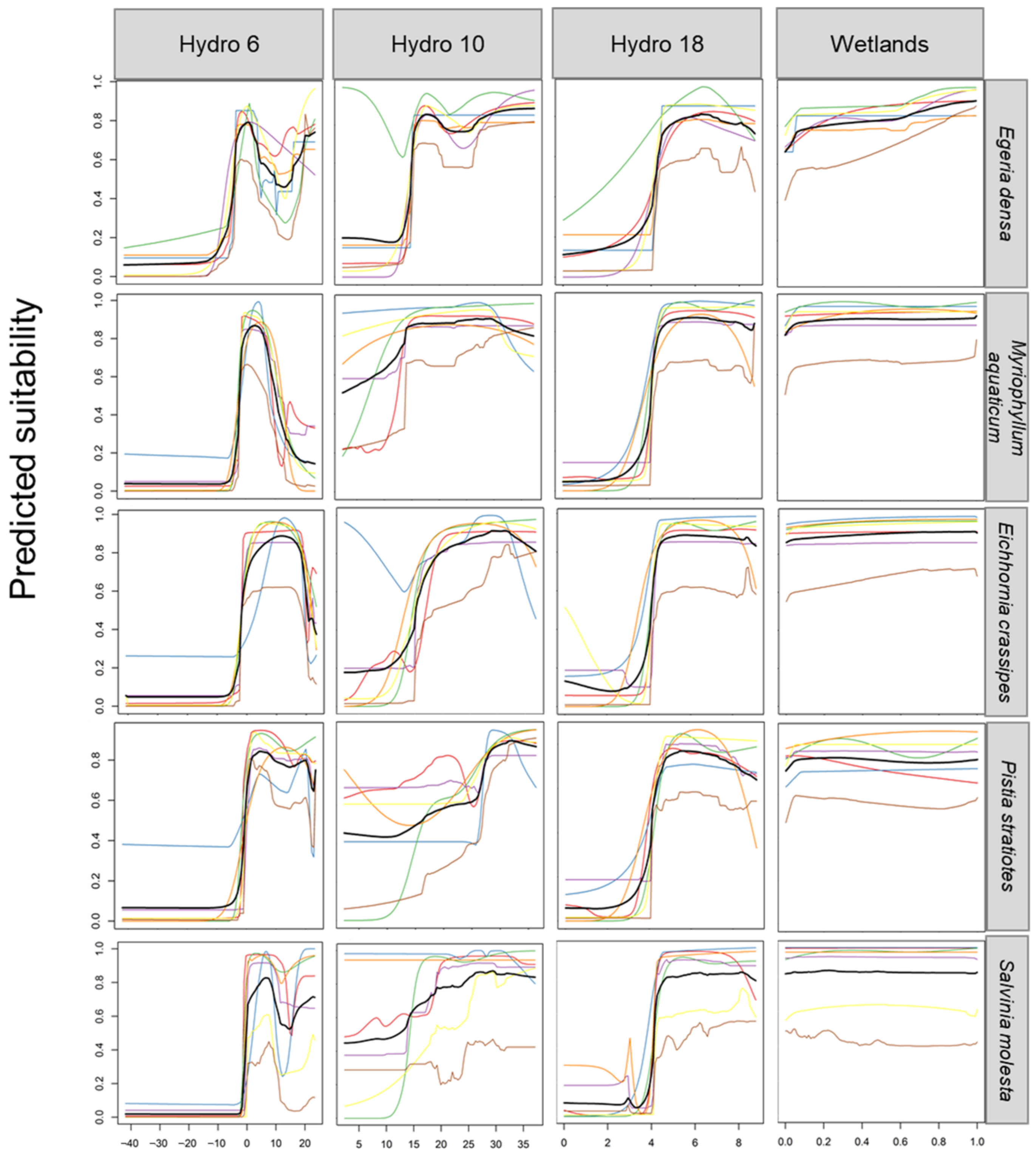
4.3. Climatic Limits to Occurrence
4.4. Implications for Climate Change
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Dudgeon, D.; Arthington, A.H.; Gessner, M.O.; Kawabata, Z.-I.; Knowler, D.J.; Lévêque, C.; Naiman, R.J.; Prieur-Richard, A.-H.; Soto, D.; Stiassny, M.L.J.; et al. Freshwater Biodiversity: Importance, Threats, Status and Conservation Challenges. Biol. Rev. 2006, 81, 163. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, P.G.; Wilby, R.L.; Battarbee, R.W.; Kernan, M.; Wade, A.J. A Review of the Potential Impacts of Climate Change on Surface Water Quality. Hydrolog. Sci. J. 2009, 54, 101–123. [Google Scholar] [CrossRef]
- Watts, G.; Battarbee, R.W.; Bloomfield, J.P.; Crossman, J.; Daccache, A.; Durance, I.; Elliott, J.A.; Garner, G.; Hannaford, J.; Hannah, D.M.; et al. Climate Change and Water in the UK—Past Changes and Future Prospects. Progr. Phys. Geogr. Earth Environ. 2015, 39, 6–28. [Google Scholar] [CrossRef]
- Jeppesen, E.; Kronvang, B.; Jørgensen, T.B.; Larsen, S.E.; Andersen, H.E.; Søndergaard, M.; Liboriussen, L.; Bjerring, R.; Johansson, L.S.; Trolle, D.; et al. Recent Climate-Induced Changes in Freshwaters in Denmark. In Climatic Change and Global Warming of Inland Waters; Goldman, C.R., Kumagai, M., Robarts, R.D., Eds.; John Wiley & Sons, Ltd: Chichester, UK, 2012; pp. 155–171. ISBN 978-1-118-47059-6. [Google Scholar]
- Jeppesen, E.; Meerhoff, M.; Davidson, T.A.; Trolle, D.; Søndergaard, M.; Lauridsen, T.L.; Beklioglu, M.; Brucet, S.; Volta, P.; González-Bergonzoni, I.; et al. Climate Change Impacts on Lakes: An Integrated Ecological Perspective Based on a Multi-Faceted Approach, with Special Focus on Shallow Lakes. J. Limnol. 2014, 73. [Google Scholar] [CrossRef]
- Parmesan, C. Ecological and Evolutionary Responses to Recent Climate Change. Annu. Rev. Ecol. Evol. Syst. 2006, 37, 637–669. [Google Scholar] [CrossRef]
- Rahel, F.J.; Olden, J.D. Assessing the Effects of Climate Change on Aquatic Invasive Species. Conserv. Biol. 2008, 22, 521–533. [Google Scholar] [CrossRef]
- Brunel, S. Pathway Analysis: Aquatic Plants Imported in 10 EPPO Countries. EPPO Bull. 2009, 39, 201–213. [Google Scholar] [CrossRef]
- Verlinden, M.; De Boeck, H.J.; Nijs, I. Climate Warming Alters Competition between Two Highly Invasive Alien Plant Species and Dominant Native Competitors. Weed Res. 2014, 54, 234–244. [Google Scholar] [CrossRef]
- Hoveka, L.N.; Bezeng, B.S.; Yessoufou, K.; Boatwright, J.S.; Van der Bank, M. Effects of Climate Change on the Future Distributions of the Top Five Freshwater Invasive Plants in South Africa. S. Afr. J. Bot. 2016, 102, 33–38. [Google Scholar] [CrossRef]
- Meis, S.; Thackeray, S.J.; Jones, I.D. Effects of Recent Climate Change on Phytoplankton Phenology in a Temperate Lake. Freshw. Biol. 2009, 54, 1888–1898. [Google Scholar] [CrossRef]
- Thackeray, S.J.; Sparks, T.H.; Frederiksen, M.; Burthe, S.; Bacon, P.J.; Bell, J.R.; Botham, M.S.; Brereton, T.M.; Bright, P.W.; Carvalho, L.; et al. Trophic Level Asynchrony in Rates of Phenological Change for Marine, Freshwater and Terrestrial Environments: Phenological Change Across Major Environments. Glob. Change Biol. 2010, 16, 3304–3313. [Google Scholar] [CrossRef]
- Andreu, J.; Vilà, M. Risk Analysis of Potential Invasive Plants in Spain. J. Nat. Conserv. 2010, 18, 34–44. [Google Scholar] [CrossRef]
- Adebayo, A.; Briski, E.; Kalaci, O.; Hernandez, M.; Ghabooli, S.; Beric, B.; Chan, F.; Zhan, A.; Fifield, E.; Leadley, T.; et al. Water Hyacinth (Eichhornia crassipes) and Water Lettuce (Pistia stratiotes) in the Great Lakes: Playing with Fire? Aquat. Invasions 2011, 6, 91–96. [Google Scholar] [CrossRef]
- Kriticos, D.J.; Brunel, S. Assessing and Managing the Current and Future Pest Risk from Water Hyacinth, (Eichhornia crassipes), an Invasive Aquatic Plant Threatening the Environment and Water Security. PLoS ONE 2016, 11, e0120054. [Google Scholar] [CrossRef] [PubMed]
- Brundu, G.; Stinca, A.; Angius, L.; Bonanomi, G.; Celesti-Grapow, L.; D’Auria, G.; Griffo, R.; Migliozzi, A.; Motti, R.; Spigno, P. Pistia stratiotes L. and Eichhornia crassipes (Mart.) Solms.: Emerging Invasive Alien Hydrophytes in Campania and Sardinia (Italy). EPPO Bull. 2012, 42, 568–579. [Google Scholar] [CrossRef]
- EPPO Pest Risk Analysis for Salvinia Molesta. 2017. Available online: https://pra.eppo.int/pra/066665df-eff1-4804-9819-13dc5f6c47b4 (accessed on 13 February 2018).
- Millane, M.; Caffrey, J. Risk Assessment of Egeria Densa. 2014. Available online: http://nonnativespecies.ie/wp-content/uploads/2014/03/Egeria-densa-Large-flowered-Waterweed1.pdf (accessed on 15 June 2014).
- Yarrow, M.; Marín, V.H.; Finlayson, M.; Tironi, A.; Delgado, L.E.; Fischer, F. The Ecology of Egeria densa Planchón (Liliopsida: Alismatales): A Wetland Ecosystem Engineer? Rev. Chil. Hist. Nat. 2009, 82. [Google Scholar] [CrossRef]
- Hussner, A. Alien Aquatic Plant Species in European Countries: Alien Aquatic Plant Species in European Countries. Weed Res. 2012, 52, 297–306. [Google Scholar] [CrossRef]
- Thouvenot, L.; Haury, J.; Thiebaut, G. A Success Story: Water Primroses, Aquatic Plant Pests: Water Primroses, Aquatic Plant Pests. Aquat. Conserv. Mar. Freshw. Ecosyst. 2013. [Google Scholar] [CrossRef]
- Brundu, G. Plant Invaders in European and Mediterranean Inland Waters: Profiles, Distribution, and Threats. Hydrobiologia 2015, 746, 61–79. [Google Scholar] [CrossRef]
- Ficetola, G.F.; Thuiller, W.; Miaud, C. Prediction and Validation of the Potential Global Distribution of a Problematic Alien Invasive Species—The American Bullfrog: Predicting the Success of Invasions. Divers. Distrib. 2007, 13, 476–485. [Google Scholar] [CrossRef]
- Broennimann, O.; Guisan, A. Predicting Current and Future Biological Invasions: Both Native and Invaded Ranges Matter. Biol. Lett. 2008, 4, 585–589. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Valverde, A.; Peterson, A.T.; Soberón, J.; Overton, J.; Aragón, P.; Lobo, J.M. Use of Niche Models in Invasive Species Risk Assessments. Biol. Invasions 2011, 13, 2785–2797. [Google Scholar] [CrossRef]
- Hulme, P.E. Weed Risk Assessment: A Way Forward or a Waste of Time? J. Appl. Ecol. 2012, 49, 10–19. [Google Scholar] [CrossRef]
- Petitpierre, B.; Kueffer, C.; Broennimann, O.; Randin, C.; Daehler, C.; Guisan, A. Climatic Niche Shifts Are Rare Among Terrestrial Plant Invaders. Science 2012, 335, 1344–1348. [Google Scholar] [CrossRef] [PubMed]
- Wellenreuther, M.; Larson, K.W.; Svensson, E.I. Climatic Niche Divergence or Conservatism? Environmental Niches and Range Limits in Ecologically Similar Damselflies. Ecology 2012, 93, 1353–1366. [Google Scholar] [CrossRef]
- Guisan, A.; Tingley, R.; Baumgartner, J.B.; Naujokaitis-Lewis, I.; Sutcliffe, P.R.; Tulloch, A.I.T.; Regan, T.J.; Brotons, L.; McDonald-Madden, E.; Mantyka-Pringle, C.; et al. Predicting Species Distributions for Conservation Decisions. Ecol. Lett. 2013, 16, 1424–1435. [Google Scholar] [CrossRef]
- Lozano, V.; Marzialetti, F.; Carranza, M.L.; Chapman, D.; Branquart, E.; Dološ, K.; Große-Stoltenberg, A.; Fiori, M.; Capece, P.; Brundu, G. Modelling Acacia Saligna Invasion in a Large Mediterranean Island Using PAB Factors: A Tool for Implementing the European Legislation on Invasive Species. Ecol. Indic. 2020, 116, 106516. [Google Scholar] [CrossRef]
- Pergl, J.; Brundu, G.; Harrower, C.A.; Cardoso, A.C.; Genovesi, P.; Katsanevakis, S.; Lozano, V.; Perglová, I.; Rabitsch, W.; Richards, G. Applying the Convention on Biological Diversity Pathway Classification to Alien Species in Europe. Neobiota 2020, 62, 333–363. [Google Scholar] [CrossRef]
- Pearman, P.B.; Guisan, A.; Broennimann, O.; Randin, C.F. Niche Dynamics in Space and Time. Trends Ecol. Evol. 2008, 23, 149–158. [Google Scholar] [CrossRef]
- Alexander, J.M.; Edwards, P.J. Limits to the Niche and Range Margins of Alien Species. Oikos 2010, 119, 1377–1386. [Google Scholar] [CrossRef]
- Espíndola, A.; Pellissier, L.; Maiorano, L.; Hordijk, W.; Guisan, A.; Alvarez, N. Predicting Present and Future Intra-Specific Genetic Structure through Niche Hindcasting across 24 Millennia: Hindcasting-Based Phylogeography. Ecol. Lett. 2012, 15, 649–657. [Google Scholar] [CrossRef] [PubMed]
- Hughes, A.C.; Satasook, C.; Bates, P.J.J.; Bumrungsri, S.; Jones, G. The Projected Effects of Climatic and Vegetation Changes on the Distribution and Diversity of Southeast Asian Bats. Glob. Change Biol. 2012, 18, 1854–1865. [Google Scholar] [CrossRef]
- Thuiller, W.; Richardson, D.M.; Pysek, P.; Midgley, G.F.; Hughes, G.O.; Rouget, M. Niche-Based Modelling as a Tool for Predicting the Risk of Alien Plant Invasions at a Global Scale. Glob. Change Biol. 2005, 11, 2234–2250. [Google Scholar] [CrossRef]
- Broennimann, O.; Treier, U.A.; Müller-Schärer, H.; Thuiller, W.; Peterson, A.T.; Guisan, A. Evidence of Climatic Niche Shift during Biological Invasion. Ecol. Lett. 2007, 10, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Guo, Z.; Ke, Z.; Wang, S.; Li, Y. Increasing Potential Risk of a Global Aquatic Invader in Europe in Contrast to Other Continents under Future Climate Change. PLoS ONE 2011, 6, e18429. [Google Scholar] [CrossRef]
- O’Donnell, J.; Gallagher, R.V.; Wilson, P.D.; Downey, P.O.; Hughes, L.; Leishman, M.R. Invasion Hotspots for Non-Native Plants in Australia under Current and Future Climates. Glob. Change Biol. 2012, 18, 617–629. [Google Scholar] [CrossRef]
- Stephens, P.A.; Mason, L.R.; Green, R.E.; Gregory, R.D.; Sauer, J.R.; Alison, J.; Aunins, A.; Brotons, L.; Butchart, S.H.; Campedelli, T. Consistent Response of Bird Populations to Climate Change on Two Continents. Science 2016, 352, 84–87. [Google Scholar] [CrossRef]
- Mandle, L.; Warren, D.L.; Hoffmann, M.H.; Peterson, A.T.; Schmitt, J.; von Wettberg, E.J. Conclusions about Niche Expansion in Introduced Impatiens Walleriana Populations Depend on Method of Analysis. PLoS ONE 2010, 5, e15297. [Google Scholar] [CrossRef]
- Broennimann, O.; Fitzpatrick, M.C.; Pearman, P.B.; Petitpierre, B.; Pellissier, L.; Yoccoz, N.G.; Thuiller, W.; Fortin, M.-J.; Randin, C.; Zimmermann, N.E.; et al. Measuring Ecological Niche Overlap from Occurrence and Spatial Environmental Data: Measuring Niche Overlap. Glob. Ecol. Biogeogr. 2012, 21, 481–497. [Google Scholar] [CrossRef]
- Luoto, M.; Virkkala, R.; Heikkinen, R.K. The Role of Land Cover in Bioclimatic Models Depends on Spatial Resolution. Glob. Ecol. Biogeogr. 2006, 16, 34–42. [Google Scholar] [CrossRef]
- Václavík, T.; Meentemeyer, R.K. Invasive Species Distribution Modeling (ISDM): Are Absence Data and Dispersal Constraints Needed to Predict Actual Distributions? Ecol. Model. 2009, 220, 3248–3258. [Google Scholar] [CrossRef]
- Guisan, A.; Thuiller, W. Predicting Species Distribution: Offering More than Simple Habitat Models. Ecol. Lett. 2005, 8, 993–1009. [Google Scholar] [CrossRef]
- Wilson, J.R.; Richardson, D.M.; Rouget, M.; Procheş, Ş.; Amis, M.A.; Henderson, L.; Thuiller, W. Residence Time and Potential Range: Crucial Considerations in Modelling Plant Invasions. Divers. Distrib. 2007, 13, 11–22. [Google Scholar] [CrossRef]
- Wiens, J.J.; Ackerly, D.D.; Allen, A.P.; Anacker, B.L.; Buckley, L.B.; Cornell, H.V.; Damschen, E.I.; Jonathan Davies, T.; Grytnes, J.-A.; Harrison, S.P.; et al. Niche Conservatism as an Emerging Principle in Ecology and Conservation Biology: Niche Conservatism, Ecology, and Conservation. Ecol. Lett. 2010, 13, 1310–1324. [Google Scholar] [CrossRef] [PubMed]
- Wiens, J.J.; Graham, C.H. Niche Conservatism: Integrating Evolution, Ecology, and Conservation Biology. Annu. Rev. Ecol. Evol. Syst. 2005, 36, 519–539. [Google Scholar] [CrossRef]
- Peterson, A.T.; Vieglais, D.A. Predicting Species Invasions Using Ecological Niche Modeling: New Approaches from Bioinformatics Attack a Pressing Problem: A New Approach to Ecological Niche Modeling, Based on New Tools Drawn from Biodiversity Informatics, Is Applied to the Challenge of Predicting Potential Species’ Invasions. BioScience 2001, 51, 363–371. [Google Scholar] [CrossRef]
- Bennett, J.R.; Cumming, B.F.; Ginn, B.K.; Smol, J.P. Broad-Scale Environmental Response and Niche Conservatism in Lacustrine Diatom Communities: Scale and Environment in Diatom Communities. Glob. Ecol. Biogeogr. 2010, 19, 724–732. [Google Scholar] [CrossRef]
- Kriticos, D.J.; Sutherst, R.W.; Brown, J.R.; Adkins, S.W.; Maywald, G.F. Climate Change and the Potential Distribution of an Invasive Alien Plant: Acacia Nilotica ssp. indica in Australia: Climate Change and Invasive Plants. J. Appl. Ecol. 2003, 40, 111–124. [Google Scholar] [CrossRef]
- Qin, Z.; DiTommaso, A.; Wu, R.S.; Huang, H.Y. Potential Distribution of Two Ambrosia Species in China under Projected Climate Change. Weed Res. 2014, 54, 520–531. [Google Scholar] [CrossRef]
- Thalmann, D.J.K.; Kikodze, D.; Khutsishvili, M.; Kharazishvili, D.; Guisan, A.; Broennimann, O.; Müller-Schärer, H. Areas of High Conservation Value in Georgia: Present and Future Threats by Invasive Alien Plants. Biol. Invasions 2015, 17, 1041–1054. [Google Scholar] [CrossRef]
- Heikkinen, R.; Leikola, N.; Fronzek, S.; Lampinen, R.; Toivonen, H. Predicting Distribution Patterns and Recent Northward Range Shift of an Invasive Aquatic Plant: Elodea Canadensis in Europe. BioRisk 2009, 2, 1–32. [Google Scholar] [CrossRef]
- Alahuhta, J.; Heino, J.; Luoto, M. Climate Change and the Future Distributions of Aquatic Macrophytes across Boreal Catchments. J. Biogeogr. 2011, 38, 383–393. [Google Scholar] [CrossRef]
- Lopes, A.; Wittmann, F.; Schöngart, J.; Householder, J.E.; Piedade, M.T.F. Modeling of Regional- and Local-Scale Distribution of the Genus Montrichardia Crueg. (Araceae). Hydrobiologia 2017, 789, 45–57. [Google Scholar] [CrossRef]
- Peterson, A.T.; Papes, M.; Kluza, D.A. Predicting the Potential Invasive Distributions of Four Alien Plant Species in North America. Weed Sci. 2003, 51, 863–868. [Google Scholar] [CrossRef]
- Gillard, M.; Thiébaut, G.; Deleu, C.; Leroy, B. Present and Future Distribution of Three Aquatic Plants Taxa across the World: Decrease in Native and Increase in Invasive Ranges. Biol. Invasions 2017, 19, 2159–2170. [Google Scholar] [CrossRef]
- Gallardo, B.; Aldridge, D.C. The ‘Dirty Dozen’: Socio-Economic Factors Amplify the Invasion Potential of 12 High-Risk Aquatic Invasive Species in Great Britain and Ireland. J. Appl. Ecol. 2013, 50, 757–766. [Google Scholar] [CrossRef]
- Kelly, R.; Leach, K.; Cameron, A.; Maggs, C.A.; Reid, N. Combining Global Climate and Regional Landscape Models to Improve Prediction of Invasion Risk. Divers. Distrib. 2014, 20, 884–894. [Google Scholar] [CrossRef]
- Koncki, N.G.; Aronson, M.F.J. Invasion Risk in a Warmer World: Modeling Range Expansion and Habitat Preferences of Three Nonnative Aquatic Invasive Plants. Invasive Plant Sci. Manag 2015, 8, 436–449. [Google Scholar] [CrossRef]
- Williams, P.A.; Champion, P.; Council, N.R. Biological Success and Weediness of Existing Terrestrial Pest Plants and Aquatic Weeds in Northland; Land Care Research New Zealand Ltd.: Nelson, New Zealand, 2008; Volume 2008, pp. 51–52. [Google Scholar]
- Coetzee, J.A.; Hill, M.P.; Ruiz-Téllez, T.; Starfinger, U.; Brunel, S. Monographs on Invasive Plants in Europe N° 2: Eichhornia crassipes (Mart.) Solms. Bot. Lett. 2017, 164, 303–326. [Google Scholar] [CrossRef]
- Wang, C.-J.; Wan, J.-Z.; Qu, H.; Zhang, Z.-X. Climatic Niche Shift of Aquatic Plant Invaders between Native and Invasive Ranges: A Test Using 10 Species across Different Biomes on a Global Scale. Knowl. Manag. Aquat. Ecosyst. 2017, 27. [Google Scholar] [CrossRef]
- Lozano, V.; Brundu, G. Prioritisation of Aquatic Invasive Alien Plants in South America with the US Aquatic Weed Risk Assessment. Hydrobiologia 2018, 812, 115–130. [Google Scholar] [CrossRef]
- Brundu, G.; Armeli Minicante, S.; Barni, E.; Bolpagni, R.; Caddeo, A.; Celesti-Grapow, L.; Cogoni, A.; Galasso, G.; Iiriti, G.; Lazzaro, L.; et al. Managing Plant Invasions Using Legislation Tools: An Analysis of the National and Regional Regulations For Non-Native Plants in Italy. Ann. Bot. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Chamberlain, S.; Ram, K.; Hart, T. Spocc: R Interface to Many Species Occurrence Data Sources. R package version 1.1.0. 2020. Available online: https://CRAN.R-project.org/package=spocc (accessed on 31 July 2018).
- Lozano, V.; Chapman, D.; Brundu, G. Native and Non-Native Aquatic Plants of South America: Comparing and Integrating GBIF Records with Literature Data. Manag. Biol. Invasion 2017, 8, 443–454. [Google Scholar] [CrossRef]
- Jocou, A.I.; Fernández, C.; Gandullo, R. Macrófitas Acuáticas Vasculares Del Sistema de Drenaje Del Alto Valle de Río Negro, Patagonia (Argentina). Rev. Mus. Plata 2018, 3, 296–308. [Google Scholar] [CrossRef]
- Huaroto, B.D.W.R. Flora Vascular y Vegetación de los Humedales de Conococha, Ancash, Perú. Bachelor’s Thesis, Facultad de Ciencias Biológicas, Universidad Nacional Mayor De San Marcos, Lima, Peru, 2011. [Google Scholar]
- Fuentes, N.; Pauchard, A.; Sánchez, P.; Esquivel, J.; Marticorena, A. A New Comprehensive Database of Alien Plant Species in Chile Based on Herbarium Records. Biol. Invasions 2013, 15, 847–858. [Google Scholar] [CrossRef]
- Hussner, A. Long-Term Macrophyte Mapping Documents a Continuously Shift from Native to Non-Native Aquatic Plant Dominance in the Thermally Abnormal River Erft (North Rhine-Westphalia, Germany). Limnologica 2014, 48, 39–45. [Google Scholar] [CrossRef]
- Tanner, R.; Branquart, E.; Brundu, G.; Buholzer, S.; Chapman, D.; Ehret, P.; Fried, G.; Starfinger, U.; van Valkenburg, J. The Prioritisation of a Short List of Alien Plants for Risk Analysis within the Framework of the Regulation (EU) No. 1143/2014. Neobiota 2017, 35, 87–118. [Google Scholar] [CrossRef]
- Hussner, A.; Van De Weyer, K.; Gross, E.M.; Hilt, S. Comments on Increasing Number and Abundance of Non-Indigenous Aquatic Macrophyte Species in Germany: Non-Indigenous Aquatic Macrophytes in Germany. Weed Res. 2010, 50, 519–526. [Google Scholar] [CrossRef]
- EPPO Pest Risk Analysis for Pistia Stratiotes. 2017. Available online: https://pra.eppo.int/pra/675eccbf-c118-4132-a83f-29ac01a2ca1b (accessed on 13 February 2018).
- Domisch, S.; Amatulli, G.; Jetz, W. Near-Global Freshwater-Specific Environmental Variables for Biodiversity Analyses in 1 Km Resolution. Sci. Data 2015, 2, 150073. [Google Scholar] [CrossRef]
- Lehner, B.; Verdin, K.; Jarvis, A. New Global Hydrography Derived From Spaceborne Elevation Data. Eos Trans. 2008, 89, 93. [Google Scholar] [CrossRef]
- Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G.; Jarvis, A. Very High Resolution Interpolated Climate Surfaces for Global Land Areas. Int. J. Climatol. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
- Lehner, B.; Döll, P. Development and Validation of a Global Database of Lakes, Reservoirs and Wetlands. J. Hydrol. 2004, 296, 1–22. [Google Scholar] [CrossRef]
- Naimi, B.; Hamm, N.A.S.; Groen, T.A.; Skidmore, A.K.; Toxopeus, A.G. Where Is Positional Uncertainty a Problem for Species Distribution Modelling? Ecography 2014, 37, 191–203. [Google Scholar] [CrossRef]
- Babak, N. Uncertainty Analysis for Species Distribution Models. Package ‘Usdm’. 2017. Available online: https://CRAN.R-project.org/package=usdm (accessed on 25 June 2017).
- Chapman, D.; Pescott, O.L.; Roy, H.E.; Tanner, R. Improving Species Distribution Models for Invasive Non-native Species with Biologically Informed Pseudo-absence Selection. J. Biogeogr. 2019, 46, 1029–1040. [Google Scholar] [CrossRef]
- Barve, N.; Barve, V.; Jiménez-Valverde, A.; Lira-Noriega, A.; Maher, S.P.; Peterson, A.T.; Soberón, J.; Villalobos, F. The Crucial Role of the Accessible Area in Ecological Niche Modeling and Species Distribution Modeling. Ecol. Model. 2011, 222, 1810–1819. [Google Scholar] [CrossRef]
- Mainali, K.P.; Warren, D.L.; Dhileepan, K.; McConnachie, A.; Strathie, L.; Hassan, G.; Karki, D.; Shrestha, B.B.; Parmesan, C. Projecting Future Expansion of Invasive Species: Comparing and Improving Methodologies for Species Distribution Modeling. Glob. Change Biol. 2015, 21, 4464–4480. [Google Scholar] [CrossRef]
- Soberon, J.; Peterson, A.T. Interpretation of Models of Fundamental Ecological Niches and Species’ Distributional Areas. Biodiv. Inf. 2005, 2. [Google Scholar] [CrossRef]
- Owens, C.S.; Smart, R.M.; Stewart, R.M. Low Temperature Limits of Giant Salvinia. J. Aquat. Plant Manag. 2004, 42, 91–94. [Google Scholar]
- Wersal, R.; Baker, E.; Larson, J.; Dettloff, K.; Fusaro, A.; Thayer, D.; Pfingsten, I. Myriophyllum aquaticum (Vell.) Verdc. US Geological Survey, Nonindigenous Aquatic Species Database. U.S. Geological Survey: Gainesville, FL, USA. 2019. Available online: https://nas.er.usgs.gov/queries/FactSheet.aspx?speciesID=235 (accessed on 5 April 2018).
- Phillips, S.J.; Dudík, M.; Elith, J.; Graham, C.H.; Lehmann, A.; Leathwick, J.; Ferrier, S. Sample Selection Bias and Presence-Only Distribution Models: Implications for Background and Pseudo-Absence Data. Ecol. Appl. 2009, 19, 181–197. [Google Scholar] [CrossRef]
- Thuiller, W.; Georges, D.; Engler, R.; Breiner, F. Biomod2: Ensemble Platform for Species Distribution Modeling. 2020. Available online: https://CRAN.R-project.org/package=biomod2 (accessed on 26 February 2018).
- Manel, S.; Williams, H.C.; Ormerod, S.J. Evaluating Presence-Absence Models in Ecology: The Need to Account for Prevalence: Presence-Absence Modelling. J. Appl. Ecol. 2002, 38, 921–931. [Google Scholar] [CrossRef]
- Gallien, L.; Münkemüller, T.; Albert, C.H.; Boulangeat, I.; Thuiller, W. Predicting Potential Distributions of Invasive Species: Where to Go from Here?: Predicting Potential Distributions of Invasive Species. Divers. Distrib. 2010, 16, 331–342. [Google Scholar] [CrossRef]
- Chapman, D.S.; Makra, L.; Albertini, R.; Bonini, M.; Páldy, A.; Rodinkova, V.; Šikoparija, B.; Weryszko-Chmielewska, E.; Bullock, J.M. Modelling the Introduction and Spread of Non-Native Species: International Trade and Climate Change Drive Ragweed Invasion. Glob. Change Biol. 2016, 22, 3067–3079. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, C.; Miranda, N.A.F.; Cumming, G.S. The Role of Waterbirds in the Dispersal of Aquatic Alien and Invasive Species. Divers. Distrib. 2015, 21, 744–754. [Google Scholar] [CrossRef]
- Clausen, P.; Nolet, B.A.; Fox, A.D.; Klaassen, M. Long-Distance Endozoochorous Dispersal of Submerged Macrophyte Seeds by Migratory Waterbirds in Northern Europe—A Critical Review of Possibilities and Limitations. Acta Oecol. 2002, 23, 191–203. [Google Scholar] [CrossRef]
- Hossain, K.; Yadav, S.; Quaik, S.; Pant, G.; Maruthi, A.Y.; Ismail, N. Vulnerabilities of Macrophytes Distribution Due to Climate Change. Theor. Appl. Climatol. 2017, 129, 1123–1132. [Google Scholar] [CrossRef]
- Téllez, T.R.; López, E.; Granado, G.; Pérez, E.; López, R.; Guzmán, J. The Water Hyacinth, Eichhornia Crassipes: An Invasive Plant in the Guadiana River Basin (Spain). Aquat. Invasions 2008, 3, 42–53. [Google Scholar] [CrossRef]
- Brundu, G.; Azzella, M.M.; Blasi, C.; Camarda, I.; Iberite, M.; Celesti-Grapow, L. The Silent Invasion of Eichhornia crassipes (Mart.) Solms. in Italy. Plant Biosyst. Int. J. Deal. Asp. Plant Biol. 2013, 147, 1120–1127. [Google Scholar] [CrossRef]
- Hussner, A.; Stiers, I.; Verhofstad, M.J.J.M.; Bakker, E.S.; Grutters, B.M.C.; Haury, J.; van Valkenburg, J.L.C.H.; Brundu, G.; Newman, J.; Clayton, J.S.; et al. Management and Control Methods of Invasive Alien Freshwater Aquatic Plants: A Review. Aquat. Bot. 2017, 136, 112–137. [Google Scholar] [CrossRef]
- Peterson, A.T.; Papeş, M.; Soberón, J. Rethinking Receiver Operating Characteristic Analysis Applications in Ecological Niche Modeling. Ecol. Model. 2008, 213, 63–72. [Google Scholar] [CrossRef]
- Zhu, G.; Gao, Y.; Zhu, L. Delimiting the Coastal Geographic Background to Predict Potential Distribution of Spartina alterniflora. Hydrobiologia 2013, 717, 177–187. [Google Scholar] [CrossRef]
- Feijoó, C.; García, M.E.; Momo, O.; Toja, J. Nutrient Absorption by the Submerged Macrophyte Egeria Dense Planch: Effect of Ammonium and Phosphorus Availability in the Water Column on Growth and Nutrient Uptake. Limnetica 2002, 2, 93–104. [Google Scholar]
- Hussner, A.; Meyer, C.; Busch, J. The Influence of Water Level and Nutrient Availability on Growth and Root System Development of Myriophyllum Aquaticum. Weed Res. 2009, 49, 73–80. [Google Scholar] [CrossRef]
- Jeppesen, E.; Kronvang, B.; Meerhoff, M.; Søndergaard, M.; Hansen, K.M.; Andersen, H.E.; Lauridsen, T.L.; Liboriussen, L.; Beklioglu, M.; Özen, A.; et al. Climate Change Effects on Runoff, Catchment Phosphorus Loading and Lake Ecological State, and Potential Adaptations. J. Environ. Qual. 2009, 38, 1930–1941. [Google Scholar] [CrossRef] [PubMed]
- Bornette, G.; Puijalon, S. Response of Aquatic Plants to Abiotic Factors: A Review. Aquat. Sci. 2011, 73, 1–14. [Google Scholar] [CrossRef]
- Heino, J.; Virkkala, R.; Toivonen, H. Climate Change and Freshwater Biodiversity: Detected Patterns, Future Trends and Adaptations in Northern Regions. Biol. Rev. 2009, 84, 39–54. [Google Scholar] [CrossRef] [PubMed]
- Kosten, S.; Kamarainen, A.; Jeppesen, E.; Van Nes, E.H.; Peeters, E.T.H.M.; Mazzeo, N.; Sass, L.; Hauxwell, J.; Hansel-Welch, N.; Lauridsen, T.L.; et al. Climate-Related Differences in the Dominance of Submerged Macrophytes in Shallow Lakes. Glob. Change Biol. 2009, 15, 2503–2517. [Google Scholar] [CrossRef]
- Yu, H.; Dong, X.; Yu, D.; Liu, C.; Fan, S. Effects of Eutrophication and Different Water Levels on Overwintering of Eichhornia Crassipes at the Northern Margin of Its Distribution in China. Front. Plant Sci. 2019, 10, 1261. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Xu, X.; Liu, S.; Song, S.; Chang, S.; Liu, C.; Yu, D. Impact of Eutrophication on Root Morphological and Topological Performance in Free-Floating Invasive and Native Plant Species. Hydrobiologia 2019, 836, 123–139. [Google Scholar] [CrossRef]
- Zettlemoyer, M.A.; Schultheis, E.H.; Lau, J.A. Phenology in a Warming World: Differences between Native and Non-native Plant Species. Ecol. Lett. 2019, 22, 1253–1263. [Google Scholar] [CrossRef]
- Gritti, E.S.; Smith, B.; Sykes, M.T. Vulnerability of Mediterranean Basin Ecosystems to Climate Change and Invasion by Exotic Plant Species. J. Biogeogr. 2006, 33, 145–157. [Google Scholar] [CrossRef]
- Valverde, P.; Serralheiro, R.; de Carvalho, M.; Maia, R.; Oliveira, B.; Ramos, V. Climate Change Impacts on Irrigated Agriculture in the Guadiana River Basin (Portugal). Agric. Water Manag. 2015, 152, 17–30. [Google Scholar] [CrossRef]
- Hayhoe, K.; Wake, C.P.; Huntington, T.G.; Luo, L.; Schwartz, M.D.; Sheffield, J.; Wood, E.; Anderson, B.; Bradbury, J.; DeGaetano, A.; et al. Past and Future Changes in Climate and Hydrological Indicators in the US Northeast. Clim. Dyn. 2007, 28, 381–407. [Google Scholar] [CrossRef]
- Rooney, N.; Kalff, J. Inter-Annual Variation in Submerged Macrophyte Community Biomass and Distribution: The Influence of Temperature and Lake Morphometry. Aquat. Bot. 2000, 68, 321–335. [Google Scholar] [CrossRef]
- Johnson, T.B.; Evans, D.O. Size-dependent Winter Mortality of Young-of-the-year White Perch: Climate Warming and Invasion of the Laurentian Great Lakes. Trans. Am. Fish. Soc. 1990, 119, 301–313. [Google Scholar] [CrossRef]
- Alahuhta, J.; Ecke, F.; Johnson, L.B.; Sass, L.; Heino, J. A Comparative Analysis Reveals Little Evidence for Niche Conservatism in Aquatic Macrophytes among Four Areas on Two Continents. Oikos 2017, 126, 136–148. [Google Scholar] [CrossRef]
- Ackerly, D.D. Community Assembly, Niche Conservatism, and Adaptive Evolution in Changing Environments. Int. J. Plant Sci. 2003, 164, S165–S184. [Google Scholar] [CrossRef]
| Species | Hydro 6 (Mean Minimum Temperature during Coldest Month) | Hydro 10 (Mean Temperature during Warmest Quarter) | Hydro 18 (Precipitation of Warmest Quarter) |
|---|---|---|---|
| Egeria densa | <−2 °C; stop growth at <6 degree but can survive at 1 degree under 15 cm of ice [62]. | >10 °C; growth is reported to commence [18]. It can survive in waters with temperatures of 3–35 °C [19]. | <4 mm; drought conditions could locally extirpate the species where water levels drop considerably [60]. |
| Eichhornia crassipes | <−3 °C; frost events limit its spread [63]. | >10 °C; limiting temperature for germination is 5–10 °C [63]. | <4 mm; germination occurs when substrates are exposed as water recedes and as dry substrates are moistened when water levels rise. Seeds survive in wet mud and are long-lived [63]. |
| Myriophyllum aquaticum | <−2 °C; growth and establishment may be curtailed subjected to icy or frosty conditions [18]. Tolerate freezing temperatures in California’s Bay [87]. | >10 °C; warm average temperature. Warmer temperatures enhance its capacity for establishment [87]. | <4 mm; precipitation driest month. |
| Pistia stratiotes | <0 °C; prolonged exposure to lethal frosts [72]. | <0 °C; below the minimum growth temperature. Consistent with the temperature inhibiting seed germination [75]. | <5 mm; seasonal drying out of waterbodies may reduce suitability [75]. |
| Salvinia molesta | <0 °C; prolonged exposure to lethal frosts [86]. Buds are killed when exposed for more than two hours to temperatures below −3 °C. | <10 °C; below the minimum growth temperature. | <5 mm; seasonal drying out of waterbodies may reduce suitability [17]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lozano, V. Distribution of Five Aquatic Plants Native to South America and Invasive Elsewhere under Current Climate. Ecologies 2021, 2, 27-42. https://doi.org/10.3390/ecologies2010003
Lozano V. Distribution of Five Aquatic Plants Native to South America and Invasive Elsewhere under Current Climate. Ecologies. 2021; 2(1):27-42. https://doi.org/10.3390/ecologies2010003
Chicago/Turabian StyleLozano, Vanessa. 2021. "Distribution of Five Aquatic Plants Native to South America and Invasive Elsewhere under Current Climate" Ecologies 2, no. 1: 27-42. https://doi.org/10.3390/ecologies2010003
APA StyleLozano, V. (2021). Distribution of Five Aquatic Plants Native to South America and Invasive Elsewhere under Current Climate. Ecologies, 2(1), 27-42. https://doi.org/10.3390/ecologies2010003





