Development of Highly Photoactive Mixed Metal Oxide (MMO) Based on the Thermal Decomposition of ZnAl-NO3-LDH
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of ZnAl-NO3 LDHs
2.2. Characterization
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lin, Y.; Hu, H.; Hu, Y.H. Role of ZnO morphology in its reduction and photocatalysis. Appl. Surf. Sci. 2020, 502, 144202. [Google Scholar] [CrossRef]
- Kitiyanan, A.; Ngamsinlapasathian, S.; Pavasupree, S.; Yoshikawa, S. The preparation and characterization of nanostructured TiO2–ZrO2 mixed oxide electrode for efficient dye-sensitized solar cells. J. Solid State Chem. 2005, 178, 1044–1048. [Google Scholar] [CrossRef]
- Fan, G.; Sun, W.; Wang, H.; Li, F. Visible-light-induced heterostructured Zn–Al–In mixed metal oxide nanocomposite photocatalysts derived from a single precursor. J. Chem. Eng. 2011, 174, 467–474. [Google Scholar] [CrossRef]
- Zahra, M.; Shariatinia, Z.; Jourshabani, M.; Mahmood, S.; Darvishi, R. ZnO photocatalyst revisited: Effective photocatalytic degradation of emerging contaminants using S-doped ZnO nanoparticles under visible light radiation. Ind. Eng. Chem. Res. 2020, 59, 15894–15911. [Google Scholar]
- Israel, W. Recent conceptual advances in the catalysis science of mixed metal oxide catalytic materials. Catal. Today 2005, 100, 79–94. [Google Scholar]
- Xin, H.; Li, Y.; Wang, H.; Zhang, Q. Controlled preparation of β-Bi2O3/Mg–Al mixed metal oxides composites with enhanced visible light photocatalytic performance. Res. Chem. Intermed. 2020, 46, 5009–5021. [Google Scholar]
- Zheng, J.; Li, W.; Tang, R.; Xiong, S.; Gong, D.; Deng, Y.; Zhou, Z.; Li, L.; Su, L.; Yang, L. Ultrafast photodegradation of nitenpyram by Ag/Ag3PO4/Zn–Al LDH composites activated by persulfate system: Removal efficiency, degradation pathway and reaction mechanism. Chemosphere 2022, 292, 133431. [Google Scholar] [CrossRef]
- Nasser, S.; Sayed, M. Stover ash-extracted mixed oxides surface-doped with Ni for photo-degradation of water organic pollutants. Int. J. Environ. Anal. 2023, 103, 8941–8956. [Google Scholar]
- Zheng, J.; Fan, C.; Li, X.; Yang, Q.; Wang, D.; Duan, A.; Ding, J. Enhanced photodegradation of tetracycline hydrochloride by hexameric AgBr/Zn-Al MMO S-scheme heterojunction photocatalysts: Low metal leaching, degradation mechanism and intermediates. J. Chem. Eng. 2022, 446, 137371. [Google Scholar] [CrossRef]
- Lorena, G.; Santos, G.; Eguiluz, K.; Salazar-Banda, G.; Lanza, M.; Saez, C.; Rodrigo, M. Towards a higher photostability of ZnO photo-electrocatalysts in the degradation of organics by using MMO substrates. Chemosphere 2021, 271, 129451. [Google Scholar]
- Parisa, E.; Wang, Q.; Razavi-Khosroshahi, H.; Fuji, M.; Ishihara, T.; Edalati, K. Photocatalytic hydrogen evolution on a high-entropy oxide. J. Mater. Chem. 2020, 8, 3814–3821. [Google Scholar]
- Parisa, E.; Shen, X.; Watanabe, M.; Ishihara, T.; Arita, M.; Fuji, M.; Edalati, K. High-entropy oxynitride as a low-bandgap and stable photocatalyst for hydrogen production. J. Mater. Chem. 2021, 9, 15076–15086. [Google Scholar]
- Iqbal, M.A.; Sun, L.; Fedel, M. Synthesis of novel cone-shaped CaAl-LDH directly on aluminum alloy by a facile urea hydrolysis method. SN Appl. Sci. 2019, 1, 1415. [Google Scholar] [CrossRef]
- Iqbal, M.A.; Asghar, H.; Iqbal, M.A.; Fedel, M. Sorption of As (V) from aqueous solution using in situ growth MgAl–NO3 layered double hydroxide thin film developed on AA6082. SN Appl. Sci. 2019, 1, 666. [Google Scholar] [CrossRef]
- Kumar, Y.D.; Uma, S.; Nagarajan, R. Microwave-assisted synthesis of ternary Li-M-Al LDHs (M = Mg, Co, Ni, Cu, Zn, and Cd) and examining their use in phenol oxidation. Appl. Clay Sci. 2022, 228, 106655. [Google Scholar]
- Yi, L.; Yu, T.; Cai, R.; Li, Y.; Yang, W.; Caro, J. One-pot synthesis of NiAl-CO3 LDH anti-corrosion coatings from CO2-saturated precursors. RSC Adv. 2015, 5, 29552. [Google Scholar]
- Guanhua, Z.; Zhang, X.; Meng, Y.; Pan, G.; Ni, Z.; Xia, S. Layered double hydroxides-based photocatalysts and visible-light driven photodegradation of organic pollutants: A review. J. Chem. Eng. 2020, 392, 123684. [Google Scholar]
- Abdul, R.; Ali, S.; Asif, M.; In, S.-I. Layered double hydroxide (LDH) based photocatalysts: An outstanding strategy for efficient photocatalytic CO2 conversion. Catalysts 2020, 10, 1185. [Google Scholar] [CrossRef]
- Hanane, B.; Da Silva, E.S.; Cherevan, A.S.; Chafik, T.; Faria, J.L.; Eder, D. Layered double hydroxide (LDH)-based materials: A mini-review on strategies to improve the performance for photocatalytic water splitting. J. Energy Chem. 2022, 64, 406–431. [Google Scholar]
- Xuanang, B.; Zhang, S.; Zhao, Y.; Shi, R.; Zhang, T. Layered double hydroxide-based photocatalytic materials toward renewable solar fuels production. InfoMat 2021, 3, 719–738. [Google Scholar]
- Jiménez-López; Leyva-Ramos, R.; Salazar-Rábago, J.; Jacobo-Azuara, A.; Aragón-Piña, A. Adsorption of selenium (iv) oxoanions on calcined layered double hydroxides of Mg-Al-CO3 from aqueous solution. Effect of calcination and reconstruction of lamellar structure. Environ. Nanotechnol. Monit. Manag. 2021, 16, 100580. [Google Scholar] [CrossRef]
- Bayu, W.; Kurniawati, P.; Purbaningtias, T.E.; Jauhari, M.H.; Yahya, A.; Tamyiz, M.; Fatimah, I.; Doong, R.-A. Assessing the effect of calcination on adsorption capability of Mg/Al layer double hydroxides (LDHs). MRX 2022, 9, 035505. [Google Scholar]
- Bin, Z.; Wang, Q.; Mo, L.; Jin, F.; Zhu, J.; Tang, M. Synthesis of Mg-Al LDH and its calcined form with natural materials for efficient Cr (VI) removal. J. Environ. Chem. Eng. 2022, 10, 108605. [Google Scholar]
- Iqbal, M.A.; Fedel, M. Ordering and disordering of in situ grown MgAl-layered double hydroxide and its effect on the structural and corrosion resistance properties. Int. J. Miner. Metall. 2019, 26, 1570. [Google Scholar] [CrossRef]
- Armaković, S.J.; Savanović, M.; Šiljegović, M.; Kisić, M.; Šćepanović, M.; Grujić-Brojčin, M.; Simić, N.; Gavanski, L.; Armaković, S. Self-Cleaning and Charge Transport Properties of Foils Coated with Acrylic Paint Containing TiO2 Nanoparticles. Inorganics 2024, 12, 35. [Google Scholar]
- Ong, C.B.; Yong Ng, L.; Mohammad, A.W. A review of ZnO nanoparticles as solar photocatalysts: Synthesis, mechanisms and applications. Renew. Sustain. Energy Rev. 2018, 81, 536–551. [Google Scholar] [CrossRef]
- Suárez-Quezada, M.G.; Romero-Ortiz, J.; Samaniego-Benítez, E.; Suárez, V.; Mantilla, A. H2 production by the water splitting reaction using photocatalysts derived from calcined ZnAl LDH. Fuel 2019, 240, 262. [Google Scholar] [CrossRef]
- Seftel, E.M.; Popovici, E.; Mertens, M.; De Witte, K.; Van Tendeloo, G.; Cool, P.; Vansant, E.F. Zn–Al layered double hydroxides: Synthesis, characterization and photocatalytic application. Microporous Mesoporous Mater. 2008, 113, 296. [Google Scholar] [CrossRef]
- Elhalil, A.; Elmoubarki, R.; Machrouhi, A.; Sadiq, M.; Abdennouri, M.; Qourzal, S.; Barka, N. Photocatalytic degradation of caffeine by ZnO-ZnAl2O4 nanoparticles derived from LDH structure. J. Environ. Chem. Eng. 2017, 5, 3719. [Google Scholar] [CrossRef]
- Zhe, Z.; Hua, Z.; Lang, J.; Song, Y.; Zhang, Q.; Han, Q.; Fan, H.; Gao, M.; Li, X.; Yang, J. Eco-friendly nanostructured Zn–Al layered double hydroxide photocatalysts with enhanced photocatalytic activity. CrystEngComm 2019, 21, 4607–4619. [Google Scholar]
- Ahmed, A.; Abidin Talib, Z.; Mohd, Z.B.H.; Zakaria, A. Improvement of the crystallinity and photocatalytic property of zinc oxide as calcination product of Zn–Al layered double hydroxide. J. Alloys Compd. 2012, 539, 154. [Google Scholar] [CrossRef]
- Thongam, D.; Gupta, J.; Kumar Sahu, N. Effect of induced defects on the properties of ZnO nanocrystals: Surfactant role and spectroscopic analysis. SN Appl. Sci. 2019, 1, 1030. [Google Scholar] [CrossRef]
- Prevot, V.; Forano, C.; Besse, J.P.; Abraham, F. Syntheses and thermal and chemical behaviors of tartrate and succinate intercalated Zn3Al and Zn2Cr layered double hydroxides. Inorg. Chem. 1998, 37, 4293. [Google Scholar] [CrossRef]
- Lagnamayee, M.; Parida, K. A review on the recent progress, challenges and perspective of layered double hydroxides as promising photocatalysts. J. Mater. Chem. 2016, 28, 10744. [Google Scholar]
- Ye, H.; Liu, S.; Yu, D.; Zhou, X.; Qin, L.; Lai, C.; Qin, F. Regeneration mechanism, modification strategy, and environment application of layered double hydroxides: Insights based on memory effect. Chem. Rev. 2022, 450, 214253. [Google Scholar] [CrossRef]
- Jiao, L.; Ding, P.; Zhu, Z.; Du, W.; Xu, X.; Hu, J.; Zhou, Y.; Zeng, H. Engineering self-reconstruction via flexible components in layered double hydroxides for superior-evolving performance. Small 2021, 17, 2101671. [Google Scholar]
- Dixit, H.; Tandon, N.; Cottenier, S.R.; Lamoen, D.; Partoens, B.; Waroquier, M. Electronic structure and band gap of zinc spinel oxides beyond LDA: ZnAl2O4, ZnGa2O4 and ZnIn2O4. New J. Phys. 2011, 13, 063002. [Google Scholar] [CrossRef]
- Vaiano, V.; Matarangolo, M.; Murcia, J.; Rojas, H.; Navío, J.; Hidalgo, M. Enhanced photocatalytic removal of phenol from aqueous solutions using ZnO modified with Ag. Appl. Catal. B 2018, 225, 197–206. [Google Scholar] [CrossRef]
- Fu, H.; Xu, T.; Zhu, S.; Zhu, Y. Photocorrosion inhibition and enhancement of photocatalytic activity for ZnO via hybridization with C60. Environ. Sci. Tech. 2008, 42, 8064–8069. [Google Scholar] [CrossRef]
- Subash, B.; Krishnakumar, B.; Swaminathan, M.; Shanthi, M. Highly efficient, solar active, and reusable photocatalyst: Zr-loaded Ag–ZnO for reactive red 120 dye degradation with synergistic effect and dye-sensitized mechanism. Langmuir 2013, 29, 939–949. [Google Scholar] [CrossRef]
- Zhang, H.; Zong, R.; Zhu, Y. Photocorrosion inhibition and photoactivity enhancement for zinc oxide via hybridization with monolayer polyaniline. J. Phys. Chem. C 2009, 113, 4605–4611. [Google Scholar] [CrossRef]
- Shukla, P.R.; Wang, S.; Ming Ang, H.; Tadé, M. Photocatalytic oxidation of phenolic compounds using zinc oxide and sulphate radicals under artificial solar light. Sep. Purif. Technol. 2010, 70, 338–344. [Google Scholar] [CrossRef]
- Salah, N.H.; Bouhelassaa, M.; Bekkouche, S.; Boultii, A. Study of photocatalytic degradation of phenol. Desalination 2004, 166, 347–354. [Google Scholar] [CrossRef]
- Hinda, L.; Houas, A.; Herrmann, J.-M. Photocatalytic degradation of polynitrophenols on various commercial suspended or deposited titania catalysts using artificial and solar light. Int. J. Photoenergy 2008, 2008, 497895. [Google Scholar]
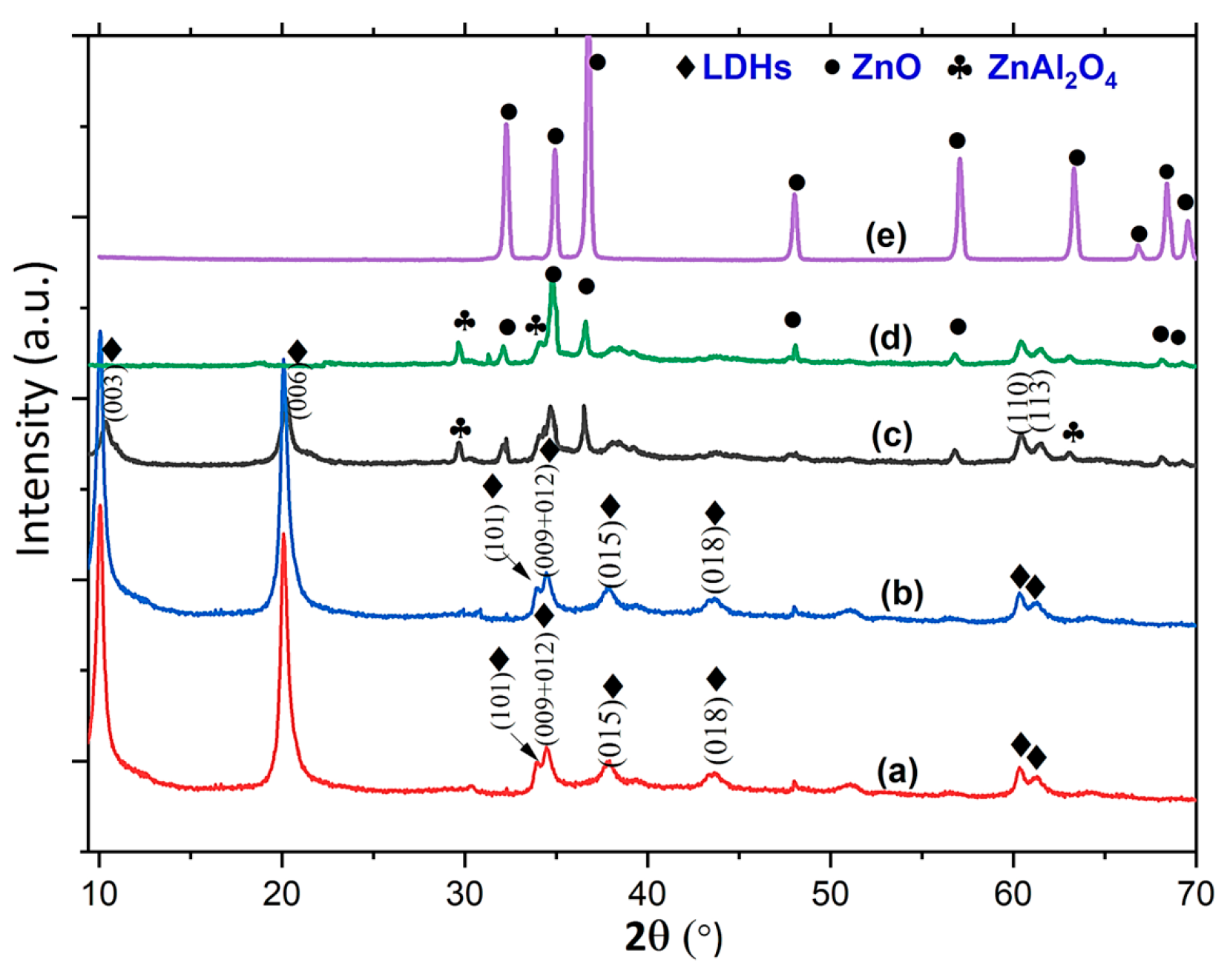

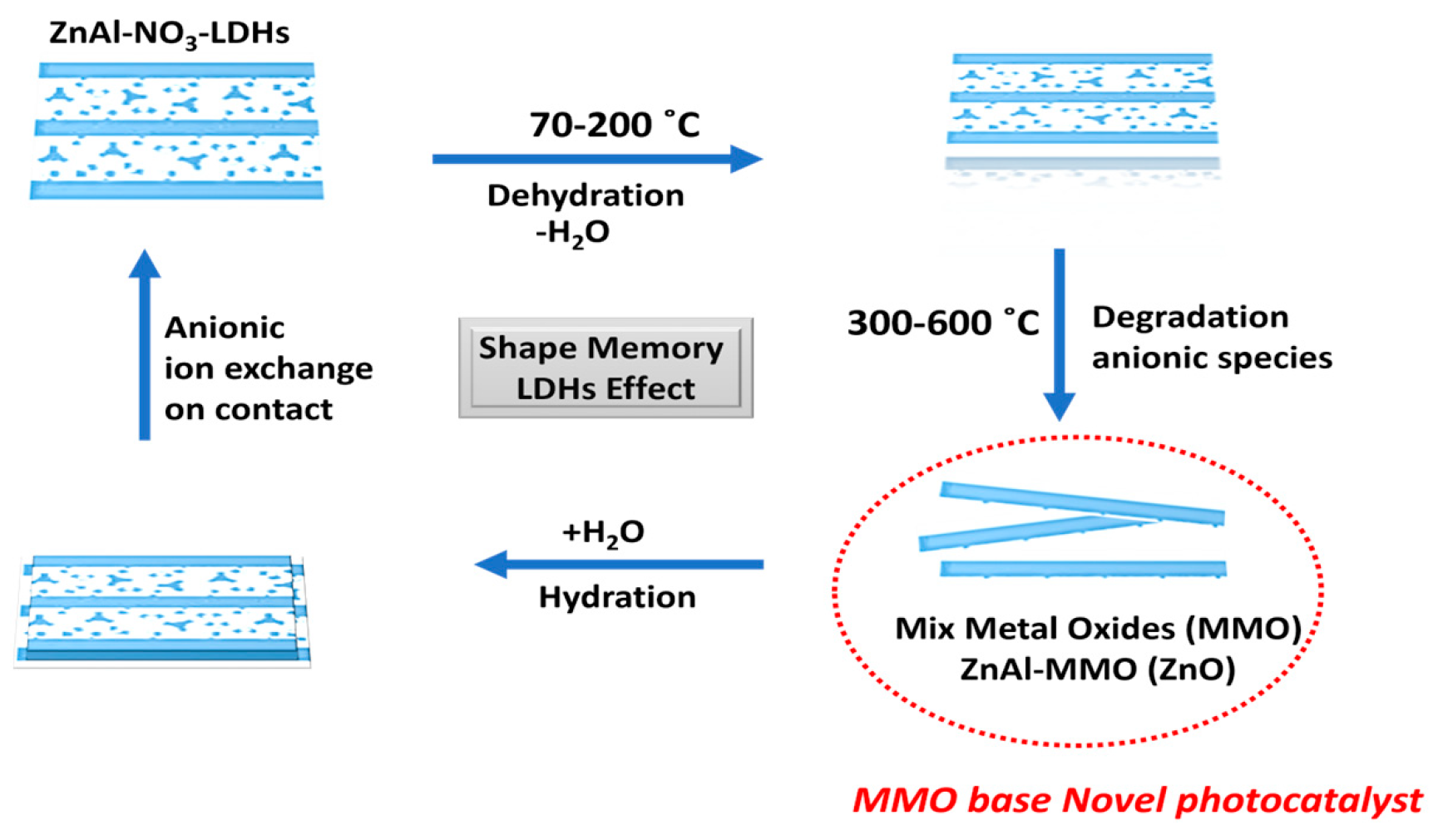
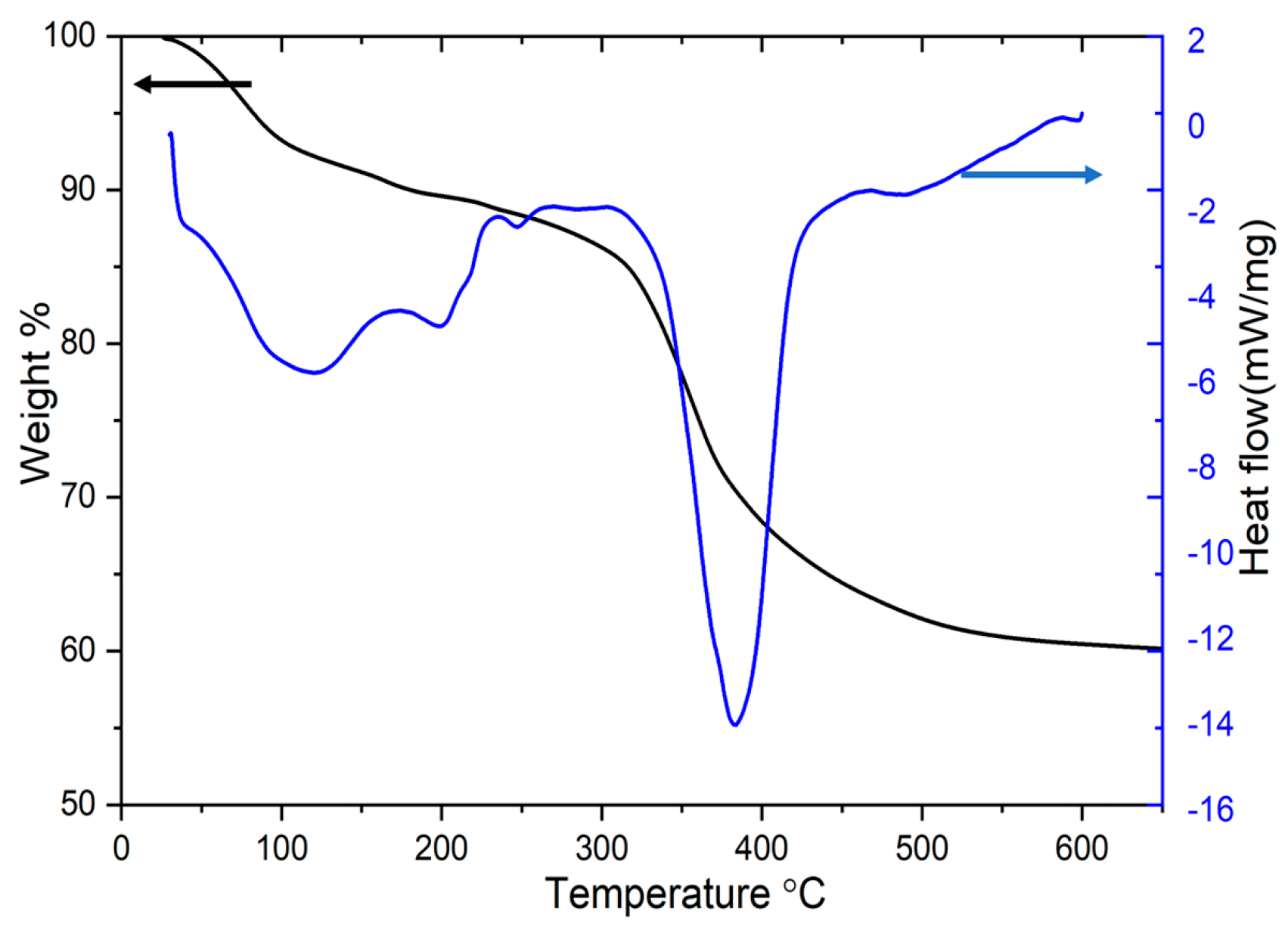

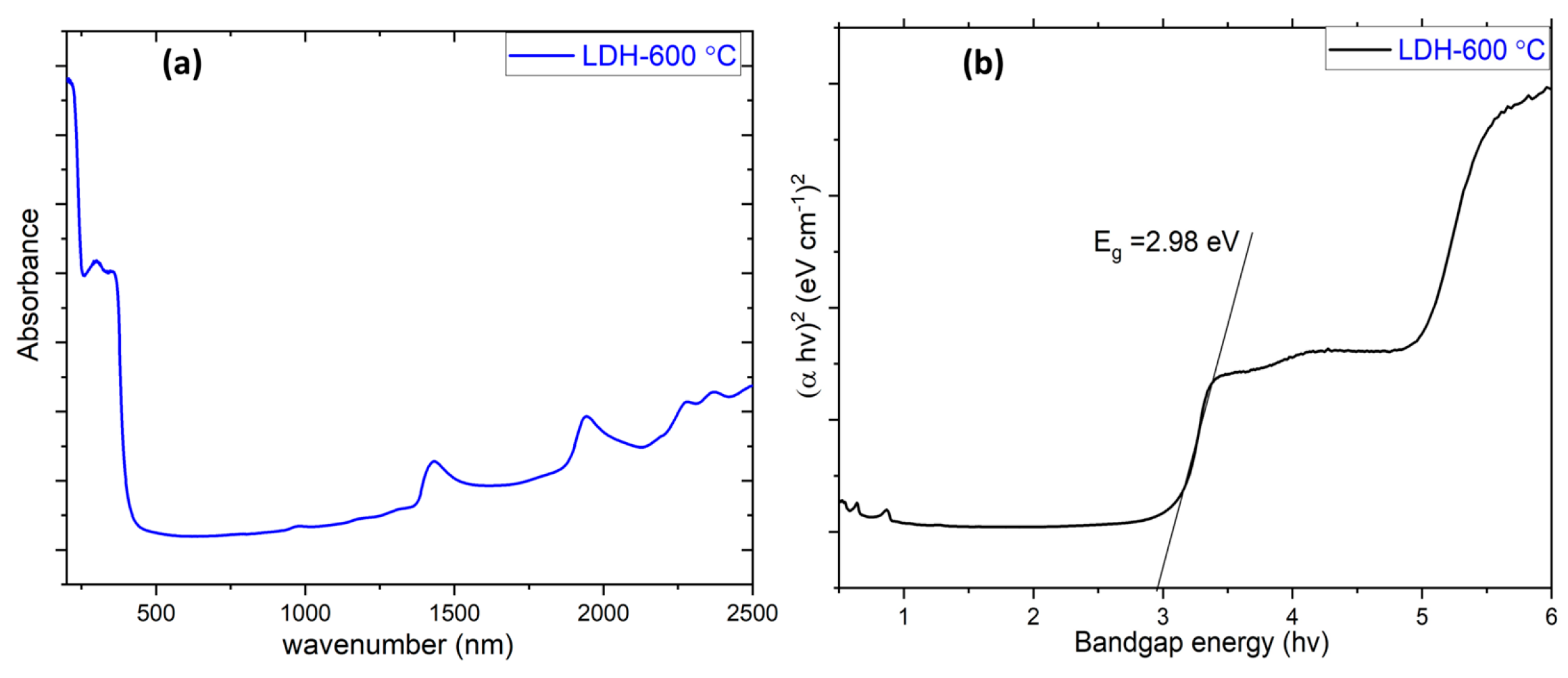

| Specimen | Lattice Parameter | Interlayer Distance | Crystallite Size, D/nm | |||
|---|---|---|---|---|---|---|
| a/nm | c/nm | d003/nm | d006/nm | d110/nm | ||
| ZnAl-LDH | 0.356 | 2.71 | 0.90 | 19.77 | 60.322 | 3.227 |
| LDH-400 °C | 0.355 | 2.64 | 0.88 | 20.01 | 60.322 | 3.147 |
| LDH-500 °C | 0.355 | 2.54 | 0.85 | 20.12 | 60.35 | 3.033 |
| Photon Energy (eV) | SBET (m2 g−1) | |
|---|---|---|
| ZnAl-LDH | 5.3 | 44.75 |
| LDH-400 °C | 4.9 | 48.09 |
| LDH-500 °C | 3.9 | 51.11 |
| LDH-600 °C | 2.98 | 71.86 |
| Material | Removal Efficiency | Time | K1 (min−1) | R2 | Pseudo-First Order Degradation Rate |
|---|---|---|---|---|---|
| (%) | (min) | (mM·min−1) | |||
| ZnAl-LDH | 20.19 | 120 | 0.0033 | 0.9694 | 0.000310 |
| LDH-400 °C | 47.63 | 120 | 0.0051 | 0.9939 | 0.000471 |
| LDH-500 °C | 59.17 | 120 | 0.0071 | 0.9951 | 0.000651 |
| LDH-600 °C | 80.12 | 120 | 0.0136 | 0.9995 | 0.001268 |
| Photocatalyst | C0 Phenol | Degradation Rate | Ref. |
|---|---|---|---|
| ZnO commercial, I = 11 W, (λmax = 254 nm) | 30 mg/L | 84% photodegradation of phenols occurs at 180 min | [41] |
| ZnO = 0.2 g/L, I = 125 W, (λmax = 270 nm) | 25 ppm | 95% degradation of phenol occurs in 130 min. | [42] |
| ZnO2 = 2 g/L, I = 125 W λmax = 366 nm) | 50 ppm | 99% degradation achieved in 360 min. | [43] |
| TiO2 = 0.5 g/L, I = 125 W, (λmax = 270 nm) | 30 mg/L | Complete degradation of phenols occurs at 300 min. | [44] |
| ZnO LDHs, I = 9 W, (λmax = 365 nm) | 30 mg/L | 83% photodegradation of phenols occurs at 120 min | This Work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asghar, H.; Maurino, V.; Iqbal, M.A. Development of Highly Photoactive Mixed Metal Oxide (MMO) Based on the Thermal Decomposition of ZnAl-NO3-LDH. Eng 2024, 5, 589-599. https://doi.org/10.3390/eng5020033
Asghar H, Maurino V, Iqbal MA. Development of Highly Photoactive Mixed Metal Oxide (MMO) Based on the Thermal Decomposition of ZnAl-NO3-LDH. Eng. 2024; 5(2):589-599. https://doi.org/10.3390/eng5020033
Chicago/Turabian StyleAsghar, Humaira, Valter Maurino, and Muhammad Ahsan Iqbal. 2024. "Development of Highly Photoactive Mixed Metal Oxide (MMO) Based on the Thermal Decomposition of ZnAl-NO3-LDH" Eng 5, no. 2: 589-599. https://doi.org/10.3390/eng5020033
APA StyleAsghar, H., Maurino, V., & Iqbal, M. A. (2024). Development of Highly Photoactive Mixed Metal Oxide (MMO) Based on the Thermal Decomposition of ZnAl-NO3-LDH. Eng, 5(2), 589-599. https://doi.org/10.3390/eng5020033







