Abstract
A 70-year-old man was admitted to the Emergency Department (ED) for marked asthenia and severe anemia. In addition, a high level of lipase was found. During hospitalization, a locally advanced gastric cancer was diagnosed, with endoscopic evidence of a large polyploid formation originating under the cardias that occupied most of the gastric lumen. A total body CT scan was performed before surgery; the tumor affected the posterior gastric wall, with tenacious infiltration of the pancreatic body. Therefore, we performed a total gastrectomy with esophageal jejunum anastomosis and reconstruction of intestinal continuity according to Roux, distal spleno-pancreatectomy, and cholecystectomy. At histology, a pancreatic acinar cell carcinoma (PACC) with full thickness infiltration of the gastric wall was diagnosed. Acinar cell carcinomas are highly aggressive neoplasms, and surgical resection, when feasible, is the treatment of choice regardless of size, also because the role of neoadjuvant or adjuvant chemo- or radiotherapy remains uncodified.
1. Introduction
Pancreatic acinar cell carcinoma (PACC) is a rare condition, representing less than 2% of exocrine pancreatic cancers, which usually show up late in adulthood [1]. Because of the difficulty in obtaining a radical surgical resection and the high rate of post-operative recurrences and metastases, PACC has a poor prognosis. Nevertheless, PACC is more indolent than pancreatic ductal adenocarcinoma and surgery is considered the elective treatment, regardless of size [2], especially at early stages; however, as PACC can show considerable dimensions at presentation, surgery could not be appropriate [3].
According to a recent article, although the reported prognosis of PACC is poor, it is better than pancreatic ductal adenocarcinoma in both resected and nonsurgical patients; the prognosis is poor and median overall survival (OS) in patients with metastatic disease, treated with chemotherapy, is between 12 and 19.6 months [4]. As for pancreatic adenocarcinoma, surgical resection appears to improve survival in the context of resectable disease [5].
A case of a 70-year-old Italian man with a preoperative diagnosis of locally advanced gastric cancer is described below; a total gastrectomy with esophageal jejunum anastomosis and reconstruction of intestinal continuity according to Roux, distal splenum-pancreatectomy, and cholecystectomy were performed. Microscopically, a pancreatic acinar cell carcinoma with full thickness infiltration of the gastric wall was diagnosed, so a “hidden wolf” was discovered, with a worse prognosis, while we were expecting a “lamb”.
2. Case Report
A 70-year-old man was admitted to the Emergency Department for marked asthenia and severe anemia without other symptoms. His primary medical comorbidities were Leriche syndrome, COPD, arterial hypertension, hypercholesterolemia, previous gastric ulcer, and benign prostatic hypertrophy; the previous gastric ulcer was an endoscopic finding, documented more than 20 years before, successfully treated with a proton pump inhibitor and without any subsequent recurrence.
Laboratory test reported a hemoglobin value of 5.9 g/dL (12.5 g/dL < n.v. < 17.2 g/dL), with AST 68 U/L (0 U/L < n.v. < 50 U/L), ALT 61 U/L (0 U/L < n.v. < 50 U/L), lipase 1204 U/L (0 U/L < n.v. < 67 U/L), and C-reactive protein (CRP) 3.51 mg/dL (0 mg/dL < n.v. < 0.5 mg/dL). The patient was subjected to multiple transfusions, due to the documented severe anemia.
During hospitalization, an upper gastrointestinal (UGI) endoscopy was performed, showing the presence of a protrudent polyploid formation of more than 6 cm type 0-Is, with origin under the cardias, occupying a great part of the gastric lumen, while the endoscopic appearance of the duodenum was normal; biopsies were performed.
The total body CT scan confirmed a mixed density gastric solid mass of at least 8 cm, hypertensive at all stages, as a possible expression of previous bleeding, without lymph node metastases or other signs of alterations (Figure 1, Figure 2, Figure 3, Figure 4, Figure 5 and Figure 6).
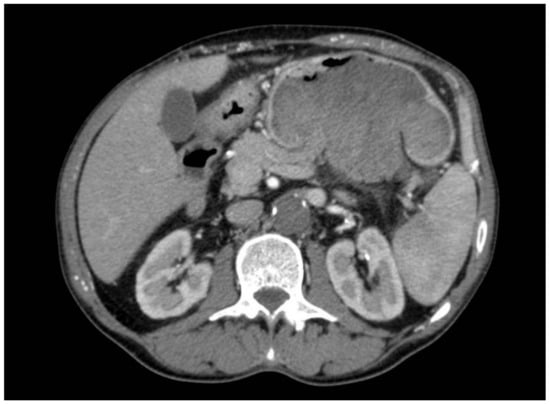
Figure 1.
Mixed density solid gastric mass, hyperdense in the arterial phase (possible previous bleeding), occupying the entire lumen of the stomach.
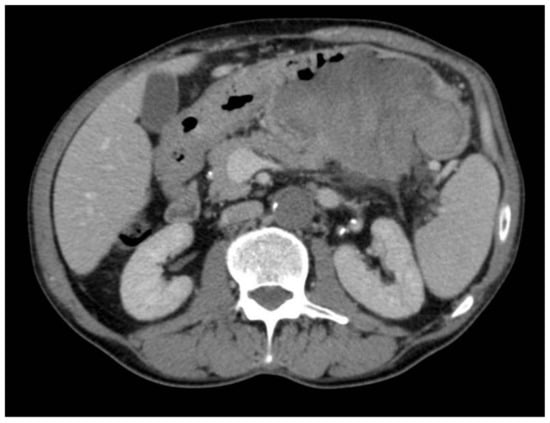
Figure 2.
Gastric lesion in the venous phase.
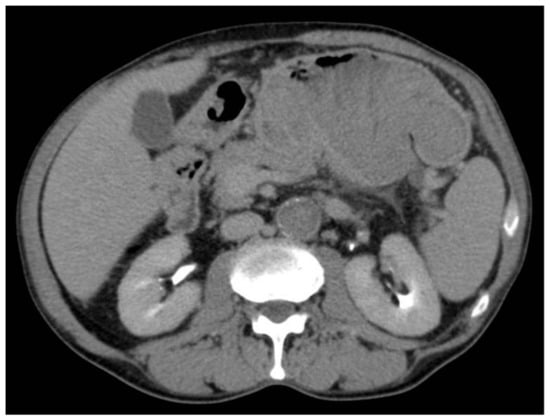
Figure 3.
Gastric lesion in the delayed phase.
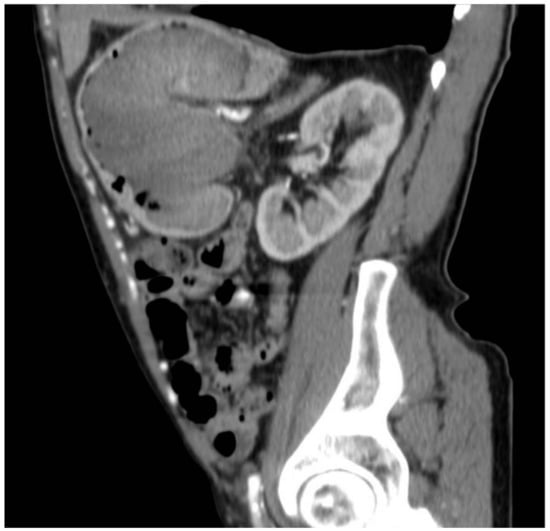
Figure 4.
Gastric lesion in the arterial phase in the sagittal section.
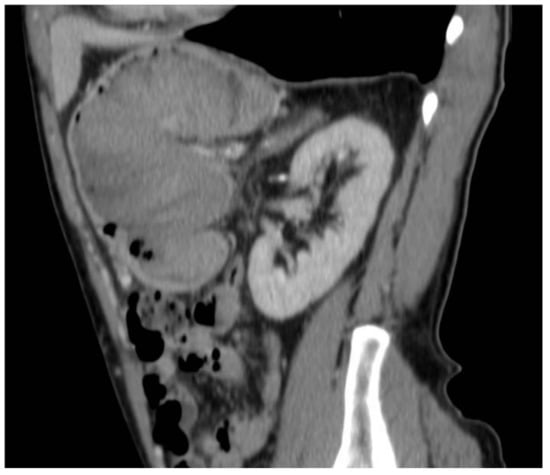
Figure 5.
Gastric lesion in the venous phase in the sagittal section.
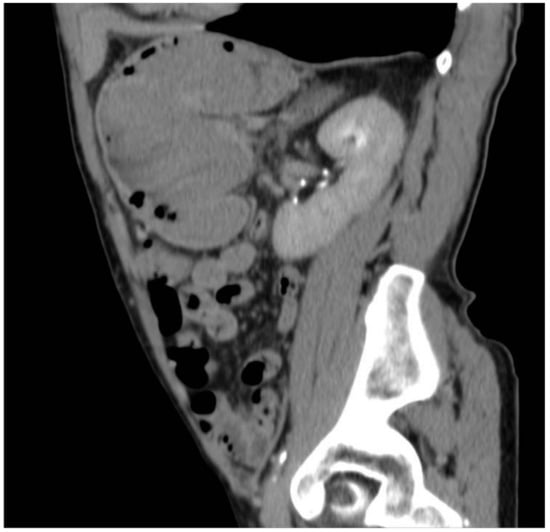
Figure 6.
Gastric lesion in the delayed phase in the sagittal section.
The biopsy showed a poorly differentiated carcinoma with solid growth and large areas of necrosis. At immunohistochemistry, neoplastic cells were diffusely positive for cytokeratin CAM 5.2 and negative for CD20 and CD3. No further immunohistochemical stains were performed owing to the small amount of viable tissue in the biopsy material.
The trend of lipases in the pre-operative period was decreasing but did not normalize before surgery. The clinical case was discussed at the Multidisciplinary Digestive System Meeting, confirming the hypothesis of an undifferentiated gastric tumor and the indication for surgery.
The patient underwent an elective surgical procedure. Laparoscopic exploration of the abdominal cavity was performed without identifying peritoneal and liver metastasis or ascites; moreover, laparoscopic peritoneal washing was performed. Taking into account the dimension of the gastric tumor and the plausible pancreatic infiltration, we opted for a laparotomic conversion that confirmed the tenacious infiltration of the pancreatic body by the posterior gastric lesion (Figure 7 and Figure 8). Otherwise, considering the hypothesis that the neoplasm might be a worse prognosis injury, maybe without a gastric origin, to be as oncologically radical as possible, we performed a total gastrectomy with esophageal jejunum anastomosis and reconstruction of intestinal continuity according to Roux, distal splenum-pancreatectomy, and cholecystectomy.
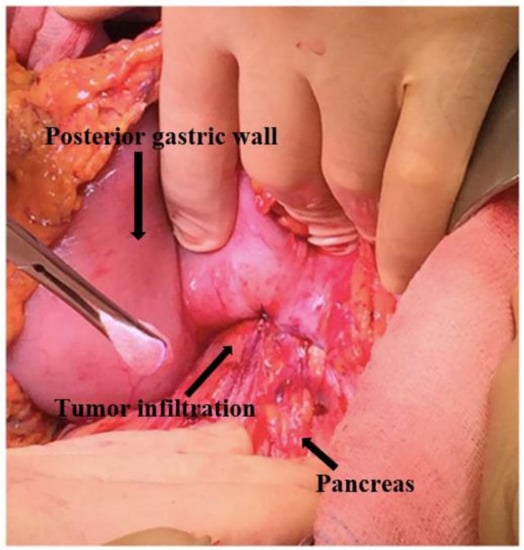
Figure 7.
Posterior gastric wall infiltration.
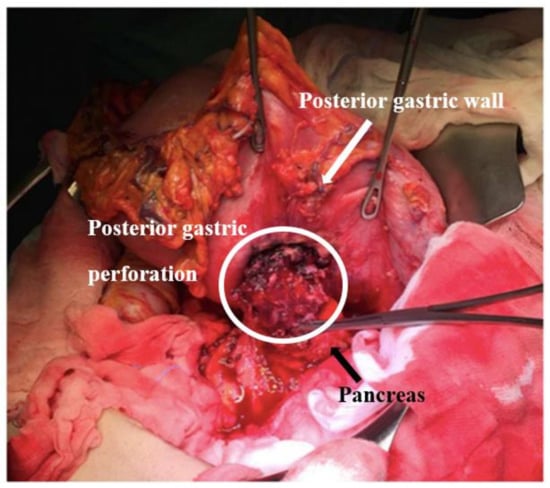
Figure 8.
Intraoperative finding of perforation of the posterior gastric wall (white circle).
Findings of the resected specimen showed, on the posterior gastric wall, a 10 cm necrotic polypoid lesion perforating and invading the pancreatic body.
At gross examination, the posterior gastric wall was occupied by a polypoid lesion measuring 10 cm along its greatest axis and showing a full thickness perforation; the pancreatic body and tail showed an irregular and hemorrhagic surface and, on slicing, was made of greyish tissue with a hard consistency and large hemorrhagic areas.
At histology, the lesion was highly cellular, with a predominant solid growth pattern; occasional acinar formation was also appreciated. There were large areas of necrosis, numerous mitoses (up to 8/10 HPF), and neoplastic vascular thromboses. No perineural neoplastic invasion was seen. The lesion invaded the gastric wall and the pancreatic parenchyma (Figure 9 and Figure 10) The omentum, spleen, and gallbladder, as well as resection margins, were free of tumor.
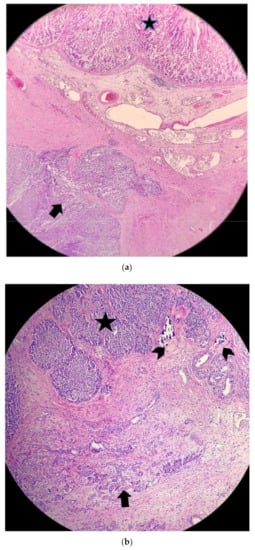
Figure 9.
(a) Above, gastric mucosa (star); below, gastric wall with neoplastic infiltration (arrow) (H&E, 100×). (b) The lesion (star) infiltrating the pancreatic parenchyma (arrow) which shows moderate atrophy. Occasional calcifications are also present (small arrows) (H&E 100×).
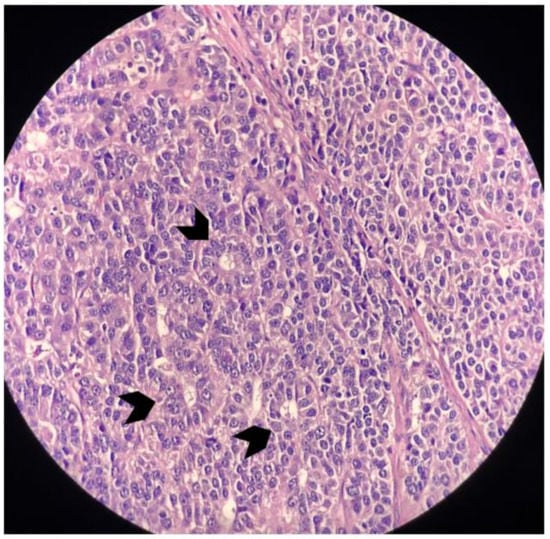
Figure 10.
Magnified image that highlights a neoplasm with occasional acinar formation (small arrows) (H&E, 400×).
At immunohistochemistry, the cells showed intense and diffuse positivity for CK CAM 5.2, CK7, CK8, and CK19, and negativity for chromogranin, synaptophysin, and CDX2. In addition, diffuse positivity for α1-antitrypsin and BCL10, as well as focal positivity for glypican 3, was highlighted, thus, documenting acinar differentiation.
Although acinar differentiation can very rarely occur in gastric carcinomas [6], the overall appearance of the tumor, specifically its proximity to the pancreas and the absence of acinar metaplasia and dysplasia in the remaining gastric mucosa, favored the diagnosis of a PACC.
The post-operative management was regular without complications and the patient was discharged on the 13th post-operative day.
The patient subsequently performed an oncological evaluation which assessed the need of adjuvant chemotherapy with gemcitabine, but the patient refused. The 5-month follow-up abdominal CT scan showed multiple hepatic metastases (Figure 11). At the following oncological evaluation, the therapy was modified with Abraxane combined with gemcitabine for palliative purposes. At the follow-up chest/abdomen CT scan after 9 months, there was a worsening of liver condition and the appearance of lung metastases (Figure 12 and Figure 13). The patient died 10 months after surgery.
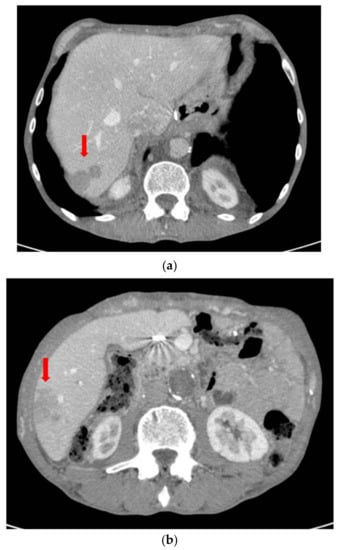
Figure 11.
(a) Follow-up CT scan after 5 months with evidence of hepatic metastases in S7. (b) Follow-up CT scan after 5 months with evidence of hepatic metastases in S6.
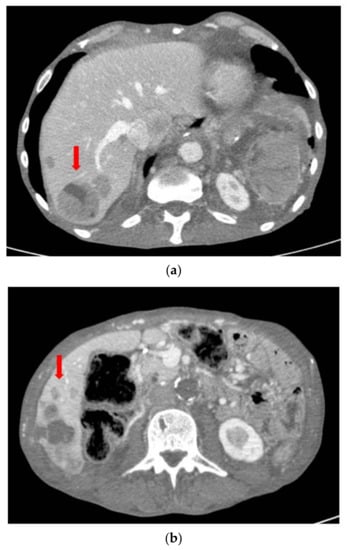
Figure 12.
(a) Follow-up CT scan after 9 months with increased size and number of liver metastases in S7. (b) Follow-up CT scan after 9 months with increased size and number of liver metastases in S6.
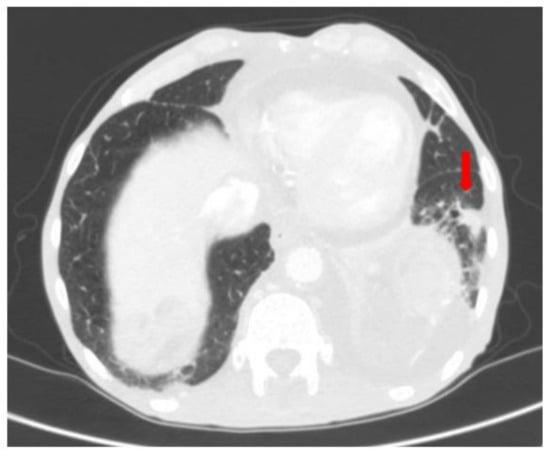
Figure 13.
Follow-up CT scan after 9 months with appearance of lung metastases in the left lower lobe.
3. Discussion and Literary Review
Pancreatic acinar cell carcinoma (PACC) is a rare condition, representing less than 2% of exocrine pancreatic cancers; they usually show up late in adulthood, at around 60–70 years of age [1], arise in any part of the organ without specific symptoms, and can produce pancreatic enzymes [3,4]. The etiology is not well understood, without any foregoer injuries, although some precursor lesions have been documented [2]. Considering that most of the pancreatic gland consists of acinar tissue, pancreatic cancers with a predominant acinar differentiation are indeed uncommon and can be found incidentally [2,4]. The overall survival in patients with PACC ranges from 18 to 47 months [7], less than in patients with gastric cancer [8]; in our case, we expected, therefore, a “lamb” while, after the surgery, we discovered an “hidden wolf”, considering the definitive diagnosis with a worse prognosis.
In fact, at the time of diagnosis, about 50% of cases already present metastases, above all in liver [9]; furthermore, paraneoplastic lipase hypersecretion syndrome (characterized by polyarthralgia, subcutaneous fat necrosis, and eosinophilia) can be clinically present in 15% of cases, with a worse prognosis and presence of diffuse metastatic disease [1,2,3,9,10] In our case, the metastases manifested at the oncological follow-up, a few months after surgery.
Regarding the radiologic findings, the MRI/CT scan features of PACC are not standardized; however, the tumor most often presents as an exophytic, oval, well-encapsulated mass with poor vascularization [11]. According to this, PACC must be differentiated from other solid pancreatic neoplasms. On the one hand, solid pancreatic pseudopapillary tumors (PTS) classically show a mass surrounded by a pseudocapsule and composed of neoplastic tissue mixed with fibrous tissue, with occasional peripheral calcifications, hemorrhage, and necrosis; on the other hand, pancreatic neuroendocrine tumors (PETs) are characterized by a well-circumscribed hyper-vascular mass [12]. The CT scan aspect in our case only demonstrated a mixed density gastric solid mass (probably due to previous bleeding) of at least 8 cm in greatest axis, hypertensive at all stages, and surrounded by a pseudocapsule.
Histopathologic diagnosis of PACC can be challenging because other rare pancreatic neoplasms, such as PTS and PETs, must be considered in the differential diagnosis. In fact, these lesions share some architectural and cytologic features in common with PACC, such as a trabecular or acinar growth pattern, as well as the presence of an eosinophilic cytoplasm and inconspicuous nucleoli. Immunohistochemistry is, therefore, essential for obtaining a correct diagnosis; PETs are positive for antibodies directed against pancytokeratin, synaptophysin, and chromogranin. Instead, PTS can be strongly positive for antibodies to vimentin, beta-catenin (nuclear positivity), CD56, and progesteron receptors [13].
The histologic diagnosis of PACC requires the detection of acinar differentiation, which is absent in PTS and PETs. It is characterized by the production of pancreatic enzymes, such as trypsin, amylase, lipase, and chymotrypsin, and, on the immunohistochemical level, is documented by the positive reaction for α1-antichymotrypsin, α1-antitrypsin, and BCL 10 antibodies. [13,14,15,16,17]. In addition, glypican 3 has recently been reported to be expressed in more than 50% of PACC [18].
The present case, along with various cytokeratin expression, showed a positive immunohistochemical reaction for α1-antitrypsin, BCL 10 and glypican 3 antibodies, thus, documenting the presence of acinar differentiation.
Acinar differentiation can also be detected in primary gastric carcinomas where it has been explained in several ways, such as arising in pancreatic heterotopia or in metaplastic tissue (e.g., acinar metaplasia). These are well recognized phenomena in gastric mucosa, either as a congenital finding or associated with chronic gastritis. However, the occurrence of acinar differentiation in gastric cancer is very rare and can be accompanied by a component of conventional gastric adenocarcinoma [6], which was not seen in the present case. In addition, the proximity to the pancreas and the absence of acinar metaplasia and dysplasia in the remaining gastric mucosa favored the pancreatic origin of the lesion. As PACC can reach huge sizes, the present tumor grew anteriorly toward the stomach, eventually perforating the visceral wall and protruding in the gastric lumen.
Given these premises, making a pre-operative diagnosis of PACC in this case would have been highly unlikely. There was not enough tissue in the biopsy material and, even if acinar differentiation had been documented, it would have been impossible to establish the origin of the neoplasm as there are no site-specific immunohistochemical markers that can distinguish a gastric or a pancreatic primary cancer.
As PACC is often large and exophytic, infiltration sometimes occurs through the extension of the intrapancreatic ducts or the main pancreatic duct, which can be associated with a vascular infiltration, resulting in vascular thrombosis (such as the portal vein or splenic vein) [19,20,21]. In other cases, the infiltration may be of greater magnitude and extend to the adjacent organs, as in our case where there was an invasion of the posterior gastric wall.
There is no standard chemotherapeutic regimen established for patients with unresectable or recurrent PACC, because PACC is a rare pancreatic cancer and no large-scale randomized controlled trials have been performed yet; platinum-containing chemotherapy regimens and irinotecan, however, are potentially useful drugs/regimens for these clinical cases [4]. Nevertheless, further studies are needed to define the role of chemoradiotherapy in palliative, adjuvant, and neoadjuvant settings [5]. In our case the patient refused any oncological treatment.
Misunderstood PACC are not so rare, and some similar case reports, with extensive involvement of adjacent organs, are described in the literature and summarized in Table 1. The first case [16] is a primary PACC, arising from the stomach, which presented a 5 cm ulcerated gastric mass onset with anemia. Poorly differentiated gastric adenocarcinoma was diagnosed at a preoperative biopsy, and partial gastrectomy with Billroth II reconstruction was performed but, during the anatomopathological examination, the diagnosis of PACC was performed, and demonstrated a positivity for antitrypsin and antichymotrypsin; no pancreatic or non-pancreatic tumors could be identified postoperatively. The second case regards a PACC with erosion of the duodenal bulb and pylorus; the preoperative diagnosis was non-functioning pancreatic endocrine tumor or gastric cancer invading both the duodenum and the pancreatic head. The patient underwent surgery and died after 6 months from multiple liver metastases [22]. The third case involved splenic flexure of the colon; a left hemicolectomy, resection of the jejunum, the pancreas body and tail, in association with splenectomy were performed, and the patient died 14 weeks after surgery of hepatic failure [23]. A particular clinical case is described in the literature with a preoperative diagnosis of a gastro-intestinal stromal tumor (GIST) or malignant lymphoma (ML), was about an acinar cell carcinoma arising from an ectopic pancreas invaded the pylorus [17]. Takagi et al. described more recently a case of PACC arising with melena; it was identified preoperatively with a jejunal mass associated with a jejunal vein thrombus. The patient underwent partial jejunostomy and partial pancreatectomy surgery and a 10-month follow-up showed no recurrences [24]. Another case present in the literature describes PACCs of 10 cm in diameter with a high level of serum alpha-fetoprotein (AFP, 1222.4 ng/mL) without being able to certainly identify the origin of the tumor; the diagnosis was hepatocellular carcinoma due to the connection with the left hepatic obo and AFP level [25]. The last case report deals with a patient without evidence of a pancreatic tumor treated with subtotal gastrectomy for a 4 cm ulcerated tumor in the distal antrum. Microscopically, intense positivity for alpha-1-antitrypsin was found, and a diagnosis of PACC originating from ectopic cells was made [26].

Table 1.
Case reports of PACC with extensive involvement of adjacent organs and misunderstood preoperative diagnosis.
4. Conclusions
Here, PACC is a rare and highly aggressive tumor that represents <2% of exocrine pancreatic cancers and most often occurs in adulthood (at 60–70 year of age) [1]. Overall survival in patients with PACC ranges from 18 to 47 months [7]. The literature shows about 50% of cases with metastases at diagnosis, often to the liver [5], although they can sometimes occur in the lungs, in ovaries, and in cervical lymph nodes [2].
If lipase hypersecretion syndrome is present, survival is shorter, as it is frequently associated with the presence of diffuse metastatic disease [2].
Surgery is the treatment of choice if PACC is diagnosed at early stage, but excision is recommended as treatment of these cancers regardless the size, when feasible, also because the role of neoadjuvant or adjuvant chemo- or radiotherapy remain unproven [3].
Considering that PACC is a rare tumor with a frequent unusual clinical presentation, it should be considered in differential diagnosis of peri-pancreatic large size tumors and unclear diagnoses.
We are aware about the limitation of our single case report and the limited data in the literature about this topic, but our paper could improve the knowledge about this rare and confusing tumor.
Author Contributions
Conceptualization M.C.; methodology N.Z.; validation V.C. and G.G.N.; resources A.F. and F.F.; data and images curation C.B.; writing-original draft preparation M.C.; writing-review and editing N.Z.; visualization C.B.; anatomic histological supervision A.F.; global supervision V.C. and G.G.N. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Patient consent was waived due to patient’s death and the absence of other members of the family.
Data Availability Statement
Data is contained within the article.
Acknowledgments
Moderate English changes have been edited; we wish to thank M. Di Matteo for her invaluable support in English language and style.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| PACC | (Pancreatic acinar cell carcinoma) |
| OS | (Overall survival) |
| ED | (Emergency Department) |
| COPD | (Chronic obstructive pulmonary disease) |
| UGI | (Upper gastrointestinal) |
| NV | (Normal value) |
| CRP | (C-reactive protein) |
| SPT | (Solid pseudopapillary tumors) |
| PET | (Pancreatic endocrine tumors) |
References
- Klimstra, D.S.; Heffess, C.S.; Oertel, J.E.; Rosai, J. Acinar cell carcinoma of the pancreas. A clinicopathologic study of 28 cases. Am. J. Surg. Pathol. 1992, 16, 815–837. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, P. Acinar Cell Carcinoma of the Pancreas: A Literature Review and Update. Indian J. Surg. 2015, 77, 226–231. [Google Scholar] [CrossRef]
- Toll, A.D.; Hruban, R.H.; Ali, S.Z. Acinar cell carcinoma of the pancreas: Clinical and cytomorphologic characteristics. Korean J. Pathol. 2013, 47, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Ikeda, M.; Shiba, S.; Imaoka, H.; Todaka, A.; Shioji, K.; Yane, K.; Kojima, Y.; Kobayashi, S.; Asagi, A.; et al. Multicenter Retrospective Analysis of Chemotherapy for Advanced Pancreatic Acinar Cell Carcinoma: Potential Efficacy of Platinum- and Irinotecan-Containing Regimens. Pancreas 2021, 50, 77–82. [Google Scholar] [CrossRef]
- Wisnoski, N.C.; Townsend, C.M., Jr.; Nealon, W.H.; Freeman, J.L.; Riall, T.S. 672 patients with acinar cell carcinoma of the pancreas: A population-based comparison to pancreatic adenocarcinoma. Surgery 2008, 144, 141–148. [Google Scholar] [CrossRef]
- Lee, H.; Tang, L.H.; Veras, E.F.; Klimstra, D.S. The prevalence of pancreatic acinar differentiation in gastric adenocarcinoma: Report of a case and immunohistochemical study of 111 additional cases. Am. J. Surg. Pathol. 2012, 36, 402–408. [Google Scholar] [CrossRef]
- Al-Hader, A.; Al-Rohil, R.N.; Han, H.; Von Hoff, D. Pancreatic acinar cell carcinoma: A review on molecular profiling of patient tumors. World J. Gastroenterol. 2017, 23, 7945–7951. [Google Scholar] [CrossRef]
- Chan, W.L.; Lam, K.O.; Lee, V.H.F.; Davidson, M.; So, T.H.; Li, J.S.; Chau, I.; Kwong, D.L.W. Gastric Cancer—From Aetiology to Management: Differences between the East and the West. Clin. Oncol. R. Coll. Radiol. 2019, 31, 570–577. [Google Scholar] [CrossRef]
- Chaudhary, P.; Ranjan, G.; Chaudhary, A.; Tiwari, A.K.; Arora, M.P. Acinar cell carcinoma: A rare pancreatic malignancy. Clin. Pract. 2013, 3, e18. [Google Scholar] [CrossRef]
- Holen, K.D.; Klimstra, D.S.; Hummer, A.; Gonen, M.; Conlon, K.; Brennan, M.; Saltz, L.B. Clinical characteristics and outcomes from an institutional series of acinar cell carcinoma of the pancreas and related tumors. J. Clin. Oncol. 2002, 20, 4673–4678. [Google Scholar] [CrossRef]
- Tatli, S.; Mortele, K.J.; Levy, A.D.; Glickman, J.N.; Ros, P.R.; Banks, P.A.; Silverman, S.G. CT and MRI features of pure acinar cell carcinoma of the pancreas in adults. Am. J. Roentgenol. 2005, 184, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Cui, W.; Wang, J.; Chen, X.; Tong, H.; Wang, Z. Differentiation between solid pseudopapillary neoplasm of the pancreas and hypovascular pancreatic neuroendocrine tumors by using computed tomography. Acta Radiol. 2019, 60, 1216–1223. [Google Scholar] [CrossRef]
- Stelow, E.B.; Bardales, R.H.; Shami, V.M.; Woon, C.; Presley, A.; Mallery, S.; Lai, R.; Stanley, M.W. Cytology of pancreatic acinar cell carcinoma. Diagn. Cytopathol. 2006, 34, 367–372. [Google Scholar] [CrossRef]
- Adams, A.L.; Siegal, G.P.; Jhala, N.C. Solid pseudopapillary tumor of the pancreas: A review of salient clinical and pathologic features. Adv. Anat. Pathol. 2008, 15, 39–45. [Google Scholar] [CrossRef] [PubMed]
- La Rosa, S.; Franzi, F.; Marchet, S.; Finzi, G.; Clerici, M.; Vigetti, D.; Chiaravalli, A.M.; Sessa, F.; Capella, C. The monoclonal anti-BCL10 antibody (clone 331.1) is a sensitive and specific marker of pancreatic acinar cell carcinoma and pancreatic metaplasia. Virchows Arch. 2009, 454, 133–142. [Google Scholar] [CrossRef]
- Sun, Y.; Wasserman, P.G. Acinar cell carcinoma arising in the stomach: A case report with literature review. Hum. Pathol. 2004, 35, 263–265. [Google Scholar] [CrossRef]
- Mizuno, Y.; Sumi, Y.; Nachi, S.; Ito, Y.; Marui, T.; Saji, S.; Matsutomo, H. Acinar cell carcinoma arising from an ectopic pancreas. Surg. Today 2007, 37, 704–707. [Google Scholar] [CrossRef] [PubMed]
- Mounajjed, T.; Zhang, L.; Wu, T.T. Glypican-3 expression in gastrointestinal and pancreatic epithelial neoplasms. Hum. Pathol. 2013, 44, 542–550. [Google Scholar] [CrossRef]
- Kida, A.; Matsuda, K.; Takegoshi, K.; Matsuda, M.; Sakai, A.; Noda, Y. Pancreatic acinar cell carcinoma with extensive tumor embolism at the trunk of portal vein and pancreatic intraductal infiltration. Clin. J. Gastroenterol. 2017, 10, 546–550. [Google Scholar] [CrossRef]
- Nishimura, M.; Wada, H.; Eguchi, H.; Iwagami, Y.; Yamada, D.; Asaoka, T.; Noda, T.; Gotoh, K.; Kobayashi, S.; Mori, M.; et al. A Case Report of Acinar Cell Carcinoma of Pancreas with Extensive Intraductal Growth to the Branch—Main Pancreatic Duct. Gan Kagaku Ryoho 2017, 44, 1568–1570. (In Japanese) [Google Scholar]
- Iwatate, M.; Matsubayashi, H.; Sasaki, K.; Kishida, N.; Yoshikawa, S.; Ono, H.; Maitra, A. Functional pancreatic acinar cell carcinoma extending into the main pancreatic duct and splenic vein. J. Gastrointest. Cancer 2012, 43, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Matsuyama, T.; Ogata, S.; Sugiura, Y.; Yoshizumi, Y.; Aiko, S.; Aida, S.; Maehara, T. Acinar cell carcinoma of the pancreas eroding the pylorus and duodenal bulb. J. Hepatobiliary Pancreat. Surg. 2004, 11, 276–279. [Google Scholar] [CrossRef] [PubMed]
- Asayama, N.; Kojima, Y.; Aoki, T.; Maeyashiki, C.; Yokoi, C.; Yanase, M.; Suda, R.; Yano, H.; Nakamura, H.; Igari, T. Acinar cell carcinoma of the pancreas with colon involvement. Case Rep. Med. 2014, 2014, 389425. [Google Scholar] [CrossRef]
- Takagi, K.; Yagi, T.; Tanaka, T.; Umeda, Y.; Yoshida, R.; Nobuoka, D.; Kuise, T.; Fujiwara, T. Primary pancreatic-type acinar cell carcinoma of the jejunum with tumor thrombus extending into the mesenteric venous system: A case report and literature review. BMC Surg. 2017, 17, 75. [Google Scholar] [CrossRef]
- Hiraoka, A.; Nakahara, H.; Kawasaki, H.; Shimizu, Y.; Hidaka, S.; Imai, Y.; Utsunomiya, H.; Tatsukawa, H.; Tazuya, N.; Yamago, H.; et al. Huge pancreatic acinar cell carcinoma with high levels of AFP and fucosylated AFP (AFP-L3). Intern. Med. 2012, 51, 1341–1349. [Google Scholar] [CrossRef] [PubMed]
- Ambrosini-Spaltro, A.; Potì, O.; De Palma, M.; Filotico, M. Pancreatic-type acinar cell carcinoma of the stomach beneath a focus of pancreatic metaplasia of the gastric mucosa. Hum. Pathol. 2009, 40, 746–749. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).