The Acceptability of Faecal Microbiota Transplant for Anterior Resection Syndrome (AFFINITY) Study
Abstract
1. Introduction
2. Materials and Methods
3. Results
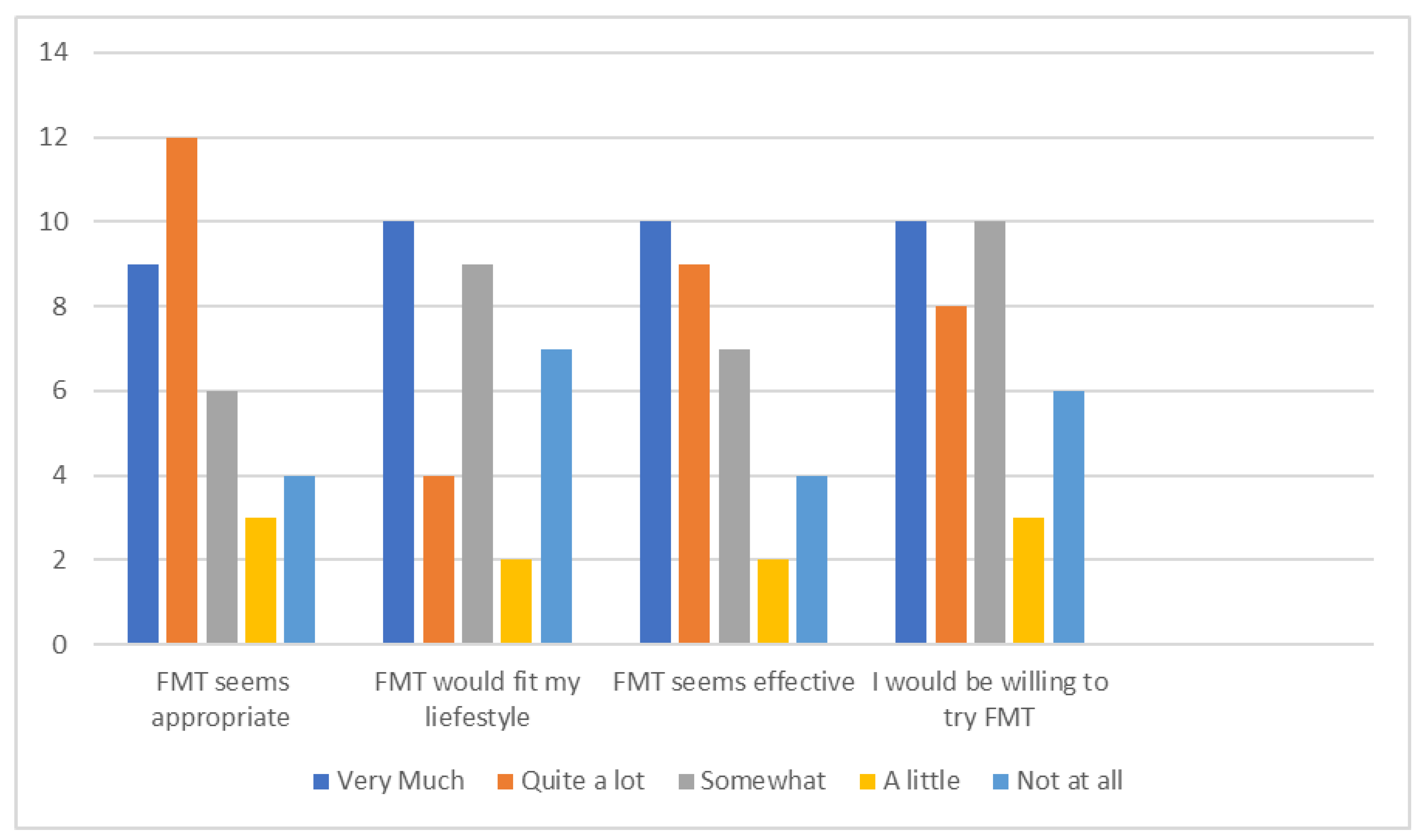
3.1. Survey Results
3.2. Focus Group Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. AFFINITY Questionnaire
- Questions about your surgery and symptoms
- Which of these best describes your surgery?
- Currently have ileostomy/stoma ☐
- Had ileostomy/stoma but now reversed☐
- Never had ileostomy/stoma ☐
- LARS Score
- Do you ever have occasions when you cannot control you flatus (wind)?
- No, never☐ (0)
- Yes, less than once per week ☐(4)
- Yes, at least once per week ☐(7)
- Do you ever have any accidental leakage of liquid stool?
- No, never☐ (0)
- Yes, less than once per week ☐ (3)
- Yes, at least once per week ☐ (3)
- How often do you open your bowels?
- More than 7 times per day (24 h) ☐ (4)
- 4–7 times per day (24 h) ☐ (2)
- 1–3 times per day (24 h) ☐ (0)
- Less than once per day (24 h) ☐ (5)
- Do you ever have to open your bowels again within one hour of the last bowel opening?
- No, never☐ (0)
- Yes, less than once per week ☐ (9)
- Yes, at least once per week ☐ (11)
- Do you ever have such a strong urge to open your bowels that you have to rush to the toilet?
- No, never☐ (0)
- Yes, less than once per week ☐ (11)
- Yes, at least once per week ☐ (16)
- Have you had treatment for symptoms of low anterior resection syndrome/LARS?
- Yes ☐ No ☐ Don’t know ☐
- If your answers suggest you may benefit for treatment for low anterior resection syndrome/LARS, would you be happy for us to contact your GP/local colorectal team?
- Yes ☐ No ☐
- Faecal Microbiota Transplantation (FMT) is putting bacteria from someone else’s faeces (poo) into the bowel. It is a recognized treatment for some bowel infections. It works by increasing the healthy bacteria in the bowel. Some people who have surgery for rectal cancer have altered bowel function after their ileostomy (stoma) is reversed. We think this may be caused by a lack of healthy bacteria in the bowel. This could be improved by FMT. The faeces from a healthy donor is mixed with saline (salt water) and would be inserted through the ileostomy before surgery to close it.
- We want to know whether this sounds like an acceptable treatment to you.
- For each statement please tick the box which fits most with your opinion.
| Not at all | A Little | Some-what | Quite a bit | Very Much | |
|---|---|---|---|---|---|
| FMT seems like an appropriate way to prevent symptoms of LARS | |||||
| FMT would fit my individual lifestyle | |||||
| FMT seems like an effective way to prevent LARS | |||||
| I would be willing to try FMT |
- Which of these would be an acceptable FMT donor? (tick all that apply)
- Partner/spouse ☐
- Blood relative ☐
- Friend ☐
- Anonymous healthy donor ☐
- None of above ☐
- Which of these routes for giving FMT is acceptable?
- Via the ileostomy/stoma ☐
- Via the rectum (bottom)/enema ☐
- Via colonoscopy (magic eye/camera test) ☐
- None of above ☐
- Questions about you
- What is your age? ………..
- What is your gender?
- Male ☐ Female ☐ Other ☐
- How would you describe your ethnic origin?
- Asian or Asian British☐ Black or Black British ☐ Mixed ☐
- White British ☐ Other ethnic group☐ Please write in ………………
- Are you…?
- Single ☐ Married/civil partnership ☐ Widowed ☐
- Co-habiting ☐ Separated/divorced ☐
- What is your level of education?
- Degree or equivalent ☐ A Level or equivalent ☐
- GCSEs or equivalent ☐ Other qualification ☐
- No qualification ☐
- Which of these activities best describes what you are doing at present?
- Employee in full time job☐ Employee in part time job☐
- Self-employed☐ Full time education☐
- Unemployed☐ Permanently sick/disabled ☐
- Retired☐ Looking after the home ☐
- Carer for spouse/relative ☐ Other ☐
- What is your current job or last job you did if not currently working?
- ……………………………………………………………….
- Had you heard of FMT before receiving this questionnaire?
- Yes- for clostridium difficile/infection ☐
- Yes- for ulcerative colitis/inflammatory bowel ☐
- No ☐
Appendix B. Focus Group Topic Guide
| Welcome and thank participants on arrival Take consent Introductions and purpose of the focus group |
The idea of FMT
|
Educational information
|
Participating in a trial
|
| Any other comments you would like to make? |
| Thank participants |
References
- Bowel Cancer UK. Facts and Figures. Available online: http://www.bowelcanceruk.org.uk/information-resources/bowel-cancer-facts-figures/ (accessed on 24 November 2019).
- Cornish, J.A.; Tilney, H.S.; Heriot, A.G.; Lavery, I.C.; Fazio, V.W.; Tekkis, P.P. A meta- analysis of quality of life for abdominoperineal excision of rectum versus anterior resection for rectal cancer. Ann. Surg. Oncol. 2007, 14, 2056–2068. [Google Scholar] [CrossRef] [PubMed]
- Association of Coloproctology of Great Britain and Ireland. Guidelines for the Management of Colorectal Cancer. 2007. Available online: https://www.acpgbi.org.uk/content/uploads/2007-CC-Management-Guidelines.pdf (accessed on 24 November 2019).
- Montedori, A.; Cirocchi, R.; Farinella, E. Covering ileo- or colostomy in anterior resection for rectal carcinoma. Cochrane Database Syst. Rev. 2010, 5, CD006878. [Google Scholar] [CrossRef] [PubMed]
- Emmertsen, K.J.; Laurberg, S. Low anterior resection syndrome score: Development and validation of a symptom-based scoring system for bowel dysfunction after low anterior resection for rectal cancer. Ann. Surg. 2012, 255, 922–928. [Google Scholar] [CrossRef]
- Bryant, C.L.; Lunniss, P.J.; Knowles, C.H.; Thaha, M.A.; Chanm, C.L. Anterior resection syndrome. Lancet Oncol. 2012, 13, e403–e408. [Google Scholar] [CrossRef]
- Ziv, Y.; Zbar, A.; Bar-Shavit, Y.; Igov, I. Low anterior resection syndrome (LARS): Cause and effect and reconstructive considerations. Tech. Coloproctol. 2013, 17, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Ihn, M.H.; Kang, S.B.; Kim, D.W.; Oh, H.K.; Lee, S.Y.; Hong, S.M. Risk factors for bowel dysfunction after sphincter-preserving rectal cancer surgery: A prospective study using the Memorial Sloan Kettering Cancer Center bowel function instrument. Dis. Colon Rectum 2014, 57, 958–966. [Google Scholar] [CrossRef] [PubMed]
- Siassi, M.; Hohenberger, W.; Lösel, F. Quality of life and patient’s expectations after closure of a temporary stoma. Int. J. Colorectal Dis. 2008, 23, 1207–1212. [Google Scholar] [CrossRef] [PubMed]
- Son, D.N.; Choi, D.J.; Woo, S.U.; Kim, J.; Keom, B.R.; Kim, C.H.; Baek, S.J.; Kim, S.H. Relationship between diversion colitis and quality of life in rectal cancer. World J. Gastroenterol. 2013, 19, 542–549. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.; Kim, S.; Lee, C.; Roh, K.; Keum, B.; Kim, C.; Kim, J. Relationship between the severity of diversion colitis and the composition of colonic bacteria: A prospective study. Gut Liver 2014, 8, 17–76. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Excellence Interventional Procedures Guidance [IPG485] Faecal Microbiota Transplant for Recurrent Clostridium Difficile Infection. 2014. Available online: https://www.nice.org.uk/guidance/ipg485 (accessed on 24 November 2019).
- Paramsothy, S.; Kamm, M.A.; Kaakoush, N.O.; Walsh, A.J.; van den Bogaerde, J.; Samuel, D.; Leong, R.W.L.; Connor, S.; Ng, W.; Paramsoth, R.; et al. Multidonor intensive faecal microbiota transplantation for active ulcerative colitis: A randomised placebo-controlled trial. Lancet 2017, 389, 1218–1228. [Google Scholar] [CrossRef]
- Pinn, D.M.; Aroniadis, O.C.; Brandt, L.J. Is fecal microbiota transplantation (FMT) an effective treatment for patients with functional gastrointestinal disorders (FGID)? Neurogastroenterol. Motil. 2015, 27, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Palmer, B.; Metcalfe, C.; Fraser, A.; Creed, T. Does education influence the acceptability of faecal microbiota transplantation in colitis: A cross-sectional study. Cogent. Med. 2016, 3, 1233685. [Google Scholar] [CrossRef]
- Leung, F.H.; Savithiri, R. Spotlight on focus groups. Can. Fam. Phys. 2009, 55, 218–219. [Google Scholar] [PubMed]
- Kreuger, R.A.; Casey, M.A. Focus Groups: A Practical Guide for Applied Research, 5th ed.; SAGE Publications: Thousand Oaks, CA, USA, 2014; pp. 63–81. [Google Scholar]
- US Food and Drug Administration. Important Safety Alert Regarding Use of Fecal Microbiota for Transplantation and Risk of Serious Adverse Reactions Due to Transmission of Multi-Drug Resistant Organisms. 2019. Available online: https://www.fda.gov/vaccines-blood-biologics/safety-availability-biologics/important-safety-alert-regarding-use-fecal-microbiota-transplantation-and-risk-serious-adverse (accessed on 24 November 2019).
- Mullish, B.H.; Nabil Quraishi, M.; Segal, J.P.; McCune, V.L.; Baxter, M.; Marsden, G.L.; Moore, D.J.; Colville, A.; Bhala, N.; Iqbal, T.H.; et al. The use of faecal microbiota transplant as treatment for recurrent or refactory Clostridium difficile infection and other potential indications: Joint British Society of Gastroenterology (BSG) and Healthcare Infection Society (HIS) guidelines. Gut 2018, 67, 1920–1941. [Google Scholar] [CrossRef] [PubMed]
- Olesen, S.W.; Leier, M.M.; Alm, E.J.; Kahn, S.A. Searching for superstool: Maximizing the therapeutic potential of FMT. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 387–388. [Google Scholar] [CrossRef] [PubMed]
| Demographic | Responses n = 40 (%) | |
|---|---|---|
| Ethnicity | White British | 38 (95) |
| Did not answer | 2 (5) | |
| Marital Status | Married/Civil Partnership | 24 (60) |
| Separated/Divorced | 6 (15) | |
| Cohabiting | 3 (7.5) | |
| Single | 3 (7.5) | |
| Widowed | 3 (7.5) | |
| Did not answer | 1 (2.5) | |
| Level of education | Degree | 8 (20) |
| A Level | 5 (12.5) | |
| GCSE | 14 (35) | |
| No qualification | 6 (15) | |
| Other | 6 (15) | |
| Did not answer | 1 (2.5) | |
| Employment | Employed full time | 4 (10) |
| Employed part time | 6 (15) | |
| Full time education | 1 (2.5) | |
| Retired | 18 (45) | |
| Self-employed | 10 (25) | |
| Unemployed | 1 (2.5) |
| LARS Score | Responses n = 29 (%) |
|---|---|
| 0–20 (No LARS) | 7 (24) |
| 21–29 (Minor LARS) | 7 (24) |
| 30–42 (Major LARS) | 15 (52) |
| Demographics | Willing to Try FMT Very Much/Quite a Bit n = 18 (%) | Not Willing to Try FMT A Little/not at all n = 9 (%) |
|---|---|---|
| Male | 10 (56) | 5 (56) |
| Median age (range) | 69 (31–79) | 73 (38–80) |
| Degree level education | 5 (28) | 1 (11) |
| No qualification | 3 (17) | 1 (11) |
| In work/education | 10 (56) | 3 (33) |
| Married/cohabiting | 11 (61) | 7 (78) |
| Previously heard of FMT | 5 (28) | 1 (11) |
| Have stoma | 8 (44) | 0 (0) |
| Major LARS | 6 (33) | 4 (44) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Powell-Chandler, A.; Withers, K.L.; Carolan-Rees, G.; Cornish, J.A. The Acceptability of Faecal Microbiota Transplant for Anterior Resection Syndrome (AFFINITY) Study. Surgeries 2020, 1, 10-20. https://doi.org/10.3390/surgeries1010003
Powell-Chandler A, Withers KL, Carolan-Rees G, Cornish JA. The Acceptability of Faecal Microbiota Transplant for Anterior Resection Syndrome (AFFINITY) Study. Surgeries. 2020; 1(1):10-20. https://doi.org/10.3390/surgeries1010003
Chicago/Turabian StylePowell-Chandler, Anna, Kathleen L. Withers, Grace Carolan-Rees, and Julie A. Cornish. 2020. "The Acceptability of Faecal Microbiota Transplant for Anterior Resection Syndrome (AFFINITY) Study" Surgeries 1, no. 1: 10-20. https://doi.org/10.3390/surgeries1010003
APA StylePowell-Chandler, A., Withers, K. L., Carolan-Rees, G., & Cornish, J. A. (2020). The Acceptability of Faecal Microbiota Transplant for Anterior Resection Syndrome (AFFINITY) Study. Surgeries, 1(1), 10-20. https://doi.org/10.3390/surgeries1010003






