Abstract
The scientific, private, and industrial sectors use a wide variety of technological platforms available to achieve protection against SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2), including vaccines. However, the virus evolves continually into new highly virulent variants, which might overcome the protection provided by vaccines and may re-expose the population to infections. Mass vaccinations should be continued in combination with more or less mandatory non-pharmaceutical interventions. Therefore, the key questions to be answered are: (i) How to identify the primary and secondary infections of SARS-CoV-2? (ii) Why are neutralizing antibodies not long-lasting in both cases of natural infections and post-vaccinations? (iii) Which are the factors responsible for this decay in neutralizing antibodies? (iv) What strategy could be adapted to develop long-term herd immunity? (v) Is the Spike protein the only vaccine target or is a vaccine cocktail better?
1. Can SARS-CoV-2 Infection Provide Lifelong Immunity?
Adaptive immune responses triggered by SARS-CoV-2 infections, including B and T lymphocyte cell response, can induce immunological memory during primary infection to prevent or decrease disease severity on repeated exposures to the same pathogen. On reinfection, antibodies produced by short-lived plasma cells derived from B lymphocytes bind to and block the recognized virus, and the long-lasting bone marrow plasma cells (BMPCs) become resident in the bone marrow and retain the ability to produce antibodies for decades [1].
The SARS-CoV-2 Immunity & REinfection EvaluatioN (SIREN) study enrolled 8278 SARS-CoV-2 seropositive and 17,383 seronegative healthcare workers in England between 18 June 2020, and 31 December 2020 [2]. The study demonstrated that individuals with a previous history of SARS-CoV-2 infection exhibited an 84% lower infection risk than the seronegative individuals, with a median protective effect lasting 7 months following primary infection [2].
However, this “long-lasting” immunity may not protect against reinfection, and despite huge numbers of infections and deaths worldwide caused by SARS-CoV-2, herd immunity could not be achieved in communities with the previous infection of most of its population. One factor which affects protection against reinfections is the severity of primary infections. In severe SARS-CoV-2 infection, the immune response may be impaired, which will decrease affinity maturation, memory quality, and quantity [3]. In contrast, in cases of mild COVID-19 (coronavirus disease 2019) disease and quick recovery, the efficient antibody immune response that cleared symptoms rapidly remains stable for a long time after infection [4]. Another factor is the decrease/waning in antibody levels with time, which was demonstrated in seven individuals five months after SARS-CoV-2 infection [5].
It has also been reported that circulating anti-spike IgG antibodies remain detectable one year after hospitalization of COVID-19 patients, and these higher antibody titers and disease severity were associated with increased durability of detectable antibodies [6]. Although the total circulating anti-SARS-CoV-2 antibodies wane over a few months, these antibodies could be detected up to one year after infection, even in patients with mild COVID-19 [7]. The levels of circulating neutralizing antibodies correlate with the duration and infection severity but not with patient age [8]. Many parameters such as but not limited to patient age, symptomatic or asymptomatic phenotype, symptomatic grade, duration of sampling, type of sample, and type of evaluation methodology used have not been broadly discussed in publications [9]. This has generated existing controversies regarding the durability of the acquired immunity.
Another concern about assessing long-lasting immunity after primary SARS-CoV-2 infection is the difficulty of identifying the precise incidence of SARS-CoV-2 reinfection [10,11]. Viral genome sequencing from primary and secondary infections is required to ensure that they are two separate events and repeated PCR (polymerase chain reaction) testing is required to confirm reinfection. Individuals who have tested positive in PCR assays should show a negative PCR result and then test positive after reinfection. Moreover, reinfections were difficult to track during the first wave of the COVID-19 pandemic due to the overloaded testing capacity [10,11].
New SARS-CoV-2 variants may be the most important explanation for the immunity loss due to the presence of mutations making them more transmissible, more efficient at avoiding the host immune system and evading immunity elicited by primary infection. In fact, throughout the COVID-19 pandemic, many SARS-CoV-2 variants have appeared. Some are variants of interest (VOI) that may be associated with reduced efficacy of available treatments and vaccinations, and predicted increase in transmissibility or disease severity (e.g., B.1.427 (Epsilon), B.1.525 (Eta), and B.1.617.1 (Kappa)), and some are variants of concern (VOC) that may cause increase in transmissibility, more severe disease (e.g., increased hospitalizations or deaths), significant reduction in neutralizing antibodies generated during previous infection or vaccination (e.g., Alpha (B.1.1.7), Beta (B.1.351), Delta (B.1.617.2 and its sub-lineages AY.1, AY.2, and AY.3), Gamma (P.1), and most recently Omicron (B1.1.529)). The seriousness of this issue can be illustrated by the case in Manaus, Brazil, where SARS-CoV-2 infected more than 70% of the population by October 2020, which should have provided herd immunity. However, a surge of reinfection occurred in late December 2020, and early January 2021; i.e., about 7 months after the first wave [12,13]. The most likely explanation for the decrease in protective antibody levels and the reinfection of the population was the appearance of new more transmissible SARS-CoV-2 variants such as the B.1.1.7 (Alpha), B.1.351 (Beta), B.1.617.2 (Delta), and AY.1, AY.2, and AY.3 sub-lineages of Delta [12,13]. Another example is given by the Omicron variant, which was demonstrated to systematically escape neutralization by the existing vaccines, convalescent serum, and most therapeutic monoclonal antibodies (mAbs) [14,15,16]. In general, the accumulating evidence suggests that the existing vaccines might have limited protection against novel SARS-CoV-2 variants, and SARS-CoV-2 infection might show limited cross-variant immunity (e.g., see [17,18,19,20,21,22,23,24,25,26]).
Novel mutations will appear as long as the virus continues to spread. This emphasizes the need for precautionary measures to reduce the risk of infection, such as mask-wearing, physical distancing, hand hygiene, and surface sanitation. Obviously, among the very important constituents of the successful strategies to control pandemics are the existing and future anti-SARS-CoV-2 vaccines.
2. Can COVID-19 Vaccines Alone Stop the Pandemic?
Available SARS-CoV-2 vaccines stimulate adaptive immune responses against different SARS-CoV-2 Spike (S) proteins and decrease both symptomatic and asymptomatic disease incidence with the development of immunological memory.
The prospective SIREN study among staff working in publicly-funded hospitals in England demonstrated that the BNT162b2 mRNA vaccine could prevent both symptomatic and asymptomatic infection caused by the B1.1.7 (Alpha) variant by 70% (95% CI 55–85) 21 days after the first dose and 85% (74–96) 7 days after the second dose [27]. For the moment, the estimated time of lasting protection from severe disease after vaccination is up to 8 months, even with the decay of neutralizing antibody titers after this period [28].
Despite the high efficacy of available COVID-19 vaccines, SARS-CoV-2 infections can still occur in vaccinated individuals due to the decline in neutralizing antibody titers 7–8 months post-vaccination. Furthermore, the emerging new SARS-CoV-2 variants are one of the most important sources of post-vaccination infections [29]. The reported efficacy against the B.1.351 (Beta) variant first identified in South Africa is 57% against moderate-to-severe COVID-19, and 89% against severe COVID-19 for the Ad26.COV2.S vaccine, and zero percent against mild-to-moderate COVID-19 for the ChAdOx1 nCoV-19 vaccine [30]. Two cases out of 417 who had received the second dose of the BNT162b2 or the mRNA-1273 vaccines developed breakthrough infection two weeks after vaccination despite evidence of vaccine efficacy. Breakthrough infections correspond to cases when individuals test positive for COVID-19 after they have been fully vaccinated against the disease [31]. Subsequent viral sequencing revealed the E484K, T95I, del142–144, and D614G mutations in SARS-CoV-2 responsible for those breakthrough infections [32].
Concerns about not acquiring post-vaccination herd immunity also include the uneven distribution of vaccines among countries and within individual countries, and the age limit of vaccination (although vaccination is now recommended for everyone ages 5 years and older by the CDC (U.S. Centers for Disease Control and Prevention)). These factors make it more difficult to reach the levels of the vaccinated population needed to achieve herd immunity [33]. A study reported sequencing of over 2000 samples from COVID-19 patients below the age of 19 years and found more than 250 VOCs, with over 70% of the VOCs in children below the age of 12, including 33 cases of B.1.1.7 (Alpha) and 119 of B.1.429/B.1.427 (Epsilon) variants [34].
Another thing to consider is the possibility of virus spread by fully vaccinated individuals without manifesting breakthrough disease. Data released by the CDC reported that vaccinated people infected by the SARS-CoV-2 Delta variant could carry viral loads similar to those of unvaccinated people (https://www.cdc.gov/coronavirus/2019-ncov/variants/delta-variant.html (accessed on 10 December 2021)). Currently, it is not recommended to test vaccinated people following exposure to infection. However, highly tested groups, such as professional sport teams, demonstrated that asymptomatic breakthrough infections among vaccinated people might be higher than reported.
A crucial question related to vaccine efficacy against current and emerging SARS-CoV-2 is whether we are looking in the wrong place. Most of the current vaccine development focuses on the S protein as a target. Unfortunately, the virus mutates faster than humans engineer vaccines, so it might be a good idea to diversify our efforts. Obviously, the inactivated whole virus vaccines can generate a broad repertoire of antibody responses and the SARS-CoV-2-based vaccine from Sinopharm was approved by the WHO (the World Health Organization) for emergency use in May 2021. The surprising observation made in several published reports is that the comparison of the inactivated whole virus vaccines with mRNA vaccines revealed that the mRNA-based vaccines were systematically more efficient and durable than the inactivated whole virus vaccines [35,36,37,38]. Additionally, other initiatives, such as the combination of S, nucleoprotein, and ORF3a sequences have elicited neutralizing antibodies and demonstrated strong CD8 T cell responses against SARS-CoV-2 in most immunized individuals (https://ir.gritstonebio.com/static-files/6a7c26ca-06a6-4295-bf76-83948a341397 (accessed on 10 December 2021)). These observations suggested that the pan-vaccine approach could potentially be a way to outsmart the virus.
3. Breakthrough Infections of SARS-CoV-2 Variants and Community Herd Immunity Build-Up
3.1. Natural Infections
Many recovered patients have been reinfected worldwide, despite being among the population that had acquired immunity against the virus. However, the best-known example of breakthrough of a population with expected established herd immunity was observed in Manaus city (the capital of Amazonas state in northern Brazil), where from June 2020 to October 2020 the SARS-CoV-2 prevalence increased from 60% to more than 70%, a condition which may mirror achieving herd immunity [12]. By January 2021, Manaus had a huge resurgence in cases due to the emergence of a new variant known as P.1 (Gamma), which was responsible for nearly 100% of the new cases [13]. Although the population may have reached a high herd immunity threshold, there is still a risk of resurgence of new variants escaping protection. Sabino et al. attributed this to the waning of protective antibodies levels and emergence of new SARS-CoV-2 variants [12,13].
3.2. Vaccination
On a personal level, recovered patients can be reinfected, and the vaccinated persons can contract new infections (despite being previously infected or not). For example, the CDC reported that Massachusetts (USA) has already fully vaccinated more than 4 million people (out of 7.03 million total) (56.899%). The total rate of fully vaccinated people in the Barnstable County is 76% [31], with the vaccination levels by age category being as follows: 86% (75+), 77% (50–64), 80% (30–49), 62% (20–29), 64% (16–19), and 43% (12–15) (https://www.wcvb.com/article/massachusetts-covid-breakthrough-cases-delta-variant-pandemic-vaccine-data-charts-maps/37089843 (accessed on 10 December 2021)). However, the Department of Public Health of Massachusetts reported 7737 total COVID-19 breakthrough cases in fully vaccinated individuals as of 3 August 2021. Among the breakthrough cases, 395 patients have been hospitalized, and 100 have died. Genomic sequencing of specimens from 133 patients revealed that this breakthrough was caused by the newly identified and highly transmissible SARS-CoV-2 variants. The B.1.617.2 (Delta) variant was identified in 119 (89%) patients, and one person was infected by the Delta AY.3 sublineage (1%). Since the youngest age group (12–15) showed the lowest coverage by one and/or two vaccine doses (43% vs. >60% in other age groups), these data indicated the need for higher vaccination levels in this group. Although many factors, such as hesitancy and shortage of vaccine supply (especially in the developing countries) [39] define low vaccination rates, vaccination is still the most important strategy to prevent severe illness, hospitalization, and death.
The emergence of breakthrough infections has urged many developed countries to call their citizens for additional vaccine booster doses (specifically for those older than 60 years of age) from the same or newly approved vaccines (combined vaccines). However, it is not clear whether such boosting will help overcoming the viral infectivity and/or transmissibility, especially related to emerging new variants. The COVID-19 pandemic incidences worldwide clearly indicate that it might not. Overall, it seems that this virus can overcome herd immunity (formed naturally and/or via vaccination), suggesting that it can prevail in the general population for a long time. Of note, young individuals may represent a live source for new infections, as most of them, when infected, are asymptomatic or show light to moderate symptoms.
As the WHO has emphasized, 90–99% of individuals infected with the SARS-CoV-2 virus develop detectable neutralizing antibodies within four weeks after infection (https://apps.who.int/iris/handle/10665/341241 (accessed on 28 April 2022)), the levels of which decline during 6–12 months post-infection. Of note, the acquired herd immunity would not protect from reinfection but would significantly enhance the chance of building up the hybrid immunity post vaccination with any type of vaccine [40]. In parallel, according to classical immunology, most vaccines can elicit durable immune responses. However, the open questions are: Why does herd immunity (from natural infection or vaccination) against SARS-CoV-2 decrease within 6–12 months? What are the mechanisms behind such a decay? Are they due to the viral factors, or host immunity factors, or vaccine ingredients or because of all these factors together?
The SARS-CoV-2-specific antibody titer kinetics and decay have been evaluated in individuals vaccinated with the BNT162b2 mRNA vaccine and in convalescent COVID-19 patients [41,42]. Although higher SARS-CoV-2 IgG antibody titers were observed in vaccinated individuals, their titers decreased by 38% each subsequent month. In contrast, less than 5% reduction was observed in convalescent patients. At 6 months post-vaccination, 16.1% of vaccinated individual exhibited antibody levels below the seropositivity threshold. In contrast, 9 months after testing positive for COVID-19, 10.8% of convalescent patients were below the <50 AU/mL threshold [41]. This suggested that different kinetics of antibody levels were detected between vaccinated individuals and convalescent patients, where the BNT162b2 mRNA vaccine provided higher initial antibody levels but faster exponential decrease. In another study, antibody titers demonstrated a 70% decrease in healthcare professionals 6 months post-vaccination, although the titers were one order of magnitude higher compared to seropositive individuals [42].
Taking all these facts into account, it is clear that the war against COVID-19 should be conducted at multiple levels (see Figure 1), and our efforts to end virus transmission, complications, lockdowns, and disruption of the world economy should not be limited to only endorsing various approaches of vaccination (e.g., promoting different vaccines, encouraging booster doses, recommending combined vaccines, re-engineering existing vaccines to target emerging variants, etc.). We should also definitely use various antibody-based immunotherapeutic approaches, such as convalescent plasma and intravenous immunoglobulins to manage COVID-19 patients [43]. Moreover, one should not neglect the important roles of monoclonal antibody therapeutics (mAb), although they are expensive and not applicable for large populations. They are the key antiviral reagents for individuals refusing vaccination, immune compromised patients, or non-hospitalized patients with laboratory-confirmed SARS-CoV-2 infection showing mild to moderate COVID-19 and who are at high risk for progressing to severe disease and/or hospitalization [44,45,46,47,48]. Moreover, mAbs might be useful in cases where vaccine efficacy against new variants is insufficient. In fact, several anti-SARS-CoV-2 mAb combinations of bamlanivimab and etesevimab, casirivimab and imdevimab (REGEN-COV), sotrovimab, and a long-acting anti-SARS-CoV-2 mAb combination, tixagevimab and cilgavimab (Evusheld), have received Emergency Use Authorizations (EUAs) from the Food and Drug Administration (FDA). However, it has been shown that bamlanivimab and etesevimab and REGEN-COV have demonstrated reduced efficiency against the B.1.1.529 (Omicron) variant of SARS-CoV-2. Furthermore, although the efficacy against the original Omicron variant was retained in the case of sotrovimab [49,50], the appearance of the Omicron BA.2 variant has changed this situation, and FDA restricted the use of sotrovimab, as the authorized dose of this mAb is unlikely to be effective against the BA.2 sub-variant [51]. All this indicates that the battle with COVID-19 is challenging, and victory requires utilization of all possible means.
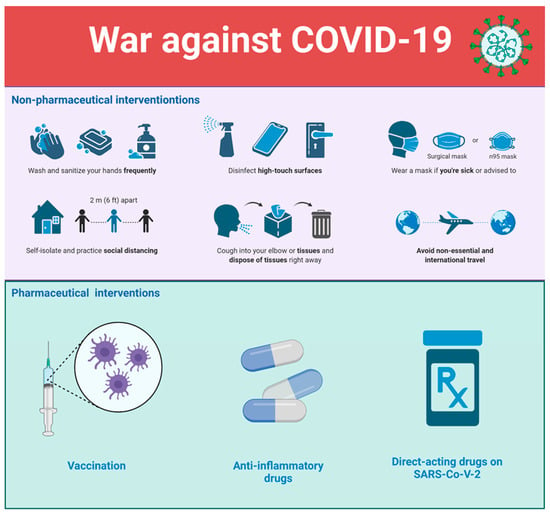
Figure 1.
The war against COVID-19 includes multiple strategies. Both pharmaceutical and non-pharmaceutical interventions are needed now and may be needed for a long time.
In light of these considerations, we also need to place greater emphasis on the various hygienic precautions. Therefore, non-pharmaceutical interventions and protective precautionary actions such as use of protective masks, frequent disinfection of public areas, and social distancing, especially indoors, should continue to remain an important and mandatory part of our daily life. One should keep in mind that when the non-pharmaceutical interventions are relaxed when the majority of the population has already been vaccinated, the probability of the emergence of a new resistant strain is greatly increased. Therefore, individuals should be encouraged to maintain the non-pharmaceutical interventions and transmission-reducing behaviors throughout the entire vaccination period [52]. Obviously, the development of novel drugs directly targeting the SARS-CoV-2 would significantly help reducing viral transmission. Furthermore, we need to understand better what represents a protective or fully protective immunity threshold for SARS-CoV-2 infection, and, similar to almost all approved vaccines for human beings, the world needs to achieve a consensus on the protective immunity threshold [53]. To this end, standard or consensus methods that would consider various correlations of different in vitro and in vivo data are needed to estimate the quality and quantity of protective immunity against SARS-CoV-2 [9,54,55].
Author Contributions
Conceptualization, E.M.R., F.E. and V.N.U.; investigation, E.M.R., F.E. and V.N.U.; data curation, E.M.R., F.E., A.A.A.A., W.B.-d.-C., D.B., A.M.B., S.S.H., K.L., Á.S.-A., K.T., M.M.T., B.D.U. and V.N.U.; writing—original draft preparation, E.M.R., F.E. and V.N.U.; writing—review and editing, E.M.R., F.E., A.A.A.A., W.B.-d.-C., D.B., A.M.B., S.S.H., K.L., Á.S.-A., K.T., M.M.T., B.D.U. and V.N.U.; visualization, M.M.T.; supervision, E.M.R., F.E. and V.N.U.; project administration, E.M.R. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Turner, J.S.; Kim, W.; Kalaidina, E.; Goss, C.W.; Rauseo, A.M.; Schmitz, A.J.; Hansen, L.; Haile, A.; Klebert, M.K.; Pusic, I.; et al. SARS-CoV-2 infection induces long-lived bone marrow plasma cells in humans. Nature 2021, 595, 421–425. [Google Scholar] [CrossRef] [PubMed]
- Hall, V.J.; Foulkes, S.; Charlett, A.; Atti, A.; Monk, E.J.M.; Simmons, R.; Wellington, E.; Cole, M.J.; Saei, A.; Oguti, B.; et al. SARS-CoV-2 infection rates of antibody-positive compared with antibody-negative health-care workers in England: A large, multicentre, prospective cohort study (SIREN). Lancet 2021, 397, 1459–1469. [Google Scholar] [CrossRef]
- Quast, I.; Tarlinton, D. B cell memory: Understanding COVID-19. Immunity 2021, 54, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zuiani, A.; Fischinger, S.; Mullur, J.; Atyeo, C.; Travers, M.; Lelis, F.J.N.; Pullen, K.M.; Martin, H.; Tong, P.; et al. Quick COVID-19 Healers Sustain Anti-SARS-CoV-2 Antibody Production. Cell 2020, 183, 1496–1507.e1416. [Google Scholar] [CrossRef] [PubMed]
- Choe, P.G.; Kang, C.K.; Suh, H.J.; Jung, J.; Song, K.H.; Bang, J.H.; Kim, E.S.; Kim, H.B.; Park, S.W.; Kim, N.J.; et al. Waning Antibody Responses in Asymptomatic and Symptomatic SARS-CoV-2 Infection. Emerg. Infect. Dis. 2021, 27, 327. [Google Scholar] [CrossRef] [PubMed]
- Masia, M.; Fernandez-Gonzalez, M.; Telenti, G.; Agullo, V.; Garcia, J.A.; Padilla, S.; Garcia-Abellan, J.; Galiana, A.; Gonzalo-Jimenez, N.; Gutierrez, F. Durable antibody response one year after hospitalization for COVID-19: A longitudinal cohort study. J. Autoimmun. 2021, 123, 102703. [Google Scholar] [CrossRef]
- Choe, P.G.; Kim, K.H.; Kang, C.K.; Suh, H.J.; Kang, E.; Lee, S.Y.; Kim, N.J.; Yi, J.; Park, W.B.; Oh, M.D. Antibody Responses One Year after Mild SARS-CoV-2 Infection. J. Korean Med. Sci. 2021, 36, e157. [Google Scholar] [CrossRef]
- Bosnjak, B.; Stein, S.C.; Willenzon, S.; Cordes, A.K.; Puppe, W.; Bernhardt, G.; Ravens, I.; Ritter, C.; Schultze-Florey, C.R.; Godecke, N.; et al. Low serum neutralizing anti-SARS-CoV-2 S antibody levels in mildly affected COVID-19 convalescent patients revealed by two different detection methods. Cell Mol. Immunol. 2021, 18, 936–944. [Google Scholar] [CrossRef]
- Perez-Saez, J.; Zaballa, M.E.; Yerly, S.; Andrey, D.O.; Meyer, B.; Eckerle, I.; Balavoine, J.F.; Chappuis, F.; Pittet, D.; Trono, D.; et al. Persistence of anti-SARS-CoV-2 antibodies: Immunoassay heterogeneity and implications for serosurveillance. Clin. Microbiol. Infect. 2021, 27, 1695-e7. [Google Scholar] [CrossRef]
- Boyton, R.J.; Altmann, D.M. Risk of SARS-CoV-2 reinfection after natural infection. Lancet 2021, 397, 1161–1163. [Google Scholar] [CrossRef]
- Ledford, H. Coronavirus reinfections: Three questions scientists are asking. Nature 2020, 585, 168–169. [Google Scholar] [CrossRef] [PubMed]
- Faria, N.R.; Mellan, T.A.; Whittaker, C.; Claro, I.M.; Candido, D.D.S.; Mishra, S.; Crispim, M.A.E.; Sales, F.C.S.; Hawryluk, I.; McCrone, J.T.; et al. Genomics and epidemiology of the P.1 SARS-CoV-2 lineage in Manaus, Brazil. Science 2021, 372, 815–821. [Google Scholar] [CrossRef] [PubMed]
- Sabino, E.C.; Buss, L.F.; Carvalho, M.P.S.; Prete, C.A., Jr.; Crispim, M.A.E.; Fraiji, N.A.; Pereira, R.H.M.; Parag, K.V.; da Silva Peixoto, P.; Kraemer, M.U.G.; et al. Resurgence of COVID-19 in Manaus, Brazil, despite high seroprevalence. Lancet 2021, 397, 452–455. [Google Scholar] [CrossRef]
- Cele, S.; Jackson, L.; Khoury, D.S.; Khan, K.; Moyo-Gwete, T.; Tegally, H.; San, J.E.; Cromer, D.; Scheepers, C.; Amoako, D.G.; et al. Omicron extensively but incompletely escapes Pfizer BNT162b2 neutralization. Nature 2022, 602, 654–656. [Google Scholar] [CrossRef]
- Collie, S.; Champion, J.; Moultrie, H.; Bekker, L.G.; Gray, G. Effectiveness of BNT162b2 Vaccine against Omicron Variant in South Africa. N. Engl. J. Med. 2022, 386, 494–496. [Google Scholar] [CrossRef]
- Khandia, R.; Singhal, S.; Alqahtani, T.; Kamal, M.A.; El-Shall, N.A.; Nainu, F.; Desingu, P.A.; Dhama, K. Emergence of SARS-CoV-2 Omicron (B.1.1.529) variant, salient features, high global health concerns and strategies to counter it amid ongoing COVID-19 pandemic. Environ. Res. 2022, 209, 112816. [Google Scholar] [CrossRef]
- Araf, Y.; Akter, F.; Tang, Y.D.; Fatemi, R.; Parvez, M.S.A.; Zheng, C.; Hossain, M.G. Omicron variant of SARS-CoV-2: Genomics, transmissibility, and responses to current COVID-19 vaccines. J. Med. Virol. 2022, 94, 1825–1832. [Google Scholar] [CrossRef]
- Cao, Y.; Wang, J.; Jian, F.; Xiao, T.; Song, W.; Yisimayi, A.; Huang, W.; Li, Q.; Wang, P.; An, R.; et al. Omicron escapes the majority of existing SARS-CoV-2 neutralizing antibodies. Nature 2022, 602, 657–663. [Google Scholar] [CrossRef]
- Chemaitelly, H.; Abu-Raddad, L.J. Waning effectiveness of COVID-19 vaccines. Lancet 2022, 399, 771–773. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kruger, N.; Schulz, S.; Cossmann, A.; Rocha, C.; Kempf, A.; Nehlmeier, I.; Graichen, L.; Moldenhauer, A.S.; Winkler, M.S.; et al. The Omicron variant is highly resistant against antibody-mediated neutralization: Implications for control of the COVID-19 pandemic. Cell 2022, 185, 447–456.e411. [Google Scholar] [CrossRef]
- Malik, J.A.; Ahmed, S.; Mir, A.; Shinde, M.; Bender, O.; Alshammari, F.; Ansari, M.; Anwar, S. The SARS-CoV-2 mutations versus vaccine effectiveness: New opportunities to new challenges. J. Infect. Public Health 2022, 15, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Sievers, B.L.; Chakraborty, S.; Xue, Y.; Gelbart, T.; Gonzalez, J.C.; Cassidy, A.G.; Golan, Y.; Prahl, M.; Gaw, S.L.; Arunachalam, P.S.; et al. Antibodies elicited by SARS-CoV-2 infection or mRNA vaccines have reduced neutralizing activity against Beta and Omicron pseudoviruses. Sci. Transl. Med. 2022, 14, eabn7842. [Google Scholar] [CrossRef] [PubMed]
- Suryawanshi, R.K.; Chen, I.P.; Ma, T.; Syed, A.M.; Brazer, N.; Saldhi, P.; Simoneau, C.R.; Ciling, A.; Khalid, M.M.; Sreekumar, B.; et al. Limited Cross-Variant Immunity after Infection with the SARS-CoV-2 Omicron Variant Without Vaccination. medRxiv 2022. [Google Scholar] [CrossRef]
- Syed, A.M.; Ciling, A.; Khalid, M.M.; Sreekumar, B.; Chen, P.Y.; Kumar, G.R.; Silva, I.; Milbes, B.; Kojima, N.; Hess, V.; et al. Omicron mutations enhance infectivity and reduce antibody neutralization of SARS-CoV-2 virus-like particles. medRxiv 2022. [Google Scholar] [CrossRef]
- Tseng, H.F.; Ackerson, B.K.; Luo, Y.; Sy, L.S.; Talarico, C.A.; Tian, Y.; Bruxvoort, K.J.; Tubert, J.E.; Florea, A.; Ku, J.H.; et al. Effectiveness of mRNA-1273 against SARS-CoV-2 Omicron and Delta variants. Nat. Med. 2022. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.Y.; Wang, W.B.; Gao, R.D.; Zhou, A.M. Omicron variant (B.1.1.529) of SARS-CoV-2: Mutation, infectivity, transmission, and vaccine resistance. World J. Clin. Cases 2022, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hall, V.J.; Foulkes, S.; Saei, A.; Andrews, N.; Oguti, B.; Charlett, A.; Wellington, E.; Stowe, J.; Gillson, N.; Atti, A.; et al. COVID-19 vaccine coverage in health-care workers in England and effectiveness of BNT162b2 mRNA vaccine against infection (SIREN): A prospective, multicentre, cohort study. Lancet 2021, 397, 1725–1735. [Google Scholar] [CrossRef]
- Khoury, D.S.; Cromer, D.; Reynaldi, A.; Schlub, T.E.; Wheatley, A.K.; Juno, J.A.; Subbarao, K.; Kent, S.J.; Triccas, J.A.; Davenport, M.P. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat. Med. 2021, 27, 1205–1211. [Google Scholar] [CrossRef]
- Irfan, N.; Chagla, Z. In South Africa, a 2-dose Oxford/AZ vaccine did not prevent mild to moderate COVID-19 (cases mainly B.1.351 variant). Ann. Intern. Med. 2021, 174, JC50. [Google Scholar] [CrossRef]
- Madhi, S.A.; Baillie, V.; Cutland, C.L.; Voysey, M.; Koen, A.L.; Fairlie, L.; Padayachee, S.D.; Dheda, K.; Barnabas, S.L.; Bhorat, Q.E.; et al. Efficacy of the ChAdOx1 nCoV-19 COVID-19 Vaccine against the B.1.351 Variant. N. Engl. J. Med. 2021, 384, 1885–1898. [Google Scholar] [CrossRef]
- Brown, C.M.; Vostok, J.; Johnson, H.; Burns, M.; Gharpure, R.; Sami, S.; Sabo, R.T.; Hall, N.; Foreman, A.; Schubert, P.L.; et al. Outbreak of SARS-CoV-2 Infections, Including COVID-19 Vaccine Breakthrough Infections, Associated with Large Public Gatherings—Barnstable County, Massachusetts, July 2021. Morb. Mortal. Wkly. Rep. 2021, 70, 1059–1062. [Google Scholar] [CrossRef] [PubMed]
- Hacisuleyman, E.; Hale, C.; Saito, Y.; Blachere, N.E.; Bergh, M.; Conlon, E.G.; Schaefer-Babajew, D.J.; DaSilva, J.; Muecksch, F.; Gaebler, C.; et al. Vaccine Breakthrough Infections with SARS-CoV-2 Variants. N. Engl. J. Med. 2021, 384, 2212–2218. [Google Scholar] [CrossRef] [PubMed]
- Aschwanden, C. Five reasons why COVID herd immunity is probably impossible. Nature 2021, 591, 520–522. [Google Scholar] [CrossRef] [PubMed]
- Bard, J.D.; Bootwalla, M.; Leber, A.; Planet, P.; Moustafa, A.M.; Harris, R.; Chen, P.Y.; Shen, L.; Ostrow, D.; Maglinte, D.; et al. Emergence of SARS-CoV-2 variants of concern in the pediatric population of the United States. medRxiv 2021. [Google Scholar] [CrossRef]
- Sester, M.; Becker, S.L. Boosting immunity after CoronaVac. Lancet 2022, 399, 496–497. [Google Scholar] [CrossRef]
- Costa Clemens, S.A.; Weckx, L.; Clemens, R.; Almeida Mendes, A.V.; Ramos Souza, A.; Silveira, M.B.V.; da Guarda, S.N.F.; de Nobrega, M.M.; de Moraes Pinto, M.I.; Gonzalez, I.G.S.; et al. Heterologous versus homologous COVID-19 booster vaccination in previous recipients of two doses of CoronaVac COVID-19 vaccine in Brazil (RHH-001): A phase 4, non-inferiority, single blind, randomised study. Lancet 2022, 399, 521–529. [Google Scholar] [CrossRef]
- Kwok, S.L.; Cheng, S.M.; Leung, J.N.; Leung, K.; Lee, C.K.; Peiris, J.M.; Wu, J.T. Waning antibody levels after COVID-19 vaccination with mRNA Comirnaty and inactivated CoronaVac vaccines in blood donors, Hong Kong, April 2020 to October 2021. Eurosurveillance 2022, 27, 2101197. [Google Scholar] [CrossRef]
- Saure, D.; O’Ryan, M.; Torres, J.P.; Zuniga, M.; Santelices, E.; Basso, L.J. Dynamic IgG seropositivity after rollout of CoronaVac and BNT162b2 COVID-19 vaccines in Chile: A sentinel surveillance study. Lancet Infect. Dis. 2022, 22, 56–63. [Google Scholar] [CrossRef]
- Solis Arce, J.S.; Warren, S.S.; Meriggi, N.F.; Scacco, A.; McMurry, N.; Voors, M.; Syunyaev, G.; Malik, A.A.; Aboutajdine, S.; Adeojo, O.; et al. COVID-19 vaccine acceptance and hesitancy in low- and middle-income countries. Nat. Med. 2021, 27, 1385–1394. [Google Scholar] [CrossRef]
- Goldblatt, D. SARS-CoV-2: From herd immunity to hybrid immunity. Nat. Rev. Immunol. 2022. [Google Scholar] [CrossRef]
- Israel, A.; Shenhar, Y.; Green, I.; Merzon, E.; Golan-Cohen, A.; Schaffer, A.A.; Ruppin, E.; Vinker, S.; Magen, E. Large-Scale Study of Antibody Titer Decay following BNT162b2 mRNA Vaccine or SARS-CoV-2 Infection. Vaccines 2021, 10, 64. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, D.; Clementi, N.; Criscuolo, E.; Ambrosi, A.; Corea, F.; Di Resta, C.; Tomaiuolo, R.; Mancini, N.; Locatelli, M.; Plebani, M.; et al. Antibody Titer Kinetics and SARS-CoV-2 Infections Six Months after Administration with the BNT162b2 Vaccine. Vaccines 2021, 9, 1357. [Google Scholar] [CrossRef] [PubMed]
- Sharun, K.; Tiwari, R.; Iqbal Yatoo, M.; Patel, S.K.; Natesan, S.; Dhama, J.; Malik, Y.S.; Harapan, H.; Singh, R.K.; Dhama, K. Antibody-based immunotherapeutics and use of convalescent plasma to counter COVID-19: Advances and prospects. Expert Opin. Biol. Ther. 2020, 20, 1033–1046. [Google Scholar] [CrossRef] [PubMed]
- Ahangarzadeh, S.; Payandeh, Z.; Arezumand, R.; Shahzamani, K.; Yarian, F.; Alibakhshi, A. An update on antiviral antibody-based biopharmaceuticals. Int. Immunopharmacol. 2020, 86, 106760. [Google Scholar] [CrossRef]
- Klasse, P.J.; Moore, J.P. Antibodies to SARS-CoV-2 and their potential for therapeutic passive immunization. Elife 2020, 9, e57877. [Google Scholar] [CrossRef]
- Jahanshahlu, L.; Rezaei, N. Monoclonal antibody as a potential anti-COVID-19. Biomed. Pharmacother. 2020, 129, 110337. [Google Scholar] [CrossRef]
- Sempowski, G.D.; Saunders, K.O.; Acharya, P.; Wiehe, K.J.; Haynes, B.F. Pandemic Preparedness: Developing Vaccines and Therapeutic Antibodies For COVID-19. Cell 2020, 181, 1458–1463. [Google Scholar] [CrossRef]
- Saghazadeh, A.; Rezaei, N. Towards treatment planning of COVID-19: Rationale and hypothesis for the use of multiple immunosuppressive agents: Anti-antibodies, immunoglobulins, and corticosteroids. Int. Immunopharmacol. 2020, 84, 106560. [Google Scholar] [CrossRef]
- Deng, J.; Heybati, K.; Ramaraju, H.B.; Zhou, F.; Rayner, D.; Heybati, S. Differential efficacy and safety of anti-SARS-CoV-2 antibody therapies for the management of COVID-19: A systematic review and network meta-analysis. Infection 2022, 1–15. [Google Scholar] [CrossRef]
- Aggarwal, N.R.; Beaty, L.E.; Bennett, T.D.; Carlson, N.E.; Davis, C.B.; Kwan, B.M.; Mayer, D.A.; Ong, T.C.; Russell, S.; Steele, J.; et al. Real World Evidence of the Neutralizing Monoclonal Antibody Sotrovimab for Preventing Hospitalization and Mortality in COVID-19 Outpatients. medRxiv 2022. [Google Scholar] [CrossRef]
- COVID-19 updates: FDA restricts use of sotrovimab. Med. Lett. Drugs Ther. 2022, 64, 64.
- Rella, S.A.; Kulikova, Y.A.; Dermitzakis, E.T.; Kondrashov, F.A. Rates of SARS-CoV-2 transmission and vaccination impact the fate of vaccine-resistant strains. Sci. Rep. 2021, 11, 15729. [Google Scholar] [CrossRef]
- Redwan, E.M. COVID-19 pandemic and vaccination build herd immunity. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 577–579. [Google Scholar] [CrossRef]
- Valdivia, A.; Torres, I.; Latorre, V.; Frances-Gomez, C.; Albert, E.; Gozalbo-Rovira, R.; Alcaraz, M.J.; Buesa, J.; Rodriguez-Diaz, J.; Geller, R.; et al. Inference of SARS-CoV-2 spike-binding neutralizing antibody titers in sera from hospitalized COVID-19 patients by using commercial enzyme and chemiluminescent immunoassays. Eur. J. Clin. Microbiol. Infect. Dis. 2021, 40, 485–494. [Google Scholar] [CrossRef]
- World Health Organization. COVID-19 Natural Immunity: Scientific Brief, 10 May 2021. Available online: https://apps.who.int/iris/handle/10665/341241 (accessed on 10 December 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).