Metal–Insulator Transition in Doped Barium Plumbates
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Density Measurement
2.3. Scanning Electron Microscope (Sem) Analysis
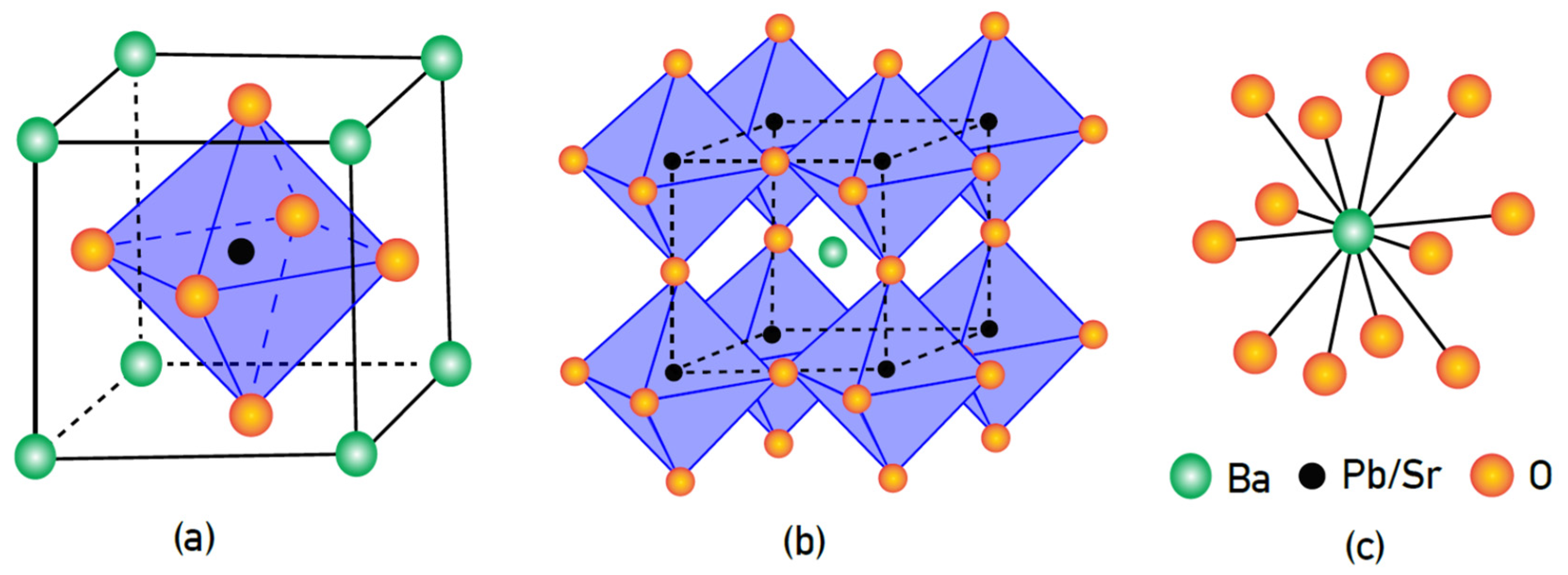
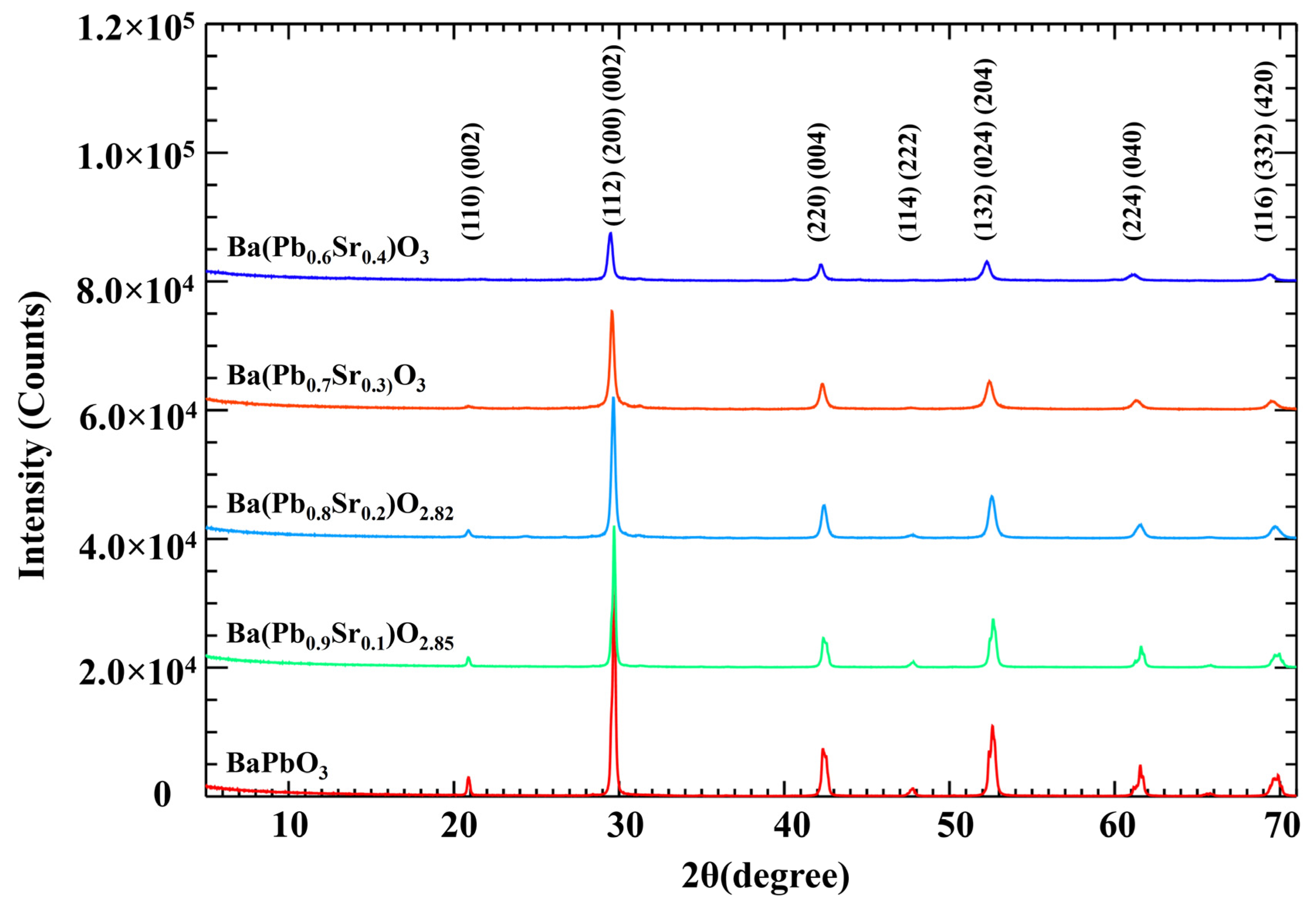
2.4. X-ray Diffraction (Xrd) Analysis
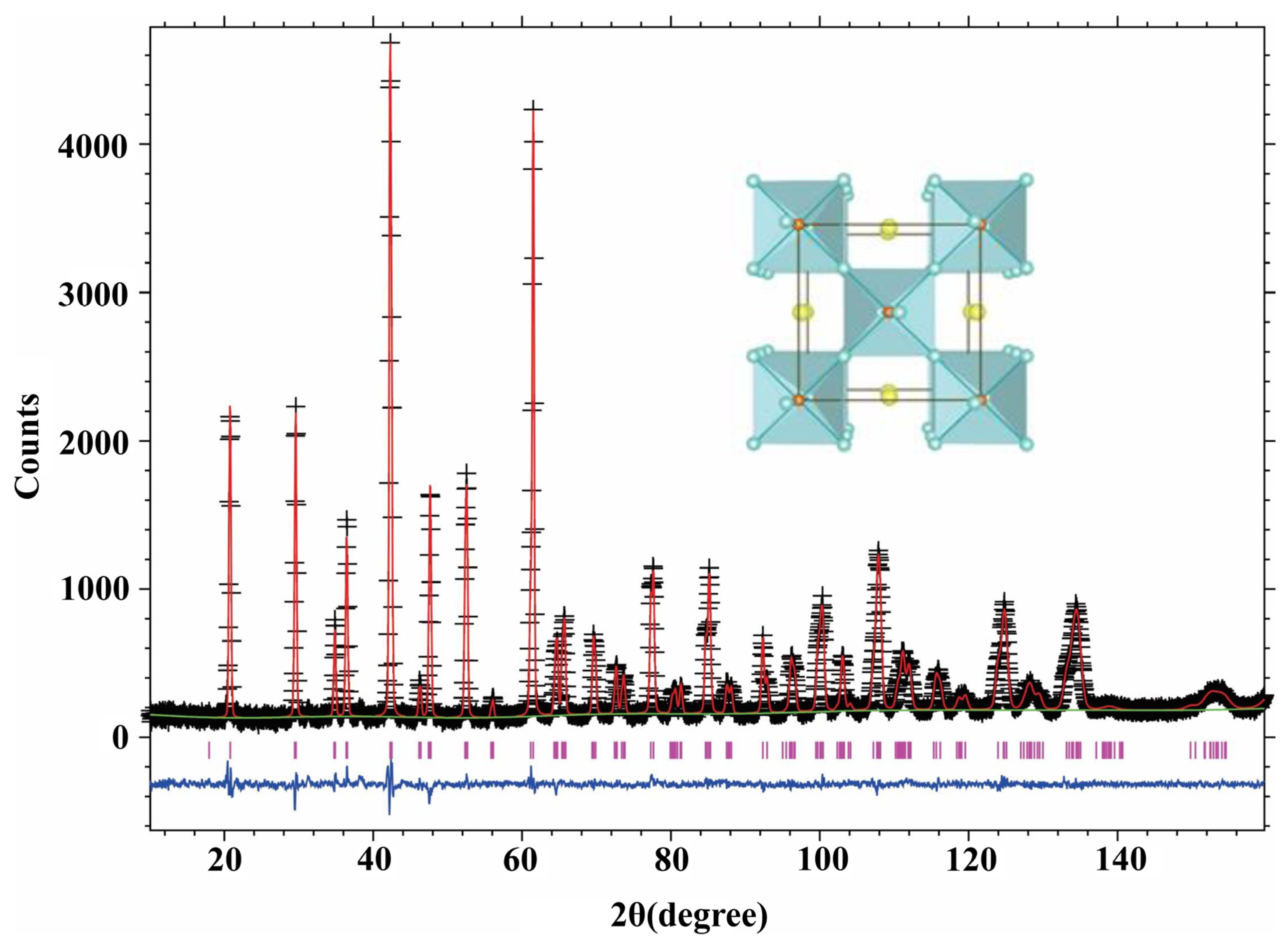
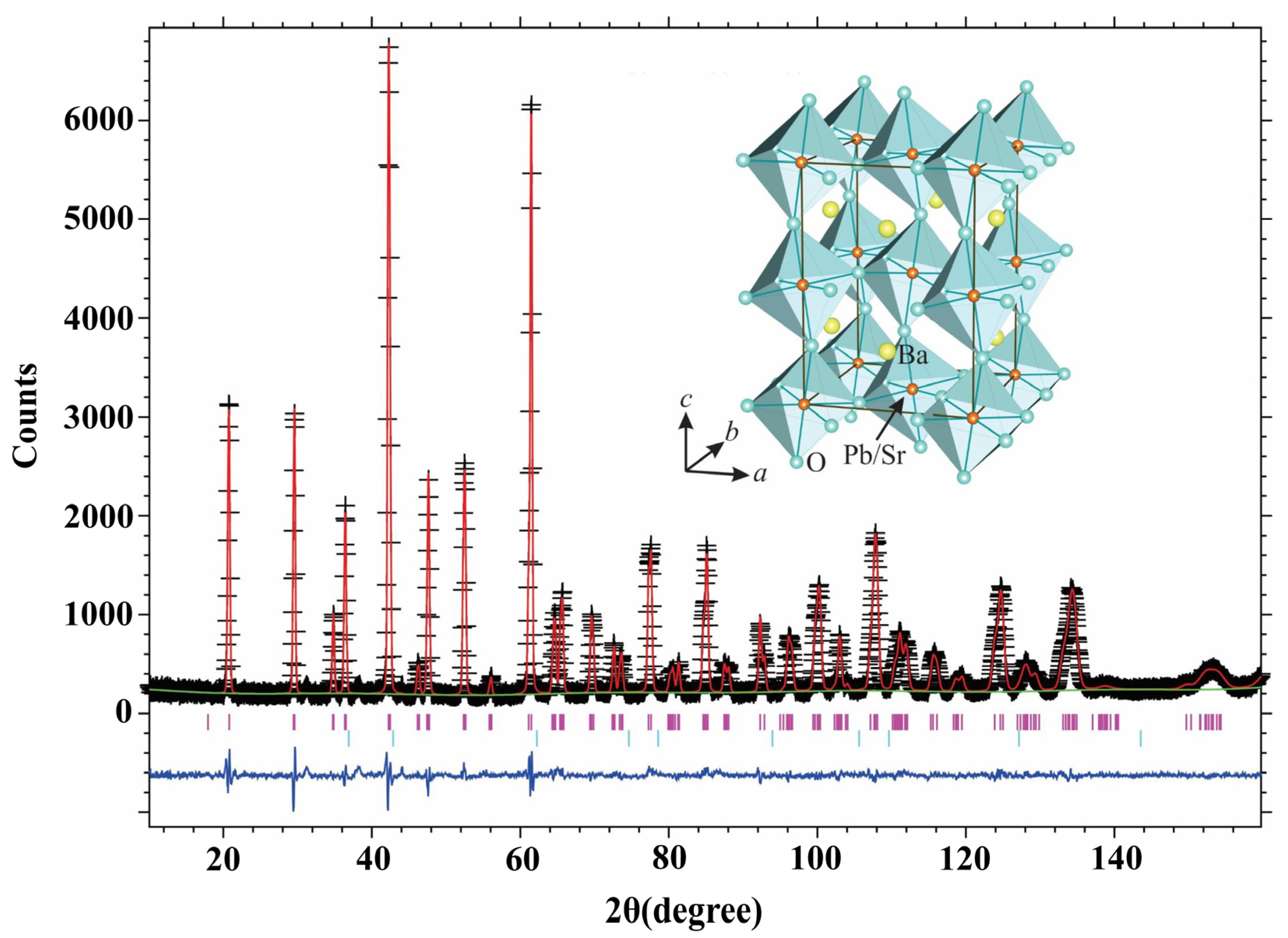
2.5. Neutron Powder Diffraction Analysis
2.6. Electrical Conductivity
2.7. Thermopower
3. Results and Discussion
3.1. Scanning Electron Microscope
3.2. XRD
3.3. Neutron Powder Diffraction
3.4. Thermopower
3.5. Electrical Conductivity
- (i)
- Carriers get increasingly trapped in the lattice, forming polarons, which are known to have considerably less mobility than bare/itinerant electrons. Such polarons favor formation at higher temperatures because of enhanced electron–phonon interaction.
- (ii)
- Sr-doping introduces holes to the valence band that may recombine with the electrons, thereby reducing electron/polaron (carrier) concentration. Reduced carrier concentration also leads to inevitable reduction in conductivity.
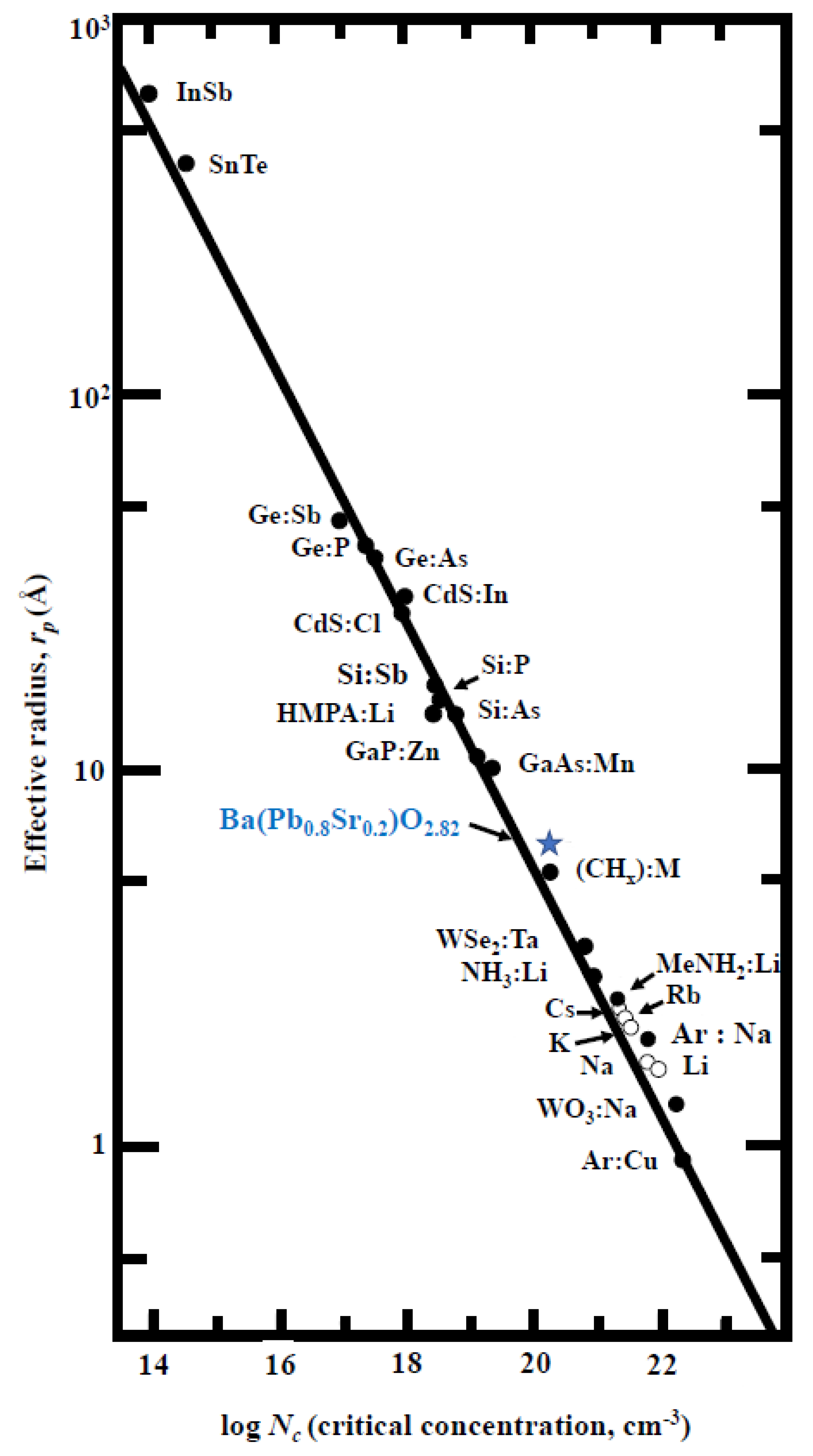
3.5.1. MIT in Ba(Pb1−xSrx)O3−z
Hubbard Model of MIT
Mott Model of MIT
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yasukawa, M.; Murayama, N. High-temperature thermoelectric properties of the oxide material: Ba1−xSrxPbO3 (x = 0–0.6). J. Mater. Sci. Lett. 1997, 16, 1731–1734. [Google Scholar] [CrossRef]
- Zhang, J.; Hu, B.; Xu, J.; Gu, Z.; Zhang, S. Phase, microstructure and conductivity of electron-doped Ba1−xLaxPbO3 ceramics. Ceram. Int. 2014, 40, 10825–10830. [Google Scholar] [CrossRef]
- Fu, W.T.; Ijdo, D.J.W.; Visser, D. High-resolution neutron powder diffraction study on the structure of BaPbO3. Solid State Commun. 2005, 134, 647–652. [Google Scholar] [CrossRef]
- Roth, R.S. Classification of Perovskite and Other ABO3-Type Compounds. J. Res. Natl. Bur. Stand. 1957, 58, 75–78. [Google Scholar] [CrossRef]
- Xin, W.; Yudong, L.; Zhiqiang, Z. Defect Compensating Mechanism Undoped Barium Plumbate. J. Chin. Ceram. Soc. 2007, 35, 701–704. [Google Scholar]
- Fu, W.T.; Ijdo, D.J.W. A Comparative Study on the structure of APbO3 (A = Ba,Sr). Solid State Commun. 1995, 95, 581–585. [Google Scholar] [CrossRef]
- Kodenkandath, T. Structure and properties of substituted BaPbO3 and Ba2PbO4 compounds. Mater. Chem. Phys. 2000, 62, 23–28. [Google Scholar] [CrossRef]
- Menushenkov, A.P.; Klementev, K.V. Extended x-ray absorption fine-structure indication of a double-well potential for oxygen vibration in Ba1−xKxBiO3. J. Phys. Condens. Matter 2000, 12, 3767–3786. [Google Scholar] [CrossRef]
- Yasukawa, M.; Murayama, N. A promising oxide material for high-temperature thermoelectric energy conversion: Ba1−xSrxPbO3 solid solution system. Mater. Sci. Eng. 1998, B54, 64–69. [Google Scholar] [CrossRef]
- Cava, R.J.; Batlogg, B.; Espinosa, G.P.; Ramirez, A.P.; Krajewski, J.J.; Peck, W.F.; Rupp, L.W., Jr.; Cooper, A.S. Superconductivity at 3.5 K in BaPb0.75Sb0.25O3: Why is Tc so low? Nature 1989, 339, 291–293. [Google Scholar] [CrossRef]
- Sleight, A.W.; Gillson, J.L.; Bierstedt, P.E. High-Temperature Superconductivity in The BaPb1−xBixO3 System. Solid State Commun. 1975, 17, 27–28. [Google Scholar] [CrossRef]
- Wignacourt, J.P.; Swinnea, J.S.; Steinfink, H.; Goodenough, J.B. Oxygen atom thermal vibration anisotropy in Ba0.87K0.13BiO3. Appl. Phys. Lett. 1988, 53, 1753–1755. [Google Scholar] [CrossRef]
- Itoh, M.; Sawada, T.; Kim, I.S.; Inaguma, Y.; Nakamura, T. Composition Dependence of Carrier Concentration and Conductivity in (Ba1−xSrx)PbO3-δ. Solid State Commun. 1992, 83, 33–36. [Google Scholar] [CrossRef]
- Kharton, V.V.; Viskup, A.P.; Kovalevsky, A.V.; Naumovich, E.N.; Marques, F.M.B. Ionic transport in oxygen-hyperstoichiometric phases with K2NiF4-type structure. Solid State Ion. 2001, 143, 337–353. [Google Scholar] [CrossRef]
- Mattheiss, L.F. Electronic structure of the Ban+1PbnO3n+1 homologous series. Phys. Rev. B 1990, B42, 359–365. [Google Scholar] [CrossRef]
- Ikushima, H.; Hayakawa, S. Electrical Properties of BaPbO3 Ceramics. Solid State Electron. 1996, 9, 921–925. [Google Scholar] [CrossRef]
- Edwards, P.P.; Johnston, R.L.; Rao, C.N.R.; Tunstall, D.P.; Hensel, F. The metal-insulator transition: A perspective. Philos. Trans. R. Soc. Lond. 1998, 356, 5–22. [Google Scholar] [CrossRef]
- Mott, N.F. The transition to the Metallic State. Philos. Mag. Lett. 1961, 6, 287–309. [Google Scholar] [CrossRef]
- Hubbard, J. Electron Correlations in Narrow Energy Bands. Proc. R. Soc. Lond. 1963, 276, 238–257. [Google Scholar]
- Mott, N. Conduction in Non-Crystalline Materials, 2nd ed.; Oxford Science Publications: Oxford, UK, 1993. [Google Scholar]
- Greaves, G.N. Small Polaron Conduction in V2O5-P2O5 Glasses. J. Non-Cryst. Solids 1973, 11, 427–446. [Google Scholar] [CrossRef]
- Toby, B.H. EXPGUI, A graphical user interface for GSAS. J. Appl. Cryst. 2001, 34, 210–213. [Google Scholar] [CrossRef] [Green Version]
- Larson, A.C.; Von Dreele, R.B. General Structure Analysis System (GSAS); Los Alamos National Laboratory Report LAUR: Los Alamos, NM, USA, 2000; pp. 86–748. [Google Scholar]
- Bhatta, R.P.; Annamalai, S.; Mohr, R.K.; Brandys, M.; Pegg, I.L.; Dutta, B. High temperature thermal conductivity of platinum microwire by 3ω method. Rev. Sci. Instrum. 2010, 81, 114904. [Google Scholar] [CrossRef] [PubMed]
- Eufrasio, A. Electrical and Thermal Transport in Cation Substituted Barium Plumbate. Ph.D. Thesis, The Catholic University of America, Washington, DC, USA, 2019. [Google Scholar]
- Edwards, P.P.; Sienko, M.J. Universality aspects of the metal-nonmetal transition in condensed media. Phys. Rev. 1978, B17, 2575–2581. [Google Scholar] [CrossRef]

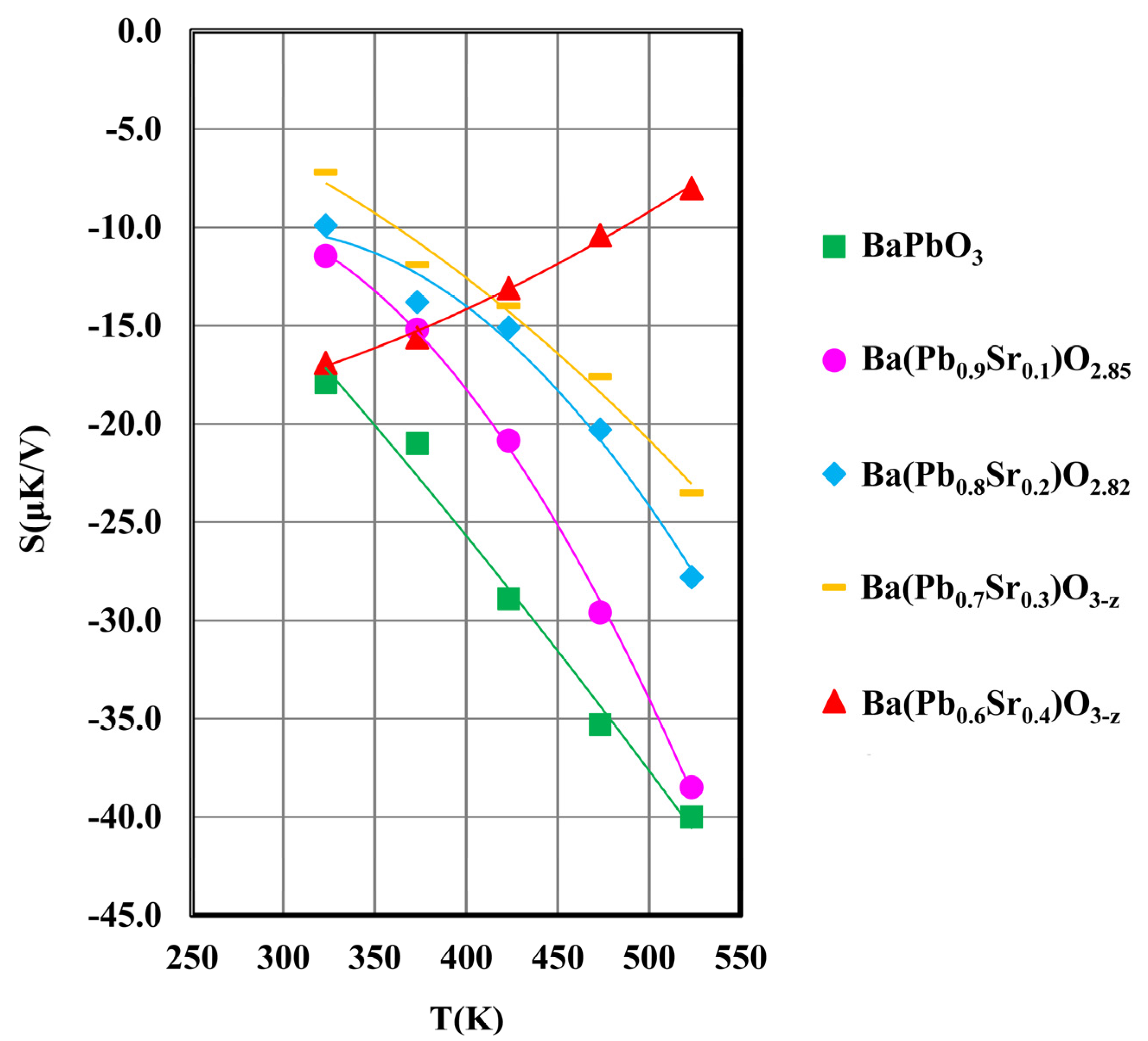

| Composition | Activation Energy, Eg(eV) * | Optical Band Gap (eV) | Density (g/cm3) | Ln(σ) (Ω.m)−1 at 473 K | Thermopower (μV/K) at 473 K |
|---|---|---|---|---|---|
| BaPbO3−z | - | - | 6.23 | 12.21 | −35.3 |
| Ba(Pb0.9Sr0.1)O2.85 | - | - | 6.22 | 11.50 | −29.6 |
| Ba(Pb0.8Sr0.2)O2.82 | 0.096 | 0.191 | 5.86 | 7.65 | −20.3 |
| Ba(Pb0.7Sr0.3)O3−z | 0.101 | 0.202 | 5.68 | 6.72 | −17.6 |
| Ba(Pb0.6Sr0.4)O3−z | 0.240 | 0.480 | 5.50 | 4.36 | −10.4 |
| Atom | Site | x | y | z | n | B11 (Å2) | B22 (Å2) | B33 (Å2) | B12 (Å2) | B13 (Å2) | B23 (Å2) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ba | 4e | 0.5001 (7) | 0 | ¼ | 1 | 1.09 (6) | 1.1 (1) | 0.068 (9) | 0 | 0 | 0 |
| Pb/Sr | 4a | 0 | 0 | 0 | 0.9/0.1 | 0.71 (5) | 0.92 (6) | 0.3 (1) | 0 | −0.10 (6) | - |
| O(1) | 4e | 0.0515 (5) | 0 | ¼ | 0.94 (1) | 1.6 (1) | 3.0 (2) | 0.3 (1) | 0 | 0 | 0 |
| O(2) | 8g | ¼ | ¼ | −0.0275 (2) | 0.956 (8) | 1.76 (8) | 1.8 (1) | 1.86 (8) | −0.86 (9) | 0 | 0 |
| Atom | Site | x | y | z | n | B11 (Å2) | B22 (Å2) | B33 (Å2) | B12 (Å2) | B13 (Å2) | B23 (Å2) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ba | 4e | 0.5002 (6) | 0 | ¼ | 1 | 1.14 (7) | 1.1 (1) | 0.90 (9) | 0 | 0 | 0 |
| Pb/Sr | 4a | 0 | 0 | 0 | 0.79/0.21 (2) | 0.65 (5) | 0.80 (6) | 0.24 (6) | 0 | −0.10 (6) | - |
| O(1) | 4e | 0.0510 (5) | 0 | ¼ | 0.89 (1) | 1.4 (1) | 2.6 (2) | 0.2 (1) | 0 | 0 | 0 |
| O(2) | 8g | ¼ | ¼ | −0.0269 (2) | 0.963 (9) | 1.60 (8) | 2.1 (2) | 1.90 (9) | −0.91(6) | 0 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eufrasio, A.M.; Pegg, I.; Kafle, A.; Wong-Ng, W.; Huang, Q.; Dutta, B. Metal–Insulator Transition in Doped Barium Plumbates. Electron. Mater. 2021, 2, 428-444. https://doi.org/10.3390/electronicmat2030029
Eufrasio AM, Pegg I, Kafle A, Wong-Ng W, Huang Q, Dutta B. Metal–Insulator Transition in Doped Barium Plumbates. Electronic Materials. 2021; 2(3):428-444. https://doi.org/10.3390/electronicmat2030029
Chicago/Turabian StyleEufrasio, Andreza M., Ian Pegg, Amrit Kafle, Winnie Wong-Ng, Qingzhen Huang, and Biprodas Dutta. 2021. "Metal–Insulator Transition in Doped Barium Plumbates" Electronic Materials 2, no. 3: 428-444. https://doi.org/10.3390/electronicmat2030029
APA StyleEufrasio, A. M., Pegg, I., Kafle, A., Wong-Ng, W., Huang, Q., & Dutta, B. (2021). Metal–Insulator Transition in Doped Barium Plumbates. Electronic Materials, 2(3), 428-444. https://doi.org/10.3390/electronicmat2030029






