Resveratrol Effects on Metabolic Syndrome Features: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Eligibility Criteria
2.3. Search Strategy
2.4. Study Selection
2.5. Data Extraction and Quality Assessment
2.6. Publication Bias Assessment
2.7. Statistical Analysis
3. Results
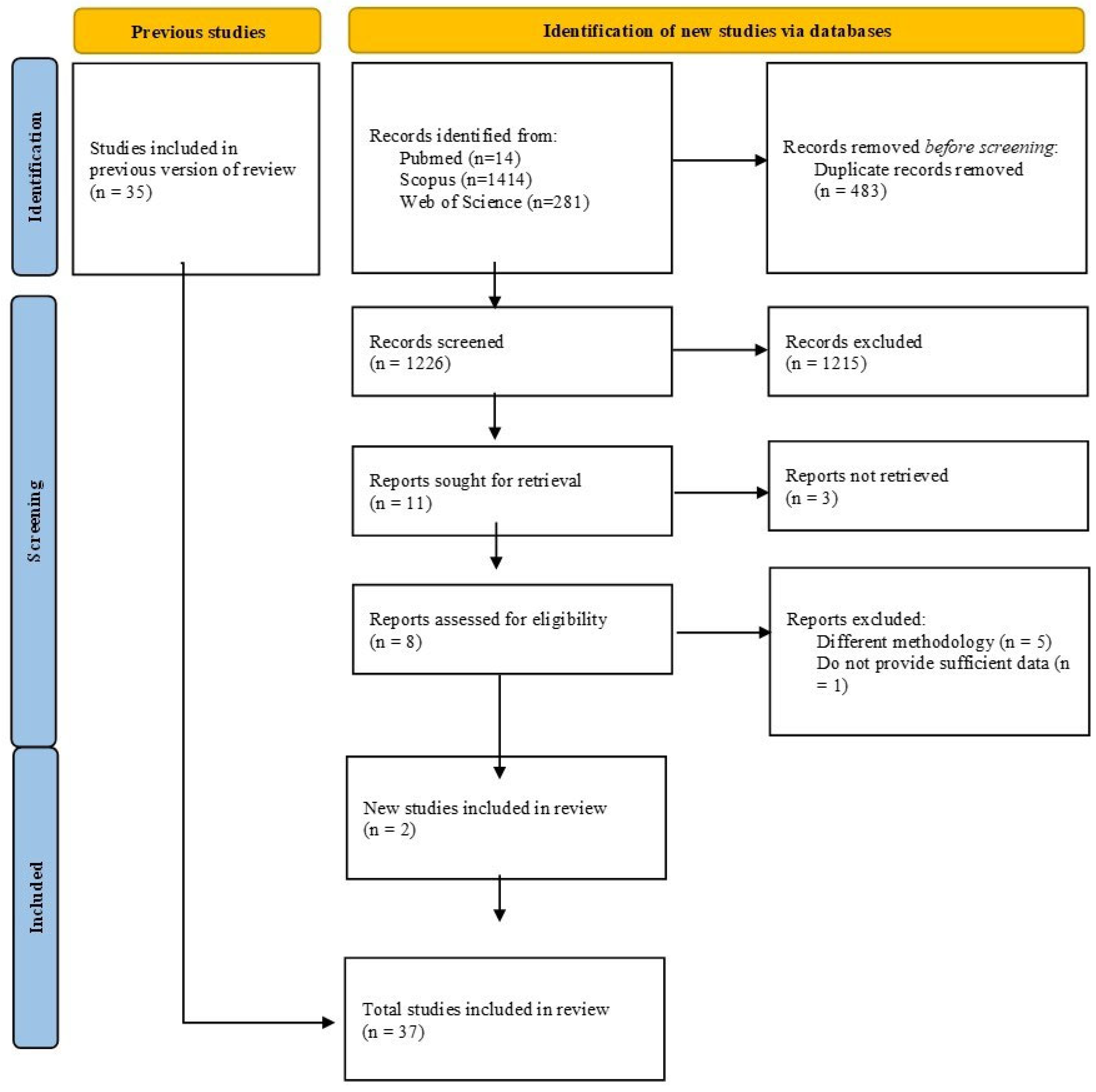
3.1. Eligibility Criteria and Search Strategy
3.2. Characteristics of the Selected Studies
3.3. Quality of Evidence of the Included Studies
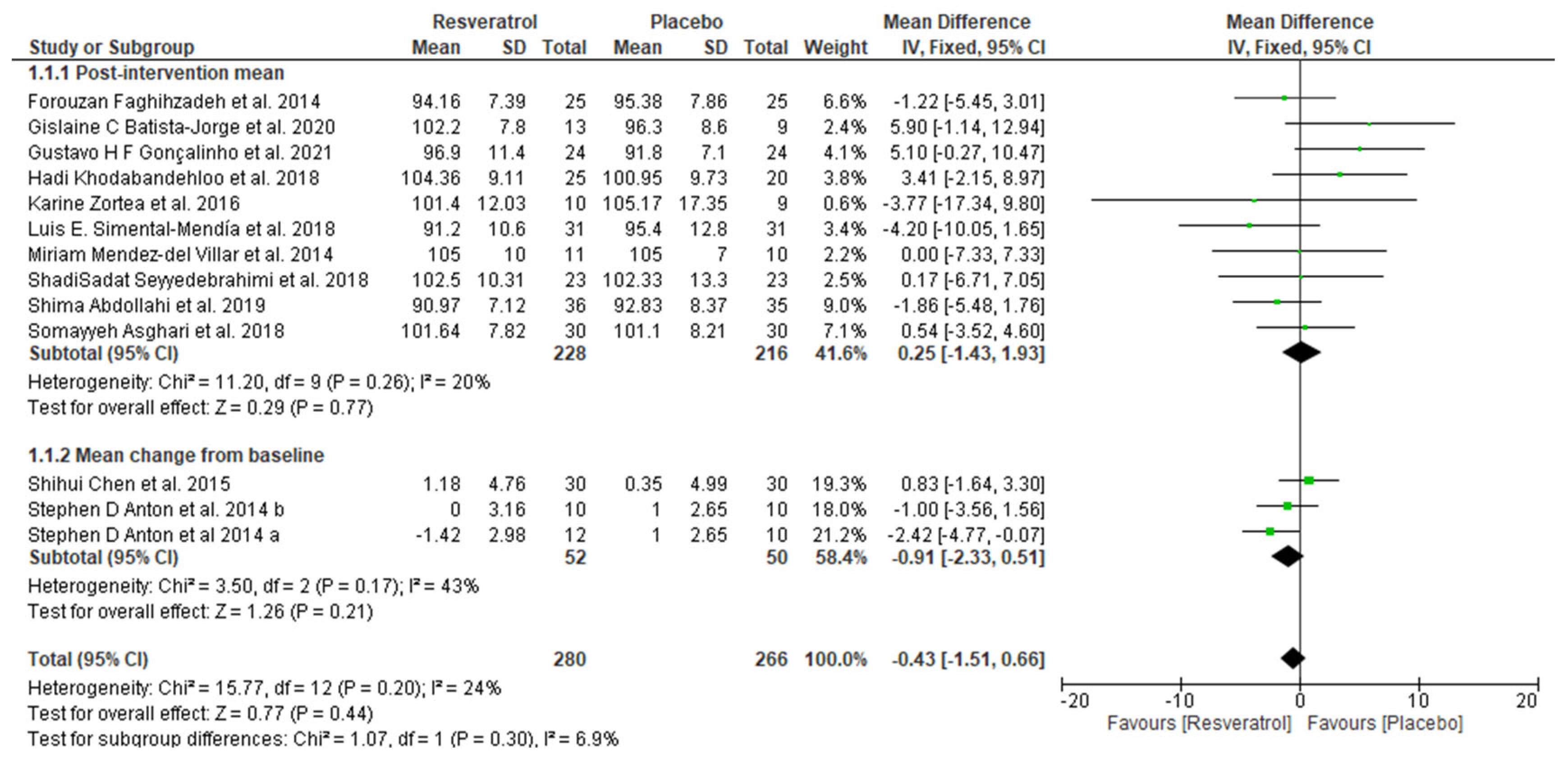
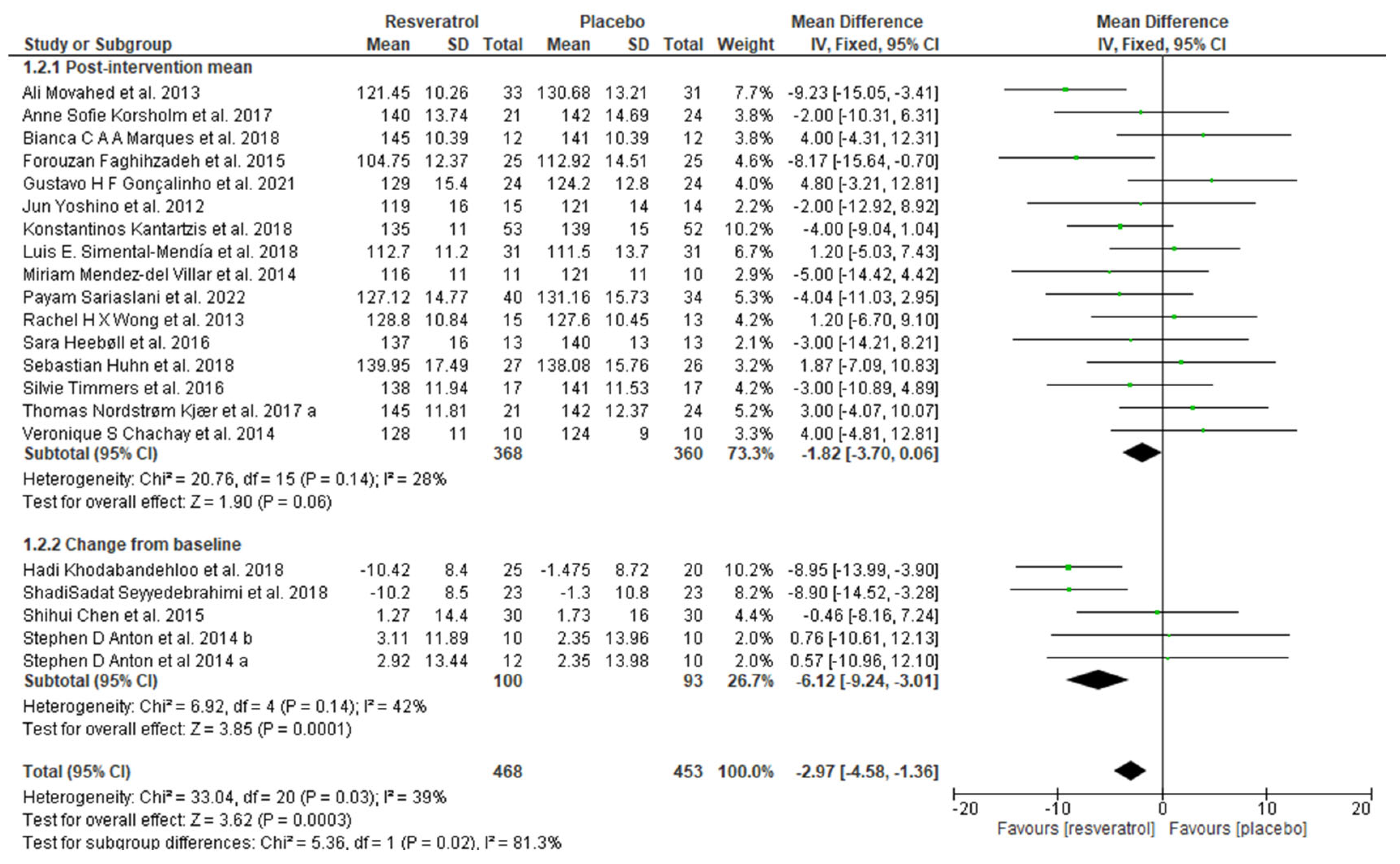
3.4. Meta-Analysis
3.5. Publication Bias Assessment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Saklayen, M.G. The Global Epidemic of the Metabolic Syndrome. Curr. Hypertens. Rep. 2018, 20, 12. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, C.; Williams, K.; Hunt, K.J.; Haffner, S.M. The National Cholesterol Education Program—Adult Treatment Panel III, International Diabetes Federation, and World Health Organization Definitions of the Metabolic Syndrome as Predictors of Incident Cardiovascular Disease and Diabetes. Diabetes Care 2007, 30, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.; Joo, M.; Hong, H.; Kang, H. Eating Speed, Physical Activity, and Cardiorespiratory Fitness Are Independent Predictors of Metabolic Syndrome in Korean University Students. Nutrients 2021, 13, 2420. [Google Scholar] [CrossRef] [PubMed]
- González-González, J.G.; Violante-Cumpa, J.R.; Zambrano-Lucio, M.; Burciaga-Jimenez, E.; Castillo-Morales, P.L.; Garcia-Campa, M.; Solis, R.C.; González-Colmenero, A.D.; Rodríguez-Gutiérrez, R. HOMA-IR as a Predictor of Health Outcomes in Patients with Metabolic Risk Factors: A Systematic Review and Meta-Analysis. High Blood Press. Cardiovasc. Prev. 2022, 29, 547–564. [Google Scholar] [CrossRef] [PubMed]
- Safaei, M.; Sundararajan, E.A.; Driss, M.; Boulila, W.; Shapi’i, A. A Systematic Literature Review on Obesity: Understanding the Causes & Consequences of Obesity and Reviewing Various Machine Learning Approaches Used to Predict Obesity. Comput. Biol. Med. 2021, 136, 104754. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M.; Cleeman, J.I.; Daniels, S.R.; Donato, K.A.; Eckel, R.H.; Franklin, B.A.; Gordon, D.J.; Krauss, R.M.; Savage, P.J.; Smith, S.C.; et al. Diagnosis and Management of the Metabolic Syndrome: An American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation 2005, 112, 2735–2752. [Google Scholar] [CrossRef] [PubMed]
- Catelli De Carvalho, M.H. I Diretriz Brasileira de Diagnóstico e Tratamento Da Síndrome Metabólica. Arq. Bras. Cardiol. 2005, 84 (Suppl. S1), 3–28. [Google Scholar] [CrossRef]
- Liese, A.D.; Mayer-Davis, E.J.; Haffner, S.M. Development of the Multiple Metabolic Syndrome: An Epidemiologic Perspective. Epidemiol. Rev. 1998, 20, 157–172. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, M.M.; Vestergaard, P.F.; Clasen, B.F.; Radko, Y.; Christensen, L.P.; Stødkilde-Jørgensen, H.; Møller, N.; Jessen, N.; Pedersen, S.B.; Jørgensen, J.O.L. High-Dose Resveratrol Supplementation in Obese Men an Investigator- Initiated, Randomized, Placebo-Controlled Clinical Trial of Substrate Metabolism, Insulin Sensitivity, and Body Composition. Diabetes 2013, 62, 1186–1195. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Chen, J.; Wang, Q.; Gál, R.; Halmosi, R.; Gallyas, F.; Tschida, M.; Mutirangura, P.; Tóth, K.; Alexy, T.; et al. Resveratrol and beyond: The Effect of Natural Polyphenols on the Cardiovascular System: A Narrative Review. Biomedicines 2023, 11, 2888. [Google Scholar] [CrossRef]
- Bahramzadeh, A.; Bolandnazar, K.; Meshkani, R. Resveratrol as a Potential Protective Compound against Skeletal Muscle Insulin Resistance. Heliyon 2023, 9, 2405–8440. [Google Scholar] [CrossRef] [PubMed]
- Alberdi, G.; Rodríguez, V.M.; Miranda, J.; Macarulla, M.T.; Arias, N.; Andrés-Lacueva, C.; Portillo, M.P. Changes in White Adipose Tissue Metabolism Induced by Resveratrol in Rats. Nutr. Metab. 2011, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Moustaid-Moussa, N.; Chen, L.; Mo, H.; Shastri, A.; Su, R.; Bapat, P.; Kwun, I.; Shen, C.-L. Novel Insights of Dietary Polyphenols and Obesity. J. Nutr. Biochem. 2014, 25, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Calmasini, F.B.; Silva, F.H.; Alexandre, E.C.; Antunes, E. Efficacy of Resveratrol in Male Urogenital Tract Dysfunctions: An Evaluation of Pre-Clinical Data. Nutr. Res. Rev. 2023, 36, 86–97. [Google Scholar] [CrossRef] [PubMed]
- Zivarpour, P.; Reiner, Ž.; Hallajzadeh, J.; Mirsafaei, L. Resveratrol and Cardiac Fibrosis Prevention and Treatment. Curr. Pharm. Biotechnol. 2022, 23, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Barger, J.L.; Kayo, T.; Vann, J.M.; Arias, E.B.; Wang, J.; Hacker, T.A.; Wang, Y.; Raederstorff, D.; Morrow, J.D.; Leeuwenburgh, C.; et al. A Low Dose of Dietary Resveratrol Partially Mimics Caloric Restriction and Retards Aging Parameters in Mice. PLoS ONE 2008, 3, e2264. [Google Scholar] [CrossRef]
- Faghihzadeh, F.; Adibi, P.; Rafiei, R.; Hekmatdoost, A. Resveratrol Supplementation Improves Inflammatory Biomarkers in Patients with Nonalcoholic Fatty Liver Disease. Nutr. Res. 2014, 34, 837–843. [Google Scholar] [CrossRef]
- Xiao, Y.; Zhang, X.; Yi, D.; Qiu, F.; Wu, L.; Tang, Y.; Wang, N. Mediterranean Diet Affects the Metabolic Outcome of Metabolic Dysfunction-Associated Fatty Liver Disease. Front. Nutr. 2023, 10, 1225946. [Google Scholar] [CrossRef] [PubMed]
- Faghihzadeh, F.; Adibi, P.; Hekmatdoost, A. The Effects of Resveratrol Supplementation on Cardiovascular Risk Factors in Patients with Non-Alcoholic Fatty Liver Disease: A Randomised, Double-Blind, Placebo-Controlled Study. Br. J. Nutr. 2015, 114, 796–803. [Google Scholar] [CrossRef]
- Machado, I.F.; Miranda, R.G.; Dorta, D.J.; Rolo, A.P.; Palmeira, C.M. Targeting Oxidative Stress with Polyphenols to Fight Liver Diseases. Antioxidants 2023, 12, 1212. [Google Scholar] [CrossRef]
- Lee, C.M.Y.; Huxley, R.R.; Wildman, R.P.; Woodward, M. Indices of Abdominal Obesity Are Better Discriminators of Cardiovascular Risk Factors than BMI: A Meta-Analysis. J. Clin. Epidemiol. 2008, 61, 646–653. [Google Scholar] [CrossRef] [PubMed]
- Abdi Dezfouli, R.; Mohammadian Khonsari, N.; Hosseinpour, A.; Asadi, S.; Ejtahed, H.S.; Qorbani, M. Waist to Height Ratio as a Simple Tool for Predicting Mortality: A Systematic Review and Meta-Analysis. Int. J. Obes. 2023, 47, 1286–1301. [Google Scholar] [CrossRef]
- Méndez-Del Villar, M.; González-Ortiz, M.; Martínez-Abundis, E.; Pérez-Rubio, K.G.; Lizárraga-Valdez, R. Effect of Resveratrol Administration on Metabolic Syndrome, Insulin Sensitivity, and Insulin Secretion. Metab. Syndr. Relat. Disord. 2014, 12, 497–501. [Google Scholar] [CrossRef] [PubMed]
- García-Martínez, B.I.; Ruiz-Ramos, M.; Pedraza-Chaverri, J.; Santiago-Osorio, E.; Mendoza-Núñez, V.M. Effect of Resveratrol on Markers of Oxidative Stress and Sirtuin 1 in Elderly Adults with Type 2 Diabetes. Int. J. Mol. Sci. 2023, 24, 7422. [Google Scholar] [CrossRef] [PubMed]
- Batista-Jorge, G.C.; Barcala-Jorge, A.S.; Silveira, M.F.; Lelis, D.F.; Andrade, J.M.O.; de Paula, A.M.B.; Guimarães, A.L.S.; Santos, S.H.S. Oral Resveratrol Supplementation Improves Metabolic Syndrome Features in Obese Patients Submitted to a Lifestyle-Changing Program. Life Sci. 2020, 256, 117962. [Google Scholar] [CrossRef]
- Walker, J.M.; Eckardt, P.; Aleman, J.O.; Rosa, J.C.d.; Liang, Y.; Iizumi, T.; Etheve, S.; Blaser, M.J.; Breslow, J.L.; Holt, P.R. The Effects of Trans-Resveratrol on Insulin Resistance, Inflammation, and Microbiota in Men with the Metabolic Syndrome: A Pilot Randomized, Placebo-Controlled Clinical Trial. J. Clin. Transl. Res. 2019, 4, 122. [Google Scholar] [PubMed]
- Mendes, K.L.; Lelis, D.d.F.; Souza, L.A.A.; Ronize Viviane Jorge Brito, M.C.A.; Nobre, S.A.M.; Guimarães, A.L.S.; Paula, A.M.B.d.; Lima, J.P.d.; Hilzendeger, A.M.; Santos, S.H.S. Lactococcus Lactis and Resveratrol Decrease Body Weight and Increase Benefic Gastrointestinal Microbiota in Mice. Protein Pept. Lett. 2020, 28, 761–768. [Google Scholar] [CrossRef] [PubMed]
- Bird, J.K.; Raederstorff, D.; Weber, P.; Steinert, R.E. Cardiovascular and Antiobesity Effects of Resveratrol Mediated through the Gut Microbiota. Adv. Nutr. 2017, 8, 839. [Google Scholar] [CrossRef] [PubMed]
- Pang, J.; Raka, F.; Heirali, A.A.; Shao, W.; Liu, D.; Gu, J.; Feng, J.N.; Mineo, C.; Shaul, P.W.; Qian, X.; et al. Resveratrol Intervention Attenuates Chylomicron Secretion via Repressing Intestinal FXR-Induced Expression of Scavenger Receptor SR-B1. Nat. Commun. 2023, 14, 2656. [Google Scholar] [CrossRef] [PubMed]
- Rethlefsen, M.L.; Kirtley, S.; Waffenschmidt, S.; Ayala, A.P.; Moher, D.; Page, M.J.; Koffel, J.B. PRISMA-S: An Extension to the PRISMA Statement for Reporting Literature Searches in Systematic Reviews. Syst. Rev. 2021, 10, 39. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Javid, A.Z.; Hormoznejad, R.; Yousefimanes, H.A.; Zakerkish, M.; Haghighi, M.H.; Ravanbakhsh, M. The Impact of Resveratrol Supplementation on Blood Glucose, Insulin, Insulin Resistance, Triglyceride and Periodontal Markers in Type 2 Diabetic Patients with Chronic Periodontitis. J. Am. Coll. Cardiol. 2016, 68, C183. [Google Scholar] [CrossRef]
- Movahed, A.; Nabipour, I.; Lieben Louis, X.; Thandapilly, S.J.; Yu, L.; Kalantarhormozi, M.; Rekabpour, S.J.; Netticadan, T. Antihyperglycemic Effects of Short Term Resveratrol Supplementation in Type 2 Diabetic Patients. J. Evid. Based Complement. Altern. Med. 2013, 2013, 851267. [Google Scholar] [CrossRef] [PubMed]
- Korsholm, A.S.; Kjær, T.N.; Ornstrup, M.J.; Pedersen, S.B. Comprehensive Metabolomic Analysis in Blood, Urine, Fat, and Muscle in Men with Metabolic Syndrome: A Randomized, Placebo-Controlled Clinical Trial on the Effects of Resveratrol after Four Months’ Treatment. Int. J. Mol. Sci. 2017, 18, 554. [Google Scholar] [CrossRef] [PubMed]
- Hoseini, A.; Namazi, G.; Farrokhian, A.; Reiner, Ž.; Aghadavod, E.; Bahmani, F.; Asemi, Z. The Effects of Resveratrol on Metabolic Status in Patients with Type 2 Diabetes Mellitus and Coronary Heart Disease. Food Funct. 2019, 10, 6042–6051. [Google Scholar] [CrossRef]
- Marques, B.C.A.A.; Trindade, M.; Aquino, J.C.F.; Cunha, A.R.; Gismondi, R.O.; Neves, M.F.; Oigman, W. Beneficial Effects of Acute Trans-Resveratrol Supplementation in Treated Hypertensive Patients with Endothelial Dysfunction. Clin. Exp. Hypertens. 2018, 40, 218–223. [Google Scholar] [CrossRef]
- Banaszewska, B.; Wrotyńska-Barczyńska, J.; Spaczynski, R.Z.; Pawelczyk, L.; Duleba, A.J. Effects of Resveratrol on Polycystic Ovary Syndrome: A Double-Blind, Randomized, Placebo-Controlled Trial. J. Clin. Endocrinol. Metab. 2016, 101, 4322–4328. [Google Scholar] [CrossRef] [PubMed]
- Gonçalinho, G.H.F.; Roggerio, A.; Goes, M.F.d.S.; Avakian, S.D.; Leal, D.P.; Strunz, C.M.C.; Mansur, A.d.P. Comparison of Resveratrol Supplementation and Energy Restriction Effects on Sympathetic Nervous System Activity and Vascular Reactivity: A Randomized Clinical Trial. Molecules 2021, 26, 3168. [Google Scholar] [CrossRef] [PubMed]
- Khodabandehloo, H.; Seyyedebrahimi, S.S.; Esfahani, E.N.; Razi, F.; Meshkani, R. Resveratrol Supplementation Decreases Blood Glucose without Changing the Circulating CD14 + CD16 + Monocytes and Inflammatory Cytokines in Patients with Type 2 Diabetes: A Randomized, Double-Blind, Placebo-Controlled Study. Nutr. Res. 2018, 54, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Zaw, J.J.T.; Howe, P.R.C.; Wong, R.H.X. Sustained Cerebrovascular and Cognitive Benefits of Resveratrol in Postmenopausal Women. Nutrients 2020, 12, 828. [Google Scholar] [CrossRef]
- Andrade, J.M.O.; Barcala-Jorge, A.S.; Batista-Jorge, G.C.; Paraíso, A.F.; Freitas, K.M.d.; Lelis, D.d.F.; Guimarães, A.L.S.; de Paula, A.M.B.; Santos, S.H.S. Effect of Resveratrol on Expression of Genes Involved Thermogenesis in Mice and Humans. Biomed. Pharmacother. 2019, 112, 108634. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, J.; Conte, C.; Fontana, L.; Mittendorfer, B.; Imai, S.; Schechtman, K.B.; Gu, C.; Kunz, I.; Fanelli, F.R.; Patterson, B.W.; et al. Resveratrol Supplementation Does Not Improve Metabolic Function in Nonobese Women with Normal Glucose Tolerance. Cell Metab. 2012, 16, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Magyar, K.; Halmosi, R.; Palfi, A.; Feher, G.; Czopf, L.; Fulop, A.; Battyany, I.; Sumegi, B.; Toth, K.; Szabados, E. Cardioprotection by Resveratrol: A Human Clinical Trial in Patients with Stable Coronary Artery Disease. Clin. Hemorheol. Microcirc. 2012, 50, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Zortea, K.; Franco, V.C.; Francesconi, L.P.; Cereser, K.M.M.; Lobato, M.I.R.; Belmonte-De-Abreu, P.S. Resveratrol Supplementation in Schizophrenia Patients: A Randomized Clinical Trial Evaluating Serum Glucose and Cardiovascular Risk Factors. Nutrients 2016, 8, 73. [Google Scholar] [CrossRef] [PubMed]
- Goh, K.P.; Lee, H.Y.; Lau, D.P.; Supaat, W.; Chan, Y.H.; Koh, A.F.Y. Effects of Resveratrol in Patients with Type 2 Diabetes Mellitus on Skeletal Muscle SIRT1 Expression and Energy Expenditure. Int. J. Sport. Nutr. Exerc. Metab. 2014, 24, 2–13. [Google Scholar] [CrossRef] [PubMed]
- Kantartzis, K.; Fritsche, L.; Bombrich, M.; Machann, J.; Schick, F.; Staiger, H.; Kunz, I.; Schoop, R.; Lehn-Stefan, A.; Heni, M.; et al. Effects of Resveratrol Supplementation on Liver Fat Content in Overweight and Insulin-Resistant Subjects: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Diabetes Obes. Metab. 2018, 20, 1793–1797. [Google Scholar] [CrossRef] [PubMed]
- Gliemann, L.; Schmidt, J.F.; Olesen, J.; Biensø, R.S.; Peronard, S.L.; Grandjean, S.U.; Mortensen, S.P.; Nyberg, M.; Bangsbo, J.; Pilegaard, H.; et al. Resveratrol Blunts the Positive Effects of Exercise Training on Cardiovascular Health in Aged Men. J. Physiol. 2013, 591, 5047–5059. [Google Scholar] [CrossRef]
- Simental-Mendía, L.E.; Guerrero-Romero, F. Effect of Resveratrol Supplementation on Lipid Profile in Subjects with Dyslipidemia: A Randomized Double-Blind, Placebo-Controlled Trial. Nutrition 2019, 58, 7–10. [Google Scholar] [CrossRef]
- De Ligt, M.; Bergman, M.; Fuentes, R.M.; Essers, H.; Moonen-Kornips, E.; Havekes, B.; Schrauwen-Hinderling, V.B.; Schrauwen, P. No Effect of Resveratrol Supplementation after 6 Months on Insulin Sensitivity in Overweight Adults: A Randomized Trial. Am. J. Clin. Nutr. 2020, 112, 1029–1038. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.H.X.; Berry, N.M.; Coates, A.M.; Buckley, J.D.; Bryan, J.; Kunz, I.; Howe, P.R.C. Chronic Resveratrol Consumption Improves Brachial Flow-Mediated Dilatation in Healthy Obese Adults. J. Hypertens. 2013, 31, 1819–1827. [Google Scholar] [CrossRef]
- Heebøll, S.; Kreuzfeldt, M.; Hamilton-Dutoit, S.; Kjær Poulsen, M.; Stødkilde-Jørgensen, H.; Møller, H.J.; Jessen, N.; Thorsen, K.; Kristina Hellberg, Y.; Bønløkke Pedersen, S.; et al. Placebo-Controlled, Randomised Clinical Trial: High-Dose Resveratrol Treatment for Non-Alcoholic Fatty Liver Disease. Scand. J. Gastroenterol. 2016, 51, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Huhn, S.; Beyer, F.; Zhang, R.; Lampe, L.; Grothe, J.; Kratzsch, J.; Willenberg, A.; Breitfeld, J.; Kovacs, P.; Stumvoll, M.; et al. Effects of Resveratrol on Memory Performance, Hippocampus Connectivity and Microstructure in Older Adults—A Randomized Controlled Trial. Neuroimage 2018, 174, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Seyyedebrahimi, S.S.; Khodabandehloo, H.; Nasli Esfahani, E.; Meshkani, R. The Effects of Resveratrol on Markers of Oxidative Stress in Patients with Type 2 Diabetes: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Acta Diabetol. 2018, 55, 341–353. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhao, X.; Ran, L.; Wan, J.; Wang, X.; Qin, Y.; Shu, F.; Gao, Y.; Yuan, L.; Zhang, Q.; et al. Resveratrol Improves Insulin Resistance, Glucose and Lipid Metabolism in Patients with Non-Alcoholic Fatty Liver Disease: A Randomized Controlled Trial. Dig. Liver Dis. 2015, 47, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Abdollahi, S.; Salehi-Abargouei, A.; Toupchian, O.; Sheikhha, M.H.; Fallahzadeh, H.; Rahmanian, M.; Tabatabaie, M.; Mozaffari-Khosravi, H. The Effect of Resveratrol Supplementation on Cardio-Metabolic Risk Factors in Patients with Type 2 Diabetes: A Randomized, Double-Blind Controlled Trial. Phytother. Res. 2019, 33, 3153–3162. [Google Scholar] [CrossRef] [PubMed]
- Timmers, S.; De Ligt, M.; Phielix, E.; Van De Weijer, T.; Hansen, J.; Moonen-Kornips, E.; Schaart, G.; Kunz, I.; Hesselink, M.K.C.; Schrauwen-Hinderling, V.B.; et al. Resveratrol as Add-on Therapy in Subjects with Well-Controlled Type 2 Diabetes: A Randomized Controlled Trial. Diabetes Care 2016, 39, 2211–2217. [Google Scholar] [CrossRef] [PubMed]
- Asghari, S.; Asghari-Jafarabadi, M.; Somi, M.H.; Ghavami, S.M.; Rafraf, M. Comparison of Calorie-Restricted Diet and Resveratrol Supplementation on Anthropometric Indices, Metabolic Parameters, and Serum Sirtuin-1 Levels in Patients With Nonalcoholic Fatty Liver Disease: A Randomized Controlled Clinical Trial. J. Am. Coll. Nutr. 2018, 37, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Thazhath, S.S.; Wu, T.; Bound, M.J.; Checklin, H.L.; Standfield, S.; Jones, K.L.; Horowitz, M.; Rayner, C.K. Administration of Resveratrol for 5 Wk Has No Effect on Glucagon-like Peptide 1 Secretion, Gastric Emptying, or Glycemic Control in Type 2 Diabetes: A Randomized Controlled Trial. Am. J. Clin. Nutr. 2016, 103, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Anton, S.D.; Embry, C.; Marsiske, M.; Lu, X.; Doss, H.; Leeuwenburgh, C.; Manini, T.M. Safety and Metabolic Outcomes of Resveratrol Supplementation in Older Adults: Results of a Twelve-Week, Placebo-Controlled Pilot Study. Exp. Gerontol. 2014, 57, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Kjær, T.N.; Ornstrup, M.J.; Poulsen, M.M.; Stødkilde-Jørgensen, H.; Jessen, N.; Jørgensen, J.O.L.; Richelsen, B.; Pedersen, S.B. No Beneficial Effects of Resveratrol on the Metabolic Syndrome: A Randomized Placebo-Controlled Clinical Trial. J. Clin. Endocrinol. Metab. 2017, 102, 1642–1651. [Google Scholar] [CrossRef] [PubMed]
- Chachay, V.S.; Macdonald, G.A.; Martin, J.H.; Whitehead, J.P.; O’Moore-Sullivan, T.M.; Lee, P.; Franklin, M.; Klein, K.; Taylor, P.J.; Ferguson, M.; et al. Resveratrol Does Not Benefit Patients With Nonalcoholic Fatty Liver Disease. Clin. Gastroenterol. Hepatol. 2014, 12, 2092–2103.e6. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zeng, Y.; Pan, Z.; Jin, Y.; Li, Q.; Pang, J.; Wang, X.; Chen, Y.; Yang, Y.; Ling, W. A Randomized Trial on Resveratrol Supplement Affecting Lipid Profile and Other Metabolic Markers in Subjects with Dyslipidemia. Nutrients 2023, 15, 492. [Google Scholar] [CrossRef]
- Sariaslani, P.; Asgharzadeh, S.; Mohammadi, H.; Ghanbari, A.; Hezarkhani, L.; Shahbazi, F.; Mirzaeei, S. Does Resveratrol Enhance Recovery from Acute Ischemic Stroke? A Randomized, Double-Blinded, Placebo-Controlled Trial. J. Rep. Pharm. Sci. 2022, 11, 192. [Google Scholar] [CrossRef]
- Jojima, K.; Tanaka, A.; Node, K. Resveratrol Supplementation: A Therapeutic Potential for Cardiac Remodeling in Hypertensive Heart Disease. Hypertens. Res. 2023, 46, 1596–1598. [Google Scholar] [CrossRef] [PubMed]
- Rush, J.W.E.; Quadrilatero, J.; Levy, A.S.; Ford, R.J. Chronic Resveratrol Enhances Endothelium-Dependent Relaxation but Does Not Alter ENOS Levels in Aorta of Spontaneously Hypertensive Rats. Exp. Biol. Med. 2017, 232, 814–822. [Google Scholar] [CrossRef]
- Mizutani, K.; Ikeda, K.; Kawai, Y.; Yamori, Y. Protective Effect of Resveratrol on Oxidative Damage in Male and Female Stroke-Prone Spontaneously Hypertensive Rats. Clin. Exp. Pharmacol. Physiol. 2001, 28, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Timmers, S.; Konings, E.; Bilet, L.; Houtkooper, R.H.; van de Weijer, T.; Goossens, G.H.; Hoeks, J.; van der Krieken, S.; Ryu, D.; Kersten, S.; et al. Calorie Restriction-like Effects of 30 Days of Resveratrol Supplementation on Energy Metabolism and Metabolic Profile in Obese Humans. Cell Metab. 2011, 14, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Miatello, R.; Vázquez, M.; Renna, N.; Cruzado, M.; Zumino, A.P.; Risler, N. Chronic Administration of Resveratrol Prevents Biochemical Cardiovascular Changes in Fructose-Fed Rats. Am. J. Hypertens. 2005, 18, 864–870. [Google Scholar] [CrossRef]
- Liu, Z.; Song, Y.; Zhang, X.; Liu, Z.; Zhang, W.; Mao, W.; Wang, W.; Cui, W.; Zhang, X.; Jia, X.; et al. Effects of Trans-Resveratrol on Hypertension-Induced Cardiac Hypertrophy Using the Partially Nephrectomized Rat Model. Clin. Exp. Pharmacol. Physiol. 2005, 32, 1049–1054. [Google Scholar] [CrossRef] [PubMed]
- Gu, W.; Geng, J.; Zhao, H.; Li, X.; Song, G. Effects of Resveratrol on Metabolic Indicators in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis. Int. J. Clin. Pract. 2022, 2022, 9734738. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Wu, C.; Qiu, S.; Yuan, X.; Li, L. Effects of Resveratrol on Glucose Control and Insulin Sensitivity in Subjects with Type 2 Diabetes: Systematic Review and Meta-Analysis. Nutr. Metab. 2017, 14, 60. [Google Scholar] [CrossRef] [PubMed]
- Hausenblas, H.A.; Schoulda, J.A.; Smoliga, J.M. Resveratrol Treatment as an Adjunct to Pharmacological Management in Type 2 Diabetes Mellitus--Systematic Review and Meta-Analysis. Mol. Nutr. Food Res. 2015, 59, 147–159. [Google Scholar] [CrossRef]
- Faghihzadeh, F.; Hekmatdoost, A.; Adibi, P. Resveratrol and Liver: A Systematic Review. J. Res. Med. Sci. 2015, 20, 797. [Google Scholar] [CrossRef] [PubMed]
- Shamardl, H.A.M.A.; Ibrahim, N.A.; Merzeban, D.H.; Elamir, A.M.; Golam, R.M.; Elsayed, A.M. Resveratrol and Dulaglutide Ameliorate Adiposity and Liver Dysfunction in Rats with Diet-Induced Metabolic Syndrome: Role of SIRT-1/Adipokines/PPARγ and IGF-1. Daru 2023, 31, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Reda, D.; Elshopakey, G.E.; Mahgoub, H.A.; Risha, E.F.; Khan, A.A.; Rajab, B.S.; El-Boshy, M.E.; Abdelhamid, F.M. Effects of Resveratrol Against Induced Metabolic Syndrome in Rats: Role of Oxidative Stress, Inflammation, and Insulin Resistance. Evid.-Based Complement. Altern. Med. 2022, 2022, 3362005. [Google Scholar] [CrossRef] [PubMed]
- Oh, E.; Cho, N.-J.; Kang, H.; Kim, S.H.; Park, H.K.; Kwon, S.H. Computed Tomography Evaluation of Skeletal Muscle Quality and Quantity in People with Morbid Obesity with and without Metabolic Abnormality. PLoS ONE 2023, 18, e0296073. [Google Scholar] [CrossRef] [PubMed]
- Wharton, S.; Lau, D.C.W.; Vallis, M.; Sharma, A.M.; Biertho, L.; Campbell-Scherer, D.; Adamo, K.; Alberga, A.; Bell, R.; Boulé, N.; et al. Obesity in Adults: A Clinical Practice Guideline. CMAJ 2020, 192, E875–E891. [Google Scholar] [CrossRef] [PubMed]
- Wiklund, P.; Toss, F.; Weinehall, L.; Hallmans, G.; Franks, P.W.; Nordstrom, A.; Nordstrom, P. Abdominal and Gynoid Fat Mass Are Associated with Cardiovascular Risk Factors in Men and Women. J. Clin. Endocrinol. Metab. 2008, 93, 4360–4366. [Google Scholar] [CrossRef] [PubMed]
- Seyedhoseinpour, A.; Barzin, M.; Mahdavi, M.; Valizadeh, M.; Azizi, F.; Ghareh, S.; Hosseinpanah, F. BMI Category-Specific Waist Circumference Thresholds Based on Cardiovascular Disease Outcomes and All-Cause Mortality: Tehran Lipid and Glucose Study (TLGS). BMC Public Health 2023, 23, 1297. [Google Scholar] [CrossRef]
- Silva de Lima Loureiro, N.I.; Lameira Maciel Amaral, T.I.; de Araújo Amaral, C., III; Torres Rego Monteiro, G.I.; Teixeira Leite de Vasconcellos, M.V.; Junior Sordi Bortolini, M.V.; Lameira Maciel Amaral, T. Relationship between Anthropometric Indicators and Risk Factors for Cardiovascular Disease in Adults and Older Adults of Rio Branco, Acre. Rev. Saude Publica 2020, 54, 24. [Google Scholar] [CrossRef]
- Bell, M.; Duncan, M.J.; Patte, K.A.; Roy, B.D.; Ditor, D.S.; Klentrou, P. Changes in Body Mass, Physical Activity, and Dietary Intake during the COVID-19 Pandemic Lockdowns in Canadian University Students. Biology 2023, 12, 326. [Google Scholar] [CrossRef]
- Alexander, L.; Christensen, S.M.; Richardson, L.; Ingersoll, A.B.; Burridge, K.; Golden, A.; Karjoo, S.; Cortez, D.; Shelver, M.; Bays, H.E. Nutrition and Physical Activity: An Obesity Medicine Association (OMA) Clinical Practice Statement 2022. Obes. Pillars 2022, 1, 100005. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, M.M.; Fjeldborg, K.; Ornstrup, M.J.; Kjaer, T.; Nohr, M.K.; Pedersen, S.B. Resveratrol and Inflammation: Challenges in Translating Pre-Clinical Findings to Improved Patient Outcomes. Biochim. Biophys. Acta 2015, 1852, 1124–1136. [Google Scholar] [CrossRef] [PubMed]
- Sahebkar, A. Effects of Resveratrol Supplementation on Plasma Lipids: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutr. Rev. 2013, 71, 822–835. [Google Scholar] [CrossRef] [PubMed]
- Catapano, A.L.; Graham, I.; De Backer, G.; Wiklund, O.; Chapman, M.J.; Drexel, H.; Hoes, A.W.; Jennings, C.S.; Landmesser, U.; ESC Scientific Document Group; et al. 2016 ESC/EAS guidelines for the management of dyslipidaemias. Eur. Heart J. 2016, 2999–3058. [Google Scholar] [CrossRef] [PubMed]
- Tomé-Carneiro, J.; Gonzálvez, M.; Larrosa, M.; García-Almagro, F.J.; Avilés-Plaza, F.; Parra, S.; Yáñez-Gascón, M.J.; Ruiz-Ros, J.A.; García-Conesa, M.T.; Tomás-Barberán, F.A.; et al. Consumption of a Grape Extract Supplement Containing Resveratrol Decreases Oxidized LDL and ApoB in Patients Undergoing Primary Prevention of Cardiovascular Disease: A Triple-Blind, 6-Month Follow-up, Placebo-Controlled, Randomized Trial. Mol. Nutr. Food Res. 2012, 56, 810–821. [Google Scholar] [CrossRef] [PubMed]
- Haghighatdoost, F.; Hariri, M. Effect of Resveratrol on Lipid Profile: An Updated Systematic Review and Meta-Analysis on Randomized Clinical Trials. Pharmacol. Res. 2018, 129, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Zamora-Ros, R.; Urpi-Sarda, M.; Lamuela-Raventós, R.M.; Martínez-González, M.Á.; Salas-Salvadó, J.; Arós, F.; Fitó, M.; Lapetra, J.; Estruch, R.; Andres-Lacueva, C.; et al. High Urinary Levels of Resveratrol Metabolites Are Associated with a Reduction in the Prevalence of Cardiovascular Risk Factors in High-Risk Patients. Pharmacol. Res. 2012, 65, 615–620. [Google Scholar] [CrossRef] [PubMed]
- Mendes, K.L.; Pinho, L.d.; João Marcus Oliveira Andrade, A.F.P.; Lula, J.F.; Macedo, S.M.; Feltenberger, J.D.; Guimarães, A.L.S.; Paula, A.M.B.d.; Santos, S.H.S. Distinct Metabolic Effects of Resveratrol on Lipogenesis Markers in Mice Adipose Tissue Treated with High-Polyunsaturated Fat and High-Protein Diets. Life Sci. 2016, 153, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Andrade, J.M.O.; Paraíso, A.F.; Oliveira, M.V.M.d.; Martins, A.M.E.; Neto, J.F.; Guimarães, A.L.S.; Paula, A.M.d.; Qureshi, M.; Santos, S.H.S. Resveratrol Attenuates Hepatic Steatosis in High-Fat Fed Mice by Decreasing Lipogenesis and Inflammation. Nutrition 2014, 30, 915–919. [Google Scholar] [CrossRef] [PubMed]
- Vitaglione, P.; Sforza, S.; Galaverna, G.; Ghidini, C.; Caporaso, N.; Vescovi, P.P.; Fogliano, V.; Marchelli, R. Bioavailability of Trans-Resveratrol from Red Wine in Humans. Mol. Nutr. Food Res. 2005, 49, 495–504. [Google Scholar] [CrossRef]

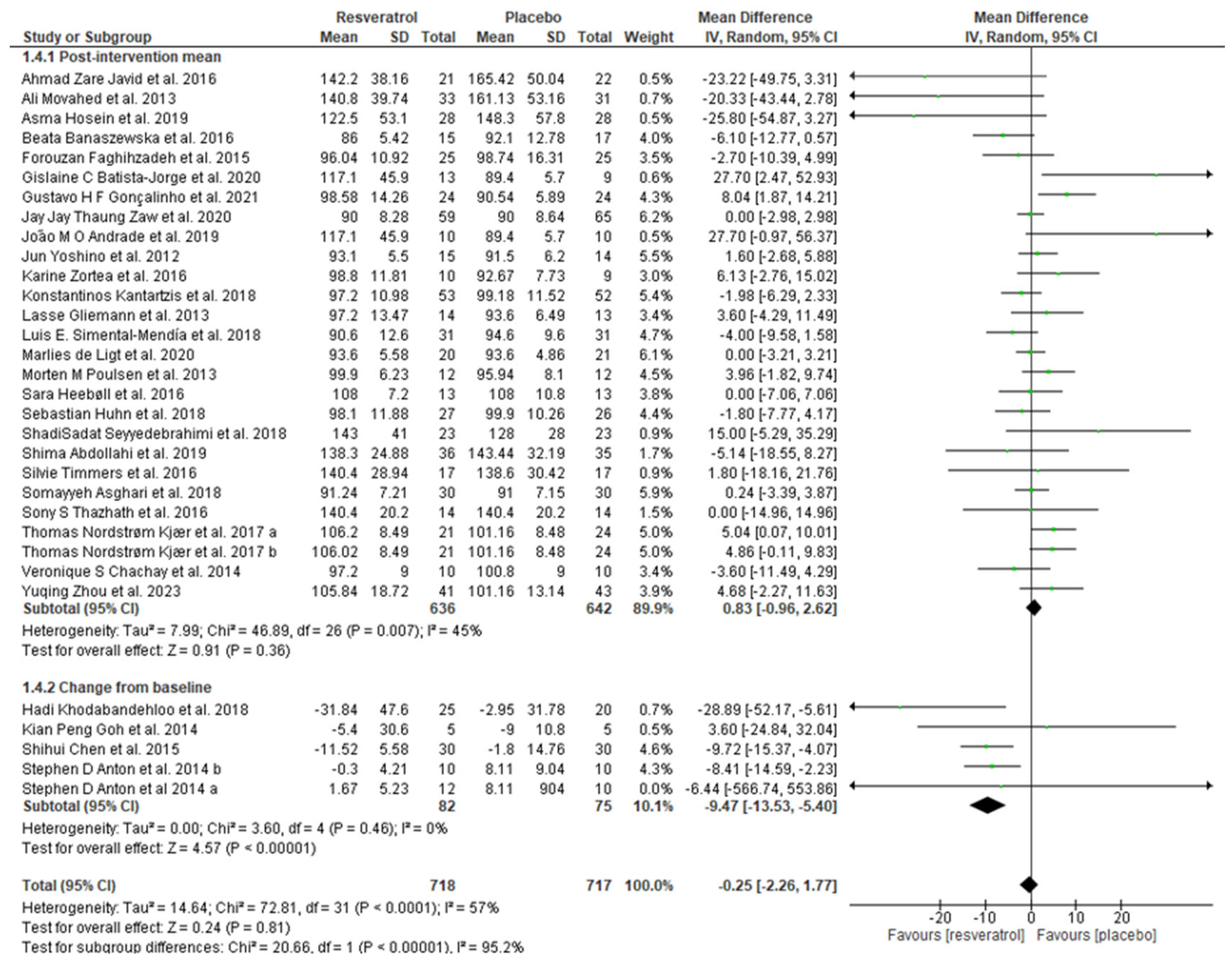
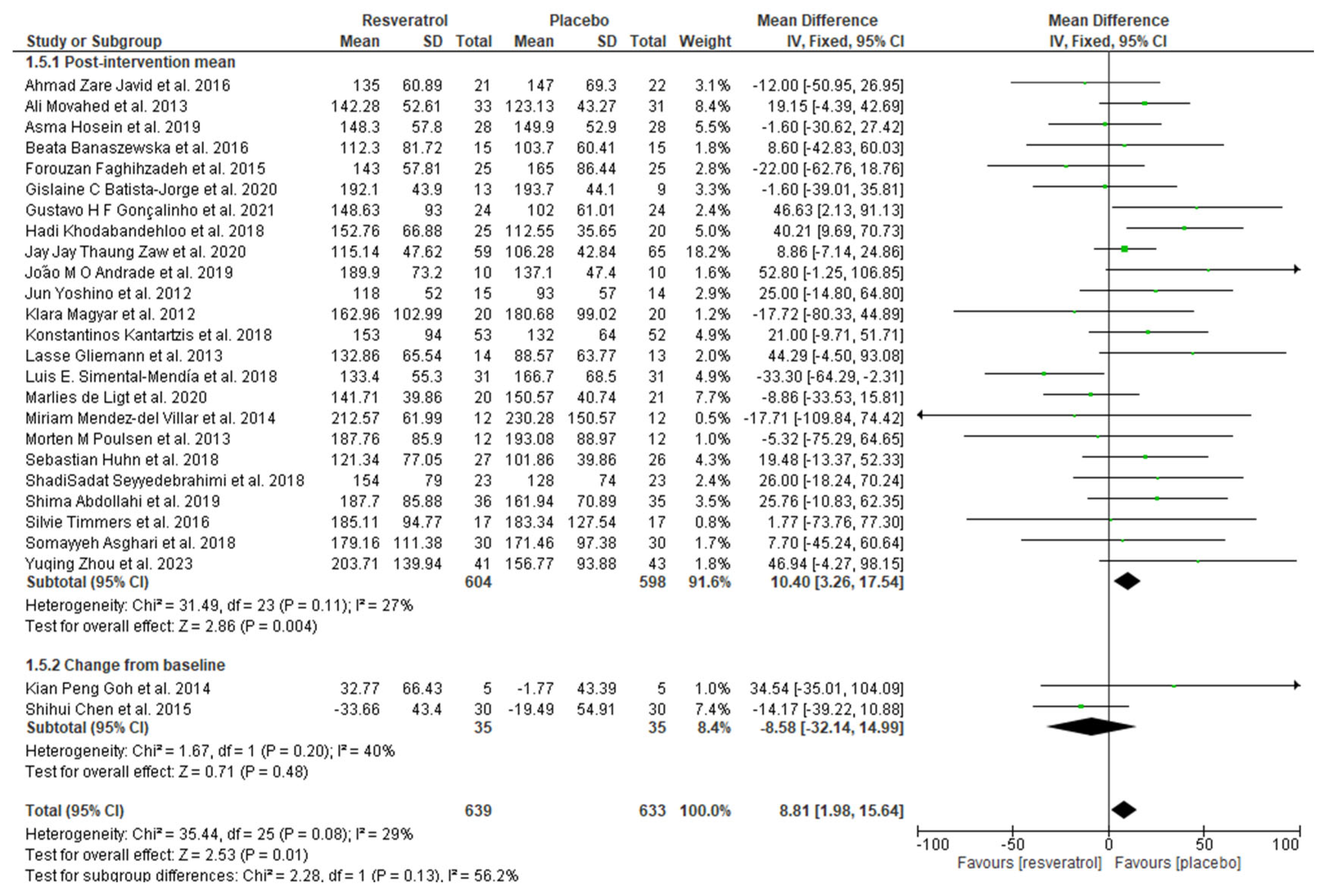
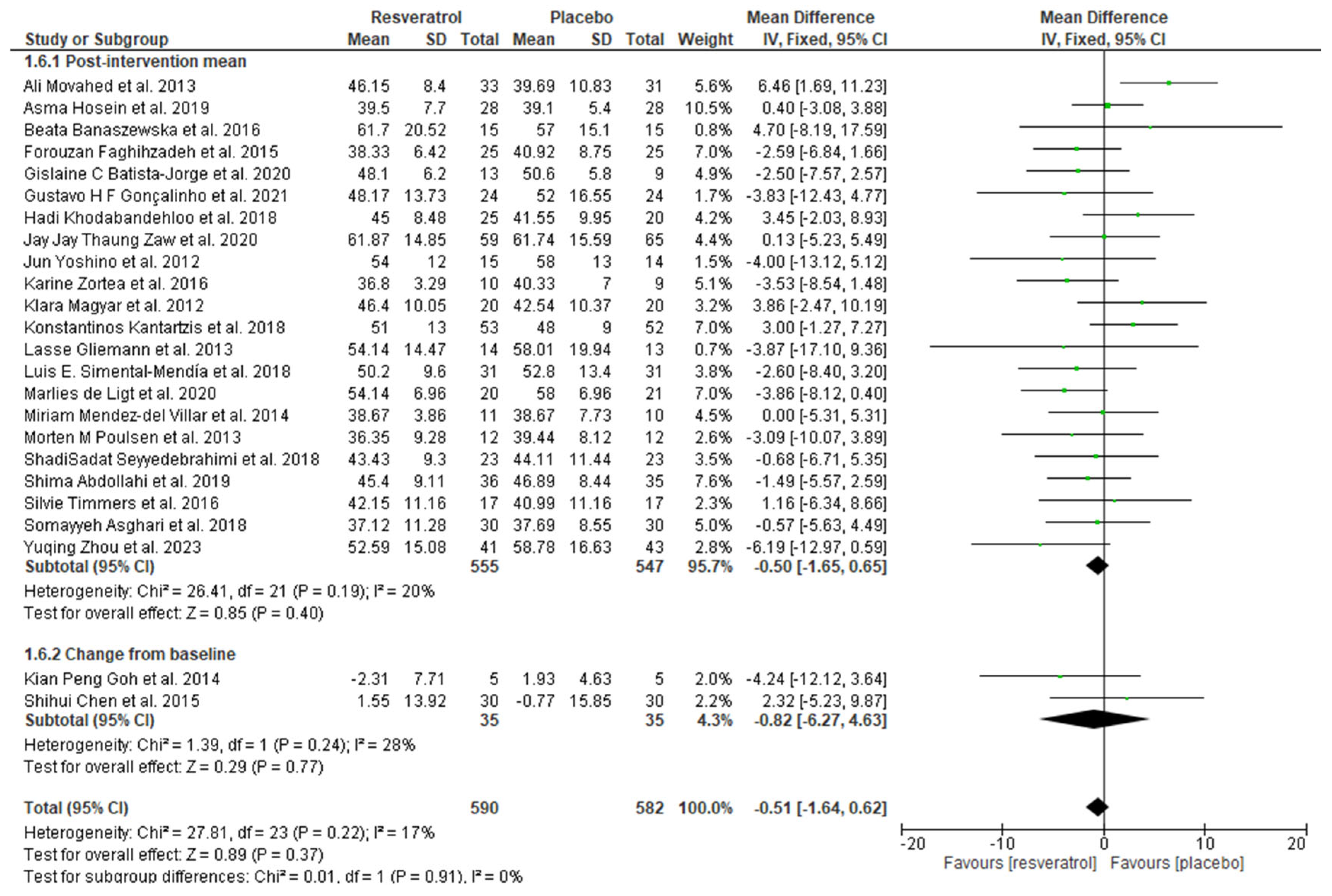
| Author/Year | Sample Size (Placebo/ Intervention) | Population/ Country | Resveratrol (Daily Dosage) | Intervention Duration | Data Retrieved |
|---|---|---|---|---|---|
| Javid et al., 2016 [32] | 22/21 | Adult men and women with diabetes/Iran | 480 mg | 28 days | Glucose/TG |
| Movahed et al., 2013 [33] | 31/33 | Adult men and women with diabetes/Iran | 1000 mg | 45 days | Glucose/TGHDL/SBP/ DBP |
| Korsholm et al., 2017 [34] | 24/21 | Adult men with metabolic syndrome (MetS)/Denmark | 1000 mg | 120 days | SBP/DBP |
| Hoseini et al., 2019 [35] | 28/28 | Adults with type 2 diabetes mellitus and coronary heart disease/Iran | 500 mg | 28 days | Glucose/TG/HDL |
| Marques et al., 2018 [36] | 12/12 | Adult men with hypertension/Brazil | 300 mg | 1 day | SBP/DBP |
| Banaszewska et al., 2016 [37] | 15/15 | Women with polycystic ovary syndrome/United States | 1500 mg | 90 days | Glucose/TG/HDL |
| Faghihzadeh et al., 2015 [19] | 25/25 | Adults with non-alcoholic fatty liver disease/Iran | 500 mg | 84 days | Glucose/TG/HDL SBP/DBP |
| Faghihzade et al., 2014 [17] | 25/25 | Adults with non-alcoholic fatty liver disease/Iran | 500 mg | 84 days | WC |
| Batista-Jorge et al., 2020 [25] | 9/13 | Adults with obesity/Brazil | 250 mg | 90 days | WC/Glucose/TG/HDL |
| Gonçalinho et al., 2021 [38] | 24/24 | Healthy men and women/Brazil | 500 mg | 30 days | Glucose/TG/HDL SBP/DBP |
| Khodabandehloo et al., 2018 [39] | 20/25 | Adults with diabetes/Iran | 800 mg | 56 days | Glucose/ WC/TG/ HDL /SBP/DBP |
| Zaw et al., 2020 [40] | 65/59 | Post-menopausal women in Australian/New Zealand | 150 mg | 360 days | Glucose/TG/HDL |
| Andrade et al., 2019 [41] | 10/10 | Adults with obesity/Brazil | 500 mg | 56 days | Glucose/TG |
| Yoshino et al., 2012 [42] | 14/15 | Adult lean and overweight women/United States | 75 mg | 84 days | Glucose/TG/HDL SBP/DBP |
| Magyar et al., 2012 [43] | 20/20 | Adults after myocardial infarction/Hungary | 10 mg | 90 days | TG/HDL |
| Zortea et al., 2016 [44] | 9/10 | Adults with schizophrenia/Brazil | 200 mg | 30 days | Glucose/WC/HDL |
| Goh et al., 2014 [45] | 5/5 | Adults with diabetes/Singapore | 500 mg | 84 days | Glucose/TG/HDL |
| Kantartzis et al. 2018 [46] | 52/53 | Adults with overweight and insulin resistance/Germany | 150 mg | 84 days | Glucose/TG/HDL SBP/DBP |
| Gliemann et al., 2013 [47] | 13/14 | Healthy older men/Denmark | 250 mg | 56 days | Glucose/TG/HDL |
| Simental-Mendía et al., 2018 [48] | 31/31 | Adults with dyslipidemia/Mexico | 100 mg | 60 days | Glucose/WC/TG/ HDL SBP/DBP |
| Ligt et al., 2020 [49] | 21/20 | Overweight men and women/The Netherlands | 150 mg | 180 days | Glucose/TG/HDL |
| Villar et al., 2014 [23] | 10/11 | Adults with metabolic syndrome/ Mexico | 1500 mg | 90 days | WC/TG/ HDL SBP/DBP |
| Poulsen et al., 2013 [9] | 12/12 | Adult obese men/Denmark | 1500 mg | 28 days | Glucose/TG/HDL |
| Wong et al., 2013 [50] | 15/13 | Healthy obese adults/Australia | 75 mg | 42 days | SBP/DBP |
| Heebøll et al. 2016 [51] | 13/13 | Patients with non-alcoholic fatty liver disease/Denmark | 1500 mg | 42 days | Glucose/ SBP/DBP |
| Huhn et al., 2018 [52] | 26/27 | Older adults/Amsterdam | 200 mg | 182 days | Glucose/TG/SBP/DBP |
| Seyyedebrahimi et al., 2018 [53] | 23/23 | Adults with diabetes/Iran | 800 mg | 60 days | Glucose WC/TG/ HDL /SBP/DBP |
| Chen et al., 2015 [54] | 30/30 | Adults with non-alcoholic fatty liver disease/China | 600 mg | 90 days | Glucose WC/TG/ HDL /SBP/DBP |
| Abdollahi et al., 2019 [55] | 35/36 | Patients with type 2 diabetes/Iran | 1000 mg | 56 days | Glucose WC/TG/ HDL |
| Timmers et al., 2016 [56] | 17/17 | Adults with diabetes/Netherlands | 150 mg | 30 days | Glucose TG/HDL/ SBP/DBP |
| Asghari et al., 2018 [57] | 30/30 | Adults with non-alcoholic fatty liver disease/United States | 600 mg | 84 days | Glucose WC/TG /HDL |
| Thazhath et al., 2016 [58] | 14/14 | Adults with diabetes/Australia | 1000 mg | 35 days | Glucose |
| Anton et al., 2014 [59] | 10/12 | Healthy overweight older adults /United States | 300 mg 1000 mg | 90 days | Glucose WC/ SBP/DBP |
| Kjær et al., 2017 [60] | 24/20 | Adults with metabolic syndrome/Denmark | 150 mg 1000 mg | 42 days | Glucose SBP/DBP |
| Chachay et al., 2014 [61] | 10/10 | Adults with non-alcoholic fatty liver disease/Australia | 3000 mg | 56 days | Glucose SBP/DBP |
| Zhou et al., 2023 [62] | 43/41 | Adults with dyslipidemia | 600 mg | 56 days | Glucose/TG/HDL |
| Sariaslane et al., 2022 [63] | 34/40 | Patients with ischemic stroke | 510 mg | 30 days | SBP/DBP |
| Summary of Findings | Quality of Evidence Assessment (Grade) | |||||
|---|---|---|---|---|---|---|
| Outcome | Time | No Patients | Limitation | Inconsistency | Imprecision | Quality |
| IMMS-IE | Short term | 363 [32,33,35,36,41,47,50,53,56,58,61] | Yes | Yes | Yes | Low |
| IMMS-IE | Short term | 19 [44] | Yes | Yes | Yes | Very low |
| IMMS-IE | Long term | 105 [34,54] | Yes | Yes | No | Moderate |
| IMMS-IE | Long term | 506 [19,37,40,43,45,46,51,52,59] | Yes | Yes | Yes | Low |
| IMMS-IE | Long term | 74 [60] | Yes | No | Yes | High |
| IMMS-DE | Short term | 93 [38,39,63] | Yes | Yes | Yes | Low |
| IMMS-DE | Short term | 71 [48] | Yes | Yes | Yes | Very low |
| IMMS-DE | Short term | 24 [9] | Yes | No | Yes | High |
| IMMS-DE | Long term | 25 [25] | Yes | Yes | Yes | Low |
| IMMS-DE | Long term | 114 [23,57] | Yes | No | Yes | Moderate |
| IMMS-DE | Long term | 45 [42] | Yes | Yes | No | Moderate |
| IMMS-DE | Long term | 50 [17] | Yes | Yes | Yes | Moderate |
| IMMS-DE | Long term | 71 [55,62] | Yes | No | Yes | High |
| IMMS-DE | Long term | 41 [49] | Yes | No | No | Very high |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Batista-Jorge, G.C.; Barcala-Jorge, A.S.; Lelis, D.F.; Santos, D.E.; Jorge, A.H.; Monteiro-Junior, R.S.; Santos, S.H.S. Resveratrol Effects on Metabolic Syndrome Features: A Systematic Review and Meta-Analysis. Endocrines 2024, 5, 225-243. https://doi.org/10.3390/endocrines5020016
Batista-Jorge GC, Barcala-Jorge AS, Lelis DF, Santos DE, Jorge AH, Monteiro-Junior RS, Santos SHS. Resveratrol Effects on Metabolic Syndrome Features: A Systematic Review and Meta-Analysis. Endocrines. 2024; 5(2):225-243. https://doi.org/10.3390/endocrines5020016
Chicago/Turabian StyleBatista-Jorge, Gislaine C., Antônio S. Barcala-Jorge, Deborah F. Lelis, Daniel E. Santos, Antônio H. Jorge, Renato S. Monteiro-Junior, and Sérgio H. S. Santos. 2024. "Resveratrol Effects on Metabolic Syndrome Features: A Systematic Review and Meta-Analysis" Endocrines 5, no. 2: 225-243. https://doi.org/10.3390/endocrines5020016
APA StyleBatista-Jorge, G. C., Barcala-Jorge, A. S., Lelis, D. F., Santos, D. E., Jorge, A. H., Monteiro-Junior, R. S., & Santos, S. H. S. (2024). Resveratrol Effects on Metabolic Syndrome Features: A Systematic Review and Meta-Analysis. Endocrines, 5(2), 225-243. https://doi.org/10.3390/endocrines5020016








