Abstract
Maturity Onset Diabetes of the Young (MODY) encompasses a group of rare monogenic forms of diabetes distinct in etiology and clinical presentation from the more common forms of Type 1 (autoimmune) and Type 2 diabetes. Since its initial description as a clinical entity nearly 50 years ago, the underlying genetic basis for the various forms of MODY has been increasingly better elucidated. Clinically, the diagnosis may be made in childhood or young adulthood and can present as overt hyperglycemia requiring insulin therapy or as a subtle form of slowly progressive glucose impairment. Due to the heterogeneity of clinical symptoms, patients with MODY may be misdiagnosed as possessing another form of diabetes, resulting in potentially inappropriate treatment and delays in screening of affected family members and associated comorbidities. In this review, we highlight the various known genetic mutations associated with MODY, clinical presentation, indications for testing, and the treatment options available.
1. Introduction
In children, particularly those under age 10 years, the predominant form of diabetes mellitus is type 1 diabetes [1], which results from autoimmune destruction of insulin-producing beta cells and results in insulinopenia and a lifelong requirement for insulin therapy. Among children over age 10 years, type 2 diabetes is increasingly more prevalent and may be the predominant form of diabetes mellitus among certain ethnic and racial groups [1]. Compared to adults, children with type 2 diabetes are more likely to present with insulinopenia and symptoms of overt hyperglycemia and often require insulin therapy at the time of their diagnosis [2].
Maturity-Onset Diabetes of the Young “MODY” is a rare, monogenic form of diabetes mellitus [3]. Initially described in a seminal publication by Tattersall and Fajans in 1975, MODY has a reported prevalence ranging from 1 to 5% among all patients diagnosed with diabetes mellitus. Over the last two decades, the number of genetic mutations and different forms of MODY has increased from an initial description of 6 subtypes [4] to as many as 14 currently described subtypes [3]. There is wide phenotypic variability both at clinical presentation and over the course of the affected individual’s life, ranging from mild asymptomatic hyperglycemia and mild Hba1c elevations to overt insulinopenia and hyperglycemia requiring insulin therapy. The age of onset is typically younger than 35 years of age, and there is frequently a strong family history of diabetes mellitus distributed in a pattern suggesting an autosomal dominant inheritance.
Given the rarity of this condition and its wide clinical presentation, it is not uncommon for MODY to be misdiagnosed as a different form of diabetes. Considering the important comorbidities associated with various forms of MODY and the potential for changes in treatment options, it is critically important that MODY is appropriately diagnosed. In this review, we will discuss the genetic basis underling various forms of MODY and the common clinical presentation of the various forms. We will highlight the indications for testing for MODY in the individual with newly diagnosed diabetes mellitus and briefly discuss the various treatment options available.
2. MODY Subtypes and Associated Genetic Mutations
The various subtypes of MODY have been described in various publications by both their underlying genetic mutation as well as by a numerical designation (i.e., MODY 1–MODY 14). With an increasingly more sophisticated understanding of the underlying cause, the various MODY subtypes can be even further subdivided based on defects in transcription, enzyme function, protein folding, ion channel function, or signal transduction [3]. Below, we describe the various subtypes of MODY by both genetic mutation and numerical designation, starting with the more common subtypes (HNF1a, GCK, HNF4a, and HNF1b) and progressing to the rarer subtypes that individually account for less than 1% of all forms of MODY. Table 1 provides a summary of the MODY subtype, gene location, and the expected effect of gene mutation on beta-cell function.

Table 1.
MODY Subtypes, associated genes, chromosome locus, and effect of gene mutation on beta cell function.
2.1. HNF1A MODY (MODY 3)
HNF1A MODY, also known as MODY 3, is the most common form of MODY overall, but the second most common form of MODY observed in children. The reported prevalence of HNF1A MODY among all forms of MODY ranges from as low as 15% in Polish populations to as high as 52% among Scandinavian and British populations [5,6]. The HNF1A gene is located on chromosome 12, region 12q24.2, and encodes for the hepatocyte nuclear factor 1 alpha (HNF1alpha) protein, which is a tissue-specific transcription factor for which its expression has been documented in adult pancreas, kidney, liver, and gut [5]. HNF1alpha regulates intestinal cell growth and cell differentiation during embryonic development, is involved in the transcriptional regulation of bile acid transporters in the intestines and kidneys (important in HDL metabolism) and acute phase proteins such as CRP and fibrinogen, and directly regulates the expression of renal glucose-transported SGLT2 [7].
A higher number of mutations have been observed in HNF1A exon 2 and exon 4, and the lowest number has been observed in exon 5 and exon 10. Mutations in the HNF1A promoter can alter or disrupt the binding site for other transcription factors in the liver and pancreas [8]. The site of promoter mutation reportedly influences its activity to differing degrees, declining as little as 30% and as much as 70% [9,10]. Various studies have cited numerous HNFA1 gene variants [11] including missense, frame shift, nonsense, splicing mutations, in-frame amino acid deletions, insertions, duplications, or partial and whole-gene deletions [12]. It is important to attempt to differentiate variants of unknown significance to truly pathogenic mutations, particularly when screening unaffected relatives of a proband.
Interestingly, mutations in different HNF1A gene isoforms may influence the age of disease onset. Patients with HNF1A mutations in exons 8–10 (affecting only isoform A) have later disease onset than compared to patients with the mutations in exons 1–6 (affecting isoforms A, B, and C) by as much as 12 years. Patients carrying missense mutations affecting the transactivation domain have later onsets than those with truncating mutations or with missense mutations in the dimerization/DBD domain. [11,13,14,15,16]. In one study of 362 MODY family members, the age of onset of HNF1A-MODY in a child was younger when the mother was diagnosed before pregnancy, and if the child was exposed to maternal MODY 3 in utero and inherited the HNF1A mutation from their mother [17]. Conversely, 50% of carriers of HNF1A mutation from their fathers have MODY 3 onset after age of 25 years [18].
Mutated HNF1alpha may alter gene expression and alter pancreatic development [19]. Abnormal structure of Islets of Langerhans and insufficient insulin secretion occur under conditions of insufficient production of HNF1alpha protein [20].
HNF1A MODY penetration in patients is approximately 63% by age 25 years, 93.6% by age 50 years, and 98.7% by age 75 years [21].
2.2. GCK MODY (MODY 2)
GCK MODY, also known as MODY 2, is the most common form of MODY in childhood, but the second most common form of MODY observed in adults. The reported prevalence of the GCK MODY subtype among all forms of MODY is reported to be approximately 22% in all age groups [6], but more selective screening of pediatric patients identified GCK MODY accounting for 88–95% of all pediatric MODY cases [22,23].
The GCK gene is located on chromosome 17, region 7p15–p13, and encodes for glucokinase, a key regulatory enzyme involved in the control of insulin secretion from the pancreatic beta cells [24]. Glucokinase is expressed in the pancreas, liver, brain, and pancreas and may play a role in glucose sensing in the hypothalamic, pituitary, and gut endocrine cells [24].
In the pancreatic beta cell, glucokinase catalyzes the conversion of glucose to glucose-6-phosphate, which is the first reaction in glycolysis and a key regulator of insulin secretion. Interestingly, activating mutations in GCK can cause hypoglycemia secondary to constitutive hyperinsulinism, while inactivating mutations in GCK result in hyperglycemia. In patients with inactivating mutations, heterozygous mutations result in GCK MODY [25], while compound heterozygous or homozygous mutations result in permanent neonatal diabetes mellitus [26,27].
To date, over 600 mutations throughout the 10 exons of the pancreatic beta cell isoform of glucokinase have been reported. There are no clear “hot spots” for these mutations [24], and partial or complete gene deletions are rare [28,29]. Functional studies of various glucokinase mutations have resulted in the creation of a mathematical model that predicts the glucose threshold associated with insulin release in various mutations [30]. However, there remain inconsistencies between the expected enzyme activity based on the mutation and the clinical phenotype, a discrepancy which may be due to alterations in enzyme stability or binding as additional variables beyond simply enzyme activity [24]. Although larger studies have argued that most mutations in GCK MODY are private, there are reports of mutations suggesting a founder effect in parts of France, the UK, Norway, and Spain [31,32,33,34].
2.3. HNF4A MODY (MODY 1)
HNF4A MODY, also known as MODY 1, is the third most common form of MODY overall, with a reported prevalence ranging from 5 to 10% among all forms of MODY [6,35].
The HNF4A gene is located on chromosome 20, region 20q13.2, and encodes for the hepatocyte nuclear factor 4 alpha (HNF4alpha) protein, a member of the nuclear receptor family of ligand-activated transcription factors [11]. The HNF4A gene encodes for nine different isoforms that are differentially expressed in the liver, kidney, and pancreas [3,11]. In the pancreas, HNF1A activates transcription of the HNF4A gene [11], and in turn the HNF4alpha protein regulates the expression of the HNF1A gene [36].
Over 400 mutations of HNF4A have been described, including missense mutations (most common), frameshifts, splice-site mutations, and partial or whole gene deletions. The mutations may be found throughout the gene but are most common in exons 7 and 8 and the least common in exons 9 and 10. The majority of mutations in HNF4A are private, without clear evidence of a founder effect in this subtype [11]. Similar to HNF1A, the position of a mutation in HNF4A may be prognostic for the age of diabetes onset, with mutations in the P2 promoter, exon 1D, exon 9, and exon 10 resulting in a later age of diabetes onset compared to mutations in exons 2–8 [37].
2.4. HNF1B MODY (MODY 5)
HNF1B MODY, also known as MODY 5 [38], is the fourth most common form of MODY overall, with a reported prevalence of approximately 2–6% among all forms of MODY [6,39]. The HNF1B gene is located on chromosome 17, region 17q12, and encodes for the hepatocyte nuclear factor 1 beta (HNF1beta) protein (also known as Transcription Factor 2 or TCF2), a member of the homeobox-containing transcription factor family that regulates development of the liver, kidney, and pancreas [40]. The HNF1B gene encodes for three different isoforms and is highly expressed in the kidney relative to liver and pancreatic islet cells. Interestingly, although both liver and islet cells appear to contain significant amounts of HNF1B mRNA, HNF1-beta protein levels are barely detectable in these tissues, suggesting significant post-translational modification [41].
Approximately 50% of cases of MODY 5 result from mutations within the HNF1B gene, while the remaining 50% result from a heterozygous whole HNF1B gene deletion [42,43]. Over 50 different HNF1B gene mutations have been described, and these mutations commonly result in a phenotype of beta cell dysfunction, diabetes, and renal cysts often referred to as RCAD (Renal Cysts And Diabetes) Syndrome [44]. The 17q12 deletion syndrome, resulting from a large gene deletion on the long arm of chromosome 17, is characterized by neurodevelopmental abnormalities, diabetes, and renal or urinary tract malformations and is nearly always associated with a whole HNF1B gene deletion [45,46]. There appear to be differences in BMI and degree of renal dysfunction among patients with gene mutations compared to those with whole gene deletions [47], rendering it clinically relevant to identify the underlying genetic alteration.
2.5. PDX1/IPF1-MODY (MODY 4)
Mutations in the PDX1 (also known as IPF1) gene result in PDX1/IPF1-MODY, also known as MODY4 [48]. The PDX-1 (Pancreatic and Duodenal Homeobox-1) gene is located on chromosome 13, region 13q12.2, and encodes for the PDX1 protein. The protein encoded by PDX1 is a transcription factor involved in the regulation of pancreatic, beta cell, and duodenal development and is one of the earliest markers of pancreatic differentiation [49]. PDX1 is also involved in transcriptional activation of several genes, including insulin, somatostatin, glucokinase, islet amyloid polypeptide, and glucose transporter type 2 [50]. In mouse models, homozygous PDX1 gene deletion results in pancreatic agenesis, while the haploinsufficient model results in impaired insulin secretion and impaired glucose tolerance [51,52]. Human studies have identified various missense mutations in the PDX1 associated with increased risk of developing diabetes [48]. In vitro models using common mutations in amino acid positions 18 and 33 suggests that heterozygous mutations impair in vitro β-cell differentiation and function, while homozygous point mutations result in downregulation of several key transcription factors such as NEUROD1and ISLI; diminished β-cell responsiveness through mis-regulation of several PDX1 target genes involved in β-cell development, maturation, and function; and decreased glucose-sensitive responsiveness in differentiated beta cells [50].
2.6. NEUROD1 MODY (MODY 6)
Mutations in the NEUROD1 gene result in NEUROD1 MODY, also known as MODY 6 [53]. The NEUROD1 gene is located on chromosome 2, region 2q32, and encodes for the neurogenic differentiation 1 (NeuroD1) protein, also called β2 [54]. NeuroD1 is a transcription factor that regulates the expression of the insulin gene and is involved in the regulation of beta cell development. In mice, homozygous NEUROD1 disruption results in inadequate expression of the insulin gene, severe beta cell mass reduction, diabetes, and early death [55]. In humans, both heterozygous and homozygous mutations are described as being associated with an increased risk of developing diabetes mellitus [53,56].
2.7. KLF11 MODY (MODY 7)
Mutations in the KLF11 gene result in KLF11 MODY, also known as MODY 7 [57]. The krueppel-like 11(KLF11) gene is located on chromosome 2, region 2q25, and encodes for the KLF11 protein, a transcription factor that regulates PDX1 and INS gene expression in pancreatic beta cells, the latter via binding to the insulin promoter and regulating its activity in beta cells [57,58]. Various studies demonstrated that KLF11 also regulates genes involved in cholesterol, prostaglandin, neurotransmitter, fat, and sugar metabolism [59,60,61,62,63]. Various mutations in KLF11 have been described in families with very early onset diabetes mellitus type 1B [64], while others are associated with later onset diabetes [65,66].
2.8. CEL MODY (MODY 8)
Mutations in the CEL gene result in CEL MODY, also known as MODY 8 [67]. The CEL (carboxyl-ester lipase) gene is located on chromosome 9, region 9q34, and is expressed in the acinar tissues of the pancreas and in mammary glands [68,69]. Human CEL contains 11 exons, with a variable number of tandem repeats (VNTR) present in the coding region of exon 11 [67]. The protein product, which is the pancreatic CEL enzyme, is involved in cholesterol and lipid-soluble vitamin absorption [70]. Endocrine dysfunction appears to be associated with protein misfolding and intracellular aggregation rather that a secretory defect [71]. A phenotype of diabetes and exocrine dysfunction has been described in association with deletions within the VNTR region of the CEL gene [72]. Interestingly, mouse models with global CEL gene knockout do not exhibit the disease phenotype, suggesting that the mutations result in a negative gain-of-function effect [73].
2.9. PAX4 MODY (MODY 9)
Mutations in the PAX4 gene result in PAX4 MODY, also known as MODY 9 [74]. The PAX4 (Paired Homeobox 4) gene is located on chromosome 7, region 7q32, and encodes for the PAX 4 protein. PAX 4 is a transcription factor that is critical for the formation of beta cells, as it is required to maintain the expression of PDX1 and NKX 6.1, which are essential regulators of beta cell development and function [75]. PAX4 is important as a regulator in commitment of progenitor cells to mature islet cells, as evidenced by a few mature alpha and beta cells in heterozygous PAX 4 knockout mouse models [76]. In adults, PAX 4 mutations may result in diabetes due to impaired beta cell regeneration [77].
2.10. INS MODY (MODY 10)
Mutations in the INS gene result in INS MODY, also known as MODY 10 [78,79]. The INS gene is located on chromosome 11, region 11p15.5, and encodes for preproinsulin protein. Preproinsulin is then further cleaved to proinsulin and subsequently insulin. Autoimmune destruction of insulin producing beta cells results in autoimmune type 1 diabetes while various genetic and environmental factors result in progressive insulin resistance, insulinopenia, and type 2 diabetes mellitus.
Mutations in the insulin gene appear to result in misfolding and intracellular accumulation of proinsulin, defective insulin synthesis, and ultimately beta cell apoptosis. Although described in the literature, INS gene mutations are less likely to result in MODY and more likely to result in neonatal diabetes mellitus, particularly when the diagnosis is made under 1 year of age [80].
2.11. BLK MODY (MODY 11)
Mutations in the BLK gene result in BLK MODY, also known as MODY 11 [81]. The BLK (B Lymphocyte Kinase) gene is located on chromosome 8, region 8p23, and is a member of the SRC family of proto-oncogenes that is preferentially expressed in B-lymphocyte and beta cells. The gene encodes for the BLK protein, which has a role in B cell signaling and B cell development and upregulates the transcription factors PDX1 and NKX6.1, which promote insulin synthesis and secretion [81]. Heterozygous gene mutations are purported used to greatly attenuate this process, resulting in MODY 11. More recent analysis of a European cohort did not find a high association between BLK gene mutations and MODY, but the analysis did note a possible weak association with type 2 diabetes, particularly in the context of obesity [82].
2.12. ABCC8 MODY (MODY 12)
Mutations in the ABCC8 gene result in ABCC MODY, also known as MODY 12 [83]. The ABCC8 (ATP-binding cassette transporter subfamily C member 8) gene is located on chromosome 11, region 11p15, and is highly expressed in the pancreas. The gene encodes for the SUR1 (Sulfonylurea Receptor-1) protein, a subunit of the ATP-sensitive potassium (K-ATP) channel, which regulates insulin secretion from pancreatic beta cells. Activating mutations in the ABCC8 gene result in neonatal diabetes mellitus, whereas loss-of-function mutations cause congenital hyperinsulinism [84]. Heterozygous mutations in ABCC8 are associated with impaired insulin secretion and the subsequent development of diabetes mellitus [83,85].
2.13. KCNJ11 MODY (MODY 13)
Mutations in the KCJN11 gene result in KCJN11 MODY, also known as MODY 13 [86,87]. The KCJN11 (Potassium Inwardly Rectifying Channel Subfamily J Member 11) gene is located on chromosome 11, region 11p15, and is highly expressed in the pancreas. The gene encodes for the KIR 6.2 (Sulfonylurea Receptor-1) protein, a subunit of the ATP-sensitive potassium (K-ATP) channel, which regulates insulin secretion from pancreatic beta cells. Activating mutations in the KCJN11 gene result in neonatal diabetes mellitus, whereas loss-of-function mutations cause congenital hyperinsulinism [84]. Heterozygous mutations in KCJN11 are associated with impaired insulin secretion and subsequent development of diabetes mellitus [86,87].
2.14. APPL1 MODY (MODY 14)
Mutations in the APPL1 gene result in APPL1 MODY, also known as MODY 14 [88,89]. The APPL1 (the adaptor protein, phosphotyrosine interaction, PH domain, and leucine zipper-containing protein 1) gene is located on chromosome 3, region 3p14.3, and is highly expressed in skeletal muscle, pancreas, liver, and adipose tissue [90]. APPL1 protein is a mediator of adiponectin signaling and plays a critical role in cell proliferation, apoptosis, and cell survival. APPL1 is involved in insulin-mediated glucose uptake in adipose tissue and is an important mediator of adiponectin-dependent insulin sensitization in skeletal muscles [90,91,92]. Heterozygous mutations in APPL1 gene result in defects in insulin secretion and reduced beta cell survival [88,89,93].
3. Clinical Presentation
There is significant variability in clinical presentation between people with a shared subtype of MODY and between people with different MODY subtypes. The sections below highlight important clinical features of each subtype of MODY.
3.1. HNF1a (MODY 3)
The age of onset of diabetes is typically under age 25 years. Phenotypically, patients are typically not obese and do not demonstrate acanthosis nigricans or other signs of insulin resistance. There may be a history of transient neonatal hyperinsulinemic hypoglycemia, and glycosuria may be noted before overt hyperglycemia. Starting at the end of the first decade of life, there is progressive asymptomatic hyperglycemia and progressive decline in insulin secretion over time. Clinical diagnosis ranges from asymptomatic hyperglycemia to overt insulinopenia with ketosis and hyperglycemia. Patients often have higher serum ghrelin and HDL levels and lower serum hsCRP compared to patients with type 1 and type 2 diabetes mellitus. A family history of diabetes will be reported in a manner resembling autosomal dominant inheritance. Earlier age of diabetes onset is associated with maternal gene inheritance and exposure to diabetes in utero [5].
3.2. GCK (MODY 2)
Patients with GCK MODY usually present prior to age 25 years with mild asymptomatic hyperglycemia or may be diagnosed with gestational diabetes while undergoing routine prenatal care. Phenotypically, patients are typically not obese and lack evidence of insulin resistance. Hyperglycemia, whether fasting or post-prandial, is typically mild and non-progressive. Blood glucose levels rarely rise above 200 mg/dL post-prandially, and Hba1c values are typically less than 7.5%. Therefore, modest dietary and lifestyle modifications can be beneficial, but mediation treatment is not routinely recommended outside of gestational diabetes. During pregnancy, unaffected infants born to mothers with MODY 2 are at risk of exposure to maternal hyperglycemia and secondary macrosomia, and there is an increased risk of maternal development of microvascular complications and type 2 diabetes. Therefore, insulin treatment is recommended in these women for gestational diabetes. Interestingly, babies with GCK MODY who inherit the maternal gene mutation may be born small for gestational age due to relatively deficient fetal insulin secretion in utero and are at greater risk for severe hypoglycemia. Treatment of hyperglycemia with insulin in these women during pregnancy is controversial and needs to be individualized. Family history will reflect an autosomal dominant inheritance pattern. There are reports of patients with GCK MODY developing positive diabetes autoantibodies and presenting with type 1 diabetes later in life. Although rare, this warrants ongoing clinical evaluation and follow-up [3,23,24].
3.3. HNF4a (MODY 1)
Diabetes due to HNF4a MODY typically has its onset during the second to third decade of life but may be diagnosed later in some patients. There may be a history of fetal macrosomia and/or a report of transient neonatal hyperinsulinemic hypoglycemia. Although they typically lack evidence of obesity and insulin resistance, patients will have elevated serum LDL and triglyceride and decreased HDL levels, features that overlap with type 2 diabetes and result in misclassification in diagnosis. Although the initial presentation may be mild to modest hyperglycemia, there is eventually progressive decline in insulin secretion and a requirement for exogenous insulin therapy. Family history reflects an autosomal dominant inheritance, and earlier onset of diabetes may be observed in patients with specific mutations [4,94].
3.4. HNF1b (MODY 5)
The age of onset for diagnosis of diabetes in patients with MODY 5 is typically the third decade of life. Renal disease, in the form of renal cysts, renal or urinary tract anomalies, and decreased renal function, often precede the diagnosis of diabetes. Phenotypically, patients with HNF1b gene mutations are more likely to have decreased renal function, elevated BMI, and insulin resistance as compared to to patients with a whole gene deletion. Whole gene deletions may also be associated with the 17q12 deletion syndrome, which includes neurodevelopmental disorders, diabetes, and renal or urinary tract malformations. HNF1b MODY is associated with pancreatic agenesis and atrophy and near universal requirement for exogenous insulin therapy. Inheritance is in an autosomal dominant pattern [47].
3.5. PDX1 (MODY 4)
The age of diabetes onset for patients with PDX1 is more variable, and cases have reported ages at diagnosis ranging from adolescence to the sixth decade of life. Due to the small number of overall identified cases, the clinical phenotype is difficult to classify. Infants with homozygous mutations were noted to have neonatal diabetes and pancreatic agenesis, while adults with heterozygous mutations and hyperglycemia are reported to have a BMI within the normal range [48,95].
3.6. NEUROD1 (MODY 6)
The age of onset of diabetes in patients with NEUROD1 MODY varies widely, from the second to the sixth decade of life, with average ages of onset in the third to fourth decade of life. Phenotypically, patients who were obese had elevated insulin levels and were more similar to patients with type 2 diabetes, while those without obesity had low insulin levels and were more likely to be on insulin therapy [53,96].
3.7. KLF11 (MODY 7)
Diabetes in patients with KLF11 MODY was initially limited to a study of two mutations in three families with early onset diabetes. The ages of diabetes onset ranged from 23 to 56 years, and treatment was described as including oral hypoglycemic agents as well as insulin. A more recent report identified a KLF 11 gene mutation in a family with very early onset insulin dependent diabetes (labeled a Type 1B diabetes), with two children requiring insulin from age 1 year old (autoantibody negative) and their mother diagnosed with insulin dependent diabetes herself at age 4 years [58,64].
3.8. CEL (MODY 8)
The mean age of diabetes onset in CEL MODY is during the fourth decade of life. Affected patients develop exocrine pancreatic dysfunction due to fecal elastase deficiency by the second decade of life and present signs and symptoms of malabsorption. Pancreatic cysts develop in affected patients who also subsequently develop diabetes mellitus, and treatment may involve oral hypoglycemic agents and ultimately insulin therapy due to progressive beta cell dysfunction and pancreatic atrophy [72,97].
3.9. PAX4 (MODY 9)
In patients of West African heritage, PAX4 gene mutations are predisposed to a phenotype of ketosis-prone diabetes [98]. This form of diabetes presents with insulinopenia and absolute insulin dependence at diagnosis, followed by a course of either continued insulin dependence or a period of treatment which can be achieved without insulin. Mean age of onset is during the third decade of life. Ultimately, the majority of subjects (>75%) require insulin therapy within 10 years of diabetes diagnosis. In a small Thai cohort, PAX 4 MODY presented with impaired glucose tolerance in the second decade of life, diabetes was reported to develop between ages 20 and 50 years, and management included diet and oral hypoglycemic agents [74].
3.10. INS (MODY 10)
Mutations in INS MODY have been more commonly described as causing neonatal diabetes associated with small size for gestational age at birth. Outside of the neonatal period, INS gene mutations are associated with early onset diabetes in the second decade of life. Cases have been described as having hyperglycemia and ketoacidosis due to the insulin deficiency resulting from progressive loss of insulin secretion. The described cases have largely been described to require insulin therapy [78,79,80].
3.11. BLK (MODY 11)
Patients with BLK MODY appear to develop diabetes at approximately the third to fourth decade of life. Phenotypically, affected patients are overweight/obese, and among those with a genetic mutation, obesity is associated with an increased risk of diabetes. Among 21 individuals with BLK MODY from three families, approximately two-third required insulin therapy, while the remainder reported adequate diabetes control with diet and oral hypoglycemic agents [81].
3.12. ABCC8 (MODY 12) and KCNJ11 (MODY 13)
The clinical presentations of patients with ABCC8 and KCNJ11 MODY are similar. There may be a reported history of transient hyperinsulinemic hypoglycemia at birth. Age of onset of diabetes is in the second to third decade of life, although interestingly some case reports describe patients who were initially diagnosed in very early childhood and infancy with hyperglycemia transiently requiring insulin treatment. Affected individuals with ABCC8 or JKCN11 MODY have a normal BMI, and they may initially be misdiagnosed as having type 1 diabetes due to low serum insulin levels despite having no detectable diabetes autoantibodies. Family history often identifies early onset diabetes in young adults in an autosomal dominant pattern. Sulfonylureas are the treatment of choice [83,85,87,99].
3.13. APPL1 (MODY 14)
Among two large family cohorts, one in Italy and the other in the United States, the median age of onset of diabetes in those with APPL1 MODY was 38 years, with a range of 20 to 50 years of age. Affected individuals with diabetes were overweight or obese, and the majority were treated with insulin [88,89].
4. Indications for Testing
4.1. General Considerations
Historical recommendations for MODY testing focused on a young age at diagnosis (under 25 years of age), negative pancreatic autoantibodies, no insulin requirement, and a family history of diabetes in an autosomal dominant pattern. [100]. An additionally important group to consider is women with newly diagnosed gestational diabetes mellitus (GDM). While testing all women with GDM for MODY is neither practical nor cost-effective, there is evidence suggesting that a higher fasting glucose level (i.e., >5.5 mmol/L) and a normal BMI increases the likelihood of a diagnosis of MODY in women diagnosed with GDM, which would have important implications on the need for and intensity of treatment during pregnancy [101].
With an increasing number of MODY subtypes identified and greater reporting of the various phenotypic associations of the various subtypes, a broader net may be appropriate to cast when considering testing. Clinicians should be knowledgeable about the co-existing findings in various forms of MODY, including exocrine pancreatic dysfunction, pancreatic atrophy, low serum hsCRP levels, renal cysts or glycosuria preceding the diabetes diagnosis, lower serum HDL and triglyceride levels, and neurodevelopmental delays, findings which would increase clinical suspicions of MODY. Given the obesity epidemic in both children and adults, it would behoove the clinician to use judgement when using the presence of an elevated BMI as an exclusion to MODY testing in a subject with otherwise high clinical suspicion.
Given the factors that need to be considered in proceeding with MODY testing, various prediction models and a MODY calculator have been developed to help predict the likelihood of a positive MODY test [102,103,104]. It appears that strong family history, negative diabetes autoantibodies, residual beta cell function, and early age at diagnosis (<25–30 years) remain strongly predictive variables [105].
4.2. Patients with Fasting Hyperglycemia or IGT
In a study of 100 Italian children with incidentally identified fasting or illness-related hyperglycemia, Aloi and colleagues identified a high prevalence of GCK MODY (88%) when strict clinical criteria were applied for testing (hyperglycemia diagnosed at age under 25 years, impaired fasting glucose or impaired 2 h glucose on OGTT, family history of diabetes, absence of obesity, and absence of diabetes autoantibodies), and a modest but clinically relevant prevalence of 32% with less strict clinical criteria for testing (hyperglycemia and absence of diabetes autoantibodies) [23]. Haliloglu and colleagues, in a cohort of 54 Turkish children diagnosed with IGTor T2DM without insulin requirement under 17 years of age, identified CGK MODY mutations in 24% of subjects [106]. Among an older cohort of adults with IGT and a mean age of 52 years, the incidence of CGK MODY was 0.76%, indicating that IGT in older adults is more likely to be due to T2DM rather than GCK MODY [107]. These studies suggest that MODY testing, particularly the GCK subtype, should be pursued in children and young adults with persistent asymptomatic hyperglycemia or glucose impairment and relevant clinical findings.
4.3. Patients with Positive Diabetes Autoantibodies
Diabetes-specific autoantibodies are present in less than 2% of the general population compared to 80–90% in those with newly diagnosed type 1 diabetes. [108]. The prevalence of islet-specific autoantibodies in patients with MODY appears to be low and similar to that in the general population, resulting in the recommendation that the presence of diabetes autoantibodies can serve as a negative predictive factor toward testing patients for MODY [109,110]. Contrary to this recommendation, various reports have demonstrated the co-existence of islet-specific autoantibodies and MODY, [111,112,113,114]. Therefore, clinicians may consider testing for MODY in select groups of patients who may have a single positive diabetes autoantibody in the appropriate clinical context, particularly if there is a strong family history, although the likelihood of a diagnosis of MODY in those with multiple positive diabetes autoantibodies remains low.
5. Treatment
At the time of initial diagnosis, the etiology of the hyperglycemia is generally unknown. Therefore, the degree of hyperglycemia, hydration status, and presence or absence of ketosis determines initial therapy. Type 1 diabetes in both children and adults requires lifelong insulin therapy, while type 2 diabetes requires an individualized approach. The ADA and ISPAD have outlined recommendations for the management of type 2 diabetes [115,116], including guidelines for the initiation of insulin treatment, which is more likely to be required in children and adolescents with type 2 diabetes both at diagnosis and relatively soon thereafter [117,118].
Over time, additional studies have improved our understanding of the natural history of MODY. There is great interest among both providers and patients in data suggesting that people with a diagnosis of MODY may be able to convert their treatment, in part or in its entirety, from insulin therapy to oral hypoglycemic agents.
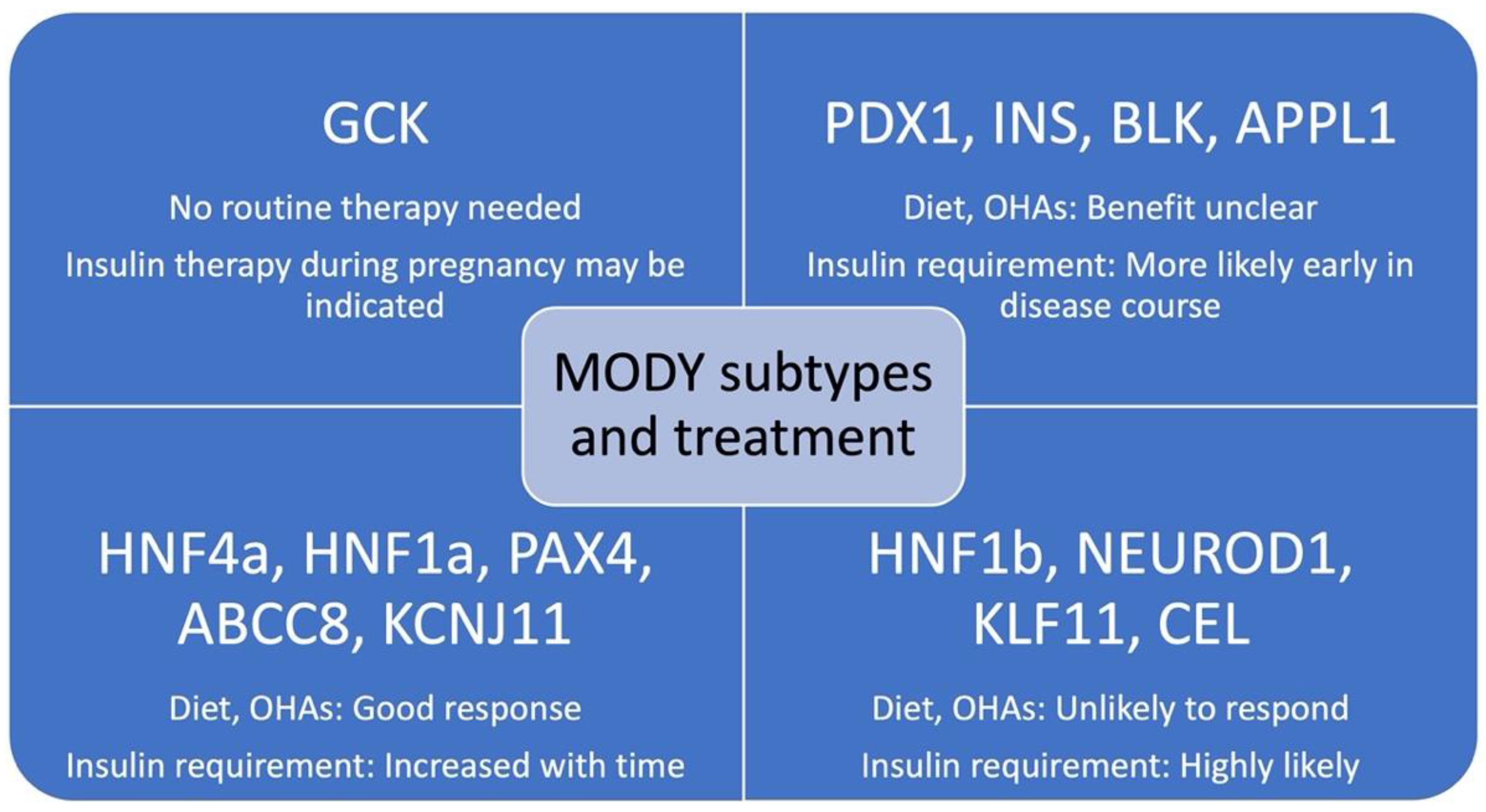
Figure 1 summarizes treatment options and recommendations for various forms of MODY. Key recommendations for GCK MODY include the lack of treatment requirement in the majority of GCK MODY given the stable mild hyperglycemia and lack of vascular complications [119]. During pregnancy, the decision for insulin treatment should be considered in the context of whether the fetus is suspected to have inherited the maternal GCK gene mutation. If a fetus is suspected of being affected by the maternal GCK mutation, insulin treatment during pregnancy is not routinely recommended as insulin therapy has been associated with small fetal birth size, early delivery, and increased risk of severe neonatal hypoglycemia. In the case where the fetus is unaffected, insulin therapy is generally recommended to avoid fetal macrosomia [120,121]. For HNF1a and HNF4a MODY, patients are expected to respond readily to oral sulfonylureas with a higher insulin sensitivity than patients with T2DM. Of interest, newer modalities of therapy in this latter group with GLP1 receptor agonists show early promise. It should be noted that the most common forms of MODY will have a greater level of available data to guide these recommendations, and future studies will continue to inform and improve the precision and validity of these recommendations.

Figure 1.
MODY subtypes and pharmacologic treatment options. OHAs = oral hypoglycemic agents; GDM = gestational diabetes mellitus. [47,119,120,121,122,123,124,125,126,127,128,129].
6. Conclusions
Maturity Onset Diabetes of the Young (MODY) encompasses a group of monogenic forms of diabetes with a wide array of clinical phenotypes. Clinicians should be vigilant and consider genetic testing in those who meet criteria for a high likelihood of poessing a form of MODY, particularly given the potential for a successful change in treatment modality from insulin to an oral hypoglycemic agent. With improvements in both access to and accuracy of genetic testing, genotype/phenotype correlations will continually be refined and can improve our understanding of the treatment and prognosis of the various forms of MODY, particularly those that are less common. As additional forms are undoubtedly discovered, patients and clinicians will benefit from these discoveries by improved outcomes in those with MODY.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Conflicts of Interest
The author declares no conflict of interest.
References
- Centers for Disease Control and Prevention. National Diabetes Statistics Report 2020. Available online: Cdc.gov/diabetes/data/statistics-report/index.html (accessed on 26 November 2021).
- Van Name, M.A.; Cheng, P.; Gal, R.L.; Kollman, C.; Lynch, J.; Nelson, B.; Tamborlane, W.V. Pediatric Diabetes Consortium. Children and adolescents with type 1 and type 2 diabetes mellitus in the Pediatric Diabetes Consortium Registries: Comparing clinical characteristics and glycaemic control. Diabet. Med. 2020, 37, 863–867. [Google Scholar] [CrossRef]
- Nkonge, K.M.; Nkonge, D.K.; Nkonge, T.N. The epidemiology, molecular pathogenesis, diagnosis, and treatment of maturity-onset diabetes of the young (MODY). Clin. Diabetes Endocrinol. 2020, 6, 20. [Google Scholar] [CrossRef]
- Fajans, S.S.; Bell, G.I.; Polonsky, K.S. Molecular mechanisms and clinical pathophysiology of maturity-onset diabetes of the young. N. Engl. J. Med. 2001, 345, 971–980. [Google Scholar] [CrossRef]
- Valkovicova, T.; Skopkova, M.; Stanik, J.; Gasperikova, D. Novel insights into genetics and clinics of the HNF1A-MODY. Endocr. Regul. 2019, 53, 110–134. [Google Scholar] [CrossRef] [PubMed]
- Sperling, M.A.; Garg, A. Monogenic Forms of Diabetes. In Diabetes in America, 3rd ed.; Cowie, C.C., Casagrande, S.S., Menke, A., Cissell, M.A., Eberhardt, M.S., Meigs, J.B., Gregg, E.W., Knowler, W.C., Barrett-Connor, E., Becker, D.J., Eds.; National Institute of Diabetes and Digestive and Kidney Diseases: Bethesda, MD, USA, 2018; pp. 1–27. [Google Scholar]
- Pontoglio, M.; Prie, D.; Cheret, C.; Doyen, A.; Leroy, C.; Froguel, P.; Velho, G.; Yaniv, M.; Friedlander, G. HNF1α controls renal glucose reabsorption in mouse and man. EMBO Rep. 2000, 1, 359–365. [Google Scholar] [CrossRef]
- Lau, H.H.; Ng, N.H.J.; Loo, L.S.W.; Jasmen, J.B.; Teo, A.K.K. The molecular functions of hepatocyte nuclear factors—In and beyond the liver. J. Hepatol. 2018, 68, 1033–1048. [Google Scholar] [CrossRef]
- Godart, F.; Bellanne-Chantelot, C.; Clauin, S.; Gragnoli, C.; Abderrahmani, A.; Blanche, H.; Boutin, P.; Chevre, J.C.; Froguel, P.; Bailleul, B. Identification of seven novel nucleotide variants in the hepatocyte nuclear factor-1α (TCF1) promoter region in MODY patients. Hum. Mutat. 2000, 15, 173–180. [Google Scholar] [CrossRef]
- Lausen, J.; Thomas, H.; Lemm, I.; Bulman, M.; Borgschulze, M.; Lingott, A.; Hattersley, A.T.; Ryffel, G.U. Naturally occurring mutations in the human HNF4alpha gene impair the function of the transcription factor to a varying degree. Nucleic Acids Res. 2000, 28, 430–437. [Google Scholar] [CrossRef][Green Version]
- Colclough, K.; Bellanne-Chantelot, C.; Saint-Martin, C.; Flanagan, S.E.; Ellard, S. Mutations in the genes encoding the transcription factors hepatocyte nuclear factor 1 alpha and 4 alpha in maturity-onset diabetes of the young and hyperinsulinemic hypoglycemia. Hum. Mutat. 2013, 34, 669–685. [Google Scholar] [CrossRef] [PubMed]
- Lek, M.; Karczewski, K.J.; Minikel, E.V.; Samocha, K.E.; Banks, E.; Fennell, T.; O’Donnell-Luria, A.H.; Ware, J.S.; Hill, A.J.; Cummings, B.B.; et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature 2016, 536, 285–291. [Google Scholar] [CrossRef]
- Vaxillaire, M.; Abderrahmani, A.; Boutin, P.; Bailleul, B.; Froguel, P.; Yaniv, M.; Pontoglio, M. Anatomy of a homeoprotein revealed by the analysis of human MODY3 mutations. J. Biol. Chem. 1999, 274, 35639–35646. [Google Scholar] [CrossRef] [PubMed]
- Harries, L.W.; Ellard, S.; Stride, A.; Morgan, N.G.; Hattersley, A.T. Isomers of the TCF1 gene encoding hepatocyte nuclear factor-1 alpha show differential expression in the pancreas and define the relationship between mutation position and clinical phenotype in monogenic diabetes. Hum. Mol. Genet. 2006, 15, 2216–2224. [Google Scholar] [CrossRef]
- Bellanné-Chantelot, C.; Carette, C.; Riveline, J.P.; Valéro, R.; Gautier, J.F.; Larger, E.; Reznik, Y.; Ducluzeau, P.H.; Sola, A.; Hartemann-Heurtier, A.; et al. The type and the position of HNF1A mutation modulate age at diagnosis of diabetes in patients with maturity-onset diabetes of the young (MODY)-3. Diabetes 2008, 57, 503–508. [Google Scholar] [CrossRef]
- Awa, W.L.; Thon, A.; Raile, K.; Grulich-Henn, J.; Meissner, T.; Schober, E.; Holl, R.W.; DPV-Wiss. Study Group. Genetic and clinical characteristics of patients with HNF1A gene variations from the German-Austrian DPV database. Eur. J. Endocrinol. 2011, 164, 513–520. [Google Scholar] [CrossRef]
- Stride, A.; Vaxillaire, M.; Tuomi, T.; Barbetti, F.; Njølstad, P.R.; Hansen, T.; Costa, A.; Conget, I.; Pedersen, O.; Søvik, O.; et al. The genetic abnormality in the beta cell determines the response to an oral glucose load. Diabetologia 2002, 45, 427–435. [Google Scholar] [CrossRef]
- Klupa, T.; Warram, J.H.; Antonellis, A.; Pezzolesi, M.; Nam, M.; Malecki, M.T.; Doria, A.; Rich, S.S.; Krolewski, A.S. Determinants of the development of diabetes (maturity-onset diabetes of the young-3) in carriers of HNF-1alpha mutations: Evidence for parent-of-origin effect. Diabetes Care 2002, 25, 2292–2301. [Google Scholar] [CrossRef]
- Naqvi, A.A.T.; Hasan, G.M.; Hassan, M.I. Investigating the role of transcription factors of pancreas development in pancreatic cancer. Pancreatology 2018, 18, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Vesterhus, M.; Haldorsen, I.S.; Raeder, H.; Molven, A.; Njølstad, P.R. Reduced pancreatic volume in hepatocyte nuclear factor 1A-maturity-onset diabetes of the young. J. Clin. Endocrinol. Metab. 2008, 93, 3505–3509. [Google Scholar] [CrossRef]
- Pearson, E.R.; Starkey, B.J.; Powell, R.J.; Gribble, F.M.; Clark, P.M.; Hattersley, A.T. Genetic cause of hyperglycaemia and response to treatment in diabetes. Lancet 2003, 362, 1275–1281. [Google Scholar] [CrossRef]
- Małachowska, B.; Borowiec, M.; Antosik, K.; Michalak, A.; Baranowska-Jaźwiecka, A.; Deja, G.; Jarosz-Chobot, P.; Brandt, A.; Myśliwiec, M.; Stelmach, M.; et al. Monogenic diabetes prevalence among Polish children-Summary of 11 years-long nationwide genetic screening program. Pediatr. Diabetes 2018, 19, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Aloi, C.; Salina, A.; Minuto, N.; Tallone, R.; Lugani, F.; Mascagni, A.; Mazza, O.; Cassanello, M.; Maghnie, M.; d’Annunzio, G. Glucokinase mutations in pediatric patients with impaired fasting glucose. Acta Diabetol. 2017, 54, 913–923. [Google Scholar] [CrossRef] [PubMed]
- Osbak, K.K.; Colclough, K.; Saint-Martin, C.; Beer, N.L.; Bellanné-Chantelot, C.; Ellard, S.; Gloyn, A.L. Update on mutations in glucokinase (GCK), which cause maturity-onset diabetes of the young, permanent neonatal diabetes, and hyperinsulinemic hypoglycemia. Hum. Mutat. 2009, 30, 1512–1526. [Google Scholar] [CrossRef] [PubMed]
- Froguel, P.; Zouali, H.; Vionnet, N.; Velho, G.; Vaxillaire, M.; Sun, F.; Lesage, S.; Stoffel, M.; Takeda, J.; Passa, P. Familial hyperglycemia due to mutations in glucokinase—definition of a subtype of diabetes mellitus. N. Engl. J. Med. 1993, 328, 697–702. [Google Scholar] [CrossRef]
- Njølstad, P.R.; Søvik, O.; Cuesta-Muñoz, A.; Bjørkhaug, L.; Massa, O.; Barbetti, F.; Undlien, D.E.; Shiota, C.; Magnuson, M.A.; Molven, A.; et al. Neonatal diabetes mellitus due to complete glucokinase deficiency. N. Engl. J. Med. 2001, 344, 1588–1592. [Google Scholar] [CrossRef]
- Hughes, A.E.; De Franco, E.; Globa, E.; Zelinska, N.; Hilgard, D.; Sifianou, P.; Hattersley, A.T.; Flanagan, S.E. Identification of GCK-maturity-onset diabetes of the young in cases of neonatal hyperglycemia: A case series and review of clinical features. Pediatr. Diabetes 2021, 22, 876–881. [Google Scholar] [CrossRef]
- Ellard, S.; Thomas, K.; Edghill, E.L.; Owens, M.; Ambye, L.; Cropper, J.; Little, J.; Strachan, M.; Stride, A.; Ersoy, B.; et al. Partial and whole gene deletion mutations of the GCK and HNF1A genes in maturity-onset diabetes of the young. Diabetologia 2007, 50, 2313–2317. [Google Scholar] [CrossRef]
- Garin, I.; Rica, I.; Estalella, I.; Oyarzabal, M.; Rodríguez-Rigual, M.; San Pedro, J.I.; Pérez-Nanclares, G.; Fernández-Rebollo, E.; Busturia, M.A.; Castaño, L.; et al. Haploinsufficiency at GCK gene is not a frequent event in MODY2 patients. Clin. Endocrinol. (Oxf.) 2008, 68, 873–878. [Google Scholar] [CrossRef]
- Gloyn, A.L.; Odili, S.; Buettger, C.; Njolstad, P.R.; Shiota, C.; Magnuson, M.A.; Matschinsky, F.M. Glucokinase and the regulation of blood sugar: A mathematical model predicts the threshold for glucose stimulated insulin release for GCK gene mutations that cause hyper- and hypoglycemia. In Glucokinase and Glycemic Diseases: From the Basics to Novel Therapies; Magnuson, M., Matschinsky, F.M., Eds.; Karger: Basel, Switzerland, 2004; pp. 92–109. [Google Scholar] [CrossRef]
- Dussoix, P.; Vaxillaire, M.; Iynedjian, P.B.; Tiercy, J.M.; Ruiz, J.; Spinas, G.A.; Berger, W.; Zahnd, G.; Froguel, P.; Philippe, J. Diagnostic heterogeneity of diabetes in lean young adults: Classification based on immunological and genetic parameters. Diabetes 1997, 46, 622–631. [Google Scholar] [CrossRef]
- Saker, P.J.; Hattersley, A.T.; Barrow, B.; Hammersley, M.S.; McLellan, J.A.; Lo, Y.M.; Olds, R.J.; Gillmer, M.D.; Holman, R.R.; Turner, R.C. High prevalence of a missense mutation of the glucokinase gene in gestational diabetic patients due to a founder-effect in a local population. Diabetologia 1996, 39, 1325–1328. [Google Scholar] [CrossRef] [PubMed]
- Estalella, I.; Rica, I.; Perez de Nanclares, G.; Bilbao, J.R.; Vazquez, J.A.; San Pedro, J.I.; Busturia, M.A.; Castaño, L.; Spanish MODY Group. Mutations in GCK and HNF-1alpha explain the majority of cases with clinical diagnosis of MODY in Spain. Clin. Endocrinol. (Oxf.) 2007, 67, 538–546. [Google Scholar] [CrossRef]
- Sagen, J.V.; Bjørkhaug, L.; Molnes, J.; Raeder, H.; Grevle, L.; Søvik, O.; Molven, A.; Njølstad, P.R. Diagnostic screening of MODY2/GCK mutations in the Norwegian MODY Registry. Pediatr. Diabetes 2008, 9, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Shields, B.M.; Hicks, S.; Shepherd, M.H.; Colclough, K.; Hattersley, A.T.; Ellard, S. Maturity-onset diabetes of the young (MODY): How many cases are we missing? Diabetologia 2010, 53, 2504. [Google Scholar] [CrossRef] [PubMed]
- Yamagata, K.; Furuta, H.; Oda, N.; Kaisaki, P.J.; Menzel, S.; Cox, N.J.; Fajans, S.S.; Signorini, S.; Stoffel, M.; Bell, G.I. Mutations in the hepatocyte nuclear factor-4alpha gene in maturity-onset diabetes of the young (MODY1). Nature 1996, 384, 458–460. [Google Scholar] [CrossRef]
- Harries, L.W.; Locke, J.M.; Shields, B.; Hanley, N.A.; Hanley, K.P.; Steele, A.; Njølstad, P.R.; Ellard, S.; Hattersley, A.T. The diabetic phenotype in HNF4A mutation carriers is moderated by the expression of HNF4A isoforms from the P1 promoter during fetal development. Diabetes 2008, 57, 1745–1752. [Google Scholar] [CrossRef] [PubMed]
- Horikawa, Y.; Iwasaki, N.; Hara, M.; Furuta, H.; Hinokio, Y.; Cockburn, B.N.; Lindner, T.; Yamagata, K.; Ogata, M.; Tomogana, O.; et al. Mutation in hepatocyte nuclear factor-1 beta gene (TCF-2) associated with MODY. Nat. Genet. 1997, 17, 384–385. [Google Scholar] [CrossRef]
- Clissold, R.L.; Hamilton, A.J.; Hattersley, A.T.; Ellard, S.; Bingham, C. HNF1B-associated renal and extrarenal disease: An expanding clinical spectrum. Nat. Rev. Nephrol. 2015, 11, 101–112. [Google Scholar] [CrossRef] [PubMed]
- El-Khairi, R.; Vallier, L. The role of hepatocyte nuclear factor 1beta in disease and development. Diabetes Obes. Metab. 2016, 18, 23–32. [Google Scholar] [CrossRef]
- Harries, L.W.; Brown, J.E.; Gloyn, A.L. Species-specific differences in the expression of the HNF1A, HNF1B and HNF4A genes. PLoS ONE 2009, 4, e785. [Google Scholar] [CrossRef]
- Bellanne-Chantelot, C.; Clauin, S.; Chauveau, D.; Colin, P.; Daumont, M.; Douillard, C.; Dubois-Laforgue, D.; Dusselier, L.; Gautier, J.F.; Jadoul, M.; et al. Large genomic rearrangements in the hetapocyte nuclear factor-1beta (TCF2) gene are the most frequent cause of maturity onset diabetes of the young (MODY)5. Diabetes 2005, 54, 3126–3132. [Google Scholar] [CrossRef]
- Edghill, E.L.; Oram, R.A.; Owens, M.; Stals, K.L.; Harrier, L.W.; Hattersley, A.T.; Ellard, S.; Bingham, C. Hepatocyte nuclear factor-1beta gene deletions—a common cause of renal disease. Nephrol. Dial. Transpl. 2008, 23, 627–635. [Google Scholar] [CrossRef]
- Lim, S.H.; Kim, J.H.; Han, K.H.; Ahn, Y.H.; Kang, H.G.; Ha, I.S.; Cheong, H.I. Genotype and phenotype analyses in pediatric patients with HNF1B mutations. J. Clin. Med. 2020, 9, 2320. [Google Scholar] [CrossRef]
- Laffargue, F.; Bourthoumieu, S.; Llanas, B.; Baudouin, V.; Lahoche, A.; Morin, D.; Bessanay, L.; De Parscau, L.; Cloarec, S.; Delrue, M.A.; et al. Toward a new point of view on the phenotype of patients with 17q12 microdeletion syndrome. Arch. Dis. Child. 2015, 100, 259–264. [Google Scholar] [CrossRef]
- Mitchel, M.W.; Moreno-De-Luca, D.; Myers, S.M.; Finucane, B.; Ledbetter, D.H.; Martin, C.L. 17q12 recurrent deletion syndrome. In Genereviews®; Pagon, R.A., Adam, M.P., Ardinger, H.H., Eds.; University of Washington: Seattle, WA, USA, 2016. Available online: ncbi.nlm.nih.gov/books/NBK1116 (accessed on 25 November 2021).
- Dubois-Laforgue, D.; Cornu, E.; Saint-Martin, C.; Coste, J.; Bellanné-Chantelot, C.; Timsit, J.; Monogenic Diabetes Study Group of the Société Francophone du Diabète. Diabetes, Associated Clinical Spectrum, Long-term Prognosis, and Genotype/Phenotype Correlations in 201 Adult Patients with Hepatocyte Nuclear Factor 1B (HNF1B) Molecular Defects. Diabetes Care 2017, 40, 1436–1443. [Google Scholar] [CrossRef] [PubMed]
- Stoffers, D.A.; Stanojevic, V.; Habener, J.F. Insulin promoter factor-1 gene mutation linked to early-onset type 2 diabetes mellitus directs expression of a dominant negative isoprotein. J. Clin. Invest. 1998, 102, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Liew, C.G.; Shah, N.N.; Briston, S.J.; Shepherd, R.M.; Khoo, C.P.; Dunne, M.J.; Moore, H.D.; Cosgrove, K.E.; Andrews, P.W. PAX4 enhances beta-cell differentiation of human embryonic stem cells. PLoS ONE 2008, 3, e1783. [Google Scholar] [CrossRef]
- Wang, X.; Sterr, M.; Burtscher, I.; Böttcher, A.; Beckenbauer, J.; Siehler, J.; Meitinger, T.; Häring, H.U.; Staiger, H.; Cernilogar, F.M.; et al. Point mutations in the PDX1 transactivation domain impair human β-cell development and function. Mol. Metab. 2019, 24, 80–97. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, J.; Carlsson, L.; Edlund, T.; Edlund, H. Insulin-promoter-factor 1 is required for pancreas development in mice. Nature 1994, 371, 606–609. [Google Scholar] [CrossRef]
- Brissova, M.; Shiota, M.; Nicholson, W.E.; Gannon, M.; Knobel, S.M.; Piston, D.W.; Wright, C.V.E.; Powers, A.C. Reduction in pancreatic transcription factor PDX-1 impairs glucose-stimulated insulin secretion. J. Biol. Chem. 2002, 277, 11225–11232. [Google Scholar] [CrossRef]
- Malecki, M.T.; Jhala, U.S.; Antonellis, A.; Fields, L.; Doria, A.; Orban, T.; Saad, M.; Warram, J.H.; Montminy, M.; Krolewski, A.S. Mutations in NEUROD1 are associated with the development of type 2 diabetes mellitus. Nat. Genet. 1999, 23, 323–328. [Google Scholar] [CrossRef]
- Tamimi, R.; Steingrimsson, E.; Copeland, N.G.; Dyer-Montgomery, K.; Lee, J.E.; Hernandez, R.; Jenkins, N.A.; Tapscott, S.J. The NEUROD gene maps to human chromosome 2q32 and mouse chromosome 2. Genomics 1996, 34, 418–421. [Google Scholar] [CrossRef]
- Naya, F.J.; Huang, H.P.; Qiu, Y.; Mutoh, H.; DeMayo, F.J.; Leiter, A.B.; Tsai, M.J. Diabetes, defective pancreatic morphogenesis, and abnormal enteroendocrine differentiation in BETA2/neuroD-deficient mice. Genes Dev. 1997, 11, 2323–2334. [Google Scholar] [CrossRef]
- Kanatsuka, A.; Tokuyama, Y.; Nozaki, O.; Matsui, K.; Egashira, T. Beta-cell dysfunction in late-onset diabetic subjects carrying homozygous mutation in transcription factors NeuroD1 and Pax4. Metabolism 2002, 51, 1161–1165. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Zapico, M.E.; van Velkinburgh, J.C.; Gutiérrez-Aguilar, R.; Neve, B.; Froguel, P.; Urrutia, R.; Stein, R. MODY7 gene, KLF11, is a novel p300-dependent regulator of Pdx-1 (MODY4) transcription in pancreatic islet beta cells. J. Biol. Chem. 2009, 284, 36482–36490. [Google Scholar] [CrossRef] [PubMed]
- Neve, B.; Fernandez-Zapico, M.E.; Ashkenazi-Katalan, V.; Dina, C.; Hamid, Y.H.; Joly, E.; Vaillant, E.; Benmezroua, Y.; Durand, E.; Bakaher, N.; et al. Role of transcription factor KLF11 and its diabetes-associated gene variants in pancreatic beta cell function. Proc. Natl. Acad. Sci. USA 2005, 102, 4807–4812. [Google Scholar] [CrossRef]
- Buttar, N.S.; DeMars, C.J.; Lomberk, G.; Rizvi, S.; Bonilla-Velez, J.; Achra, S.; Rashtak, S.; Wang, K.K.; Fernandez-Zapico, M.E.; Urrutia, R. Distinct role of Kruppel-like factor 11 in the regulation of prostaglandin E2 biosynthesis. J. Biol. Chem. 2010, 285, 11433–11444. [Google Scholar] [CrossRef]
- Cao, S.; Fernandez-Zapico, M.E.; Jin, D.; Puri, V.; Cook, T.A.; Lerman, L.O.; Zhu, X.Y.; Urrutia, R.; Shah, V. KLF11-mediated repression antagonizes Sp1/sterol-responsive element-binding protein-induced transcriptional activation of caveolin-1 in response to cholesterol signaling. J. Biol. Chem. 2005, 280, 1901–1910. [Google Scholar] [CrossRef] [PubMed]
- Grunewald, M.; Johnson, S.; Lu, D.; Wang, Z.; Lomberk, G.; Albert, P.R.; Stockmeier, C.A.; Meyer, J.H.; Urrutia, R.; Miczek, K.A.; et al. Mechanistic role for a novel glucocorticoid-KLF11 (TIEG2) protein pathway in stress-induced monoamine oxidase A expression. J. Biol. Chem. 2012, 287, 24195–24206. [Google Scholar] [CrossRef]
- Ou, X.M.; Chen, K.; Shih, J.C. Dual functions of transcription factors, transforming growth factor-β-inducible early gene (TIEG)2 and Sp3, are mediated by CACCC element and Sp1 sites of human monoamine oxidase (MAO) B gene. J. Biol. Chem. 2004, 279, 21021–21028. [Google Scholar] [CrossRef]
- Yamamoto, K.; Sakaguchi, M.; Medina, R.J.; Niida, A.; Sakaguchi, Y.; Miyazaki, M.; Kataoka, K.; Huh, N.H. Transcriptional regulation of a brown adipocyte-specific gene, UCP1, by KLF11 and KLF15. Biochem. Biophys. Res. Commun. 2010, 400, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Ushijima, K.; Narumi, S.; Ogata, T.; Yokota, I.; Sugihara, S.; Kaname, T.; Horikawa, Y.; Matsubara, Y.; Fukami, M.; Kawamura, T.; et al. KLF11 variant in a family clinically diagnosed with early childhood-onset type 1B diabetes. Pediatr. Diabetes 2019, 20, 712–719. [Google Scholar] [CrossRef]
- Tanahashi, T.; Shinohara, K.; Keshavarz, P.; Yamaguchi, Y.; Miyawaki, K.; Kunika, K.; Moritani, M.; Nakamura, N.; Yoshikawa, T.; Shiota, H.; et al. The association of genetic variants in Krüppel-like factor 11 and Type 2 diabetes in the Japanese population. Diabet Med. 2008, 25, 19–26. [Google Scholar] [CrossRef]
- Ma, L.; Hanson, R.L.; Que, L.N.; Mack, J.L.; Franks, P.W.; Infante, A.M.; Kobes, S.; Bogardus, C.; Baier, L.J. Association analysis of Krüppel-like factor 11 variants with type 2 diabetes in Pima Indians. J. Clin. Endocrinol. Metab. 2008, 93, 3644–3649. [Google Scholar] [CrossRef]
- Torsvik, J.; Johansson, S.; Johansen, A.; Ek, J.; Minton, J.; Raeder, H.; Ellard, S.; Hattersley, A.; Pedersen, O.; Hansen, T.; et al. Mutations in the VNTR of the carboxyl-ester lipase gene (CEL) are a rare cause of monogenic diabetes. Hum. Genet. 2010, 127, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, D. Bile salt-dependent lipase: Its pathophysiological implications. Biochim. Biophys. Acta 2001, 1533, 1–28. [Google Scholar] [CrossRef]
- Blackberg, L.; Angquist, K.A.; Hernell, O. Bile-salt-stimulated lipase in human milk: Evidence for its synthesis in the lactating mammary gland. FEBS Lett. 1987, 217, 37–41. [Google Scholar] [CrossRef]
- Lombardo, D.; Guy, O. Studies on the substrate specificity of a carboxyl ester hydrolase from human pancreatic juice. II. Action on cholesterol esters and lipid-soluble vitamin esters. Biochim. Biophys. Acta 1980, 611, 147–155. [Google Scholar] [CrossRef]
- Dalva, M.; Lavik, I.K.; El Jellas, K.; Gravdal, A.; Lugea, A.; Pandol, S.J.; Njølstad, P.R.; Waldron, R.T.; Fjeld, K.; Johansson, B.B.; et al. Pathogenic Carboxyl Ester Lipase (CEL) Variants Interact with the Normal CEL Protein in Pancreatic Cells. Cells 2020, 9, 244. [Google Scholar] [CrossRef]
- Raeder, H.; Johansson, S.; Holm, P.I.; Haldorsen, I.S.; Mas, E.; Sbarra, V.; Nermoen, I.; Eide, S.A.; Grevle, L.; Bjorkhaug, L.; et al. Mutations in the CEL VNTR cause a syndrome of diabetes and pancreatic exocrine dysfunction. Nat. Genet. 2006, 38, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Ræder, H.; Vesterhus, M.; El Ouaamari, A.; Paulo, J.A.; McAllister, F.E.; Liew, C.W.; Hu, J.; Kawamori, D.; Molven, A.; Gygi, S.P.; et al. Absence of diabetes and pancreatic exocrine dysfunction in a transgenic model of carboxyl-ester lipase-MODY (maturity-onset diabetes of the young). PLoS ONE 2013, 8, e60229. [Google Scholar] [CrossRef]
- Plengvidhya, N.; Kooptiwut, S.; Songtawee, N.; Doi, A.; Furuta, H.; Nishi, M.; Nanjo, K.; Tantibhedhyangkul, W.; Boonyasrisawat, W.; Yenchitsomanus, P.T.; et al. PAX4 mutations in Thais with maturity onset diabetes of the young. J. Clin. Endocrinol. Metab. 2007, 92, 2821–2826. [Google Scholar] [CrossRef]
- Wang, J.; Elghazi, L.; Parker, S.E.; Kizilocak, H.; Asano, M.; Sussel, L.; Sosa-Pineda, B. The concerted activities of Pax4 and Nkx2.2 are essential to initiate pancreatic beta-cell differentiation. Dev. Biol. 2004, 266, 178–189. [Google Scholar] [CrossRef]
- Sosa-Pineda, B.; Chowdhury, K.; Torres, M.; Oliver, G.; Gruss, P. The Pax4 gene is essential for differentiation of insulin-producing beta cells in the mammalian pancreas. Nature 1997, 386, 399–402. [Google Scholar] [CrossRef] [PubMed]
- Biason-Lauber, A.; Boehm, B.; Lang-Muritano, M.; Gauthier, B.R.; Brun, T.; Wollheim, C.B.; Schoenle, E.J. Association of childhood type 1 diabetes mellitus with a variant of PAX4: Possible link to beta-cell regenerative capacity. Diabetologia 2005, 48, 900–905. [Google Scholar] [CrossRef] [PubMed]
- Molven, A.; Ringdal, M.; Nordbø, A.M.; Raeder, H.; Støy, J.; Lipkind, G.M.; Steiner, D.F.; Philipson, L.H.; Bergmann, I.; Aarskog, D.; et al. Mutations in the insulin gene can cause MODY and autoantibody-negative type 1 diabetes. Diabetes 2008, 57, 1131–1135. [Google Scholar] [CrossRef]
- Boesgaard, T.W.; Pruhova, S.; Andersson, E.A.; Cinek, O.; Obermannova, B.; Lauenborg, J.; Damm, P.; Bergholdt, R.; Pociot, F.; Pisinger, C.; et al. Further evidence that mutations in INS can be a rare cause of Maturity-Onset Diabetes of the Young (MODY). BMC Med. Genet. 2010, 1211, 42. [Google Scholar] [CrossRef]
- Edghill, E.L.; Flanagan, S.E.; Patch, A.M.; Boustred, C.; Parrish, A.; Shields, B.; Shepherd, M.H.; Hussain, K.; Kapoor, R.R.; Malecki, M.; et al. Insulin mutation screening in 1044 patients with diabetes: Mutations in the INS gene are a common cause of neonatal diabetes but a rare cause of diabetes diagnosed in childhood or adulthood. Diabetes 2008, 57, 1034–1042. [Google Scholar] [CrossRef]
- Borowiec, M.; Liew, C.W.; Thompson, R.; Boonyasrisawat, W.; Hu, J.; Mlynarski, W.M.; El Khattabi, I.; Kim, S.H.; Marselli, L.; Rich, S.S.; et al. Mutations at the BLK locus linked to maturity onset diabetes of the young and beta-cell dysfunction. Proc. Natl. Acad. Sci. USA 2009, 106, 14460–14465. [Google Scholar] [CrossRef] [PubMed]
- Bonnefond, A.; Yengo, L.; Philippe, J.; Dechaume, A.; Ezzidi, I.; Vaillant, E.; Gjesing, A.P.; Andersson, E.A.; Czernichow, S.; Hercberg, S.; et al. Reassessment of the putative role of BLK-p.A71T loss-of-function mutation in MODY and type 2 diabetes. Diabetologia 2013, 56, 492–496. [Google Scholar] [CrossRef]
- Bowman, P.; Flanagan, S.E.; Edghill, E.L.; Damhuis, A.; Shepherd, M.H.; Paisey, R. Heterozygous ABCC8 mutations are a cause of MODY. Diabetologia 2012, 55, 123–127. [Google Scholar] [CrossRef]
- De Franco, E.; Saint-Martin, C.; Brusgaard, K.; Knight Johnson, A.E.; Aguilar-Bryan, L.; Bowman, P.; Arnoux, J.B.; Larsen, A.R.; Sanyoura, M.; Greeley, S.A.W.; et al. Update of variants identified in the pancreatic β-cell KATP channel genes KCNJ11 and ABCC8 in individuals with congenital hyperinsulinism and diabetes. Hum. Mutat. 2020, 41, 884–905. [Google Scholar] [CrossRef]
- Huopio, H.; Otonkoski, T.; Vauhkonen, I.; Reimann, F.; Ashcroft, F.M.; Laakso, M. A new subtype of autosomal dominant diabetes attributable to a mutation in the gene for sulfonylurea receptor 1. Lancet 2003, 361, 301–307. [Google Scholar] [CrossRef]
- Bonnefond, A.; Philippe, J.; Durand, E.; Dechaume, A.; Huyvaert, M.; Montagne, L.; Marre, M.; Balkau, B.; Fajardi, I.; Vambergue, A.; et al. Whole-exome sequencing and high throughput genotyping identified KCNJ11 as the thirteenth MODY gene. PLoS ONE 2012, 7, e37423. [Google Scholar] [CrossRef]
- Yorifuji, T.; Nagashima, K.; Kurokawa, K.; Kawai, M.; Oishi, M.; Hosokawa, M.; Yamada, Y.; Inagaki, N.; Nakahata, T. The C42R mutation in the Kir6.2 (KCNJ11) gene as a cause of transient neonatal diabetes, childhood diabetes, or later-onset, apparently type 2 diabetes mellitus. J. Clin. Endocrinol. Metab. 2005, 90, 3174–3178. [Google Scholar] [CrossRef]
- Ivanoshchuk, D.E.; Shakhtshneider, E.V.; Rymar, O.D.; Ovsyannikova, A.K.; Mikhailova, S.V.; Orlov, P.S.; Yi, R.; Mi, V. Analysis of APPL1 gene polymorphisms in patients with a phenotype of maturity onset diabetes of the young. J. Pers. Med. 2020, 10, E100. [Google Scholar] [CrossRef]
- Prudente, S.; Jungtrakoon, P.; Marucci, A.; Ludovico, O.; Buranasupkajorn, P.; Mazza, T.; Hastings, T.; Milano, T.; Morini, E.; Mercuri, L.; et al. Loss-of-function mutations in APPL1 in familial diabetes mellitus. Am. J. Hum. Genet. 2015, 97, 177–185. [Google Scholar] [CrossRef]
- Deepa, S.S.; Dong, L.Q. APPL1: Role in adiponectin signaling and beyond. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E22–E36. [Google Scholar] [CrossRef]
- Ryu, J.; Galan, A.K.; Xin, X.; Dong, F.; Abdul-Ghani, M.A.; Zhou, L.; Wang, C.; Li, C.; Holmes, B.M.; Sloane, L.B.; et al. APPL1 potentiates insulin sensitivity by facilitating the binding of IRS1/2 to the insulin receptor. Cell Rep. 2014, 7, 1227–1238. [Google Scholar] [CrossRef]
- Cheng, K.K.; Lam, K.S.; Wu, D.; Wang, Y.; Sweeney, G.; Hoo, R.L.; Zhang, J.; Hu, A. APPL1 potentiates insulin secretion in pancreatic β cells by enhancing protein kinase Akt-dependent expression of SNARE proteins in mice. Proc. Natl. Acad. Sci. USA 2012, 109, 8919–8924. [Google Scholar] [CrossRef] [PubMed]
- Schenck, A.; Goto-Silva, L.; Collinet, C.; Rhinn, M.; Giner, A.; Habermann, B.; Brand, M.; Zerial, M. The endosomal protein Appl1 mediates Akt substrate specificity and cell survival in vertebrate development. Cell 2008, 133, 486–497. [Google Scholar] [CrossRef]
- Pearson, E.R.; Boj, S.F.; Steele, A.M.; Barrett, T.; Stals, K.; Shield, J.P.; Ellard, S.; Ferrer, J.; Hattersley, A.T. Macrosomia and hyperinsulinaemic hypoglycaemia in patients with heterozygous mutations in the HNF4A gene. PLoS Med. 2007, 4, e118. [Google Scholar] [CrossRef] [PubMed]
- Gragnoli, C.; Stanojevic, V.; Gorini, A.; Von Preussenthal, G.M.; Thomas, M.K.; Habener, J.F. IPF-1/MODY4 gene missense mutation in an Italian family with type 2 and gestational diabetes. Metabolism 2005, 54, 983–988. [Google Scholar] [CrossRef] [PubMed]
- Kristinsson, S.Y.; Thorolfsdottir, E.T.; Talseth, B.; Steingrimsson, E.; Thorsson, A.V.; Helgason, T.; Hreidarsson, A.B.; Arngrimsson, R. MODY in Iceland is associated with mutations in HNF-1alpha and a novel mutation in NeuroD1. Diabetologia 2001, 44, 2098–2103. [Google Scholar] [CrossRef] [PubMed]
- Ræder, H.; McAllister, F.E.; Tjora, E.; Bhatt, S.; Haldorsen, I.; Hu, J.; Willems, S.M.; Vesterhus, M.; El Ouaamari, A.; Liu, M.; et al. Carboxyl-ester lipase maturity-onset diabetes of the young is associated with development of pancreatic cysts and upregulated MAPK signaling in secretin-stimulated duodenal fluid. Diabetes 2014, 63, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Mauvais-Jarvis, F.; Smith, S.B.; Le May, C.; Leal, S.M.; Gautier, J.F.; Molokhia, M.; Riveline, J.P.; Rajan, A.S.; Kevorkian, J.P.; Zhang, S.; et al. PAX4 gene variations predispose to ketosis-prone diabetes. Hum. Mol. Genet. 2004, 13, 3151–3159. [Google Scholar] [CrossRef]
- Riveline, J.P.; Rousseau, E.; Reznik, Y.; Fetita, S.; Philippe, J.; Dechaume, A.; Hartemann, A.; Polak, M.; Petit, C.; Charpentier, G.; et al. Clinical and metabolic features of adult-onset diabetes caused by ABCC8 mutations. Diabetes Care 2012, 35, 248–251. [Google Scholar] [CrossRef]
- Naylor, R.; Philipson, L.H. Who should have genetic testing for maturity-onset diabetes of the young? Clin. Endocrinol. (Oxf.) 2011, 75, 422–426. [Google Scholar] [CrossRef]
- Chakera, A.J.; Spyer, G.; Vincent, N.; Ellard, S.; Hattersley, A.T.; Dunne, F.P. The 0.1% of the population with glucokinase monogenic diabetes can be recognized by clinical characteristics in pregnancy: The Atlantic Diabetes in Pregnancy cohort. Diabetes Care 2014, 37, 1230–1236. [Google Scholar] [CrossRef]
- Shields, B.M.; McDonald, T.J.; Ellard, S.; Campbell, M.J.; Hyde, C.; Hattersley, A.T. The development and validation of a clinical prediction model to determine the probability of MODY in patients with young-onset diabetes. Diabetologia 2012, 55, 1265–1272. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Ping, F.; Wang, T.; Liu, Y.; Wang, X.; Yu, J.; Deng, M.; Liu, J.; Zhang, Q.; Yu, M.; et al. A Clinical Prediction Model to Distinguish Maturity-Onset Diabetes of the Young from Type 1 and Type 2 Diabetes in the Chinese Population. Endocr. Pr. 2021, 27, 776–782. [Google Scholar] [CrossRef] [PubMed]
- Pinelli, M.; Acquaviva, F.; Barbetti, F.; Caredda, E.; Cocozza, S.; Delvecchio, M.; Mozzillo, E.; Pirozzi, D.; Prisco, F.; Rabbone, I.; et al. Identification of candidate children for maturity-onset diabetes of the young type 2 (MODY2) gene testing: A seven-item clinical flowchart (7-iF). PLoS ONE 2013, 8, e79933. [Google Scholar] [CrossRef]
- Peixoto-Barbosa, R.; Reis, A.F.; Giuffrida, F.M.A. Update on clinical screening of maturity-onset diabetes of the young (MODY). Diabetol. Metab. Syndr. 2020, 12, 50. [Google Scholar] [CrossRef] [PubMed]
- Haliloglu, B.; Hysenaj, G.; Atay, Z.; Guran, T.; Abalı, S.; Turan, S.; Bereket, A.; Ellard, S. GCK gene mutations are a common cause of childhood-onset MODY (maturity-onset diabetes of the young) in Turkey. Clin. Endocrinol. (Oxf.) 2016, 85, 393–399. [Google Scholar] [CrossRef]
- Gloyn, A.L.; van de Bunt, M.; Stratton, I.M.; Lonie, L.; Tucker, L.; Ellard, S.; Holman, R.R. Prevalence of GCK mutations in individuals screened for fasting hyperglycaemia. Diabetologia 2009, 52, 172–174. [Google Scholar] [CrossRef] [PubMed]
- Leslie, R.D.; Atkinson, M.A.; Notkins, A.L. Autoantigens IA-2 and GAD in Type I (insulin-dependent) diabetes. Diabetologia 1999, 42, 3–14. [Google Scholar] [CrossRef][Green Version]
- McDonald, T.J.; Colclough, K.; Brown, R.; Shields, B.; Shepherd, M.; Bingley, P.; Williams, A.; Hattersley, A.T.; Ellard, S. Islet autoantibodies can discriminate maturity-onset diabetes of the young (MODY) from Type 1 diabetes. Diabet. Med. 2011, 28, 1028–1033. [Google Scholar] [CrossRef]
- Carlsson, A.; Shepherd, M.; Ellard, S.; Weedon, M.; Lernmark, Å.; Forsander, G.; Colclough, K.; Brahimi, Q.; Valtonen-Andre, C.; Ivarsson, S.A.; et al. Absence of Islet Autoantibodies and Modestly Raised Glucose Values at Diabetes Diagnosis Should Lead to Testing for MODY: Lessons From a 5-Year Pediatric Swedish National Cohort Study. Diabetes Care 2020, 43, 82–89. [Google Scholar] [CrossRef]
- Urbanová, J.; Rypáčková, B.; Kučera, P.; Anděl, M.; Heneberg, P. Should the negativity for islet cell autoantibodies be used in a prescreening for genetic testing in maturity-onset diabetes of the young? The case of autoimmunity-associated destruction of pancreatic β-cells in a family of HNF1A-MODY subjects. Int. Arch. Allergy Immunol. 2013, 161, 279–284. [Google Scholar] [CrossRef]
- Bowden, S.A.; Hoffman, R.P. Triple diabetes: Coexistence of type 1 diabetes mellitus and a novel mutation in the gene responsible for MODY3 in an overweight adolescent. Pediatr. Diabetes 2008, 9, 162–164. [Google Scholar] [CrossRef] [PubMed]
- Urbanová, J.; Rypáčková, B.; Procházková, Z.; Kučera, P.; Cerná, M.; Anděl, M.; Heneberg, P. Positivity for islet cell autoantibodies in patients with monogenic diabetes is associated with later diabetes onset and higher HbA1c level. Diabet. Med. 2014, 31, 466–471. [Google Scholar] [CrossRef]
- Lindgren, C.M.; Widén, E.; Tuomi, T.; Li, H.; Almgren, P.; Kanninen, T.; Melander, O.; Weng, J.; Lehto, M.; Groop, L.C. Contribution of known and unknown susceptibility genes to early-onset diabetes in scandinavia: Evidence for heterogeneity. Diabetes 2002, 51, 1609–1617. [Google Scholar] [CrossRef][Green Version]
- American Diabetes Association. 13. Children and adolescents: Standards of Medical Care in Diabetes 2021. Diabetes Care 2021, 44, S180–S199. [Google Scholar] [CrossRef] [PubMed]
- DiMeglio, L.A.; Acerini, C.L.; Codner, E.; Craig, M.E.; Hofer, S.E.; Pillay, K.; Maahs, D.M. ISPAD Clinical Practice Consensus Guidelines 2018: Glycemic control targets and glucose monitoring for children, adolescents, and young adults with diabetes. Pediatric. Diabetes 2018, 19, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Meyerovitch, J.; Zlotnik, M.; Yackobovitch-Gavan, M.; Phillip, M.; Shalitin, S. Real-Life Glycemic Control in Children with Type 2 Diabetes: A Population-Based Study. J. Pediatr. 2017, 188, 173–180. [Google Scholar] [CrossRef] [PubMed]
- TODAY Study Group; Zeitler, P.; Hirst, K.; Pyle, L.; Linder, B.; Copeland, K.; Arslanian, S.; Cuttler, L.; Nathan, D.M.; Tollefsen, S.; et al. A clinical trial to maintain glycemic control in youth with type 2 diabetes. N. Engl. J. Med. 2012, 366, 2247–2256. [Google Scholar] [CrossRef]
- Steele, A.M.; Shields, B.M.; Wensley, K.J.; Colclough, K.; Ellard, S.; Hattersley, A.T. Prevalence of vascular complications among patients with glucokinase mutations and prolonged, mild hyperglycemia. JAMA 2014, 311, 279–286. [Google Scholar] [CrossRef]
- Dickens, L.T.; Letourneau, L.R.; Sanyoura, M.; Greeley, S.A.W.; Philipson, L.H.; Naylor, R.N. Management and pregnancy outcomes of women with GCK-MODY enrolled in the US Monogenic Diabetes Registry. Acta Diabetol. 2019, 56, 405–411. [Google Scholar] [CrossRef]
- Broome, D.T.; Pantalone, K.M.; Kashyap, S.R.; Philipson, L.H. Approach to the Patient with MODY-Monogenic Diabetes. J. Clin. Endocrinol. Metab. 2021, 106, 237–250. [Google Scholar] [CrossRef]
- Delvecchio, M.; Mozzillo, E.; Salzano, G.; Iafusco, D.; Frontino, G.; Patera, P.I.; Rabbone, I.; Cherubini, V.; Grasso, V.; Tinto, N.; et al. Monogenic diabetes accounts for 6.3% of cases referred to 15 Italian pediatric diabetes centers during 2007 to 2012. J. Clin. Endocrinol. Metab. 2017, 102, 1826–1834. [Google Scholar] [CrossRef]
- Hattersley, A.T.; Greeley, S.A.W.; Polak, M.; Rubio-Cabezas, O.; Njolstad, P.R.; Mlynarski, W.; Castano, L.; Carlsson, A.; Raile, K.; Chi, D.V.; et al. ISPAD Clinical Practice Consensus Guidelines 2018: The diagnosis and management of monogenic diabetes in children and adolescents. Pediatr. Diabetes 2018, 19, 47–63. [Google Scholar] [CrossRef]
- Pearson, E.; Liddell, W.; Shepherd, M.; Corrall, R.; Hattersley, A. Sensitivity to sulphonylureas in patients with HNF-1a gene mutations: Evidence for pharmacogenetics in diabetes. Diabet. Med. 2000, 17, 543–545. [Google Scholar] [CrossRef]
- Broome, D.T.; Tekin, Z.; Pantalone, K.M.; Mehta, A.E. Novel use of GLP-1 receptor agonist therapy in HNF4A-MODY. Diabet. Care 2020, 43, e65. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, M.; Shields, B.; Ellard, S.; Rubio-Cabezas, O.; Hattersley, A.T. A genetic diagnosis of HNF1A diabetes alters treatment and improves glycaemic control in the majority of insulin-treated patients. Diabet. Med. 2009, 26, 437–441. [Google Scholar] [CrossRef]
- Carrillo, E.; Lomas, A.; Pines, P.J.; Lamas, C. Long-lasting response to oral therapy in a young male with monogenic diabetes as part of HNF1B-related disease. Endocrinol. Diabetes Metab. Case Rep. 2017, 17, 0052. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Delvecchio, M.; Pastore, C.; Giordano, P. Treatment Options for MODY Patients: A Systematic Review of Literature. Diabetes 2020, 11, 1667–1685. [Google Scholar] [CrossRef] [PubMed]
- Fajans, S.S.; Bell, G.I. MODY: History, genetics, pathophysiology, and clinical decision making. Diabetes Care 2011, 34, 1878–1884. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).