Paradoxical Relationships between Serum 25(OH)D and Ferritin with Body Composition and Burnout: Variation by Sex and Sports Team
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schork, N.J. Personalized medicine: Time for one-person trials. Nature 2015, 520, 609–611. [Google Scholar] [CrossRef]
- Dahlquist, D.T.; Dieter, B.P.; Koehle, M.S. Plausible ergogenic effects of vitamin D on athletic performance and recovery. J. Int. Soc. Sports Nutr. 2015, 12, 33. [Google Scholar] [CrossRef]
- Pasricha, S.R.; Low, M.; Thompson, J.; Farrell, A.; De-Regil, L.M. Iron supplementation benefits physical performance in women of reproductive age: A systematic review and meta-analysis. J. Nutr. 2014, 144, 906–914. [Google Scholar] [CrossRef] [PubMed]
- Lansdowne, A.T.; Provost, S.C. Vitamin D3 enhances mood in healthy subjects during winter. Psychopharmacology 1998, 135, 319–323. [Google Scholar] [CrossRef] [PubMed]
- Ljungqvist, A.; Jenoure, P.J.; Engebretsen, L.; Alonso, J.M.; Bahr, R.; Clough, A.F.; de Bondt, G.; Dvorak, J.; Maloley, R.; Matheson, G.; et al. The International Olympic Committee (IOC) consensus statement on periodic health evaluation of elite athletes, March 2009. Clin. J. Sport Med. 2009, 19, 347–365. [Google Scholar] [CrossRef]
- Rockwell, M.; Hulver, M.; Eugene, E. Vitamin D Practice Patterns in National Collegiate Athletic Association Division I Collegiate Athletics Programs. J. Athl. Train. 2020, 55, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Parks, R.B.; Hetzel, S.J.; Brooks, M.A. Iron Deficiency and Anemia among Collegiate Athletes: A Retrospective Chart Review. Med. Sci. Sports Exerc. 2017, 49, 1711–1715. [Google Scholar] [CrossRef]
- Hackney, A.C.; Elliott-Sale, K.J. Exercise Endocrinology: “What Comes Next?”. Endocrines 2021, 2, 17. [Google Scholar] [CrossRef]
- Grieshober, J.A.; Mehran, N.; Photopolous, C.; Fishman, M.; Lombardo, S.J.; Kharrazi, F.D. Vitamin D Insufficiency Among Professional Basketball Players: A Relationship to Fracture Risk and Athletic Performance. Orthop. J. Sports Med. 2018, 6, 2325967118774329. [Google Scholar] [CrossRef]
- Villacis, D.; Yi, A.; Jahn, R.; Kephart, C.J.; Charlton, T.; Gamradt, S.C.; Romano, R.; Tibone, J.E.; Hatch, G.F., 3rd. Prevalence of Abnormal Vitamin D Levels Among Division I NCAA Athletes. Sports Health 2014, 6, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Fields, J.B.; Payne, D.C.; Gallo, S.; Busteed, D.R.; Jones, M.T. Vitamin D Status Differs by Sex, Sport-Season, and Skin Pigmentation among Elite Collegiate Basketball Players. Sports 2019, 7, 239. [Google Scholar] [CrossRef] [PubMed]
- Fields, J.B.; Gallo, S.; Worswick, J.M.; Busteed, D.R.; Jones, M.T. 25-Hydroxyvitamin D, Vitamin D Binding Protein, Bioavailable 25-Hydroxyvitamin D, and Body Composition in a Diverse Sample of Women Collegiate Indoor Athletes. J. Funct. Morphol. Kinesiol. 2020, 5, 32. [Google Scholar] [CrossRef] [PubMed]
- Weber, A.E.; Bolia, I.K.; Korber, S.; Mayfield, C.K.; Lindsay, A.; Rosen, J.; McMannes, S.; Romano, R.; Tibone, J.E.; Gamradt, S.C. Five-Year Surveillance of Vitamin D Levels in NCAA Division I Football Players: Risk Factors for Failed Supplementation. Orthop J. Sports Med. 2021, 9, 2325967120975100. [Google Scholar] [CrossRef] [PubMed]
- Halliday, T.M.; Peterson, N.J.; Thomas, J.J.; Kleppinger, K.; Hollis, B.W.; Larson-Meyer, D.E. Vitamin D status relative to diet, lifestyle, injury, and illness in college athletes. Med. Sci. Sports Exerc. 2011, 43, 335–343. [Google Scholar] [CrossRef]
- Cowell, B.S.; Rosenbloom, C.A.; Skinner, R.; Summers, S.H. Policies on screening female athletes for iron deficiency in NCAA division I-A institutions. Int. J. Sport Nutr. Exerc. Metab. 2003, 13, 277–285. [Google Scholar] [CrossRef]
- Cook, J.D.; Skikne, B.S. Iron deficiency: Definition and diagnosis. J. Intern. Med. 1989, 226, 349–355. [Google Scholar] [CrossRef]
- Eichner, E.R. Perennial questions: On fatigue, on iron, and on anemia. Curr Sports Med. Rep. 2012, 11, 274–275. [Google Scholar] [CrossRef]
- Pilch, W.; Kita, B.; Piotrowska, A.; Tota, Ł.; Maciejczyk, M.; Czerwińska-Ledwig, O.; Sadowska-Krepa, E.; Kita, S.; Pałka, T. The effect of vitamin D supplementation on the muscle damage after eccentric exercise in young men: A randomized, control trial. J. Int. Soc. Sports Nutr. 2020, 17, 53. [Google Scholar] [CrossRef]
- Miller, J.R.; Dunn, K.W.; Ciliberti, L.J., Jr.; Patel, R.D.; Swanson, B.A. Association of Vitamin D With Stress Fractures: A Retrospective Cohort Study. J. Foot Ankle Surg. 2016, 55, 117–120. [Google Scholar] [CrossRef]
- Risser, W.L.; Lee, E.J.; Poindexter, H.B.; West, M.S.; Pivarnik, J.M.; Risser, J.M.; Hickson, J.F. Iron deficiency in female athletes: Its prevalence and impact on performance. Med. Sci. Sports Exerc. 1988, 20, 116–121. [Google Scholar] [CrossRef]
- Barakat, C.; Pearson, J.; Escalante, G.; Campbell, B.; De Souza, E.O. Body Recomposition: Can Trained Individuals Build Muscle and Lose Fat at the Same Time? Strength Cond. J. 2020, 42, 7–21. [Google Scholar] [CrossRef]
- Bennell, K.L.; Malcolm, S.A.; Khan, K.M.; Thomas, S.A.; Reid, S.J.; Brukner, P.D.; Ebeling, P.R.; Wark, J.D. Bone mass and bone turnover in power athletes, endurance athletes, and controls: A 12-month longitudinal study. Bone 1997, 20, 477–484. [Google Scholar] [CrossRef]
- Stanfa, M.R.; Silles, N.N.; Cooper, A.; Arena, S.; Landis-Piwowar, K.; Aprik, C.; Hew-Butler, T. Risk Factors for Collegiate Swimmers Hospitalized With Exertional Rhabdomyolysis. Clin. J. Sport Med. 2017, 27, 37–45. [Google Scholar] [CrossRef]
- Mountjoy, M.; Sundgot-Borgen, J.; Burke, L.; Carter, S.; Constantini, N.; Lebrun, C.; Meyer, N.; Sherman, R.; Steffen, K.; Budgett, R.; et al. The IOC consensus statement: Beyond the Female Athlete Triad--Relative Energy Deficiency in Sport (RED-S). Br. J. Sports Med. 2014, 48, 491–497. [Google Scholar] [CrossRef]
- Tenforde, A.S.; Carlson, J.L.; Chang, A.; Sainani, K.L.; Shultz, R.; Kim, J.H.; Cutti, P.; Golden, N.H.; Fredericson, M. Association of the Female Athlete Triad Risk Assessment Stratification to the Development of Bone Stress Injuries in Collegiate Athletes. Am. J. Sports Med. 2017, 45, 302–310. [Google Scholar] [CrossRef]
- Scheid, J.L.; Stefanik, M.E. Drive for Thinness Predicts Musculoskeletal Injuries in Division II NCAA Female Athletes. J. Funct. Morphol. Kinesiol. 2019, 4, 52. [Google Scholar] [CrossRef] [PubMed]
- Polak, M.A.; Houghton, L.A.; Reeder, A.I.; Harper, M.J.; Conner, T.S. Serum 25-hydroxyvitamin D concentrations and depressive symptoms among young adult men and women. Nutrients 2014, 6, 4720–4730. [Google Scholar] [CrossRef] [PubMed]
- Vahdat Shariatpanaahi, M.; Vahdat Shariatpanaahi, Z.; Moshtaaghi, M.; Shahbaazi, S.H.; Abadi, A. The relationship between depression and serum ferritin level. Eur. J. Clin. Nutr. 2007, 61, 532–535. [Google Scholar] [CrossRef]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Nielsen, P.; Nachtigall, D. Iron supplementation in athletes. Current recommendations. Sports Med. 1998, 26, 207–216. [Google Scholar] [CrossRef]
- Hologic. QDR Reference Manual, 2nd ed.; MAN-00732; Hologic, Inc.: Bedford, MA, USA, 2007. [Google Scholar]
- Raedeke, T.D.; Smith, A.L. Development and Preliminary Validation of an Athlete Burnout Measure. J. Sport Exerc. Psychol. 2001, 23, 281–306. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, A.; Zahediasl, S. Normality tests for statistical analysis: A guide for non-statisticians. Int. J. Endocrinol. Metab. 2012, 10, 486–489. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, G.M.; Feinn, R. Using Effect Size-or Why the P Value Is Not Enough. J. Grad. Med. Educ. 2012, 4, 279–282. [Google Scholar] [CrossRef]
- Lerchbaum, E.; Pilz, S.; Trummer, C.; Schwetz, V.; Pachernegg, O.; Heijboer, A.C.; Obermayer-Pietsch, B. Vitamin D and Testosterone in Healthy Men: A Randomized Controlled Trial. J. Clin. Endocrinol. Metab. 2017, 102, 4292–4302. [Google Scholar] [CrossRef]
- Krzywański, J.; Pokrywka, A.; Młyńczak, M.; Mikulski, T. Is vitamin D status reflected by testosterone concentration in elite athletes? Biol. Sport 2020, 37, 229–237. [Google Scholar] [CrossRef]
- Fraser, I.S.; Warner, P.; Marantos, P.A. Estimating menstrual blood loss in women with normal and excessive menstrual fluid volume. Obstet. Gynecol. 2001, 98, 806–814. [Google Scholar] [CrossRef]
- Wilson-Barnes, S.L.; Hunt, J.E.A.; Williams, E.L.; Allison, S.J.; Wild, J.J.; Wainwright, J.; Lanham-New, S.A.; Manders, R.J.F. Seasonal variation in vitamin D status, bone health and athletic performance in competitive university student athletes: A longitudinal study. J. Nutr. Sci. 2020, 9, e8. [Google Scholar] [CrossRef]
- Constantini, N.W.; Arieli, R.; Chodick, G.; Dubnov-Raz, G. High prevalence of vitamin D insufficiency in athletes and dancers. Clin. J. Sport Med. 2010, 20, 368–371. [Google Scholar] [CrossRef] [PubMed]
- Powe, C.E.; Evans, M.K.; Wenger, J.; Zonderman, A.B.; Berg, A.H.; Nalls, M.; Tamez, H.; Zhang, D.; Bhan, I.; Karumanchi, S.A.; et al. Vitamin D-binding protein and vitamin D status of black Americans and white Americans. N. Engl. J. Med. 2013, 369, 1991–2000. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.M.; Redzic, M.; Thomas, D.T. The effects of season-long vitamin D supplementation on collegiate swimmers and divers. Int J. Sport Nutr. Exerc. Metab. 2013, 23, 431–440. [Google Scholar] [CrossRef]
- Webb, A.R.; Kline, L.; Holick, M.F. Influence of season and latitude on the cutaneous synthesis of vitamin D3: Exposure to winter sunlight in Boston and Edmonton will not promote vitamin D3 synthesis in human skin. J. Clin. Endocrinol. Metab. 1988, 67, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Klesges, R.C.; Ward, K.D.; Shelton, M.L.; Applegate, W.B.; Cantler, E.D.; Palmieri, G.M.; Harmon, K.; Davis, J. Changes in bone mineral content in male athletes. Mechanisms of action and intervention effects. JAMA 1996, 276, 226–230. [Google Scholar] [CrossRef]
- Heller, J.E.; Thomas, J.J.; Hollis, B.W.; Larson-Meyer, D.E. Relation between vitamin D status and body composition in collegiate athletes. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 128–135. [Google Scholar] [CrossRef]
- LeFevre, M.L.; LeFevre, N.M. Vitamin D Screening and Supplementation in Community-Dwelling Adults: Common Questions and Answers. Am. Fam. Physician 2018, 97, 254–260. [Google Scholar]
- Jin, J. Screening for Vitamin D Deficiency in Adults. JAMA 2021, 325, 1480. [Google Scholar] [CrossRef] [PubMed]
- Camaschella, C. Iron deficiency. Blood 2019, 133, 30–39. [Google Scholar] [CrossRef]
- Kell, D.B.; Pretorius, E. Serum ferritin is an important inflammatory disease marker, as it is mainly a leakage product from damaged cells. Metallomics 2014, 6, 748–773. [Google Scholar] [CrossRef] [PubMed]
- Comassi, M.; Vitolo, E.; Pratali, L.; Del Turco, S.; Dellanoce, C.; Rossi, C.; Santini, E.; Solini, A. Acute effects of different degrees of ultra-endurance exercise on systemic inflammatory responses. Intern. Med. J. 2015, 45, 74–79. [Google Scholar] [CrossRef]
- Deruisseau, K.C.; Roberts, L.M.; Kushnick, M.R.; Evans, A.M.; Austin, K.; Haymes, E.M. Iron status of young males and females performing weight-training exercise. Med. Sci. Sports Exerc. 2004, 36, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Zürcher, S.J.; Quadri, A.; Huber, A.; Thomas, L.; Close, G.L.; Brunner, S.; Noack, P.; Gojanovic, B.; Kriemler, S. Predictive Factors for Vitamin D Concentrations in Swiss Athletes: A Cross-sectional Study. Sports Med. Int. Open 2018, 2, E148–E156. [Google Scholar] [CrossRef][Green Version]

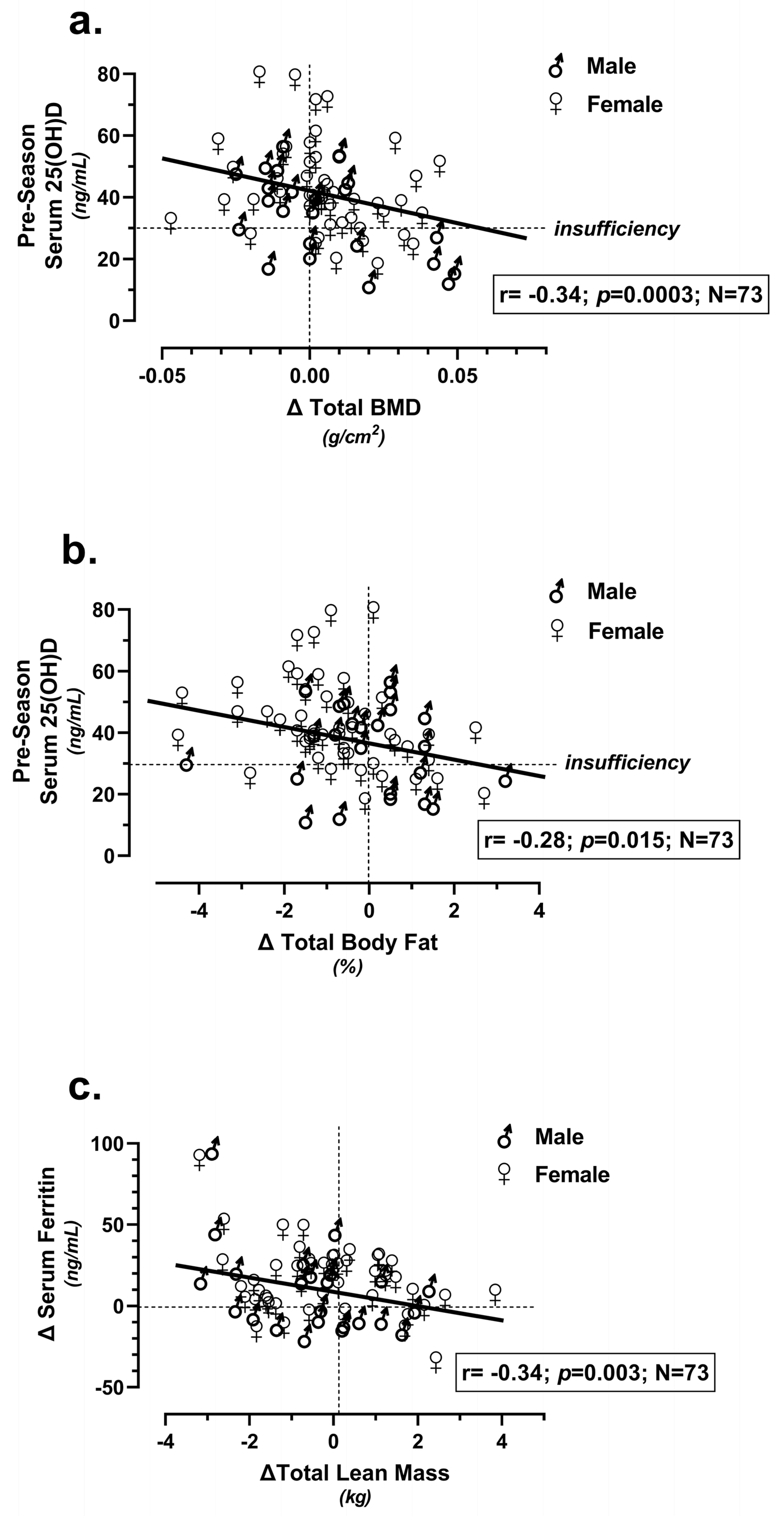
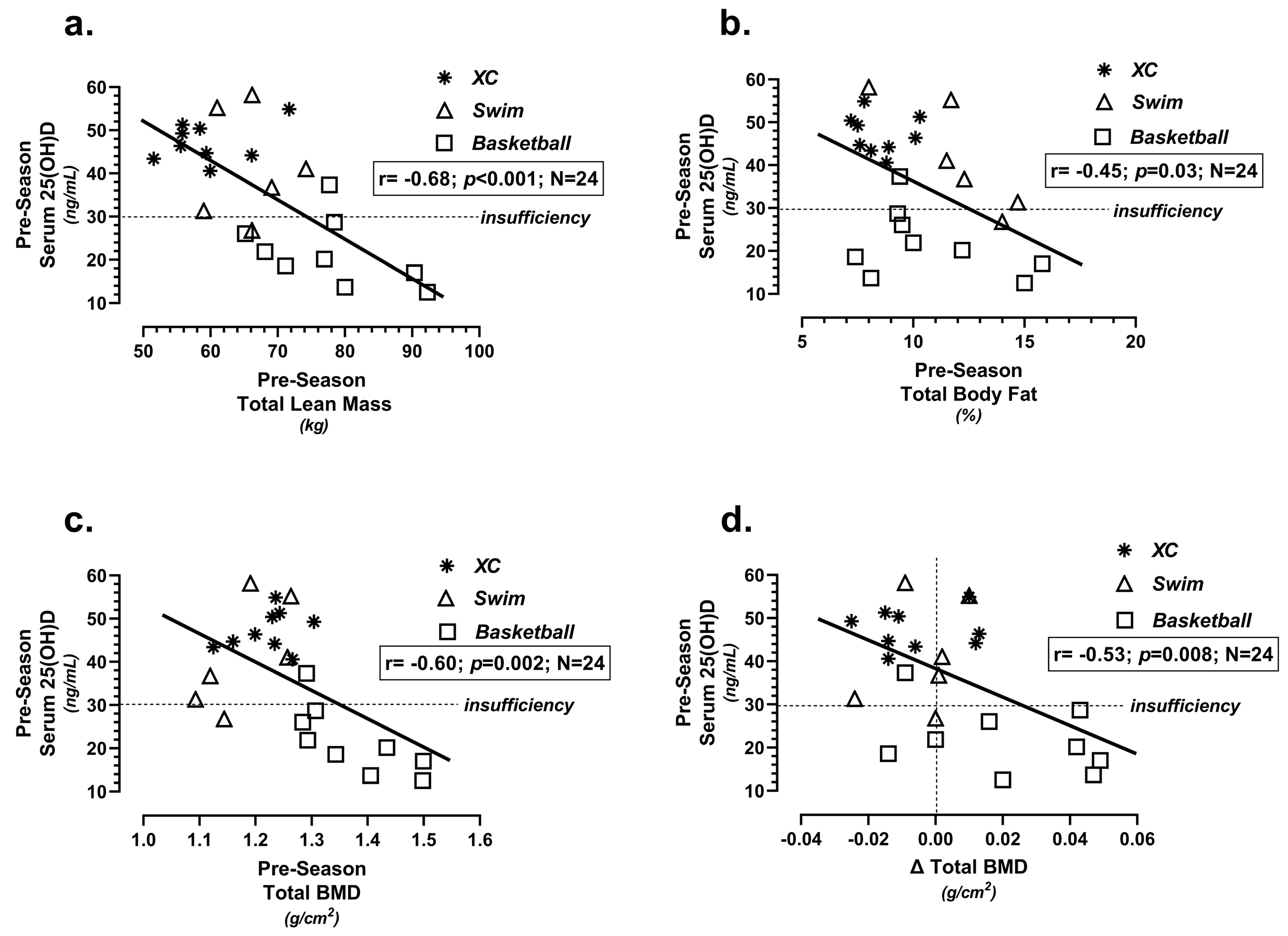

| Team (N = 73) | Age (years) | Height (m) | Weight (kg) | BMI (kg/m2) |
|---|---|---|---|---|
| ♀XC (n = 16) | 19.4 ± 1.5 | 1.64 ± 0.1 | 56.6 ± 4.9 | 20.8 ± 1.8 |
| ♂XC (n = 9) | 20.3 ± 0.9 | 1.80 ± 0.1 | 68.8 ± 6.9 | 21.3 ± 0.9 |
| ♀Swim (n-12) | 19.6 ± 1.1 | 1.70 ± 0.1 | 67.2 ± 5.8 | 23.2 ± 2.1 |
| ♂Swim (n = 6) | 20.7 ± 1.2 | 1.84 ± 0.1 | 78.5 ± 5.2 | 23.3 ± 1.6 |
| ♀Basketball (n = 10) | 19.7 ± 1.4 | 1.76 ± 0.1 | 75.1 ± 12.0 | 24.3 ± 3.4 |
| ♂Basketball (n = 9) | 20.1 ± 1.5 | 1.96 ± 0.1 | 94.5 ± 14.3 | 24.6 ± 2.8 |
| ♀Soccer (n = 11) | 19.5 ± 1.2 | 1.69 ± 0.1 | 64.9 ± 14.4 | 22.6 ± 3.6 |
| ♀COMBINED (n = 49) | 19.5 ± 1.3 | 1.69 ± 0.1 | 64.8 ± 11.5 | 22.6 ± 2.0 |
| ♂COMBINED (n = 24) | 20.3 ± 0.1 | 1.87 ± 0.1 | 80.9 ± 15.0 | 23.0 ± 2.4 |
| Team (N = 73) | Pre Body Fat (%) | ∆ Body Fat (%) | Pre BMD (g/cm2) | ∆ BMD (g/cm2) | Pre Lean Mass (kg) | ∆ Lean Mass (kg) | T or Z-Score |
|---|---|---|---|---|---|---|---|
| ♀XC (n = 16) | 18.7 ± 3.1 | −0.8 ± 1.1 | 1.08 ± 0.06 | −0.00 ± 0.02 | 43.7 ± 4.3 | −0.4 ± 0.9 | −0.34 ± 0.82 |
| ♂XC (n = 9) | 8.5 ± 1.1 | −0.1 ± 0.8 | 1.22 ± 0.05 | −0.01 ± 0.01 | 59.4 ± 6.1 | −0.3 ± 0.9 | −0.19 ± 0.46 |
| ♀Swim (n-12) | 21.2 ± 3.8 | −1.7 ± 1.8 | 1.08 ± 0.07 | 0.00 ± 0.02 | 49.0 ± 3.0 | 1.8 ± 1.3 | −0.22 ± 0.76 |
| ♂Swim (n = 6) | 12.0 ± 2.4 | −1.3 ± 1.7 | 1.17 ± 0.07 | 0.00 ± 0.01 | 66.0 ± 5.5 | 0.5 ± 2.1 | −0.57 ± 0.75 |
| ♀Basketball (n = 10) | 18.2 ± 5.1 | −0.7 ± 1.1 | 1.19 ± 0.09 | 0.01 ± 0.02 | 56.1 ± 5.9 | −0.6 ± 1.9 | 1.10 ± 0.55 |
| ♂Basketball (n = 9) | 10.7 ± 3.0 | 0.8 ± 1.4 | 1.37 ± 0.09 | 0.02 ± 0.02 | 77.8 ± 9.1 | −1.3 ± 1.4 | 1.14 ± 0.79 |
| ♀Soccer (n = 11) | 19.9 ± 5.0 | 0.2 ± 1.5 | 1.09 ± 0.07 | 0.01 ± 0.01 | 48.7 ± 6.5 | −0.8 ± 1.5 | 0.76 ± 0.43 |
| ♀COMBINED (n = 49) | 19.5 ± 4.2 | −0.8 ± 1.5 | 1.10 ± 0.08 | 0.00 ± 0.02 | 48.7 ± 6.6 | −0.2 ± 1.6 | 0.18 ± 0.90 |
| ♂COMBINED (n = 24) | 10.2 ± 2.6 | −0.1 ± 1.5 | 1.27 ± 0.11 | 0.01 ± 0.02 | 67.9 ± 10.8 | 0.3 ± 3.8 | 0.17 ± 0.97 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hew-Butler, T.; Aprik, C.; Byrd, B.; Landis-Piwowar, K.; Smith-Hale, V.; VanSumeren, M.; Sabourin, J.; Byrd, G.; Martin, J. Paradoxical Relationships between Serum 25(OH)D and Ferritin with Body Composition and Burnout: Variation by Sex and Sports Team. Endocrines 2021, 2, 320-333. https://doi.org/10.3390/endocrines2030030
Hew-Butler T, Aprik C, Byrd B, Landis-Piwowar K, Smith-Hale V, VanSumeren M, Sabourin J, Byrd G, Martin J. Paradoxical Relationships between Serum 25(OH)D and Ferritin with Body Composition and Burnout: Variation by Sex and Sports Team. Endocrines. 2021; 2(3):320-333. https://doi.org/10.3390/endocrines2030030
Chicago/Turabian StyleHew-Butler, Tamara, Carrie Aprik, Brigid Byrd, Kristin Landis-Piwowar, Valerie Smith-Hale, Matthew VanSumeren, Jordan Sabourin, Gregory Byrd, and Jeffrey Martin. 2021. "Paradoxical Relationships between Serum 25(OH)D and Ferritin with Body Composition and Burnout: Variation by Sex and Sports Team" Endocrines 2, no. 3: 320-333. https://doi.org/10.3390/endocrines2030030
APA StyleHew-Butler, T., Aprik, C., Byrd, B., Landis-Piwowar, K., Smith-Hale, V., VanSumeren, M., Sabourin, J., Byrd, G., & Martin, J. (2021). Paradoxical Relationships between Serum 25(OH)D and Ferritin with Body Composition and Burnout: Variation by Sex and Sports Team. Endocrines, 2(3), 320-333. https://doi.org/10.3390/endocrines2030030








