Involvement of the MEN1 Gene in Hormone-Related Cancers: Clues from Molecular Studies, Mouse Models, and Patient Investigations
Abstract
1. Introduction
2. Background about Breast and Prostate Cancers
2.1. Histopathology and Classification
2.1.1. Breast Cancer
2.1.2. Prostate Cancer
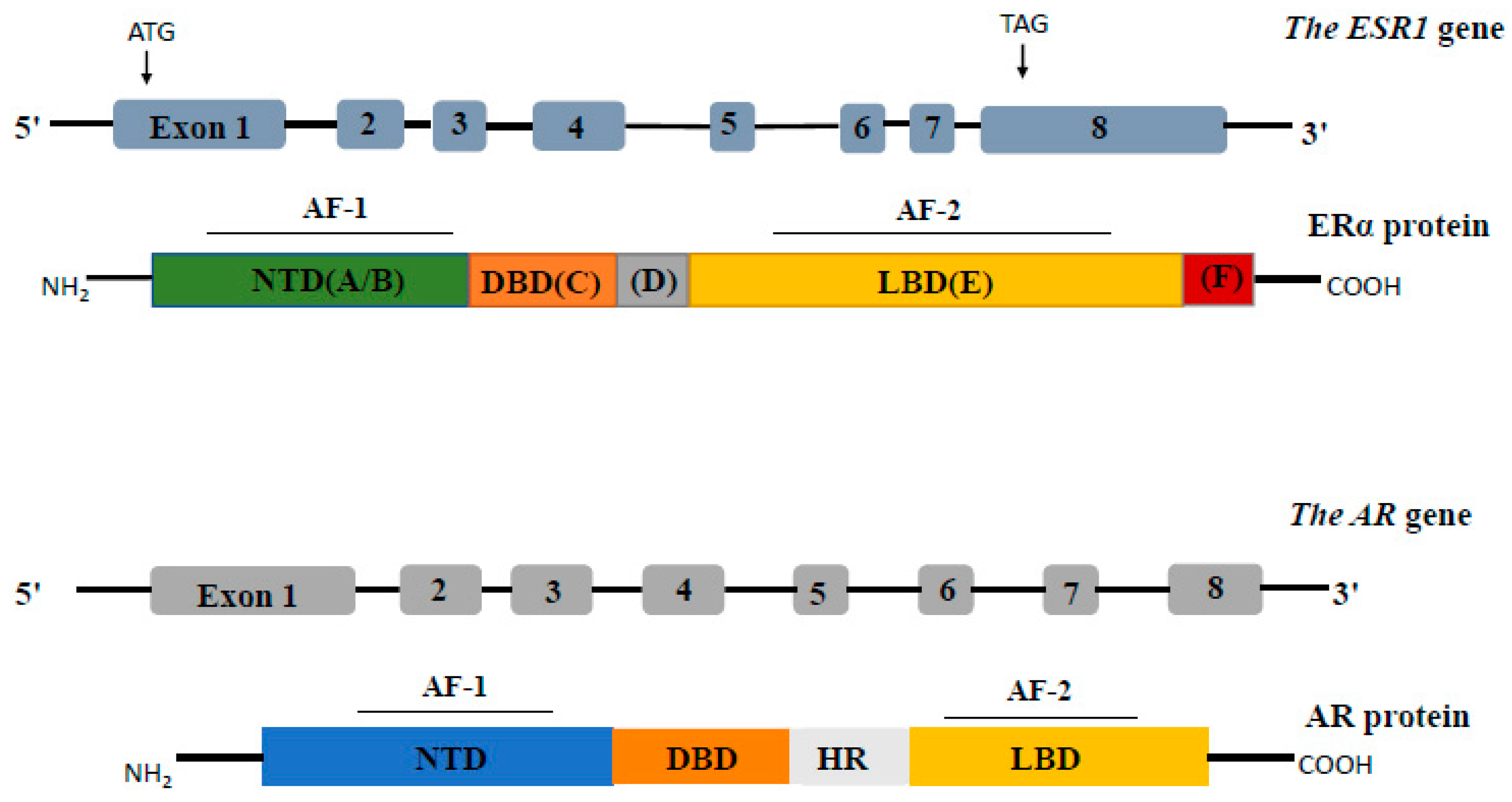
2.2. Estrogen Receptor-Alpha and Androgen Receptor
2.2.1. Estrogen Receptor-Alpha (ERα)
Structure
ESR1 Gene Regulation
Gene Targets and Gene Functions
2.2.2. Androgen Receptor (AR)
Structure
AR Gene Regulation
Gene Targets and Functions
3. The Involvement of the MEN1 Gene in Breast and Prostate Cancers
3.1. Molecular Studies
3.2. Mouse Models
3.2.1. Mammary Gland Lesions in Mouse Men1 Models
3.2.2. Prostate Lesions in Mouse Men1 Models
3.3. Human Studies
3.3.1. MEN1 in Human Breast Cancer
3.3.2. MEN1 in Human Prostate Cancer
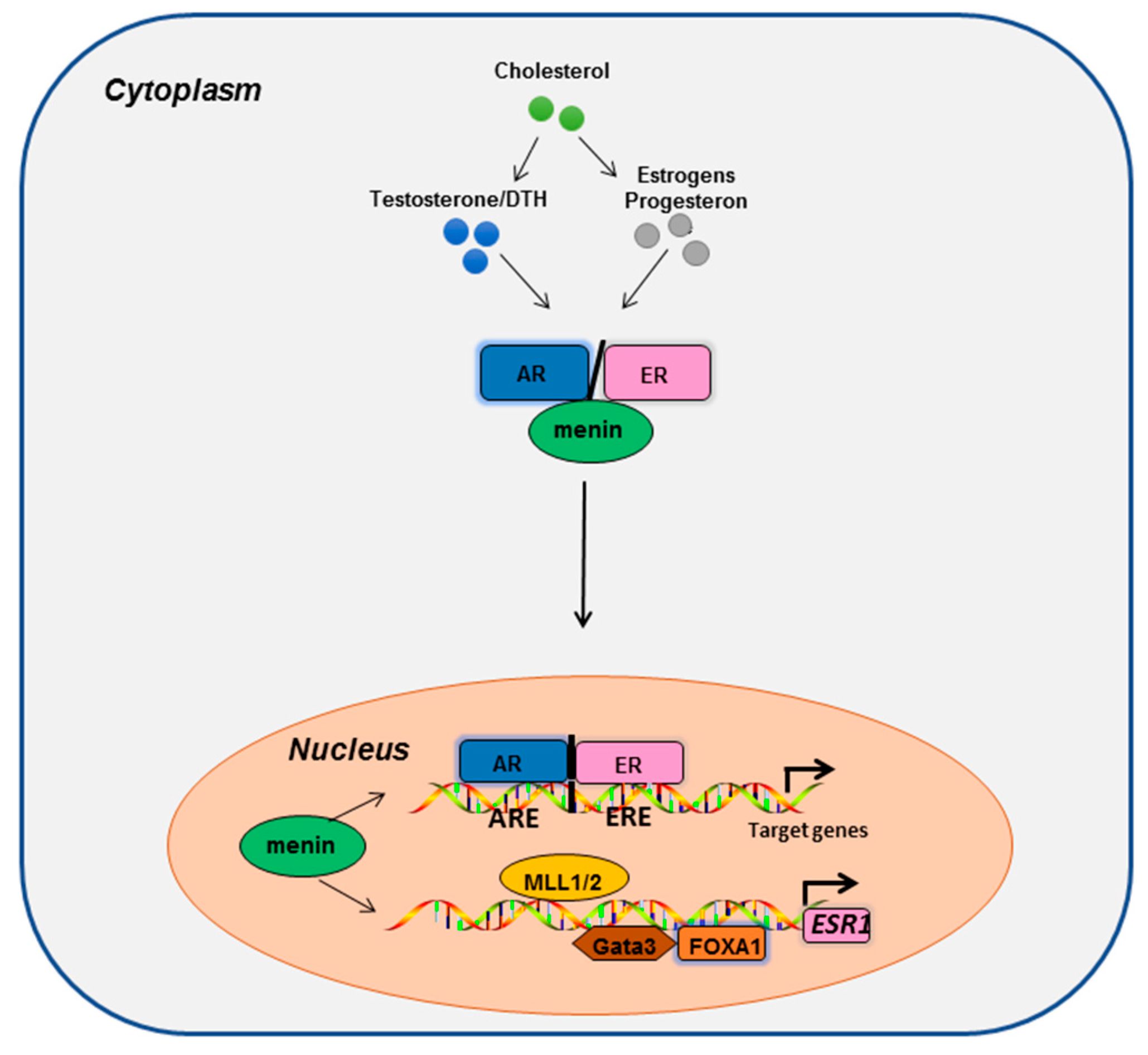
4. Further Clues for the Role of Menin in Breast and Prostate Cancers
4.1. Epigenetic Factors
4.2. Transcription Factors
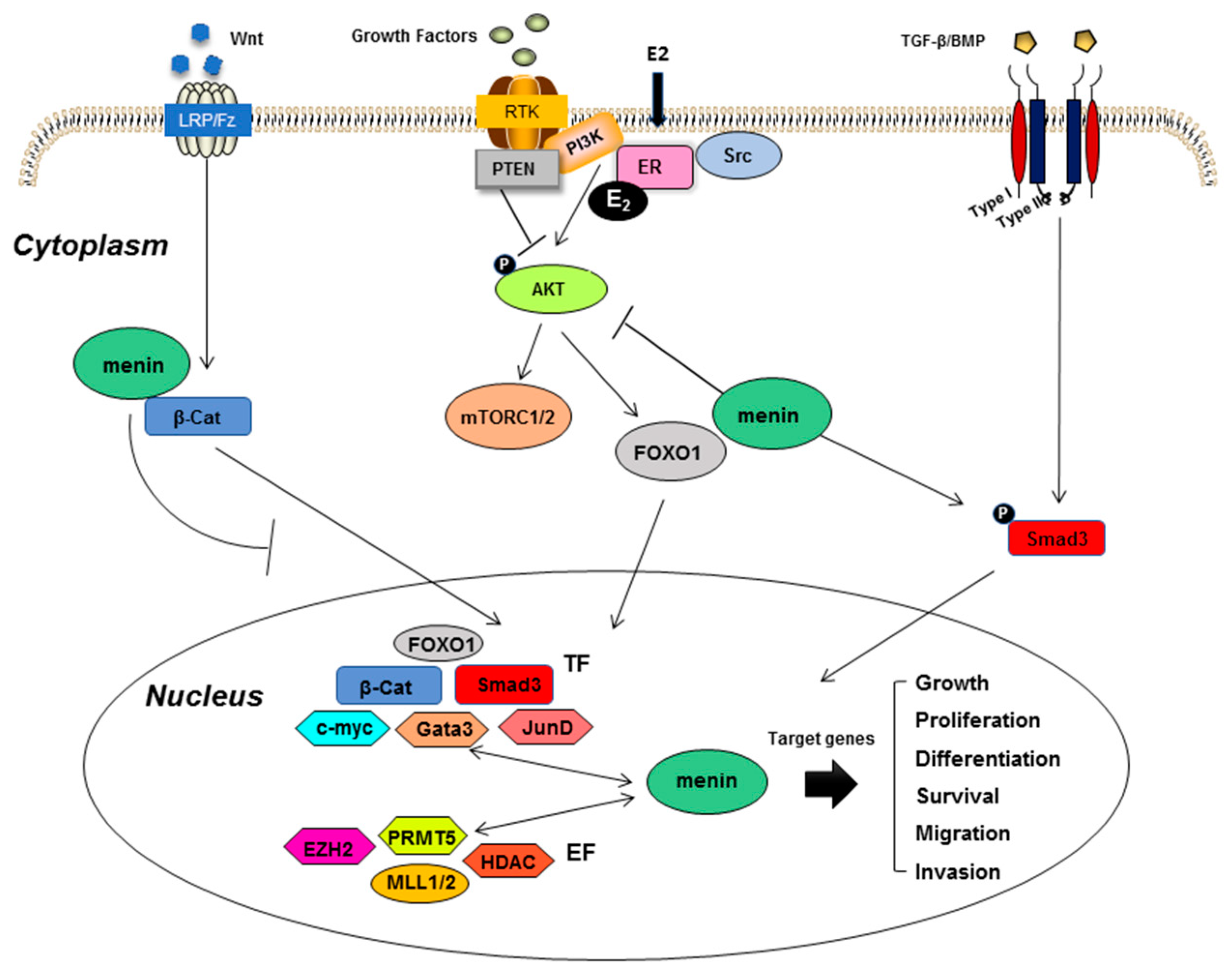
4.3. Signal Transduction Pathways
4.3.1. The PI3K/PTEN/AKT/mTOR Pathways
4.3.2. Cell Cycle, Growth, and Death Control
4.3.3. Wnt Signaling
5. Finishing Words
5.1. The Dual Role of Menin
5.2. Remaining Questions
6. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ADT | Androgen deprivation therapies |
| AP-1 | Activator protein 1 |
| AR | Androgen receptor |
| AREs | Androgen response elements |
| ASK | Activator of S-phase kinase |
| BAX | BCL2 Associated X |
| BC | Breast cancer |
| BRCA1&2 | Breast cancers 1 and 2 |
| CDK | Cyclin-dependent kinase |
| CRPC | Castration-resistant prostate cancer |
| DBD | DNA-binding domain |
| DHT | Dihydrotestosterone |
| DNMTs | DNA methyltransferases |
| EIT-6 | Estrogen Induced Tag-6 |
| ER | Estrogen receptor |
| ERE | Estrogen response element |
| EZH2 | Enhancer of zeste homolog 2 |
| FOX | Forkhead box |
| H3K4me3 | Tri-methylation at the 4th lysine residue of the histone H3 protein |
| HCC | Hepatocellular carcinoma |
| HDAC | Histone deacetylase |
| HER2 | Human epidermal growth factor receptor 2 |
| HOX | Homeobox |
| Hsp | Heat-shock proteins |
| IGF-1 | insulin-like growth factor-1 |
| IGFBP-3 | Insulin-like growth factor-binding protein 3 |
| LAR | Luminal-androgen receptor |
| LBD | Ligand-binding domain |
| LEDGF | Lens epithelium-derived growth factor |
| LOH | Loss of heterozygosity |
| mCRPC | Metastatic CRPC |
| MEF | Mouse embryonic fibroblast |
| MEN1 | Multiple Endocrine Neoplasia type 1 |
| MI | Molecule inhibitor of menin-MLL interaction |
| MIN | Mammary intraepithelial neoplasia |
| MLL1&2 | mixed lineage leukemia 1&2 (KMT2A and 2B) |
| MTA1 | metastasis-associated protein 1 |
| mTOR | Mammalian target of rapamycin |
| NEPC | Neuroendocrine prostate cancer |
| NF-κB | nuclear factor-κB |
| NLS | Nuclear localization sequence |
| NTD | N-terminal Domain |
| PCa | Prostate cancer |
| PR | Progesterone receptor |
| PRMT5 | Protein arginine N-methyltransferase 5 |
| PSA | Prostate-specific antigen |
| PTEN | Phosphatase and TENsin homolog |
| ROS | Reactive oxygen species |
| SCC | Small cell carcinomas |
| SERDs | Selective estrogen receptor downregulators |
| SERMs | Selective estrogen receptor modulators |
| TGF-β | Transforming growth factor beta |
| Th2 | T- helper type 2 |
| TIT-5 | Tamoxifen Induced Tag-5 |
| TMPRSS2 | Transmembrane protease serine 2 |
| TNBC | Triple negative breast cancer |
| TNFα | Tumor necrosis factor alpha |
| TSS | Transcription start site |
| Wap | Whey acidic protein |
References
- Latest Global Cancer Data: Cancer Burden Rises to 18.1 Million New Cases and 9.6 Million Cancer Deaths in 2018; The International Agency for Research on Cancer (IARC): Geneva, Switzerland, 2018; Volume 263, p. 3.
- Siegel, R.L.; Miller, K.D. Cancer Statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef]
- Boyle, P.; Ferlay, J. Cancer Incidence and Mortality in Europe, 2004. Ann. Oncol. 2005, 16, 481–488. [Google Scholar] [CrossRef]
- Malhotra, G.K.; Zhao, X.; Band, H.; Band, V. Histological, Molecular and Functional Subtypes of Breast Cancers. Cancer Biol. Ther. 2010, 10, 955–960. [Google Scholar] [CrossRef] [PubMed]
- Eliyatkın, N.; Yalçın, E.; Zengel, B.; Aktaş, S.; Vardar, E. Molecular Classification of Breast Carcinoma: From Traditional, Old-Fashioned Way to A New Age, and A New Way. J. Breast Health 2015, 11, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, B.D.; Bauer, J.A.; Chen, X.; Sanders, M.E.; Chakravarthy, A.B.; Shyr, Y.; Pietenpol, J.A. Identification of Human Triple-Negative Breast Cancer Subtypes and Preclinical Models for Selection of Targeted Therapies. J. Clin. Investig. 2011, 121, 2750–2767. [Google Scholar] [CrossRef] [PubMed]
- Tong, C.W.S.; Wu, M.; Cho, W.C.S.; To, K.K.W. Recent Advances in the Treatment of Breast Cancer. Front. Oncol. 2018, 8, 227. [Google Scholar] [CrossRef]
- Gleason, D.F. Classification of Prostatic Carcinomas. Cancer Chem. Rep. 1966, 50, 125–128. [Google Scholar]
- Gleason, D.F.; Mellinger, G.T. Prediction of Prognosis for Prostatic Adenocarcinoma by Combined Histological Grading and Clinical Staging. J. Urol. 1974, 111, 58–64. [Google Scholar] [CrossRef]
- Humphrey, P.A. Gleason Grading and Prognostic Factors in Carcinoma of the Prostate. Mod. Pathol. Off. J. U.S. Can. Acad. Pathol. Inc. 2004, 17, 292–306. [Google Scholar] [CrossRef]
- Nelson, P.S.; Clegg, N.; Arnold, H.; Ferguson, C.; Bonham, M.; White, J.; Hood, L.; Lin, B. The Program of Androgen-Responsive Genes in Neoplastic Prostate Epithelium. Proc. Natl. Acad. Sci. USA 2002, 99, 11890–11895. [Google Scholar] [CrossRef]
- Montgomery, R.B.; Mostaghel, E.A.; Vessella, R.; Hess, D.L.; Kalhorn, T.F.; Higano, C.S.; True, L.D.; Nelson, P.S. Maintenance of Intratumoral Androgens in Metastatic Prostate Cancer: A Mechanism for Castration-Resistant Tumor Growth. Cancer Res. 2008, 68, 4447–4454. [Google Scholar] [CrossRef] [PubMed]
- Taylor, B.S.; Schultz, N.; Hieronymus, H.; Gopalan, A.; Xiao, Y.; Carver, B.S.; Arora, V.K.; Kaushik, P.; Cerami, E.; Reva, B.; et al. Integrative Genomic Profiling of Human Prostate Cancer. Cancer Cell 2010, 18, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Bluemn, E.G.; Coleman, I.M.; Lucas, J.M.; Coleman, R.T.; Hernandez-Lopez, S.; Tharakan, R.; Bianchi-Frias, D.; Dumpit, R.F.; Kaipainen, A.; Corella, A.N.; et al. Androgen Receptor Pathway-Independent Prostate Cancer Is Sustained through FGF Signaling. Cancer Cell 2017, 32, 474–489.e6. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.R.; Cook, R.; Lee, K.A.; Nelson, J.B. Disease and Host Characteristics as Predictors of Time to First Bone Metastasis and Death in Men with Progressive Castration-Resistant Nonmetastatic Prostate Cancer. Cancer 2011, 117, 2077–2085. [Google Scholar] [CrossRef]
- Roudier, M.P.; True, L.D.; Higano, C.S.; Vesselle, H.; Ellis, W.; Lange, P.; Vessella, R.L. Phenotypic Heterogeneity of End-Stage Prostate Carcinoma Metastatic to Bone. Hum. Pathol. 2003, 34, 646–653. [Google Scholar] [CrossRef]
- Wang, W.; Epstein, J.I. Small Cell Carcinoma of the Prostate. A Morphologic and Immunohistochemical Study of 95 Cases. Am. J. Surg. Pathol. 2008, 32, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Aparicio, A.; Tzelepi, V.; Araujo, J.C.; Guo, C.C.; Liang, S.; Troncoso, P.; Logothetis, C.J.; Navone, N.M.; Maity, S.N. Neuroendocrine Prostate Cancer Xenografts with Large-Cell and Small-Cell Features Derived from a Single Patient’s Tumor: Morphological, Immunohistochemical, and Gene Expression Profiles. Prostate 2011, 71, 846–856. [Google Scholar] [CrossRef]
- Tzelepi, V.; Zhang, J.; Lu, J.F.; Kleb, B.; Wu, G.; Wan, X.; Hoang, A.; Efstathiou, E.; Sircar, K.; Navone, N.M.; et al. Modeling a Lethal Prostate Cancer Variant with Small-Cell Carcinoma Features. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2012, 18, 666–677. [Google Scholar] [CrossRef]
- Turbat-Herrera, E.A.; Herrera, G.A.; Gore, I.; Lott, R.L.; Grizzle, W.E.; Bonnin, J.M. Neuroendocrine Differentiation in Prostatic Carcinomas. A Retrospective Autopsy Study. Arch. Pathol. Lab. Med. 1988, 112, 1100–1105. [Google Scholar]
- Tanaka, M.; Suzuki, Y.; Takaoka, K.; Suzuki, N.; Murakami, S.; Matsuzaki, O.; Shimazaki, J. Progression of Prostate Cancer to Neuroendocrine Cell Tumor. Int. J. Urol. Off. J. Jpn. Urol. Assoc. 2001, 8, 431–436. [Google Scholar] [CrossRef]
- Shah, R.B.; Mehra, R.; Chinnaiyan, A.M.; Shen, R.; Ghosh, D.; Zhou, M.; Macvicar, G.R.; Varambally, S.; Harwood, J.; Bismar, T.A.; et al. Androgen-Independent Prostate Cancer Is a Heterogeneous Group of Diseases: Lessons from a Rapid Autopsy Program. Cancer Res. 2004, 64, 9209–9216. [Google Scholar] [CrossRef] [PubMed]
- Têtu, B.; Ro, J.Y.; Ayala, A.G.; Johnson, D.E.; Logothetis, C.J.; Ordonez, N.G. Small Cell Carcinoma of the Prostate. Part I. A Clinicopathologic Study of 20 Cases. Cancer 1987, 59, 1803–1809. [Google Scholar] [CrossRef]
- Oesterling, J.E.; Hauzeur, C.G.; Farrow, G.M. Small Cell Anaplastic Carcinoma of the Prostate: A Clinical, Pathological and Immunohistological Study of 27 Patients. J. Urol. 1992, 147 Pt 2, 804–807. [Google Scholar] [CrossRef]
- Kos, M.; Reid, G.; Denger, S.; Gannon, F. Minireview: Genomic Organization of the Human ERalpha Gene Promoter Region. Mol. Endocrinol. 2001, 15, 2057–2063. [Google Scholar] [CrossRef] [PubMed]
- Koike, S.; Sakai, M.; Muramatsu, M. Molecular Cloning and Characterization of Rat Estrogen Receptor CDNA. Nucleic Acids Res. 1987, 15, 2499–2513. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Zakharov, M.N.; Khan, S.H.; Miki, R.; Jang, H.; Toraldo, G.; Singh, R.; Bhasin, S.; Jasuja, R. The Dynamic Structure of the Estrogen Receptor. J. Amino Acids 2011, 2011, 812540. [Google Scholar] [CrossRef]
- Flouriot, G.; Brand, H.; Denger, S.; Metivier, R.; Kos, M.; Reid, G.; Sonntag-Buck, V.; Gannon, F. Identification of a New Isoform of the Human Estrogen Receptor-Alpha (HER-Alpha) That Is Encoded by Distinct Transcripts and That Is Able to Repress HER-Alpha Activation Function 1. EMBO J. 2000, 19, 4688–4700. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, X.; Shen, P.; Loggie, B.W.; Chang, Y.; Deuel, T.F. A Variant of Estrogen Receptor-{alpha}, HER-{alpha}36: Transduction of Estrogen- and Antiestrogen-Dependent Membrane-Initiated Mitogenic Signaling. Proc. Natl. Acad. Sci. USA 2006, 103, 9063–9068. [Google Scholar] [CrossRef]
- Omarjee, S.; Jacquemetton, J.; Poulard, C.; Rochel, N.; Dejaegere, A.; Chebaro, Y.; Treilleux, I.; Marangoni, E.; Corbo, L.; Romancer, M.L. The Molecular Mechanisms Underlying the ERα-36-Mediated Signaling in Breast Cancer. Oncogene 2017, 36, 2503–2514. [Google Scholar] [CrossRef]
- Zhang, Q.X.; Borg, A.; Wolf, D.M.; Oesterreich, S.; Fuqua, S.A. An Estrogen Receptor Mutant with Strong Hormone-Independent Activity from a Metastatic Breast Cancer. Cancer Res. 1997, 57, 1244–1249. [Google Scholar]
- Basudan, A.; Priedigkeit, N.; Hartmaier, R.J.; Sokol, E.S.; Bahreini, A.; Watters, R.J.; Boisen, M.M.; Bhargava, R.; Weiss, K.R.; Karsten, M.M.; et al. Frequent ESR1 and CDK Pathway Copy-Number Alterations in Metastatic Breast Cancer. Mol. Cancer Res. 2019, 17, 457–468. [Google Scholar] [CrossRef] [PubMed]
- Ottaviano, Y.L.; Issa, J.P.; Parl, F.F.; Smith, H.S.; Baylin, S.B.; Davidson, N.E. Methylation of the Estrogen Receptor Gene CpG Island Marks Loss of Estrogen Receptor Expression in Human Breast Cancer Cells. Cancer Res. 1994, 54, 2552–2555. [Google Scholar] [PubMed]
- Kawai, H.; Li, H.; Avraham, S.; Jiang, S.; Avraham, H.K. Overexpression of Histone Deacetylase HDAC1 Modulates Breast Cancer Progression by Negative Regulation of Estrogen Receptor Alpha. Int. J. Cancer 2003, 107, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Macaluso, M.; Cinti, C.; Russo, G.; Russo, A.; Giordano, A. PRb2/P130-E2F4/5-HDAC1-SUV39H1-P300 and PRb2/P130-E2F4/5-HDAC1-SUV39H1-DNMT1 Multimolecular Complexes Mediate the Transcription of Estrogen Receptor-Alpha in Breast Cancer. Oncogene 2003, 22, 3511–3517. [Google Scholar] [CrossRef] [PubMed]
- Paech, K.; Webb, P.; Kuiper, G.G.; Nilsson, S.; Gustafsson, J.; Kushner, P.J.; Scanlan, T.S. Differential Ligand Activation of Estrogen Receptors ERalpha and ERbeta at AP1 Sites. Science 1997, 277, 1508–1510. [Google Scholar] [CrossRef]
- Belguise, K.; Guo, S.; Sonenshein, G.E. Activation of FOXO3a by the Green Tea Polyphenol Epigallocatechin-3-Gallate Induces Estrogen Receptor Alpha Expression Reversing Invasive Phenotype of Breast Cancer Cells. Cancer Res. 2007, 67, 5763–5770. [Google Scholar] [CrossRef]
- Madureira, P.A.; Varshochi, R.; Constantinidou, D.; Francis, R.E.; Coombes, R.C.; Yao, K.-M.; Lam, E.W.-F. The Forkhead Box M1 Protein Regulates the Transcription of the Estrogen Receptor Alpha in Breast Cancer Cells. J. Biol. Chem. 2006, 281, 25167–25176. [Google Scholar] [CrossRef]
- Campbell, T.M.; Castro, M.A.A.; de Oliveira, K.G.; Ponder, B.A.J.; Meyer, K.B. ERα Binding by Transcription Factors NFIB and YBX1 Enables FGFR2 Signaling to Modulate Estrogen Responsiveness in Breast Cancer. Cancer Res. 2018, 78, 410–421. [Google Scholar] [CrossRef]
- Lacroix, M.; Leclercq, G. About GATA3, HNF3A, and XBP1, Three Genes Co-Expressed with the Oestrogen Receptor-Alpha Gene (ESR1) in Breast Cancer. Mol. Cell. Endocrinol. 2004, 219, 1–7. [Google Scholar] [CrossRef]
- Bernardo, G.M.; Lozada, K.L.; Miedler, J.D.; Harburg, G.; Hewitt, S.C.; Mosley, J.D.; Godwin, A.K.; Korach, K.S.; Visvader, J.E.; Kaestner, K.H.; et al. FOXA1 Is an Essential Determinant of ERalpha Expression and Mammary Ductal Morphogenesis. Development 2010, 137, 2045–2054. [Google Scholar] [CrossRef]
- van de Vijver, M.J.; He, Y.D.; van’t Veer, L.J.; Dai, H.; Hart, A.A.M.; Voskuil, D.W.; Schreiber, G.J.; Peterse, J.L.; Roberts, C.; Marton, M.J.; et al. A Gene-Expression Signature as a Predictor of Survival in Breast Cancer. N. Engl. J. Med. 2002, 347, 1999–2009. [Google Scholar] [CrossRef]
- Oh, D.S.; Troester, M.A.; Usary, J.; Hu, Z.; He, X.; Fan, C.; Wu, J.; Carey, L.A.; Perou, C.M. Estrogen-Regulated Genes Predict Survival in Hormone Receptor-Positive Breast Cancers. J. Clin. Oncol. 2006, 24, 1656–1664. [Google Scholar] [CrossRef]
- Dreijerink, K.M.A.; Groner, A.C.; Vos, E.S.M.; Font-Tello, A.; Gu, L.; Chi, D.; Reyes, J.; Cook, J.; Lim, E.; Lin, C.Y.; et al. Enhancer-Mediated Oncogenic Function of the Menin Tumor Suppressor in Breast Cancer. Cell Rep. 2017, 18, 2359–2372. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, A.; Wang, Z.; Wysocka, J.; Sanyal, M.; Aufiero, D.J.; Kitabayashi, I.; Herr, W.; Cleary, M.L. Leukemia Proto-Oncoprotein MLL Forms a SET1-like Histone Methyltransferase Complex with Menin to Regulate Hox Gene Expression. Mol. Cell. Biol. 2004, 24, 5639–5649. [Google Scholar] [CrossRef] [PubMed]
- Korach, K.S.; Emmen, J.M.A.; Walker, V.R.; Hewitt, S.C.; Yates, M.; Hall, J.M.; Swope, D.L.; Harrell, J.C.; Couse, J.F. Update on Animal Models Developed for Analyses of Estrogen Receptor Biological Activity. J. Steroid Biochem. Mol. Biol. 2003, 86, 387–391. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Ström, A.; Vega, V.B.; Kong, S.L.; Yeo, A.L.; Thomsen, J.S.; Chan, W.C.; Doray, B.; Bangarusamy, D.K.; Ramasamy, A.; et al. Discovery of Estrogen Receptor Alpha Target Genes and Response Elements in Breast Tumor Cells. Genome Biol. 2004, 5, R66. [Google Scholar] [CrossRef] [PubMed]
- Marino, M.; Galluzzo, P.; Ascenzi, P. Estrogen Signaling Multiple Pathways to Impact Gene Transcription. Curr. Genom. 2006, 7, 497–508. [Google Scholar] [CrossRef]
- Brown, A.M.; Jeltsch, J.M.; Roberts, M.; Chambon, P. Activation of PS2 Gene Transcription Is a Primary Response to Estrogen in the Human Breast Cancer Cell Line MCF-7. Proc. Natl. Acad. Sci. USA 1984, 81, 6344–6348. [Google Scholar] [CrossRef]
- Jakowlew, S.B.; Breathnach, R.; Jeltsch, J.M.; Masiakowski, P.; Chambon, P. Sequence of the PS2 MRNA Induced by Estrogen in the Human Breast Cancer Cell Line MCF-7. Nucleic Acids Res. 1984, 12, 2861–2878. [Google Scholar] [CrossRef]
- Charpentier, A.H.; Bednarek, A.K.; Daniel, R.L.; Hawkins, K.A.; Laflin, K.J.; Gaddis, S.; MacLeod, M.C.; Aldaz, C.M. Effects of Estrogen on Global Gene Expression: Identification of Novel Targets of Estrogen Action. Cancer Res. 2000, 60, 5977–5983. [Google Scholar]
- Cunliffe, H.E.; Ringnér, M.; Bilke, S.; Walker, R.L.; Cheung, J.M.; Chen, Y.; Meltzer, P.S. The Gene Expression Response of Breast Cancer to Growth Regulators: Patterns and Correlation with Tumor Expression Profiles. Cancer Res. 2003, 63, 7158–7166. [Google Scholar] [PubMed]
- Frasor, J.; Danes, J.M.; Komm, B.; Chang, K.C.N.; Lyttle, C.R.; Katzenellenbogen, B.S. Profiling of Estrogen Up- and down-Regulated Gene Expression in Human Breast Cancer Cells: Insights into Gene Networks and Pathways Underlying Estrogenic Control of Proliferation and Cell Phenotype. Endocrinology 2003, 144, 4562–4574. [Google Scholar] [CrossRef] [PubMed]
- Inoue, A.; Yoshida, N.; Omoto, Y.; Oguchi, S.; Yamori, T.; Kiyama, R.; Hayashi, S. Development of CDNA Microarray for Expression Profiling of Estrogen-Responsive Genes. J. Mol. Endocrinol. 2002, 29, 175–192. [Google Scholar] [CrossRef] [PubMed]
- Seth, P.; Krop, I.; Porter, D.; Polyak, K. Novel Estrogen and Tamoxifen Induced Genes Identified by SAGE (Serial Analysis of Gene Expression). Oncogene 2002, 21, 836–843. [Google Scholar] [CrossRef]
- Altucci, L.; Addeo, R.; Cicatiello, L.; Dauvois, S.; Parker, M.G.; Truss, M.; Beato, M.; Sica, V.; Bresciani, F.; Weisz, A. 17beta-Estradiol Induces Cyclin D1 Gene Transcription, P36D1-P34cdk4 Complex Activation and P105Rb Phosphorylation during Mitogenic Stimulation of G(1)-Arrested Human Breast Cancer Cells. Oncogene 1996, 12, 2315–2324. [Google Scholar]
- Dubik, D.; Dembinski, T.C.; Shiu, R.P. Stimulation of C-Myc Oncogene Expression Associated with Estrogen-Induced Proliferation of Human Breast Cancer Cells. Cancer Res. 1987, 47 Pt 1, 6517–6521. [Google Scholar]
- Dubik, D.; Shiu, R.P. Transcriptional Regulation of C-Myc Oncogene Expression by Estrogen in Hormone-Responsive Human Breast Cancer Cells. J. Biol. Chem. 1988, 263, 12705–12708. [Google Scholar]
- Umayahara, Y.; Kawamori, R.; Watada, H.; Imano, E.; Iwama, N.; Morishima, T.; Yamasaki, Y.; Kajimoto, Y.; Kamada, T. Estrogen Regulation of the Insulin-like Growth Factor I Gene Transcription Involves an AP-1 Enhancer. J. Biol. Chem. 1994, 269, 16433–16442. [Google Scholar]
- Elangovan, S.; Moulton, B.C. Progesterone and Estrogen Control of Rates of Synthesis of Uterine Cathepsin D. J. Biol. Chem. 1980, 255, 7474–7479. [Google Scholar]
- Yu, W.C.; Leung, B.S.; Gao, Y.L. Effects of 17 Beta-Estradiol on Progesterone Receptors and the Uptake of Thymidine in Human Breast Cancer Cell Line CAMA-1. Cancer Res. 1981, 41 Pt 1, 5004–5009. [Google Scholar]
- Jenster, G.; van der Korput, H.A.; van Vroonhoven, C.; van der Kwast, T.H.; Trapman, J.; Brinkmann, A.O. Domains of the Human Androgen Receptor Involved in Steroid Binding, Transcriptional Activation, and Subcellular Localization. Mol. Endocrinol. (Baltim. Md.) 1991, 5, 1396–1404. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.; Nonomura, N. Role of Androgen Receptor in Prostate Cancer: A Review. World J. Men’s Health 2019, 37, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Bryce, A.H.; Antonarakis, E.S. Androgen Receptor Splice Variant 7 in Castration-Resistant Prostate Cancer: Clinical Considerations. Int. J. Urol. Off. J. Jpn. Urol. Assoc. 2016, 23, 646–653. [Google Scholar] [CrossRef] [PubMed]
- Hu, R.; Dunn, T.A.; Wei, S.; Isharwal, S.; Veltri, R.W.; Humphreys, E.; Han, M.; Partin, A.W.; Vessella, R.L.; Isaacs, W.B.; et al. Ligand-Independent Androgen Receptor Variants Derived from Splicing of Cryptic Exons Signify Hormone-Refractory Prostate Cancer. Cancer Res. 2009, 69, 16–22. [Google Scholar] [CrossRef]
- Van der Steen, T.; Tindall, D.J.; Huang, H. Posttranslational Modification of the Androgen Receptor in Prostate Cancer. Int. J. Mol. Sci. 2013, 14, 14833–14859. [Google Scholar] [CrossRef] [PubMed]
- Hoang, D.T.; Iczkowski, K.A.; Kilari, D.; See, W.; Nevalainen, M.T. Androgen Receptor-Dependent and -Independent Mechanisms Driving Prostate Cancer Progression: Opportunities for Therapeutic Targeting from Multiple Angles. Oncotarget 2017, 8, 3724–3745. [Google Scholar] [CrossRef]
- Koochekpour, S. Androgen Receptor Signaling and Mutations in Prostate Cancer. Asian J. Androl. 2010, 12, 639–657. [Google Scholar] [CrossRef]
- Grasso, C.S.; Wu, Y.M.; Robinson, D.R.; Cao, X.; Dhanasekaran, S.M.; Khan, A.P.; Quist, M.J.; Jing, X.; Lonigro, R.J.; Brenner, J.C.; et al. The Mutational Landscape of Lethal Castration-Resistant Prostate Cancer. Nature 2012, 487, 239–243. [Google Scholar] [CrossRef]
- Robinson, D.; Van Allen, E.M.; Wu, Y.M.; Schultz, N.; Lonigro, R.J.; Mosquera, J.M.; Montgomery, B.; Taplin, M.E.; Pritchard, C.C.; Attard, G.; et al. Integrative Clinical Genomics of Advanced Prostate Cancer. Cell 2015, 161, 1215–1228. [Google Scholar] [CrossRef]
- Hay, C.W.; Hunter, I.; MacKenzie, A.; McEwan, I.J. An Sp1 Modulated Regulatory Region Unique to Higher Primates Regulates Human Androgen Receptor Promoter Activity in Prostate Cancer Cells. PLoS ONE 2015, 10, e0139990. [Google Scholar] [CrossRef][Green Version]
- Deng, X.; Shao, G.; Zhang, H.T.; Li, C.; Zhang, D.; Cheng, L.; Elzey, B.D.; Pili, R.; Ratliff, T.L.; Huang, J.; et al. Protein Arginine Methyltransferase 5 Functions as an Epigenetic Activator of the Androgen Receptor to Promote Prostate Cancer Cell Growth. Oncogene 2017, 36, 1223–1231. [Google Scholar] [CrossRef] [PubMed]
- Grad, J.M.; Lyons, L.S.; Robins, D.M.; Burnstein, K.L. The Androgen Receptor (AR) Amino-Terminus Imposes Androgen-Specific Regulation of AR Gene Expression via an Exonic Enhancer. Endocrinology 2001, 142, 1107–1116. [Google Scholar] [CrossRef] [PubMed]
- Shafi, A.A.; Yen, A.E.; Weigel, N.L. Androgen Receptors in Hormone-Dependent and Castration-Resistant Prostate Cancer. Pharmacol. Ther. 2013, 140, 223–238. [Google Scholar] [CrossRef] [PubMed]
- Davey, R.A.; Grossmann, M. Androgen Receptor Structure, Function and Biology: From Bench to Bedside. Clin. Biochem. Rev. 2016, 37, 3–15. [Google Scholar]
- Tan, M.H.; Li, J.; Xu, H.E.; Melcher, K.; Yong, E.L. Androgen Receptor: Structure, Role in Prostate Cancer and Drug Discovery. Acta Pharmacol. Sin. 2015, 36, 3–23. [Google Scholar] [CrossRef]
- Fu, M.; Wang, C.; Reutens, A.T.; Wang, J.; Angeletti, R.H.; Siconolfi-Baez, L.; Ogryzko, V.; Avantaggiati, M.-L.; Pestell, R.G. p300 and p300/cAMP-response element-binding protein-associated factor acetylate the androgen receptor at sites governing hormone-dependent transactivation. J. Biol. Chem. 2000, 275, 20853–20860. [Google Scholar] [CrossRef]
- Bakin, R.E.; Gioeli, D.; Bissonette, E.A.; Weber, M.J. Attenuation of Ras Signaling Restores Androgen Sensitivity to Hormone-Refractory C4-2 Prostate Cancer Cells. Cancer Res. 2003, 63, 1975–1980. [Google Scholar]
- Bakin, R.E.; Gioeli, D.; Sikes, R.A.; Bissonette, E.A.; Weber, M.J. Constitutive Activation of the Ras/Mitogen-Activated Protein Kinase Signaling Pathway Promotes Androgen Hypersensitivity in LNCaP Prostate Cancer Cells. Cancer Res. 2003, 63, 1981–1989. [Google Scholar]
- Gregory, C.W.; Fei, X.; Ponguta, L.A.; He, B.; Bill, H.M.; French, F.S.; Wilson, E.M. Epidermal Growth Factor Increases Coactivation of the Androgen Receptor in Recurrent Prostate Cancer. J. Biol. Chem. 2004, 279, 7119–7130. [Google Scholar] [CrossRef]
- Wu, J.D.; Haugk, K.; Woodke, L.; Nelson, P.; Coleman, I.; Plymate, S.R. Interaction of IGF Signaling and the Androgen Receptor in Prostate Cancer Progression. J. Cell. Biochem. 2006, 99, 392–401. [Google Scholar] [CrossRef]
- Schweizer, L.; Rizzo, C.A.; Spires, T.E.; Platero, J.S.; Wu, Q.; Lin, T.A.; Gottardis, M.M.; Attar, R.M. The Androgen Receptor Can Signal through Wnt/Beta-Catenin in Prostate Cancer Cells as an Adaptation Mechanism to Castration Levels of Androgens. BMC Cell Boil. 2008, 9, 4. [Google Scholar] [CrossRef]
- Leung, J.K.; Sadar, M.D. Non-Genomic Actions of the Androgen Receptor in Prostate Cancer. Front. Endocrinol. 2017, 8, 2. [Google Scholar] [CrossRef] [PubMed]
- Mounir, Z.; Korn, J.M.; Westerling, T.; Lin, F.; Kirby, C.A.; Schirle, M.; McAllister, G.; Hoffman, G.; Ramadan, N.; Hartung, A.; et al. ERG Signaling in Prostate Cancer Is Driven through PRMT5-Dependent Methylation of the Androgen Receptor. eLife 2016, 5, e13964. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Chen, S.Y.; Ross, K.N.; Balk, S.P. Androgens induce prostate cancer cell proliferation through mammalian target of rapamycin activation and post-transcriptional increases in cyclin D proteins. Cancer Res. 2006, 66, 7783–7792. [Google Scholar]
- Lu, S.; Liu, M.; Epner, D.E.; Tsai, S.Y.; Tsai, M.J. Androgen Regulation of the Cyclin-Dependent Kinase Inhibitor P21 Gene through an Androgen Response Element in the Proximal Promoter. Mol. Endocrinol. (Baltim. Md.) 1999, 13, 376–384. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Aaltomaa, S.; Lipponen, P.; Eskelinen, M.; Ala-Opas, M.; Kosma, V.M. Prognostic Value and Expression of P21(Waf1/Cip1) Protein in Prostate Cancer. Prostate 1999, 39, 8–15. [Google Scholar] [CrossRef]
- Baretton, G.B.; Klenk, U.; Diebold, J.; Schmeller, N.; Löhrs, U. Proliferation- and Apoptosis-Associated Factors in Advanced Prostatic Carcinomas before and after Androgen Deprivation Therapy: Prognostic Significance of P21/WAF1/CIP1 Expression. Br. J. Cancer 1999, 80, 546–555. [Google Scholar] [CrossRef]
- Knudsen, K.E.; Arden, K.C.; Cavenee, W.K. Multiple G1 Regulatory Elements Control the Androgen-Dependent Proliferation of Prostatic Carcinoma Cells. J. Boil. Chem. 1998, 273, 20213–20222. [Google Scholar] [CrossRef]
- Rokhlin, O.W.; Taghiyev, A.F.; Guseva, N.V.; Glover, R.A.; Chumakov, P.M.; Kravchenko, J.E.; Cohen, M.B. Androgen Regulates Apoptosis Induced by TNFR Family Ligands via Multiple Signaling Pathways in LNCaP. Oncogene 2005, 24, 6773–6784. [Google Scholar] [CrossRef]
- Frezza, M.; Yang, H.; Dou, Q.P. Modulation of the Tumor Cell Death Pathway by Androgen Receptor in Response to Cytotoxic Stimuli. J. Cell. Physiol. 2011, 226, 2731–2739. [Google Scholar] [CrossRef]
- Liao, X.; Tang, S.; Thrasher, J.B.; Griebling, T.L.; Li, B. Small-Interfering RNA-Induced Androgen Receptor Silencing Leads to Apoptotic Cell Death in Prostate Cancer. Mol. Cancer Ther. 2005, 4, 505–515. [Google Scholar] [CrossRef]
- Godfrey, B.; Lin, Y.; Larson, J.; Haferkamp, B.; Xiang, J. Proteasomal Degradation Unleashes the Pro-Death Activity of Androgen Receptor. Cell Res. 2010, 20, 1138–1147. [Google Scholar] [CrossRef]
- Zhao, J.C.; Yu, J.; Runkle, C.; Wu, L.; Hu, M.; Wu, D.; Liu, J.S.; Wang, Q.; Qin, Z.S.; Yu, J. Cooperation between Polycomb and Androgen Receptor during Oncogenic Transformation. Genome Res. 2012, 22, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Yu, Z.; Sun, X.; Feng, J.; Yu, Q.; Khan, H.; Zhu, X.; Huang, L.; Li, M.; Mok, M.T.S.; et al. Androgen Receptor Drives Hepatocellular Carcinogenesis by Activating Enhancer of Zeste Homolog 2-Mediated Wnt/β-Catenin Signaling. EBioMedicine 2018, 35, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Thakker, R.V. Multiple Endocrine Neoplasia Type 1 (MEN1) and Type 4 (MEN4). Mol. Cell. Endocrinol. 2014, 386, 2–15. [Google Scholar] [CrossRef] [PubMed]
- Lemmens, I.; Van de Ven, W.J.; Kas, K.; Zhang, C.X.; Giraud, S.; Wautot, V.; Buisson, N.; De Witte, K.; Salandre, J.; Lenoir, G.; et al. Identification of the Multiple Endocrine Neoplasia Type 1 (MEN1) Gene. The European Consortium on MEN1. Hum. Mol. Genet. 1997, 6, 1177–1183. [Google Scholar] [CrossRef]
- Chandrasekharappa, S.C. Positional Cloning of the Gene for Multiple Endocrine Neoplasia-Type 1. Science 1997, 276, 404–407. [Google Scholar] [CrossRef]
- Wautot, V.; Khodaei, S.; Frappart, L.; Buisson, N.; Baro, E.; Lenoir, G.M.; Calender, A.; Zhang, C.X.; Weber, G. Expression Analysis of Endogenous Menin, the Product of the Multiple Endocrine Neoplasia Type 1 Gene, in Cell Lines and Human Tissues. Int. J. Cancer 2000, 85, 877–881. [Google Scholar] [CrossRef]
- Matkar, S.; Thiel, A.; Hua, X. Menin: A Scaffold Protein That Controls Gene Expression and Cell Signaling. Trends Biochem. Sci. 2013, 38, 394–402. [Google Scholar] [CrossRef]
- Lu, J.; Hamze, Z.; Bonnavion, R.; Herath, N.; Pouponnot, C.; Assade, F.; Fontanière, S.; Bertolino, P.; Cordier-Bussat, M.; Zhang, C.X. Reexpression of Oncoprotein MafB in Proliferative β-Cells and Men1 Insulinomas in Mouse. Oncogene 2012, 31, 3647–3654. [Google Scholar] [CrossRef]
- Hamze, Z.; Vercherat, C.; Bernigaud-Lacheretz, A.; Bazzi, W.; Bonnavion, R.; Lu, J.; Calender, A.; Pouponnot, C.; Bertolino, P.; Roche, C.; et al. Altered MENIN Expression Disrupts the MAFA Differentiation Pathway in Insulinoma. Endocr. Relat. Cancer 2013, 20, 833–848. [Google Scholar] [CrossRef]
- Bonnavion, R.; Teinturier, R.; Gherardi, S.; Leteurtre, E.; Yu, R.; Cordier-Bussat, M.; Du, R.; Pattou, F.; Vantyghem, M.-C.; Bertolino, P.; et al. Foxa2, a Novel Protein Partner of the Tumour Suppressor Menin, Is Deregulated in Mouse and Human MEN1 Glucagonomas. J. Pathol. 2017, 242, 90–101. [Google Scholar] [CrossRef]
- Cao, Y.; Liu, R.; Jiang, X.; Lu, J.; Jiang, J.; Zhang, C.; Li, X.; Ning, G. Nuclear-Cytoplasmic Shuttling of Menin Regulates Nuclear Translocation of {beta}-Catenin. Mol. Cell. Biol. 2009, 29, 5477–5487. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Cao, Y.; Li, F.; Su, Y.; Li, Y.; Peng, Y.; Cheng, Y.; Zhang, C.; Wang, W.; Ning, G. Targeting β-Catenin Signaling for Therapeutic Intervention in MEN1-Deficient Pancreatic Neuroendocrine Tumours. Nat. Commun. 2014, 5, 5809. [Google Scholar] [CrossRef] [PubMed]
- Bazzi, W.; Renon, M.; Vercherat, C.; Hamze, Z.; Lacheretz-Bernigaud, A.; Wang, H.; Blanc, M.; Roche, C.; Calender, A.; Chayvialle, J.-A.; et al. MEN1 Missense Mutations Impair Sensitization to Apoptosis Induced by Wild-Type Menin in Endocrine Pancreatic Tumor Cells. Gastroenterology 2008, 135, 1698–1709.e2. [Google Scholar] [CrossRef] [PubMed]
- Gherardi, S.; Ripoche, D.; Mikaelian, I.; Chanal, M.; Teinturier, R.; Goehrig, D.; Cordier-Bussat, M.; Zhang, C.X.; Hennino, A.; Bertolino, P. Menin Regulates Inhbb Expression through an Akt/Ezh2-Mediated H3K27 Histone Modification. Biochim. Biophys. Acta (BBA) Gene Regul. Mech. 2017, 1860, 427–437. [Google Scholar] [CrossRef]
- Dreijerink, K.M.A.; Varier, R.A.; van Beekum, O.; Jeninga, E.H.; Höppener, J.W.M.; Lips, C.J.M.; Kummer, J.A.; Kalkhoven, E.; Timmers, H.T.M. The Multiple Endocrine Neoplasia Type 1 (MEN1) Tumor Suppressor Regulates Peroxisome Proliferator-Activated Receptor Gamma-Dependent Adipocyte Differentiation. Mol. Cell. Biol. 2009, 29, 5060–5069. [Google Scholar] [CrossRef] [PubMed]
- Aziz, A.; Miyake, T.; Engleka, K.A.; Epstein, J.A.; McDermott, J.C. Menin Expression Modulates Mesenchymal Cell Commitment to the Myogenic and Osteogenic Lineages. Dev. Biol. 2009, 332, 116–130. [Google Scholar] [CrossRef]
- Maillard, I.; Chen, Y.-X.; Friedman, A.; Yang, Y.; Tubbs, A.T.; Shestova, O.; Pear, W.S.; Hua, X. Menin Regulates the Function of Hematopoietic Stem Cells and Lymphoid Progenitors. Blood 2009, 113, 1661–1669. [Google Scholar] [CrossRef]
- Dreijerink, K.M.A.; Mulder, K.W.; Winkler, G.S.; Höppener, J.W.M.; Lips, C.J.M.; Timmers, H.T.M. Menin Links Estrogen Receptor Activation to Histone H3K4 Trimethylation. Cancer Res. 2006, 66, 4929–4935. [Google Scholar] [CrossRef]
- Imachi, H.; Murao, K.; Dobashi, H.; Bhuyan, M.M.; Cao, X.; Kontani, K.; Niki, S.; Murazawa, C.; Nakajima, H.; Kohno, N.; et al. Menin, a Product of the MENI Gene, Binds to Estrogen Receptor to Enhance Its Activity in Breast Cancer Cells: Possibility of a Novel Predictive Factor for Tamoxifen Resistance. Breast Cancer Res. Treat. 2010, 122, 395–407. [Google Scholar] [CrossRef]
- Malik, R.; Khan, A.P.; Asangani, I.A.; Cieślik, M.; Prensner, J.R.; Wang, X.; Iyer, M.K.; Jiang, X.; Borkin, D.; Escara-Wilke, J.; et al. Targeting the MLL Complex in Castration-Resistant Prostate Cancer. Nat. Med. 2015, 21, 344–352. [Google Scholar] [CrossRef]
- Bertolino, P.; Tong, W.-M.; Galendo, D.; Wang, Z.-Q.; Zhang, C.-X. Heterozygous Men1 Mutant Mice Develop a Range of Endocrine Tumors Mimicking Multiple Endocrine Neoplasia Type 1. Mol. Endocrinol. 2003, 17, 1880–1892. [Google Scholar] [CrossRef] [PubMed]
- Seigne, C.; Auret, M.; Treilleux, I.; Bonnavion, R.; Assade, F.; Carreira, C.; Goddard-Léon, S.; Lavergne, E.; Chabaud, S.; Garcia, A.; et al. High Incidence of Mammary Intraepithelial Neoplasia Development in Men1-Disrupted Murine Mammary Glands: Men—1 and Pre-Cancerous Mammary Glands Lesions. J. Pathol. 2013, 229, 546–558. [Google Scholar] [CrossRef] [PubMed]
- Seigne, C.; Fontanière, S.; Carreira, C.; Lu, J.; Tong, W.M.; Fontanière, B.; Wang, Z.Q.; Zhang, C.X.; Frappart, L. Characterisation of Prostate Cancer Lesions in Heterozygous Men1 Mutant Mice. BMC Cancer 2010, 10, 395. [Google Scholar] [CrossRef]
- Honda, M.; Tsukada, T.; Horiuchi, T.; Tanaka, R.; Yamaguchi, K.; Obara, T.; Miyakawa, H.; Yamaji, T.; Ishibashi, M. Primary Hyperparathyroidism Associatiated with Aldosterone-Producing Adrenocortical Adenoma and Breast Cancer: Relation to MEN1 Gene. Intern. Med. 2004, 43, 310–314. [Google Scholar] [CrossRef]
- Jeong, Y.J.; Oh, H.K.; Bong, J.G. Multiple Endocrine Neoplasia Type 1 Associated with Breast Cancer: A Case Report and Review of the Literature. Oncol. Lett. 2014, 8, 230–234. [Google Scholar] [CrossRef]
- Herranz-Antolín, S.; Gil-García, S.; Álvarez-de Frutos, V. Multiple Endocrine Neoplasia Type 1 and Breast Cancer. An Association to Consider. Endocrinol. Diabetes Nutr. 2018, 65, 468–469. [Google Scholar] [CrossRef]
- Dreijerink, K.M.A.; Goudet, P.; Burgess, J.R.; Valk, G.D. International Breast Cancer in MEN1 Study Group. Breast-Cancer Predisposition in Multiple Endocrine Neoplasia Type 1. N. Engl. J. Med. 2014, 371, 583–584. [Google Scholar] [CrossRef]
- Nik-Zainal, S.; Davies, H.; Staaf, J.; Ramakrishna, M.; Glodzik, D.; Zou, X.; Martincorena, I.; Alexandrov, L.B.; Martin, S.; Wedge, D.C.; et al. Landscape of Somatic Mutations in 560 Breast Cancer Whole-Genome Sequences. Nature 2016, 534, 47–54. [Google Scholar] [CrossRef]
- Inic, Z.M.; Inic, M.; Dzodic, R.; Pupic, G.; Damjanovic, S. Breast Cancer in a Patient with Multiple Endocrine Neoplasia Type 1 (MEN 1): A Case Report and Review of the Literature. JCO 2012, 30 (Suppl. 15), e21136. [Google Scholar] [CrossRef]
- Papi, L.; Palli, D.; Masi, L.; Putignano, A.L.; Congregati, C.; Zanna, I.; Marini, F.; Giusti, F.; Luzi, E.; Tonelli, F.; et al. Germline Mutations in MEN1 and BRCA1 Genes in a Woman with Familial Multiple Endocrine Neoplasia Type 1 and Inherited Breast-Ovarian Cancer Syndromes: A Case Report. Cancer Genet. Cytogenet. 2009, 195, 75–79. [Google Scholar] [CrossRef]
- Ghataorhe, P.; Kurian, A.W.; Pickart, A.; Trapane, P.; Norton, J.A.; Kingham, K.; Ford, J.M. A Carrier of Both MEN1 and BRCA2 Mutations: Case Report and Review of the Literature. Cancer Genet. Cytogenet. 2007, 179, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Imachi, H.; Yu, X.; Nishiuchi, T.; Miyai, Y.; Masugata, H.; Murao, K. Raloxifene Inhibits Menin-Dependent Estrogen Receptor Activation in Breast Cancer Cells. J. Endocrinol. Investig. 2011, 34, 813–815. [Google Scholar] [CrossRef]
- Perakakis, N.; Flohr, F.; Kayser, G.; Thomusch, O.; Parsons, L.; Billmann, F.; von Dobschuetz, E.; Rondot, S.; Seufert, J.; Laubner, K. Multiple Endocrine Neoplasia Type 1 Associated with a New Germline Men1 Mutation in a Family with Atypical Tumor Phenotype. Hormones 2016, 15, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Manson-Bahr, D.; Ball, R.; Gundem, G.; Sethia, K.; Mills, R.; Rochester, M.; Goody, V.; Anderson, E.; O’Meara, S.; Flather, M.; et al. Mutation Detection in Formalin-Fixed Prostate Cancer Biopsies Taken at the Time of Diagnosis Using next-Generation DNA Sequencing. J. Clin. Pathol. 2015, 68, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Varier, R.A.; Timmers, H.T. Histone Lysine Methylation and Demethylation Pathways in Cancer. Biochim. Biophys. Acta 2011, 1815, 75–89. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Zhang, J.; Sampieri, K.; Clohessy, J.G.; Mendez, L.; Gonzalez-Billalabeitia, E.; Liu, X.S.; Lee, Y.R.; Fung, J.; Katon, J.M.; et al. An Aberrant SREBP-Dependent Lipogenic Program Promotes Metastatic Prostate Cancer. Nat. Genet. 2018, 50, 206–218. [Google Scholar] [CrossRef]
- Paris, P.L.; Andaya, A.; Fridlyand, J.; Jain, A.N.; Weinberg, V.; Kowbel, D.; Brebner, J.H.; Simko, J.; Watson, J.E.V.; Volik, S.; et al. Whole Genome Scanning Identifies Genotypes Associated with Recurrence and Metastasis in Prostate Tumors. Hum. Mol. Genet. 2004, 13, 1303–1313. [Google Scholar] [CrossRef] [PubMed]
- Heselmeyer-Haddad, K.M.; Berroa Garcia, L.Y.; Bradley, A.; Hernandez, L.; Hu, Y.; Habermann, J.K.; Dumke, C.; Thorns, C.; Perner, S.; Pestova, E.; et al. Single-Cell Genetic Analysis Reveals Insights into Clonal Development of Prostate Cancers and Indicates Loss of PTEN as a Marker of Poor Prognosis. Am. J. Pathol. 2014, 184, 2671–2686. [Google Scholar] [CrossRef] [PubMed]
- Paris, P.L.; Sridharan, S.; Hittelman, A.B.; Kobayashi, Y.; Perner, S.; Huang, G.; Simko, J.; Carroll, P.; Rubin, M.A.; Collins, C. An Oncogenic Role for the Multiple Endocrine Neoplasia Type 1 Gene in Prostate Cancer. Prostate Cancer Prostatic Dis. 2009, 12, 184–191. [Google Scholar] [CrossRef]
- Zhu, S.; Xu, Y.; Song, M.; Chen, G.; Wang, H.; Zhao, Y.; Wang, Z.; Li, F. PRDM16 Is Associated with Evasion of Apoptosis by Prostatic Cancer Cells According to RNA Interference Screening. Mol. Med. Rep. 2016, 14, 3357–3361. [Google Scholar] [CrossRef] [PubMed]
- Ansari, K.I.; Shrestha, B.; Hussain, I.; Kasiri, S.; Mandal, S.S. Histone Methylases MLL1 and MLL3 Coordinate with Estrogen Receptors in Estrogen-Mediated HOXB9 Expression. Biochemistry 2011, 50, 3517–3527. [Google Scholar] [CrossRef]
- Yokoyama, A.; Cleary, M.L. Menin Critically Links MLL Proteins with LEDGF on Cancer-Associated Target Genes. Cancer Cell 2008, 14, 36–46. [Google Scholar] [CrossRef]
- Gao, S.-B.; Feng, Z.-J.; Xu, B.; Wu, Y.; Yin, P.; Yang, Y.; Hua, X.; Jin, G.-H. Suppression of Lung Adenocarcinoma through Menin and Polycomb Gene-Mediated Repression of Growth Factor Pleiotrophin. Oncogene 2009, 28, 4095–4104. [Google Scholar] [CrossRef]
- Gurung, B.; Feng, Z.; Hua, X. Menin Directly Represses Gli1 Expression Independent of Canonical Hedgehog Signaling. Mol. Cancer Res. 2013, 11, 1215–1222. [Google Scholar] [CrossRef]
- Hwang, C.; Giri, V.N.; Wilkinson, J.C.; Wright, C.W.; Wilkinson, A.S.; Cooney, K.A.; Duckett, C.S. EZH2 Regulates the Transcription of Estrogen-Responsive Genes through Association with REA, an Estrogen Receptor Corepressor. Breast Cancer Res. Treat. 2008, 107, 235–242. [Google Scholar] [CrossRef]
- Powers, M.A.; Fay, M.M.; Factor, R.E.; Welm, A.L.; Ullman, K.S. Protein Arginine Methyltransferase 5 Accelerates Tumor Growth by Arginine Methylation of the Tumor Suppressor Programmed Cell Death 4. Cancer Res. 2011, 71, 5579–5587. [Google Scholar] [CrossRef]
- Agarwal, S.K.; Guru, S.C.; Heppner, C.; Erdos, M.R.; Collins, R.M.; Park, S.Y.; Saggar, S.; Chandrasekharappa, S.C.; Collins, F.S.; Spiegel, A.M.; et al. Menin Interacts with the AP1 Transcription Factor JunD and Represses JunD-Activated Transcription. Cell 1999, 96, 143–152. [Google Scholar] [CrossRef]
- Huang, J.; Gurung, B.; Wan, B.; Matkar, S.; Veniaminova, N.A.; Wan, K.; Merchant, J.L.; Hua, X.; Lei, M. The Same Pocket in Menin Binds Both MLL and JUND but Has Opposite Effects on Transcription. Nature 2012, 482, 542–546. [Google Scholar] [CrossRef]
- Kharman-Biz, A.; Gao, H.; Ghiasvand, R.; Zhao, C.; Zendehdel, K.; Dahlman-Wright, K. Expression of Activator Protein-1 (AP-1) Family Members in Breast Cancer. BMC Cancer 2013, 13, 441. [Google Scholar] [CrossRef]
- Hipkaeo, W.; Sakulsak, N.; Wakayama, T.; Yamamoto, M.; Nakaya, M.-A.; Keattikunpairoj, S.; Kurobo, M.; Iseki, S. Coexpression of Menin and JunD during the Duct Cell Differentiation in Mouse Submandibular Gland. Tohoku J. Exp. Med. 2008, 214, 231–245. [Google Scholar] [CrossRef][Green Version]
- Chen, D.; Reierstad, S.; Fang, F.; Bulun, S.E. JunD and JunB Integrate Prostaglandin E2 Activation of Breast Cancer-Associated Proximal Aromatase Promoters. Mol. Endocrinol. 2011, 25, 767–775. [Google Scholar] [CrossRef]
- Millena, A.C.; Vo, B.T.; Khan, S.A. JunD Is Required for Proliferation of Prostate Cancer Cells and Plays a Role in Transforming Growth Factor-β (TGF-β)-Induced Inhibition of Cell Proliferation. J. Boil. Chem. 2016, 291, 17964–17976. [Google Scholar] [CrossRef]
- Perou, C.M.; Sørlie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; Rees, C.A.; Pollack, J.R.; Ross, D.T.; Johnsen, H.; Akslen, L.A.; et al. Molecular Portraits of Human Breast Tumours. Nature 2000, 406, 747–752. [Google Scholar] [CrossRef]
- Elliott, B.; Millena, A.C.; Matyunina, L.; Zhang, M.; Zou, J.; Wang, G.; Zhang, Q.; Bowen, N.; Eaton, V.; Webb, G.; et al. Essential Role of JunD in Cell Proliferation Is Mediated via MYC Signaling in Prostate Cancer Cells. Cancer Lett. 2019, 448, 155–167. [Google Scholar] [CrossRef]
- Mehraein-Ghomi, F.; Basu, H.S.; Church, D.R.; Hoffmann, F.M.; Wilding, G. Androgen Receptor Requires JunD as a Coactivator to Switch on an Oxidative Stress Generation Pathway in Prostate Cancer Cells. Cancer Res. 2010, 70, 4560–4568. [Google Scholar] [CrossRef]
- Shang, Y.; Hu, X.; DiRenzo, J.; Lazar, M.A.; Brown, M. Cofactor Dynamics and Sufficiency in Estrogen Receptor-Regulated Transcription. Cell 2000, 103, 843–852. [Google Scholar] [CrossRef]
- Shang, Y.; Brown, M. Molecular Determinants for the Tissue Specificity of SERMs. Science 2002, 295, 2465–2468. [Google Scholar] [CrossRef]
- Bièche, I.; Laurendeau, I.; Tozlu, S.; Olivi, M.; Vidaud, D.; Lidereau, R.; Vidaud, M. Quantitation of MYC Gene Expression in Sporadic Breast Tumors with a Real-Time Reverse Transcription-PCR Assay. Cancer Res. 1999, 59, 2759–2765. [Google Scholar]
- Cheng, A.S.L.; Jin, V.X.; Fan, M.; Smith, L.T.; Liyanarachchi, S.; Yan, P.S.; Leu, Y.-W.; Chan, M.W.Y.; Plass, C.; Nephew, K.P.; et al. Combinatorial Analysis of Transcription Factor Partners Reveals Recruitment of C-MYC to Estrogen Receptor-Alpha Responsive Promoters. Mol. Cell 2006, 21, 393–404. [Google Scholar] [CrossRef]
- Fleming, W.H.; Hamel, A.; MacDonald, R.; Ramsey, E.; Pettigrew, N.M.; Johnston, B.; Dodd, J.G.; Matusik, R.J. Expression of the C-Myc Protooncogene in Human Prostatic Carcinoma and Benign Prostatic Hyperplasia. Cancer Res. 1986, 46, 1535–1538. [Google Scholar]
- Sato, H.; Minei, S.; Hachiya, T.; Yoshida, T.; Takimoto, Y. Fluorescence in Situ Hybridization Analysis of C-Myc Amplification in Stage TNM Prostate Cancer in Japanese Patients. Int. J. Urol. Off. J. Jpn. Urol. Assoc. 2006, 13, 761–766. [Google Scholar] [CrossRef]
- Wu, G.; Yuan, M.; Shen, S.; Ma, X.; Fang, J.; Zhu, L.; Sun, L.; Liu, Z.; He, X.; Huang, D.; et al. Menin Enhances C-Myc-Mediated Transcription to Promote Cancer Progression. Nat. Commun. 2017, 8, 15278. [Google Scholar] [CrossRef]
- Zaman, S.; Sukhodolets, K.; Wang, P.; Qin, J.; Levens, D.; Agarwal, S.K.; Marx, S.J. FBP1 Is an Interacting Partner of Menin. Int. J. Endocrinol. 2014, 2014, 535401. [Google Scholar] [CrossRef]
- Carroll, J.S.; Brown, M. Estrogen Receptor Target Gene: An Evolving Concept. Mol. Endocrinol. 2006, 20, 1707–1714. [Google Scholar] [CrossRef]
- Carroll, J.S.; Liu, X.S.; Brodsky, A.S.; Li, W.; Meyer, C.A.; Szary, A.J.; Eeckhoute, J.; Shao, W.; Hestermann, E.V.; Geistlinger, T.R.; et al. Chromosome-Wide Mapping of Estrogen Receptor Binding Reveals Long-Range Regulation Requiring the Forkhead Protein FoxA1. Cell 2005, 122, 33–43. [Google Scholar] [CrossRef]
- Nakata, Y.; Brignier, A.C.; Jin, S.; Shen, Y.; Rudnick, S.I.; Sugita, M.; Gewirtz, A.M. C-Myb, Menin, GATA-3, and MLL Form a Dynamic Transcription Complex That Plays a Pivotal Role in Human T Helper Type 2 Cell Development. Blood 2010, 116, 1280–1290. [Google Scholar] [CrossRef]
- Obinata, D.; Takayama, K.; Takahashi, S.; Inoue, S. Crosstalk of the Androgen Receptor with Transcriptional Collaborators: Potential Therapeutic Targets for Castration-Resistant Prostate Cancer. Cancers 2017, 9, 22. [Google Scholar] [CrossRef]
- Castaneda, C.A.; Cortes-Funes, H.; Gomez, H.L.; Ciruelos, E.M. The Phosphatidyl Inositol 3-Kinase/AKT Signaling Pathway in Breast Cancer. Cancer Metastasis Rev. 2010, 29, 751–759. [Google Scholar] [CrossRef]
- Ellis, M.J.; Perou, C.M. The Genomic Landscape of Breast Cancer as a Therapeutic Roadmap. Cancer Discov. 2013, 3, 27–34. [Google Scholar] [CrossRef]
- Stemke-Hale, K.; Gonzalez-Angulo, A.M.; Lluch, A.; Neve, R.M.; Kuo, W.L.; Davies, M.; Carey, M.; Hu, Z.; Guan, Y.; Sahin, A.; et al. An integrative genomic and proteomic analysis of PIK3CA, PTEN, and AKT mutations in breast cancer. Cancer Res. 2008, 68, 6084–6091. [Google Scholar] [CrossRef]
- Jones, N.; Bonnet, F.; Sfar, S.; Lafitte, M.; Lafon, D.; Sierankowski, G.; Brouste, V.; Banneau, G.; Tunon de Lara, C.; Debled, M.; et al. Comprehensive Analysis of PTEN Status in Breast Carcinomas. Int. J. Cancer 2013, 133, 323–334. [Google Scholar] [CrossRef]
- Zhang, H.; Li, W.; Wang, Q.; Wang, X.; Li, F.; Zhang, C.; Wu, L.; Long, H.; Liu, Y.; Li, X.; et al. Glucose-Mediated Repression of Menin Promotes Pancreatic β-Cell Proliferation. Endocrinology 2012, 153, 602–611. [Google Scholar] [CrossRef]
- Li, S.; Shen, Y.; Wang, M.; Yang, J.; Lv, M.; Li, P.; Chen, Z.; Yang, J. Loss of PTEN Expression in Breast Cancer: Association with Clinicopathological Characteristics and Prognosis. Oncotarget 2017, 8, 32043–32054. [Google Scholar] [CrossRef]
- Wang, S.; Gao, J.; Lei, Q.; Rozengurt, N.; Pritchard, C.; Jiao, J.; Thomas, G.V.; Li, G.; Roy-Burman, P.; Nelson, P.S.; et al. Prostate-Specific Deletion of the Murine Pten Tumor Suppressor Gene Leads to Metastatic Prostate Cancer. Cancer Cell 2003, 4, 209–221. [Google Scholar] [CrossRef]
- Shen, M.M.; Abate-Shen, C. Pten Inactivation and the Emergence of Androgen-Independent Prostate Cancer. Cancer Res. 2007, 67, 6535–6538. [Google Scholar] [CrossRef]
- Sircar, K.; Yoshimoto, M.; Monzon, F.A.; Koumakpayi, I.H.; Katz, R.L.; Khanna, A.; Alvarez, K.; Chen, G.; Darnel, A.D.; Aprikian, A.G.; et al. PTEN Genomic Deletion Is Associated with P-Akt and AR Signalling in Poorer Outcome, Hormone Refractory Prostate Cancer. J. Pathol. 2009, 218, 505–513. [Google Scholar] [CrossRef]
- Jiao, Y.; Shi, C.; Edil, B.H.; de Wilde, R.F.; Klimstra, D.S.; Maitra, A.; Schulick, R.D.; Tang, L.H.; Wolfgang, C.L.; Choti, M.A.; et al. DAXX/ATRX, MEN1, and MTOR Pathway Genes Are Frequently Altered in Pancreatic Neuroendocrine Tumors. Science 2011, 331, 1199–1203. [Google Scholar] [CrossRef]
- Scarpa, A.; Chang, D.K.; Nones, K.; Corbo, V.; Patch, A.-M.; Bailey, P.; Lawlor, R.T.; Johns, A.L.; Miller, D.K.; Mafficini, A.; et al. Whole-Genome Landscape of Pancreatic Neuroendocrine Tumours. Nature 2017, 543, 65–71. [Google Scholar] [CrossRef]
- Boulay, A.; Rudloff, J.; Ye, J.; Zumstein-Mecker, S.; O’Reilly, T.; Evans, D.B.; Chen, S.; Lane, H.A. Dual Inhibition of MTOR and Estrogen Receptor Signaling in Vitro Induces Cell Death in Models of Breast Cancer. Clin. Cancer Res. 2005, 11, 5319–5328. [Google Scholar] [CrossRef]
- Crowder, R.J.; Phommaly, C.; Tao, Y.; Hoog, J.; Luo, J.; Perou, C.M.; Parker, J.S.; Miller, M.A.; Huntsman, D.G.; Lin, L.; et al. PIK3CA and PIK3CB Inhibition Produce Synthetic Lethality When Combined with Estrogen Deprivation in Estrogen Receptor-Positive Breast Cancer. Cancer Res. 2009, 69, 3955–3962. [Google Scholar] [CrossRef]
- Miller, T.W.; Hennessy, B.T.; González-Angulo, A.M.; Fox, E.M.; Mills, G.B.; Chen, H.; Higham, C.; García-Echeverría, C.; Shyr, Y.; Arteaga, C.L. Hyperactivation of Phosphatidylinositol-3 Kinase Promotes Escape from Hormone Dependence in Estrogen Receptor-Positive Human Breast Cancer. J. Clin. Investig. 2010, 120, 2406–2413. [Google Scholar] [CrossRef]
- Nagata, Y.; Lan, K.-H.; Zhou, X.; Tan, M.; Esteva, F.J.; Sahin, A.A.; Klos, K.S.; Li, P.; Monia, B.P.; Nguyen, N.T.; et al. PTEN Activation Contributes to Tumor Inhibition by Trastuzumab, and Loss of PTEN Predicts Trastuzumab Resistance in Patients. Cancer Cell 2004, 6, 117–127. [Google Scholar] [CrossRef]
- Wang, Y.; Ozawa, A.; Zaman, S.; Prasad, N.B.; Chandrasekharappa, S.C.; Agarwal, S.K.; Marx, S.J. The Tumor Suppressor Protein Menin Inhibits AKT Activation by Regulating Its Cellular Localization. Cancer Res. 2011, 71, 371–382. [Google Scholar] [CrossRef]
- Wuescher, L.; Angevine, K.; Hinds, T.; Ramakrishnan, S.; Najjar, S.M.; Mensah-Osman, E.J. Insulin Regulates Menin Expression, Cytoplasmic Localization, and Interaction with FOXO1. Am. J. Physiol. Endocrinol. Metab. 2011, 301, E474–E483. [Google Scholar] [CrossRef]
- Li, H.; Liu, X.; Wang, Z.; Lin, X.; Yan, Z.; Cao, Q.; Zhao, M.; Shi, K. MEN1/Menin Regulates Milk Protein Synthesis through MTOR Signaling in Mammary Epithelial Cells. Sci. Rep. 2017, 7, 5479. [Google Scholar] [CrossRef]
- Kaji, H.; Canaff, L.; Lebrun, J.J.; Goltzman, D.; Hendy, G.N. Inactivation of menin, a Smad3-interacting protein, blocks transforming growth factor type beta signaling. Proc. Natl. Acad. Sci. USA 2001, 98, 3837–3842. [Google Scholar]
- Ratineau, C.; Bernard, C.; Poncet, G.; Blanc, M.; Josso, C.; Fontanière, S.; Calender, A.; Chayvialle, J.A.; Zhang, C.X.; Roche, C. Reduction of Menin Expression Enhances Cell Proliferation and Is Tumorigenic in Intestinal Epithelial Cells. J. Biol. Chem. 2004, 279, 24477–24484. [Google Scholar] [CrossRef]
- Fontanière, S.; Tost, J.; Wierinckx, A.; Lachuer, J.; Lu, J.; Hussein, N.; Busato, F.; Gut, I.; Wang, Z.Q.; Zhang, C.X. Gene Expression Profiling in Insulinomas of Men1 Beta-Cell Mutant Mice Reveals Early Genetic and Epigenetic Events Involved in Pancreatic Beta-Cell Tumorigenesis. Endocr. Relat. Cancer 2006, 13, 1223–1236. [Google Scholar] [CrossRef]
- Schnepp, R.W.; Hou, Z.; Wang, H.; Petersen, C.; Silva, A.; Masai, H.; Hua, X. Functional Interaction between Tumor Suppressor Menin and Activator of S-Phase Kinase. Cancer Res. 2004, 64, 6791–6796. [Google Scholar] [CrossRef]
- Pei, X.-H.; Bai, F.; Smith, M.D.; Usary, J.; Fan, C.; Pai, S.-Y.; Ho, I.-C.; Perou, C.M.; Xiong, Y. CDK Inhibitor P18(INK4c) Is a Downstream Target of GATA3 and Restrains Mammary Luminal Progenitor Cell Proliferation and Tumorigenesis. Cancer Cell 2009, 15, 389–401. [Google Scholar] [CrossRef]
- Stephens, P.J.; Tarpey, P.S.; Davies, H.; Van Loo, P.; Greenman, C.; Wedge, D.C.; Nik-Zainal, S.; Martin, S.; Varela, I.; Bignell, G.R.; et al. The Landscape of Cancer Genes and Mutational Processes in Breast Cancer. Nature 2012, 486, 400–404. [Google Scholar] [CrossRef]
- Karnik, S.K.; Hughes, C.M.; Gu, X.; Rozenblatt-Rosen, O.; McLean, G.W.; Xiong, Y.; Meyerson, M.; Kim, S.K. Menin Regulates Pancreatic Islet Growth by Promoting Histone Methylation and Expression of Genes Encoding P27Kip1 and P18INK4c. Proc. Natl. Acad. Sci. USA 2005, 102, 14659–14664. [Google Scholar] [CrossRef]
- Milne, T.A.; Hughes, C.M.; Lloyd, R.; Yang, Z.; Rozenblatt-Rosen, O.; Dou, Y.; Schnepp, R.W.; Krankel, C.; LiVolsi, V.A.; Gibbs, D.; et al. Menin and MLL Cooperatively Regulate Expression of Cyclin-Dependent Kinase Inhibitors. Proc. Natl. Acad. Sci. USA 2005, 102, 749–754. [Google Scholar] [CrossRef]
- Schnepp, R.W.; Chen, Y.-X.; Wang, H.; Cash, T.; Silva, A.; Diehl, J.A.; Brown, E.; Hua, X. Mutation of Tumor Suppressor Gene Men1 Acutely Enhances Proliferation of Pancreatic Islet Cells. Cancer Res. 2006, 66, 5707–5715. [Google Scholar] [CrossRef]
- Schnepp, R.W.; Mao, H.; Sykes, S.M.; Zong, W.X.; Silva, A.; La, P.; Hua, X. Menin Induces Apoptosis in Murine Embryonic Fibroblasts. J. Biol. Chem. 2004, 279, 10685–10691. [Google Scholar] [CrossRef]
- Lindsten, T.; Ross, A.J.; King, A.; Zong, W.X.; Rathmell, J.C.; Shiels, H.A.; Ulrich, E.; Waymire, K.G.; Mahar, P.; Frauwirth, K.; et al. The Combined Functions of Proapoptotic Bcl-2 Family Members Bak and Bax Are Essential for Normal Development of Multiple Tissues. Mol. Cell 2000, 6, 1389–1399. [Google Scholar] [CrossRef]
- Yeh, Y.; Guo, Q.; Connelly, Z.; Cheng, S.; Yang, S.; Prieto-Dominguez, N.; Yu, X. Wnt/Beta-Catenin Signaling and Prostate Cancer Therapy Resistance. Adv. Exp. Med. Biol. 2019, 1210, 351–378. [Google Scholar] [CrossRef]
- Kim, B.; Song, T.Y.; Jung, K.Y.; Kim, S.G.; Cho, E.J. Direct Interaction of Menin Leads to Ubiquitin-Proteasomal Degradation of β-Catenin. Biochem. Biophys. Res. Commun. 2017, 492, 128–134. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abou Ziki, R.; Luo, Y.; Vlaeminck-Guillem, V.; Le Romancer, M.; Zhang, C.X. Involvement of the MEN1 Gene in Hormone-Related Cancers: Clues from Molecular Studies, Mouse Models, and Patient Investigations. Endocrines 2020, 1, 58-81. https://doi.org/10.3390/endocrines1020007
Abou Ziki R, Luo Y, Vlaeminck-Guillem V, Le Romancer M, Zhang CX. Involvement of the MEN1 Gene in Hormone-Related Cancers: Clues from Molecular Studies, Mouse Models, and Patient Investigations. Endocrines. 2020; 1(2):58-81. https://doi.org/10.3390/endocrines1020007
Chicago/Turabian StyleAbou Ziki, Razan, Yakun Luo, Virginie Vlaeminck-Guillem, Muriel Le Romancer, and Chang Xian Zhang. 2020. "Involvement of the MEN1 Gene in Hormone-Related Cancers: Clues from Molecular Studies, Mouse Models, and Patient Investigations" Endocrines 1, no. 2: 58-81. https://doi.org/10.3390/endocrines1020007
APA StyleAbou Ziki, R., Luo, Y., Vlaeminck-Guillem, V., Le Romancer, M., & Zhang, C. X. (2020). Involvement of the MEN1 Gene in Hormone-Related Cancers: Clues from Molecular Studies, Mouse Models, and Patient Investigations. Endocrines, 1(2), 58-81. https://doi.org/10.3390/endocrines1020007





