Steroid-Induced Iatrogenic Adrenal Insufficiency in Children: A Literature Review
Abstract
:1. Mainstem Concepts of Adrenal Insufficiency
1.1. Primary, Secondary, and Tertiary Adrenal Insufficiency
1.2. Iatrogenic Adrenal Insufficiency
2. Case Presentations of SIAI
3. Effect of Steroid Dosage and Administration Duration on SIAI
3.1. Dose Dependency
3.2. Dose Threshold for SIAI
3.3. Adverse Effects of Glucocorticoid Pulse Therapy
3.4. Factors Associated with SIAI Development
4. Clinical Manifestations of SIAI
4.1. Chronic Symptoms
4.2. Symptoms as Side Effects of Glucocorticoids
4.3. Acute Symptoms in the Presence of Stressors
5. Diagnostic Approaches to SIAI
5.1. Variation in Diagnostic Approaches
5.2. Low-Dose ACTH Stimulation Test
5.3. Early Morning Serum Cortisol Level
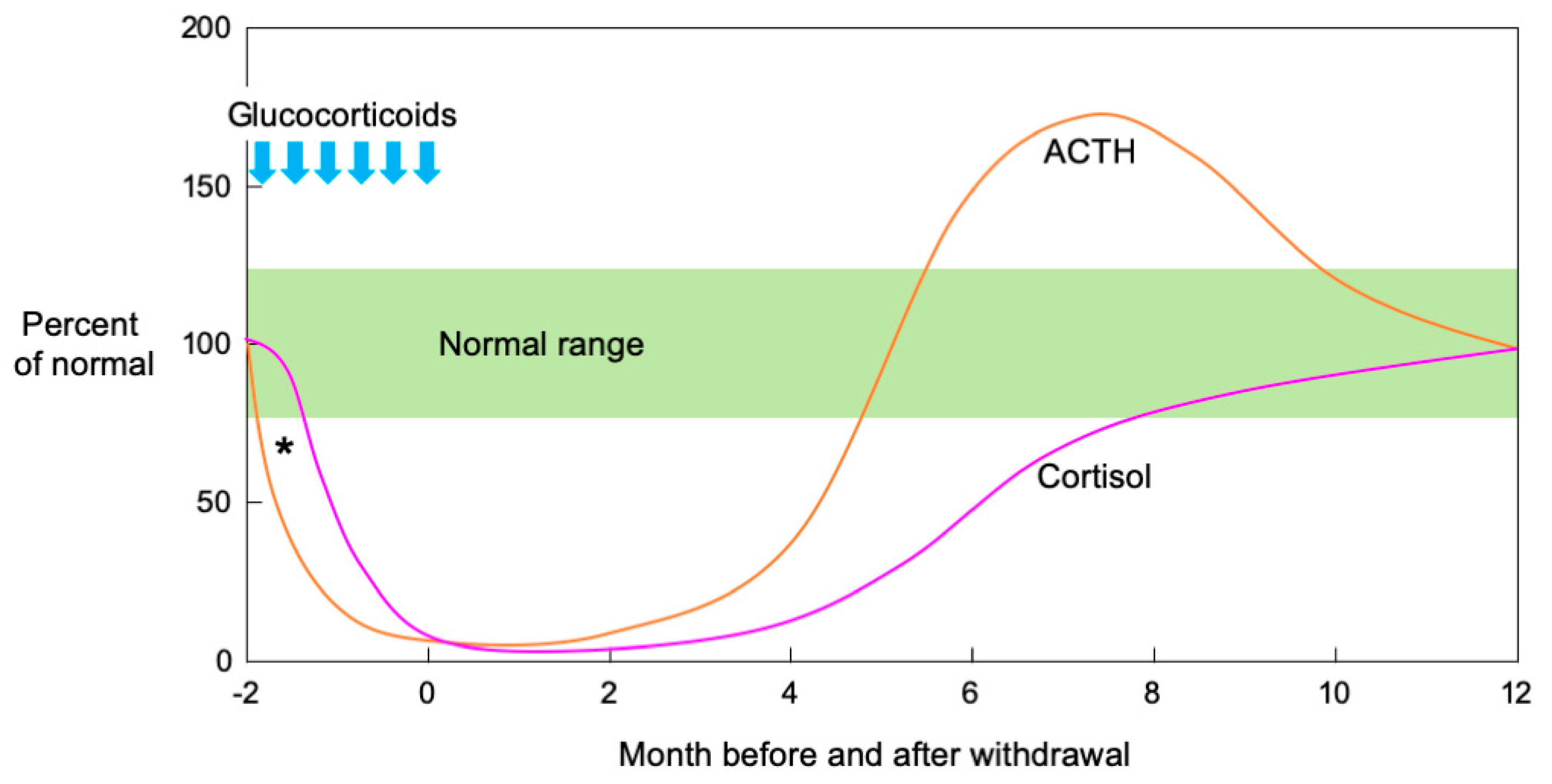
6. Recovery Course in SIAI
6.1. Long-Term Administration
6.2. Short-Term Administration
7. Practical Management of SIAI
7.1. Tapering from the Therapeutic to Physiological Replacement Dose
7.2. Stress Doses
7.3. Patient Education
8. Further Considerations Regarding SIAI
8.1. Pathophysiological and Pharmacological Research
8.2. Clinical and Epidemiological Research
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oelkers, W. Adrenal insufficiency. N. Engl. J. Med. 1996, 335, 1206–1212. [Google Scholar] [CrossRef]
- Charmandari, E.; Nicolaides, N.C.; Chrousos, G.P. Adrenal insufficiency. Lancet (Lond. Engl.) 2014, 383, 2152–2167. [Google Scholar] [CrossRef]
- Crowley, R.K.; Argese, N.; Tomlinson, J.W.; Stewart, P.M. Central hypoadrenalism. J. Clin. Endocrinol. Metab. 2014, 99, 4027–4036. [Google Scholar] [CrossRef]
- Cooper, M.S.; Stewart, P.M. Corticosteroid insufficiency in acutely ill patients. N. Engl. J. Med. 2003, 348, 727–734. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomez-Sanchez, C.E.; Gomez-Sanchez, E.P.; Yamakita, N. Endocrine causes of hypertension. Semin. Nephrol. 1995, 15, 106–115. [Google Scholar] [PubMed]
- Patti, G.; Guzzeti, C.; Di Iorgi, N.; Maria Allegri, A.E.; Napoli, F.; Loche, S.; Maghnie, M. Central adrenal insufficiency in children and adolescents. Best Pract. Res. Clin. Endocrinol. Metab. 2018, 32, 425–444. [Google Scholar] [CrossRef] [PubMed]
- Cemeroglu, A.P.; Coulas, T.; Kleis, L. Spectrum of clinical presentations and endocrinological findings of patients with septo-optic dysplasia: A retrospective study. J. Pediatr. Endocrinol. Metab. 2015, 28, 1057–1063. [Google Scholar] [CrossRef]
- Miyamoto, J.; Hasegawa, Y.; Ohnami, N.; Onigata, K.; Kinoshita, E.; Nishi, Y.; Tachibana, K.; Hasegawa, T. Development of growth hormone and adrenocorticotropic hormone deficiencies in patients with prenatal or perinatal-onset hypothalamic hypopituitarism having invisible or thin pituitary stalk on magnetic resonance imaging. Endocr. J. 2001, 48, 355–362. [Google Scholar] [CrossRef] [Green Version]
- Oberfield, S.E.; Chin, D.; Uli, N.; David, R.; Sklar, C. Endocrine late effects of childhood cancers. J. Pediatr. 1997, 131, S37–S41. [Google Scholar] [CrossRef]
- Benvenga, S.; Campenni, A.; Ruggeri, R.M.; Trimarchi, F. Clinical review 113: Hypopituitarism secondary to head trauma. J. Clin. Endocrinol. Metab. 2000, 85, 1353–1361. [Google Scholar] [CrossRef]
- Einaudi, S.; Bondone, C. The effects of head trauma on hypothalamic-pituitary function in children and adolescents. Curr. Opin. Pediatr. 2007, 19, 465–470. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.; Sun, G.; Tang, Z.; Hou, X. Congenital Isolated ACTH Deficiency Caused by TBX19 Gene Mutation: A Family Report. Front. Pediatr. 2019, 7, 546. [Google Scholar] [CrossRef] [PubMed]
- Broersen, L.H.; Pereira, A.M.; Jorgensen, J.O.; Dekkers, O.M. Adrenal Insufficiency in Corticosteroids Use: Systematic Review and Meta-Analysis. J. Clin. Endocrinol. Metab. 2015, 100, 2171–2180. [Google Scholar] [CrossRef] [PubMed]
- Schlaghecke, R.; Kornely, E.; Santen, R.T.; Ridderskamp, P. The effect of long-term glucocorticoid therapy on pituitary-adrenal responses to exogenous corticotropin-releasing hormone. N. Engl. J. Med. 1992, 326, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Loose, D.S.; Kan, P.B.; Hirst, M.A.; Marcus, R.A.; Feldman, D. Ketoconazole blocks adrenal steroidogenesis by inhibiting cytochrome P450-dependent enzymes. J. Clin. Investig. 1983, 71, 1495–1499. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reimondo, G.; Puglisi, S.; Zaggia, B.; Basile, V.; Saba, L.; Perotti, P.; De Francia, S.; Volante, M.; Zatelli, M.C.; Cannavo, S.; et al. Effects of mitotane on the hypothalamic-pituitary-adrenal axis in patients with adrenocortical carcinoma. Eur. J. Endocrinol. 2017, 177, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Majesko, A.; Darby, J.M. Etomidate and adrenal insufficiency: The controversy continues. Crit. Care 2010, 14, 338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DeCherney, G.S.; DeBold, C.R.; Jackson, R.V.; Sheldon, W.R., Jr.; Island, D.P.; Orth, D.N. Diurnal variation in the response of plasma adrenocorticotropin and cortisol to intravenous ovine corticotropin-releasing hormone. J. Clin. Endocrinol. Metab. 1985, 61, 273–279. [Google Scholar] [CrossRef]
- Rensen, N.; Gemke, R.J.; van Dalen, E.C.; Rotteveel, J.; Kaspers, G.J. Hypothalamic-pituitary-adrenal (HPA) axis suppression after treatment with glucocorticoid therapy for childhood acute lymphoblastic leukaemia. Cochrane Database Syst. Rev. 2017, 11, CD008727. [Google Scholar] [CrossRef] [Green Version]
- Zollner, E.W. Hypothalamic-pituitary-adrenal axis suppression in asthmatic children on inhaled corticosteroids (Part 2)—the risk as determined by gold standard adrenal function tests: A systematic review. Pediatr. Allergy Immunol. 2007, 18, 469–474. [Google Scholar] [CrossRef]
- Newell-Price, J.D.C.; Auchus, R.J. The Adrenal Cortex. Williams Textb. Endocrinol. 2019, 15, 1792. [Google Scholar]
- Bulus, A.D.; Andiran, N.; Kocak, M. Cushing’s syndrome: Hidden risk in usage of topical corticosteroids. J. Pediatr. Endocrinol. Metab. 2014, 27, 977–981. [Google Scholar] [CrossRef] [PubMed]
- Wood Heickman, L.K.; Davallow Ghajar, L.; Conaway, M.; Rogol, A.D. Evaluation of Hypothalamic-Pituitary-Adrenal Axis Suppression following Cutaneous Use of Topical Corticosteroids in Children: A Meta-Analysis. Horm. Res. Paediatr. 2018, 89, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Ahmet, A.; Kim, H.; Spier, S. Adrenal suppression: A practical guide to the screening and management of this under-recognized complication of inhaled corticosteroid therapy. Allergy Asthma Clin. Immunol. 2011, 7, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dolan, L.M.; Kesarwala, H.H.; Holroyde, J.C.; Fischer, T.J. Short-term, high-dose, systemic steroids in children with asthma: The effect on the hypothalamic-pituitary-adrenal axis. J. Allergy Clin. Immunol. 1987, 80, 81–87. [Google Scholar] [CrossRef]
- Vaz, R.; Senior, B.; Morris, M.; Binkiewicz, A. Adrenal effects of beclomethasone inhalation therapy in asthmatic children. J. Pediatr. 1982, 100, 660–662. [Google Scholar] [CrossRef]
- Joseph, R.M.; Hunter, A.L.; Ray, D.W.; Dixon, W.G. Systemic glucocorticoid therapy and adrenal insufficiency in adults: A systematic review. Semin. Arthritis Rheum. 2016, 46, 133–141. [Google Scholar] [CrossRef]
- Aljebab, F.; Choonara, I.; Conroy, S. Systematic review of the toxicity of short-course oral corticosteroids in children. Arch. Dis. Child. 2016, 101, 365–370. [Google Scholar] [CrossRef] [Green Version]
- Spiegel, R.J.; Vigersky, R.A.; Oliff, A.I.; Echelberger, C.K.; Bruton, J.; Poplack, D.G. Adrenal suppression after short-term corticosteroid therapy. Lancet (Lond. Engl.) 1979, 1, 630–633. [Google Scholar] [CrossRef]
- Zora, J.A.; Zimmerman, D.; Carey, T.L.; O’Connell, E.J.; Yunginger, J.W. Hypothalamic-pituitary-adrenal axis suppression after short-term, high-dose glucocorticoid therapy in children with asthma. J. Allergy Clin. Immunol. 1986, 77, 9–13. [Google Scholar] [CrossRef]
- Sinha, A.; Bagga, A. Pulse steroid therapy. Indian J. Pediatr. 2008, 75, 1057–1066. [Google Scholar] [CrossRef] [PubMed]
- Novak, E.; Stubbs, S.S.; Seckman, C.E.; Hearron, M.S. Effects of a single large intravenous dose of methylprednisolone sodium succinate. Clin. Pharmacol. Ther. 1970, 11, 711–717. [Google Scholar] [CrossRef] [PubMed]
- Miura, M.; Ohki, H.; Yoshiba, S.; Ueda, H.; Sugaya, A.; Satoh, M.; Yamagishi, H. Adverse effects of methylprednisolone pulse therapy in refractory Kawasaki disease. Arch. Dis. Child. 2005, 90, 1096–1097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klein-Gitelman, M.S.; Pachman, L.M. Intravenous corticosteroids: Adverse reactions are more variable than expected in children. J. Rheumatol. 1998, 25, 1995–2002. [Google Scholar]
- Manosroi, W.; Phimphilai, M.; Khorana, J.; Atthakomol, P.; Pipanmekaporn, T. Predictive Factors of Adrenal Insufficiency in Outpatients with Indeterminate Serum Cortisol Levels: A Retrospective Study. Med. Kaunas 2020, 56. [Google Scholar] [CrossRef] [Green Version]
- Hollanders, J.J.; van der Voorn, B.; Rotteveel, J.; Finken, M.J.J. Is HPA axis reactivity in childhood gender-specific? A systematic review. Biol. Sex. Differ. 2017, 8, 23. [Google Scholar] [CrossRef]
- Menon, K.; Ward, R.E.; Lawson, M.L.; Gaboury, I.; Hutchison, J.S.; Hebert, P.C.; Canadian Critical Care Trials, G. A prospective multicenter study of adrenal function in critically ill children. Am. J. Respir. Crit. Care Med. 2010, 182, 246–251. [Google Scholar] [CrossRef] [Green Version]
- Foisy, M.M.; Yakiwchuk, E.M.; Chiu, I.; Singh, A.E. Adrenal suppression and Cushing’s syndrome secondary to an interaction between ritonavir and fluticasone: A review of the literature. HIV Med. 2008, 9, 389–396. [Google Scholar] [CrossRef]
- Schacke, H.; Schottelius, A.; Docke, W.D.; Strehlke, P.; Jaroch, S.; Schmees, N.; Rehwinkel, H.; Hennekes, H.; Asadullah, K. Dissociation of transactivation from transrepression by a selective glucocorticoid receptor agonist leads to separation of therapeutic effects from side effects. Proc. Natl. Acad. Sci. USA 2004, 101, 227–232. [Google Scholar] [CrossRef] [Green Version]
- Bornstein, S.R.; Allolio, B.; Arlt, W.; Barthel, A.; Don-Wauchope, A.; Hammer, G.D.; Husebye, E.S.; Merke, D.P.; Murad, M.H.; Stratakis, C.A.; et al. Diagnosis and Treatment of Primary Adrenal Insufficiency: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2016, 101, 364–389. [Google Scholar] [CrossRef] [Green Version]
- Barthel, A.; Willenberg, H.S.; Gruber, M.; Bornstein, S.R. Adrenal Insufficiency. Endocrinol. Adult Pediatr. 2016, 102, 2704. [Google Scholar]
- Allolio, B.; Arlt, W. DHEA treatment: Myth or reality? Trends Endocrinol. Metab. 2002, 13, 288–294. [Google Scholar] [CrossRef]
- Rimsza, M.E. Complications of corticosteroid therapy. Am. J. Dis. Child. 1978, 132, 806–810. [Google Scholar] [CrossRef] [PubMed]
- Joshi, R.R.; Maresh, A. Iatrogenic Cushing’s syndrome and adrenal insufficiency in infants on intranasal dexamethasone drops for nasal obstruction—Case series and literature review. Int. J. Pediatr. Otorhinolaryngol. 2018, 105, 123–126. [Google Scholar] [CrossRef]
- Aso, K.; Izawa, M.; Higuchi, A.; Kotoh, S.; Hasegawa, Y. Stress doses of glucocorticoids cannot prevent progression of all adrenal crises. Clin. Pediatr. Endocrinol. 2009, 18, 23–27. [Google Scholar] [CrossRef] [Green Version]
- Oelkers, W. Hyponatremia and inappropriate secretion of vasopressin (antidiuretic hormone) in patients with hypopituitarism. N. Engl. J. Med. 1989, 321, 492–496. [Google Scholar] [CrossRef]
- Kim, R.J.; Shah, R. Disorders of the Adrenal Gland. Netter Pediatr. 2011, 70, 864. [Google Scholar]
- Todd, G.R.; Acerini, C.L.; Ross-Russell, R.; Zahra, S.; Warner, J.T.; McCance, D. Survey of adrenal crisis associated with inhaled corticosteroids in the United Kingdom. Arch. Dis. Child. 2002, 87, 457–461. [Google Scholar] [CrossRef] [Green Version]
- Wallace, I.; Cunningham, S.; Lindsay, J. The diagnosis and investigation of adrenal insufficiency in adults. Ann. Clin. Biochem. 2009, 46, 351–367. [Google Scholar] [CrossRef] [Green Version]
- Lindholm, J.; Kehlet, H. Re-evaluation of the clinical value of the 30 min ACTH test in assessing the hypothalamic-pituitary-adrenocortical function. Clin. Endocrinol. (Oxf.) 1987, 26, 53–59. [Google Scholar] [CrossRef]
- Stewart, P.M.; Corrie, J.; Seckl, J.R.; Edwards, C.R.; Padfield, P.L. A rational approach for assessing the hypothalamo-pituitary-adrenal axis. Lancet (Lond. Engl.) 1988, 1, 1208–1210. [Google Scholar] [CrossRef]
- White, P.C. Adrenocortical Insufficiency. Nelson Textb. Pediatr. 2020, 593, 4264. [Google Scholar]
- Claahsen-van der Grinten, H.L.; Otten, B.J. Adrenal function: A gold standard test for adrenal insufficiency in children? Nat. Rev. Endocrinol. 2010, 6, 605–606. [Google Scholar] [CrossRef] [PubMed]
- Kazlauskaite, R.; Maghnie, M. Pitfalls in the diagnosis of central adrenal insufficiency in children. Endocr. Dev. 2010, 17, 96–107. [Google Scholar] [CrossRef] [Green Version]
- Montes-Villarreal, J.; Perez-Arredondo, L.A.; Rodriguez-Gutierrez, R.; Gonzalez-Colmenero, A.D.; Solis, R.C.; Gonzalez-Gonzalez, J.G.; Mancillas-Adame, L.G. Serum Morning Cortisol as a Screening Test for Adrenal Insufficiency. Endocr. Pract. 2020, 26, 30–35. [Google Scholar] [CrossRef]
- Karangizi, A.H.K.; Al-Shaghana, M.; Logan, S.; Criseno, S.; Webster, R.; Boelaert, K.; Hewins, P.; Harper, L. Glucocorticoid induced adrenal insufficiency is common in steroid treated glomerular diseases—proposed strategy for screening and management. BMC Nephrol. 2019, 20, 154. [Google Scholar] [CrossRef]
- Schmidt, I.L.; Lahner, H.; Mann, K.; Petersenn, S. Diagnosis of adrenal insufficiency: Evaluation of the corticotropin-releasing hormone test and Basal serum cortisol in comparison to the insulin tolerance test in patients with hypothalamic-pituitary-adrenal disease. J. Clin. Endocrinol. Metab. 2003, 88, 4193–4198. [Google Scholar] [CrossRef] [Green Version]
- Bowden, S.A.; Henry, R. Pediatric Adrenal Insufficiency: Diagnosis, Management, and New Therapies. Int. J. Pediatr. 2018, 2018, 1739831. [Google Scholar] [CrossRef]
- Ospina, N.S.; Al Nofal, A.; Bancos, I.; Javed, A.; Benkhadra, K.; Kapoor, E.; Lteif, A.N.; Natt, N.; Murad, M.H. ACTH Stimulation Tests for the Diagnosis of Adrenal Insufficiency: Systematic Review and Meta-Analysis. J. Clin. Endocrinol. Metab. 2016, 101, 427–434. [Google Scholar] [CrossRef] [Green Version]
- Kazlauskaite, R.; Evans, A.T.; Villabona, C.V.; Abdu, T.A.; Ambrosi, B.; Atkinson, A.B.; Choi, C.H.; Clayton, R.N.; Courtney, C.H.; Gonc, E.N.; et al. Corticotropin tests for hypothalamic-pituitary- adrenal insufficiency: A metaanalysis. J. Clin. Endocrinol. Metab. 2008, 93, 4245–4253. [Google Scholar] [CrossRef] [Green Version]
- Dorin, R.I.; Qualls, C.R.; Crapo, L.M. Diagnosis of adrenal insufficiency. Ann. Intern. Med. 2003, 139, 194–204. [Google Scholar] [CrossRef] [PubMed]
- Gill, H.; Barrowman, N.; Webster, R.; Ahmet, A. Evaluating the Low-Dose ACTH Stimulation Test in Children: Ideal Times for Cortisol Measurement. J. Clin. Endocrinol. Metab. 2019, 104, 4587–4593. [Google Scholar] [CrossRef] [PubMed]
- SRL Inc. Serum Cortisol. Available online: http://test-guide.srl.info/hachioji/test/detail/004100902 (accessed on 25 November 2020).
- SRL Inc. Plasma Cortisol. Available online: http://test-guide.srl.info/hachioji/test/detail/004100906 (accessed on 25 November 2020).
- Siskos, A.P.; Jain, P.; Romisch-Margl, W.; Bennett, M.; Achaintre, D.; Asad, Y.; Marney, L.; Richardson, L.; Koulman, A.; Griffin, J.L.; et al. Interlaboratory Reproducibility of a Targeted Metabolomics Platform for Analysis of Human Serum and Plasma. Anal. Chem. 2017, 89, 656–665. [Google Scholar] [CrossRef] [PubMed]
- Grinspoon, S.K.; Biller, B.M. Clinical review 62: Laboratory assessment of adrenal insufficiency. J. Clin. Endocrinol. Metab. 1994, 79, 923–931. [Google Scholar] [CrossRef]
- Goto, M.; Miyagawa, N.; Kikunaga, K.; Miura, M.; Hasegawa, Y. Single serum cortisol values at 09:00 h can be indices of adrenocortical function in children with Kawasaki disease treated with intravenous immunoglobulin plus prednisolone. Clin. Pediatr. Endocrinol. 2015, 24, 69–75. [Google Scholar] [CrossRef]
- Graber, A.L.; Ney, R.L.; Nicholson, W.E.; Island, D.P.; Liddle, G.W. Natural History of Pituitary-Adrenal Recovery Following Long-Term Suppression with Corticosteroids. J. Clin. Endocrinol. Metab. 1965, 25, 11–16. [Google Scholar] [CrossRef]
- Younes, A.K.; Younes, N.K. Recovery of steroid induced adrenal insufficiency. Transl. Pediatr. 2017, 6, 269–273. [Google Scholar] [CrossRef] [Green Version]
- Vestergaard, T.R.; Juul, A.; Lausten-Thomsen, U.; Lausen, B.; Hjalgrim, H.; Kvist, T.K.; Andersen, E.W.; Schmiegelow, K. Duration of adrenal insufficiency during treatment for childhood acute lymphoblastic leukemia. J. Pediatr. Hematol. Oncol. 2011, 33, 442–449. [Google Scholar] [CrossRef] [Green Version]
- Huber, B.M.; Bolt, I.B.; Sauvain, M.J.; Fluck, C.E. Adrenal insufficiency after glucocorticoid withdrawal in children with rheumatic diseases. Acta Paediatr. 2010, 99, 1889–1893. [Google Scholar] [CrossRef]
- Streck, W.F.; Lockwood, D.H. Pituitary adrenal recovery following short-term suppression with corticosteroids. Am. J. Med. 1979, 66, 910–914. [Google Scholar] [CrossRef]
- Kim, E.B.; Susan, M.B.; Scott, B.; Heddwen, L.B. The Adrenal Medulla & Adrenal Cortex. In Ganong’s Review of Medical Physiology, 24th ed.; McGraw Hill Education: New York, NY, USA, 2012; Chapter 20. [Google Scholar]
- Esteban, N.V.; Loughlin, T.; Yergey, A.L.; Zawadzki, J.K.; Booth, J.D.; Winterer, J.C.; Loriaux, D.L. Daily cortisol production rate in man determined by stable isotope dilution/mass spectrometry. J. Clin. Endocrinol. Metab. 1991, 72, 39–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Linder, B.L.; Esteban, N.V.; Yergey, A.L.; Winterer, J.C.; Loriaux, D.L.; Cassorla, F. Cortisol production rate in childhood and adolescence. J. Pediatr. 1990, 117, 892–896. [Google Scholar] [CrossRef]
- Esteban, N.V.; Yergey, A.L. Cortisol production rates measured by liquid chromatography/mass spectrometry. Steroids 1990, 55, 152–158. [Google Scholar] [CrossRef]
- Kerrigan, J.R.; Veldhuis, J.D.; Leyo, S.A.; Iranmanesh, A.; Rogol, A.D. Estimation of daily cortisol production and clearance rates in normal pubertal males by deconvolution analysis. J. Clin. Endocrinol. Metab. 1993, 76, 1505–1510. [Google Scholar] [CrossRef]
- Ahmet, A.; Brienza, V.; Tran, A.; Lemieux, J.; Aglipay, M.; Barrowman, N.; Duffy, C.; Roth, J.; Jurencak, R. Frequency and Duration of Adrenal Suppression Following Glucocorticoid Therapy in Children With Rheumatic Diseases. Arthritis Care Res. (Hoboken) 2017, 69, 1224–1230. [Google Scholar] [CrossRef]
- Prete, A.; Taylor, A.E.; Bancos, I.; Smith, D.J.; Foster, M.A.; Kohler, S.; Fazal-Sanderson, V.; Komninos, J.; O’Neil, D.M.; Vassiliadi, D.A.; et al. Prevention of Adrenal Crisis: Cortisol Responses to Major Stress Compared to Stress Dose Hydrocortisone Delivery. J. Clin. Endocrinol. Metab. 2020, 105. [Google Scholar] [CrossRef] [Green Version]
- Mass Screening, C.; Japanese Society for Pediatric, E.; Japanese Society for Mass, S.; Ishii, T.; Anzo, M.; Adachi, M.; Onigata, K.; Kusuda, S.; Nagasaki, K.; Harada, S.; et al. Guidelines for diagnosis and treatment of 21-hydroxylase deficiency (2014 revision). Clin. Pediatr. Endocrinol. 2015, 24, 77–105. [Google Scholar] [CrossRef] [Green Version]
- Shulman, D.I.; Palmert, M.R.; Kemp, S.F.; Lawson Wilkins, D.; Therapeutics, C. Adrenal insufficiency: Still a cause of morbidity and death in childhood. Pediatrics 2007, 119, e484–e494. [Google Scholar] [CrossRef] [Green Version]
- Woodcock, T.; Barker, P.; Daniel, S.; Fletcher, S.; Wass, J.A.H.; Tomlinson, J.W.; Misra, U.; Dattani, M.; Arlt, W.; Vercueil, A. Guidelines for the management of glucocorticoids during the peri-operative period for patients with adrenal insufficiency: Guidelines from the Association of Anaesthetists, the Royal College of Physicians and the Society for Endocrinology UK. Anaesthesia 2020, 75, 654–663. [Google Scholar] [CrossRef]
- Wass, J.A.; Arlt, W. How to avoid precipitating an acute adrenal crisis. BMJ 2012, 345, e6333. [Google Scholar] [CrossRef]
- Grossman, A.; Johannsson, G.; Quinkler, M.; Zelissen, P. Therapy of endocrine disease: Perspectives on the management of adrenal insufficiency: Clinical insights from across Europe. Eur. J. Endocrinol. 2013, 169, R165–R175. [Google Scholar] [CrossRef] [PubMed]
- Quinkler, M.; Hahner, S.; Johannsson, G.; Stewart, P.M. Saving lives of patients with adrenal insufficiency: A pan-European initiative? Clin. Endocrinol. (Oxf.) 2014, 80, 319–321. [Google Scholar] [CrossRef] [PubMed]
- Johannsson, G.; Nilsson, A.G.; Bergthorsdottir, R.; Burman, P.; Dahlqvist, P.; Ekman, B.; Engstrom, B.E.; Olsson, T.; Ragnarsson, O.; Ryberg, M.; et al. Improved cortisol exposure-time profile and outcome in patients with adrenal insufficiency: A prospective randomized trial of a novel hydrocortisone dual-release formulation. J. Clin. Endocrinol. Metab. 2012, 97, 473–481. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whitaker, M.; Debono, M.; Huatan, H.; Merke, D.; Arlt, W.; Ross, R.J. An oral multiparticulate, modified-release, hydrocortisone replacement therapy that provides physiological cortisol exposure. Clin. Endocrinol. (Oxf.) 2014, 80, 554–561. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gross, K.L.; Cidlowski, J.A. Tissue-specific glucocorticoid action: A family affair. Trends Endocrinol. Metab. 2008, 19, 331–339. [Google Scholar] [CrossRef] [Green Version]
- Luhder, F.; Reichardt, H.M. Novel Drug Delivery Systems Tailored for Improved Administration of Glucocorticoids. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef]
- Timmermans, S.; Souffriau, J.; Libert, C. A General Introduction to Glucocorticoid Biology. Front. Immunol. 2019, 10, 1545. [Google Scholar] [CrossRef] [Green Version]
- Whirledge, S.; DeFranco, D.B. Glucocorticoid Signaling in Health and Disease: Insights From Tissue-Specific GR Knockout Mice. Endocrinology 2018, 159, 46–64. [Google Scholar] [CrossRef]
| Primary Adrenal Insufficiency | |
| Autoimmune adrenalitis | APS type 1, 2, 3 |
| Infectious adrenalitis | Tuberculosis, HIV-1, cryptococcosis, Treponema pallidum |
| Bilateral adrenal hemorrhage | Meningococcal sepsis |
| Bilateral adrenal infiltration | Primary adrenal lymphoma, hemochromatosis |
| Drug-induced | Ketoconazole, fluconazole, phenobarbital, phenytoin, rifampicin |
| Genetic disorders | CAH, adrenoleukodystrophy, adrenal hypoplasia congenita |
| Secondary adrenal insufficiency | |
| Pituitary tumor | Craniopharyngiomas, adenomas, cysts |
| Pituitary injury | Trauma, surgery, irradiation, pituitary apoplexy |
| Pituitary infiltration | Lymphocytic hypophysitis, tuberculosis, meningitis |
| Genetic disorders | Combined pituitary hormone deficiency, isolated ACTH deficiency |
| Tertiary adrenal insufficiency | |
| Hypothalamic tumor | Craniopharyngiomas, metastasis |
| Hypothalamic injury | Trauma, surgery, irradiation |
| Hypothalamic infiltration | Hemochromatosis, tuberculosis, meningitis |
| Steroid-induced | Systemic, inhalation, topical, intra-articular |
| Other drug-induced | Chlorpromazine, imipramine |
| Abbreviations: ACTH, adrenocorticotropic hormone; APS, autoimmune polyendocrinopathy syndrome; CAH, congenital adrenal hyperplasia. | |
| (Modified from [2] E. Charmandari, et al. Adrenal insufficiency. Lancet (London, England) 2014, 383, 2152–2167.) | |
| Risk Factors | |
| Predisposing diseases | Asthma, leukemia, collagen disease, inflammatory bowel disease |
| Glucocorticoid dose | Doses higher than the physiological equivalent HDC (6–8 mg/m2) and durations exceeding two weeks are associated with SIAI risk. |
| Clinical symptoms | |
| Chronic symptoms | Weakness, fatigue, anorexia, weight loss |
| Acute symptoms | Vomiting, diarrhea, abdominal pain, hypoglycemia, hypotension |
| Diagnostic evaluations | |
| Screening test | Serum cortisol level at 8–9 a.m. with cutoff <10 µg/dL |
| Dynamic test | Low-dose ACTH stimulation test with cutoff <18 µg/dL serum cortisol level 30 min after stimulation |
| Follow-up schedules | Initial testing 1–3 months after the end of pharmacological GC followed by dynamic testing every 3–6 months until recovery |
| Glucocorticoid supplementation | |
| Physiological replacement dose | po HDC 6–8 mg/m2/day unless serum cortisol >5 µg/dL on morning testing |
| Stress dose | im/iv HDC 50–100 mg/m2/day according to the physical stress unless peak cortisol >18 µg/dL on dynamic testing |
| Abbreviations: ACTH, adrenocorticotropic hormone; GC, glucocorticoid; HDC, hydrocortisone; im, intramuscular; iv, intravenous; po, orally; SIAI, steroid-induced iatrogenic adrenal insufficiency. | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akahoshi, S.; Hasegawa, Y. Steroid-Induced Iatrogenic Adrenal Insufficiency in Children: A Literature Review. Endocrines 2020, 1, 125-137. https://doi.org/10.3390/endocrines1020012
Akahoshi S, Hasegawa Y. Steroid-Induced Iatrogenic Adrenal Insufficiency in Children: A Literature Review. Endocrines. 2020; 1(2):125-137. https://doi.org/10.3390/endocrines1020012
Chicago/Turabian StyleAkahoshi, Shogo, and Yukihiro Hasegawa. 2020. "Steroid-Induced Iatrogenic Adrenal Insufficiency in Children: A Literature Review" Endocrines 1, no. 2: 125-137. https://doi.org/10.3390/endocrines1020012
APA StyleAkahoshi, S., & Hasegawa, Y. (2020). Steroid-Induced Iatrogenic Adrenal Insufficiency in Children: A Literature Review. Endocrines, 1(2), 125-137. https://doi.org/10.3390/endocrines1020012





