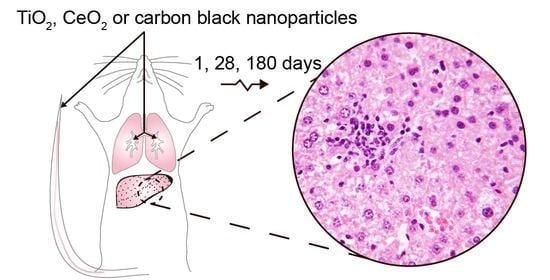
Effect on Mouse Liver Morphology of CeO2, TiO2 and Carbon Black Nanoparticles Translocated from Lungs or Deposited Intravenously
Abstract
1. Introduction
2. Materials and Methods
2.1. Characterization of Nanoparticles and Nanoparticle Suspensions
2.2. Animal Handling and Exposure to Nanoparticles
2.3. Selection of the Dose
2.4. Necropsy and Sample Collection
2.5. Hematology Parameters and Activity of Transaminases and Alkaline Phosphatase
2.6. Histological Examination
2.7. Detection of Foreign Material in Lung and Liver by Enhanced Darkfield and Brightfield Microscopy
2.8. Statistical Analysis
3. Results
3.1. General Appearance, Body Weight, Feed Intake and Liver Weight
3.2. Hematology
3.3. Activities of Enzymes Indicative of Liver Injury
3.4. Lung Histology
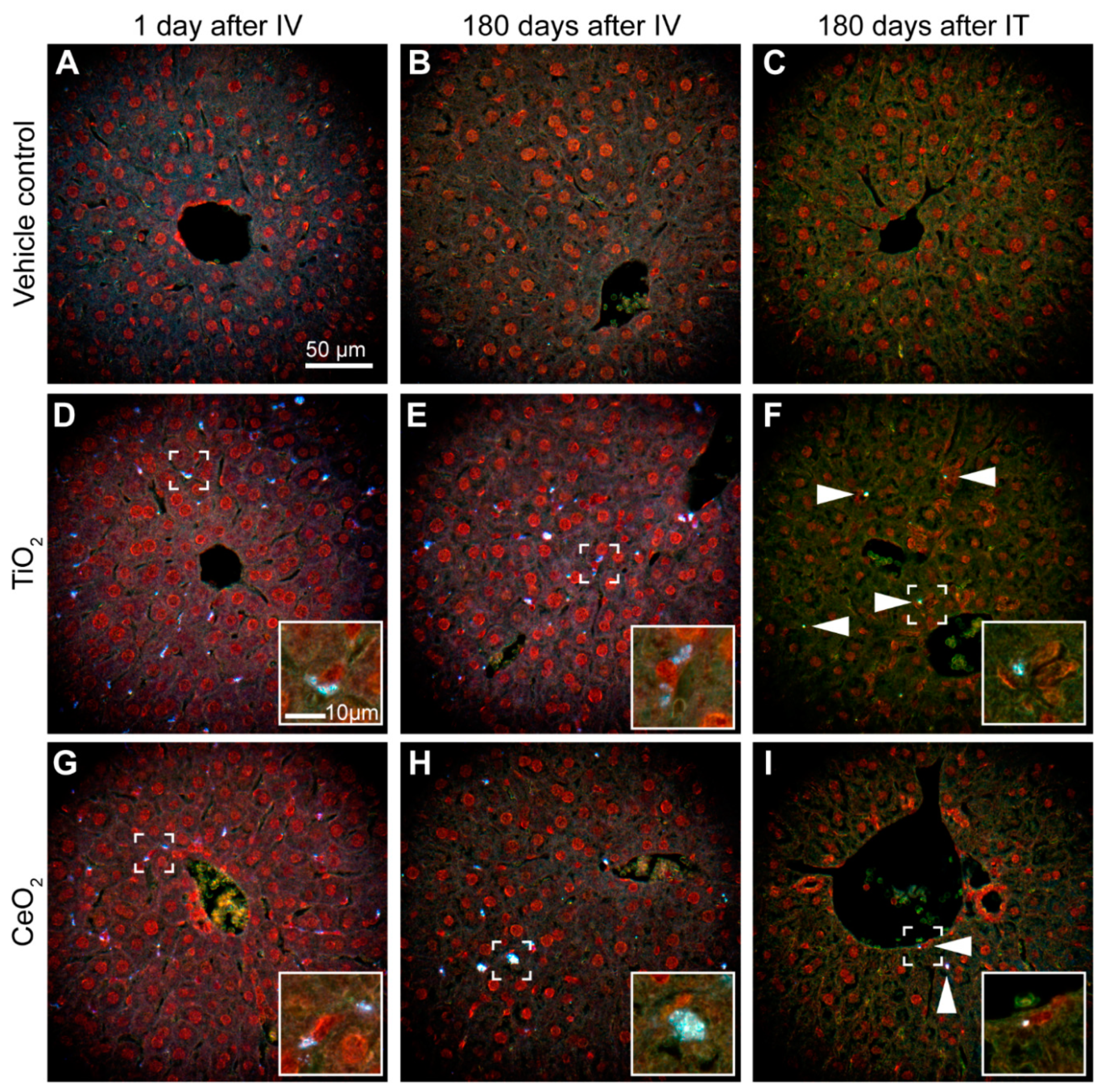
3.5. Localization of Nanoparticles in Liver
3.5.1. Localization in Liver after Intravenous Exposure
3.5.2. Localization in Liver after Intratracheal Instillation
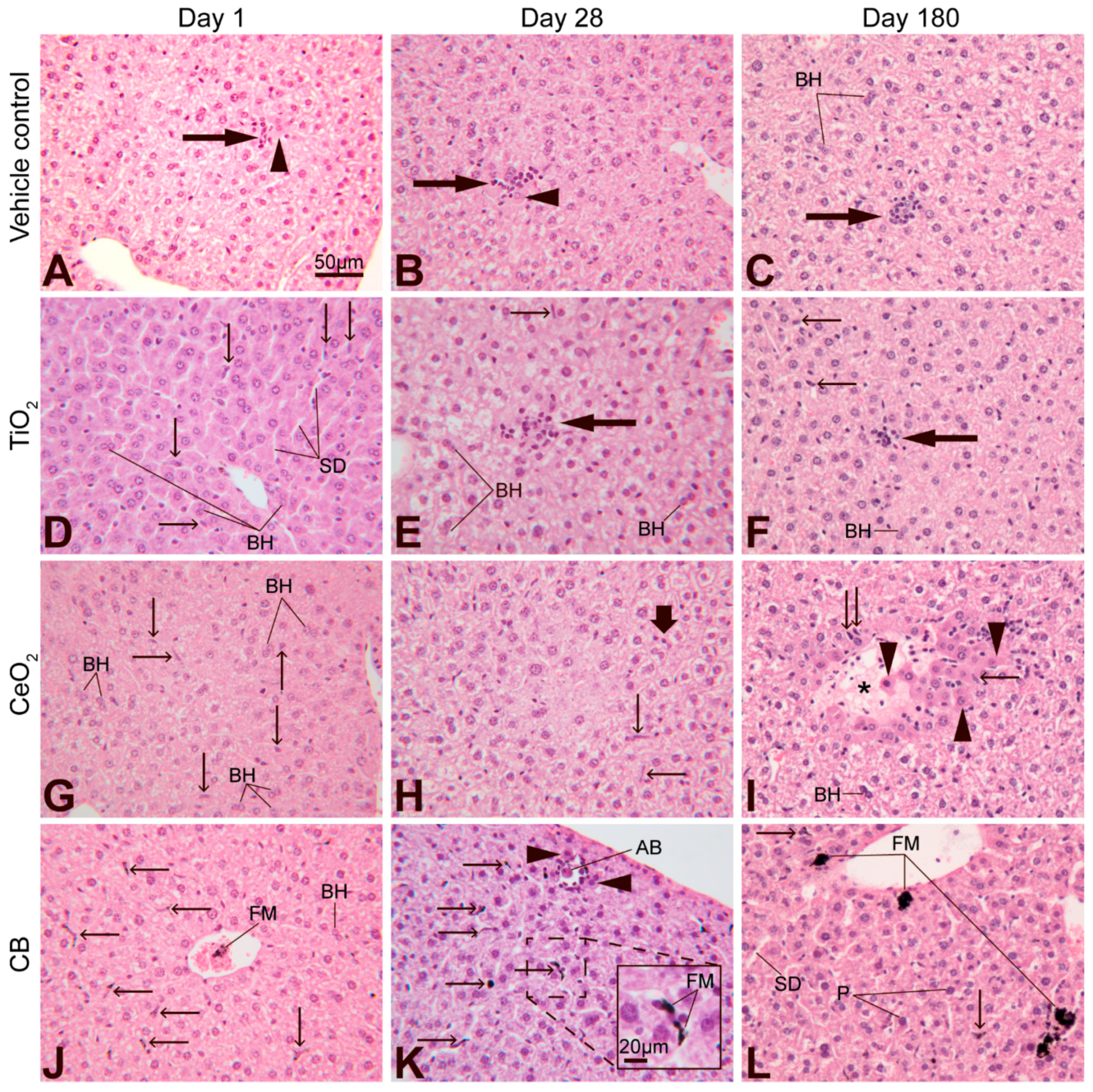
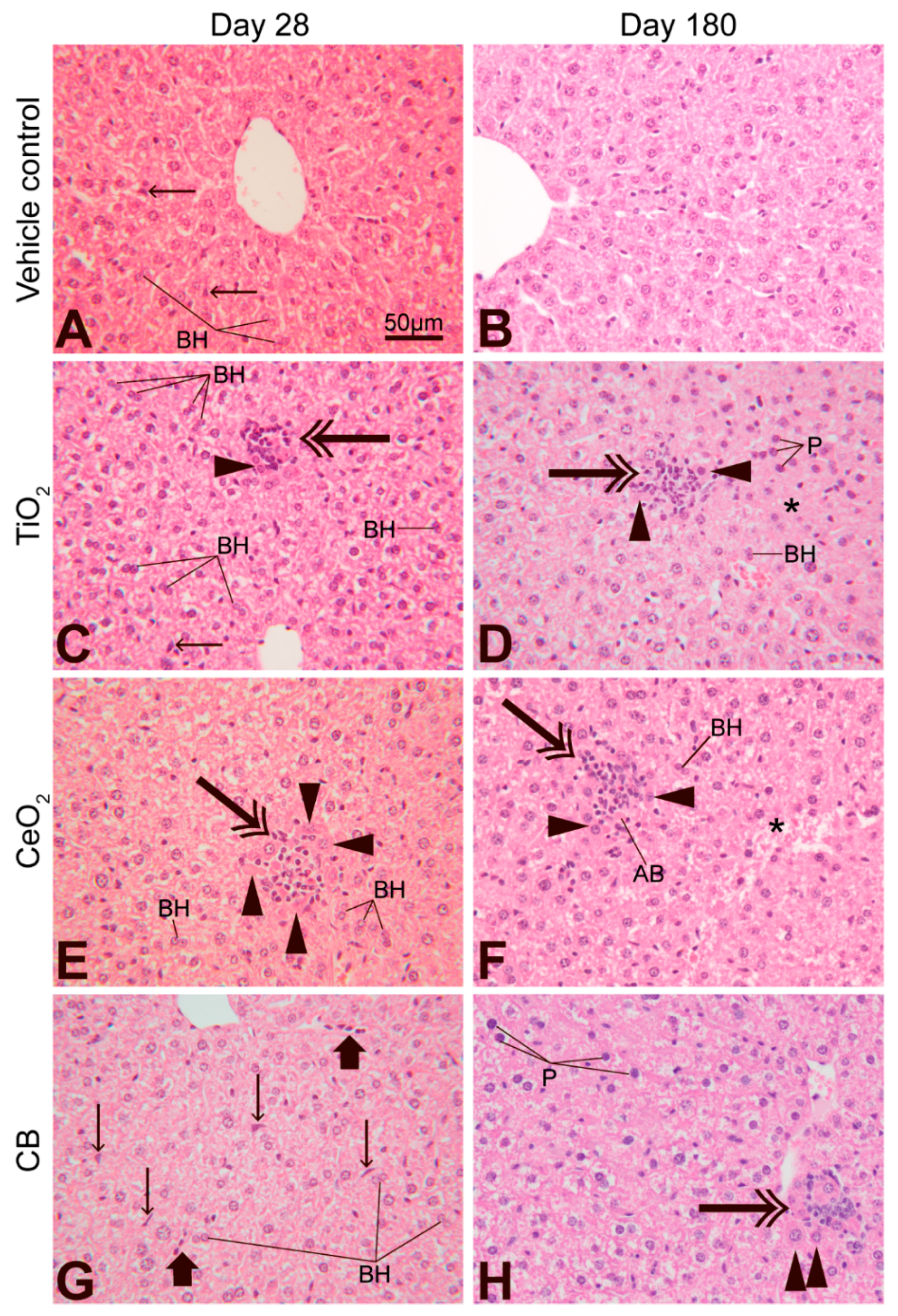
3.6. Effect of Nanoparticle Exposure on Liver Histology
4. Discussion
4.1. Relevance of the Exposure by Intratracheal Instillation
4.2. Liver Burden of Nanoparticles
4.3. Body Weight, Feed Intake, Hematology, Transaminases and Alkaline Phosphatase, and Liver Weight
4.4. Liver Histology
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Oberdörster, G.; Oberdörster, E.; Oberdörster, J. Nanotoxicology: An Emerging Discipline Evolving from Studies of Ultrafine Particles. Environ. Health Perspect. 2005, 113, 823–839. [Google Scholar] [CrossRef]
- Stone, V.; Miller, M.R.; Clift, M.; Elder, A.; Mills, N.L.; Møller, P.; Schins, R.P.; Vogel, U.; Kreyling, W.; Jensen, K.A.; et al. Nanomaterials Versus Ambient Ultrafine Particles: An Opportunity to Exchange Toxicology Knowledge. Environ. Health Perspect. 2017, 125, 106002. [Google Scholar] [CrossRef]
- Semmler-Behnke, M.; Takenaka, S.; Fertsch, S.; Wenk, A.; Seitz, J.; Mayer, P.; Oberdörster, G.; Kreyling, W. Efficient Elimination of Inhaled Nanoparticles from the Alveolar Region: Evidence for Interstitial Uptake and Subsequent Reentrainment onto Airways Epithelium. Environ. Health Perspect. 2007, 115, 728–733. [Google Scholar] [CrossRef] [PubMed]
- Oberdörster, G.; Sharp, Z.; Atudorei, V.; Elder, A.; Gelein, R.; Lunts, A.; Kreyling, W.; Cox, C. Extrapulmonary translocation of ultrafine carbon particles following whole-body inhalation exposure of rats. J. Toxicol. Environ. Health Part A 2002, 65, 1531–1543. [Google Scholar] [CrossRef] [PubMed]
- Husain, M.; Wu, D.; Saber, A.T.; Decan, N.; Jacobsen, N.R.; Williams, A.; Yauk, C.; Wallin, H.; Vogel, U.; Halappanavar, S. Intratracheally instilled titanium dioxide nanoparticles translocate to heart and liver and activate complement cascade in the heart of C57BL/6 mice. Nanotoxicology 2015, 9, 1013–1022. [Google Scholar] [CrossRef] [PubMed]
- Kreyling, W.G.; Semmler-Behnke, M.; Seitz, J.; Scymczak, W.; Wenk, A.; Mayer, P.; Takenaka, S.; Oberdörster, G. Size dependence of the translocation of inhaled iridium and carbon nanoparticle aggregates from the lung of rats to the blood and secondary target organs. Inhal. Toxicol. 2009, 21, 55–60. [Google Scholar] [CrossRef]
- Geiser, M.; Kreyling, W.G. Deposition and biokenetics of inhaled nanoparticles. Part. Fibre Toxicol. 2010, 7, 2–19. [Google Scholar] [CrossRef]
- Lipka, J.; Semmler-Behnke, M.; Sperling, R.; Wenk, A.; Takenaka, S.; Schleh, C.; Kissel, T.; Parak, W.J.; Kreyling, W.G. Biodistribution of PEG-modified gold nanoparticles following intratracheal instillation and intravenous injection. Biomaterials 2010, 31, 6574–6581. [Google Scholar] [CrossRef]
- Sadauskas, E.; Wallin, H.; Stoltenberg, M.; Vogel, U.; Doering, P.; Larsen, A.; Danscher, G. Kupffer cells are central in the removal of nanoparticles from the organism. Part. Fibre Toxicol. 2007, 4, 10. [Google Scholar] [CrossRef]
- Sadauskas, E.; Danscher, G.; Stoltenberg, M.; Vogel, U.; Larsen, A.; Wallin, H. Protracted elimination of gold nanoparticles from mouse liver. Nanomed. Nanotechnol. Biol. Med. 2009, 5, 162–169. [Google Scholar] [CrossRef]
- Kermanizadeh, A.; Powell, L.G.; Stone, V. A review of hepatic nanotoxicology—Summation of recent findings and considerations for the next generation of study designs. J. Toxicol. Environ. Health Part B 2020, 23, 137–176. [Google Scholar] [CrossRef]
- Sun, T.; Kang, Y.; Liu, J.; Zhang, Y.; Ou, L.; Liu, X.; Lai, R.; Shao, L. Nanomaterials and hepatic disease: Toxicokinetics, disease types, intrinsic mechanisms, liver susceptibility, and influencing factors. J. Nanobiotechnol. 2021, 19, 1–23. [Google Scholar] [CrossRef]
- Lipshutz, G.S.; Brennan, T.V.; Warren, R.S. Thorotrast-induced liver neoplasia: A collective review. J. Am. Coll. Surg. 2002, 195, 713–718. [Google Scholar] [CrossRef]
- Andersson, M.; Vyberg, M.; Visfeldt, J.; Carstensen, B.; Storm, H.H. Primary Liver Tumors among Danish Patients Exposed to Thorotrast. Radiat. Res. 1994, 137, 262. [Google Scholar] [CrossRef]
- Hansen, S.F.; Heggelund, L.R.; Besora, P.R.; Mackevica, A.; Boldrin, A.; Baun, A. Nanoproducts—What is actually available to European consumers? Environ. Sci. Nano 2015, 3, 169–180. [Google Scholar] [CrossRef]
- EFSA Panel on Food Additives and Flavourings. Safety assessment of titanium dioxide (E171) as a food additive. EFSA J. 2021, 19, 6585. [Google Scholar] [CrossRef]
- Rzigalinski, B.A.; Carfagna, C.S.; Ehrich, M. Cerium oxide nanoparticles in neuroprotection and considerations for efficacy and safety. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2017, 9, e1444. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Sayle, D.C.; Wang, Z.L.; Paras, M.S.; Santora, B.; Sutorik, A.C.; Sayle, T.X.T.; Yang, Y.; Ding, Y.; Wang, X.; et al. Converting Ceria Polyhedral Nanoparticles into Single-Crystal Nanospheres. Science 2006, 312, 1504–1508. [Google Scholar] [CrossRef] [PubMed]
- Cassee, F.R.; Van Balen, E.C.; Singh, C.; Green, D.; Muijser, H.; Weinstein, J.; Dreher, K. Exposure, Health and Ecological Effects Review of Engineered Nanoscale Cerium and Cerium Oxide Associated with its Use as a Fuel Additive. Crit. Rev. Toxicol. 2011, 41, 213–229. [Google Scholar] [CrossRef] [PubMed]
- Casals, G.; Perramón, M.; Casals, E.; Portolés, I.; Fernández-Varo, G.; Morales-Ruiz, M.; Puntes, V.; Jiménez, W. Cerium Oxide Nanoparticles: A New Therapeutic Tool in Liver Diseases. Antioxidants 2021, 10, 660. [Google Scholar] [CrossRef] [PubMed]
- Manne, N.D.; Arvapalli, R.; Graffeo, V.A.; Bandarupalli, V.V.K.; Shokuhfar, T.; Patel, S.; Rice, K.; Ginjupalli, G.K.; Blough, E.R. Prophylactic Treatment with Cerium Oxide Nanoparticles Attenuate Hepatic Ischemia Reperfusion Injury in Sprague Dawley Rats. Cell. Physiol. Biochem. 2017, 42, 1837–1846. [Google Scholar] [CrossRef]
- Jacobsen, N.R.; Saber, A.T.; White, P.; Møller, P.; Pojana, G.; Vogel, U.; Loft, S.; Gingerich, J.; Soper, L.; Douglas, G.R.; et al. Increased mutant frequency by carbon black, but not quartz, in thelacZ andcII transgenes of muta™mouse lung epithelial cells. Environ. Mol. Mutagen. 2007, 48, 451–461. [Google Scholar] [CrossRef]
- Jacobsen, N.R.; Pojana, G.; White, P.; Møller, P.; Cohn, C.A.; Korsholm, K.S.; Vogel, U.; Marcomini, A.; Loft, S.; Wallin, H. Genotoxicity, cytotoxicity, and reactive oxygen species induced by single-walled carbon nanotubes and C60fullerenes in the FE1-Muta™Mouse lung epithelial cells. Environ. Mol. Mutagen. 2008, 49, 476–487. [Google Scholar] [CrossRef] [PubMed]
- Modrzynska, J.; Berthing, T.; Ravn-Haren, G.; Jacobsen, N.R.; Weydahl, I.K.; Loeschner, K.; Mortensen, A.; Saber, A.T.; Vogel, U. Primary genotoxicity in the liver following pulmonary exposure to carbon black nanoparticles in mice. Part. Fibre Toxicol. 2018, 15, 1–12. [Google Scholar] [CrossRef]
- Wallin, H.; Kyjovska, Z.O.; Poulsen, S.S.; Jacobsen, N.R.; Saber, A.T.; Bengtson, S.; Jackson, P.; Vogel, U. Surface modification does not influence the genotoxic and inflammatory effects of TiO2 nanoparticles after pulmonary exposure by instillation in mice. Mutagenesis 2017, 32, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Saber, A.T.; Jacobsen, N.R.; Mortensen, A.; Szarek, J.; Jackson, P.; Madsen, A.M.; Jensen, K.A.; Koponen, I.K.; Brunborg, G.; Gützkow, K.B.; et al. Nanotitanium dioxide toxicity in mouse lung is reduced in sanding dust from paint. Part. Fibre Toxicol. 2012, 9, 4. [Google Scholar] [CrossRef] [PubMed]
- Bourdon, J.A.; Saber, A.T.; Jacobsen, N.R.; Jensen, K.A.; Madsen, A.M.; Lamson, J.S.; Wallin, H.; Møller, P.; Loft, S.; Yauk, C.; et al. Carbon black nanoparticle instillation induces sustained inflammation and genotoxicity in mouse lung and liver. Part. Fibre Toxicol. 2012, 9, 5. [Google Scholar] [CrossRef]
- Warheit, D.B.; Webb, T.R.; Reed, K.L.; Frerichs, S.; Sayes, C. Pulmonary toxicity study in rats with three forms of ultrafine-TiO2 particles: Differential responses related to surface properties. Toxicology 2007, 230, 90–104. [Google Scholar] [CrossRef]
- Liu, R.; Yin, L.; Pu, Y.; Liang, G.; Zhang, J.; Su, Y.; Xiao, Z.; Ye, B. Pulmonary toxicity induced by three forms of titanium dioxide nanoparticles via intra-tracheal instillation in rats. Prog. Nat. Sci. 2009, 19, 573–579. [Google Scholar] [CrossRef]
- Kwon, S.; Yang, Y.-S.; Yang, H.-S.; Lee, J.; Kang, M.-S.; Lee, B.-S.; Lee, K.; Song, C.-W. Nasal and Pulmonary Toxicity of Titanium Dioxide Nanoparticles in Rats. Toxicol. Res. 2012, 28, 217–224. [Google Scholar] [CrossRef]
- Keller, J.; Wohlleben, W.; Ma-Hock, L.; Strauss, V.; Gröters, S.; Küttler, K.; Wiench, K.; Herden, C.; Oberdörster, G.; Van Ravenzwaay, B.; et al. Time course of lung retention and toxicity of inhaled particles: Short-term exposure to nano-Ceria. Arch. Toxicol. 2014, 88, 2033–2059. [Google Scholar] [CrossRef]
- Saputra, D.; Yoon, J.-H.; Park, H.; Heo, Y.; Yang, H.; Lee, E.J.; Lee, S.; Song, C.-W.; Lee, K. Inhalation of Carbon Black Nanoparticles Aggravates Pulmonary Inflammation in Mice. Toxicol. Res. 2014, 30, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Yoshiura, Y.; Izumi, H.; Oyabu, T.; Hashiba, M.; Kambara, T.; Mizuguchi, Y.; Lee, B.W.; Okada, T.; Tomonaga, T.; Myojo, T.; et al. Pulmonary toxicity of well-dispersed titanium dioxide nanoparticles following intratracheal instillation. J. Nanoparticle Res. 2015, 17, 241. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, Y.; Izumi, H.; Yoshiura, Y.; Tomonaga, T.; Oyabu, T.; Myojo, T.; Kawai, K.; Yatera, K.; Shimada, M.; Kubo, M.; et al. Pulmonary toxicity of well-dispersed cerium oxide nanoparticles following intratracheal instillation and inhalation. J. Nanoparticle Res. 2015, 17, 1–16. [Google Scholar] [CrossRef]
- Schwotzer, D.; Ernst, H.; Schaudien, D.; Kock, H.; Pohlmann, G.; Dasenbrock, C.; Creutzenberg, O. Effects from a 90-day inhalation toxicity study with cerium oxide and barium sulfate nanoparticles in rats. Part. Fibre Toxicol. 2017, 14, 23. [Google Scholar] [CrossRef] [PubMed]
- Danielsen, P.H.; Knudsen, K.B.; Štrancar, J.; Umek, P.; Koklič, T.; Garvas, M.; Vanhala, E.; Savukoski, S.; Ding, Y.; Madsen, A.M.; et al. Effects of physicochemical properties of TiO2 nanomaterials for pulmonary inflammation, acute phase response and alveolar proteinosis in intratracheally exposed mice. Toxicol. Appl. Pharmacol. 2020, 386, 114830. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.Y.; Zhao, H.; Mercer, R.R.; Barger, M.; Rao, M.; Meighan, T.; Schwegler-Berry, D.; Castranova, V.; Ma, J.K. Cerium oxide nanoparticle-induced pulmonary inflammation and alveolar macrophage functional change in rats. Nanotoxicology 2010, 5, 312–325. [Google Scholar] [CrossRef] [PubMed]
- Heringa, M.B.; Peters, R.J.B.; Bleys, R.L.A.W.; Van Der Lee, M.K.; Tromp, P.C.; Van Kesteren, P.C.E.; Van Eijkeren, J.C.H.; Undas, A.; Oomen, A.G.; Bouwmeester, H. Detection of titanium particles in human liver and spleen and possible health implications. Part. Fibre Toxicol. 2018, 15, 1–9. [Google Scholar] [CrossRef]
- Peters, R.J.B.; Oomen, A.G.; Van Bemmel, G.; Van Vliet, L.; Undas, A.; Munniks, S.; Bleys, R.L.A.W.; Tromp, P.C.; Brand, W.; Van Der Lee, M. Silicon dioxide and titanium dioxide particles found in human tissues. Nanotoxicology 2020, 14, 420–432. [Google Scholar] [CrossRef]
- Guillard, A.; Gaultier, E.; Cartier, C.; Devoille, L.; Noireaux, J.; Chevalier, L.; Morin, M.; Grandin, F.; Lacroix, M.Z.; Coméra, C.; et al. Basal Ti level in the human placenta and meconium and evidence of a materno-foetal transfer of food-grade TiO2 nanoparticles in an ex vivo placental perfusion model. Part. Fibre Toxicol. 2020, 17, 51. [Google Scholar] [CrossRef]
- Heringa, M.B.; Geraets, L.; Van Eijkeren, J.C.H.; Vandebriel, R.; De Jong, W.H.; Oomen, A.G. Risk assessment of titanium dioxide nanoparticles via oral exposure, including toxicokinetic considerations. Nanotoxicology 2016, 10, 1515–1525. [Google Scholar] [CrossRef]
- Modrzynska, J.; Berthing, T.; Ravn-Haren, G.; Kling, K.; Mortensen, A.; Rasmussen, R.R.; Larsen, E.H.; Saber, A.T.; Vogel, U.; Loeschner, K. In vivo-induced size transformation of cerium oxide nanoparticles in both lung and liver does not affect long-term hepatic accumulation following pulmonary exposure. PLoS ONE 2018, 13, e0202477. [Google Scholar] [CrossRef]
- Hougaard, K.S.; Jackson, P.; Jensen, K.A.; Sloth, J.J.; Löschner, K.; Larsen, E.H.; Birkedal, R.K.; Vibenholt, A.; Boisen, A.-M.Z.; Wallin, H.; et al. Effects of prenatal exposure to surface-coated nanosized titanium dioxide (UV-Titan). A study in mice. Part. Fibre Toxicol. 2010, 7, 16. [Google Scholar] [CrossRef]
- Halappanavar, S.; Saber, A.T.; Decan, N.; Jensen, K.A.; Wu, D.; Jacobsen, N.R.; Guo, C.; Rogowski, J.; Koponen, I.K.; Levin, M.; et al. Transcriptional profiling identifies physicochemical properties of nanomaterials that are determinants of the in vivo pulmonary response. Environ. Mol. Mutagen. 2015, 56, 245–264. [Google Scholar] [CrossRef]
- Saber, A.T.; Jensen, K.A.; Jacobsen, N.R.; Birkedal, R.; Mikkelsen, L.; Møller, P.; Loft, S.; Wallin, H.; Vogel, U. Inflammatory and genotoxic effects of nanoparticles designed for inclusion in paints and lacquers. Nanotoxicology 2011, 6, 453–471. [Google Scholar] [CrossRef]
- Graham, U.M.; Tseng, M.T.; Jasinski, J.B.; Yokel, R.; Unrine, J.; Davis, B.H.; Dozier, A.K.; Hardas, S.S.; Sultana, R.; Grulke, E.A.; et al. In Vivo Processing of Ceria Nanoparticles inside Liver: Impact on Free-Radical Scavenging Activity and Oxidative Stress. ChemPlusChem 2014, 79, 1083–1088. [Google Scholar] [CrossRef] [PubMed]
- Kyjovska, Z.O.; Jacobsen, N.R.; Saber, A.T.; Bengtson, S.; Jackson, P.; Wallin, H.; Vogel, U. DNA damage following pulmonary exposure by instillation to low doses of carbon black (Printex 90) nanoparticles in mice. Environ. Mol. Mutagen. 2014, 56, 41–49. [Google Scholar] [CrossRef]
- Lide, R.C. Handbook of Chemistry and Physics, 84th ed.; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Levin, M.; Rojas, E.; Vanhala, E.; Vippola, M.; Liguori, B.; Kling, K.I.; Koponen, I.K.; Mølhave, K.; Tuomi, T.; Gregurec, D.; et al. Influence of relative humidity and physical load during storage on dustiness of inorganic nanomaterials: Implications for testing and risk assessment. J. Nanoparticle Res. 2015, 17, 337. [Google Scholar] [CrossRef]
- Saber, A.T.; Bornholdt, J.; Dybdahl, M.; Sharma, A.K.; Loft, S.; Vogel, U.; Wallin, H. Tumor necrosis factor is not required for particle-induced genotoxicity and pulmonary inflammation. Arch. Toxicol. 2004, 79, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, S.S.; Saber, A.T.; Williams, A.; Andersen, O.; Købler, C.; Atluri, R.; Pozzebon, M.E.; Mucelli, S.P.; Simion, M.; Rickerby, D.; et al. MWCNTs of different physicochemical properties cause similar inflammatory responses, but differences in transcriptional and histological markers of fibrosis in mouse lungs. Toxicol. Appl. Pharmacol. 2015, 284, 16–32. [Google Scholar] [CrossRef]
- Mikkelsen, L.; Sheykhzade, M.; Jensen, K.A.; Saber, A.T.; Jacobsen, N.R.; Vogel, U.; Wallin, H.; Loft, S.; Møller, P. Modest effect on plaque progression and vasodilatory function in atherosclerosis-prone mice exposed to nanosized TiO2. Part. Fibre Toxicol. 2011, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, U.; Fuhst, R.; Rittinghausen, S.; Creutzenberg, O.; Bellmann, B.; Koch, W.; Levsen, K. Chronic Inhalation Exposure of Wistar Rats and two Different Strains of Mice to Diesel Engine Exhaust, Carbon Black, and Titanium Dioxide. Inhal. Toxicol. 1995, 7, 533–556. [Google Scholar] [CrossRef]
- Haschek, W.M.; Rousseaux, C.G.; Wallig, M.A. Fundamentals of Toxicologic Pathology, 2nd ed.; Academic Press: London, UK, 2010. [Google Scholar] [CrossRef]
- Renne, R.; Brix, A.; Harkema, J.; Herbert, R.; Kittel, B.; Lewis, D.; March, T.; Nagano, K.; Pino, M.; Rittinghausen, S.; et al. Proliferative and Nonproliferative Lesions of the Rat and Mouse Respiratory Tract. Toxicol. Pathol. 2009, 37, 5S–73S. [Google Scholar] [CrossRef] [PubMed]
- Thoolen, B.; Maronpot, R.R.; Harada, T.; Nyska, A.; Rousseaux, C.; Nolte, T.; Malarkey, D.E.; Kaufmann, W.; Küttler, K.; Deschl, U.; et al. Proliferative and Nonproliferative Lesions of the Rat and Mouse Hepatobiliary System. Toxicol. Pathol. 2010, 38, 5S–81S. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Shi, H.; Ruth, M.; Yu, H.; Lazar, L.; Zou, B.; Yang, C.; Wu, A.; Zhao, J. Acute Toxicity of Intravenously Administered Titanium Dioxide Nanoparticles in Mice. PLoS ONE 2013, 8, e70618. [Google Scholar] [CrossRef] [PubMed]
- Aalapati, S.; Ganapathy, S.; Manapuram, S.; Anumolu, G.; Prakya, B.M. Toxicity and bio-accumulation of inhaled cerium oxide nanoparticles in CD1 mice. Nanotoxicology 2013, 8, 786–798. [Google Scholar] [CrossRef] [PubMed]
- Srinivas, A.; Rao, P.J.; Selvam, G.S.; Murthy, P.B.; Reddy, P.N. Acute inhalation toxicity of cerium oxide nanoparticles in rats. Toxicol. Lett. 2011, 205, 105–115. [Google Scholar] [CrossRef]
- Gosens, I.; Mathijssen, L.E.; Bokkers, B.G.; Muijser, H.; Cassee, F.R. Comparative hazard identification of nano- and micro-sized cerium oxide particles based on 28-day inhalation studies in rats. Nanotoxicology 2013, 8, 643–653. [Google Scholar] [CrossRef] [PubMed]
- Okada, T.; Ogami, A.; Lee, B.W.; Kadoya, C.; Oyabu, T.; Myojo, T. Pulmonary responses in rat lungs after intratracheal instillation of 4 crystal forms of titanium dioxide nanoparticles. J. Occup. Health 2016, 58, 602–611. [Google Scholar] [CrossRef]
- Rahman, L.; Wu, N.; Johnston, M.; William, A.; Halappanavar, S.; Williams, A. Toxicogenomics analysis of mouse lung responses following exposure to titanium dioxide nanomaterials reveal their disease potential at high doses. Mutagenesis 2016, 32, 59–76. [Google Scholar] [CrossRef]
- Husain, M.; Saber, A.T.; Guo, C.; Jacobsen, N.R.; Jensen, K.A.; Yauk, C.L.; Williams, A.; Vogel, U.; Wallin, H.; Halappanavar, S. Pulmonary instillation of low doses of titanium dioxide nanoparticles in mice leads to particle retention and gene expression changes in the absence of inflammation. Toxicol. Appl. Pharmacol. 2013, 269, 250–262. [Google Scholar] [CrossRef] [PubMed]
- Semmler, M.; Seitz, J.; Erbe, F.; Mayer, P.; Heyder, J.; Oberdörster, G.; Kreyling, W.G. Long-Term Clearance Kinetics of Inhaled Ultrafine Insoluble Iridium Particles from the Rat Lung, Including Transient Translocation into Secondary Organs. Inhal. Toxicol. 2004, 16, 453–459. [Google Scholar] [CrossRef]
- Kreyling, W.; Holzwarth, U.; Haberl, N.; Kozempel, J.; Hirn, S.; Wenk, A.; Schleh, C.; Schäffler, M.; Lipka, J.; Semmler-Behnke, M.; et al. Quantitative biokinetics of titanium dioxide nanoparticles after intravenous injection in rats: Part 1. Nanotoxicology 2017, 11, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Kreyling, W.G.; Holzwarth, U.; Haberl, N.; Kozempel, J.; Wenk, A.; Hirn, S.; Schleh, C.; Schäffler, M.; Lipka, J.; Semmler-Behnke, M.; et al. Quantitative biokinetics of titanium dioxide nanoparticles after intratracheal instillation in rats: Part 3. Nanotoxicology 2017, 11, 454–464. [Google Scholar] [CrossRef]
- Gaté, L.; Knudsen, K.B.; Seidel, C.; Berthing, T.; Chézeau, L.; Jacobsen, N.R.; Valentino, S.; Wallin, H.; Bau, S.; Wolff, H.; et al. Pulmonary toxicity of two different multi-walled carbon nanotubes in rat: Comparison between intratracheal instillation and inhalation exposure. Toxicol. Appl. Pharmacol. 2019, 375, 17–31. [Google Scholar] [CrossRef]
- Kinaret, P.; Ilves, M.; Fortino, V.; Rydman, E.; Karisola, P.; Lähde, A.; Koivisto, A.J.; Jokiniemi, J.; Wolff, H.; Savolainen, K.; et al. Inhalation and Oropharyngeal Aspiration Exposure to Rod-Like Carbon Nanotubes Induce Similar Airway Inflammation and Biological Responses in Mouse Lungs. ACS Nano 2017, 11, 291–303. [Google Scholar] [CrossRef]
- Baisch, B.L.; Corson, N.M.; Wade-Mercer, P.; Gelein, R.; Kennell, A.J.; Oberdörster, G.; Elder, A. Equivalent titanium dioxide nanoparticle deposition by intratracheal instillation and whole body inhalation: The effect of dose rate on acute respiratory tract inflammation. Part. Fibre Toxicol. 2014, 11, 5. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, G.; Tiancheng, W.; Yu, H.; Wang, T.; Ma, Y.; Jiangxue, W.; Gao, Y.; Li, Y.-F.; Sun, J. Acute toxicity and biodistribution of different sized titanium dioxide particles in mice after oral administration. Toxicol. Lett. 2007, 168, 176–185. [Google Scholar] [CrossRef]
- Tseng, M.T.; Fu, Q.; Lor, K.; Fernandez-Botran, G.R.; Deng, Z.-B.; Graham, U.; Butterfield, D.A.; Grulke, E.A.; Yokel, R. Persistent Hepatic Structural Alterations Following Nanoceria Vascular Infusion in the Rat. Toxicol. Pathol. 2014, 42, 984–996. [Google Scholar] [CrossRef] [PubMed]
- Park, E.-J.; Park, Y.-K.; Park, K.-S. Acute Toxicity and Tissue Distribution of Cerium Oxide Nanoparticles by a Single Oral Administration in Rats. Toxicol. Res. 2009, 25, 79–84. [Google Scholar] [CrossRef]
- Nalabotu, S.K.; Kolli, M.B.; Triest, W.; Ma, J.Y.; Manne, N.D.; Katta, A.; Addagarla, H.; Rice, K.; Blough, E.R. Intratracheal instillation of cerium oxide nanoparticles induces hepatic toxicity in male Sprague-Dawley rats. Int. J. Nanomed. 2011, 6, 2327–2335. [Google Scholar] [CrossRef]
- Nalabotu, S.K.; Manne, N.D.; Kolli, M.B.; Nandyala, G.; Para, R.K.; Rice, K.M.; Jones, C.D.; Blough, E.R. Role of Oxidative Stress and Apoptosis in the Hepatic Toxicity Induced by Cerium Oxide Nanoparticles Following Intratracheal Instillation in Male Sprague-Dawley Rats. J. Toxicol. Risk Assess. 2019, 5, 26. [Google Scholar] [CrossRef]
- Harada, T.; Enomoto, A.; Boorman, G.A.; Maronpot, R.A. Liver and gallbladder. In Pathology of the Mouse; Maronpot, R.R., Boorman, G.A., Gaul, B.W., Eds.; Cache River Press: Vienna, IL, USA, 1999; pp. 119–183. [Google Scholar]
- Saber, A.T.; Mortensen, A.; Szarek, J.; Jacobsen, N.R.; Levin, M.; Koponen, I.K.; Jensen, K.A.; Vogel, U.; Wallin, H. Toxicity of pristine and paint-embedded TiO2 nanomaterials. Hum. Exp. Toxicol. 2019, 38, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Bourdon, J.A.; Halappanavar, S.; Saber, A.T.; Jacobsen, N.R.; Williams, A.; Wallin, H.; Vogel, U.; Yauk, C.L. Hepatic and Pulmonary Toxicogenomic Profiles in Mice Intratracheally Instilled with Carbon Black Nanoparticles Reveal Pulmonary Inflammation, Acute Phase Response, and Alterations in Lipid Homeostasis. Toxicol. Sci. 2012, 127, 474–484. [Google Scholar] [CrossRef] [PubMed]
- Hirouchi, Y.; Iwata, H.; Yamakawa, S.; Kato, M.; Kobayashi, K.; Yamamoto, T.; Inoue, H.; Enomoto, M.; Shiga, A.; Koike, Y. Historical data of neoplastic and non-neoplastic lesions in B6C3F1(C57BL/6CrSlc*C3H/HeSlc) mice. J. Toxicol. Pathol. 1994, 7, 153–177. [Google Scholar] [CrossRef][Green Version]
- Talamini, L.; Gimondi, S.; Violatto, M.B.; Fiordaliso, F.; Pedica, F.; Tran, N.L.; Sitia, G.; Aureli, F.; Raggi, A.; Nelissen, I.; et al. Repeated administration of the food additive E171 to mice results in accumulation in intestine and liver and promotes an inflammatory status. Nanotoxicology 2019, 13, 1087–1101. [Google Scholar] [CrossRef]
- Liu, Y.; Ji, J.; Ji, L.; Li, Y.; Zhang, B.; Yang, T.; Yang, J.; Lv, L.; Wu, G. Translocation of intranasal (i.n.) instillation of different-sized cerium dioxide (CeO2) particles: Potential adverse effects in mice. J. Toxicol. Environ. Health Part A 2019, 82, 1069–1075. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Lum, J.T.-S.; Huang, Y.; Hu, B.; Leung, K.S.-Y. Integration of sub-organ quantitative imaging LA-ICP-MS and fractionation reveals differences in translocation and transformation of CeO2 and Ce3+ in mice. Anal. Chim. Acta 2019, 1082, 18–29. [Google Scholar] [CrossRef]
- Kermanizadeh, A.; Chauché, C.; Balharry, D.; Brown, D.M.; Kanase, N.; Boczkowski, J.; Lanone, S.; Stone, V. The role of Kupffer cells in the hepatic response to silver nanoparticles. Nanotoxicology 2013, 8, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Park, J.-K.; Utsumi, T.; Seo, Y.-E.; Deng, Y.; Satoh, A.; Saltzman, W.M.; Iwakiri, Y. Cellular distribution of injected PLGA-nanoparticles in the liver. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 1365–1374. [Google Scholar] [CrossRef]
- Kreyling, W.G.; Holzwarth, U.; Schleh, C.; Kozempel, J.; Wenk, A.; Haberl, N.; Hirn, S.; Schäffler, M.; Lipka, J.; Semmler-Behnke, M.; et al. Quantitative biokinetics of titanium dioxide nanoparticles after oral application in rats: Part 2. Nanotoxicology 2017, 11, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.-J.; Chen, F.; Lau, J.T.Y.; Hu, Y.-P. Hepatocyte polyploidization and its association with pathophysiological processes. Cell Death Dis. 2017, 8, e2805. [Google Scholar] [CrossRef] [PubMed]
| Tested Nanomaterial | Source | Primary Particle Size | Specific Surface Area | Particle Density |
|---|---|---|---|---|
| TiO2 (rutile) | NanoAmor [44] | 10.5 nm [44] | 139.1 m2/g [44] | 4.23 g/cm3 [47] |
| CeO2 | Degussa/Quimidroga [48] | 13.0 ± 12.1 nm [48] | 56.7 m2/g [48] | 7.29 g/cm3 [48] |
| Carbon black | Evonik-Degussa | 14 nm [45] | 295 m2/g [49] | 2.1 g/cm3 [49] |
| 1 Day | 28 Days | 180 Days | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | TiO2 | CeO2 | CB | Control | TiO2 | CeO2 | CB | Control | TiO2 | CeO2 | CB | |
| Intratracheal instillation | ||||||||||||
| Initial body weight (g) | 18.63 ± 1.66 | 17.44 ± 1.74 | 19.02 ± 0.91 | 17.86 ± 1.44 | 18.30 ± 0.9 | 18.20 ± 1.40 | 18.50 ± 0.90 | 18.00 ± 1.00 | 18.60 ± 0.52 | 18.00 ± 1.03 | 18.30 ± 0.94 | 18.00 ± 0.98 |
| Final body weight (g) | 17.79 ± 1.35 | 16.61 ± 1.73 | 17.84 ± 0.69 | 17.67 ± 1.35 | 21.18 ± 0.89 | 21.52 ± 1.24 | 21.54 ± 0.74 | 21.10 ± 1.27 | 24.70 ± 0.85 | 24.90 ± 0.99 | 25.40 ± 1.93 | 25.05 ± 1.50 |
| Feed intake (g/kg bw/day) A | - | - | - | - | 181.8 ± 21.6 | 187.8 ± 19.8 | 185.9 ± 3.8 | 196.5 ± 23.4 | 168.5 ± 13.3 | 171.3 ± 7.6 | 161.8 ± 10.0 | 187.1 ± 40.7 |
| Absolute liver weight (g) | 0.9 ± 0.09 | 0.77 ± 0.09 | 0.89 ± 0.04 | 0.84 ± 0.08 | 1.04 ± 0.04 | 1.05 ± 0.12 | 1.10 ± 0.06 | 1.06 ± 0.07 | 1.15 ± 0.07 | 1.21 ± 0.12 | 1.20 ± 0.17 | 1.21 ± 0.15 |
| Relative liver weight (g/100 g) | 4.79 ± 0.27 | 4.66 ± 0.33 | 4.99 ± 0.21 | 4.78 ± 0.39 | 4.90 ± 0.16 | 4.88 ± 0.49 | 5.10 ± 0.20 | 5.05 ± 0.17 | 4.67 ± 0.21 | 4.85 ± 0.30 | 4.69 ± 0.36 | 4.80 ± 0.37 |
| Ti content in liver (µg/g) B | 0.41 ± 0.20 | 0.25 ± 0.15 | n.a. | n.a. | 0.30 ± 0.06 | 0.54 ± 0.31 | n.a. | n.a. | 0.18 ± 0.11 | 1.57 ± 1.38 | n.a. | n.a. |
| Ce content in liver (µg/g) B | 0.02 ±0.01 | n.a. | 0.03 ± 0.02 | n.a. | 0.02 ± 0.02 | n.a. | 0.82 ± 0.71 | n.a. | 0.00 ±0.00 | n.a. | 2.03 ± 2.63 | n.a. |
| Intravenous injection | ||||||||||||
| Initial body weight (g) | 19.14 ± 0.81 | 18.72 ± 0.65 | 19.56 ± 0.96 | 19.42 ± 0.81 | 18.20 ± 1.3 | 18.20 ± 1.2 | 18.20 ± 1.0 | 18.80 ± 1.1 | 18.56 ± 0.95 | 18.30 ± 1.33 | 17.18 ± 1.10 | 18.30 ± 1.42 |
| Final body weight (g) | 18.08 ± 0.79 | 18.03 ± 0.69 | 18.49 ± 0.75 | 18.10 ± 0.75 | 21.29 ± 1.32 | 21.31 ± 0.87 | 20.74 ± 1.40 | 21.39 ± 1.22 | 25.40 ± 1.71 | 24.10 ± 1.14 | 25.70 ± 2.25 | 26.00 ± 1.41 |
| Feed intake (g/kg bw/day) A | - | - | - | - | 182.1 ± 3.9 | 181.8 ± 9.2 | 192.4 ± 17.4 | 173.5 ± 6.9 | 180.2 ± 4.9 | 201.1 ± 21.4 | 183.4 ± 8.4 | 186.0 ± 19.3 |
| Absolute liver weight (g) | 0.90 ± 0.07 | 0.86 ± 0.07 | 0.94 ± 0.10 | 0.88 ± 0.09 | 1.00 ± 0.07 | 1.06 ± 0.07 | 1.01 ± 0.11 | 1.05 ± 0.08 | 1.22 ± 0.16 | 1.17 ± 0.10 | 1.18 ± 0.16 | 1.20 ± 0.19 |
| Relative liver weight (g/100 g) | 4.91 ± 0.44 | 4.76 ± 0.28 | 5.08 ± 0.38 | 4.83 ± 0.34 | 4.71 ± 0.08 | 4.96 ± 0.22 | 4.88 ± 0.33 | 4.89 ± 0.19 | 4.80 ± 0.38 | 4.82 ± 0.26 | 4.56 ± 0.33 | 4.62 ± 0.67 |
| Ti content in liver (µg/g) B | 0.42 ± 0.14 | 40.4 ± 33.0 | n.a. | n.a. | 0.49 ± 0.15 | 33.1 ± 27.1 | n.a. | n.a. | 0.40 ± 0.19 | 22.2 ± 22.3 | n.a. | n.a. |
| Ce content in liver (µg/g) B | 0.03 ± 0.03 | n.a. | 40.0 ± 47.8 | n.a. | 0.01 ±0.01 | n.a. | 36.9 ± 53.0 | n.a. | 0.02 ±0.03 | n.a. | 48.0 ±37.5 | n.a. |
| Enzyme | Intratracheal Instillation | Intravenous Injection | ||||||
|---|---|---|---|---|---|---|---|---|
| (U/L) | Control | TiO2 | CeO2 | CB | Control | TiO2 | CeO2 | CB |
| ALP | 119 ± 17 | 138 ± 16 | 113 ± 13 | 133 ± 19 | 125 ± 17 | 121 ± 13 | 119 ± 13 | 128 ± 14 |
| AST | 185 ± 141 | 132 ± 78 | 293 ± 179 | 213 ± 119 | 151 ± 85 | 91 ± 20 | 136 ± 25 | 120 ± 56 |
| ALT | 35 ± 8 | 38 ± 3 | 41 ± 9 | 47 ± 13 | 39 ± 5 | 34 ± 7 | 39 ± 8 | 38 ± 8 |
| Days Post-Exposure | Intratracheal Instillation | Intravenous Injection | ||||||
|---|---|---|---|---|---|---|---|---|
| Control | TiO2 | CeO2 | CB | Control | TiO2 | CeO2 | CB | |
| Focal infiltration of inflammatory cells (small) | ||||||||
| day 1 | 5/9 | 2/9 | 6/9 | 1/9 | 3/9 | 7/9 | 8/9 * | 6/9 |
| (8) | (2) | (12) | (1) # | (6) | (11) | (12) | (10) | |
| day 28 | 9/9 | 9/9 | 8/9 | 4/9 * | 5/8 | 8/9 | 6/9 | 1/9 * |
| (25) | (17) # | (25) | (11) # | (13) | (12) | (15) | (1) # | |
| day 180 | 6/9 | 5/9 | 5/9 | 2/8 | 5/9 | 3/8 | 6/9 | 3/9 |
| (11) | (9) | (8) | (2) | (10) | (9) | (13) | (3) | |
| Focal infiltration of inflammatory cells (big) | ||||||||
| day 1 | 3/9 | 4/9 | 3/9 | 3/9 | 6/9 | 2/9 | 6/9 | 1/9 * |
| (5) | (5) | (8) | (3) | (10) | (2) | (11) | (3) # | |
| day 28 | 4/9 | 4/9 | 3/9 | 6/9 | 4/8 | 7/9 | 3/9 | 6/9 |
| (6) | (5) | (4) | (13) | (4) | (11) | (3) | (15) | |
| day 180 | 6/9 | 3/9 | 6/9 | 2/8 | 4/9 | 4/8 | 4/9 | 5/9 |
| (11) | (6) | (10) | (2) | (8) | (10) | (9) | (9) | |
| Hepatocytes with pyknotic nuclei (single) | ||||||||
| day 1 | 0/9 | 0/9 | 0/9 | 1/9 | 1/9 | 0/9 | 1/9 | 0/9 |
| day 28 | 0/9 | 1/9 | 5/9 * | 2/9 | 3/8 | 1/9 | 0/9 | 2/9 |
| day 180 | 1/9 | 0/9 | 0/9 | 1/8 | 1/9 | 3/8 | 5/9 | 2/9 |
| Hepatocytes with pyknotic nuclei (area) | ||||||||
| day 1 | 0/9 | 0/9 | 1/9 | 0/9 | 3/9 | 7/9 | 1/9 | 4/9 |
| day 28 | 1/9 | 1/9 | 2/9 | 0/9 | 0/8 | 2/9 | 7/9 ** | 2/9 |
| day 180 | 3/9 | 8/9 * | 8/9 * | 3/8 | 0/9 | 2/8 | 1/9 | 5/9 * |
| Necrosis (area) | ||||||||
| day 1 | 0/9 | 0/9 | 0/9 | 2/9 | 8/9 | 9/9 | 3/9 | 4/9 |
| day 28 | 3/9 | 4/9 | 6/9 | 2/9 | 1/8 | 1/9 | 6/9 * | 4/9 |
| day 180 | 5/9 | 8/9 | 9/9 | 6/8 | 2/9 | 1/9 | 5/9 | 6/9 |
| Prominent nuclei of Kupffer cells | ||||||||
| day 1 | 0/9 | 0/9 | 0/9 | 0/9 | 3/9 | 9/9 ** | 3/9 | 9/9 ** |
| day 28 | 2/9 | 0/9 | 0/9 | 2/9 | 5/8 | 4/9 | 4/9 | 7/9 |
| day 180 | 1/9 | 0/9 | 1/9 | 1/8 | 0/9 | 1/8 | 0/9 | 2/9 |
| Apparent increase in binucleate hepatocytes | ||||||||
| day 1 | 0/9 | 6/9 ** | 1/9 | 1/9 | 0/9 | 5/9 * | 3/9 | 5/9* |
| day 28 | 0/9 | 4/9 | 2/9 | 4/9 | 1/8 | 2/9 | 0/9 | 0/9 |
| day 180 | 0/9 | 2/9 | 0/9 | 0/8 | 0/9 | 0/8 | 0/9 | 2/9 |
| Cytoplasmic vacuolation (hydropic degeneration) in hepatocytes | ||||||||
| day 1 | 0/9 | 0/9 | 0/9 | 0/9 | 0/9 | 0/9 | 2/9 | 0/9 |
| day 28 | 2/9 | 4/9 | 8/9 * | 0/9 | 6/8 | 4/9 | 6/9 | 5/9 |
| day 180 | 2/9 | 7/9 | 3/9 | 6/8 | 5/9 | 0/8 | 4/9 | 6/9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Modrzynska, J.; Mortensen, A.; Berthing, T.; Ravn-Haren, G.; Szarek, J.; Saber, A.T.; Vogel, U. Effect on Mouse Liver Morphology of CeO2, TiO2 and Carbon Black Nanoparticles Translocated from Lungs or Deposited Intravenously. Appl. Nano 2021, 2, 222-241. https://doi.org/10.3390/applnano2030016
Modrzynska J, Mortensen A, Berthing T, Ravn-Haren G, Szarek J, Saber AT, Vogel U. Effect on Mouse Liver Morphology of CeO2, TiO2 and Carbon Black Nanoparticles Translocated from Lungs or Deposited Intravenously. Applied Nano. 2021; 2(3):222-241. https://doi.org/10.3390/applnano2030016
Chicago/Turabian StyleModrzynska, Justyna, Alicja Mortensen, Trine Berthing, Gitte Ravn-Haren, Józef Szarek, Anne Thoustrup Saber, and Ulla Vogel. 2021. "Effect on Mouse Liver Morphology of CeO2, TiO2 and Carbon Black Nanoparticles Translocated from Lungs or Deposited Intravenously" Applied Nano 2, no. 3: 222-241. https://doi.org/10.3390/applnano2030016
APA StyleModrzynska, J., Mortensen, A., Berthing, T., Ravn-Haren, G., Szarek, J., Saber, A. T., & Vogel, U. (2021). Effect on Mouse Liver Morphology of CeO2, TiO2 and Carbon Black Nanoparticles Translocated from Lungs or Deposited Intravenously. Applied Nano, 2(3), 222-241. https://doi.org/10.3390/applnano2030016