The Effects of Tetrabromobisphenol A (TBBPA) on the Mussel Mytilus galloprovincialis: A Multi-Biomarker Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. TBBPA Stock Solution
2.2. Experimental Trials
Ethics
2.3. Sample Treatment
2.4. Total Protein
2.5. Biomarkers Analyses
2.5.1. Glutathione S-Transferase (GST) Activity
2.5.2. Superoxide Dismutase (SOD) Activity
2.5.3. Catalase (CAT) Activity
2.5.4. Acetylcholinesterase (AChE) Activity
2.5.5. Lipid Peroxidation (LPO)
2.5.6. Total Antioxidant Capacity (TAC)
2.5.7. Caspase-3 (CASP-3)
2.5.8. Total Ubiquitin (UBI)
2.6. Statistical Analysis
3. Results
3.1. Mortality Rate
3.2. Biomarkers of Oxidative Stress
3.2.1. Glutathione-S-Transferase (GST)
3.2.2. Superoxide Dismutase (SOD)
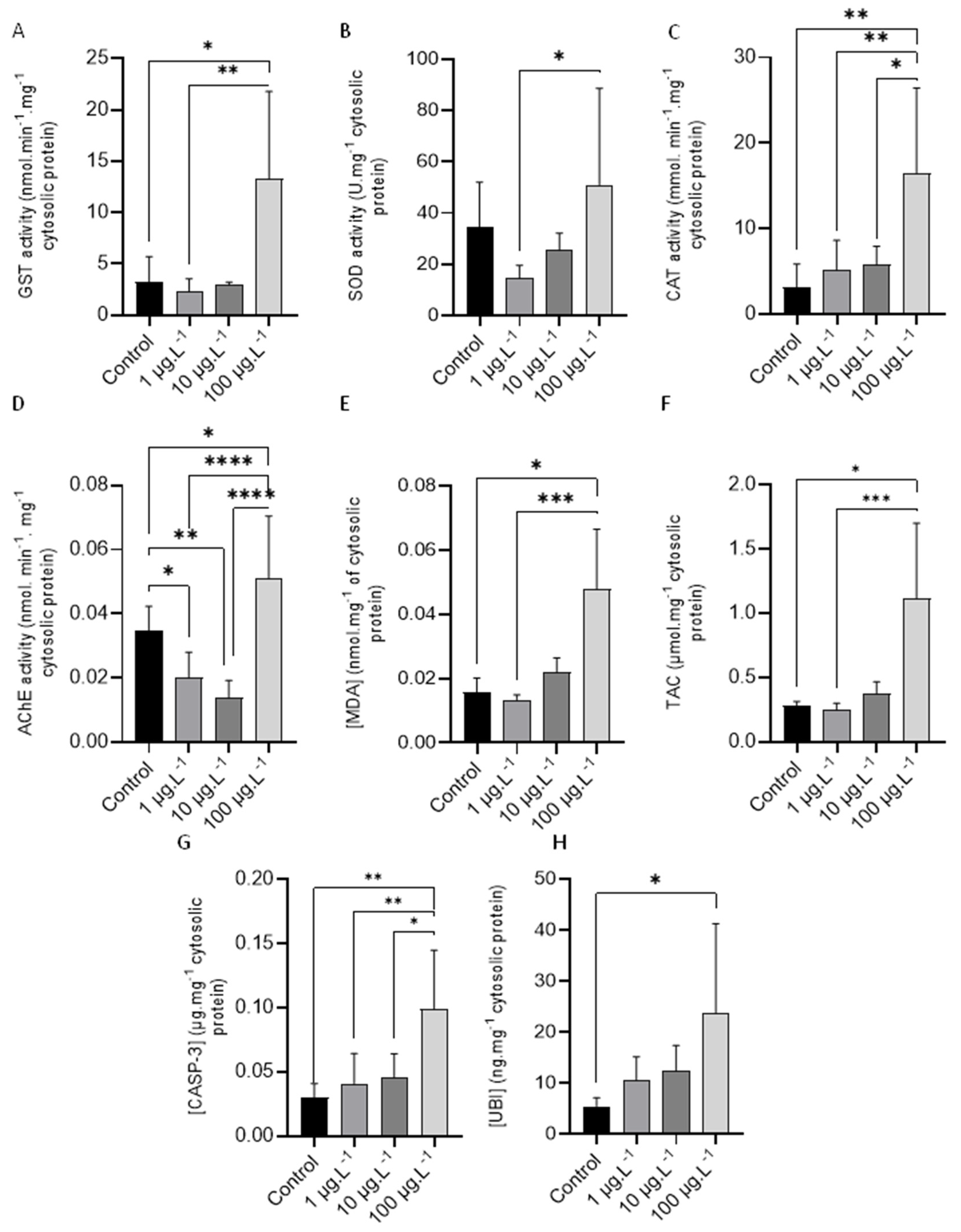
3.2.3. Catalase (CAT)
3.2.4. Acetylcholinesterase (AChE)
3.2.5. Lipid Peroxidation (LPO)
3.2.6. Total Antioxidant Capacity (TAC)
3.2.7. Caspase-3 (CASP-3)
3.2.8. Total Ubiquitin (UBI)
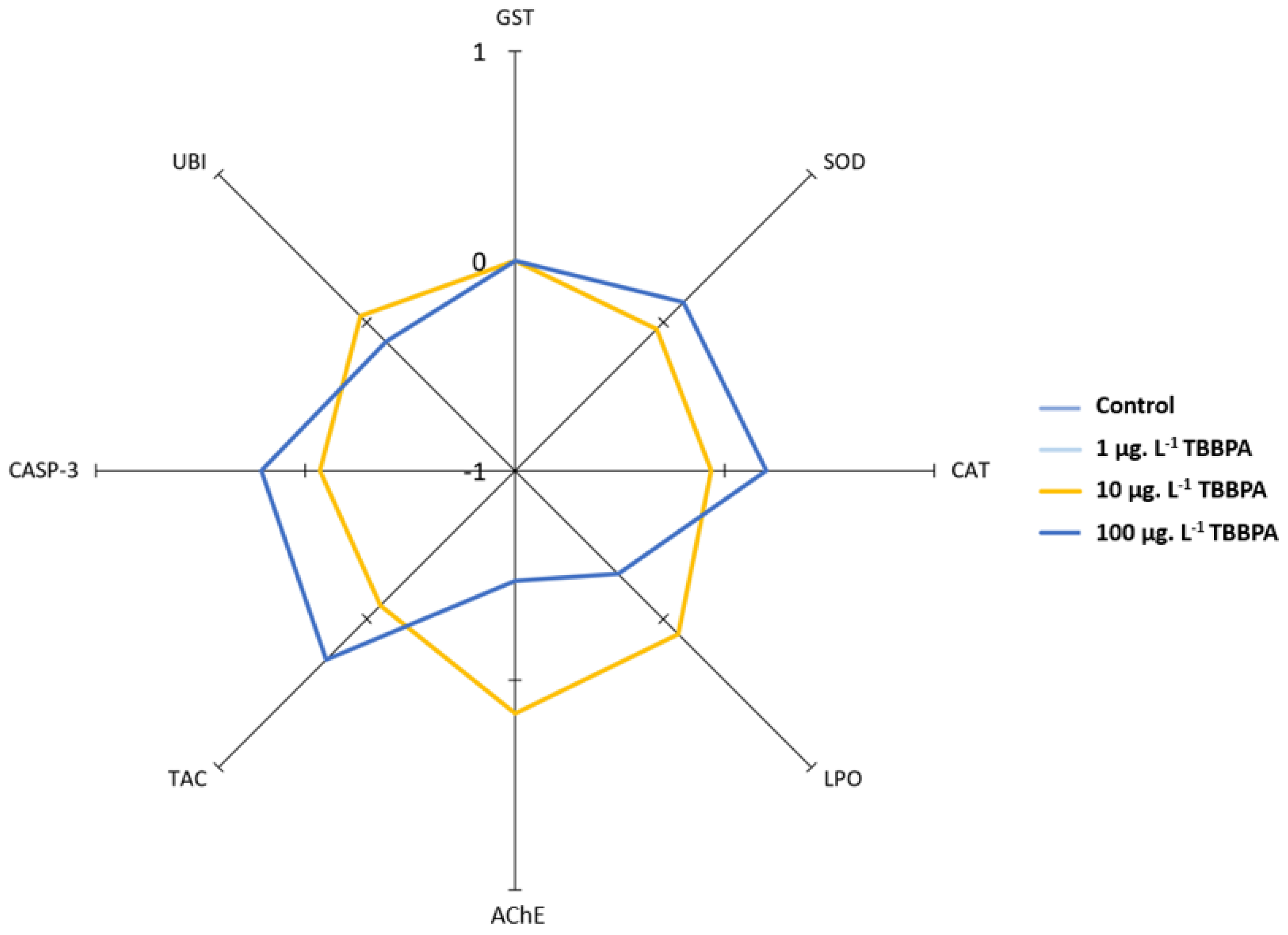
3.3. IBR Index
3.4. Correlation Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Gereckea, A.C.; Schmid, P.; Bogdal, C.; Kohler, M.; Zennegg, M.; Heeb, N.V. Brominated Flame Retardants—Endocrine-Disrupting Chemicals in the Swiss Environment. Chimia 2008, 62, 352. [Google Scholar] [CrossRef]
- Fabbri, R.; Montagna, M.; Balbi, T.; Raffo, E.; Palumbo, F.; Canesi, L. Adaptation of the bivalve embryotoxicity assay for the high throughput screening of emerging contaminants in Mytilus galloprovincialis. Mar. Environ. Res. 2014, 99, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Poma, G.; Malysheva, S.V.; Goscinny, S.; Malarvannan, G.; Voorspoels, S.; Covaci, A.; Van Loco, J. Occurrence of selected halogenated flame retardants in Belgian foodstuff. Chemosphere 2018, 194, 256–265. [Google Scholar] [CrossRef]
- Wang, R.; Zhang, C.; Li, X.; Sha, W.; Xue, Z.; Zhou, Z.; Ma, Y.; Zhu, S.; Guo, Z.; Zhao, B.; et al. Toxicological evaluation of TBBPA by common carp (Cyprinus carpio) about the in vivo/vitro disturbance of the AHR pathway. Sci. Total Environ. 2023, 904, 166622. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Guo, H.; Yu, H.; Wang, X.; Wu, J.; Xue, Y. Bioaccumulation and physiological effects of tetrabromobisphenol A in coontail Ceratophyllum demersum L. Chemosphere 2008, 70, 1787–1795. [Google Scholar] [CrossRef]
- Liu, A.; Zhao, Z.; Qu, G.; Shen, Z.; Liang, X.; Shi, J.; Jiang, G. Identification of transformation/degradation products of tetrabromobisphenol A and its derivatives. TrAC Trends Anal. Chem. 2019, 111, 85–99. [Google Scholar] [CrossRef]
- Blanco, E.; Casais, M.C.; Mejuto, M.C.; Cela, R. Analysis of tetrabromobisphenol A and other phenolic compounds in water samples by non-aqueous capillary electrophoresis coupled to photodiode array ultraviolet detection. J. Chromatogr. A 2005, 1071, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-W.; Yan, Z.-G.; Xu, F.-F.; Wang, S.-R.; Wu, F.-C. Development of freshwater aquatic life criteria for Tetrabromobisphenol A in China. Environ. Pollut. 2012, 169, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Saint-Louis, R.; Pelletier, E. LC-ESI-MS-MS method for the analysis of tetrabromobisphenol A in sediment and sewage sludge. Analyst 2004, 129, 724–730. [Google Scholar] [CrossRef]
- OSPAR Convention (Convention for the Protection of the Marine Environment of the North-East Atlantic). Available online: https://www.ospar.org/site/assets/files/1169/ospar_convention.pdf (accessed on 5 January 2024).
- Álvarez-Muñoz, D.; Rodríguez-Mozaz, S.; Maulvault, A.L.; Tediosi, A.; Fernández-Tejedor, M.; Van den Heuvel, F.; Kotterman, M.; Marques, A.; Barceló, D. Occurrence of pharmaceuticals and endocrine disrupting compounds in macroalgaes, bivalves, and fish from coastal areas in Europe. Environ. Res. 2015, 143, 56–64. [Google Scholar] [CrossRef]
- Kotthoff, M.; Rüdel, H.; Jürling, H. Detection of tetrabromobisphenol A and its mono- and dimethyl derivatives in fish, sediment and suspended particulate matter from European freshwaters and estuaries. Anal. Bioanal. Chem. 2017, 409, 3685–3694. [Google Scholar] [CrossRef]
- Sun, C.-S.; Yuan, S.-W.; Hou, R.; Zhang, S.-Q.; Huang, Q.-Y.; Lin, L.; Li, H.-X.; Liu, S.; Cheng, Y.-Y.; Li, Z.-H.; et al. First insights into the bioaccumulation, biotransformation and trophic transfer of typical tetrabromobisphenol A (TBBPA) analogues along a simulated aquatic food chain. J. Hazard. Mater. 2024, 465, 133390. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-G.; Jeong, Y.; Kim, D.; Kang, G.-J.; Kang, Y. Assessment of Tetrabromobisphenol and Hexabromocyclododecanes exposure and risk characterization using occurrence data in foods. Food Chem. Toxicol. 2020, 137, 111121. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ji, C.; Li, F.; Zhan, J.; Sun, T.; Tang, J.; Wu, H. Tetrabromobisphenol A induced reproductive endocrine-disrupting effects in mussel Mytilus galloprovincialis. J. Hazard. Mater. 2021, 416, 126228. [Google Scholar] [CrossRef]
- Mariussen, E.; Fonnum, F. The effect of brominated flame retardants on neurotransmitter uptake into rat brain synaptosomes and vesicles. Neurochem. Int. 2003, 43, 533–542. [Google Scholar] [CrossRef]
- Pullen, S.; Boecker, R.; Tiegs, G. The flame retardants tetrabromobisphenol A and tetrabromobisphenol A–bisallylether suppress the induction of interleukin-2 receptor α chain (CD25) in murine splenocytes. Toxicology 2003, 184, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, N.; Ito, Y.; Yamaguchi, M.; Mitumori, K.; Koizumi, M.; Hasegawa, R.; Kamata, E.; Ema, M. Unexpected nephrotoxicity induced by tetrabromobisphenol A in newborn rats. Toxicol. Lett. 2004, 150, 145–155. [Google Scholar] [CrossRef]
- He, Q.; Wang, X.; Sun, P.; Wang, Z.; Wang, L. Acute and chronic toxicity of tetrabromobisphenol A to three aquatic species under different pH conditions. Aquat. Toxicol. 2015, 164, 145–154. [Google Scholar] [CrossRef]
- Pittinger, C.A.; Pecquet, A.M. Review of historical aquatic toxicity and bioconcentration data for the brominated flame retardant tetrabromobisphenol A (TBBPA): Effects to fish, invertebrates, algae, and microbial communities. Environ. Sci. Pollut. R. 2018, 25, 14361–14372. [Google Scholar] [CrossRef]
- Wang, S.; Sun, Z.; Ren, C.; Li, F.; Xu, Y.; Wu, H.; Ji, C. Time- and dose-dependent detoxification and reproductive endocrine disruption induced by tetrabromobisphenol A (TBBPA) in mussel Mytilus galloprovincialis. Mar. Environ. Res. 2023, 183, 105839. [Google Scholar] [CrossRef]
- Jiang, S.; Miao, J.; Wang, X.; Liu, P.; Pan, L. Inhibition of growth in juvenile manila clam Ruditapes philippinarum: Potential adverse outcome pathway of TBBPA. Chemosphere 2019, 224, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Boukadida, K.; Mlouka, R.; Clerandeau, C.; Banni, M.; Cachot, J. Natural distribution of pure and hybrid Mytilus sp. along the south Mediterranean and North-east Atlantic coasts and sensitivity of D-larvae stages to temperature increases and metal pollution. Sci. Total Environ. 2021, 756, 143675. [Google Scholar] [CrossRef] [PubMed]
- Beyer, J.; Green, N.W.; Brooks, S.; Allan, I.J.; Ruus, A.; Gomes, T.; Bråte, I.L.N.; Schøyen, M. Blue mussels (Mytilus edulis spp.) as sentinel organisms in coastal pollution monitoring: A review. Mar. Environ. Res. 2017, 130, 338–365. [Google Scholar] [CrossRef] [PubMed]
- Farrington, J.W.; Tripp, B.W.; Tanabe, S.; Subramanian, A.; Sericano, J.L.; Wade, T.L.; Knap, A.H.; Edward, D. Goldberg’s proposal of “the Mussel Watch”: Reflections after 40 years. Mar. Pollut. Bull. 2016, 110, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Cajaraville, M.P.; Bebianno, M.J.; Blasco, J.; Porte, C.; Sarasquete, C.; Viarengo, A. The use of biomarkers to assess the impact of pollution in coastal environments of the Iberian Peninsula: A practical approach. Sci. Total Environ. 2000, 247, 295–311. [Google Scholar] [CrossRef] [PubMed]
- Matozzo, V.; Gagné, F.; Marin, M.G.; Ricciardi, F.; Blaise, C. Vitellogenin as a biomarker of exposure to estrogenic compounds in aquatic invertebrates: A review. Environ. Int. 2008, 34, 531–545. [Google Scholar] [CrossRef] [PubMed]
- van der Oost, R.; Beyer, J.; Vermeulen, N.P.E. Fish bioaccumulation and biomarkers in environmental risk assessment: A review. Environ. Toxicol. Phar 2003, 13, 57–149. [Google Scholar] [CrossRef] [PubMed]
- Almroth, B.C.; Sturve, J.; Berglund, Å.; Förlin, L. Oxidative damage in eelpout (Zoarces viviparus), measured as protein carbonyls and TBARS, as biomarkers. Aquat. Toxicol. 2005, 73, 171–180. [Google Scholar] [CrossRef]
- Borković, S.S.; Šaponjić, J.S.; Pavlović, S.Z.; Blagojević, D.P.; Milošević, S.M.; Kovačević, T.B.; Radojičić, R.M.; Spasić, M.B.; Žikić, R.V.; Saičić, Z.S. The activity of antioxidant defence enzymes in the mussel Mytilus galloprovincialis from the Adriatic Sea. Comp. Biochem. Phys. C 2005, 141, 366–374. [Google Scholar] [CrossRef]
- Rodrigues, I.; Ferreira, I.J.; Duarte, R.M.B.O.; Diniz, M. Effects of Exposure to Urban Atmospheric Particulate Matter Suspended in Seawater on the Mussel Mytilus galloprovincialis. Environments 2024, 11, 12. [Google Scholar] [CrossRef]
- Afsa, S.; De Marco, G.; Giannetto, A.; Parrino, V.; Cappello, T.; ben Mansour, H.; Maisano, M. Histological endpoints and oxidative stress transcriptional responses in the Mediterranean mussel Mytilus galloprovincialis exposed to realistic doses of salicylic acid. Environ. Toxicol. Phar 2022, 92, 103855. [Google Scholar] [CrossRef] [PubMed]
- Chora, S.; McDonagh, B.; Sheehan, D.; Starita-Geribaldi, M.; Roméo, M.; Bebianno, M.J. Ubiquitination and carbonylation as markers of oxidative-stress in Ruditapes decussatus. Mar. Environ. Res. 2008, 66, 95–97. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Umeno, A.; Shichiri, M. Lipid peroxidation biomarkers for evaluating oxidative stress and assessing antioxidant capacity in vivo. J. Clin. Biochem. Nutr. 2013, 52, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione S-Transferases: The first enzymatic step in mercapturic acid formation. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar] [CrossRef]
- Sun, Y.; Oberley, L.W.; Li, Y. A simple method for clinical assay of superoxide dismutase. Clin. Chem. 1988, 34, 497–500. [Google Scholar] [CrossRef] [PubMed]
- Johansson, L.H.; Håkan Borg, L.A. A spectrophotometric method for determination of catalase activity in small tissue samples. Anal. Biochem. 1988, 174, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Madeira, C.; Leal, M.C.; Diniz, M.S.; Cabral, H.N.; Vinagre, C. Thermal stress and energy metabolism in two circumtropical decapod crustaceans: Responses to acute temperature events. Mar. Environ. Res. 2018, 141, 148–158. [Google Scholar] [CrossRef]
- Kambayashi, Y.; Binh, N.T.; Asakura, H.W.; Hibino, Y.; Hitomi, Y.; Nakamura, H.; Ogino, K. Efficient Assay for Total Antioxidant Capacity in Human Plasma Using a 96-Well Microplate. J. Clin. Biochem. Nutr. 2008, 44, 46–51. [Google Scholar] [CrossRef]
- Lopes, A.R.; Sampaio, E.; Santos, C.; Couto, A.; Pegado, M.R.; Diniz, M.; Munday, P.L.; Rummer, J.L.; Rosa, R. Absence of cellular damage in tropical newly hatched sharks (Chiloscyllium plagiosum) under ocean acidification conditions. Cell Stress Chaperon 2018, 23, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Beliaeff, B.; Burgeot, T. Integrated biomarker response: A useful tool for ecological risk assessment. Environ. Toxicol. Chem. 2002, 21, 1316–1322. [Google Scholar] [CrossRef] [PubMed]
- Canesi, L.; Lorusso, L.C.; Ciacci, C.; Betti, M.; Gallo, G. Effects of the brominated flame retardant tetrabromobisphenol-A (TBBPA) on cell signaling and function of Mytilus hemocytes: Involvement of MAP kinases and protein kinase C. Aquat. Toxicol. 2005, 75, 277–287. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Ji, G.; Liu, J.; Zhang, S.; Gong, Y.; Shi, L. TBBPA induces developmental toxicity, oxidative stress, and apoptosis in embryos and zebrafish larvae (Danio rerio). Environ. Toxicol. 2016, 31, 1241–1249. [Google Scholar] [CrossRef] [PubMed]
- Jarosiewicz, M.; Krokosz, A.; Marczak, A.; Bukowska, B. Changes in the activities of antioxidant enzymes and reduced glutathione level in human erythrocytes exposed to selected brominated flame retardants. Chemosphere 2019, 227, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Hao, C.; Xiang, M.; Tian, J.; Kuang, H.; Li, Z. Potential obesogenic effects of TBBPA and its alternatives TBBPS and TCBPA revealed by metabolic perturbations in human hepatoma cells. Sci. Total Environ. 2022, 832, 154847. [Google Scholar] [CrossRef] [PubMed]
- Feng, W.; Xu, T.; Zuo, J.; Luo, M.; Mao, G.; Chen, Y.; Ding, Y.; Okeke, E.S.; Wu, X.; Yang, L. The potential mechanisms of TBBPA bis(2-hydroxyethyl) ether induced developmental neurotoxicity in juvenile zebrafish (Danio rerio). Comp. Biochem. Phys. C 2023, 265, 109530. [Google Scholar] [CrossRef] [PubMed]
- Crowley, L.C.; Waterhouse, N.J. Detecting Cleaved Caspase-3 in Apoptotic Cells by Flow Cytometry. Cold Spring Harb. Protoc. 2016, 2016, pdb.prot087312. [Google Scholar] [CrossRef]
- Abbas, R.; Larisch, S. Killing by Degradation: Regulation of Apoptosis by the Ubiquitin-Proteasome-System. Cells 2021, 10, 3465. [Google Scholar] [CrossRef]
- Gil, F.; Pla, A. Biomarkers as biological indicators of xenobiotic exposure. J. Appl. Toxicol. 2001, 21, 245–255. [Google Scholar] [CrossRef]
- Hook, S.E.; Gallagher, E.P.; Batley, G.E. The role of biomarkers in the assessment of aquatic ecosystem health. Integr. Environ. Assess. Manag. 2014, 10, 327–341. [Google Scholar] [CrossRef] [PubMed]
- Lomartire, S.; Marques, J.C.; Gonçalves, A.M.M. Biomarkers based tools to assess environmental and chemical stressors in aquatic systems. Ecol. Indic. 2021, 122, 107207. [Google Scholar] [CrossRef]
- Yang, S.; Wang, S.; Liu, H.; Yan, Z. Tetrabromobisphenol A: Tissue distribution in fish, and seasonal variation in water and sediment of Lake Chaohu, China. Environ. Sci. Pollut. R. 2012, 19, 4090–4096. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Pan, L.; Xiu, M.; Jin, Q. Exposure of Chlamys farreri to tetrabromobisphenol A: Accumulation and multibiomarker responses. Environ. Sci. Pollut. R. 2015, 22, 12224–12234. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Pan, L.; Xiu, M.; Liu, D. Dietary accumulation of tetrabromobisphenol A and its effects on the scallop Chlamys farreri. Comp. Biochem. Phys. C 2015, 167, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Xu, F.; Zheng, B.; Wu, F.; Wang, S. Multibiomarker responses upon exposure to tetrabromobisphenol A in the freshwater fish Carassius auratus. Aquat. Toxicol. 2013, 142–143, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.; Pan, L.; Miao, J.; Liu, N. Application of SSH and quantitative real time PCR to construction of gene expression profiles from scallop Chlamys farreri in response to exposure to tetrabromobisphenol A. Environ. Toxicol. Phar 2012, 34, 911–918. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Pan, L.; Xiu, M.; Jin, Q.; Wang, G.; Wang, C. Bioaccumulation and detoxification responses in the scallop Chlamys farreri exposed to tetrabromobisphenol A (TBBPA). Environ. Toxicol. Phar 2015, 39, 997–1007. [Google Scholar] [CrossRef] [PubMed]
- Fini, J.-B.; Riu, A.; Debrauwer, L.; Hillenweck, A.; Le Mével, S.; Chevolleau, S.; Boulahtouf, A.; Palmier, K.; Balaguer, P.; Cravedi, J.-P.; et al. Parallel Biotransformation of Tetrabromobisphenol A in Xenopus laevis and Mammals: Xenopus as a Model for Endocrine Perturbation Studies. Toxicol. Sci. 2012, 125, 359–367. [Google Scholar] [CrossRef] [PubMed]
- Almeida, E.A.; Bainy, A.C.D.; Dafre, A.L.; Gomes, O.F.; Medeiros, M.H.G.; Di Mascio, P. Oxidative stress in digestive gland and gill of the brown mussel (Perna perna) exposed to air and re-submersed. J. Exp. Mar. Biol. Ecol. 2005, 318, 21–30. [Google Scholar] [CrossRef]
- López-Barea, J.; Pueyo, C. Mutagen content and metabolic activation of promutagens by molluscs as biomarkers of marine pollution. Mutat. Res. 1998, 399, 3–15. [Google Scholar] [CrossRef]
- Ji, C.; Li, F.; Wang, Q.; Zhao, J.; Sun, Z.; Wu, H. An integrated proteomic and metabolomic study on the gender-specific responses of mussels Mytilus galloprovincialis to tetrabromobisphenol A (TBBPA). Chemosphere 2016, 144, 527–539. [Google Scholar] [CrossRef] [PubMed]
- Oruç, E.Ö.; Usta, D. Evaluation of oxidative stress responses and neurotoxicity potential of diazinon in different tissues of Cyprinus carpio. Environ. Toxicol. Phar 2007, 23, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, I.J.; Meneses, L.; Paiva, A.; Diniz, M.; Duarte, A.R.C. Assessment of deep eutectic solvents toxicity in zebrafish (Danio rerio). Chemosphere 2022, 299, 134415. [Google Scholar] [CrossRef]
- Emmanouil, C.; Green, R.M.; Willey, F.R.; Chipman, J.K. Oxidative damage in gill of Mytilus edulis from Merseyside, UK, and reversibility after depuration. Environ. Pollut. 2008, 151, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Liang, Y.; Chen, M.; Wang, X. Assessing the toxicity of TBBPA and HBCD by zebrafish embryo toxicity assay and biomarker analysis. Environ. Toxicol. 2009, 24, 334–342. [Google Scholar] [CrossRef]
- Matozzo, V.; Tomei, A.; Marin, M.G. Acetylcholinesterase as a biomarker of exposure to neurotoxic compounds in the clam Ruditapes philippinarum from the Lagoon of Venice. Mar. Pollut. Bull. 2005, 50, 1686–1693. [Google Scholar] [CrossRef]
- Zhu, B.; Zhao, G.; Yang, L.; Zhou, B. Tetrabromobisphenol A caused neurodevelopmental toxicity via disrupting thyroid hormones in zebrafish larvae. Chemosphere 2018, 197, 353–361. [Google Scholar] [CrossRef]
- Liu, Q.; Ren, X.; Long, Y.; Hu, L.; Qu, G.; Zhou, Q.; Jiang, G. The potential neurotoxicity of emerging tetrabromobisphenol A derivatives based on rat pheochromocytoma cells. Chemosphere 2016, 154, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Melo, J.B.; Agostinho, P.; Oliveira, C.R. Involvement of oxidative stress in the enhancement of acetylcholinesterase activity induced by amyloid beta-peptide. Neurosci. Res. 2003, 45, 117–127. [Google Scholar] [CrossRef]
- Barboza, L.G.A.; Lopes, C.; Oliveira, P.; Bessa, F.; Otero, V.; Henriques, B.; Raimundo, J.; Caetano, M.; Vale, C.; Guilhermino, L. Microplastics in wild fish from North East Atlantic Ocean and its potential for causing neurotoxic effects, lipid oxidative damage, and human health risks associated with ingestion exposure. Sci. Total Environ. 2020, 717, 134625. [Google Scholar] [CrossRef]
- De Marco, G.; Eliso, M.C.; Oliveri Conti, G.; Galati, M.; Billè, B.; Maisano, M.; Ferrante, M.; Cappello, T. Short-term exposure to polystyrene microplastics hampers the cellular function of gills in the Mediterranean mussel Mytilus galloprovincialis. Aquat. Toxicol. 2023, 264, 106736. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.B.; Liu, Y.Q.; Cui, Y.F. Pathways to caspase activation. Cell Biol. Int. 2005, 29, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Hochstrasser, M. Ubiquitin and intracellular protein degradation. Curr. Opin. Cell Biol. 1992, 4, 1024–1031. [Google Scholar] [CrossRef]
- Shang, F.; Gong, X.; Taylor, A. Activity of Ubiquitin-dependent Pathway in Response to Oxidative Stress: Ubiquitin-activating enzyme is transiently up-regulated. J. Biol. Chem. 1997, 272, 23086–23093. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Yang, N.; Liu, H.; Yao, Y.; Xu, S. TBBPA causes apoptosis in grass carp hepatocytes involving destroyed ER-mitochondrial function. Chemosphere 2023, 341, 139974. [Google Scholar] [CrossRef] [PubMed]
- Suyama, K.; Kesamaru, H.; Okubo, T.; Kasatani, K.; Tomohara, K.; Matsushima, A.; Nose, T. High cytotoxicity of a degraded TBBPA, dibromobisphenol A, through apoptotic and necrosis pathways. Heliyon 2023, 9, e13003. [Google Scholar] [CrossRef]
- Liu, H.; Ma, Z.; Zhang, T.; Yu, N.; Su, G.; Giesy, J.P.; Yu, H. Pharmacokinetics and effects of tetrabromobisphenol a (TBBPA) to early life stages of zebrafish (Danio rerio). Chemosphere 2018, 190, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Miao, B.; Yakubu, S.; Zhu, Q.; Issaka, E.; Zhang, Y.; Adams, M. A Review on Tetrabromobisphenol A: Human Biomonitoring, Toxicity, Detection and Treatment in the Environment. Molecules 2023, 28, 2505. [Google Scholar] [CrossRef]
- Cunha, S.C.; Menezes-Sousa, D.; Mello, F.V.; Miranda, J.A.T.; Fogaca, F.H.S.; Alonso, M.B.; Torres, J.P.M.; Fernandes, J.O. Survey on endocrine-disrupting chemicals in seafood: Occurrence and distribution. Environ. Res. 2022, 210, 112886. [Google Scholar] [CrossRef]
| SOD | CAT | LPO | AChE | TAC | CASP-3 | UBI | |
|---|---|---|---|---|---|---|---|
| GST | 0.26 | 0.37 | 0.71 *** | 0.27 | 0.59 *** | 0.52 * | 0.10 |
| SOD | 0.14 | 0.60 ** | 0.33 | 0.78 **** | 0.41 | 0.44 * | |
| CAT | 0.65 ** | −0.05 | 0.55 * | 0.40 | 0.31 | ||
| LPO | 0.48 * | 0.87 **** | 0.64 ** | 0.75 *** | |||
| AChE | 0.50 * | 0.20 | 0.18 | ||||
| TAC | 0.69 **** | 0.59 ** | |||||
| CASP-3 | 0.64 ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Copeto, S.; Ganço, S.; Ferreira, I.J.; Silva, M.; Motta, C.; Diniz, M. The Effects of Tetrabromobisphenol A (TBBPA) on the Mussel Mytilus galloprovincialis: A Multi-Biomarker Approach. Oceans 2024, 5, 181-195. https://doi.org/10.3390/oceans5020011
Copeto S, Ganço S, Ferreira IJ, Silva M, Motta C, Diniz M. The Effects of Tetrabromobisphenol A (TBBPA) on the Mussel Mytilus galloprovincialis: A Multi-Biomarker Approach. Oceans. 2024; 5(2):181-195. https://doi.org/10.3390/oceans5020011
Chicago/Turabian StyleCopeto, Sandra, Sara Ganço, Inês João Ferreira, Marco Silva, Carla Motta, and Mário Diniz. 2024. "The Effects of Tetrabromobisphenol A (TBBPA) on the Mussel Mytilus galloprovincialis: A Multi-Biomarker Approach" Oceans 5, no. 2: 181-195. https://doi.org/10.3390/oceans5020011
APA StyleCopeto, S., Ganço, S., Ferreira, I. J., Silva, M., Motta, C., & Diniz, M. (2024). The Effects of Tetrabromobisphenol A (TBBPA) on the Mussel Mytilus galloprovincialis: A Multi-Biomarker Approach. Oceans, 5(2), 181-195. https://doi.org/10.3390/oceans5020011









