Erythrocyte, Whole Blood, Plasma, and Blubber Fatty Acid Profiles in Oceanaria-Based versus Wild Alaskan Belugas (Delphinapterus leucas)
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Laboratory Processing and Analysis of PLASMA, RBC, WB, Blubber and Dried Blood Spots
2.3. Fish FA Analysis
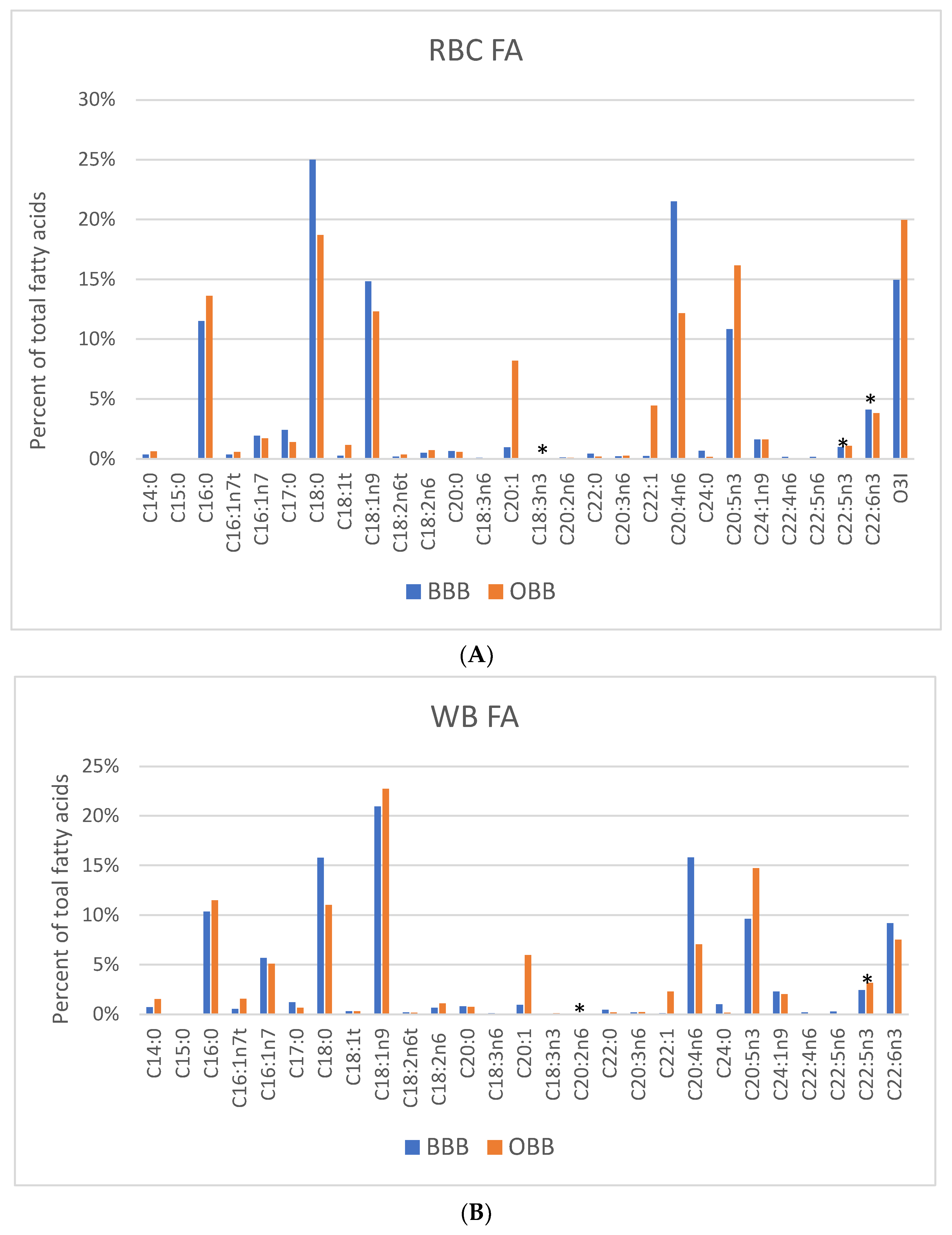
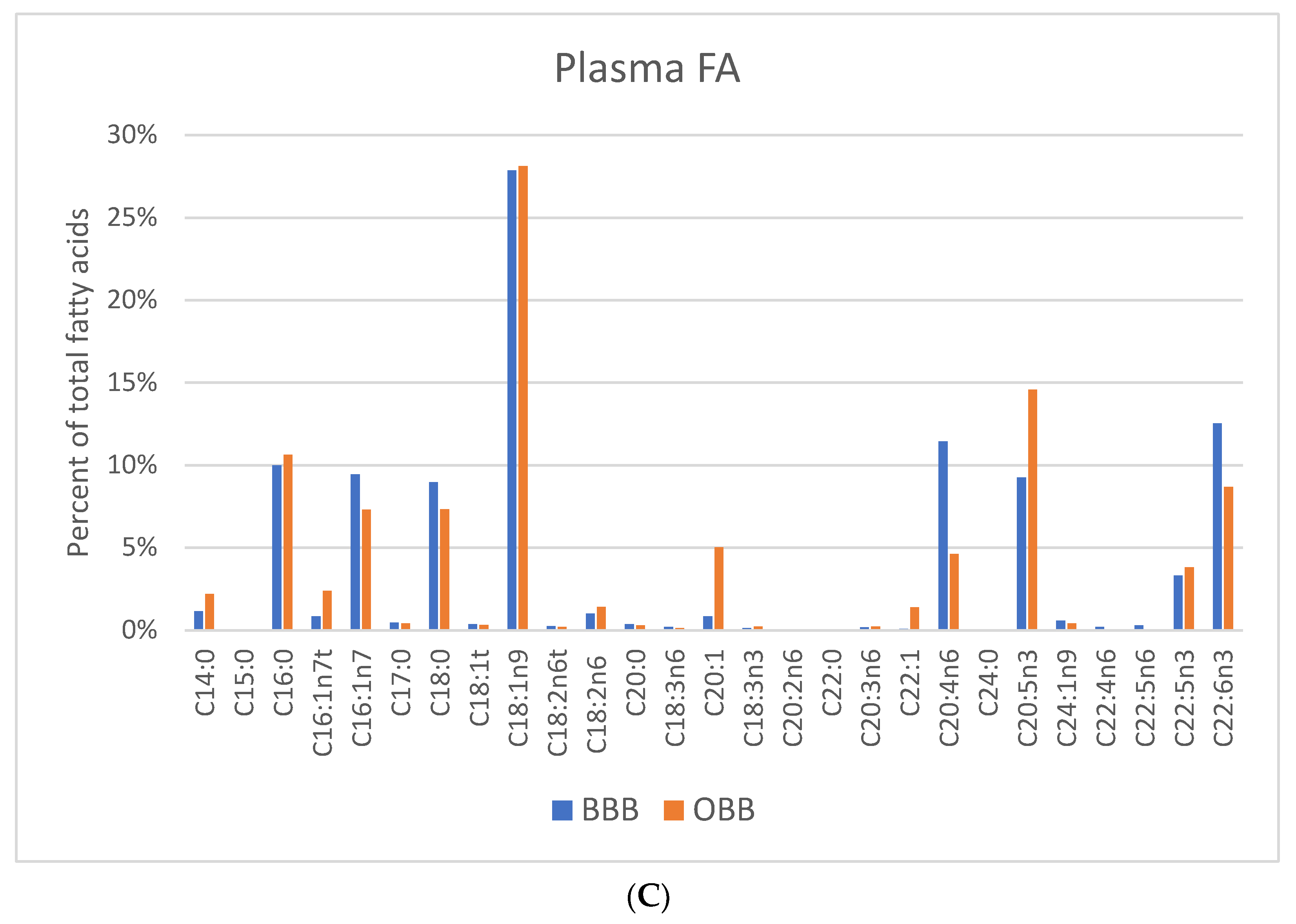
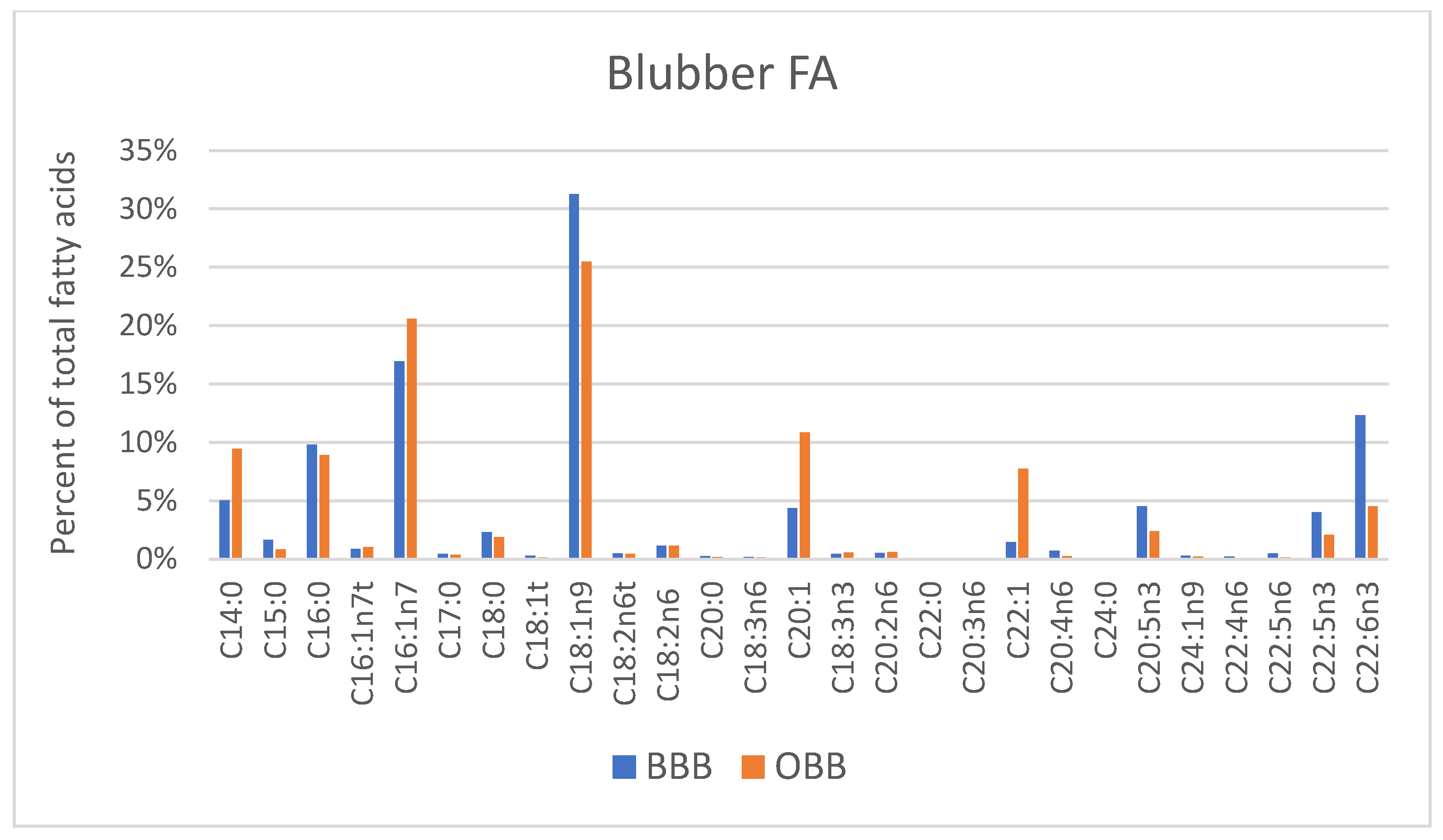
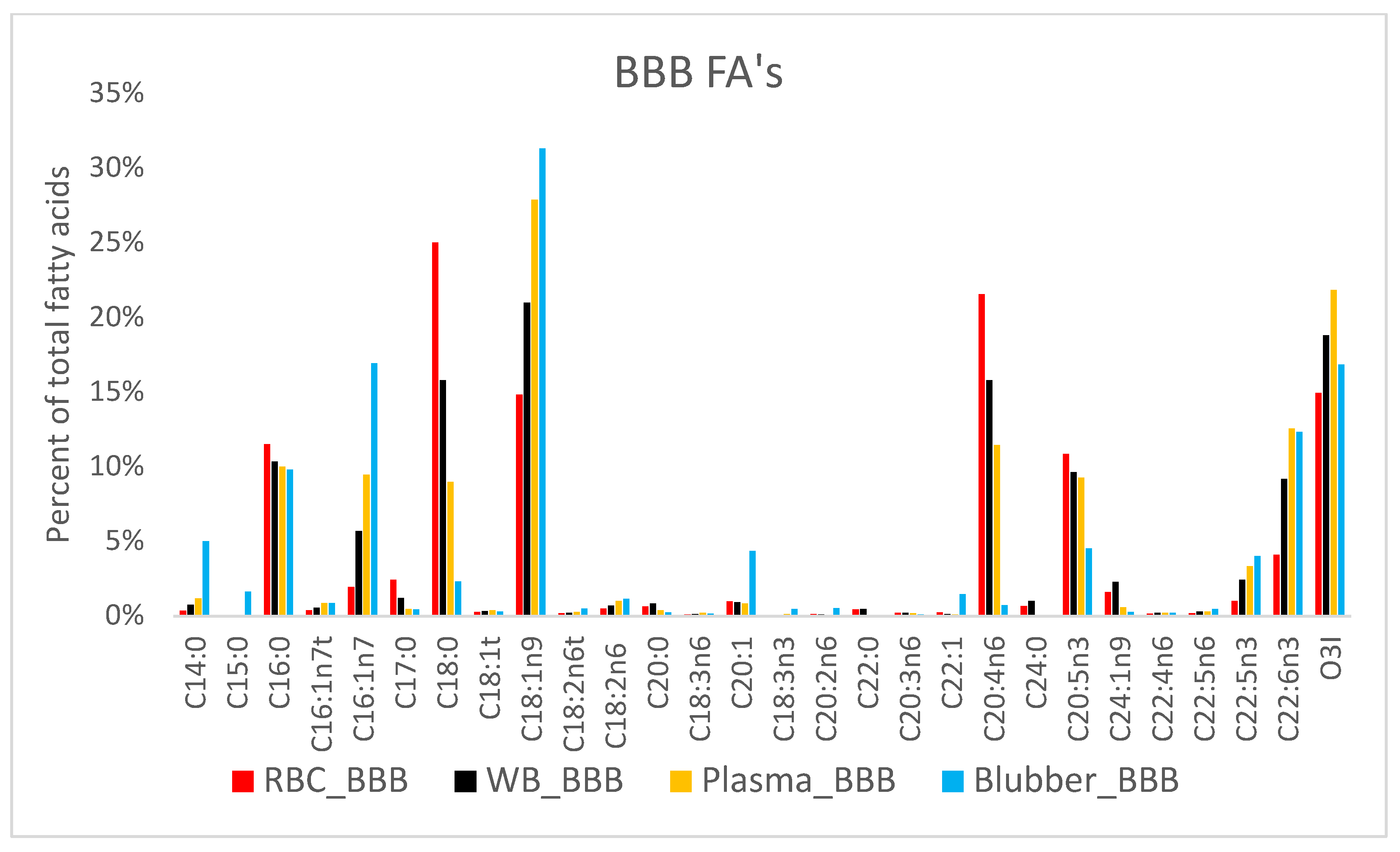
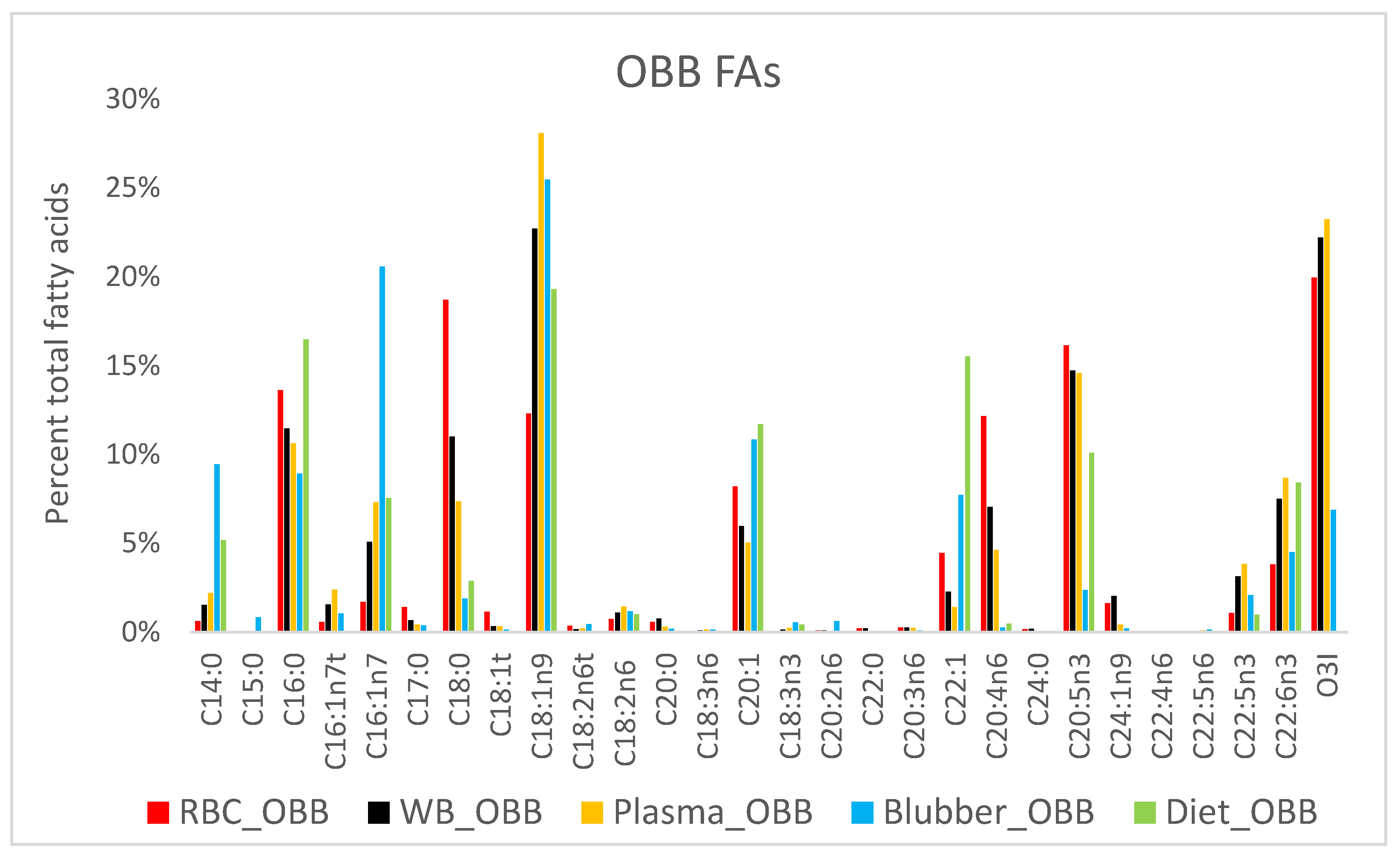
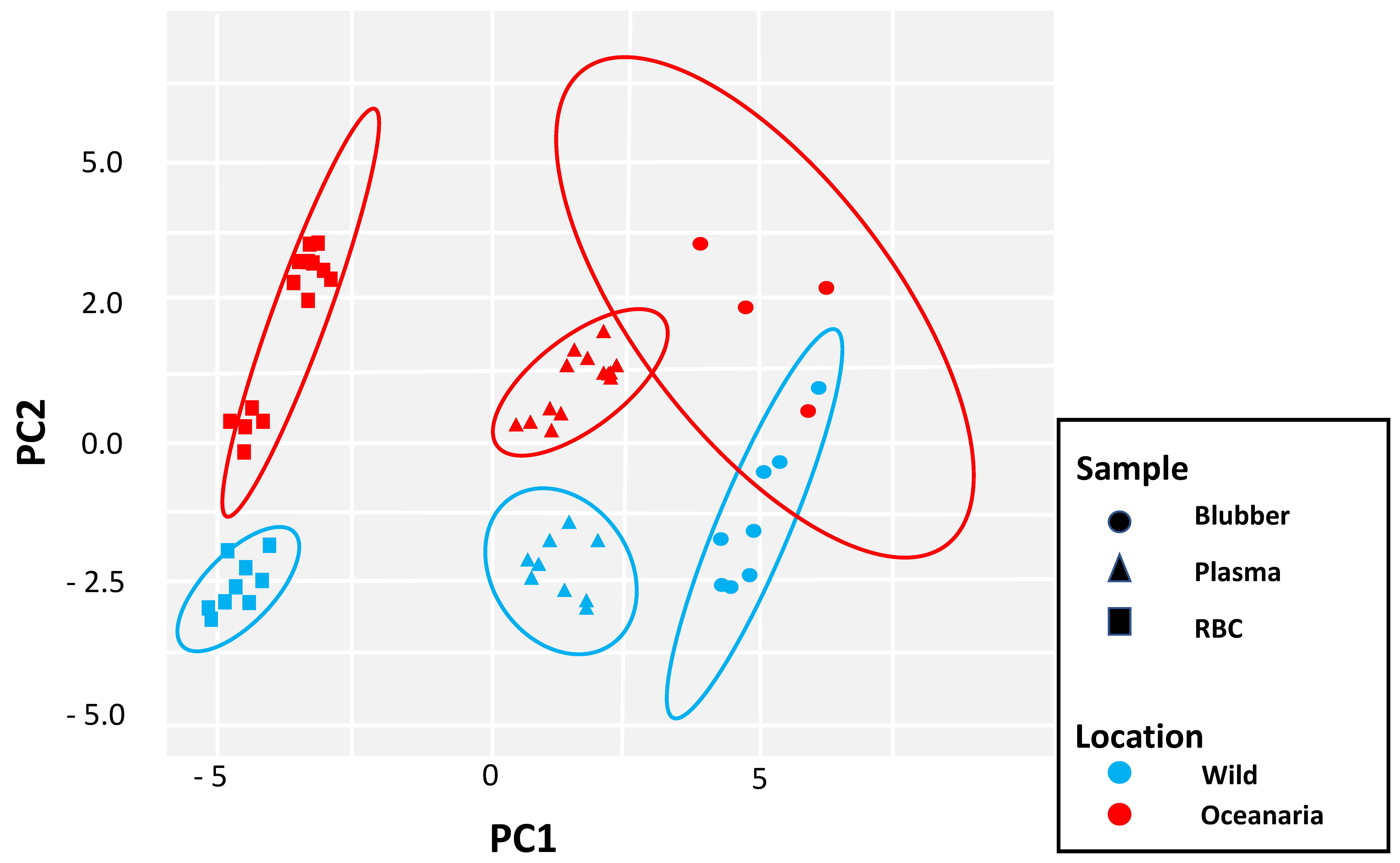
3. Results
4. Discussion
4.1. The Omega-3 Index
4.2. Potential Utility of Dried Blood Spot Testing in Marine Research
4.3. Fatty Acid Composition of Blubber
4.4. Potential Health Consequences of Fatty Acids
4.5. Ecological Dietary Differences
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dahl, T.M.; Lydersen, C.; Kovacs, K.M.; Falk-Petersen, S.; Sargent, J.; Gjertz, I.; Gulliksen, B. Fatty acid composition of the blubber in white whales (Delphinapterus leucas). Polar Biol. 2000, 23, 401–409. [Google Scholar] [CrossRef]
- Herman, D.P.; Burrow, D.G.; Wade, P.R.; Durban, J.W.; Matkin, C.O.; LeDuc, R.G.; Barrett-Lennard, L.G.; Krahn, M.M. Feeding ecology of eastern North Pacific killer whales Orcinus orca from fatty acid, stable isotope and organochlorine analyses of blubber biopsies. Mar. Ecol. Prog. Ser. 2005, 302, 275–291. [Google Scholar] [CrossRef]
- Koopman, H.N. Phylogenetic, ecological, and ontogenetic factors influencing the biochemical structure of the blubber of odontocetes. Mar. Biol. 2007, 151, 277–291. [Google Scholar] [CrossRef]
- Smith, H.R. Fatty Acid Variation in Beluga (Delphinapterus leucas) Blubber: Implications for Estimating Diet Using Fatty Acids. Ph.D. Dissertation, University of Washington, Seattle, WA, USA, 2009. [Google Scholar]
- Bourque, J.; Dietz, R.; Sonne, C.; St Leger, J.; Iverson, S.; Rosing-Asvid, A.; Hansen, M.; McKinney, M.A. Fedding habits of a new Arctic predator: Insight from full-depth blubber fatty acid signatures of Greenland, Faroe Islands, Denmark, and managed-care killer whales Orcinus orca. Mar. Ecol. Prog. Ser. 2018, 603, 1–12. [Google Scholar] [CrossRef]
- Iverson, S.J.; Field, C.; Bowen, W.D.; Blanchard, W. Quantitative fatty acid signature analysis: A new method of estimating predator diets. Ecol. Monogr. 2004, 74, 211–235. [Google Scholar] [CrossRef]
- Cornick, L.A.; Quakenbush, L.T.; Norman, S.A.; Pasi, C.; Maslyk, P.; Burek, K.A.; Goertz, C.E.C.; Hobbs, R.C. Seasonal and developmental differences in blubber stores of beluga whales in Bristol Bay, Alaska, using high-resolution ultrasound. J. Mammal. 2016, 97, 1238–1248. [Google Scholar] [CrossRef]
- Fenton, J.I.; Gurzell, E.A.; Davidson, E.A.; Harris, W.S. Red blood cell PUFAs reflect the phospholipid PUFA composition of major organs. Prostaglandins Leukot. Essent. Fat. Acids 2016, 112, 12–23. [Google Scholar] [CrossRef]
- Jackson, K.H.; Harris, W.S. Blood fatty acid profiles: New biomarkers for cardiometabolic disease risk. Curr. Atheroscler. Rep. 2018, 20, 22. [Google Scholar] [CrossRef]
- King, M.W. Omega Fatty-Acids. 2019. Available online: themedicalbiochemistrypage.org (accessed on 18 December 2019).
- Saini, R.K.; Keum, Y.-S. Omega-3 and omega-6 polyunsaturated fatty acids: Dietary sources, metabolisms and significance—A review. Life Sci. 2018, 203, 255–267. [Google Scholar] [CrossRef]
- Surette, M.E. The science behind dietary omega-3 fatty acids. Can. Med. Assoc. J. 2008, 178, 177–180. [Google Scholar] [CrossRef]
- Calder, P.C. Polyunsaturated fatty acids and inflammation. Prostaglandins Leukot. Essent. Fat. Acids 2006, 75, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Loseto, L.; Stern, G.; Connelly, T.L.; Deibel, D.; Gemmill, B.; Prokopowicz, A.; Fortier, L.; Ferguson, S. Summer diet of beluga whale inferred by fatty acid analysis of eastern Beaufort sea food web. J. Exp. Mar. Biol. Ecol. 2009, 374, 12–18. [Google Scholar] [CrossRef]
- Quakenbush, L.T.S.; Suydam, R.S.; Bryan, A.L.; Lowry, L.F.; Frost, K.J.; Mahoney, B.A. Diet of beluga whales (Delphinapterus leucas) in Alaska from stomach contents, March–November. Mar. Fish. Rev. 2015, 77, 70–74. [Google Scholar] [CrossRef]
- Seaman, G.A.; Lowry, L.F.; Frost, K.J. Foods of belukha whales (Delphinapterus leucas) in western Alaska. Cetology 1982, 44, 3–7. [Google Scholar]
- Harris, W.S.; Schmitt, T.L. Unexpected similarity in RBC, DHA and AA levels between bottlenose dolphins and humans. Prostaglandins Leukot. Essent. Fat. Acids 2014, 90, 55–59. [Google Scholar] [CrossRef]
- Citta, J.J.; Quakenbush, L.T.; Frost, K.J.; Lowry, L.; Hobbs, R.C.; Aderman, H. Movements of beluga whales (Delphinapterus leucas) in Bristol Bay, Alaska. Mar. Mammal. Sci 2016, 32, 1272–1298. [Google Scholar] [CrossRef]
- Norman, S.A.; Goertz, C.E.; Burek, K.A.; Quakenbush, L.T.; Cornick, L.A.; Romano, T.A.; Spoon, T.; Miller, W.; Beckett, L.A.; Hobbs, R.C. Seasonal hematology and serum chemistry of wild beluga whales (Delphinapterus leucas) in Bristol Bay, Alaska, USA. J. Wildl. Dis. 2012, 48, 21–32. [Google Scholar] [CrossRef]
- Harris, W.S.; Polreis, J. Measurement of the Omega-3 Index in Dried Blood Spots. Ann. Clin. Lab. Res. 2016, 4, 137–144. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane-Stanley, G.H. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Harris, W.S.; Von Schacky, C. The Omega-3 Index: A new risk factor for death from coronary artery disease? Prev. Med. 2004, 39, 212–220. [Google Scholar] [CrossRef]
- Kelley, T.C.; Loseto, L.L.; Stewart, R.E.A.; Yurkowski, M.; Ferguson, S.H. Importance of eating capelin: Unique dietary habits of Hudson Bay beluga. In A Little Less Arctic; Ferguson, S.H., Loseto, L.L., Mallory, M.L., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 53–70. [Google Scholar]
- Louis, M.; Skovrind, M.; Garde, E.; Heide-Jorgensen, M.P.; Szpak, P.; Lorenzen, E.D. Population-specific sex and size variation in long-term foraging ecology of belugas and narwhals. R. Soc. Open Sci. 2021, 8, 202226. [Google Scholar] [CrossRef] [PubMed]
- Choy, E.S.; Sheehan, B.; Haulena, M.; Rosenberg, B.; Roth, J.D.; Loseto, L.L. A comparison of diet estimates of captive beluga whales using fatty acid mixing models with their true diets. J. Exp. Mar. Biol. Ecol. 2019, 516, 132–139. [Google Scholar] [CrossRef]
- Cooper, M.H.; Iverson, S.J.; Rouvinen-Watt, K. Metabolism of dietary cetoleic acid (22:1n-11) in mink (Mustela vison) and gray seals (Halichoerus grypus) studied using radiolabeled fatty acids. Physiol. Biochem. Zool. 2006, 79, 820–829. [Google Scholar] [CrossRef]
- Sargent, J.R.; Falk-Petersen, S. The lipid biochemistry of calanoid copepods. Hydrobiologia 1988, 167, 101–114. [Google Scholar] [CrossRef]
- Bremer, J.; Norum, K.R. Metabolism of very long-chain monounsaturated fatty acids (22:1) and the adaptation to their presence in the diet. J. Lipid Res. 1982, 23, 243–256. [Google Scholar] [CrossRef]
- Ostbye, T.K.; Berge, G.M.; Nilsson, A.; Romarheim, O.H.; Bou, M.; Ruyter, B. The long-chain monounsaturated cetoleic acid improves the efficiency of the n-3 fatty acid metabolic pathway in Atlantic salmon and human HepG2 cells. Br. J. Nutr. 2019, 122, 755–768. [Google Scholar] [CrossRef] [PubMed]
- Elvevoll, E.O.; Eilertsen, K.-E.; Brox, J.; Dragnes, B.T.; Falkenberg, P.; Olsen, J.O.; Kirkhus, B.; Lamglait, A.; Osterud, B. Seafood diets: Hypolipidemic and antiatherogenic effects of taurine and n-3 fatty acids. Atherosclerosis 2008, 200, 396–402. [Google Scholar] [CrossRef]
- Strandberg, U.; Kakela, A.; Lydersen, C.; Kovacs, K.M.; Grahl-Nielsen, O.; Hyvarinen, H.; Kakela, R. Stratification, composition, and function of marine mammal blubber: The ecology of fatty acids in marine mammals. Phys. Biochem. Zool. 2008, 81, 473–485. [Google Scholar] [CrossRef]
- Hana, V.S.; Hafez, E.A.A. Synopsis of arachidonic acid metabolism: A review. J. Adv. Res. 2018, 11, 23–32. [Google Scholar] [CrossRef]
- Kusumoto, A.; Ishikura, Y.; Kawashima, H.; Kiso, Y.; Takai, S.; Miyazaki, M. Effects of arachidonate-enriched triacylglycerol supplementation on serum fatty acids and platelet aggregation in healthy male subjects with a fish diet. Br. Nutr. J. 2007, 98, 626–635. [Google Scholar] [CrossRef]
- Nelson, G.J.; Schmidt, P.C.; Bartolini, G.; Kelley, D.S.; Phinney, S.D.; Kyle, D.; Silbermann, S.; Schaefer, E.J. The effect of dietary arachidonic acid on plasma lipoprotein distributions, apoproteins, blood lipid levels, and tissue fatty acid composition in humans. Lipids 1997, 32, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Pottala, J.V.; Varvel, S.A.; Borowski, J.J.; Ward, J.N.; McConnell, J.P. Erythrocyte omega-3 fatty acids increase and linoleic acid decreases with age: Observations from 160,000 patients. Prostaglandins Leukot. Essent. Fat. Acids 2013, 88, 257–263. [Google Scholar] [CrossRef] [PubMed]
- De Caterina, R. N-3 fatty acids in cardiovascular disease. N. Engl. J. Med. 2011, 364, 2439–2450. [Google Scholar] [CrossRef]
- Fritsche, K. Fatty acids as modulators of the immune response. Annu. Rev. Nutr. 2006, 26, 45–73. [Google Scholar] [CrossRef] [PubMed]
- Whelan, J.; Rust, C. Innovative dietary sources of N-3 fatty acids. Annu. Rev. Nutr. 2006, 26, 75–103. [Google Scholar] [CrossRef]
- Wood, J.; Minter, L.J.; Bibus, D.; Stoskopf, M.K.; Fellner, V.; Ange-van Heugten, K. Comparison of African savanna elephant (Loxodonta africana) fatty acid profiles in whole blood, whole blood dried on blood spot cards, serum, and plasma. PeerJ 2021, 9, e12650. [Google Scholar] [CrossRef]
- Monge, A.; Harris, W.S.; Ortiz-Panozo, E.; Yunes, E.; Cantu-Brito, C.; Catzin-Kuhlmann, A.; Lopez-Ridaura, R.; Lajous, M. Whole Blood omega-3 Fatty Acids Are Inversely Associated with Carotid Intima-Media Thickness in Indigenous Mexican Women. Nutr. J. 2016, 146, 1365–1372. [Google Scholar] [CrossRef]
- Adjepong, M.; Yakah, W.; Harris, W.S.; Annan, R.A.; Pontifex, M.B.; Fenton, J.I. Whole blood n-3 fatty acids are associated with executive function in 2-6-year-old Northern Ghanaian children. J. Nutr. Biochem. 2018, 57, 287–293. [Google Scholar] [CrossRef]
- Jumbe, T.; Comstock, S.S.; Harris, W.S.; Kinabo, J.; Pontifex, M.B.; Fenton, J.I. Whole-blood fatty acids are associated with executive function in Tanzanian children aged 4-6 years: A cross-sectional study. Br. J. Nutr. 2016, 116, 1537–1545. [Google Scholar] [CrossRef]
- Nayak, U.; Kanungo, S.; Zhang, D.; Ross Colgate, E.; Carmolli, M.P.; Dey, A.; Alam, M.; Manna, B.; Nandy, R.K.; Kim, D.R.; et al. Influence of maternal and socioeconomic factors on breast milk fatty acid composition in urban, low-income families. Matern. Child Nutr. 2017, 13, e12423. [Google Scholar] [CrossRef]
- Krahn, M.M.; Herman, D.P.; Ylitalo, G.M.; Sloan, C.A.; Burrows, D.G.; Hobbs, R.C.; Mahoney, B.A.; Yanagida, G.K.; Calambokidis, J.; Moore, S.E. Stratification of lipids, fatty acids and organochlorine contaminants in blubber of white whales and killer whales. J. Cetacean Res. Manag. 2004, 6, 175–189. [Google Scholar]
- Fontes, J.D.; Rahman, F.; Lacey, S.; Larson, M.G.; Vasan, R.S.; Benjamin, E.J.; Harris, W.S.; Robins, S.J. Red blood cell fatty acids and biomarkers of inflammation: A cross-sectional study in a community-based cohort. Atherosclerosis 2015, 240, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Harris, W.S.; Chung, M.; Lichtenstein, A.H.; Balk, E.M.; Kupelnick, B.; Jordan, H.S.; Lau, J. N-3 fatty acids from fish or fish-oil supplements, but not α-linolenic acid, benefit cardiovascular disease outcomes in primary- and secondary-prevention studies: A systematic review. Am. J. Clin. Nutr. 2006, 84, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Marcoux, M.; McMeans, B.C.; Fisk, A.T.; Ferguson, S.H. Composition and temporal variation in the diet of beluga whales, derived from stable isotopes. Mar. Ecol. Prog. Ser. 2012, 471, 283–291. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schmitt, T.L.; Goertz, C.E.C.; Hobbs, R.C.; Osborn, S.; DiRocco, S.; Bissell, H.; Harris, W.S. Erythrocyte, Whole Blood, Plasma, and Blubber Fatty Acid Profiles in Oceanaria-Based versus Wild Alaskan Belugas (Delphinapterus leucas). Oceans 2022, 3, 464-479. https://doi.org/10.3390/oceans3040031
Schmitt TL, Goertz CEC, Hobbs RC, Osborn S, DiRocco S, Bissell H, Harris WS. Erythrocyte, Whole Blood, Plasma, and Blubber Fatty Acid Profiles in Oceanaria-Based versus Wild Alaskan Belugas (Delphinapterus leucas). Oceans. 2022; 3(4):464-479. https://doi.org/10.3390/oceans3040031
Chicago/Turabian StyleSchmitt, Todd L., Caroline E. C. Goertz, Roderick C. Hobbs, Steve Osborn, Stacy DiRocco, Heidi Bissell, and William S. Harris. 2022. "Erythrocyte, Whole Blood, Plasma, and Blubber Fatty Acid Profiles in Oceanaria-Based versus Wild Alaskan Belugas (Delphinapterus leucas)" Oceans 3, no. 4: 464-479. https://doi.org/10.3390/oceans3040031
APA StyleSchmitt, T. L., Goertz, C. E. C., Hobbs, R. C., Osborn, S., DiRocco, S., Bissell, H., & Harris, W. S. (2022). Erythrocyte, Whole Blood, Plasma, and Blubber Fatty Acid Profiles in Oceanaria-Based versus Wild Alaskan Belugas (Delphinapterus leucas). Oceans, 3(4), 464-479. https://doi.org/10.3390/oceans3040031







