Analysis of Dairy Cow Behavior during Milking Associated with Lameness
Abstract
:1. Introduction
2. Materials and Methods
2.1. Farm Recruitment and Selected Milkings
2.2. Data collection and Parameters
2.2.1. Lameness Assessment
2.2.2. Hind Leg Activity
Technical Details
Limitations of the Sensor-Measurement
2.2.3. Front Leg Activity and Head Movements
2.3. Statistical Analyses
3. Results
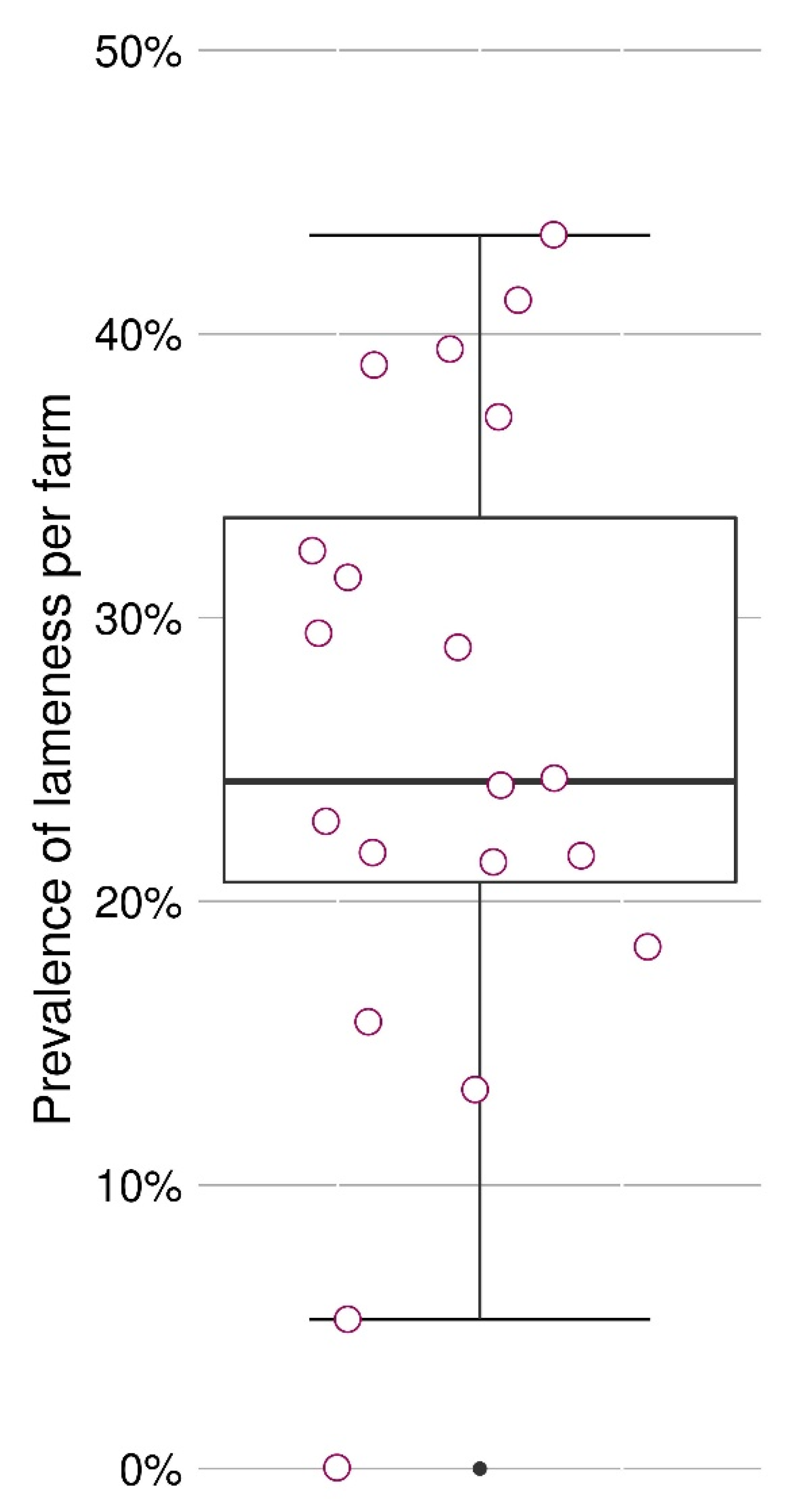
3.1. Prevalence of Lameness
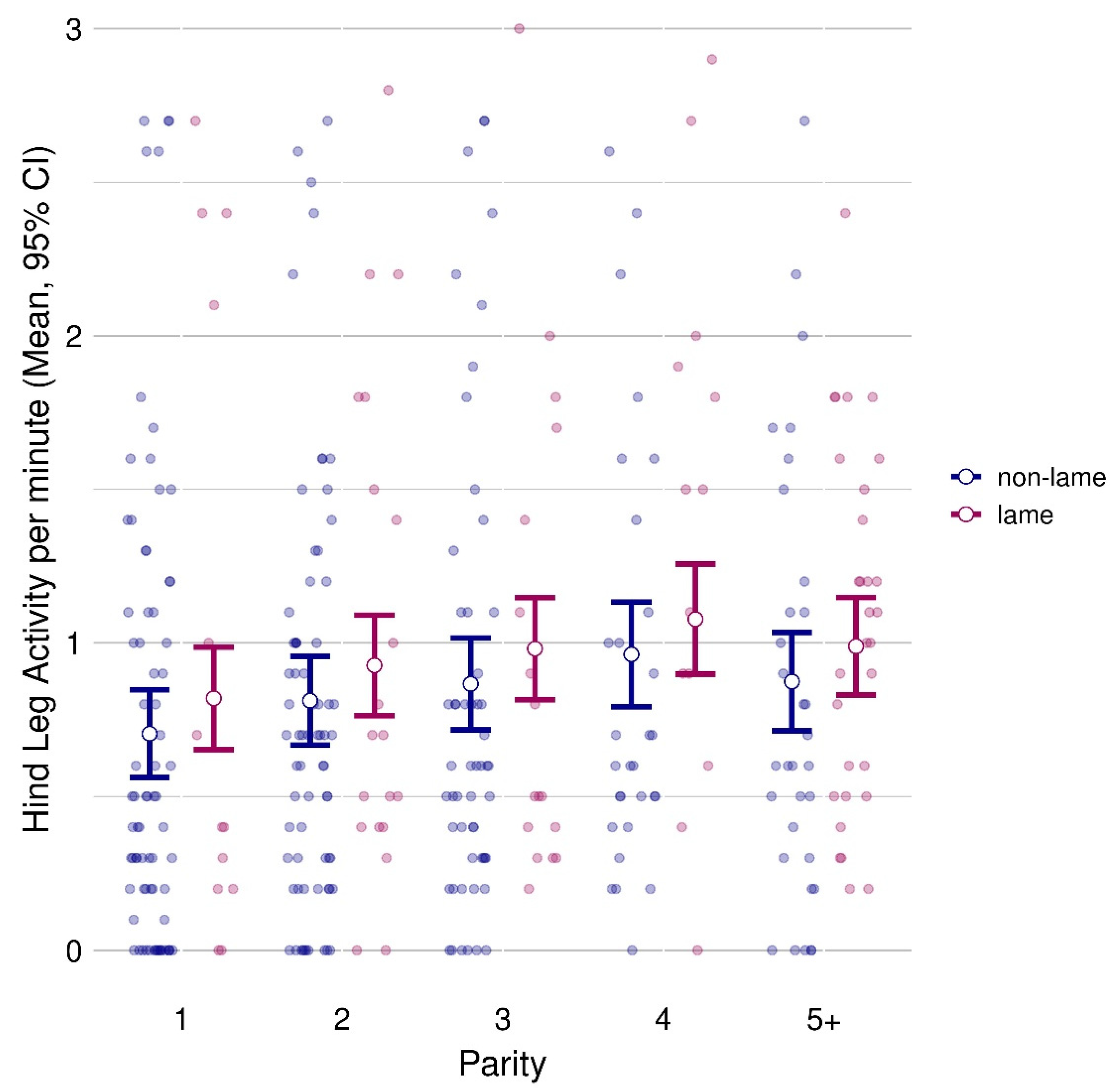
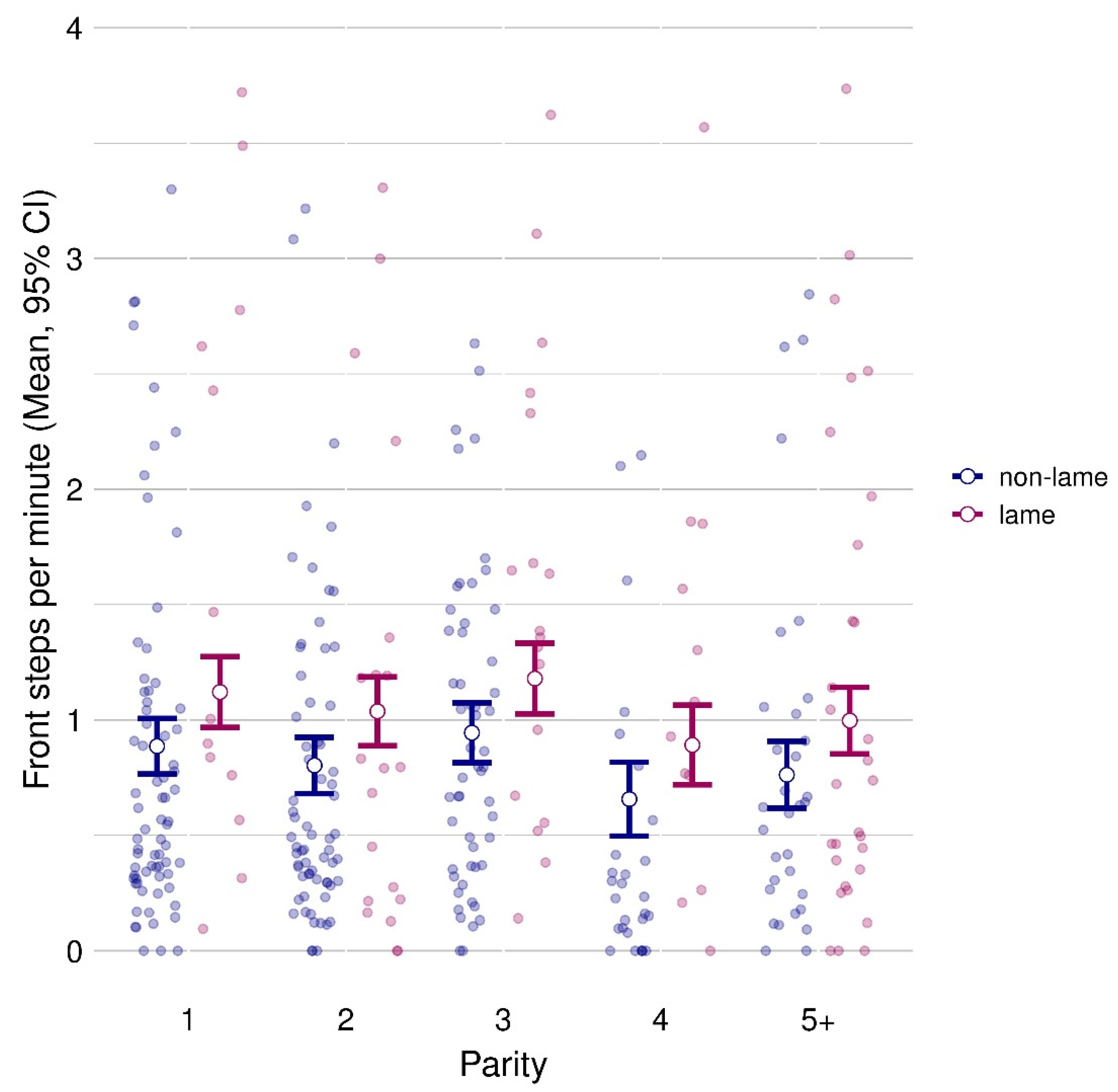
3.2. Influence of Parity on Lameness
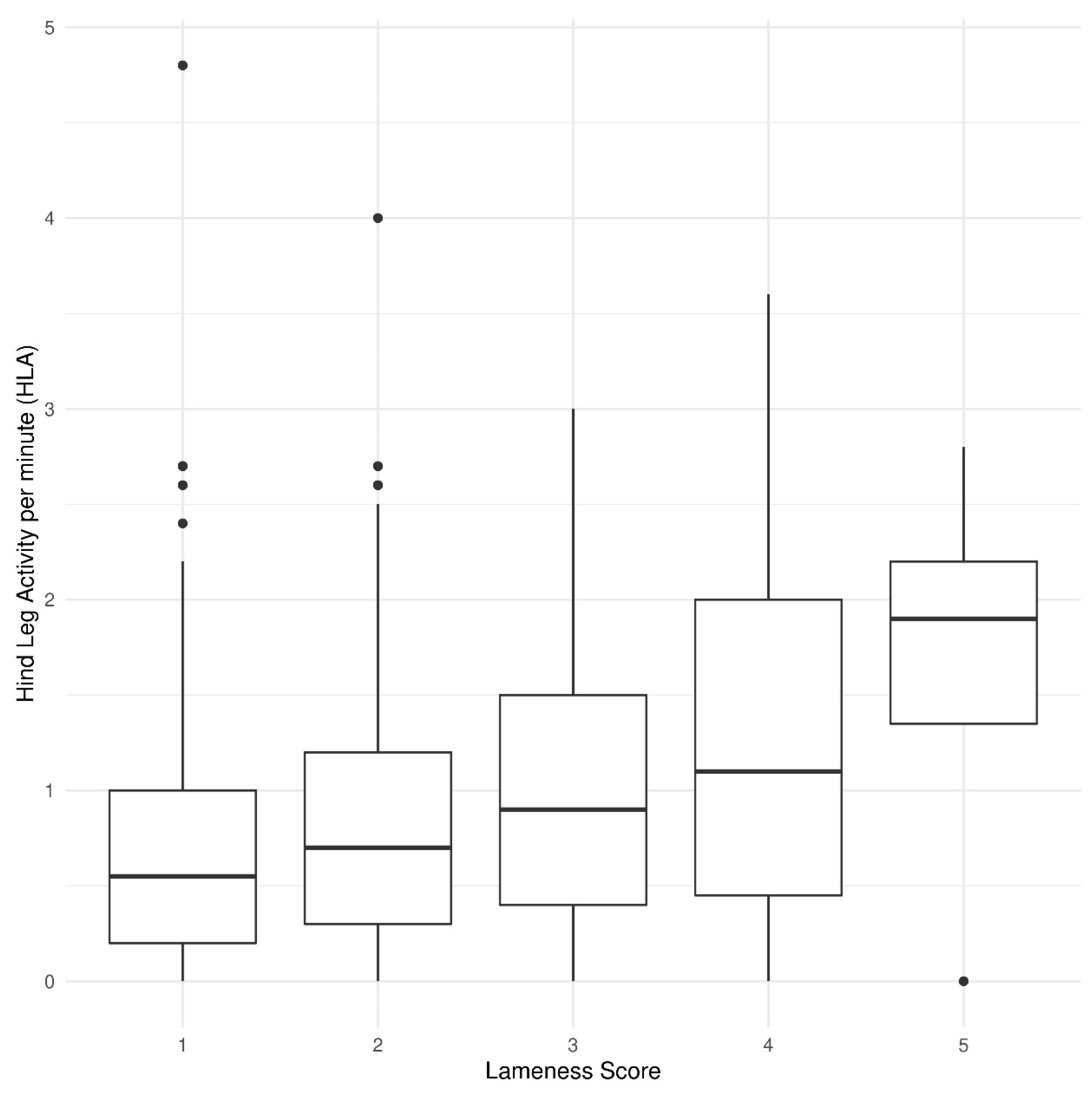
3.3. Hind Leg Activity and Front Steps
3.4. Head Movements
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
Appendix A
| Threshold [g] | TN | FN | FP | TP | Sensitivity | Specificity | Positive Predictive Value | Negative Predictive Value |
|---|---|---|---|---|---|---|---|---|
| 0.15 | 3825 | 18 | 432 | 246 | 0.93 | 0.90 | 0.36 | 1.00 |
| 0.2 | 4051 | 48 | 242 | 210 | 0.81 | 0.94 | 0.46 | 0.99 |
| 0.25 | 4197 | 76 | 125 | 181 | 0.70 | 0.97 | 0.59 | 0.98 |
| 0.3 | 4289 | 97 | 58 | 156 | 0.62 | 0.99 | 0.73 | 0.98 |
| 0.35 | 4346 | 118 | 27 | 130 | 0.52 | 0.99 | 0.83 | 0.97 |
| 0.4 | 4250 | 128 | 13 | 116 | 0.48 | 1.00 | 0.90 | 0.97 |
Appendix B
References
- Becker, J.; Steiner, A.; Kohler, S.; Koller-Bähler, A.; Wüthrich, M.; Reist, M. Lameness and foot lesions in Swiss dairy cows: I. Prevalence. Schweiz. Arch. Tierheilkd. 2014, 156, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Sarjokari, K.; Kaustell, K.O.; Hurme, T.; Kivinen, T.; Peltoniemi, O.A.T.; Saloniemi, H.; Rajala-Schultz, P.J. Prevalence and risk factors for lameness in insulated free stall barns in Finland. Livest. Sci. 2013, 156, 44–52. [Google Scholar] [CrossRef]
- Matson, R.D.; King, M.T.M.; Duffield, T.F.; Santschi, D.E.; Orsel, K.; Pajor, E.A.; Penner, G.B.; Mutsvangwa, T.; DeVries, T.J. Farm-level factors associated with lameness prevalence, productivity, and milk quality in farms with automated milking systems. J. Dairy Sci. 2022, 105, 793–806. [Google Scholar] [CrossRef]
- Juarez, S.T.; Robinson, P.H.; DePeters, E.J.; Price, E.O. Impact of lameness on behavior and productivity of lactating Holstein cows. Appl. Anim. Behav. Sci. 2003, 83, 1–14. [Google Scholar] [CrossRef]
- Green, L.E.; Hedges, V.J.; Schukken, Y.H.; Blowey, R.W.; Packington, A.J. The Impact of Clinical Lameness on the Milk Yield of Dairy Cows. J. Dairy Sci. 2002, 85, 2250–2256. [Google Scholar] [CrossRef]
- Sprecher, D.J.; Hostetler, D.E.; Kaneene, J.B. A lameness scoring system that uses posture and gait to predict dairy cattle reproductive performance. Theriogenology 1997, 47, 1179–1187. [Google Scholar] [CrossRef]
- Garbarino, E.J.; Hernandez, J.A.; Shearer, J.K.; Risco, C.A.; Thatcher, W.W. Effect of Lameness on Ovarian Activity in Postpartum Holstein Cows. J. Dairy Sci. 2004, 87, 4123–4131. [Google Scholar] [CrossRef]
- Huxley, J.N. Impact of lameness and claw lesions in cows on health and production. Livest. Sci. 2013, 156, 64–70. [Google Scholar] [CrossRef]
- Whay, H.R.; Shearer, J.K. The Impact of Lameness on Welfare of the Dairy Cow. Vet. Clin. N. Am. Food Anim. Pract. 2017, 33, 153–164. [Google Scholar] [CrossRef]
- Weigele, H.C.; Gygax, L.; Steiner, A.; Wechsler, B.; Burla, J.B. Moderate lameness leads to marked behavioral changes in dairy cows. J. Dairy Sci. 2018, 101, 2370–2382. [Google Scholar] [CrossRef]
- Bulgarelli-Jimenez, G.; Dercks, K.; van Amerongen, J.; Schukken, Y.; van Nielen, M. A Hind feet position scoring-system to monitor subclinical lameness in Dutch Holstein-Friesian cows. In Proceedings of the 9th International Symposium on Disorders of the Ruminant Digit, Jerusalem, Israel, 15–18 April 1996. [Google Scholar]
- Hoffman, A.C.; Moore, D.A.; Vanegas, J.; Wenz, J.R. Association of abnormal hind-limb postures and back arch with gait abnormality in dairy cattle. J. Dairy Sci. 2014, 97, 2178–2185. [Google Scholar] [CrossRef] [PubMed]
- Raven, E.T. The Principles of Claw Trimming. Vet. Clin. N. Am. Food Anim. Pract. 1985, 1, 93–107. [Google Scholar] [CrossRef] [PubMed]
- Chapinal, N.; Tucker, C.B. Validation of an automated method to count steps while cows stand on a weighing platform and its application as a measure to detect lameness. J. Dairy Sci. 2012, 95, 6523–6528. [Google Scholar] [CrossRef] [PubMed]
- Leach, K.A.; Dippel, S.; Huber, J.; March, S.; Winckler, C.; Whay, H.R. Assessing lameness in cows kept in tie-stalls. J. Dairy Sci. 2009, 92, 1567–1574. [Google Scholar] [CrossRef] [PubMed]
- Rushen, J.; Pombourcq, E.; de Passillé, A.M. Local anaesthetic as a means to validate measures of lameness in dairy cows. In Proceedings of the 40th International Congress of ISAE, Bristol, UK, 8–12 August 2006; p. 53. [Google Scholar]
- Werema, C.W.; Yang, D.A.; Laven, L.J.; Mueller, K.R.; Laven, R.A. Evaluating Alternatives to Locomotion Scoring for Detecting Lameness in Pasture-Based Dairy Cattle in New Zealand: In-Parlour Scoring. Animals 2022, 12, 703. [Google Scholar] [CrossRef]
- Alsaaod, M.; Fadul, M.; Steiner, A. Automatic lameness detection in cattle. Vet. J. 2019, 246, 35–44. [Google Scholar] [CrossRef]
- Han, S.; Zhang, J.; Cheng, G.; Peng, Y.; Zhang, J.; Wu, J. Current State and Challenges of Automatic Lameness Detection in Dairy Cattle. Smart Agric. 2020, 2, 21–36. [Google Scholar] [CrossRef]
- Rajkondawar, P.G.; Lefcourt, A.M.; Neerchal, N.K.; Dyer, R.M.; Varner, M.A.; Erez, B.; Tasch, U. The development of an objective lameness scoring system for dairy herds: Pilot study. Trans. ASAE 2002, 45, 1123–1125. [Google Scholar] [CrossRef]
- Pastell, M.; Aisla, A.-M.; Hautala, M.; Poikalainen, V.; Praks, J.; Veermäe, I.; Ahokas, J. Contactless measurement of cow behavior in a milking robot. Behav. Res. Methods 2006, 38, 479–486. [Google Scholar] [CrossRef]
- Poursaberi, A.; Bahr, C.; Pluk, A.; Van Nuffel, A.; Berckmans, D. Real-time automatic lameness detection based on back posture extraction in dairy cattle: Shape analysis of cow with image processing techniques. Comput. Electron. Agric. 2010, 74, 110–119. [Google Scholar] [CrossRef]
- Kang, X.; Zhang, X.D.; Liu, G. Accurate detection of lameness in dairy cattle with computer vision: A new and individualized detection strategy based on the analysis of the supporting phase. J. Dairy Sci. 2020, 103, 10628–10638. [Google Scholar] [CrossRef] [PubMed]
- Abdul Jabbar, A.; Hansen, M.F.; Smith, M.L.; Smith, L.N. Early and non-intrusive lameness detection in dairy cows using 3-dimensional video. Biosyst. Eng. 2017, 153, 63–69. [Google Scholar] [CrossRef]
- Jiang, B.; Song, H.; Wang, H.; Li, C. Dairy cow lameness detection using a back curvature feature. Comput. Electron. Agric. 2022, 194, 106729. [Google Scholar] [CrossRef]
- O’Leary, N.W.; Byrne, D.T.; O’Connor, A.H.; Shalloo, L. Invited review: Cattle lameness detection with accelerometers. J. Dairy Sci. 2020, 103, 3895–3911. [Google Scholar] [CrossRef]
- Thorup, V.M.; Munksgaard, L.; Robert, P.E.; Erhard, H.W.; Thomsen, P.T.; Friggens, N.C. Lameness detection via leg-mounted accelerometers on dairy cows on four commercial farms. Animal 2015, 9, 1704–1712. [Google Scholar] [CrossRef]
- Riaboff, L.; Relun, A.; Petiot, C.E.; Feuilloy, M.; Couvreur, S.; Madouasse, A. Identification of discriminating behavioural and movement variables in lameness scores of dairy cows at pasture from accelerometer and GPS sensors using a Partial Least Squares Discriminant Analysis. Prev. Vet. Med. 2021, 193, 105383. [Google Scholar] [CrossRef]
- García-Muñoz, A.; Vidal, G.; Singh, N.; Silva-del-Río, N. Evaluation of two methodologies for lameness detection in dairy cows based on postural and gait abnormalities observed during milking and while restrained at headlock stanchions. Prev. Vet. Med. 2016, 128, 33–40. [Google Scholar] [CrossRef]
- Thomsen, P.T.; Munksgaard, L.; Tøgersen, F.A. Evaluation of a Lameness Scoring System for Dairy Cows. J. Dairy Sci. 2008, 91, 119–126. [Google Scholar] [CrossRef]
- Winckler, C.; Willen, S. The Reliability and Repeatability of a Lameness Scoring System for Use as an Indicator of Welfare in Dairy Cattle. Acta Agric. Scand. A Anim. Sci. 2001, 51, 103–107. [Google Scholar] [CrossRef]
- Raoult, C.M.C.; Margerit, A.A.; Fricker, S.; Blümel, F.E.; Savary, P.E. Detection of cow hind-leg activity during milking by using a 3-dimensional accelerometer attached to the milking cluster. JDS Commun. 2021, 2, 55–60. [Google Scholar] [CrossRef]
- Friard, O.; Gamba, M. BORIS: A free, versatile open-source event-logging software for video/audio coding and live observations. Methods Ecol. Evol. 2016, 7, 1325–1330. [Google Scholar] [CrossRef]
- de Oliveira, D.; Keeling, L.J. Routine activities and emotion in the life of dairy cows: Integrating body language into an affective state framework. PLoS ONE 2018, 13, e0195674. [Google Scholar] [CrossRef] [PubMed]
- Posit-Team. RStudio: Integrated Development Environment for R, Posit Software; PBC: Boston, MA, USA, 2022.
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- R-Core-Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2022. [Google Scholar]
- Fox, J.; Weisberg, S. An R Companion to Applied Regression, 3rd ed.; SAGE Publications, Inc.: Thousand Oaks, CA, USA, 2019. [Google Scholar]
- Fox, J. Effect Displays in R for Generalised Linear Models. J. Stat. Softw. 2003, 8, 1–27. [Google Scholar] [CrossRef]
- Hartig, F. DHARMa: Residual Diagnostics for Hierarchical (Multi-Level/Mixed) Regression Models, R Package Version 0.4.5. 2022. Available online: https://cran.r-project.org/package=DHARMa (accessed on 15 January 2023).
- Lenth, R.V. emmeans: Estimated Marginal Means, aka Least-Squares Means, R Package Version 1.8.4-1. 2023. Available online: https://cran.r-project.org/package=emmeans (accessed on 15 January 2023).
- Griffiths, B.E.; Grove White, D.; Oikonomou, G. A Cross-Sectional Study Into the Prevalence of Dairy Cattle Lameness and Associated Herd-Level Risk Factors in England and Wales. Front. Vet. Sci. 2018, 5, 65. [Google Scholar] [CrossRef]
- Manske, T.; Hultgren, J.; Bergsten, C. The effect of claw trimming on the hoof health of Swedish dairy cattle. Prev. Vet. Med. 2002, 54, 113–129. [Google Scholar] [CrossRef]
- Olechnowicz, J.; Jaśkowski, J.M. Relationship between clinical lameness and somatic cell counts, and fat and protein contents in the milk of dairy cows. Med. Weter. 2012, 68, 740–743. [Google Scholar]
- Archer, S.C.; Green, M.J.; Madouasse, A.; Huxley, J.N. Association between somatic cell count and serial locomotion score assessments in UK dairy cows. J. Dairy Sci. 2011, 94, 4383–4388. [Google Scholar] [CrossRef]
- Klindworth, H.-P.; Schranner, A.; Ullrich, A.; Kaske, M. Prevalence of lameness in dairy herds in Lower Saxony—Investigations as part of the Animal Welfare Plan. Berl. Münchener Tierärztliche Wochenschr. 2018, 131, 465. [Google Scholar] [CrossRef]
- Köck, A.; Fuerst-Waltl, B.; Kofler, J.; Burgstaller, J.; Steininger, F.; Fuerst, C.; Egger-Danner, C. Short communication: Use of lameness scoring to genetically improve claw health in Austrian Fleckvieh, Brown Swiss, and Holstein cattle. J. Dairy Sci. 2019, 102, 1397–1401. [Google Scholar] [CrossRef]
- Holzhauer, M.; Hardenberg, C.; Bartels, C.J.M.; Frankena, K. Herd- and Cow-Level Prevalence of Digital Dermatitis in The Netherlands and Associated Risk Factors. J. Dairy Sci. 2006, 89, 580–588. [Google Scholar] [CrossRef]
- Solano, L.; Barkema, H.W.; Pajor, E.A.; Mason, S.; LeBlanc, S.J.; Zaffino Heyerhoff, J.C.; Nash, C.G.R.; Haley, D.B.; Vasseur, E.; Pellerin, D.; et al. Prevalence of lameness and associated risk factors in Canadian Holstein-Friesian cows housed in freestall barns. J. Dairy Sci. 2015, 98, 6978–6991. [Google Scholar] [CrossRef]
- Olechnowicz, J.; Jaśkowski, J.M. Reasons for culling, culling due to lameness, and economic losses in dairy cows. Med. Weter. 2011, 67, 618–621. [Google Scholar]
- Länge der Nutzungsdauer [Internet]. Plattform Tierstatistik, Identitas AG. 2023. Available online: https://tierstatistik.identitas.ch/de/cattle-productivelife.html (accessed on 22 February 2023).
- Weaver, A.D.; Andersson, L.; De Laistre Banting, A.; Demerzis, P.N.; Knezevic, P.F.; Peterse, D.J.; Sankovic, F. Review of disorders of the ruminant digit with proposals for anatomical and pathological terminology and recording. Vet. Rec. 1981, 108, 117–120. [Google Scholar] [CrossRef]
- Murray, R.D.; Downham, D.Y.; Clarkson, M.J.; Faull, W.B.; Hughes, J.W.; Manson, F.J.; Merritt, J.B.; Russell, W.B.; Sutherst, J.E.; Ward, W.R. Epidemiology of lameness in dairy cattle: Description and analysis of foot lesions. Vet. Rec. 1996, 138, 586–591. [Google Scholar] [CrossRef]
- Endres, M.I. The Relationship of Cow Comfort and Flooring to Lameness Disorders in Dairy Cattle. Vet. Clin. N. Am. Food Anim. Pract. 2017, 33, 227–233. [Google Scholar] [CrossRef]
- Becker, J.; Steiner, A.; Kohler, S.; Koller-Bähler, A.; Wüthrich, M.; Reist, M. Lameness and foot lesions in Swiss dairy cows: II. Risk Factors. Schweiz. Arch. Tierheilkd. 2014, 156, 79–89. [Google Scholar] [CrossRef]
- Lean, I.J.; Westwood, C.T.; Golder, H.M.; Vermunt, J.J. Impact of nutrition on lameness and claw health in cattle. Livest. Sci. 2013, 156, 71–87. [Google Scholar] [CrossRef]
- Gygax, L.; Neuffer, I.; Kaufmann, C.; Hauser, R.; Wechsler, B. Restlessness behaviour, heart rate and heart-rate variability of dairy cows milked in two types of automatic milking systems and auto-tandem milking parlours. Appl. Anim. Behav. Sci. 2008, 109, 167–179. [Google Scholar] [CrossRef]
- Cerqueira, J.O.L.; Araújo, J.P.P.; Blanco-Penedo, I.; Cantalapiedra, J.; Sørensen, J.T.; Niza-Ribeiro, J.J.R. Relationship between stepping and kicking behavior and milking management in dairy cattle herds. J. Vet. Behav. 2017, 19, 72–77. [Google Scholar] [CrossRef]
- ICAR. International Agreement of Recording Practices; Guidelines approved by the general assembly, Riga, Latvia, June 2010; International Committee for Animal Recording: Rome, Italy, 2012. [Google Scholar]
- Oshin, M.; Deb, S.; Kadyan, S.; Pb, N.; Kumar, S. Non genetic factors affecting udder type traits in Sahiwal cattle. Indian J. Dairy Sci. 2022, 75, 347–352. [Google Scholar] [CrossRef]
- Kuczaj, M. Analysis of changes in udder size of high-yelding cows in subsequent lactations with regard to mastitis. Electron. J. Pol. Agric. Univ. 2003, 6, 1–10. [Google Scholar]
- Rousing, T.; Bonde, M.; Badsberg, J.H.; Sørensen, J.T. Stepping and kicking behaviour during milking in relation to response in human–animal interaction test and clinical health in loose housed dairy cows. Livest. Prod. Sci. 2004, 88, 1–8. [Google Scholar] [CrossRef]
- Fallahi, S. Behavioral genetics in Cattle—A review. J. Livest. Sci. 2019, 10, 102–108. [Google Scholar] [CrossRef]
- Manske, T.; Hultgren, J.; Bergsten, C. Prevalence and interrelationships of hoof lesions and lameness in Swedish dairy cows. Prev. Vet. Med. 2002, 54, 247–263. [Google Scholar] [CrossRef]
- Neveux, S.; Weary, D.M.; Rushen, J.; Von Keyserlingk, M.A.G.; De Passillé, A.M. Hoof discomfort changes how dairy cattle distribute their body weight. J. Dairy Sci. 2006, 89, 2503–2509. [Google Scholar] [CrossRef]
- Proudfoot, K.L.; Huzzey, J.M. A first time for everything: The influence of parity on the behavior of transition dairy cows. JDS Commun. 2022, 3, 467–471. [Google Scholar] [CrossRef]

| Category | Score and Brief Term | Description |
|---|---|---|
| Non-lame | 1. Normal | Normal gait pattern with no signs of irregularities or uneven weight bearing between legs. In most cases flat back while walking and standing. |
| 2. Uneven gait | Cow walks almost normally but with a slightly uneven gait. No evident signs of lameness. Back may be arched while walking but flat when standing. | |
| Lame | 3. Mild lameness | Deviation from the regular gait rhythm owing to uneven load on the legs and shortened stride on one or more legs. In most cases arched back while walking and head carried lower than normal. Often difficult to be certain which leg is affected. |
| 4. Lameness | Obvious lameness with in most cases arched back while walking and standing with clear attempt to unload one or more legs. In most cases head bob while walking. Usually obvious which legs are affected. | |
| 5. Severe lameness | Cow is reluctant or unable to bear weight on the affected leg while walking or standing. In most cases arched back while walking or standing and head bob while walking. |
| Body Part | Behavior | Definition |
|---|---|---|
| Head | Horizontal movement | Head movement either to the left or the right side or back to the center, no matter how far the movement reaches |
| Vertical movement | Head movement up or down into another range 1, no matter how many ranges the head crosses. Therefor dividing the cow’s head position into four ranges 1: 1. Head and neck are held above the horizontal line of the back 2. Head and neck are equal or nearly equal to the imaginary straight line of the back 3. Head and neck are bent down at chest level 4. Head and neck are bent down nearly completely | |
| Hoicking | Big rapid expulsive head movement upwards | |
| Twitching | Tiny choppy head movement without changing the head position | |
| Shaking | Fast rhythmic circular head movements | |
| Scratching | Rubbing the head against a component of the milking box | |
| Front legs | Stepping | Lifting or shifting the claw |
| Kicking | Rapid expulsive movement of the claw with the aim of striking towards the abdomen or the milking cluster |
| Behavior (per Minute) | Lameness | Parity | Farm-to-Farm Variability (%) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean ± SE | p-Value | Mean ± SE | p-Value | |||||||
| Non-Lame | Lame | 1 | 2 | 3 | 4 | 5+ | ||||
| Hind leg activity | 0.844 ± 0.064 | 0.959 ± 0.073 | 0.026 | 0.762 ± 0.075 | 0.869 ± 0.075 | 0.924 ± 0.077 | 1.019 ± 0.086 | 0.932 ± 0.077 | 0.008 | 23.9 |
| Front steps | 0.810 ± 0.048 | 1.045 ± 0.061 | 0.00005 | 1.003 ± 0.065 | 0.919 ± 0.064 | 1.062 ± 0.067 | 0.774 ± 0.080 | 0.879 ± 0.068 | 0.009 | 8.7 |
| Horizontal head movements | 1.429 ± 0.061 | 1.465 ± 0.072 | 0.551 | 1.636 ± 0.076 | 1.498 ± 0.075 | 1.449 ± 0.078 | 1.282 ± 0.089 | 1.372 ± 0.079 | 0.0006 | 13.4 |
| Vertical head movements | 1.054 ± 0.046 | 1.086 ± 0.061 | 0.600 | 1.197 ± 0.065 | 1.153 ± 0.064 | 1.127 ± 0.068 | 0.879 ± 0.083 | 0.995 ± 0.069 | 0.004 | 6.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schönberger, D.; Berthel, R.M.; Savary, P.; Bodmer, M. Analysis of Dairy Cow Behavior during Milking Associated with Lameness. Dairy 2023, 4, 554-570. https://doi.org/10.3390/dairy4040038
Schönberger D, Berthel RM, Savary P, Bodmer M. Analysis of Dairy Cow Behavior during Milking Associated with Lameness. Dairy. 2023; 4(4):554-570. https://doi.org/10.3390/dairy4040038
Chicago/Turabian StyleSchönberger, Diana, Roxanne Magali Berthel, Pascal Savary, and Michèle Bodmer. 2023. "Analysis of Dairy Cow Behavior during Milking Associated with Lameness" Dairy 4, no. 4: 554-570. https://doi.org/10.3390/dairy4040038
APA StyleSchönberger, D., Berthel, R. M., Savary, P., & Bodmer, M. (2023). Analysis of Dairy Cow Behavior during Milking Associated with Lameness. Dairy, 4(4), 554-570. https://doi.org/10.3390/dairy4040038






