Bovine Colostrum for Human Consumption—Improving Microbial Quality and Maintaining Bioactive Characteristics through Processing
Abstract
1. Background on Bovine Colostrum, Contaminants, and Processing
1.1. Contamination
1.2. Processing
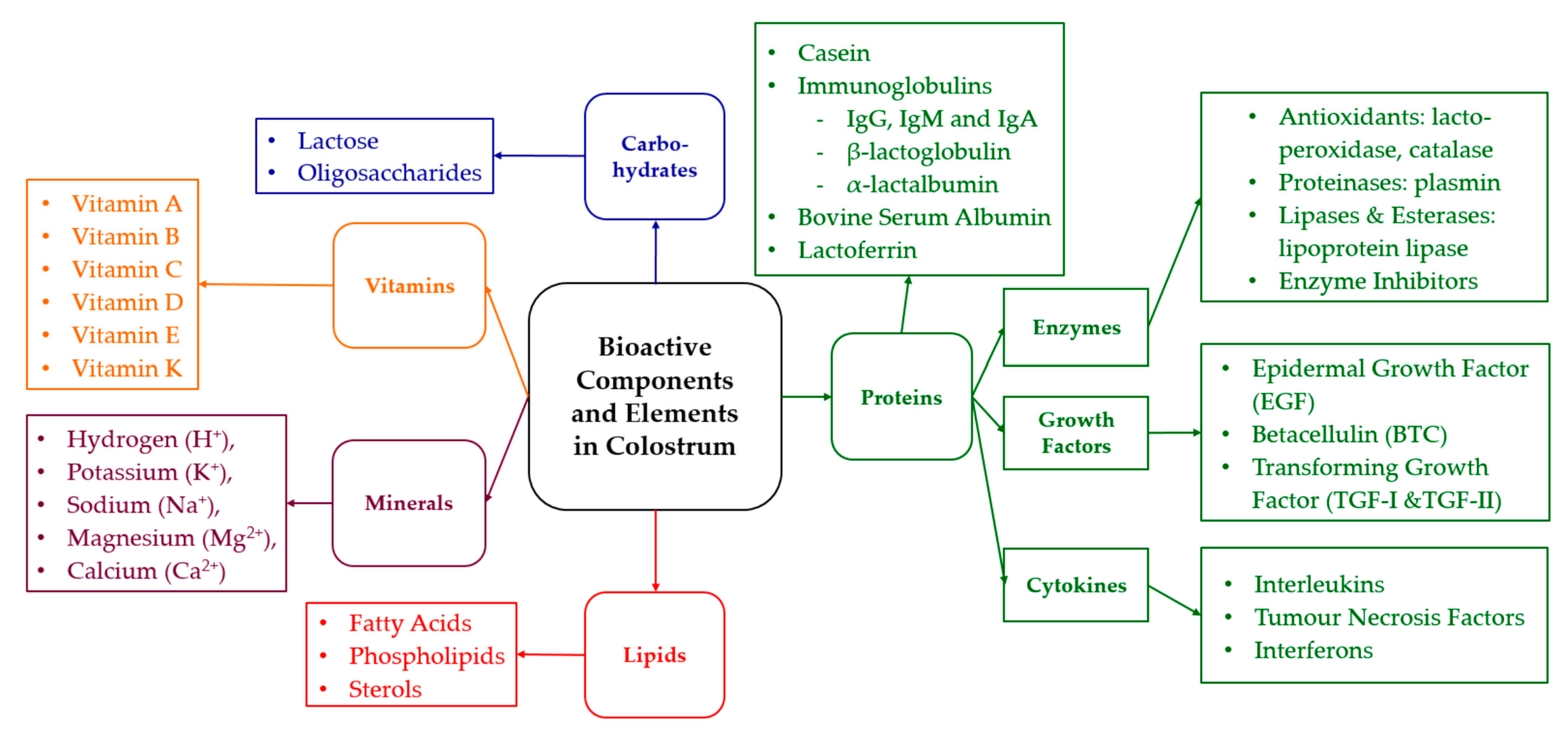
2. Bioactive Components in Colostrum
2.1. Bioactive Compounds
2.2. Immunoglobulins
2.3. Enzymes
2.4. Lactoperoxidase
2.5. Non-Enzymatic Bioactive Components
3. Microbiological Quality of Colostrum
3.1. Regulations on Bacterial Counts
3.2. Bacteria Occuring in Colostrum
3.3. Bovine Pathogens
3.4. Probiotic Bacteria
3.5. Bacterial Effects in Colostrum
4. Products of Bovine Colostrum
5. Contamination Control On-Farm and in Processing
5.1. Contamination Risk On-Farm
5.2. Risks in Processing of Bovine Colostrum
5.3. Heat Treatment
5.4. Fermentation
5.5. Microfiltration, High-Pressure Processing, and Subsequent Processing
6. Process Design
6.1. Process Synthesis
6.2. Process Alternatives
6.3. Economic Considerations
6.3.1. Equipment Costs and Efficiency, Non-Dimensional
6.3.2. Bacterial Inactivation and Loss of Bioactive Agents
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- McGrath, B.A.; Fox, P.F.; McSweeney, P.L.H.; Kelly, A.L. Composition and properties of bovine colostrum: A review. Dairy Sci. Technol. 2016, 96, 133–158. [Google Scholar] [CrossRef]
- Korhonen, H.J. Bioactive milk proteins and peptides: From science to functional applications. Aust. J. Dairy Technol. 2009, 64, 16. [Google Scholar]
- Foley, J.A.; Otterby, D.E. Availability, storage, treatment, composition, and feeding value of surplus colostrum: A review. J. Dairy Sci. 1978, 61, 1033–1060. [Google Scholar] [CrossRef]
- Gosch, T.; Apprich, S.; Kneifel, W.; Novalin, S. A combination of microfiltration and high pressure treatment for the elimination of bacteria in bovine colostrum. Int. Dairy J. 2014, 34, 41–46. [Google Scholar] [CrossRef]
- El-Loly, M. Bovine milk immunoglobulins in relation to human health. Int. J. Dairy Sci. 2007, 2, 183–195. [Google Scholar] [CrossRef][Green Version]
- dos Santos Oliveira Silva, E.G.; do Nascimento Rangel, A.H.; Mürmam, L.; Bezerra, M.F.; de Oliveira, J.P.F. Bovine colostrum: Benefits of its use in human food. Food Sci. Technol. 2019, 39, 355–362. [Google Scholar] [CrossRef]
- Playford, R.J.; Weiser, M.J. Bovine colostrum: Its constituents and uses. Nutrients 2021, 13, 265. [Google Scholar] [CrossRef] [PubMed]
- Borad, S.G.; Singh, A.K. Colostrum immunoglobulins: Processing, preservation and application aspects. Int. Dairy J. 2018, 85, 201–210. [Google Scholar] [CrossRef]
- Fecteau, G.; Baillargeon, P.; Higgins, R.; Paré, J.; Fortin, M. Bacterial contamination of colostrum fed to newborn calves in Québec dairy herds. Can. Vet. J. 2002, 43, 523–527. [Google Scholar]
- Houser, B.A.; Donaldson, S.C.; Kehoe, S.I.; Heinrichs, A.J.; Jayarao, B.M. A survey of bacteriological quality and the occurrence of Salmonella in raw bovine colostrum. Foodborne Pathog. Dis. 2008, 5, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Morrill, K.M.; Conrad, E.; Lago, A.; Campbell, J.; Quigley, J.; Tyler, H. Nationwide evaluation of quality and composition of colostrum on dairy farms in the United States. J. Dairy Sci. 2012, 95, 3997–4005. [Google Scholar] [CrossRef]
- Godden, S.M.; Lombard, J.E.; Woolums, A.R. Colostrum management for dairy calves. Vet. Clinics. N. Am. Food Anim. Pract. 2019, 35, 535–556. [Google Scholar] [CrossRef]
- Lora, I.; Gottardo, F.; Bonfanti, L.; Stefani, A.L.; Soranzo, E.; Dall’Ava, B.; Capello, K.; Martini, M.; Barberio, A. Transfer of passive immunity in dairy calves: The effectiveness of providing a supplementary colostrum meal in addition to nursing from the dam. Animal 2019, 13, 2621–2629. [Google Scholar] [CrossRef]
- Lima, S.F.; Teixeira, A.G.V.; Lima, F.S.; Ganda, E.K.; Higgins, C.H.; Oikonomou, G.; Bicalho, R.C. The bovine colostrum microbiome and its association with clinical mastitis. J. Dairy Sci. 2017, 100, 3031–3042. [Google Scholar] [CrossRef]
- Derakhshani, H.; Plaizier, J.C.; de Buck, J.; Barkema, H.W.; Khafipour, E. Composition of the teat canal and intramammary microbiota of dairy cows subjected to antimicrobial dry cow therapy and internal teat sealant. J. Dairy Sci. 2018, 101, 10191–10205. [Google Scholar] [CrossRef]
- Klein-Jöbstl, D.; Quijada, N.M.; Dzieciol, M.; Feldbacher, B.; Wagner, M.; Drillich, M.; Schmitz-Esser, S.; Mann, E. Microbiota of newborn calves and their mothers reveals possible transfer routes for newborn calves’ gastrointestinal microbiota. PLoS ONE 2019, 14, e0220554. [Google Scholar] [CrossRef]
- Stewart, S.; Godden, S.M.; Bey, R.; Rapnicki, P.; Fetrow, J.; Farnsworth, R.; Scanlon, M.; Arnold, Y.; Clow, L.; Mueller, K.; et al. Preventing bacterial contamination and proliferation during the harvest, storage, and feeding of fresh bovine colostrum. J. Dairy Sci. 2005, 88, 2571–2578. [Google Scholar] [CrossRef]
- Claeys, W.L.; Cardoen, S.; Daube, G.; de Block, J.; Dewettinck, K.; Dierick, K.; de Zutter, L.; Huyghebaert, A.; Imberechts, H.; Thiange, P.; et al. Raw or heated cow milk consumption: Review of risks and benefits. Food Control 2013, 31, 251–262. [Google Scholar] [CrossRef]
- Fernandes, R. Microbiology Handbook: Dairy Products; Royal Society of Chemistry: Leatherhead, UK, 2009. [Google Scholar]
- Elizondo-Salazar, J.A.; Heinrichs, A.J. Heat treating bovine colostrum. Appl. Anim. Sci. 2008, 24, 530–538. [Google Scholar] [CrossRef]
- Stelwagen, K.; Carpenter, E.; Haigh, B.; Hodgkinson, A.; Wheeler, T.T. Immune components of bovine colostrum and milk. J. Anim. Sci. 2009, 87 (Suppl. 1), 3–9. [Google Scholar] [CrossRef]
- Park, Y.W.; Nam, M.S. Bioactive peptides in milk and dairy products: A review. Korean J. Food Sci. Anim. Resour. 2015, 35, 831–840. [Google Scholar] [CrossRef]
- Grand View Research. Colostrum Market Size, Share & Trends Analysis Report by Product (Whole Powder, Skimmed Powder, Specialty), by Application (Nutritional Supplementation, Animal Feed), by Region, and Segment Forecasts, 2019–2025. 2019. Available online: https://www.grandviewresearch.com/industry-analysis/colostrum-market (accessed on 12 November 2020).
- Future Market Insights. Colostrum Market to Surge at 6.4% CAGR, Rising Health Concern due to COVID-19 Will Promote Overall Growth, Says FMI. 2020. Available online: https://www.futuremarketinsights.com/press-release/colostrum-market (accessed on 20 December 2020).
- Korhonen, H.J. Bioactive milk proteins, peptides and lipids and other functional components derived from milk and bovine colostrum. In Functional Foods, 2nd ed.; Saarela, M., Ed.; Woodhead Publishing Series in Food Science, Technology and Nutrition; Woodhead Publishing: Cambridge, UK, 2011; pp. 471–511. [Google Scholar]
- Contarini, G.; Povolo, M.; Pelizzola, V.; Monti, L.; Bruni, A.; Passolungo, L.; Abeni, F.; Degano, L. Bovine colostrum: Changes in lipid constituents in the first 5 days after parturition. J. Dairy Sci. 2014, 97, 5065–5072. [Google Scholar] [CrossRef]
- Korhonen, H.J.; Marnila, P.; Gill, H.S. Milk immunoglobulins and complement factors. Br. J. Nutr. 2000, 84 (Suppl. 1), S75–S80. [Google Scholar] [CrossRef]
- Mayasari, N.; de Vries Reilingh, G.; Nieuwland, M.; Remmelink, G.J.; Parmentier, H.K.; Kemp, B.; van Knegsel, A. Effect of maternal dry period length on colostrum immunoglobulin content and on natural and specific antibody titers in calves. J. Dairy Sci. 2015, 98, 3969–3979. [Google Scholar] [CrossRef]
- Muller, L.D.; Ellinger, D.K. Colostral immunoglobulin concentrations among breeds of dairy cattle. J. Dairy Sci. 1981, 64, 1727–1730. [Google Scholar] [CrossRef]
- Saad, K.; Abo-Elela, M.G.M.; El-Baseer, K.A.A.; Ahmed, A.E.; Ahmad, F.-A.; Tawfeek, M.S.K.; El-Houfey, A.A.; Aboul Khair, M.D.; Abdel-Salam, A.M.; Abo-elgheit, A.; et al. Effects of bovine colostrum on recurrent respiratory tract infections and diarrhea in children. Medicine 2016, 95, e4560. [Google Scholar] [CrossRef]
- Alsayed, A.; Al-Doori, A.; Al-Dulaimi, A.; Alnaseri, A.; Abuhashish, J.; Aliasin, K.; Alfayoumi, I. Influences of bovine colostrum on nasal swab microbiome and viral upper respiratory tract infections—A case report. Respir. Med. Case Rep. 2020, 31, 101189. [Google Scholar] [CrossRef]
- Kanekanian, A. Milk and Dairy Products as Functional Foods; John Wiley & Sons Inc.: Chichester, UK, 2014. [Google Scholar]
- Roos, N.; Mahé, S.; Benamouzig, R.; Sick, H.; Rautureau, J.; Tomé, D. 15N-labeled immunoglobulins from bovine colostrum are partially resistant to digestion in human intestine. J. Nutr. 1995, 125, 1238–1244. [Google Scholar] [CrossRef]
- Ulfman, L.H.; Leusen, J.H.W.; Savelkoul, H.F.J.; Warner, J.O.; van Neerven, R.J.J. Effects of bovine immunoglobulins on immune function, allergy, and infection. Front. Nutr. 2018, 5, 52. [Google Scholar] [CrossRef]
- Anderson, R.C.; Dalziel, J.E.; Haggarty, N.W.; Dunstan, K.E.; Gopal, P.K.; Roy, N.C. Short communication: Processed bovine colostrum milk protein concentrate increases epithelial barrier integrity of Caco-2 cell layers. J. Dairy Sci. 2019, 102, 10772–10778. [Google Scholar] [CrossRef]
- Fox, P.F.; Kelly, A.L. Indigenous enzymes in milk: Overview and historical aspects—Part 1. Int. Dairy J. 2006, 16, 500–516. [Google Scholar] [CrossRef]
- Fox, P.F.; Kelly, A.L. Indigenous enzymes in milk: Overview and historical aspects—Part 2. Int. Dairy J. 2006, 16, 517–532. [Google Scholar] [CrossRef]
- Pakkanen, R.; Aalto, J. Growth factors and antimicrobial factors of bovine colostrum. Int. Dairy. J. 1997, 7, 285–297. [Google Scholar] [CrossRef]
- Korhonen, H.J. Antimicrobial factors in bovine colostrum. Agric. Food Sci. 1977, 49, 434–447. [Google Scholar] [CrossRef]
- Lie, O.; Solbu, H.; Syed, M. A genetic association between bovine serum and colostrum lysozyme levels. Anim. Genet. 1986, 17, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Reiter, B. Review of nonspecific antimicrobial factors in colostrum. J. Vet. Res. 1978, 9, 205–224. [Google Scholar]
- Korhonen, H.J. Production and properties of health-promoting proteins and peptides from bovine colostrum and milk. Cell. Mol. Biol. 2013, 59, 12–24. [Google Scholar] [PubMed]
- Uruakpa, F.; Ismond, M.; Akobundu, E. Colostrum and its benefits: A review. Nutr. Res. 2002, 22, 755–767. [Google Scholar] [CrossRef]
- Plaut, A.G.; St. Geme, J. Lactoferrin. In Handbook of Proteolytic Enzymes, 3rd ed.; Rawlings, N.D., Salvesen, G., Eds.; Academic Press: London, UK, 2013; pp. 3635–3640. [Google Scholar]
- Korhonen, H.J.; Pihlanto, A. Technological options for the production of health-promoting proteins and peptides derived from milk and colostrum. Curr. Pharm. Des. 2007, 13, 829–843. [Google Scholar] [CrossRef]
- Tung, Y.-T.; Chen, H.-L.; Yen, C.-C.; Lee, P.-Y.; Tsai, H.-C.; Lin, M.-F.; Chen, C.-M. Bovine lactoferrin inhibits lung cancer growth through suppression of both inflammation and expression of vascular endothelial growth factor. J. Dairy Sci. 2013, 96, 2095–2106. [Google Scholar] [CrossRef] [PubMed]
- Kehoe, S.I.; Jayarao, B.M.; Heinrichs, A.J. A survey of bovine colostrum composition and colostrum management practices on Pennsylvania dairy farms. J. Dairy Sci. 2007, 90, 4108–4116. [Google Scholar] [CrossRef]
- Madsen, B.D.; Rasmussen, M.D.; Nielsen, M.O.; Wiking, L.; Larsen, L.B. Physical properties of mammary secretions in relation to chemical changes during transition from colostrum to milk. J. Dairy Res. 2004, 71, 263–272. [Google Scholar] [CrossRef] [PubMed]
- O’Kennedy, B.T. Caseins. In Handbook of Food Proteins; Woodhead Publishing: Cambridge, UK, 2011; pp. 13–29. [Google Scholar] [CrossRef]
- Holt, C. Structure and stability of bovine casein micelles. Adv. Protein Chem. 1992, 43, 63–151. [Google Scholar] [CrossRef] [PubMed]
- Lönnerdal, B. Effects of milk and milk components on calcium, magnesium, and trace element absorption during infancy. Physiol. Rev. 1997, 77, 643–669. [Google Scholar] [CrossRef]
- Phelan, M.; Aherne, A.; FitzGerald, R.J.; O’Brien, N.M. Casein-derived bioactive peptides: Biological effects, industrial uses, safety aspects and regulatory status. Int. Dairy J. 2009, 19, 643–654. [Google Scholar] [CrossRef]
- Isaacs, C. Antimicrobial function of milk lipids. Adv. Nutr. Res. 2001, 10, 271–285. [Google Scholar] [CrossRef]
- Laakso, P.; Manninen, P.; Mäkinen, J.; Kallio, H. Postparturition changes in the triacylglycerols of cow colostrum. Lipids 1996, 31, 937–943. [Google Scholar] [CrossRef]
- Bitzan, M.M.; Gold, B.D.; Philpott, D.J.; Huesca, M.; Sherman, P.M.; Karch, H.; Lissner, R.; Lingwood, C.A.; Karmali, M.A. Inhibition of Helicobacter pylori and Helicobacter mustelae binding to lipid receptors by bovine colostrum. J. Infect. Dis. 1998, 177, 955–961. [Google Scholar] [CrossRef]
- Sacerdote, P.; Mussano, F.; Franchi, S.; Panerai, A.E.; Bussolati, G.; Carossa, S.; Bartorelli, A.; Bussolati, B. Biological components in a standardized derivative of bovine colostrum. J. Dairy Sci. 2013, 96, 1745–1754. [Google Scholar] [CrossRef]
- Hantsis-Zacharov, E.; Halpern, M. Culturable psychrotrophic bacterial communities in raw milk and their proteolytic and lipolytic traits. Appl. Environ. Microbiol. 2007, 73, 7162–7168. [Google Scholar] [CrossRef]
- Gelsinger, S.L.; Jones, C.M.; Heinrichs, A.J. Effect of colostrum heat treatment and bacterial population on immunoglobulin G absorption and health of neonatal calves. J. Dairy Sci. 2015, 98, 4640–4645. [Google Scholar] [CrossRef]
- European Parliament and the Council of the European Union. Regulation (EC) no 853/2004 of the European Parliament and of the Council of 29 April 2004 laying down specific hygiene rules for on the hygiene of foodstuffs. 2004. Available online: https://eur-lex.europa.eu/eli/reg/2004/853/oj (accessed on 10 June 2021).
- Godden, S.M.; Smolenski, D.J.; Donahue, M.; Oakes, J.M.; Bey, R.; Wells, S.; Sreevatsan, S.; Stabel, J.; Fetrow, J. Heat-treated colostrum and reduced morbidity in preweaned dairy calves: Results of a randomized trial and examination of mechanisms of effectiveness. J. Dairy Sci. 2012, 95, 4029–4040. [Google Scholar] [CrossRef]
- Phipps, A.J.; Beggs, D.S.; Murray, A.J.; Mansell, P.D.; Stevenson, M.A.; Pyman, M.F. Survey of bovine colostrum quality and hygiene on northern Victorian dairy farms. J. Dairy Sci. 2016, 99, 8981–8990. [Google Scholar] [CrossRef]
- Jans, C.; Meile, L.; Kaindi, D.W.M.; Kogi-Makau, W.; Lamuka, P.; Renault, P.; Kreikemeyer, B.; Lacroix, C.; Hattendorf, J.; Zinsstag, J.; et al. African fermented dairy products–Overview of predominant technologically important microorganisms focusing on African Streptococcus infantarius variants and potential future applications for enhanced food safety and security. Int. J. Food Microbiol. 2017, 250, 27–36. [Google Scholar] [CrossRef]
- Abebe, E.; Gugsa, G.; Ahmed, M. Review on major food-borne zoonotic bacterial pathogens. J. Trop. Med. 2020, 2020, 4674235. [Google Scholar] [CrossRef]
- Fritsche, O. Mikrobiologie; Springer Spektrum: Wiesbaden, Germany, 2016. [Google Scholar]
- Hoorfar, J. Rapid Detection, Characterization, and Enumeration of Foodborne Pathogens; ASM Press: Washington, DC, USA, 2011. [Google Scholar]
- Zadoks, R.N.; Middleton, J.R.; McDougall, S.; Katholm, J.; Schukken, Y.H. Molecular epidemiology of mastitis pathogens of dairy cattle and comparative relevance to humans. J. Mammary Gland Biol. Neoplasia 2011, 16, 357–372. [Google Scholar] [CrossRef]
- Lindner, J.D.D.; Santarelli, M.; Yamaguishi, C.T.; Soccol, C.R.; Neviani, E. Recovery and identification of bovine colostrum microflora using traditional and molecular approaches. Food Technol. Biotechnol. 2011, 49, 364–368. [Google Scholar]
- Lailler, R.; Sanaa, M.; Chadoeuf, J.; Fontez, B.; Brisabois, A.; Colmin, C.; Millemann, Y. Prevalence of multidrug resistant (MDR) Salmonella in bovine dairy herds in western France. Prev. Vet. Med. 2005, 70, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Gyles, C.L.; Prescott, J.F.; Prescott, J.F.; Songer, J.G.; Thoen, C.O.; Songer, G. Pathogenesis of Bacterial Infections in Animals; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2004. [Google Scholar]
- Hasegawa, M.; Iwabuchi, E.; Yamamoto, S.; Esaki, H.; Kobayashi, K.; Ito, M.; Hirai, K. Prevalence and characteristics of Listeria monocytogenes in bovine colostrum in Japan. J. Food Prot. 2013, 76, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Janzen, J.J. Economic losses resulting from mastitis: A review. J. Dairy Sci. 1970, 53, 1151–1160. [Google Scholar] [CrossRef]
- Moretti, A.; Pasquali, P.; Mencaroni, G.; Boncio, L.; Piergili Fioretti, D. Relationship between cell counts in bovine milk and the presence of mastitis pathogens (yeasts and bacteria). J. Vet. Med. B 1998, 45, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Maunsell, F.P.; Woolums, A.R.; Francoz, D.; Rosenbusch, R.F.; Step, D.L.; Wilson, D.; Janzen, E. Mycoplasma bovis infections in cattle. J. Vet. Intern. Med./Am. Coll. Vet. Intern. Med. 2011, 25, 772–783. [Google Scholar] [CrossRef] [PubMed]
- Zastempowska, E.; Lassa, H. Genotypic characterization and evaluation of an antibiotic resistance of Trueperella pyogenes (Arcanobacterium pyogenes) isolated from milk of dairy cows with clinical mastitis. Vet. Microbiol. 2012, 161, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Mager, D.L. Bacteria and cancer: Cause, coincidence or cure? A review. J. Transl. Med. 2006, 4, 14. [Google Scholar] [CrossRef]
- Finke, E.-J.; Tomaso, H.; Frangoulidis, D. Bioterrorismus, infektiologische Aspekte. In Lexikon der Infektionskrankheiten des Menschen: Erreger, Symptome, Diagnose, Therapie und Prophylaxe; Darai, G., Handermann, M., Sonntag, H.-G., Zöller, L., Eds.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 76–392. Available online: https://link.springer.com/book/10.1007/978-3-642-17158-1 (accessed on 7 January 2021).
- Gille, L.; Evrard, J.; Callens, J.; Supré, K.; Grégoire, F.; Boyen, F.; Haesebrouck, F.; Deprez, P.; Pardon, B. The presence of Mycoplasma bovis in colostrum. Vet. Res. 2020, 51, 54. [Google Scholar] [CrossRef]
- Bernard, K.A.; Munro, C.; Wiebe, D.; Ongsansoy, E. Characteristics of rare or recently described Corynebacterium species recovered from human clinical material in Canada. J. Clin. Microbiol. 2002, 40, 4375–4381. [Google Scholar] [CrossRef]
- Langoni, H.; da Silva, C.; Polo, C.; Troncarelli, M.Z.; Tata, A.; Belaz, K.R.A.; Eberlin, M.N.; Joaquim, S.F.; Guimarães, F.F.; Pardo, R.B.; et al. Short communication: Identification of Corynebacterium bovis by MALDI-mass spectrometry. J. Dairy Sci. 2017, 100, 4287–4289. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.-L.; Odah, K.A.; Liu, L.; Xu, Q.-J.; Gao, Y.-H.; Kong, L.-C.; Ma, H.-X. Multidrug resistance genes are associated with a 42-kb island TGI1 carrying a complex class 1 integron in Trueperella pyogenes. J. Glob. Antimicrob. Resist. 2020, 22, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Ali, Z.I.; Saudi, A.M.; Albrecht, R.; Talaat, A.M. The inhibitory effect of nisin on Mycobacterium avium ssp. paratuberculosis and its effect on mycobacterial cell wall. J. Dairy Sci. 2019, 102, 4935–4944. [Google Scholar] [CrossRef] [PubMed]
- Peterz, M.; Butot, S.; Jagadeesan, B.; Bakker, D.; Donaghy, J. Thermal inactivation of Mycobacterium avium subsp. paratuberculosis in artificially contaminated milk by direct steam injection. Appl. Environ. Microbiol. 2016, 82, 2800–2808. [Google Scholar] [CrossRef]
- Nielsen, S.S.; Bjerre, H.; Toft, N. Colostrum and milk as risk factors for infection with Mycobacterium avium subspecies paratuberculosis in dairy cattle. J. Dairy Sci. 2008, 91, 4610–4615. [Google Scholar] [CrossRef] [PubMed]
- Streeter, R.N.; Hoffsis, G.F.; Bech-Nielsen, S.; Shulaw, W.P.; Rings, D.M. Isolation of Mycobacterium paratuberculosis from colostrum and milk of subclinically infected cows. Am. J. Vet. Res. 1995, 56, 1322–1324. [Google Scholar] [PubMed]
- Pithua, P.; Godden, S.M.; Wells, S.J.; Stabel, J.R. Evaluation of the risk of paratuberculosis in adult cows fed Mycobacterium avium subsp. paratuberculosis DNA-positive or -negative colostrum as calves. Am. J. Vet. Res. 2011, 72, 1456–1464. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, C.; Gray, N.F. The coliform index and waterborne disease: Problems of microbial drinking water assessment; CRC Press: London, UK, 1997. [Google Scholar]
- Cockcroft, P. Bovine Medicine, 3rd ed.; Wiley Blackwell: Chichester, UK, 2015. [Google Scholar]
- dos Santos, G.; da Silva, J.T.; da Rocha Santos, F.H.; Machado Bittar, C.M. Nutritional and microbiological quality of bovine colostrum samples in Brazil. R. Bras. Zootec. 2017, 46, 72–79. [Google Scholar] [CrossRef]
- Nonnecke, B.J.; Smith, K.L. Biochemical and antibacterial properties of bovine mammary secretion during mammary involution and at parturition. J. Dairy Sci. 1984, 67, 2863–2872. [Google Scholar] [CrossRef]
- Wijesooriya, L.I.; Namalie, D.; Sirisena, N.; Sunil-Chandra, N. Antibiotic resistance in coliforms: Human versus livestock infections. Int. J. Infect. Dis. 2020, 101, S1–S22. [Google Scholar] [CrossRef]
- Kröger, C.; Schauer, K.; Clerkin, S.R.; Märtlbauer, E.; Fleming, A.B. Draft genome sequence and annotation of Acinetobacter junii MHI21018 isolated from bovine colostrum. Microbiol. Resour. Announc. 2019, 8, e01700–e01718. [Google Scholar] [CrossRef]
- Darai, G.; Handermann, M.; Sonntag, H.-G.; Zöller, L. (Eds.) Lexikon der Infektionskrankheiten des Menschen: Erreger, Symptome, Diagnose, Therapie und Prophylaxe; Springer: Berlin/Heidelberg, Germany, 2012; pp. 1–31. Available online: https://link.springer.com/book/10.1007/978-3-642-17158-1. (accessed on 7 January 2021).
- Ribeiro Júnior, J.C.; Tamanini, R.; de Oliveira, A.; Alfieri, A.A.; Beloti, V. Genetic diversity of thermoduric spoilage microorganisms of milk from Brazilian dairy farms. J. Dairy Sci. 2018, 101, 6927–6936. [Google Scholar] [CrossRef]
- Petkar, H.; Li, A.; Bunce, N.; Duffy, K.; Malnick, H.; Shah, J.J. Cellulosimicrobium funkei: First report of infection in a nonimmunocompromised patient and useful phenotypic tests for differentiation from Cellulosimicrobium cellulans and Cellulosimicrobium terreum. J. Clin. Microbiol. 2011, 49, 1175–1178. [Google Scholar] [CrossRef]
- Dréno, B.; Pécastaings, S.; Corvec, S.; Veraldi, S.; Khammari, A.; Roques, C. Cutibacterium acnes (Propionibacterium acnes) and acne vulgaris: A brief look at the latest updates. J. Eur. Acad. Dermatol. Venereol. 2018, 32 (Suppl. 2), 5–14. [Google Scholar] [CrossRef]
- Kim, K.K.; Lee, J.-S.; Stevens, D.A. Microbiology and epidemiology of Halomonas species. Future Microbiol. 2013, 8, 1559–1573. [Google Scholar] [CrossRef]
- Baba, T.; Kuwahara-Arai, K.; Uchiyama, I.; Takeuchi, F.; Ito, T.; Hiramatsu, K. Complete genome sequence of Macrococcus caseolyticus strain JCSCS5402, corrected reflecting the ancestral genome of the human-pathogenic staphylococci. J. Bacteriol. 2009, 191, 1180–1190. [Google Scholar] [CrossRef] [PubMed]
- Behera, S.S.; Ray, R.C.; Zdolec, N. Lactobacillus plantarum with functional properties: An approach to increase safety and shelf-life of fermented foods. BioMed Res. Int. 2018, 2018, 9361614. [Google Scholar] [CrossRef] [PubMed]
- Parodi, P.W. The role of intestinal bacteria in the causation and prevention of cancer: Modulation by diet and probiotics. Aust. J. Dairy Technol. 1999, 54, 103–121. [Google Scholar]
- Morrin, S.T.; Lane, J.A.; Marotta, M.; Bode, L.; Carrington, S.D.; Irwin, J.A.; Hickey, R.M. Bovine colostrum-driven modulation of intestinal epithelial cells for increased commensal colonisation. Appl. Microbiol. Biotechnol. 2019, 103, 2745–2758. [Google Scholar] [CrossRef]
- Kuhl, G.C.; Lindner, J.D.D. Biohydrogenation of linoleic acid by lactic acid bacteria for the production of functional cultured dairy products: A review. Foods 2016, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- Angelin, J.; Kavitha, M. Exopolysaccharides from probiotic bacteria and their health potential. Int. J. Biol. Macromol. 2020, 162, 853–865. [Google Scholar] [CrossRef]
- Rajilić-Stojanović, M.; de Vos, W.M. The first 1000 cultured species of the human gastrointestinal microbiota. FEMS Microbiol. Rev. 2014, 38, 996–1047. [Google Scholar] [CrossRef]
- Wittanalai, S.; Tanruean, K.; Mapoong, P. Inhibition of coliform bacteria by lactic acid bacteria isolated from nham hed (fermented mushroom). Appl. Mech. Mater. 2019, 886, 56–60. [Google Scholar] [CrossRef]
- Vivarelli, S.; Salemi, R.; Candido, S.; Falzone, L.; Santagati, M.; Stefani, S.; Torino, F.; Banna, G.L.; Tonini, G.; Libra, M. Gut microbiota and cancer: From pathogenesis to therapy. Cancers 2019, 11, 38. [Google Scholar] [CrossRef]
- Cummins, C.; Lorenz, I.; Kennedy, E. Short communication: The effect of storage conditions over time on bovine colostral immunoglobulin G concentration, bacteria, and pH. J. Dairy Sci. 2016, 99, 4857–4863. [Google Scholar] [CrossRef] [PubMed]
- Borad, S.G.; Singh, A.K.; Meena, G.S.; Arora, S.; Raju, P.N.; Sabikhi, L. Optimization of spray drying of colostrum protein ingredients—A rheological approach. J. Food Eng. 2021, 288, 110247. [Google Scholar] [CrossRef]
- Sats, A.; Kaart, T.; Poikalainen, V.; Aare, A.; Lepasalu, L.; Andreson, H.; Jõudu, I. Bovine colostrum whey: Postpartum changes of particle size distribution and immunoglobulin G concentration at different filtration pore sizes. J. Dairy Sci. 2020, 103, 6810–6819. [Google Scholar] [CrossRef] [PubMed]
- European Parliament and the Council of the European Union. Regulation (EC) no 852/2004 of the European Parliament and of the Council of 29 April 2004 on the Hygiene of Foodstuffs. 2004. Available online: https://eur-lex.europa.eu/eli/reg/2004/852/oj. (accessed on 10 June 2021).
- Dewulf, J.; Van Immerseel, F. (Eds.) Biosecurity in Animal Production and Veterinary Medicine from Principles to Practice; Acco: Leuven, Belgium, 2018. [Google Scholar]
- Food Standards Australia New Zealand. Primary Production and Processing (PPP) Standards. 2020. Available online: https://www.foodstandards.gov.au/foodsafety/standards/Pages/Primary-Production-and-Processing-(PPP)-Standards-(Chapter-4).aspx (accessed on 28 March 2021).
- Food Standards Australia New Zealand. Food Safety Hub. 2019. Available online: https://www.foodstandards.gov.au/foodsafety/Pages/default.aspx. (accessed on 28 March 2021).
- Bartkiene, E.; Bartkevics, V.; Ikkere, L.E.; Pugajeva, I.; Zavistanaviciute, P.; Lele, V.; Ruzauskas, M.; Bernatoniene, J.; Jakstas, V.; Klupsaite, D.; et al. The effects of ultrasonication, fermentation with Lactobacillus sp., and dehydration on the chemical composition and microbial contamination of bovine colostrum. J. Dairy Sci. 2018, 101, 6787–6798. [Google Scholar] [CrossRef] [PubMed]
- Elizondo-Salazar, J.A.; Jayarao, B.M.; Heinrichs, A.J. Effect of heat treatment of bovine colostrum on bacterial counts, viscosity, and Immunoglobulin G concentration. J. Dairy Sci. 2010, 93, 961–967. [Google Scholar] [CrossRef]
- Lewis, M.J. Thermal processing. In Food Processing Handbook; Brennan, J.G., Ed.; WILEY-VCH: Weinheim, Germany, 2006; pp. 33–70. [Google Scholar]
- Johnson, J.L.; Godden, S.M.; Molitor, T.; Ames, T.; Hagman, D. Effects of feeding heat-treated colostrum on passive transfer of immune and nutritional parameters in neonatal dairy calves. J. Dairy Sci. 2007, 90, 5189–5198. [Google Scholar] [CrossRef]
- Godden, S.M.; McMartin, S.; Feirtag, J.; Stabel, J.; Bey, R.; Goyal, S.; Metzger, L.; Fetrow, J.; Wells, S.; Chester-Jones, H. Heat-treatment of bovine colostrum. II: Effects of heating duration on pathogen viability and immunoglobulin G. J. Dairy Sci. 2006, 89, 3476–3483. [Google Scholar] [CrossRef]
- Stabel, J.R.; Hurd, S.; Calvente, L.; Rosenbusch, R.F. Destruction of Mycobacterium paratuberculosis, Salmonella spp., and Mycoplasma spp. in raw milk by a commercial on-farm high-temperature, short-time pasteurizer. J. Dairy Sci. 2004, 87, 2177–2183. [Google Scholar] [CrossRef]
- Bartkiene, E.; Lele, V.; Sakiene, V.; Zavistanaviciute, P.; Ruzauskas, M.; Stankevicius, A.; Grigas, J.; Pautienius, A.; Bernatoniene, J.; Jakstas, V.; et al. Fermented, ultrasonicated, and dehydrated bovine colostrum: Changes in antimicrobial properties and immunoglobulin content. J. Dairy Sci. 2020, 103, 1315–1323. [Google Scholar] [CrossRef]
- De Haan, A.B. Process Technology—An Introduction; De Gruyter: Eindhoven, The Netherlands, 2015; pp. 11–26. [Google Scholar] [CrossRef]
- Wanniarachchi, W.; Gopura, R.; Punchihewa, H. Development of a layout model suitable for the food processing industry. J. Ind. Eng. 2016, 2016, 1–8. [Google Scholar] [CrossRef]
- Bowser, T.J. Food processing facility design. In Handbook of Farm, Dairy and Food Machinery Engineering, 3rd ed.; Kutz, M., Ed.; Elsevier Inc.: Cambridge, MA, USA, 2019; pp. 623–649. Available online: https://app.knovel.com/hotlink/toc/id:kpHFDFME01/handbook-farm-dairy-food/handbook-farm-dairy-food (accessed on 19 February 2021).
- McGuirk, S.M.; Collins, M. Managing the production, storage, and delivery of colostrum. Vet. Clin. Food Anim. 2004, 20, 593–603. [Google Scholar] [CrossRef]
- Manohar, A.A.; Williamson, M.; Koppikar, G.V. Effect of storage of colostrum in various containers. Indian Pediatr. 1997, 34, 93–295. [Google Scholar]
- Lazaar, M.; Boughanmi, H.; Bouadila, S.; Jarraya, M. Parametric study of plate heat exchanger for eventual use in a solar pasteurization process designed for small milk collection centers in Tunisia. Sustain. Energy Technol. Assess. 2021, 45, 101174. [Google Scholar]
- Tomasula, P.M.; Yee, W.C.F.; MacAloon, A.J.; Nutter, D.W.; Bonnaillie, L.M. Computer simulation of energy use, greenhouse gas emissions, and process economics of the fluid milk process. J. Dairy Sci. 2013, 96, 3350–3368. [Google Scholar] [CrossRef]
- Meraj, M.; Mahmood, S.M.; Khan, M.E.; Azhar, M.; Tiwari, G.N. Effect of N-Photovoltaic thermal integrated parabolic concentrator on milk temperature for pasteurization: A simulation study. Renew. Energy 2021, 163, 2153–2164. [Google Scholar] [CrossRef]
- Al-Hilphy, A.R.; Abdulstar, A.R.; Gavahian, M. Moderate electric field pasteurization of milk in a continuous flow unit: Effects of process parameters, energy consumption, and shelf-life determination. Innov. Food Sci. Emerg. Technol. 2021, 67, 102568. [Google Scholar] [CrossRef]
- Maroulis, Z.B.; Saravacos, G.D. Food Process Design; Marcel Dekker: New York, NY, USA, 2003; pp. 21–57. [Google Scholar]
- Parin, M.A.; Zugarramurdi, A. Investment and production costs analysis in food processing plants. Int. J. Prod. Econ. 1994, 34, 83–89. [Google Scholar] [CrossRef]
- Couper, J.R.; Hertz, D.W.; Smith, L. Process economics. In Perry’s Chemical Engineers’ Handbook, 8th ed.; Perry, R.H., Green, D.W., Eds.; McGraw-Hill: New York, NY, USA, 2007; 56p. [Google Scholar]
- Bartholomai, A. Food Factories–Processes, Equipment, Costs; VCH Verlagsgesellshaft mbH: Hemsbach, Germany, 1987. [Google Scholar]
- STROM-REPORT. 2021 Strom-Report Blog. Electricity Prices in Europe 2019. Available online: https://strom-report.de/electricity-prices-europe/ (accessed on 22 February 2021).
- Rehfeldt, M.; Globisch, J.; Fleiter, T. Fuel choice in industrial steam generation: Empirical evidence reveals technology preferences. Energy Strategy Rev. 2019, 26, 100407. [Google Scholar] [CrossRef]

| Contaminants | Source | Pathogenic Potential in Humans |
|---|---|---|
| Alcaligenaceae | [15] | Wound infection, pneumonia, and sepsis [92] |
| Brachybacterium sp. | [67] | Thermoduric spoilage bacterium [93] |
| Cellulosimicrobium funkei | [67] | Opportunistic pathogen, endocarditis [94] |
| Cutibacterium acnes (formerly Propionibacterium acnes) | [67] | Endocarditis [76], commensal in human skin microbiome [95] |
| Enterococcus spp. | [9] | Enterococcal infections, urinary tract infection (UTI), and endocarditis [76] |
| Fusobacterium spp. | [14] | Endocarditis, UTI, and sepsis [76] |
| Halomonas spp. | [14] | Bacteremia [96] |
| Macrococcus caseolyticus | [67] | Close relation to human pathogen staphylococci [97] |
| Micrococcus spp. | [9] | Endocarditis [76] |
| Paenibacillus barcinonensis | [67] | No data on effect on humans |
| Paenibacillus graminis | [67] | No data on effect on humans |
| Pasteurella spp. | [9] | Empyema, Tularemia, and Brazilian purpuric fever [76] |
| Porphyromondaceae | [14] | Empyema and sepsis [76] |
| Proteus spp. | [9] | Endocarditis and UTI [76] |
| Stenotrophomonas spp. | [15] | Endocarditis and UTI [76] |
| Method | Inactivation Rate (log CFU/mL) | IgG Concentration Loss (%) |
|---|---|---|
| Low temperature long time (LTLT) batch pasteurization, 30 min at 60 °C [117] | Inactivation of Mycoplasma bovis, Listeria monocytogenes, Escherichia coli O157:H7 and Salmonella enteritidis, but Mycobacterium avium ssp. paratuberculosis (MAP) was detected | No significant loss detected |
| LTLT batch pasteurization, 60 min at 60 °C [117] | Inactivation of M. bovis, L. monocytogenes, E. coli O157:H7, S. enteritidis, and in three of four batches no MAP | No significant loss detected |
| LTLT batch pasteurization, 120 min at 60 °C [117] | Viable M. bovis, L. monocytogenes, E. coli O157:H7, S. enteritidis, and MAP were not detected | No significant loss detected |
| High temperature short time (HTST) pasteurization, 15 s at 67 °C [118] | MAP 4 | 22 |
| HTST pasteurization, 15 s at 72 °C [118] | Inactivation of MAP in colostrum | 27 |
| Fermentation with Lactobacillus plantarum LUHS135 [113,119] | 3 [113] | No significant loss detected [119] |
| Fermentation with Lactobacillus paracasei LUHS244 [113,119] | 3.3 [113] | 22 [119] |
| Microfiltration (MF) and high-pressure treatment (HPP) [4] | >6 | 27–64 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fasse, S.; Alarinta, J.; Frahm, B.; Wirtanen, G. Bovine Colostrum for Human Consumption—Improving Microbial Quality and Maintaining Bioactive Characteristics through Processing. Dairy 2021, 2, 556-575. https://doi.org/10.3390/dairy2040044
Fasse S, Alarinta J, Frahm B, Wirtanen G. Bovine Colostrum for Human Consumption—Improving Microbial Quality and Maintaining Bioactive Characteristics through Processing. Dairy. 2021; 2(4):556-575. https://doi.org/10.3390/dairy2040044
Chicago/Turabian StyleFasse, Sylvia, Jarmo Alarinta, Björn Frahm, and Gun Wirtanen. 2021. "Bovine Colostrum for Human Consumption—Improving Microbial Quality and Maintaining Bioactive Characteristics through Processing" Dairy 2, no. 4: 556-575. https://doi.org/10.3390/dairy2040044
APA StyleFasse, S., Alarinta, J., Frahm, B., & Wirtanen, G. (2021). Bovine Colostrum for Human Consumption—Improving Microbial Quality and Maintaining Bioactive Characteristics through Processing. Dairy, 2(4), 556-575. https://doi.org/10.3390/dairy2040044






