1. Introduction
Chemical complexity leading to the molecular origins of life relies upon the creation of molecules containing the elements carbon, hydrogen, oxygen, and nitrogen. While many such molecules are known towards various astronomical sources [
1,
2], their complexity has grown over the recent few years. Notably, ethanolamine (NH
2CH
2CH
2OH) has been observed towards the G+0.693 molecular cloud [
3]. The proposed reaction pathway for the creation of this complicated, highly-saturated, pre-biotic molecule involves the formation of a NH
2CHCO structure (aminoketene) from smaller, known interstellar molecules which themselves contain fewer than five atoms. The formation of NH
2CHCO could utilize HNCCO + 2H, NH
2CH + CO, and NH
3 + C + CO [
3,
4] and may occur in shocked regions [
5] where solid-phase formation supported by laboratory experiments [
6,
7] could then lead to gas-phase desorption and possible detection. Hence, if the formation of ethanolamine proceeds in this way, the aminoketene intermediate should be detectable in the same astronomical regions, as well. In order to potentially observe this intermediate, or any other molecule for that matter, the spectroscopic reference data for the desired compound must be on hand for comparison whether to radioastronomical observation through observatories like the IRAM 30 m telescope, the Yebes 40 m telescope, or even the Atacama Large Millimeter Array (ALMA) or through infrared observation via the
James Webb Space Telescope (
JWST) or any of its potential successors, e.g., the
Habitable Worlds Observatory (
HWO).
Previous work has shown that the
aminoketene structure is not the lowest energy arrangement of the NCCO connectivity with three additional hydrogen atoms among the possible isomers [
4,
8,
9]. This actually belongs to a formyl-imine structure, 2-iminoacetaldehyde or HN=CHC(=O)H, which itself exhibits four conformers all within 0.25 eV relative energy of one another [
4,
8]. The NH
2CHCO isomer actually lies 0.36 eV above the formyl-imine minimum, and it has two conformers: the lower-energy with the amine hydrogens pointing equatorially away from the C=C=O portion of the molecule as well another conformer where the amine hydrogens are axial pointing more towards the portion of the molecule containing the heavy atoms. There are other cyclic and even zwitterionic isomers, but those are higher in energy and exhibit strain or other destabilizing factors. While the previous work has already provided high-level quantum chemical predictions for the fundamental vibrational frequencies and the rotational constants for the two lowest formyl-imine isomers as well as the equatorial aminoketene [
4,
8], the small energy gaps between isomers and the kinetic nature of gas-phase astrochemical reactions implies that the other conformers/isomers may also be present.
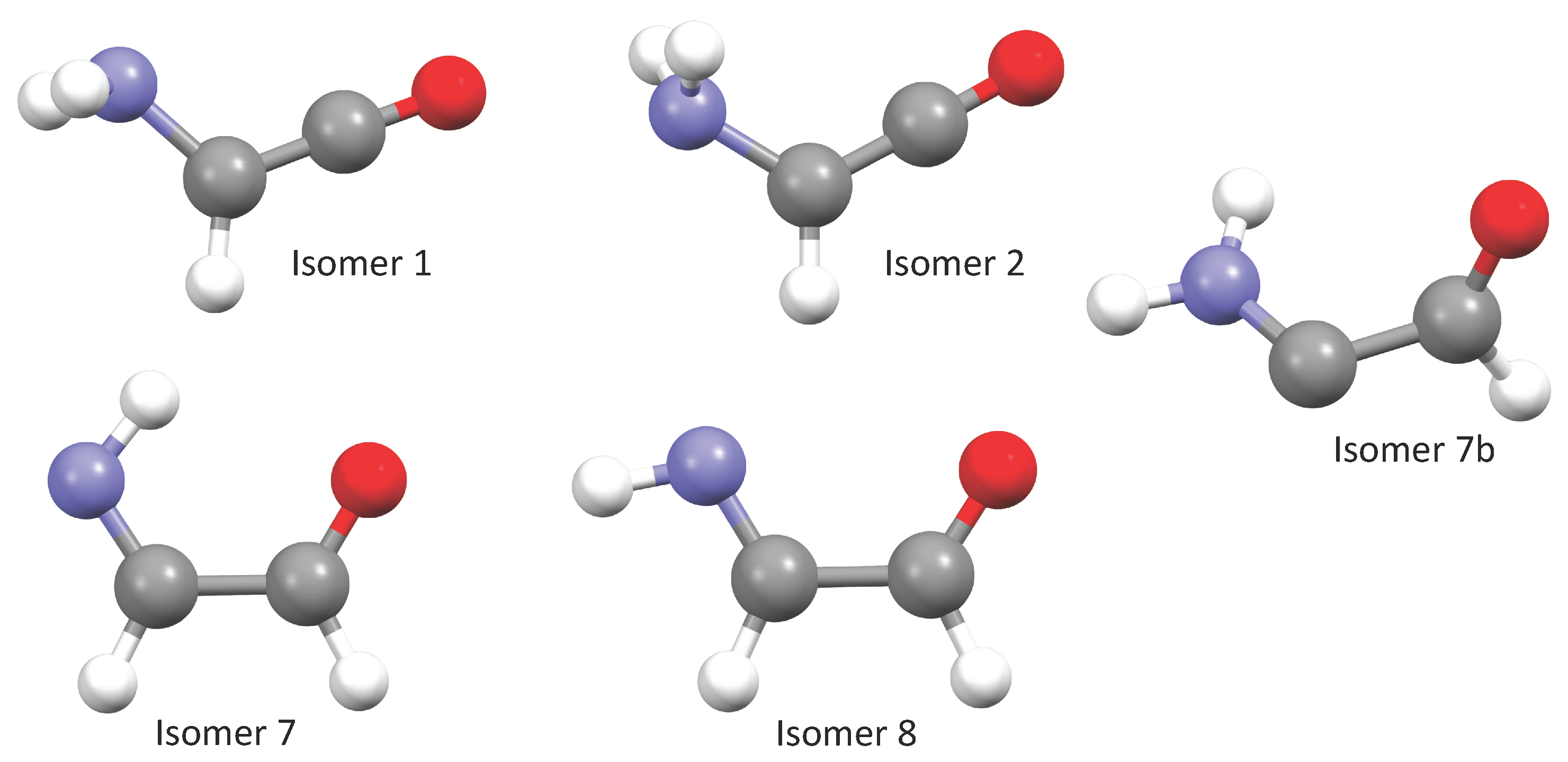
As mentioned, we have previously investigated 14 tautomers and conformers of NH
2CHCO [
4]. A full depiction of them can be found in Figure 1 of Ref. [
8]. Following a re-evaluation of the energetic properties across the entire isomeric family, a corrigendum has been published (see Figure 1 of [
8]) confirming that isomer 10 (
-(E)-2-iminoacetaldehyde) remains the lowest-energy structure, followed by its conformers 9, 7, and 8 (
-(Z)-2-iminoacetaldehyde,
-2-(Z)-iminoacetaldehyde, and
-(E)-2-iminoacetaldehyde, respectively). In that study, isomer 8 is conclusively shown to be a local minimum at the CCSD(T)-F12b/cc-pVTZ-F12 level of theory [
8]. Additionally, isomer 1 and its conformer, isomer 2, are verified to constitute the next-lowest energy set within the series. Isomers 5, 6, 11, and 12 were initially considered local minima (Figure 1 of [
4]); however, further analysis has revealed that they dissociate into van der Waals complexes of methanimine HN=CH
2 and carbon monoxide. Consequently, these structures should not be classified as tautomers and conformers of NH
2CHCO and should have been excluded from Table 1 of [
8] and are excluded from the present study, as well. Isomer 3 remains a second-order saddle point, but the relative energy reported represents the most accurate value to date. Isomer 4 does not correspond to a local minimum and instead reverts to isomer 1 or 2 upon geometry optimization, depending on the initial structural guess. Similarly, isomer 14 relaxes to isomer 13; thus, both are excluded from further consideration.
While the previous study presents accurate spectroscopic data for the lowest-energy isomer and the NH
2CHCO isomer hypothesized to be involved in ethanolamine synthesis, we now extend the spectral characterization to additional structures depicted in
Figure 1 in this work. Specifically, conformers of the
-(Z)- and
-(E)-2-iminoacetaldehyde (HN=CHC(=O)H; isomers 7 and 8), which represent conformers of the global minimum and lie within 0.250 eV (2000 cm
−1) of the lowest-energy tautomer or conformer of aminoketene, may contribute to the overall vibrational and/or rotational spectra of this system. Furthermore, the two lower-energy,
isomers have been observed in condensed-phase experiments [
10] as well as studied computationally [
11], and similar diiminoethene conformers have been explored via quantum chemistry with a similar approach as that used here [
12]. Consequently, the spectral properties of the higher-energy
-2-iminoacetaldehyde isomers (isomers 7 and 8, as defined in [
8] and depicted in Figure 1) are computed herein. While theoretically computed spectroscopic data exist for isomers 7 and 8 [
13], the present work aims to provide these at a higher level. Furthermore, new spectral data for isomer 2, as well as updated spectroscopic parameters for isomer 1, the lowest-energy conformation of aminoketene are also presented. Finally, the spectral characteristics of a previously uncharacterized aminoketene tautomer/conformer (7b) are reported in this work. Additionally, our previous work does not provide the intensities for the aminoketene fundamental frequencies, and that is addressed in this work. The revised relative energies presented in this study enhance our ability to model the interconversion and interactions of these molecular species in various environments, including diverse astrophysical regions. As such, several knowledge gaps are present for a more complete characterization of the tautomers and conformers of NH
2CHCO, and this work will provide such.
As done in the previous work, the anharmonic spectroscopic data reported herein will rely upon quartic force fields (QFFs) [
14,
15,
16,
17,
18,
19] computed with explicitly correlated coupled-cluster theory within the F12 formalism [
20,
21,
22]. The use of core electron correlation (“cC”) and scalar relativity (“R”) [
23] adds to the accuracy of the explicit correlation which, itself, better approximates the complete basis set limit for a smaller basis set size [
24,
25,
26,
27,
28,
29]. As such, the so-called “F12-TcCR” QFF (so named for the F12-based energy with a “T” triple-
basis set along with the “cC” and “R” terms) has been shown to provide accuracies for
B and
C spectroscopic constants to better than 0.1% of experiment and fundamental vibrational frequencies within 0.7%, often within 1.0 cm
−1 [
30,
31]. Furthermore, rotational constants for related nitrogen-containing hydrocarbons have been predicted to within 10 MHz of experiment for numerous molecules [
32,
33,
34]. and the rotational constants and fundamental vibrational frequencies of other prebiotic species of astrochemical and astrobiological significance have also been provided [
35,
36,
37,
38,
39]. Hence, this work will utilize this same quantum chemical approach to increase the spectroscopic reference data available for the aminoketene conformers and tautomers. These data will provide for a more complete laboratory experimental exploration of these isomers and will inform where and to what extent these isomers and conformers will be detectable with current, archival, and future multi-messenger observations [
40,
41].
3. Observational and Spectroscopic Considerations
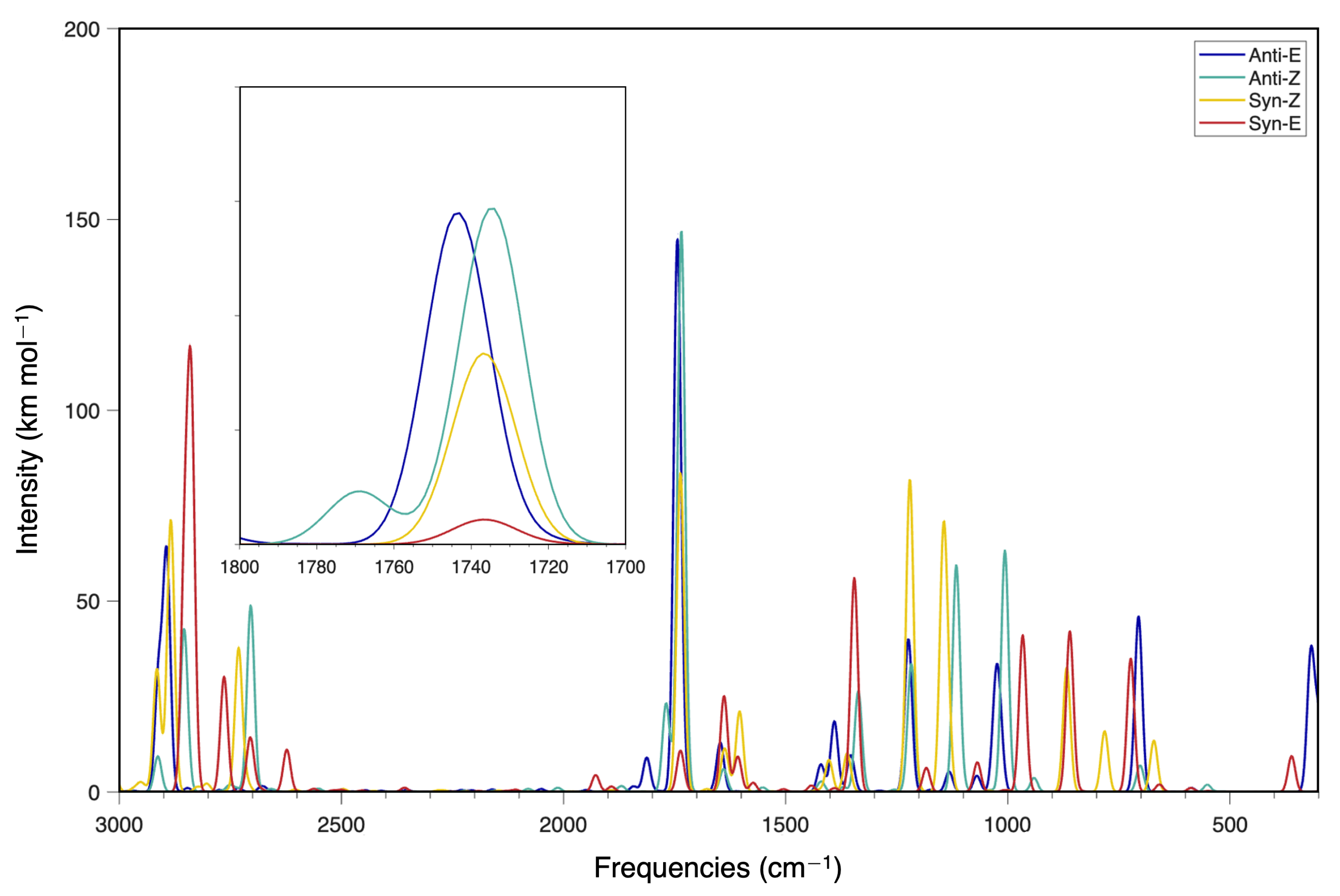
2-iminoacetaldehyde is one of the simplest prebiotic molecules yet to be observed in the ISM or other astronomical regions. Its family of conformers exhibits strong IR absorption in the range of 1720 cm
−1 to 1780 cm
−1 and notable dipole moments for all four conformers (isomers 7–10). Case in point,
Figure 2 displays the F12-TcCR QFF spectra for all four conformers. Clearly, the region from 1700 cm
−1 to 1800 cm
−1 (given as an inset) produces the most intense features as the
C=O stretch fundamental carries the largest intensity. The three lowest-energy conformers all cluster together in this region with the highest energy
-(E) conformer showcasing a blue-shifted fundamental in this region. The C=O stretch of formaldehyde also lies in this region at 1746.1 cm
−1 [
51], but F12-TcCR computations shift this frequency of H
2CO up to 1752.0 cm
−1 [
30]. Hence, the C=O stretches of the 2-iminoacetaldehyde conformers computationally predicted to lie in the region of 1735 cm
−1 should still be distinct from the C=O stretch of the more common formaldehyde molecule.
Additionally,
Figure 2 highlights that population of the first three conformational levels will not shift the frequency of this molecule as the higher energy conformers become populated at higher temperatures. Purely thermodynamically, the
-(E) conformer should be the dominant form observed in the cold ISM. From the relative energies of Ref. [
8], temperatures would have to surpass 115 K before a meager 1% of the population would convert from
-(E) to
-(Z) (isomer 9 to isomer 10) and more than 170 K before
-(Z) would provide a notable contribution to the population of 2-iminoacetaldehyde. The highest energy
-(E) (isomer 8) conformer would require more than 500 K before its population is ever greater than 1%. Hence, the
-(E) should be the primary target for observation in colder regions, but all conformers may contribute to the observed IR or JWST spectra in warmer regions if they are present.
Even with these considerations, the possible unique detection of 2-iminoacetaldehyde will likely arise from radioastronomy due to the nature of emission rotational spectroscopy and from the 2.0+ D dipole moments of the conformers of this molecular family. While the rotational constants and the fundamental vibrational frequencies of the two
-2-iminoacetaldehyde conformers have been computationally explored previously [
4,
8], this present work is expanding that to the
-2-iminoacetaldehyde (Z) and (E) pair. The
conformers are both very much of near-prolate character with
values of −0.98. Due to their similarities, high resolution rotational spectroscopy would be required to separate the rotational spectra of the
conformers. Most notably, the differences in the
constants are less than 4.0 MHz while this grows to more than 15 MHz in the
constants [
4] However, the dipole moments and rotational constants of the
conformers are notably separated from each other and from the two
conformers. Hence, they will produce other lines in the spectra as these higher-energy conformers are populated at higher temperatures. Regardless, the small number of heavy atoms (4) puts this molecule in the range of known prebiotic complex organic molecules [
1,
2,
52]. In addition to ethanolamine, the conformers of 2-iminoacetaldehyde exhibit nearly the same skeletal structure as glycine in the N-C-C-O connectivity, missing only an additional oxygen atom bonded to the aldehyde carbon and a couple of ubiquitous hydrogen atoms.
This connectivity is maintained for the aminoketene conformers (isomers 1 and 2). Only the positions of the hydrogen atoms change. While the conformers of 2-iminoacetaldehyde are the lowest energy isomers, the NH
2CHCO conformers could still be the first of these isomers synthesized in astronomical environments potentially in shocked regions [
3,
5]. As such, the spectroscopic constants provided in this work are vital for understanding the possible populations of these isomers astrophysically. Like isomers 9 and 10, the spectroscopic constants of isomers 1 and 2 differ by less than 50 MHz for the
B constant and less than 10 MHz for the
C constant. As such, the rotational signals of the two NH
2CHCO conformers may combine with lower-resolution laboratory characterization or possible astronomical observation. Hence, shifts in population between the aminoketene isomers would not greatly change the observations.
While laboratory work would need to conclusively determine the rotational constants for any of these isomers, the present work should be providing rotational constants to within 10 MHz or less of experiment [
30,
34], greatly reducing the need to comb the spectra significantly in order to make attributions. Finally, the ratio of aminoketene to 2-iminoacetaldehyde would also provide insights into the nature of the formation of these molecules themselves, and it would also be able to give a clue as to the evolution of relatively simple prebiotic molecules.
5. Conclusions
A more complete spectral picture of the tautomers and conformers of NH
2CHCO is produced in this work, notably showcasing that the two aminoketene conformers have strong IR features in the range of 4.6–4.7 μm (2125–2150 cm
−1) potentially in competition with functionalized PAHs in astronomical observations where C≡N stretches have been reported recently [
45]. Hence, these two molecules are also notable targets for their IR features as well as their radioastronomical (rotational) signatures. Since the slightly lower-energy equatorial conformer (isomer 1) has a nearly identical dipole moment as the axial conformer (isomer 2), either can be observed rotationally and would likely be observed in similar amounts. Detection of either would help to add evidence for this molecule’s role in the interstellar formation of the known prebiotic molecule ethanolamine.
The 2-iminoacetaldehyde conformers from this and previous work [
4] all exhibit notable dipole moments of at least 2.0 D if not greater than 4.0 D. Their IR fundamental vibrational frequencies are also populated with intense
C=O stretch fundamentals enabling their observation at frequencies in the range of 1700 cm
−1 to 1800 cm
−1. The presently examined
-(Z) and
-(E) conformers of 2-iminoacetaldehyde or even the lower-energy
-(E) and
-(Z) conformers are likely not part of the synthesis of ethanolamine. They would be required to migrate a hydrogen atom to create the amine group. However, they may have other roles to play astrochemically given their notable mid-IR intensities, and these data provide the necessary references for their potential observation.
Finally, a new isomer, isomer 7b NH2CCOH, is also shown to be a local minimum in this work despite being a carbene. While much higher in energy that most of the rest of the isomers examined this and the previous work, it could still form from other chemical reactions. Hence, its spectral data are produced in this work, as well.