Abstract
In recent years, transdermal drug delivery systems have gained considerable attention for their ability to enhance patient compliance and provide consistent drug release. However, the barrier function of the skin’s outermost layer remains the foremost challenge in these systems. As a result, there is a growing interest in the use of ethosomes as a cutting-edge nanocarrier in drug delivery. This review presents the concept and mechanism of action of ethosomes as well as the numerous types and preparation techniques available. It also discusses the significance of characterization methods and how they impact ethosomal properties along with recent advancements and applications in the realm of transdermal drug delivery. Ethosomes exhibit tremendous potential for the pharmaceutical industry, and optimizing preparation methods, performing comprehensive property studies, and exploring their applications are critical areas for future research.
1. Introduction
Transdermal drug delivery is a significant area of research in drug delivery due to its convenience, safety, and effectiveness as a non-invasive method of drug administration. It provides various benefits such as bypassing the first-pass effect linked to gastrointestinal absorption, improved patient compliance, sustained and controlled release, and reduced drug metabolism [1,2,3]. However, the skin’s natural barrier significantly restricts drug penetration and absorption during transdermal administration, which limits the usefulness of certain drugs. To overcome this limitation, researchers strive to improve drug permeability and absorption for effective transdermal delivery [4,5].
In recent years, novel nanocarriers have shown significant potential for drug delivery [2,6,7,8,9]. Ethosomes are drug complexes formed by combining a drug with a carrier, typically an alcohol or its derivatives. This carrier contains an active alcohol component that provides better permeability and drug loading capacity than traditional liposomes [10,11]. Ethosomes have demonstrated two major benefits: enhancing drug–skin interactions by improving drug absorption and penetration and being easily adaptable to various drug types, including water-soluble, fat-soluble, and unstable drugs. As a result, using ethosomes can increase drug stability and reduce systemic side effects and drug wastage [12]. Furthermore, ethosomes have various advantages in the realm of transdermal drug administration and have potential uses across sectors such as pharmaceuticals, biotechnology, veterinary medicine, cosmetics, and nutrition, among others [13]. Most of the products marketed to date are pharmaceuticals. A specific instance is the Decorin cream from Genomic Cosmetics, based in Pennsylvania, the USA, which aims to tackle anti-aging and pigmentation issues [14]. Noicellex and Supravir are topical creams created by Novel Therapeutic Technologies and Trima, respectively. Noicellex is an anti-cellulite cream designed to enhance the effectiveness of its active ingredient by deepening its penetration. In contrast, Supravir is a cream that encases acyclovir, a drug employed to treat herpes infections by disrupting lipids [15], whereas Cellutight EF is a topical anti-cellulite cream manufactured by Hampden Health (USA). The lotion is formulated with a potent combination of components that enhance metabolic rates and decrease fat by penetrating deeper into the skin [16].
The purpose of this review is to present a comprehensive analysis of existing research pertaining to ethosomes, a contemporary drug delivery technology employed in the field of transdermal drug delivery. The review largely centers on the many classifications of ethosomes, their underlying mechanisms, methods of manufacturing, influential variables, and clinical investigations. In this discourse, we shall explore the obstacles and opportunities related to the facilitation and enhancement of ethosomes as a mechanism for delivering drugs via the skin in transdermal drug delivery. The objective of this study is to offer a thorough comprehension of the research on transdermal medication delivery using ethosomes while also providing valuable insights and suggestions for future researchers in this particular field of study.
2. The Type of the Ethosomes
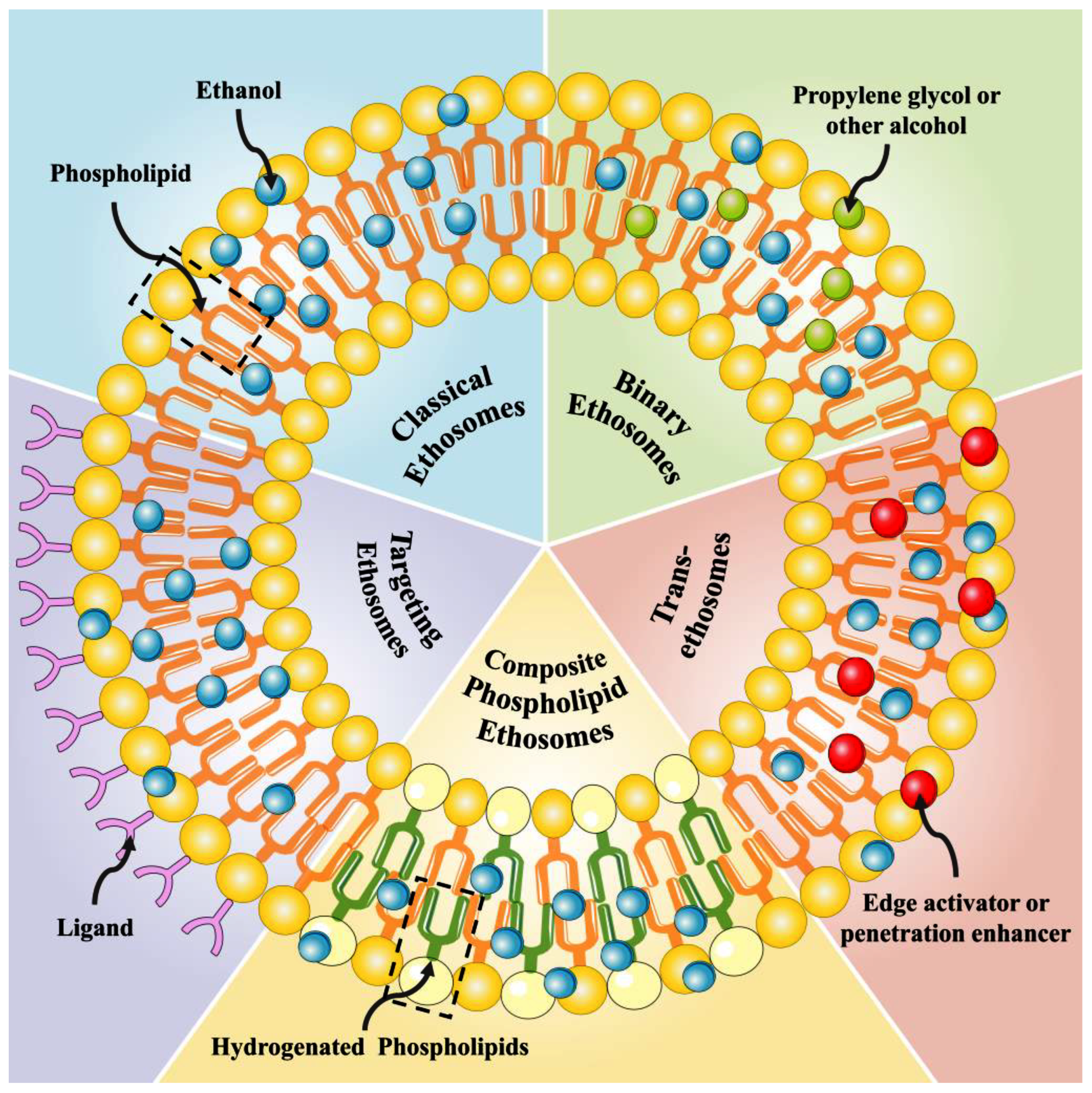
Ethosomes can be classified according to their composition into several categories, including classical ethosomes, binary ethosomes, transethosomes, composite phospholipid ethosomes, and active targeting ethosomes. Each of these categories possesses distinct traits and advantages (Figure 1).
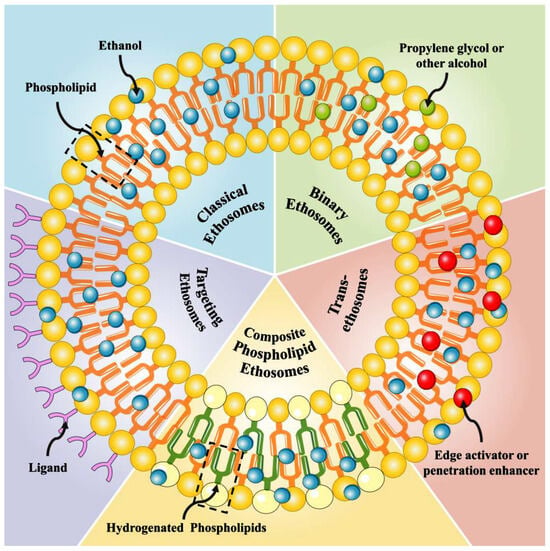
Figure 1.
The structure of different ethosomes.
2.1. Classical Ethosomes
Classical ethosomes are stable vehicles for delivering drugs and composed of phospholipids, cholesterol, ethanol, and water in varying concentrations. These vehicles can be used to transport active ingredients effectively and efficiently [17]. Compared to common liposomes, classical ethosomes have a high concentration of ethanol, resulting in better permeability. For instance, Yucel et al. developed ethosomes and liposomes loaded with rosmarinic acid. The study showed that ethosomal formulations could enhance drug permeation across human skin and had a higher transdermal flux than liposomes [18]. Feng et al. prepared ethosomes and liposomes of psoralen. The transdermal flow and skin deposition of psoralen in ethosomes were found to be 38.89 ± 0.32 mg/cm2/h and 3.87 ± 1.74 mg/cm2, respectively. These values were observed to be 3.50 and 2.15 times higher than those observed with liposomes [19]. The above results have all shown that the ethosomes have a better permeability compared to the ordinary liposomes.
2.2. Binary Ethosomes
Binary ethosomes are produced through enhancements to classical ethosomes by using a propylene glycol and ethanol mixture in the preparation process instead of single ethanol. This reduces the amount of ethanol and its volatility, increasing drug solubility and formulation stability as well as promoting drug penetration [20,21,22]. Many researchers have prepared binary ethosomes to increase formulation stability and improve skin penetration. For example, Wu et al. prepared ethosomes, binary ethosomes, and transfersomes containing terbinafine hydrochloride. The study’s findings indicated that binary ethosomes composed of ethanol and propylene glycol in a 7:3 (w/w) ratio were the most effective in enhancing drug permeation through the skin. Additionally, rhodamine B exhibited a greater depth of penetration and fluorescence intensity when delivered through binary ethosomes compared to ethosomes and transfersomes [17]. Akhtar et al. prepared ethosomal gel and binary ethosomal soy lecithin and binary mixture (1:1) gel formulations of triamcinolone. The vesicles exhibited higher zeta potential and EE and improved Rhodamine B penetration than the reference ethosomal gel [23].
2.3. Transethosomes (TEs)
TEs represent a newer generation of ethosomal systems, initially introduced by Song et al. [24]. This vesicle type has been developed to merge the advantages of both ethosomes and TEs into a single composition [10]. The formulation of TEs involves the incorporation of a penetration enhancer or surfactant, such as Tween 20, Span 60, sodium cholate, or deoxycholic acid sodium, into traditional ethosomes [25]. The surfactant is inserted into the phospholipid bilayer, which increases the distance between the phospholipid molecules, disrupts the sequence of the phospholipid bilayer and increases the fluidity of the ethosomes. Upon skin hydration, the ethosomes undergo deformation and squeeze into the stratum corneum, thus facilitating the transdermal absorption of the drug. For example, Moolakkadath et al. prepared fisetin TEs that showed better penetration than conventional fisetin gel [26]. Albash et al. prepared olmesartan TEs that showed higher permeability than drug suspensions in dermal delivery studies of Olmesartan [27].
2.4. Composite Phospholipid Ethosomes (CE)
Compared to conventional ethosomes, CE consists of both saturated and unsaturated phospholipids (soybean lecithin, phosphatidylcholine (PC), hydrogenated lecithin). This composition effectively inhibits unsaturated phospholipid oxidation [28]. For example, Chen et al. prepared a curcumin-loaded CE to improve the stability and transdermal absorption of the conventional ethosomes. The curcumin CE prepared with a 1:1 ratio of PC/hydrogenated phosphatidylcholine (HPC) demonstrates superior vesicle stability and flexibility compared to traditional ethosomes. The high stability is because of the interaction between saturated phospholipids and unsaturated phospholipids in the phospholipids, which can effectively inhibit the oxidation of unsaturated phospholipids [29].
2.5. Actively Targeted Ethosomes
Actively targeted ethosomes are prepared by modifying certain ligands, including galactosylated chitosan, hyaluronic acid (HA), polyethyleneimine, and sodium cholate, on the surface of conventional ethosomes [30]. Zhang et al. prepared ethosomes modified with hyaluronic acid (HA) to enhance their therapeutic efficacy through specific targeting of the CD44 protein, which is known to be upregulated in inflamed psoriatic skin [31]. Sagar et al. prepared a study in which they formulated ethosomes loaded with hepatitis B surface antigen (HBsAg). Their findings revealed that these ethosomes exhibited superior internalization capabilities and immunogenicity when compared to elastic liposomes [32,33].
3. Mechanisms for Penetration of Ethosomes
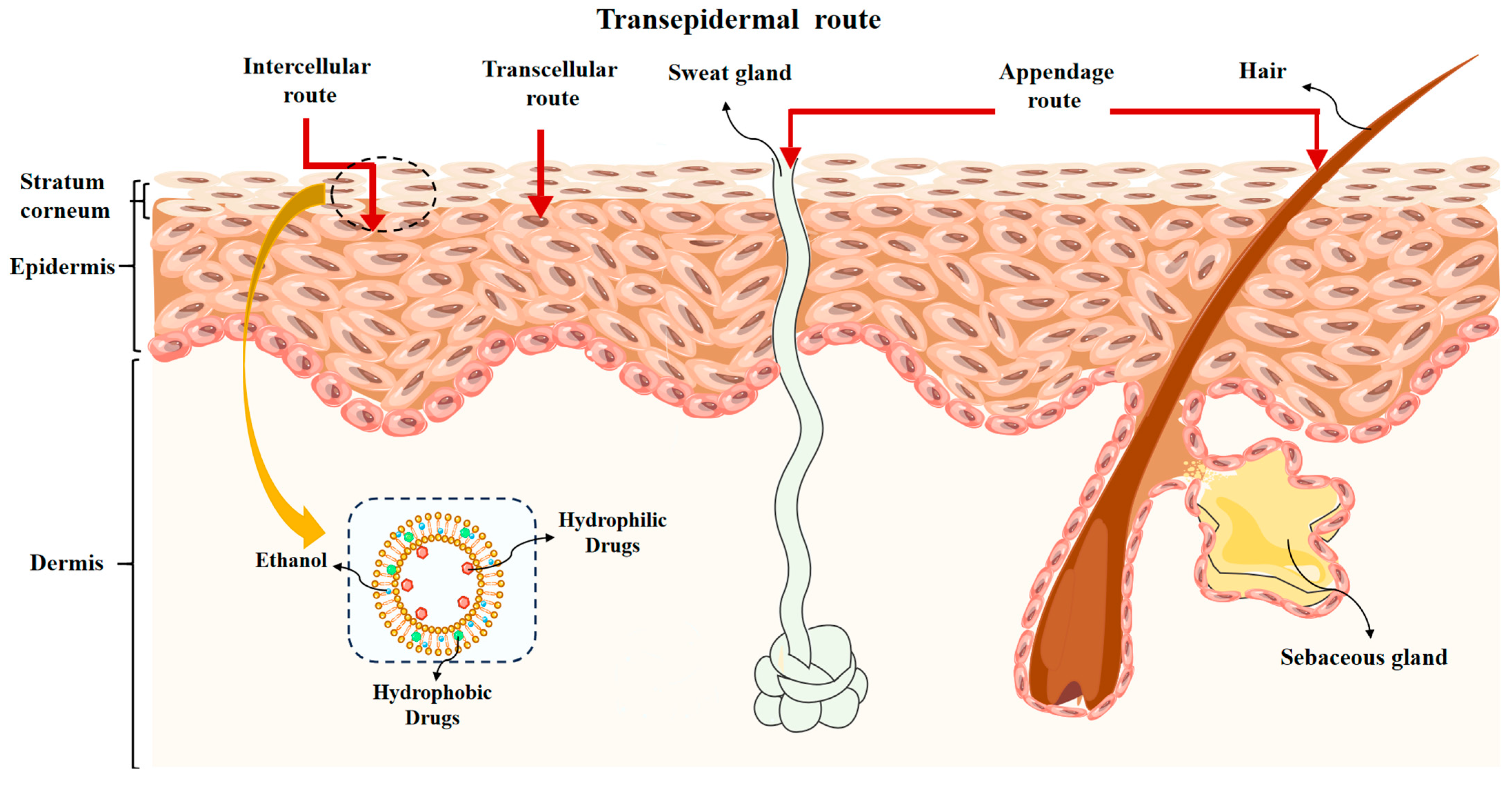
The skin, a vital organ of the body, comprises the epidermis, dermis, and subcutaneous layers. It plays a crucial role in the process of transdermal absorption. It is commonly accepted that drugs transported through the epidermis are quickly removed from the capillaries without an absorption barrier forming. The epidermis is made up of multiple layers, such as the outermost SC layer and the viable epidermal layer, consisting of the hyaline, granular, spiny, and basal layers. The SC layer, with a thickness of approximately 10~20 microns and no phospholipids, is regarded as a non-living layer of dead cells that facilitates drug penetration through a passive diffusion process regulated by physicochemical laws. Therefore, the outermost stratum corneum (SC) layer of the skin functions as the principal barrier for the process of transdermal absorption.
There are two distinct routes for drugs to penetrate the skin and enter the systemic circulation: the epidermal route and the appendage route. The drug, when applied to the skin via the epidermal route, is released from the preparation onto the skin surface. Drugs dissolve and pass through either the intercellular lipid (intercellular route), keratinocytes and intercellular lipids (transcellular route), or both (Figure 2). It then diffuses into the active epidermis and distributes to the water-active epidermis before finally diffusing into the dermis, where it is eventually absorbed by capillaries. The primary method of drug absorption through the skin is through the epidermal route. Drugs have the ability to permeate the skin appendages at an accelerated pace; these appendages only comprise about 0.1% of the skin’s total surface area, making them a less common pathway for drug absorption in most scenarios. For certain ionic medications and macromolecules that are water soluble, passage through the stratum corneum, which is lipid-rich, is a challenging task.
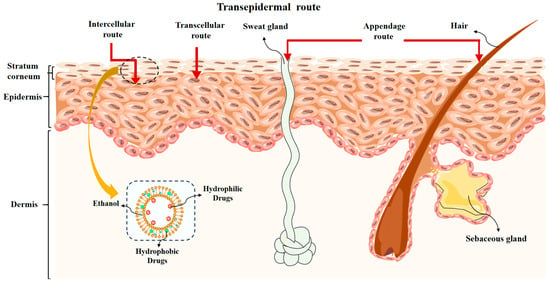
Figure 2.
The transepidermal route of drug through the skin.
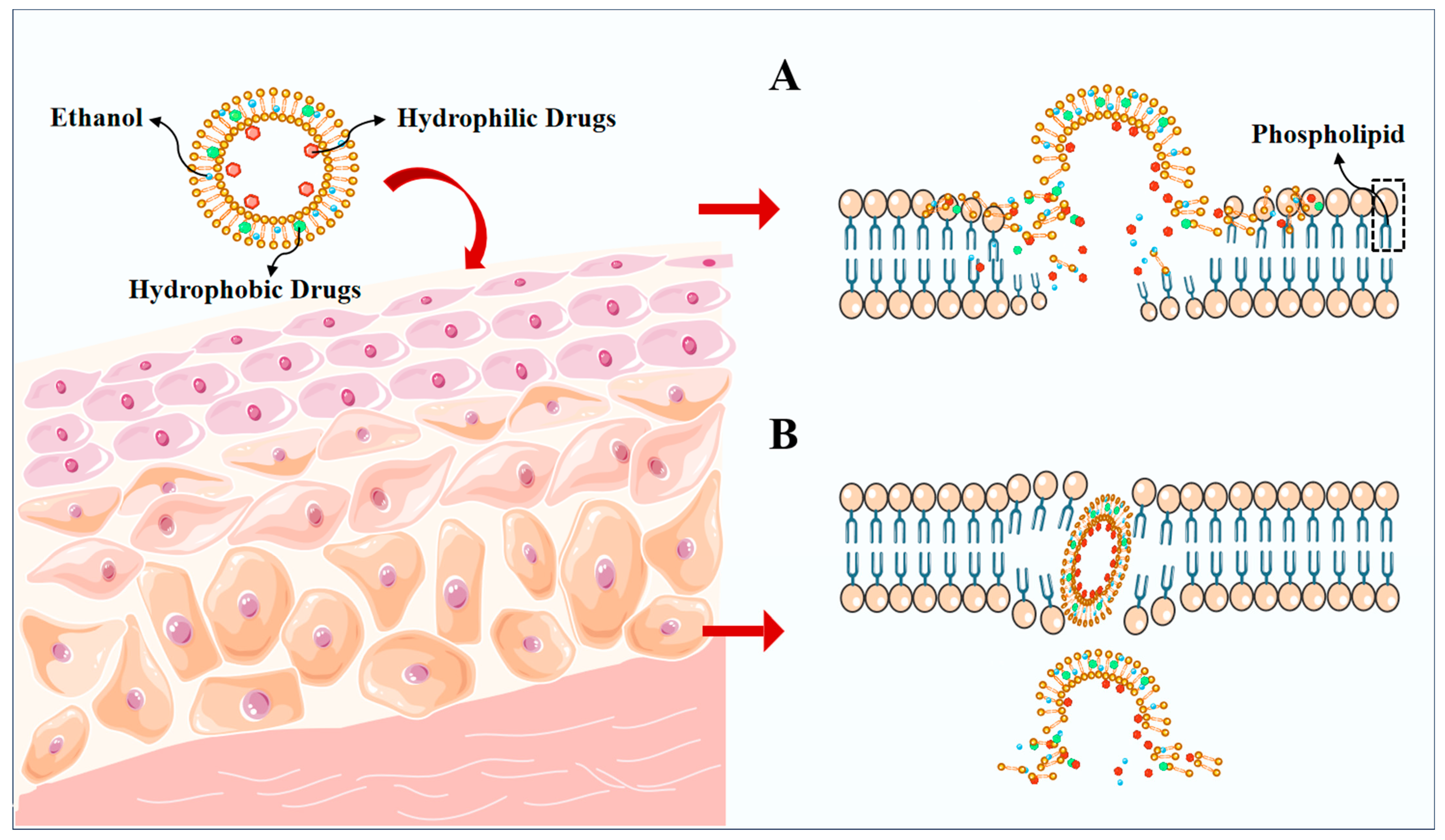
The high permeability of ethosomes is connected to the large amount of ethanol they contain, and the disruption of the lipid structure of the stratum corneum can occur. This results in an active stratum corneum, larger gaps between cells, and the increased flexibility and fluidity of the phospholipid layer. Ethosomes may potentially aid drug penetration during transdermal penetration by disrupting the skin barrier through the following mechanisms: (1) The disruption of the skin barrier through the presence of surfactants within ethosomes, which has the potential to disrupt the barrier function of the stratum corneum, hence enhancing the permeation of drugs into the skin. (2) Ethosomes contain water-soluble polymers that can form hydrates with water molecules present in the skin, augmenting the solubility and permeability of the drug in the skin. (3) Ethosomes act as a carrier for the drug. Alcohol plasmas act as drug carriers, stably encapsulate drugs within microparticles, protect against skin metabolizing enzymes, and enhance drug permeability and residence time [34,35,36,37]. The pliable and adaptable ethosomal vesicles display amplified permeability across the unordered lipid bilayers of the stratum corneum, a commonly acknowledged occurrence referred to as the “ethanol effect” (Figure 3A) [28,38,39]. The fusion of phospholipids present in ethosomes with the skin layers underneath produces the “ethosomes effect”, which aids in the improved penetration of the unbound medication (Figure 3B) [28,38,40,41]. In summary, the impact of ethanol on the lipids of the stratum corneum and the fluidity of vesicles, along with the potential for interaction between ethosomes and the stratum corneum, may result in enhanced drug delivery.
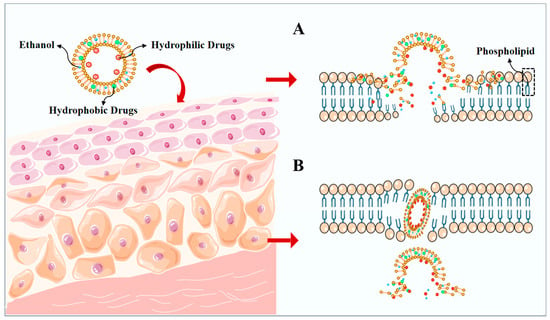
Figure 3.
A schematic representation of the main permeation mechanisms of ethosomes. (A) ethanol effect, (B) ethosomes effect.
4. Methods of Ethosomal Penetration Mechanisms
The studies typically elucidate the penetration mechanisms of ethosomes through molecular conformation, thermodynamic properties, ultrastructure, and fluorescent labeling at present. The researcher employed various analytical methods, such as attenuated total reflection Fourier transform infrared spectroscopy (ATR-FTIR), Raman spectroscopy, differential scanning calorimetry (DSC), and imaging techniques, including scanning electron microscopy (SEM), transmission electron microscopy (TEM), and confocal laser scanning microscopy (CLSM) [34,42,43].
4.1. ATR-FTIR and Raman Spectroscopy
ATR-FTIR is a method utilized for molecular-level characterization of skin SC groups. The vibrations of CH2, including the asymmetric stretching at 2920 cm−1, symmetric stretching at 2850 cm−1, and shear vibration at 1460–1470 cm−1, can be linked to the intercellular lipids of the stratum corneum [28]. The CH2 shear bands indicate the barrier effect of the orthogonal lattice on lipids, the ratio between the maximum intensity of υsym CH2 and υasym CH2 (H2920/2850), which is thought to be a measure of the lateral interaction between acyl chains in the stratum corneum [44]. After treating skin with DPH ethosomes, Xiao-Qian Niu et al. observed a notable increase in the values of both symmetric and asymmetric methylene stretching bands when compared to the control samples, with a CH2 cleavage width of 66.85 ± 2.13, indicating an ordered–disordered lipid transition. The treatment of pig skin with a formulation containing vesicles showed a significant decrease in H2920/2850 values and an increase in A2850 values compared to the control [38]. The study conducted by Virginia Campani revealed that EthoVK1 resulted in a noteworthy reduction in H2920/2850 values by 1.71 ± 0.03 compared to the untreated group. The use of ATR-FTIR spectroscopy indicates that ethosomes penetrate the stratum corneum, enhancing lipid fraction enrichment and ordered structure formation in the skin, facilitating further drug penetration [45].
Raman spectroscopy can be used to study the molecular structure and chemical properties of ethosomes to understand the effect of ethosomes on drug stability and bioavailability. Aika Sekine et al. utilized Raman spectroscopy to examine the interaction between DSPC liposomes and polyols. The results exhibited broader peaks and slightly lower wave numbers of DSPC liposomes–BG and DSPC liposomes–PG compared to DSPC liposomes–Gly. This suggests that the inclusion of BG and PG in DSPC liposomes leads to a compact stacking arrangement of the compound in the bilayer [46].
4.2. DSC
In recent years, ethosomes have been extensively studied by researchers using DSC techniques, and some important results have been obtained. The investigation of the phase change characteristics of ethosomes within the spectrum of skin temperature allows for a comprehensive comprehension of the underlying mechanism governing the interaction between ethosomes and the skin. This study aims to enhance the comprehension of the transdermal transport mechanism of ethosomes and to optimize the design and application of these ethosomes [47].
J Xiaowei et al. designed KP-loaded functionalized single-walled carbon nanotube composite ethosomes (f-SWCNTs-KP-ES). DSC indicated that KP was present in an amorphous state following adsorption and loading onto f-SWCNTs. Moreover, ethosomes were found to enhance transdermal absorption and increase the bioavailability of KP [48]. Yu Li et al. formulated and subsequently compared ethosomes loaded with curcumin (referred to as CE) that possessed varying phospholipid compositions. The use of differential scanning calorimetry (DSC) revealed a noteworthy interaction between unsaturated phosphatidylcholine (PC) and saturated hydrogenated phosphatidylcholine (HPC). This interaction led to a decrease in PC peroxidation when HPC was present, hence disturbing the lipid domain of SC [29].
4.3. SEM and TEM
SEM and TEM allow for the macroscopic observation and microstructural analysis of the skin to further visualize the inner layers of the skin and thus evaluate the potential of ethosomes for transdermal drug delivery [49,50].
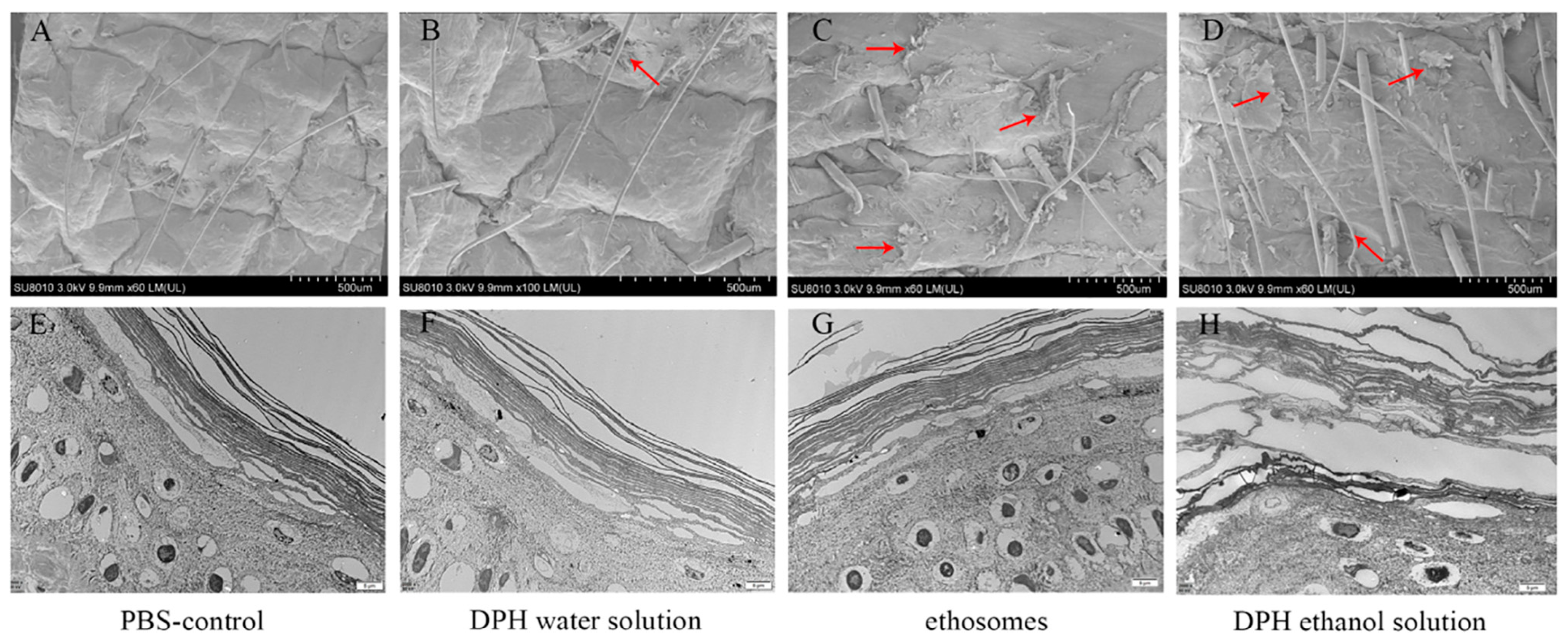
Xiaoqian Niu et al. observed by SEM that there were almost no detached SC fragments in the skin samples treated with PBS and diphenhydramine (DPH) aqueous solution; a significant number of fragments were observed to detach. Conversely, in the pig skin samples treated with DPH ethosomes and DPH ethanol, a substantial amount of fragments were also observed to detach. Thus, when porcine skin was soaked in DPH ethosomes and 30% ethanol enhancer solution, intercellular lipids were dissolved and extracted, resulting in separation from intact SC and desquamation. The ultrastructure of porcine skin was observed by TEM. The results showed that compared with the dense SC cells in the control group, the keratinocyte was destroyed, the SC cell layer was further expanded, and the intercellular gap between the cells in the stratum corneum appeared to be expanded; the results are shown in Figure 4 [38].
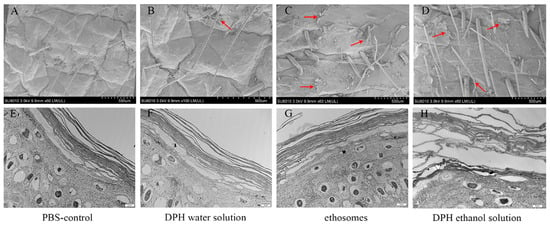
Figure 4.
SEM and TEM of different formulation-treated skin samples. (A) SEM of skin-applicated PBS for 24 h. (B) SEM of skin-applicated DPH water solution for 24 h. (C) SEM of skin-applicated DPH ethosomes for 24 h. (D) SEM of skin-applicated 30% ethanol DPH solution for 24 h. (E) TEM of SC-applicated PBS for 24 h. (F) TEM of SC-applicated DPH water solution for 24 h. (G) TEM of SC-applicated DPH ethosomes for 24 h. (H) TEM of SC-applicated 30% ethanol DPH solution for 24 h. The pictures in the same column belong to the same group, and the red arrow represents SC exfoliation. (Reproduced with permission from reference [38]. Copyright 2019, Int J Pharm X).
4.4. CLSM
CLSM is a comprehensive technique that provides high-resolution three-dimensional images and has fluorescent dye labeling technology, which is commonly used to observe and analyze the transdermal transfer process of ethosomes. By observing the distribution of ethosomes and fluorescent dyes, the distribution of ethosomes on the skin surface, the degree of penetration, and the interaction with the stratum corneum can be determined [51,52].
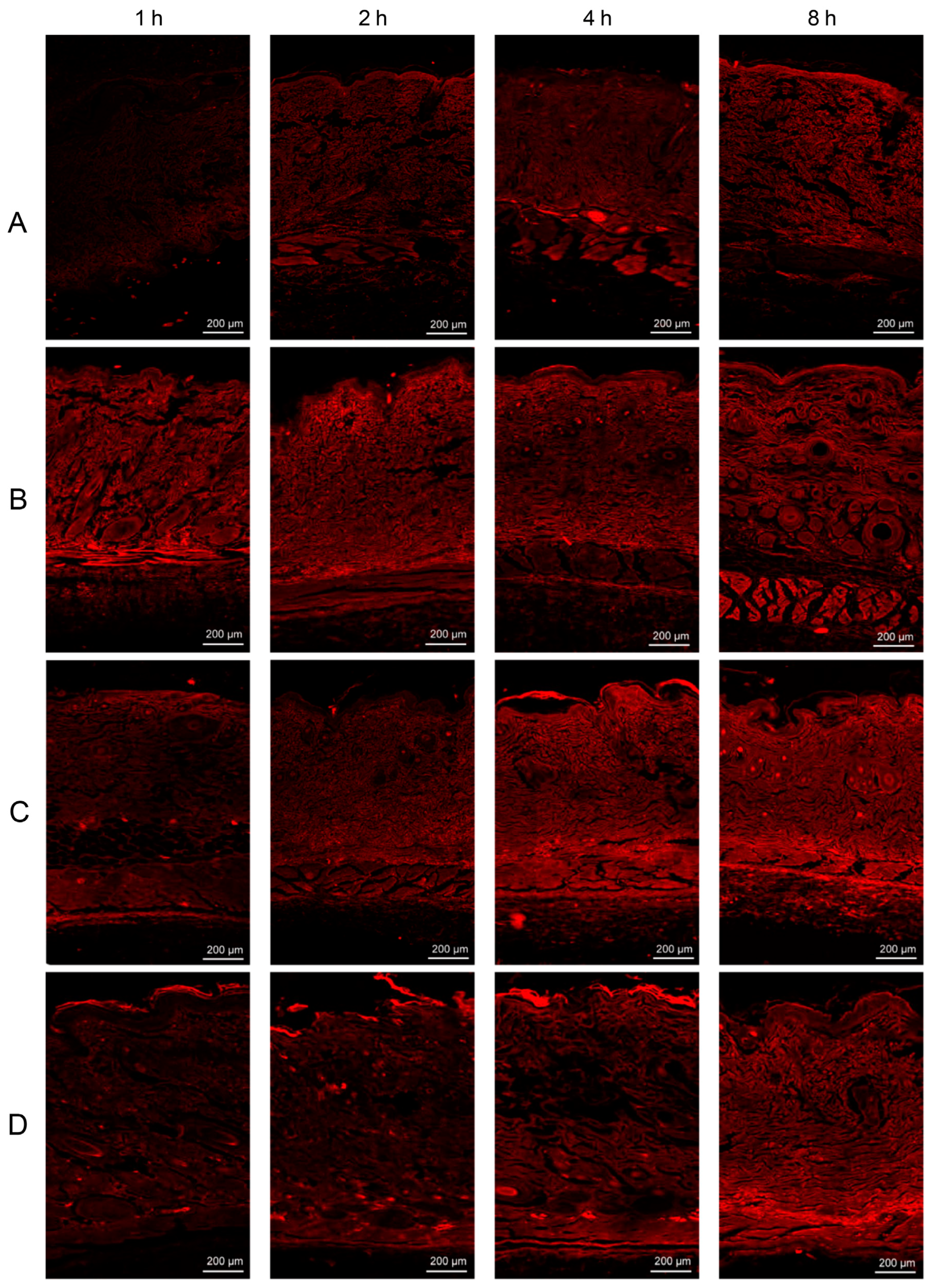
CLSM with dual-fluorescent labeled ethosomes was performed to investigate the depth of penetration of lipophilic substances contained in ethosomes into the skin’s underlying layers. The localization of the fluorescent tracer gradually increased through the skin layer over time (1, 2, 4, and 8 h) for all formulations. In addition, the three lipid vesicles facilitated the penetration of RhB into the skin compared to the plain RhB hydrogel and improved the penetration of rhodamine-loaded nanovesicles through skin layers with a wider distribution and higher fluorescence intensity; the results are shown in Figure 5 [53]. Yi-Ping Fang et al. used the CLSM technique to examine the intensity and depth of protoporphyrin IX (PpIX) in order to gain insights into the penetration behavior of PpIX synthesis produced by 5-aminolevulinic acid (ALA) in both normal skin and overproliferating mouse skin models. The findings of the study indicated that the overall concentration of ALA in an aqueous solution of ALA and ALA encapsulated in an ethosome carrier (PE) in the skin of the mice was measured to be 255.25 ± 42.09 and 930.07 ± 96.32, respectively. Furthermore, it was observed that the PpIX molecules exhibited a maximum depth of penetration of 30 μm into the skin of the mice, with an extension of around 80 μm [54].
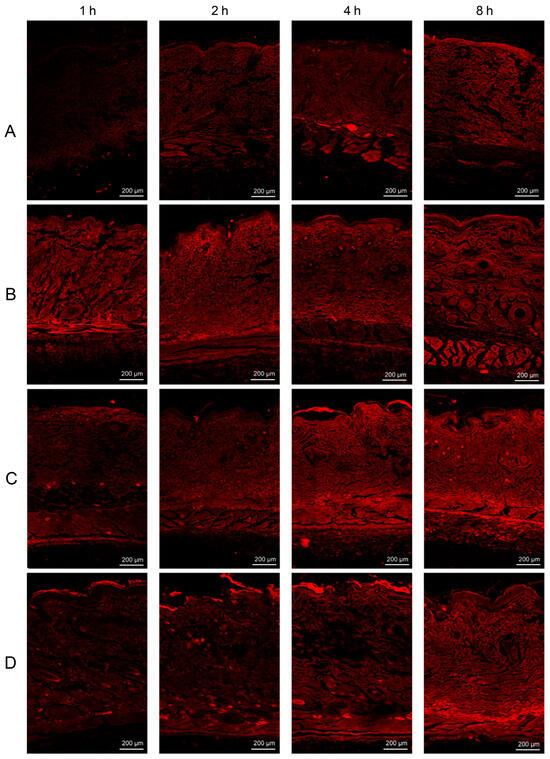
Figure 5.
CLSM images of (A) plain RhB hydrogel, (B) RhB-L hydrogel, (C) RhB-E-30 hydrogel, and (D) RhB-T-SDC hydrogel at 1, 2, 4, and 8 h post in vivo application on dorsal rat skins. (Reproduced with permission from reference [53]. Copyright 2019, Int J Pharm).
5. The Method of Preparation of Ethosomes
Researchers have used different methods to obtain the optimal vesicle of ethosomes. The preparation method of ethosomes is relatively simple compared to liposomes because ethosomes do not require the removal of organic solvents. The most common methods for preparing ethosomes include the ethanol injection method, rotary film evaporation, vortex sonication, the pH gradient method, and the microfluidic technique.
5.1. Thin Film Dispersion Method
The recommended quantity of phospholipids and cholesterol is dissolved in a limited quantity of suitable organic solvents. This solution is then subjected to a rotary evaporator to eliminate the solvents, resulting in the formation of a consistent lipid film on the inner surface of the container. The drug was first dissolved in a volume of water–ethanol mixture, then, with the rotation of the lipid film hydration, left at room temperature and filtered via ultrasound to obtain a suspension ethosomes [55,56,57]. The ethosomes prepared by this method have a large particle size, which requires an ultrasonic probe to make the particle size smaller and more uniform, but it is not easy to produce precipitation, and the system is stable.
5.2. Ethanol Injection Method
The ethanol injection method has the following steps: dissolve phospholipids and cholesterol in ethanol, place them in a closed container, slowly add the water phase while stirring, continue stirring for a certain period of time, and filter it through the microporous membrane to obtain the solution. In general, lipophilic drugs can be dissolved in ethanol first. Ursolic acid, paeonol, and benzocaine ethosomes are prepared by this method [58,59,60]. Hydrophilic drugs are first dissolved in water during preparation, phospholipids and cholesterol are dissolved in an alcohol solution, and then the aqueous solution is slowly added to the ethanol solution, stirred for a certain period of time, and cooled to obtain ethosomes [61,62,63]. Vancomycin hydrochloride and anthralin ethosomes are prepared by this method [5,64]. The ethosomes prepared by this method are difficult to pass through the microporous filter membrane, and the prepared particle size is relatively large, which is easy to adsorb and causes precipitation and delamination.
5.3. Injection–Ultrasound Combination Method
A certain amount of phospholipid and the main drug are dissolved in ethanol, placed on a magnetic stirrer, and slowly injected into the phosphate buffer while stirring under airtight conditions at room temperature; the stirring is continued for a certain period of time, and then, the probe is ultrasonicated in an ice water bath and passed through the micropore to obtain the ethosomes [65]. Compared with the injection method, the combined injection–ultrasound method adds the step of ultrasound, which may be related to the reduction in the particle size distribution of the ethosomes. The ethosomes prepared after the ultrasound probe have a small and uniform particle size, which is advantageous for permeating the microporous membrane and is not easily precipitated, and the system is stable.
5.4. pH Gradient Method
Under airtight stirring conditions, the buffer is injected into the phospholipid-dissolved ethanol solution; after the injection is completed, stirring is continued for a certain period of time, and the solution is cooled at room temperature to obtain blank ethosomes. The drug is added while stirring to dissolve it completely, and then, NaOH is added to adjust the pH of the outside of the vesicles so that a pH gradient is formed inside and outside the vesicles, and then, the whole system is fully incubated at a certain temperature. After a period of time, the main drug passes through the lipid bilayer and enters the vesicles to yield ethosomes [20,59,66]. This method involves changes in pH during the preparation process, so it is only suitable for drugs whose physical and chemical properties are not affected by pH [67]. The encapsulation rate of water-soluble drugs is very low because hydrophilic drugs are not easily adsorbed on the surface of lecithin. Therefore, the pH gradient method is suitable for lipophilic drugs.
5.5. Microfluidic Techniques
Phospholipids and cholesterol are dissolved in a certain concentration of ethanol and stirred thoroughly, followed by dissolving the drug in a lipid ethanol solution on a magnetic stirrer; the organic ethanol solution was mixed with water in a certain ratio and finally prepared by NanoAssemblr [68]. Dexamethasone-loaded liposomes were prepared by this method [69]. The liposomes prepared by microfluidics have a monolayer structure, and the liposomes obtained by this method have higher assembly capacity and efficiency. The liposomes prepared by this method had uniform, tunable, and reproducible particle size [70].
6. Factors Affecting Properties of Ethosomes
During the pursuit of an optimum formulation of ethosomes, several process variables may exert an influence on the characteristics of the ethosomes. These variables mostly pertain to the formulation preparation of ethosomal formulations, which are classified as follows.
6.1. Effect of Ethanol
Ethanol assumes a significant function within ethosomal systems, as it bestows distinctive characteristics upon the vesicles, encompassing size, zeta potential, stability, encapsulation efficiency (EE), and skin permeability. Reportedly, the percentage of ethanol in ethosomal systems typically varies between 20% and 45% (w/w). It has been observed that when the ethanol concentration decreases, the size of the vesicles in these systems tends to rise [71]. In addition, ethanol makes the vesicles negatively charged to improve the stability of the vesicles and avoid the accumulation of the vesicle system due to static electricity. A high concentration of ethanol can not only increase the solubility of the drug in the stratum corneum of the skin but also promote the penetration of the drug. In the process of transdermal penetration, the ethosomes can deform, pass through the cell gap smaller than its own particle size, and reach the deep layer of the skin [72]. Therefore, the penetration depth of ethosomes is deeper than that of liposomes, which significantly improves the efficiency of drug transdermal penetration. The presence of a high concentration of ethanol has the potential to decrease the thickness of the vesicle membrane layer, thereby impacting the EE [73].
6.2. Effect of Phospholipids
The phospholipid concentration in an ethosomal formulation falls within the range of 0.5% to 5%. It could influence the size and EE of the ethosomes [74,75]. The vesicle size increased slightly or moderately with the increase in phospholipid concentration, and the EE of ethosomes first increased and then decreased with the increase in phospholipid concentration [76]. The phospholipids also play an important role in the process of transdermal penetration. The phospholipids could fuse with the lipids in the skin stratum corneum to disrupt the dense and ordered structure of the skin stratum corneum, allowing for the drug to be released from the ethosomes and enter the skin in a free state [77].
6.3. Effect of Propylene Glycol
It was found that propylene glycol could affect the size, EE, skin permeation, and stability of the ethosomes. The incorporation of propylene glycol into the ethosomal systems could further reduce the particle size of the vesicles [72]. Propylene glycol may also improve the volatility and skin irritation of the ethanol and increase the stability of the vesicles. At the same time, when propylene glycol is used in combination with other accelerators, it can not only increase the solubility of the drug and the accelerator but also can synergistically promote the penetration of the drug. The presence of ethanol and propylene glycol in ethosomes can also increase the EE of the drug and improve the distribution of the drug in the vesicle [75,78].
6.4. Effect of Cholesterol
The use of cholesterol has been observed to enhance the EE of ethosomes while concurrently augmenting the skin penetration capability of liposomes by promoting membrane fluidity. Excessive cholesterol will result in a decrease in the ability of vesicles to encapsulate drugs. Cholesterol can also increase the spatial stability of the ethosomal system, thereby reducing vesicle aggregation and particle size [79,80].
6.5. Effect of Edge Activators
The careful choice of an appropriate edge activator or penetration enhancer is a crucial aspect in the formulation of ethosomes since it significantly impacts the characteristics of the ethosomal system. After the edge activator is inserted into the phospholipid bilayer, it can increase the distance between the phospholipid molecules, interfere with the sequence of the phospholipid phthalide chain, and increase the fluidity of the ethosomes. Under the hydration of the skin, the ethosomes deform and pass through the stratum corneum, thereby promoting the transdermal absorption of the drug [81,82]. Previous studies have indicated that the inclusion of Tween 80 in ethosomal formulations leads to a decrease in vesicle size, enhancement of system stability, and improvement of skin permeability characteristics [72,83].
7. Characterization of the Ethosomes
Various methodologies have been documented in the literature for the determination of characterisation criteria pertaining to ethosomes, including vesicle size and shape, size distribution, and polydispersity index (PDI), EE, zeta potential, stability, loading capacity and vesicle fluidity, drug release, and drug content, which are useful for optimizing the ethosomal formulation. Each of these characterization methods is discussed in detail in other articles and is not listed here [61,84].
8. Different Applications of Ethosomes
Compared with ordinary liposomes, ethosomes have many advantages, such as large drug loading, high EE, small particle size, better flexibility, high transdermal efficiency, and large skin retention. The ethosomes as transdermal drug delivery systems have been used in dermatological drugs, non-steroidal anti-inflammatory drugs, antifungal and antiviral drugs, hormone drugs, and other drugs [22,85].
8.1. Delivery of Antifungal and Antibacterial Drug
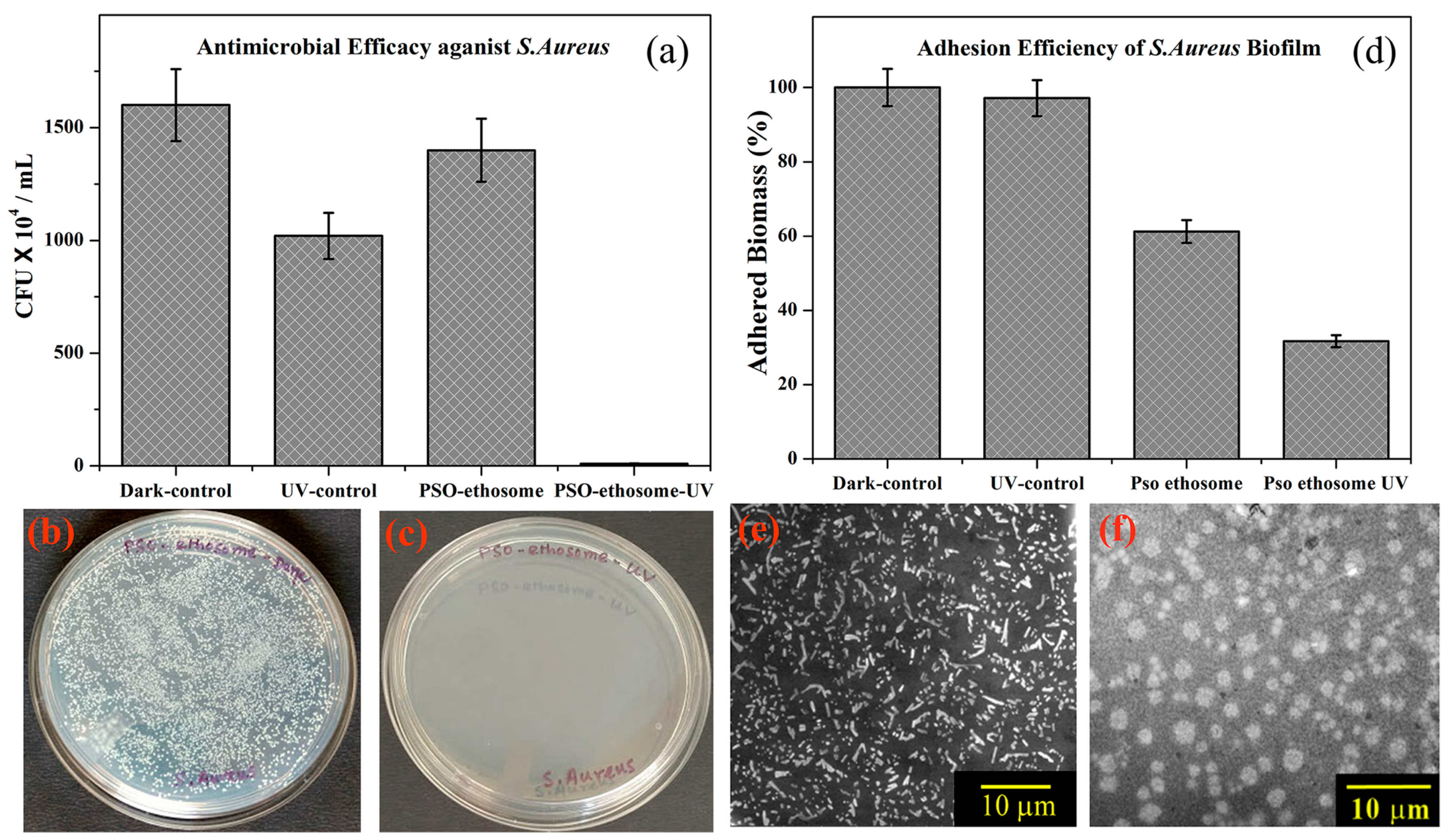
Numerous pathogenic microorganisms responsible for angioinvasive fungal infections are widely distributed in the environment and typically do not result in clinical infection among individuals with a fully functional immune system [86]. When the antifungal and antibacterial drugs are made into ethosomes, the increase in transdermal permeation can achieve the goal of maintaining a constant plasma drug concentration while reducing the number of administrations [41,65,77]. For example, Shetty S. et al. developed an ethosomal gel loaded with clove oil. The antifungal activity of the ethosomal gel against the fungus Candida albicans was found to be satisfactory in comparison to the pure clove oil [87]. Maheshwai et al. also showed that clotrimazole-loaded ethosomes have a stronger penetration enhancing effect and a higher inhibitory effect against Candida species than liposome formulations [88]. Bagchi et al. developed ethosomes loaded with psoralen. The resulting PSO–ethosomes demonstrated effective antimicrobial activity against Staphylococcus aureus and facilitated psoralen transport through the biological barrier; the results are shown in Figure 6 [89]. Zhu et al. also prepared hexyl aminolevulinate (HAL)-loaded ethosomes (HAL-ES). The application of antimicrobial photodynamic therapy (aPDT) using a 0.5% concentration of HAL resulted in a significant reduction in C. albicans biofilm activity by 69.71%. However, when aPDT was administered with 0.5% HAL-ES, an even greater reduction in biofilm activity was observed, reaching 92.95 ± 0.16%. Additionally, this treatment inhibited hyphal growth by 25.71 ± 1.61% within a 48 h timeframe. These findings were found to be associated with a 3-fold increase in C. albicans plasma membrane permeabilization [90]. In addition, there are many other drugs (terbinafine hydrochloride, vancomycin hydrochloride, griseofulvin, ketoconazole, luliconazole, eugenol, and silver sulfadiazine) that have been encapsulated in ethosomes to enhance antifungal and antibacterial activity (Table 1).
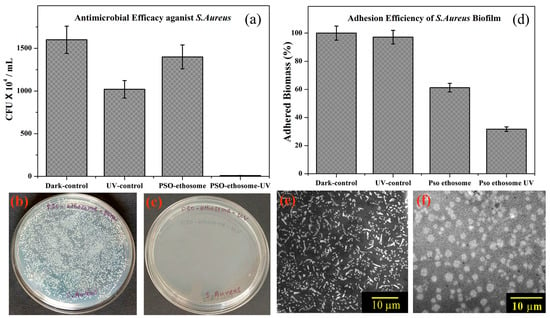
Figure 6.
(a) The antibacterial activity of PSO–ethosomes against S. aureus in the absence and presence of UVA. (b,c) Images of PSO–ethosome-treated S. aureus culture plates before and after UVA irradiation, respectively. (d) The adhesion efficiency of PSO–ethosome-treated S. aureus biofilms in the absence and presence UVA irradiation. SEM images of an S. aureus biofilm (e) without treatment and (f) treated with PSO–ethosomes followed by UVA illumination for 30 min. (Reproduced with permission from reference [86]. Copyright 2017, ACS Omega).

Table 1.
Examples of applications of ethosomes as transdermal delivery systems of antifungal and antibacterial drugs.
8.2. Delivery of Anticancer Drug
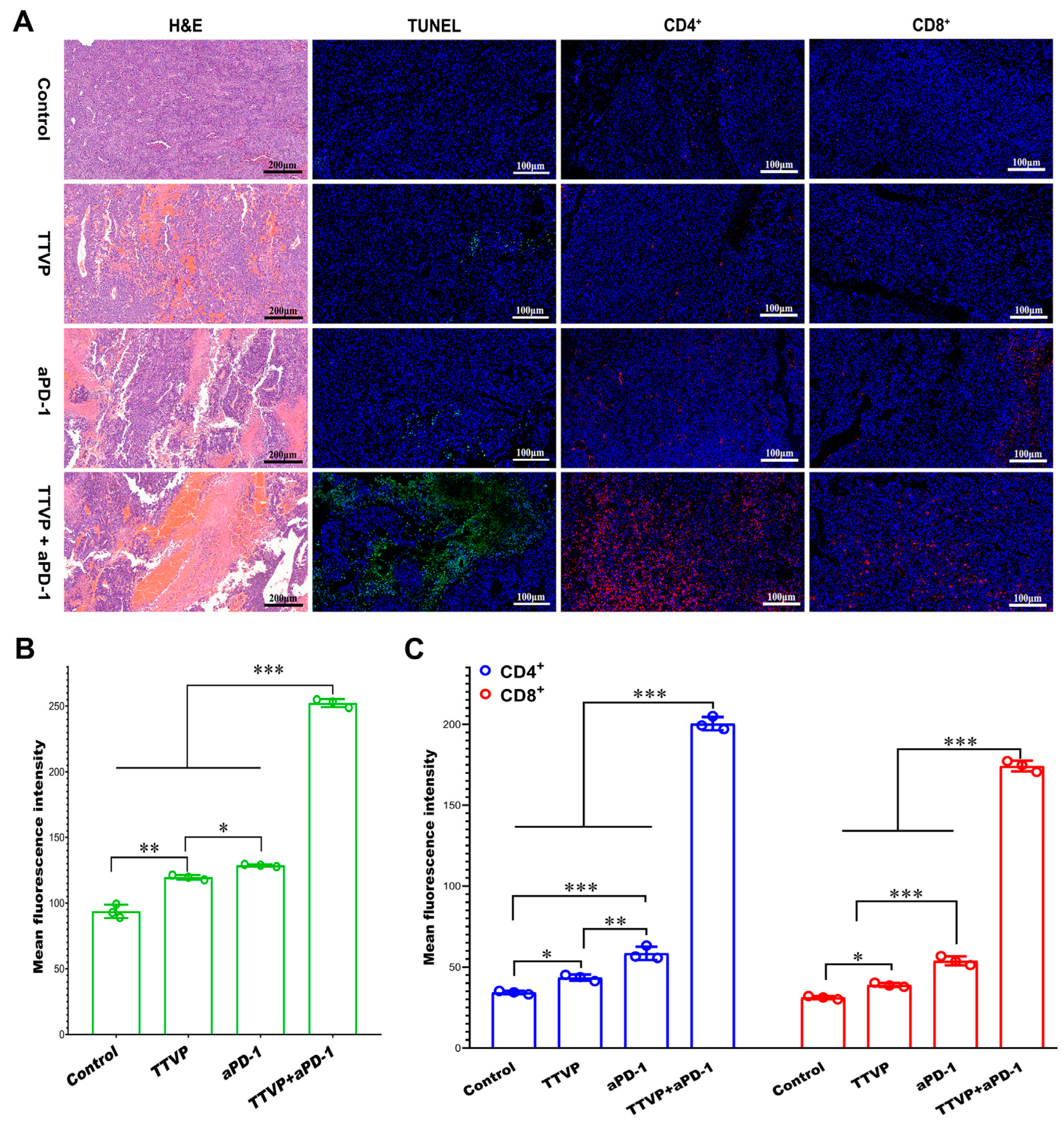
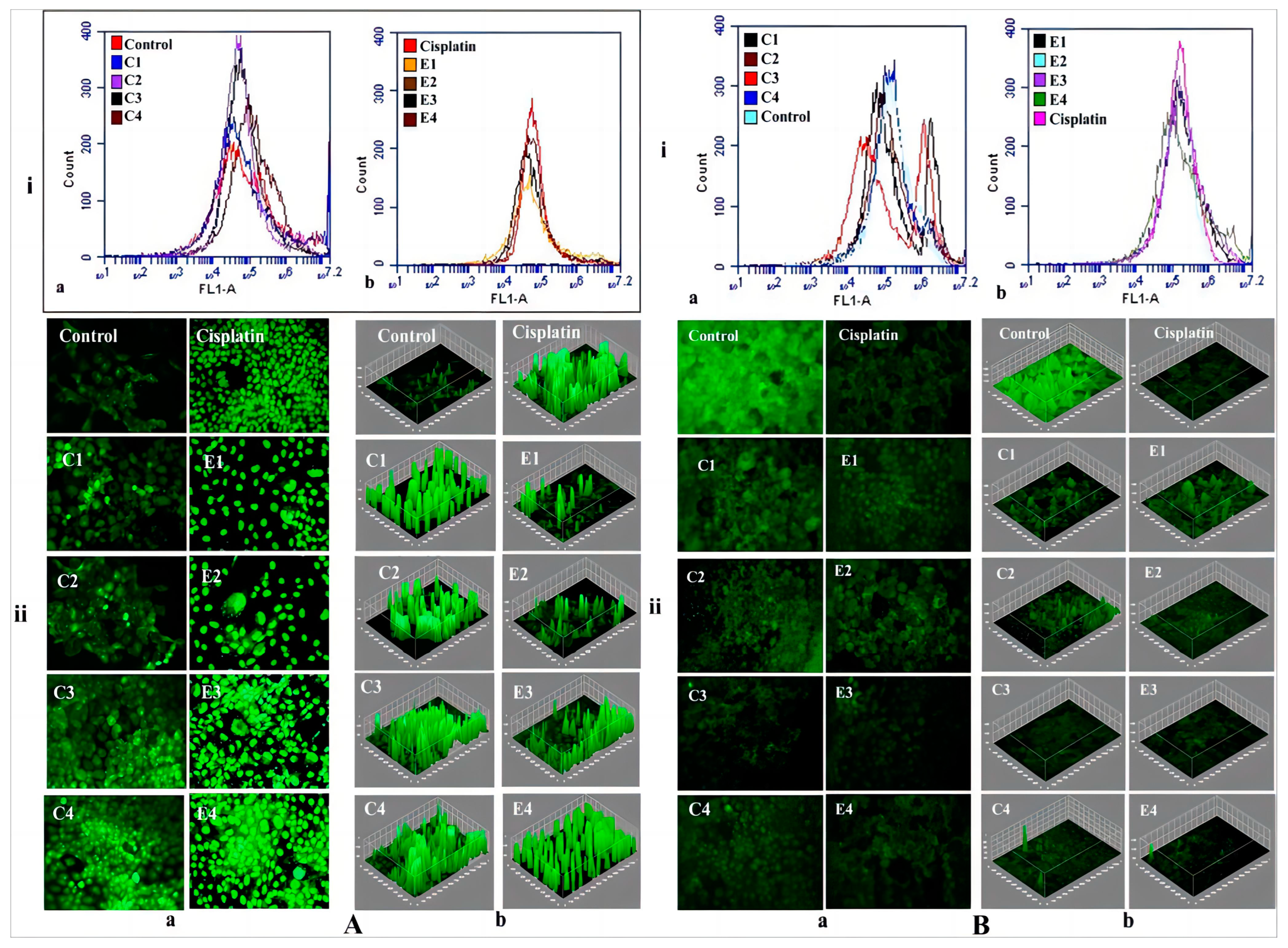
Mettlesome have emerged as a highly promising transdermal delivery technology for the treatment of skin cancer due to their non-invasive nature and low occurrence of notable adverse effects. The researchers Yu et al. formulated an ethosomal gel containing mitoxantrone (MTO) for the purpose of treating cancer. The ethosomes exhibited considerably more permeability in vitro across rat skin and demonstrated notable cytotoxicity. Furthermore, they displayed a heightened anti-melanoma impact in vivo compared to MTO solutions, achieved through the enhancement of calreticulin membrane translocation in B16 cells. F Amr Gamal et al. prepared a VMD-loaded ethosomal gel containing 65% isopropyl alcohol and 35% ethanol for the management of basal cell skin carcinoma [97]. The ethosomal gel that exhibited the highest level of effectiveness demonstrated a notable decrease in both the quantity and size of papillomas when compared to the oral VMD suspension and the ethosomal gel loaded with VMD but containing no isopropyl alcohol (IPA). Raj et al. prepared a nano-sized cytarabine-loaded ethosome to optimize the transdermal administration of drugs for the treatment of leukemia [98]. The formulation exhibited a reduced lag time and increased transdermal penetration of cytarabine within 3–12 h after injection through the skin, attributed to the flexibility of nanovesicles and the impact of ethanol on the tight junction of the lipid layer. Lin et al. formulated a co-loaded system consisting of berberine chloride and evodiamine ethosomes for the purpose of treating melanoma [99]. The results of the cell viability experiments indicated that the ethosomes that were optimized had an enhanced inhibitory effect on B16 melanoma cells. Moolakkadath et al. prepared a binary ethosomal gel formulation containing fisetin for the purpose of treating skin cancer through dermal application on mice exposed to ultraviolet radiation [100]. The results of the in vivo investigation demonstrated that mice who received pre-treatment with fisetin binary ethosomal gel had a notable reduction in the levels of TNF-α and IL-1α in comparison to the mice that were solely exposed to UV radiation. Furthermore, the group of mice treated with binary ethosomal gel had a lower percentage of tumor incidence (49%) compared to mice treated with UV only (96% tumor incidence). Cristiano et al. designed an ethosomal formulation including sulforaphane for the purpose of treating skin cancer [101]. The present study aimed to evaluate the anti-proliferative activity of ethosomes loaded with sulforaphane on SK-MEL 28 cells. The results demonstrated an enhanced anticancer effect when compared to the unencapsulated medication. El-Kayal et al. prepared (-)-epigallocatechin-3-gallate-loaded ethosomes and transethosomes for the treatment of skin cancer [102]. These two formulations showed an inhibitory effect on the epidermoid carcinoma cell line A431 and also reduced tumor size in mice. Bragagni et al. developed celecoxib-loaded transfersomes and ethosomes to improve anticancer efficacy against skin tumors [103]. These two formulations significantly improved the amount of drug penetrating the skin compared to an aqueous suspension, from 6.5- to 9.0-fold for transfersomes and ethosomes, respectively. Paolino et al. prepared a paclitaxel-loaded ethosome for the treatment of squamous cell carcinoma [104]. The results of the in vitro investigation demonstrated that the application of paclitaxel-loaded ethosomes onto the skin surface resulted in enhanced absorption of paclitaxel in a model representing the outermost layer of the skin (stratum corneum–epidermis membrane). Furthermore, this dermal application also led to greater anti-proliferative and anti-apoptotic effects of paclitaxel in DJM-1 cells as compared to the effects observed with the free form of the drug. In addition, the researchers created a new transcutaneous tumor vaccine patch (TTVP) using electrospun silk fibroin and polyvinyl alcohol nanofibrous patches. This patch included mannosylated polyethyleneimine-modified ethosome. The percutaneous injection of the Eth-PEIman chemical specifically targeted dendritic cells, inhibiting B16F10 tumor development. TTVP and aPD-1 also had a synergistic anti-melanoma effect. This effect is due to increased CD4+ and CD8+ T cell infiltration and IL-12 production in tumor tissue. This study’s findings are shown in Figure 7 [105]. Nayak et al. prepared a bioengineered ethosome with AgNPs and Tasar silk sericin proteins. This formulation was tested for treating non-melanoma skin cancer. Sericin helps wound healing by adding functional groups to AgNPs and chitosan. The main role of ethosomes is to transport encapsulated medicines. Silver nanoparticles (AgNPs) then rapidly produce reactive oxygen species (ROS), which disrupt cancer cell homeostatic equilibrium and degrade mitochondria and DNA machinery; the results are shown in Figure 8 [106]. The applications of ethosomes as transdermal delivery systems for anticancer drugs are shown in Table 2.
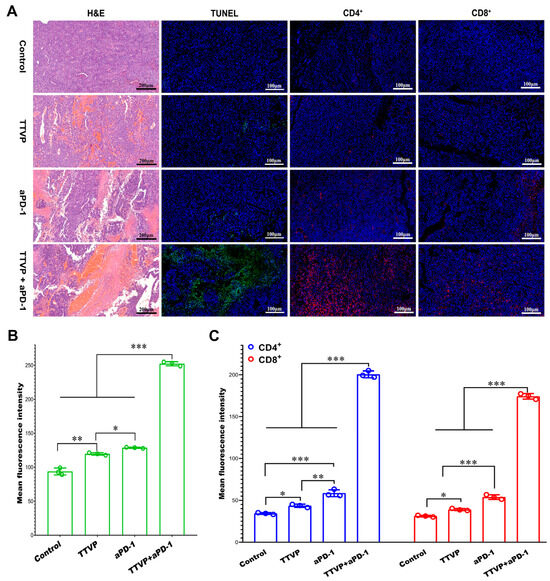
Figure 7.
Micrographs of tumor tissues stained by H&E, TUNEL, and antibodies of CD4/CD8. (A) Fluorescent intensity of (B) TUNEL and (C) CD4/CD8 staining (data are expressed as mean ± SD, n = 3; * p < 0.05, ** p < 0.01, *** p < 0.001). (Reproduced with permission from reference [105]. Copyright 2022, Acta Biomater).
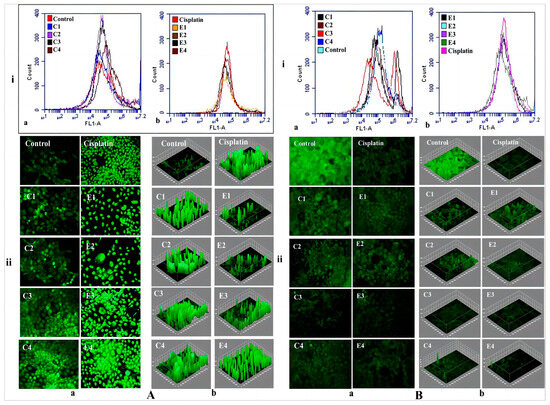
Figure 8.
(A) (i) The flow cytometric analysis of in vitro ROS productions in A431 cells after treatment with (a) chitosan formulations; (b) ethosomal formulations using DCFH-DA dye in F1 filter. (ii) Fluorescence microscopy in (a) raw fluorescence images; (b) ImageJ software analyzed images; (B) (i) Mitochondrial membrane potential (Ψm) studies observed through the FL-1 filter using Rh 123 dye in (a) chitosan formulations; (b) ethosomal formulations. (ii) Fluorescence microscopy images of using Rh 123 dye in (a) raw fluorescence images; (b) ImageJ software analyzed images. (Reproduced with permission from reference [106]. Copyright 2021, Int J Pharm).

Table 2.
Examples of applications of ethosomes as transdermal delivery systems of anticancer drugs.
8.3. Delivery of Anti-Psoriasis Drug
The chronic, non-infectious autoimmune skin disease psoriasis has red, scaly plaques. Ethosomes may improve the efficacy and reduce the side effects of psoriasis treatments. Fathalla et al. created liposomal and ethosomal Pluronic® F-127 gels with anthralin. These formulations were then tested for psoriasis efficacy and safety. The clinicaltrials.gov study ID was NCT03348462. Liposomes and ethosomes had average PASI changes of −68.66% and −81.84%, respectively, after treatment. Ethosomes were more effective than liposomes, according to these studies. No negative effects were found in either group. Anthralin ethosomes may treat psoriasis [64]. Negi et al. prepared an ethosomal hydrogel with thymoquinone as an anti-psoriatic agent in a mouse tail model. Compared to Nigella sativa extract, thymoquinone alone, and tazarotene gel, thymoquinone-loaded ethosomal gel had higher orthokeratosis and drug activity rates [110]. For simultaneous psoriasis treatment, Guo et al. prepared a unique drug delivery method. They created Cur@GA-TPGS-ES multifunctional ethosomes using curcumin (Cur) loaded with glycyrrhetinic acid-D-α-tocopherol acid-polyethylene glycol succinate (GA-TPGS). Cur@GA-TPGS-ES was effective when applied topically to imiquimod-induced psoriasis mice. Curcumin inhibited inflammatory infiltration, GA had glucocorticoid-like effects, and d-alpha-tocopheryl polyethylene glycol 1000 succinate had anti-lipid peroxidation capabilities [111]. The applications of ethosomes as transdermal delivery systems of anti-psoriasis drugs are shown in Table 3.

Table 3.
Examples of applications of ethosomes as transdermal delivery systems of anti-psoriasis drugs.
8.4. Delivery of Anti-Hypertrophic Scar (HS) Drug
A hypertrophic scar (HS), a common and unavoidable problem after cutaneous skin injury, has a much thicker fibrotic dermis than normal skin. HS management is clinically challenging. Ethosomal vesicles are a novel way to improve hypertrophic scar treatment.
Yu Z. et al. developed novel nano-photosensitizer IR-808-loaded nanoethosomes (IR-808-ES) to enhance synergistic transdermal photodynamic/photothermal therapy (PDT/PTT) for improving the therapeutic efficacy of an HS. The results showed that IR-808 hydroalcoholic solution (IR-808-HA) gel and IR-808-ES gel were uniformly distributed throughout the dermis in the IR-808-ES group after topical administration and accumulated in HS tissues. In the IR-808-HA group, IR-808 accumulated in the epidermis mainly because of its lipid solubility due to the disruption of lipid organization in the SC by ethanol. Twelve times more IR-808 accumulated in the dermis in the IR-808-ES group than in the IR-808-HA group, suggesting a significant transdermal drug delivery capacity of IR-808ES. Furthermore, in vivo delivery histology also observed that IR-808-ES crossed dense collagen fibers into the deep dermis, possibly due to the significant deformability and fluidity of the vesicle membrane and the intercellular pathway for transdermal permeation. The intact ES structure after transdermal permeation played a key role in ameliorating the PDT/PTT effect and inducing HSF apoptosis [112]. Mao et al. prepared 65 nm-diameter 5-fluorouracil (5-FU) ethosomes. Ethosomes with fluorescence penetration could penetrate the deep dermal layer of hypertrophic scars [113]. Zhang et al. demonstrated that 24 h 5-FU penetration into an HS and skin was as follows: ethosomes via HS (E-Scar) > hydroethanolic solution via an HS (H-Scar) > ethosomes via skin (E-Skin) > hydroethanolic solution via skin; ethosomes are very efficient vehicles for penetrating an HS [114]. Wo et al. also prepared ethosomal gels loaded with 5-FU (5-FU EGs). Using CLSM, rhodamine-6G-labeled ethosomal gels penetrated into the deep dermis of an HS within 24 h in the rabbit ear hypertrophic model. In addition, the Scar Elevation Index (SEI) value of the 5-FU EG group was lower than that of the 5-FU PBS gel; 5-FU EGs may be a suitable drug for hypertrophic scarring [115].
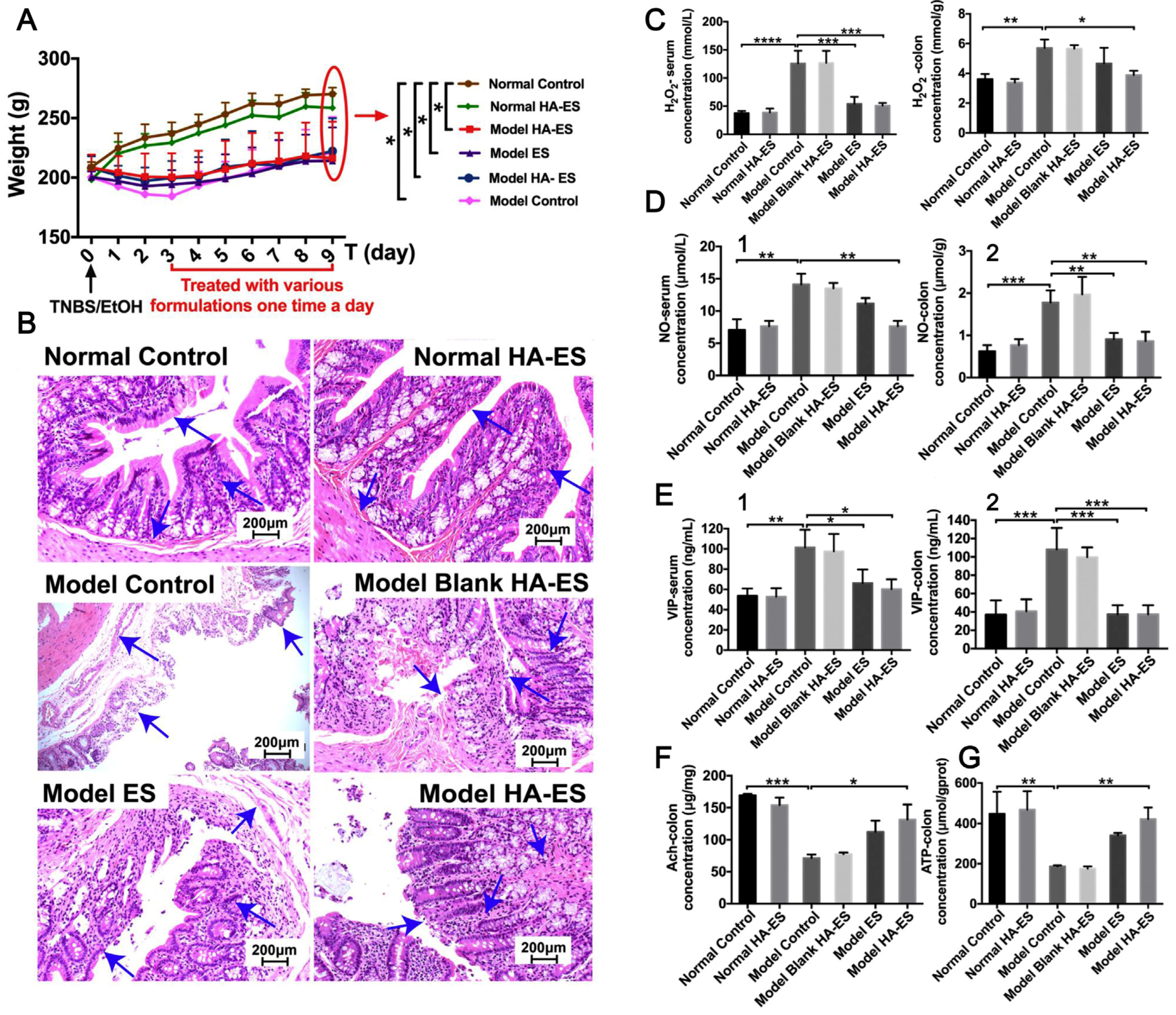
In hypertrophic scar treatment, a fractional CO2 laser may improve drug absorption. A CO2 fractional laser increases the stratum corneum permeability of 5-FU-encapsulated ethosomes according to a study. Permeability increased rapidly due to this improvement. The combined group showed significantly a decreased relative thickness and scar elevation index (SEI) of rabbit ear hypertrophic scars after 7 days of therapy compared to the group treated with 5-FU encapsulated ethosomes. Furthermore, the process by which HSs (hypertrophic scars) improved was associated with the decrease in collagen I/III levels and the suppression of TGF-β1 expression [116]. Xie et al. prepared a hyaluronic acid-containing ethosomal (HA-ES) formulation and used RB as a model medication. HA-ES-RB penetrated the deepest dermis layer better than ES-RB. HA-ES-RB’s transdermal drug delivery efficacy is due to its small size, hyaluronic acid’s hydration capabilities, and liposomal carriers’ ability to target the skin and appendages. The HA-ES delivery system was biocompatible because it did not affect normal cells [117]. The applications of ethosomes as transdermal delivery systems for anti-hypertrophic drugs are shown in Table 4.

Table 4.
Examples of applications of ethosomes as transdermal delivery systems of anti-hypertrophic scar drugs.
8.5. Delivering Drugs for Chronic Diseases
Chronic diseases are the primary contributor to global mortality, sometimes evading early detection and typically not being detected until severe stages [118]. The treatment and application of ethosomes in chronic diseases is beginning to attract the interest of researchers. Ammar et al. prepared ethosomes loaded with vardenafil hydrochloride. CLSM studies demonstrated their ability to deeply penetrate rat skin. In addition, Cmax was lower, Tmax was delayed, and AUC0–24h was greater in adults and geriatrics after the transdermal application of the ethosomal system [119]. Ibrahim et al. prepared a carvedilol-loaded ethosomal gel for the treatment of hypertension. Compared with pure carvedilol gel, the ethosomal gel showed a significant reduction in mean arterial pressure in rats (p < 0.01) at the second hour of the experiment (146.11 mmHg) and a further significant reduction at 6 h (p < 0.001) (98.88 mmHg) [56]. Liu et al. prepared a transdermal ligustrazine ethosomal patch for the treatment of myocardial ischemia. The drug patch can maintain a steady blood drug concentration, increase medication availability, and shorten drug administration. These compounds may reduce hemorheological parameters associated with myocardial ischemia in rats and protect acutely ischemic and ischemia-reperfusion-wounded myocardium [120]. Bhosale et al. prepared ethosomes filled with repaglinide (RPG) for diabetes treatment. When applied to excised rat skin, the ethosome penetrated 64% to 97% of the dose better than the free drug and its hydroalcoholic solution. Researchers found that RPG has a much longer anti-diabetic effect than oral doses [121]. Shi et al. prepared a transdermal ethosomal system containing ligustrazine phosphate (LP) as a potential therapeutic approach for the management of Alzheimer’s disease. The findings of the study indicated that the LP ethosomal system exhibited superior penetrating ability and drug deposition in the skin compared to the aqueous system. The researchers successfully reinstated the functioning of antioxidant enzymes and the levels of MDA in the brains of amnesic rats to a comparable state as that of normal rats [122]. Liu et al. prepared the pharmacokinetics of intragastric, transdermal, and standard transdermal ligustrazine patches. The ligustrazine ethosomal patch group had a 2.09-fold larger area under the curve (AUC) than the oral medication group and a 2.12-fold higher AUC than the traditional patch group. Compared to oral administration, ligustrazine ethosomes had a 209.45% relative bioavailability, while the standard transdermal patch had 98.63%. This study suggests that ligustrazine ethosomal patches may improve medicine absorption and bioavailability compared to the other two alternatives [123]. Bhosale et al. prepared ethosomal carrier-based transdermal VLT delivery. The study found that transdermal ethosomal VLT delivered better and longer-lasting antihypertensive benefits to Wistar rats than oral VLT suspension. This is because the VLT penetrates Wistar rats’ skin transdermally. The ethosome solubilized epidermal intercellular lipids to penetrate the skin but did not change its cellular architecture. In conclusion, this ethosomal formulation has improved transdermal VLT absorption, making it better than oral administration for antihypertensive therapy [124]. The applications of ethosomes as transdermal delivery systems of drugs for chronic diseases are shown in Table 5.

Table 5.
Examples of applications of ethosomes as a transdermal delivery system of chronic disease drugs.
8.6. Delivery of Other Drug from Ethosomal Systems
Ethosomes have also been used to treat other conditions such as acne, androgenic alopecia, malaria, ageing, fever and pain, menopausal syndromes, acne vulgaris, UV radiation, and so on (Table 6). Feng et al. designed a modified ethosomal system using hyaluronic acid (HA) to specifically target the CD44 protein, which is known to be upregulated in inflamed psoriatic skin. This approach aimed to enhance the transport of drugs to the intended site, resulting in increased curcumin accumulation within the inflamed skin [31]. Ahmed et al. prepared an ethosomal system to enhance the transdermal activity of tramadol to significantly prolong the analgesic effect [125]. Yongtai Zhang et al. prepared EUG/CAH-loaded HA-immobilized ethosomes (HA-ES) with the aim of possessing excellent deformability and showed improved efficacy against UC compared to ES; the results are shown in Figure 9 [126].

Table 6.
Examples of applications of ethosomes as transdermal delivery systems of other drugs.
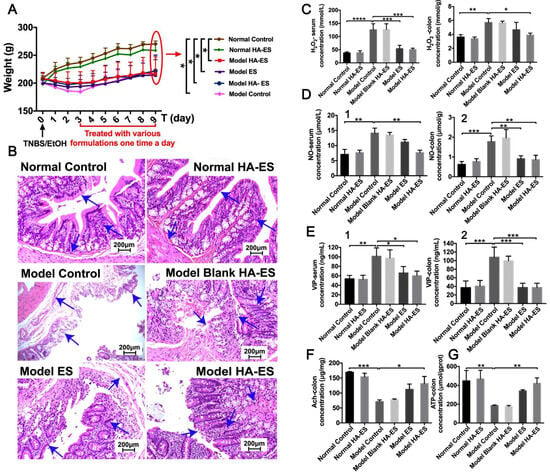
Figure 9.
Physiological and pathological changes in normal (Normal) and UC (Model) rats after treatment with blank HA-ES, EUG/CAH-loaded HA-ES (HA-ES1), and ES by transdermal administration on abdominal skin regions. (A) Body weight; (B) H&E-stained pathological colon slices (200× magnification; arrows refer to the layers of colon wall, including the mucosal layer, mucosal muscular layer, and submucosa); (C–G), respectively, refer to H2O2, NO, VIP, Ach, and ATP levels in serum (1) and/or colon tissues (2). The marks “*”, “**”, “***”, and “****”, respectively, refer to the statistical p-values 0.05, 0.01, 0.001, and 0.0001 (n = 8). (Reproduced with permission from reference [126]. Copyright 2020, Nanomedicine).
9. Discussion and Prospects
Ethosomes, a novel drug carrier with numerous benefits such as enhanced drug penetration into the skin, reduced dosage and side effects, and improved user-friendliness, have attracted significant attention in both pharmaceutical and cosmetic fields. This paper reviews the research progress on the types, mechanisms of action, preparation methods, influencing factors, and clinical applications of ethosomes, aiming to provide a reference for their development and application. Compared to traditional liposomes, ethosomes exhibit better encapsulation efficiency, exhibit higher skin penetration potential, and can overcome the challenge of low macromolecular drug transport efficiency. Despite rapid development in recent years, ethosomes still face several challenges. Ethosomes are less stable during long-term storage, affecting their drug content. The particle size of ethosomes, ranging from ten to several hundred nanometers, is a key factor affecting their skin penetration. Therefore, a suitable particle size is likely to improve transdermal efficiency and drug delivery. These factors will be the main challenges and opportunities for future research in this field. It is hoped that more researchers will conduct studies in this field, enabling ethosomes to realize their potential as a multifunctional material in material science and bioengineering. Moreover, ‘‘Chemistry’’ plays a crucial role in ethosomal technology. Continuous innovation can lead to more opportunities and breakthroughs in drug delivery. Therefore, it is worthwhile to discuss how chemistry will advance ethosomal technology and applications.
Author Contributions
Conceptualization, B.Z. and J.W.; methodology, X.F.; software, K.X.; validation, H.L.; writing–original draft preparation, B.Z.; writing–review and editing, Y.J.; visualization, B.Z.; supervision, Y.S.; project administration X.F.; funding acquisition, Y.J. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Shaanxi Administration of Traditional Chinese Medicine grant number [No. SZY-NLTL-2024-002], Shaanxi Province Drug Analysis and Drug Intelligence Monitoring Research Key Laboratory Open Subjects grant number [No. NNLS-O-202305] And The APC was funded by [No. SZY-NLTL-2024-002].
Data Availability Statement
Samples of the compounds are available from the authors.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Jafari, A.; Daneshamouz, S.; Ghasemiyeh, P.; Mohammadi-Samani, S. Ethosomes as dermal/transdermal drug delivery systems: Applications, preparation and characterization. J. Liposome Res. 2023, 33, 34–52. [Google Scholar] [CrossRef] [PubMed]
- Sguizzato, M.; Ferrara, F.; Hallan, S.S.; Baldisserotto, A.; Drechsler, M.; Malatesta, M.; Costanzo, M.; Cortesi, R.; Puglia, C.; Valacchi, G.; et al. Ethosomes and Transethosomes for Mangiferin Transdermal Delivery. Antioxidants 2021, 10, 768. [Google Scholar] [CrossRef] [PubMed]
- Nair, R.S.; Billa, N.; Leong, C.O.; Morris, A.P. An evaluation of tocotrienol ethosomes for transdermal delivery using Strat-M(®) membrane and excised human skin. Pharm. Dev. Technol. 2021, 26, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Garg, V.; Singh, H.; Bimbrawh, S.; Singh, S.K.; Gulati, M.; Vaidya, Y.; Kaur, P. Ethosomes and Transfersomes: Principles, Perspectives and Practices. Curr. Drug Deliv. 2017, 14, 613–633. [Google Scholar] [CrossRef]
- Mohammed, M.I.; Makky, A.M.; Teaima, M.H.; Abdellatif, M.M.; Hamzawy, M.A.; Khalil, M.A. Transdermal delivery of vancomycin hydrochloride using combination of nano-ethosomes and iontophoresis: In vitro and in vivo study. Drug Deliv. 2016, 23, 1558–1564. [Google Scholar] [PubMed]
- Huang, M.; Liu, J.; Fan, Y.; Sun, J.; Cheng, J.X.; Zhang, X.F.; Zhai, B.T.; Guo, D.Y. Development of curcumin-loaded galactosylated chitosan-coated nanoparticles for targeted delivery of hepatocellular carcinoma. Int. J. Biol. Macromol. 2023, 253, 127219. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Zhai, B.; Fan, Y.; Sun, J.; Cheng, J.; Zou, J.; Zhang, X.; Shi, Y.; Guo, D. Development of Paeonol Liposomes: Design, Optimization, in vitro and in vivo Evaluation. Int. J. Nanomed. 2022, 17, 5027–5046. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Lv, H.; Han, G.; Ma, K. Ethosomes Loaded with Cryptotanshinone for Acne Treatment through Topical Gel Formulation. PLoS ONE 2016, 11, e0159967. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhao, Y.; Zhai, B.; Cheng, J.; Sun, J.; Zhang, X.; Guo, D. Phloretin Transfersomes for Transdermal Delivery: Design, Optimization, and In Vivo Evaluation. Molecules 2023, 28, 6790. [Google Scholar] [CrossRef]
- Carita, A.C.; Eloy, J.O.; Chorilli, M.; Lee, R.J.; Leonardi, G.R. Recent Advances and Perspectives in Liposomes for Cutaneous Drug Delivery. Curr. Med. Chem. 2018, 25, 606–635. [Google Scholar] [CrossRef]
- Lu, J.; Guo, T.; Fan, Y.; Li, Z.; He, Z.; Yin, S.; Feng, N. Recent Developments in the Principles, Modification and Application Prospects of Functionalized Ethosomes for Topical Delivery. Curr. Drug Deliv. 2021, 18, 570–582. [Google Scholar] [CrossRef]
- Dumitriu Buzia, O.; Păduraru, A.M.; Stefan, C.S.; Dinu, M.; Cocoș, D.I.; Nwabudike, L.C.; Tatu, A.L. Strategies for Improving Transdermal Administration: New Approaches to Controlled Drug Release. Pharmaceutics 2023, 15, 1183. [Google Scholar] [CrossRef]
- Zhou, Y.; Wei, Y.H.; Zhang, G.Q.; Wu, X.A. Synergistic penetration of ethosomes and lipophilic prodrug on the transdermal delivery of acyclovir. Arch. Pharm. Res. 2010, 33, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Caberlotto, E.; Ruiz, L.; Miller, Z.; Poletti, M.; Tadlock, L. Effects of a skin-massaging device on the ex-vivo expression of human dermis proteins and in-vivo facial wrinkles. PLoS ONE 2017, 12, e0172624. [Google Scholar] [CrossRef]
- Sharon, A. Gynecare Morcellex Sigma(®): Manufacturer: ETHICON Women’s Health & Urology, A Division of ETHICON, INC., a Johnson & Johnson company, Somerville, NJ 08876-0151, USA, © ETHICON, INC. 2005. J. Obstet. Gynaecol. India 2014, 64, 226–227. [Google Scholar]
- Anderson, P. Assessment and development of executive function (EF) during childhood. Child Neuropsychol. 2002, 8, 71–82. [Google Scholar] [CrossRef]
- Zhang, J.P.; Wei, Y.H.; Zhou, Y.; Li, Y.Q.; Wu, X.A. Ethosomes, binary ethosomes and transfersomes of terbinafine hydrochloride: A comparative study. Arch. Pharm. Res. 2012, 35, 109–117. [Google Scholar] [CrossRef]
- Yucel, C.; Seker Karatoprak, G.; Degim, I.T. Anti-aging formulation of rosmarinic acid-loaded ethosomes and liposomes. J. Microencapsul. 2019, 36, 180–191. [Google Scholar] [CrossRef]
- Zhang, Y.T.; Shen, L.N.; Wu, Z.H.; Zhao, J.H.; Feng, N.P. Comparison of ethosomes and liposomes for skin delivery of psoralen for psoriasis therapy. Int. J. Pharm. 2014, 471, 449–452. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Wei, Y.; Liu, H.; Zhang, G.; Wu, X. Preparation and in vitro evaluation of ethosomal total alkaloids of Sophora alopecuroides loaded by a transmembrane pH-gradient method. AAPS PharmSciTech 2010, 11, 1350–1358. [Google Scholar] [CrossRef] [PubMed]
- Zeng, C.; Jiang, W.; Tan, M.; Yang, X.; He, C.; Huang, W.; Xing, J. Optimization of the process variables of tilianin-loaded composite phospholipid liposomes based on response surface-central composite design and pharmacokinetic study. Eur. J. Pharm. Sci. 2016, 85, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Elsayed, M.M.; Abdallah, O.Y.; Naggar, V.F.; Khalafallah, N.M. Deformable liposomes and ethosomes: Mechanism of enhanced skin delivery. Int. J. Pharm. 2006, 322, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, N.; Verma, A.; Pathak, K. Feasibility of binary composition in development of nanoethosomal glycolic vesicles of triamcinolone acetonide using Box-behnken design: In vitro and ex vivo characterization. Artif. Cells Nanomed. Biotechnol. 2017, 45, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Song, C.K.; Balakrishnan, P.; Shim, C.K.; Chung, S.J.; Chong, S.; Kim, D.D. A novel vesicular carrier, transethosome, for enhanced skin delivery of voriconazole: Characterization and in vitro/in vivo evaluation. Colloids Surf. B Biointerfaces 2012, 92, 299–304. [Google Scholar] [CrossRef]
- Ascenso, A.; Raposo, S.; Batista, C.; Cardoso, P.; Mendes, T.; Praca, F.G.; Bentley, M.V.; Simoes, S. Development, characterization, and skin delivery studies of related ultradeformable vesicles: Transfersomes, ethosomes, and transethosomes. Int. J. Nanomed. 2015, 10, 5837–5851. [Google Scholar] [CrossRef] [PubMed]
- Moolakkadath, T.; Aqil, M.; Ahad, A.; Imam, S.S.; Iqbal, B.; Sultana, Y.; Mujeeb, M.; Iqbal, Z. Development of transethosomes formulation for dermal fisetin delivery: Box-Behnken design, optimization, in vitro skin penetration, vesicles-skin interaction and dermatokinetic studies. Artif. Cells Nanomed. Biotechnol. 2018, 46, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Albash, R.; Abdelbary, A.A.; Refai, H.; El-Nabarawi, M.A. Use of transethosomes for enhancing the transdermal delivery of olmesartan medoxomil: In vitro, ex vivo, and in vivo evaluation. Int. J. Nanomed. 2019, 14, 1953–1968. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Wu, L.; Wu, D.; Shi, D.; Wang, T.; Zhu, X. Mechanism of transdermal permeation promotion of lipophilic drugs by ethosomes. Int. J. Nanomed. 2017, 12, 3357–3364. [Google Scholar] [CrossRef]
- Li, Y.; Xu, F.; Li, X.; Chen, S.Y.; Huang, L.Y.; Bian, Y.Y.; Wang, J.; Shu, Y.T.; Yan, G.J.; Dong, J.; et al. Development of curcumin-loaded composite phospholipid ethosomes for enhanced skin permeability and vesicle stability. Int. J. Pharm. 2021, 592, 119936. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Wang, X.; Wu, J.; Zhang, D.; Zhang, L.; Song, X.; Hong, H.; He, C.; Mo, X.; Wu, S.; et al. Polyethylenimine and sodium cholate-modified ethosomes complex as multidrug carrier for the treatment of melanoma through transdermal delivery. Nanomedicine 2019, 14, 2395–2408. [Google Scholar] [CrossRef]
- Zhang, Y.; Xia, Q.; Li, Y.; He, Z.; Li, Z.; Guo, T.; Wu, Z.; Feng, N. CD44 Assists the Topical Anti-Psoriatic Efficacy of Curcumin-Loaded Hyaluronan-Modified Ethosomes: A New Strategy for Clustering Drug in Inflammatory Skin. Theranostics 2019, 9, 48–64. [Google Scholar] [CrossRef] [PubMed]
- Mishra, D.; Mishra, P.K.; Dubey, V.; Nahar, M.; Dabadghao, S.; Jain, N.K. Systemic and mucosal immune response induced by transcutaneous immunization using Hepatitis B surface antigen-loaded modified liposomes. Eur. J. Pharm. Sci. 2008, 33, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Rattanapak, T.; Young, K.; Rades, T.; Hook, S. Comparative study of liposomes, transfersomes, ethosomes and cubosomes for transcutaneous immunisation: Characterisation and in vitro skin penetration. J. Pharm. Pharmacol. 2012, 64, 1560–1569. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Huang, J.; Zhang, Y.; Cao, J.; Wang, X.; Chu, X. Study on the transdermal penetration mechanism of ibuprofen nanoemulsions. Drug Dev. Ind. Pharm. 2019, 45, 465–473. [Google Scholar]
- Champeau, M.; Vignoud, S.; Mortier, L.; Mordon, S. Photodynamic therapy for skin cancer: How to enhance drug penetration? J. Photochem. Photobiol. B Biol. 2019, 197, 111544. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Shi, Y.; Le, G. [Antibacterial activity and mechanisms of a new peptide derived from cell-penetrating peptide]. Wei Sheng Wu Xue Bao = Acta Microbiol. Sin. 2013, 53, 950–956. [Google Scholar] [PubMed]
- Jia, Z.; Wu, A.; Tam, M.; Spain, J.; McKinney, J.M.; Wang, W. Caval Penetration by Inferior Vena Cava Filters: A Systematic Literature Review of Clinical Significance and Management. Circulation 2015, 132, 944–952. [Google Scholar] [CrossRef]
- Niu, X.Q.; Zhang, D.P.; Bian, Q.; Feng, X.F.; Li, H.; Rao, Y.F.; Shen, Y.M.; Geng, F.N.; Yuan, A.R.; Ying, X.Y.; et al. Mechanism investigation of ethosomes transdermal permeation. Int. J. Pharm. X 2019, 1, 100027. [Google Scholar] [CrossRef]
- Shen, S.; Liu, S.Z.; Zhang, Y.S.; Du, M.B.; Liang, A.H.; Song, L.H.; Ye, Z.G. Compound antimalarial ethosomal cataplasm: Preparation, evaluation, and mechanism of penetration enhancement. Int. J. Nanomed. 2015, 10, 4239–4253. [Google Scholar] [CrossRef]
- Mbah, C.C.; Builders, P.F.; Attama, A.A. Nanovesicular carriers as alternative drug delivery systems: Ethosomes in focus. Expert Opin. Drug Deliv. 2014, 11, 45–59. [Google Scholar] [CrossRef]
- Sala, M.; Diab, R.; Elaissari, A.; Fessi, H. Lipid nanocarriers as skin drug delivery systems: Properties, mechanisms of skin interactions and medical applications. Int. J. Pharm. 2018, 535, 1–17. [Google Scholar] [CrossRef]
- Binder, L.; Kulovits, E.M.; Petz, R.; Ruthofer, J.; Baurecht, D.; Klang, V.; Valenta, C. Penetration monitoring of drugs and additives by ATR-FTIR spectroscopy/tape stripping and confocal Raman spectroscopy—A comparative study. Eur. J. Pharm. Biopharm. 2018, 130, 214–223. [Google Scholar] [CrossRef]
- Amin, S.; Kohli, K.; Khar, R.K.; Mir, S.R.; Pillai, K.K. Mechanism of in vitro percutaneous absorption enhancement of carvedilol by penetration enhancers. Pharm. Dev. Technol. 2008, 13, 533–539. [Google Scholar] [CrossRef]
- Mollica, F.; Biondi, M.; Muzzi, S.; Ungaro, F.; Quaglia, F.; La Rotonda, M.I.; Netti, P.A. Mathematical modelling of the evolution of protein distribution within single PLGA microspheres: Prediction of local concentration profiles and release kinetics. J. Mater. Sci. Mater. Med. 2008, 19, 1587–1593. [Google Scholar] [CrossRef] [PubMed]
- Cevc, G.; Vierl, U. Nanotechnology and the transdermal route: A state of the art review and critical appraisal. J. Control. Release 2010, 141, 277–299. [Google Scholar] [CrossRef] [PubMed]
- Sekine, A.; Ogura, T.; Tsuchiya, K.; Ohishi, K.; Masubuchi, Y.; Akamatsu, M.; Sakai, K.; Abe, M.; Sakai, H. Effect of polyols on membrane structures of liposomes: A study using small-angle X-ray scattering data and generalized indirect Fourier transformation. Chem. Phys. Lipids 2022, 249, 105253. [Google Scholar] [CrossRef]
- Ji, H.; Zhao, W.; Yu, Z. Interaction mechanism of three egg protein derived ACE inhibitory tri-peptides and DPPC membrane using FS, FTIR, and DSC studies. Food Chem. X 2022, 15, 100366. [Google Scholar] [CrossRef]
- Xiaowei, J.; Lijuan, Y.; Yanling, L.; Qiuxiao, L.; Bohong, G. Design and Development of Functionalized Single-walled Carbon Nanotube-ethosomes for Transdermal Delivery of Ketoprofen. Die Pharm. 2023, 78, 31–36. [Google Scholar]
- Xie, W.J.; Zhang, Y.P.; Xu, J.; Sun, X.B.; Yang, F.F. The Effect and Mechanism of Transdermal Penetration Enhancement of Fu’s Cupping Therapy: New Physical Penetration Technology for Transdermal Administration with Traditional Chinese Medicine (TCM) Characteristics. Molecules 2017, 22, 525. [Google Scholar] [CrossRef]
- Elamin, A.; Enomoto, H.; Watanabe, M.; Sakuda, S. The Mechanism of Ochratoxin Contamination of Artificially Inoculated Licorice Roots. Toxins 2023, 15, 219. [Google Scholar] [CrossRef] [PubMed]
- Kaliyavaradhan, S.K.; Ling, T.C.; Guo, M.Z.; Mo, K.H. Waste resources recycling in controlled low-strength material (CLSM): A critical review on plastic properties. J. Environ. Manag. 2019, 241, 383–396. [Google Scholar] [CrossRef] [PubMed]
- Gleichgerrcht, E.; Fridriksson, J.; Rorden, C.; Bonilha, L. Connectome-based lesion-symptom mapping (CLSM): A novel approach to map neurological function. NeuroImage Clin. 2017, 16, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Alim, S.H.; Kassem, A.A.; Basha, M.; Salama, A. Comparative study of liposomes, ethosomes and transfersomes as carriers for enhancing the transdermal delivery of diflunisal: In vitro and in vivo evaluation. Int. J. Pharm. 2019, 563, 293–303. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.P.; Huang, Y.B.; Wu, P.C.; Tsai, Y.H. Topical delivery of 5-aminolevulinic acid-encapsulated ethosomes in a hyperproliferative skin animal model using the CLSM technique to evaluate the penetration behavior. Eur. J. Pharm. Biopharm. 2009, 73, 391–398. [Google Scholar] [CrossRef]
- Wang, S.; Li, W.; Sun, K.; Zhang, R.; Wang, S.; Geng, L. Study of release kinetics and degradation thermodynamics of ferric citrate liposomes. Chem. Phys. Lipids 2019, 225, 104811. [Google Scholar] [CrossRef]
- Ibrahim, T.M.; Abdallah, M.H.; El-Megrab, N.A.; El-Nahas, H.M. Transdermal ethosomal gel nanocarriers; a promising strategy for enhancement of anti-hypertensive effect of carvedilol. J. Liposome Res. 2019, 29, 215–228. [Google Scholar] [CrossRef]
- Yu, X.; Du, L.; Li, Y.; Fu, G.; Jin, Y. Improved anti-melanoma effect of a transdermal mitoxantrone ethosome gel. Biomed. Pharmacother. 2015, 73, 6–11. [Google Scholar] [CrossRef]
- Maestrelli, F.; Capasso, G.; Gonzalez-Rodriguez, M.L.; Rabasco, A.M.; Ghelardini, C.; Mura, P. Effect of preparation technique on the properties and in vivo efficacy of benzocaine-loaded ethosomes. J. Liposome Res. 2009, 19, 253–260. [Google Scholar] [CrossRef]
- Chen, Y.; Wiu, Q.; Zhang, Z.; Zhou, L.; Liu, X.; Du, M.; Jia, X. [Preparation and transdermal diffusion of ursolic acid ethosomes]. Zhongguo Zhong Yao Za Zhi = Zhongguo Zhongyao Zazhi = China J. Chin. Mater. Medica 2011, 36, 988–991. [Google Scholar]
- Ma, H.; Guo, D.; Fan, Y.; Wang, J.; Cheng, J.; Zhang, X. Paeonol-Loaded Ethosomes as Transdermal Delivery Carriers: Design, Preparation and Evaluation. Molecules 2018, 23, 1756. [Google Scholar] [CrossRef] [PubMed]
- Opatha, S.A.T.; Titapiwatanakun, V.; Chutoprapat, R. Transfersomes: A Promising Nanoencapsulation Technique for Transdermal Drug Delivery. Pharmaceutics 2020, 12, 855. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ou, R.; Guan, S.; Ye, X.; Hu, B.; Zhang, Y.; Lu, S.; Zhou, Y.; Yuan, Z.; Zhang, J.; et al. A novel drug delivery gel of terbinafine hydrochloride with high penetration for external use. Drug Deliv. 2015, 22, 1086–1093. [Google Scholar] [CrossRef] [PubMed]
- Balata, G.F.; Faisal, M.M.; Elghamry, H.A.; Sabry, S.A. Preparation and Characterization of Ivabradine HCl Transfersomes for Enhanced Transdermal Delivery. J. Drug Deliv. Sci. Technol. 2020, 60, 101921. [Google Scholar] [CrossRef]
- Fathalla, D.; Youssef, E.M.K.; Soliman, G.M. Liposomal and Ethosomal Gels for the Topical Delivery of Anthralin: Preparation, Comparative Evaluation and Clinical Assessment in Psoriatic Patients. Pharmaceutics 2020, 12, 446. [Google Scholar] [CrossRef]
- Pilch, E.; Musial, W. Liposomes with an Ethanol Fraction as an Application for Drug Delivery. Int. J. Mol. Sci. 2018, 19, 3806. [Google Scholar] [CrossRef]
- Fan, C.; Li, X.; Zhou, Y.; Zhao, Y.; Ma, S.; Li, W.; Liu, Y.; Li, G. Enhanced topical delivery of tetrandrine by ethosomes for treatment of arthritis. BioMed Res. Int. 2013, 2013, 161943. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Chen, Y.; Xu, H.; Wo, Y.; Zhang, Z.; Liu, Y.; Su, W.; Cui, D.; Zhang, Y. 5-Aminolevulinic acid loaded ethosomal vesicles with high entrapment efficiency for in vitro topical transdermal delivery and photodynamic therapy of hypertrophic scars. Nanoscale 2016, 8, 19270–19279. [Google Scholar] [CrossRef]
- Levy, E.S.; Yu, J.; Estevez, A.; Mao, J.; Liu, L.; Torres, E.; Leung, D.; Yen, C.W. A Systematic Approach for Liposome and Lipodisk Preclinical Formulation Development by Microfluidic Technology. AAPS J. 2021, 23, 111. [Google Scholar] [CrossRef]
- Al-Amin, M.D.; Bellato, F.; Mastrotto, F.; Garofalo, M.; Malfanti, A.; Salmaso, S.; Caliceti, P. Dexamethasone Loaded Liposomes by Thin-Film Hydration and Microfluidic Procedures: Formulation Challenges. Int. J. Mol. Sci. 2020, 21, 1611. [Google Scholar] [CrossRef]
- Wang, L.; Wang, J. Self-assembly of colloids based on microfluidics. Nanoscale 2019, 11, 16708–16722. [Google Scholar] [CrossRef]
- Ainbinder, D.; Paolino, D.; Fresta, M.; Touitou, E. Drug delivery applications with ethosomes. J. Biomed. Nanotechnol. 2010, 6, 558–568. [Google Scholar] [CrossRef]
- Abdulbaqi, I.M.; Darwis, Y.; Khan, N.A.; Assi, R.A.; Khan, A.A. Ethosomal nanocarriers: The impact of constituents and formulation techniques on ethosomal properties, in vivo studies, and clinical trials. Int. J. Nanomed. 2016, 11, 2279–2304. [Google Scholar] [CrossRef]
- Dayan, N.; Touitou, E. Carriers for skin delivery of trihexyphenidyl HCl: Ethosomes vs. liposomes. Biomaterials 2000, 21, 1879–1885. [Google Scholar] [CrossRef]
- Majeed, I.; Raza, S.A.; Akhtar, N.; Siddiqui, F.A.; Iqbal, B. Formulation and in-vitro characterization of Capsaicin loaded ethosomes. Pak. J. Pharm. Sci. 2019, 32, 2849–2857. [Google Scholar]
- Mishra, A.D.; Patel, C.N.; Shah, D.R. Formulation and optimization of ethosomes for transdermal delivery of ropinirole hydrochloride. Curr. Drug Deliv. 2013, 10, 500–516. [Google Scholar] [CrossRef] [PubMed]
- El-Shenawy, A.A.; Mahmoud, R.A.; Mahmoud, E.A.; Mohamed, M.S. Intranasal In Situ Gel of Apixaban-Loaded Nanoethosomes: Preparation, Optimization, and In Vivo Evaluation. AAPS PharmSciTech 2021, 22, 147. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, B.; Gupta, R.; Singh, S.K.; Gulati, M.; Singh, S. Prodrugs, phospholipids and vesicular delivery—An effective triumvirate of pharmacosomes. Adv. Colloid Interface Sci. 2018, 253, 35–65. [Google Scholar] [CrossRef] [PubMed]
- Kawar, D.; Abdelkader, H. Hyaluronic acid gel-core liposomes (hyaluosomes) enhance skin permeation of ketoprofen. Pharm. Dev. Technol. 2019, 24, 947–953. [Google Scholar] [CrossRef]
- López-Pinto, J.M.; González-Rodríguez, M.L.; Rabasco, A.M. Effect of cholesterol and ethanol on dermal delivery from DPPC liposomes. Int. J. Pharm. 2005, 298, 1–12. [Google Scholar] [CrossRef]
- Li, G.; Fan, Y.; Fan, C.; Li, X.; Wang, X.; Li, M.; Liu, Y. Tacrolimus-loaded ethosomes: Physicochemical characterization and in vivo evaluation. Eur. J. Pharm. Biopharm. 2012, 82, 49–57. [Google Scholar] [CrossRef]
- El Zaafarany, G.M.; Awad, G.A.; Holayel, S.M.; Mortada, N.D. Role of edge activators and surface charge in developing ultradeformable vesicles with enhanced skin delivery. Int. J. Pharm. 2010, 397, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Natsheh, H.; Touitou, E. Phospholipid Vesicles for Dermal/Transdermal and Nasal Administration of Active Molecules: The Effect of Surfactants and Alcohols on the Fluidity of Their Lipid Bilayers and Penetration Enhancement Properties. Molecules 2020, 25, 2959. [Google Scholar] [CrossRef] [PubMed]
- Ghanbarzadeh, S.; Arami, S. Enhanced transdermal delivery of diclofenac sodium via conventional liposomes, ethosomes, and transfersomes. BioMed Res. Int. 2013, 2013, 616810. [Google Scholar] [CrossRef] [PubMed]
- Zhai, B.; Wu, Q.; Wang, W.; Zhang, M.; Han, X.; Li, Q.; Chen, P.; Chen, X.; Huang, X.; Li, G.; et al. Preparation, characterization, pharmacokinetics and anticancer effects of PEGylated beta-elemene liposomes. Cancer Biol. Med. 2020, 17, 60–75. [Google Scholar] [CrossRef] [PubMed]
- Pandey, V.; Golhani, D.; Shukla, R. Ethosomes: Versatile vesicular carriers for efficient transdermal delivery of therapeutic agents. Drug Deliv. 2015, 22, 988–1002. [Google Scholar] [CrossRef]
- Shields, B.E.; Rosenbach, M.; Brown-Joel, Z.; Berger, A.P.; Ford, B.A.; Wanat, K.A. Angioinvasive fungal infections impacting the skin: Background, epidemiology, and clinical presentation. J. Am. Acad. Dermatol. 2019, 80, 869–880 e865. [Google Scholar] [CrossRef] [PubMed]
- Shetty, S.; Jose, J.; Kumar, L.; Charyulu, R.N. Novel ethosomal gel of clove oil for the treatment of cutaneous candidiasis. J. Cosmet. Dermatol. 2019, 18, 862–869. [Google Scholar] [CrossRef]
- Maheshwari, R.G.; Tekade, R.K.; Sharma, P.A.; Darwhekar, G.; Tyagi, A.; Patel, R.P.; Jain, D.K. Ethosomes and ultradeformable liposomes for transdermal delivery of clotrimazole: A comparative assessment. Saudi Pharm. J. 2012, 20, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Bagchi, D.; Dutta, S.; Singh, P.; Chaudhuri, S.; Pal, S.K. Essential Dynamics of an Effective Phototherapeutic Drug in a Nanoscopic Delivery Vehicle: Psoralen in Ethosomes for Biofilm Treatment. ACS Omega 2017, 2, 1850–1857. [Google Scholar] [CrossRef]
- Wang, Y.; Song, J.; Zhang, F.; Zeng, K.; Zhu, X. Antifungal Photodynamic Activity of Hexyl-Aminolevulinate Ethosomes Against Candida albicans Biofilm. Front. Microbiol. 2020, 11, 2052. [Google Scholar] [CrossRef]
- Marto, J.; Vitor, C.; Guerreiro, A.; Severino, C.; Eleuterio, C.; Ascenso, A.; Simoes, S. Ethosomes for enhanced skin delivery of griseofulvin. Colloids Surf. B Biointerfaces 2016, 146, 616–623. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.; Wang, J.; Ma, M.; Tan, F.; Li, N. Skin targeted lipid vesicles as novel nano-carrier of ketoconazole: Characterization, in vitro and in vivo evaluation. J. Mater. Sci. Mater. Med. 2015, 26, 175. [Google Scholar] [CrossRef]
- Dave, V.; Bhardwaj, N.; Gupta, N.; Tak, K. Herbal ethosomal gel containing luliconazole for productive relevance in the field of biomedicine. 3 Biotech 2020, 10, 97. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, N.; Pathak, K. Cavamax W7 composite ethosomal gel of clotrimazole for improved topical delivery: Development and comparison with ethosomal gel. AAPS PharmSciTech 2012, 13, 344–355. [Google Scholar] [CrossRef] [PubMed]
- Jin, P.; Yao, R.; Qin, D.; Chen, Q.; Du, Q. Enhancement in Antibacterial Activities of Eugenol-Entrapped Ethosome Nanoparticles via Strengthening Its Permeability and Sustained Release. J. Agric. Food Chem. 2019, 67, 1371–1380. [Google Scholar] [CrossRef] [PubMed]
- Razavi, S.; Partoazar, A.; Takzaree, N.; Fasihi-Ramandi, M.; Bahador, A.; Darvishi, M.H. Silver sulfadiazine nanoethogel for burn healing: Characterization and investigation of its in vivo effects. Nanomedicine 2018, 13, 1319–1331. [Google Scholar] [CrossRef] [PubMed]
- Gamal, F.A.; Sayed, O.M.; Abo El-Ela, F.I.; Kharshoum, R.M.; Salem, H.F. Treatment of Basal Cell Carcinoma Via Binary Ethosomes of Vismodegib: In Vitro and In Vivo Studies. AAPS PharmSciTech 2020, 21, 51. [Google Scholar]
- Raj, R.; Raj, P.M.; Ram, A. Lipid based noninvasive vesicular formulation of cytarabine: Nanodeformable liposomes. Eur. J. Pharm. Sci. 2016, 88, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Lin, L.; Choi, Y.; Michniak-Kohn, B. Development and in-vitro evaluation of co-loaded berberine chloride and evodiamine ethosomes for treatment of melanoma. Int. J. Pharm. 2020, 581, 119278. [Google Scholar] [CrossRef]
- Moolakkadath, T.; Aqil, M.; Ahad, A.; Imam, S.S.; Praveen, A.; Sultana, Y.; Mujeeb, M. Preparation and optimization of fisetin loaded glycerol based soft nanovesicles by Box-Behnken design. Int. J. Pharm. 2020, 578, 119125. [Google Scholar] [CrossRef]
- Cristiano, M.C.; Froiio, F.; Spaccapelo, R.; Mancuso, A.; Nisticò, S.P.; Udongo, B.P.; Fresta, M.; Paolino, D. Sulforaphane-Loaded Ultradeformable Vesicles as A Potential Natural Nanomedicine for the Treatment of Skin Cancer Diseases. Pharmaceutics 2019, 12, 6. [Google Scholar] [CrossRef]
- El Kayal, M.; Nasr, M.; Mortada, N.; Elkheshen, S. Optimization of the colloidal properties of different vesicular systems aiming to encapsulate (-)-epigallocatechin-3-gallate. Farmacia 2020, 68, 97–110. [Google Scholar] [CrossRef]
- Bragagni, M.; Mennini, N.; Maestrelli, F.; Cirri, M.; Mura, P. Comparative study of liposomes, transfersomes and ethosomes as carriers for improving topical delivery of celecoxib. Drug Deliv. 2012, 19, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Paolino, D.; Celia, C.; Trapasso, E.; Cilurzo, F.; Fresta, M. Paclitaxel-loaded ethosomes®: Potential treatment of squamous cell carcinoma, a malignant transformation of actinic keratoses. Eur. J. Pharm. Biopharm. 2012, 81, 102–112. [Google Scholar] [CrossRef]
- Song, X.; Jiang, Y.; Zhang, W.; Elfawal, G.; Wang, K.; Jiang, D.; Hong, H.; Wu, J.; He, C.; Mo, X.; et al. Transcutaneous tumor vaccination combined with anti-programmed death-1 monoclonal antibody treatment produces a synergistic antitumor effect. Acta Biomater. 2022, 140, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Nayak, D.; Thathapudi, N.C.; Ashe, S.; Nayak, B. Bioengineered ethosomes encapsulating AgNPs and Tasar silk sericin proteins for non melanoma skin carcinoma (NMSC) as an alternative therapeutics. Int. J. Pharm. 2021, 596, 120265. [Google Scholar] [CrossRef]
- Raj, R.; Raj, P.M.; Ram, A. Nanosized ethanol based malleable liposomes of cytarabine to accentuate transdermal delivery: Formulation optimization, in vitro skin permeation and in vivo bioavailability. Artif. Cells Nanomed. Biotechnol. 2018, 46, 951–963. [Google Scholar] [CrossRef]
- Moolakkadath, T.; Aqil, M.; Ahad, A.; Imam, S.S.; Praveen, A.; Sultana, Y.; Mujeeb, M.; Iqbal, Z. Fisetin loaded binary ethosomes for management of skin cancer by dermal application on UV exposed mice. Int. J. Pharm. 2019, 560, 78–91. [Google Scholar] [CrossRef]
- El-Kayal, M.; Nasr, M.; Elkheshen, S.; Mortada, N. Colloidal (-)-epigallocatechin-3-gallate vesicular systems for prevention and treatment of skin cancer: A comprehensive experimental study with preclinical investigation. Eur. J. Pharm. Sci. 2019, 137, 104972. [Google Scholar] [CrossRef]
- Negi, P.; Sharma, I.; Hemrajani, C.; Rathore, C.; Bisht, A.; Raza, K.; Katare, O.P. Thymoquinone-loaded lipid vesicles: A promising nanomedicine for psoriasis. BMC Complement. Altern. Med. 2019, 19, 334. [Google Scholar] [CrossRef]
- Guo, T.; Lu, J.; Fan, Y.; Zhang, Y.; Yin, S.; Sha, X.; Feng, N. TPGS assists the percutaneous administration of curcumin and glycyrrhetinic acid coloaded functionalized ethosomes for the synergistic treatment of psoriasis. Int. J. Pharm. 2021, 604, 120762. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Meng, X.; Zhang, S.; Wang, X.; Chen, Y.; Min, P.; Zhang, Z.; Zhang, Y. IR-808 loaded nanoethosomes for aggregation-enhanced synergistic transdermal photodynamic/photothermal treatment of hypertrophic scars. Biomater. Sci. 2021, 10, 158–166. [Google Scholar] [CrossRef]
- Gu, J.; Mao, X.; Li, C.; Ao, H.; Yang, X. A Novel Therapy for Laryngotracheal Stenosis: Treatment With Ethosomes Containing 5-Fluorouracil. Ann. Otol. Rhinol. Laryngol. 2015, 124, 561–566. [Google Scholar] [CrossRef]
- Zhang, Z.; Wo, Y.; Zhang, Y.; Wang, D.; He, R.; Chen, H.; Cui, D. In vitro study of ethosome penetration in human skin and hypertrophic scar tissue. Nanomedicine 2012, 8, 1026–1033. [Google Scholar] [CrossRef] [PubMed]
- Wo, Y.; Zhang, Z.; Zhang, Y.; Zhang, Z.; Wang, K.; Mao, X.; Su, W.; Li, K.; Cui, D.; Chen, J. Enhanced in vivo delivery of 5-fluorouracil by ethosomal gels in rabbit ear hypertrophic scar model. Int. J. Mol. Sci. 2014, 15, 22786–22800. [Google Scholar] [CrossRef]
- Huang, J.; Chen, J.; Wo, Y.; Wang, X.; Zhang, Y.; Chen, X.; Zhang, Z.; Biskup, E. CO2 Fractional Laser Combined with 5-Fluorouracil Ethosomal Gel Treatment of Hypertrophic Scar Macro-, Microscopic, and Molecular Mechanism of Action in a Rabbit Animal Model. Rejuvenation Res. 2021, 24, 131–138. [Google Scholar] [CrossRef]
- Xie, J.; Ji, Y.; Xue, W.; Ma, D.; Hu, Y. Hyaluronic acid-containing ethosomes as a potential carrier for transdermal drug delivery. Colloids Surf. B Biointerfaces 2018, 172, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Jankowska-Polanska, B.; Karniej, P.; Polanski, J.; Sen, M.; Swiatoniowska-Lonc, N.; Grochans, E. Diabetes Mellitus Versus Hypertension-Does Disease Affect Pharmacological Adherence? Front. Pharmacol. 2020, 11, 1157. [Google Scholar] [CrossRef]
- Ammar, H.O.; Tadros, M.I.; Salama, N.M.; Ghoneim, A.M. Ethosome-Derived Invasomes as a Potential Transdermal Delivery System for Vardenafil Hydrochloride: Development, Optimization and Application of Physiologically Based Pharmacokinetic Modeling in Adults and Geriatrics. Int. J. Nanomed. 2020, 15, 5671–5685. [Google Scholar] [CrossRef]
- Liu, X.; Liu, H.; Zeng, Z.; Zhou, W.; Liu, J.; He, Z. Pharmacokinetics of ligustrazine ethosome patch in rats and anti-myocardial ischemia and anti-ischemic reperfusion injury effect. Int. J. Nanomed. 2011, 6, 1391–1398. [Google Scholar] [CrossRef]
- Bodade, S.S.; Shaikh, K.S.; Kamble, M.S.; Chaudhari, P.D. A study on ethosomes as mode for transdermal delivery of an antidiabetic drug. Drug Deliv. 2013, 20, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Wang, Y.; Luo, G. Ligustrazine phosphate ethosomes for treatment of Alzheimer’s disease, in vitro and in animal model studies. AAPS PharmSciTech 2012, 13, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liu, H.; Liu, J.; He, Z.; Ding, C.; Huang, G.; Zhou, W.; Zhou, L. Preparation of a ligustrazine ethosome patch and its evaluation in vitro and in vivo. Int. J. Nanomed. 2011, 6, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Bhosale, S.S.; Avachat, A.M. Design and development of ethosomal transdermal drug delivery system of valsartan with preclinical assessment in Wistar albino rats. J. Liposome Res. 2013, 23, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Sarim Imam, S.; Zafar, A.; Ali, A.; Aqil, M.; Gull, A. In vitro and preclinical assessment of factorial design based nanoethosomes transgel formulation of an opioid analgesic. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1793–1802. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, H.; Zhang, K.; Li, Z.; Guo, T.; Wu, T.; Hou, X.; Feng, N. Co-hybridized composite nanovesicles for enhanced transdermal eugenol and cinnamaldehyde delivery and their potential efficacy in ulcerative colitis. Nanomedicine 2020, 28, 102212. [Google Scholar] [CrossRef] [PubMed]
- Wilson, V.; Siram, K.; Rajendran, S.; Sankar, V. Development and evaluation of finasteride loaded ethosomes for targeting to the pilosebaceous unit. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1892–1901. [Google Scholar] [CrossRef] [PubMed]
- Meng, S.; Chen, Z.; Yang, L.; Zhang, X.; Guo, J.; Li, M.; Li, J. High-efficient nano-carrier gel systems for testosterone propionate skin delivery. Pharm. Dev. Technol. 2015, 20, 724–729. [Google Scholar] [CrossRef]
- Shelke, S.; Shahi, S.; Jalalpure, S.; Dhamecha, D. Poloxamer 407-based intranasal thermoreversible gel of zolmitriptan-loaded nanoethosomes: Formulation, optimization, evaluation and permeation studies. J. Liposome Res. 2016, 26, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Babaie, S.; Ghanbarzadeh, S.; Davaran, S.; Kouhsoltani, M.; Hamishehkar, H. Nanoethosomes for Dermal Delivery of Lidocaine. Adv. Pharm. Bull. 2015, 5, 549–556. [Google Scholar] [CrossRef]
- Fu, X.; Shi, Y.; Wang, H.; Zhao, X.; Sun, Q.; Huang, Y.; Qi, T.; Lin, G. Ethosomal Gel for Improving Transdermal Delivery of Thymosin β-4. Int. J. Nanomed. 2019, 14, 9275–9284. [Google Scholar] [CrossRef] [PubMed]
- El-Menshawe, S.F.; Ali, A.A.; Halawa, A.A.; Srag El-Din, A.S. A novel transdermal nanoethosomal gel of betahistine dihydrochloride for weight gain control: In-vitro and in-vivo characterization. Drug Des. Dev. Ther. 2017, 11, 3377–3388. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Song, Y.K.; Hyun, S.Y.; Kim, H.T.; Kim, C.K.; Oh, J.M. Transdermal delivery of low molecular weight heparin loaded in flexible liposomes with bioavailability enhancement: Comparison with ethosomes. J. Microencapsul. 2011, 28, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Shumilov, M.; Bercovich, R.; Duchi, S.; Ainbinder, D.; Touitou, E. Ibuprofen transdermal ethosomal gel: Characterization and efficiency in animal models. J. Biomed. Nanotechnol. 2010, 6, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Campani, V.; Biondi, M.; Mayol, L.; Cilurzo, F.; Franze, S.; Pitaro, M.; De Rosa, G. Nanocarriers to Enhance the Accumulation of Vitamin K1 into the Skin. Pharm. Res. 2016, 33, 893–908. [Google Scholar] [CrossRef] [PubMed]
- Shumilov, M.; Touitou, E. Buspirone transdermal administration for menopausal syndromes, in vitro and in animal model studies. Int. J. Pharm. 2010, 387, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Limsuwan, T.; Boonme, P.; Khongkow, P.; Amnuaikit, T. Ethosomes of Phenylethyl Resorcinol as Vesicular Delivery System for Skin Lightening Applications. Biomed. Res. Int. 2017, 2017, 8310979. [Google Scholar] [CrossRef]
- Ansari, S.A.; Qadir, A.; Warsi, M.H.; Mujeeb, M.; Aqil, M.; Mir, S.R.; Sharma, S. Ethosomes-based gel formulation of karanjin for treatment of acne vulgaris: In vitro investigations and preclinical assessment. 3 Biotech 2021, 11, 456. [Google Scholar] [CrossRef]
- Goindi, S.; Dhatt, B.; Kaur, A. Ethosomes-based topical delivery system of antihistaminic drug for treatment of skin allergies. J. Microencapsul. 2014, 31, 716–724. [Google Scholar] [CrossRef]
- Shukla, R.; Tiwari, G.; Tiwari, R.; Rai, A.K. Formulation and evaluation of the topical ethosomal gel of melatonin to prevent UV radiation. J. Cosmet. Dermatol. 2020, 19, 2093–2104. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).