Promising Anticancer Activity of β-Carboline Derivatives: Design, Synthesis, and Pharmacological Evaluation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of β-Carboline Derivatives
2.2. Biological Screening
2.3. Docking Studies of Designed Compounds
3. Results and Discussion
3.1. Biological Screening Results
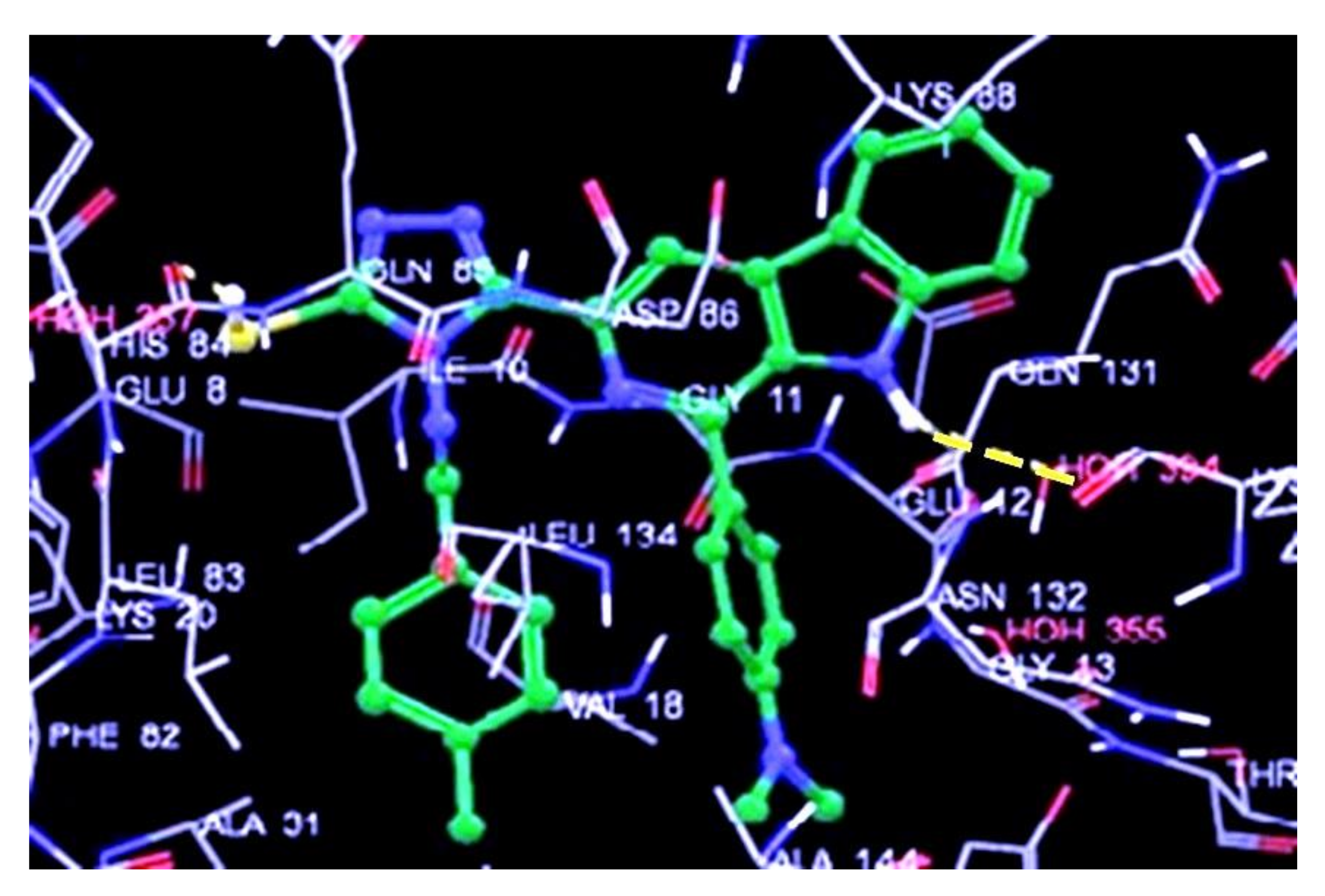
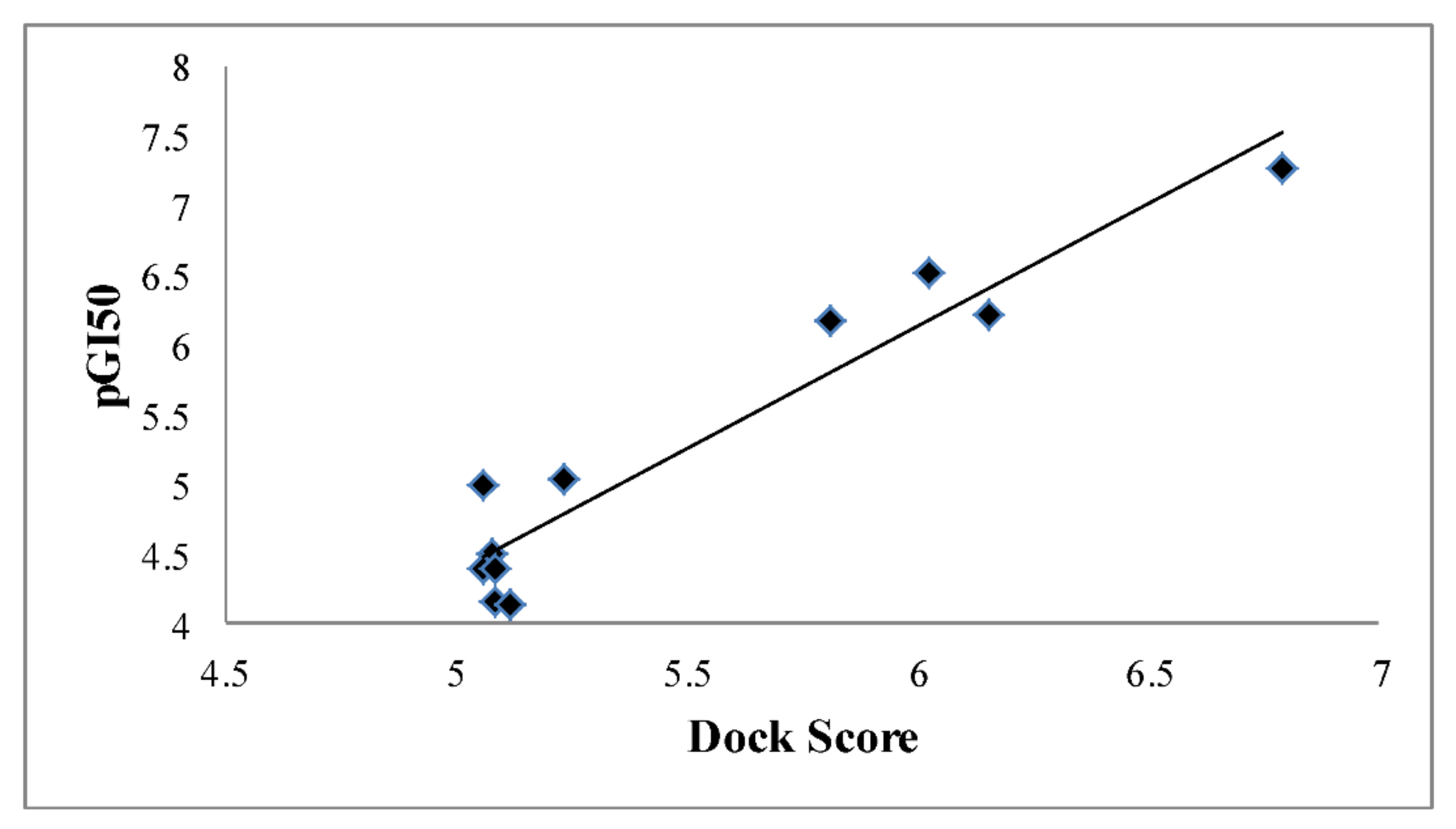
3.2. Docking Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, I.; Bray, A.F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 713, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Lopez, J.S.; Banerji, U. Combine and conquer: Challenges for targeted therapy combinations in early phase trials. Nat. Rev. Clin. Oncol. 2017, 141, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Chandra, V.; Jain, P.K.; Pathak, K.; Pathak, D.; Vaidya, A. Comprehensive review on current developments of quinoline-based anticancer agents. Arab. J. Chem. 2019, 128, 4920–4946. [Google Scholar] [CrossRef]
- Xiao, S.; Lin, W.; Wang, C.; Yang, M. Synthesis and Biological Evaluation of DNA Targeting Flexible Side-Chain Substituted β-Carboline Derivatives. Bioorg. Med. Chem. Lett. 2001, 11, 437–441. [Google Scholar] [CrossRef]
- Cao, R.; Peng, W.; Chen, H.; Hou, X.; Guan, H.; Chen, Q.; Ma, Y.; Xu, A. Synthesis and in vitro cytotoxic evaluation of 1,3-bisubstituted and 1,3,9-trisubstituted β-carboline derivatives. Eur. J. Med. Chem. 2005, 40, 249–257. [Google Scholar] [CrossRef]
- Castro, A.C.; Dang, L.C.; Soucy, F.; Grenier, L.; Mazdiyasni, H.; Hottelet, M.; Parent, L.; Pien, C.; Palombella, V.; Adams, J. Novel IKK Inhibitors: β-Carbolines. Bioorg. Med. Chem. Lett. 2003, 13, 2419–2422. [Google Scholar] [CrossRef]
- Deveau, A.M.; Labroli, M.A.; Dieckhaus, C.M.; Barthen, M.T.; Smith, K.S.; Macdonald, T.L. The Synthesis of Amino-Acid Functionalized β-Carbolines as Topoisomerase II Inhibitors. Bioorg. Med. Chem. Lett. 2001, 11, 1251–1255. [Google Scholar] [CrossRef]
- Cao, R.; Guan, X.; Shi, B.; Chen, Z.; Ren, Z.; Peng, W.; Song, H. Design, synthesis and 3D-QSAR of β-carboline derivatives as potent antitumor agents. Eur. J. Med. Chem. 2010, 45, 2503–2515. [Google Scholar] [CrossRef]
- Cao, R.; Yi, W.; Wu, Q.; Guan, X.; Feng, M.; Ma, C.; Chen, Z.; Song, H.; Peng, W. Synthesis and cytotoxic activities of 1-benzylidine substituted β-carboline derivatives. Bioorg. Med. Chem. Lett. 2008, 18, 6558–6561. [Google Scholar] [CrossRef]
- Trujillo, J.I.; Meyers, M.J.; Anderson, D.R.; Hegde, S.; Mahoney, M.W.; Vernier, W.F.; Buchler, I.P.; Wu, K.K.; Yang, S.; Hartmann, S.J.; et al. Novel tetrahydro-β-carboline-1-carboxylic acids as inhibitors of mitogen activated protein kinase-activated protein kinase 2 (MK-2). Bioorg. Med. Chem. Lett. 2007, 17, 4657–4663. [Google Scholar] [CrossRef]
- Barsanti, P.A.; Wang, W.; Ni, Z.; Duhl, D.; Brammeier, N.; Martin, E.; Bussiere, D.; Walter, A.O. The discovery of tetrahydro-β-carbolines as inhibitors of the kinesin Eg5. Bioorg. Med. Chem. Lett. 2010, 20, 157–160. [Google Scholar] [CrossRef] [PubMed]
- Cao, R.; Peng, W.; Chen, H.; Ma, Y.; Liu, X. DNA binding properties of 9-substituted harmine derivatives. Biochem. Biophys. Res. Commun. 2005, 3383, 1557–1563. [Google Scholar] [CrossRef]
- Chourasiya, K.R.; Rao, R.A.; Agrawal, K.R. Pharmacophore Modeling and QSAR Analysis of Novel β-carboline Derivatives as Antitumor Agents. Lett. Drug Des. Discov. 2013, 107, 572–584. [Google Scholar]
- Formagio, A.S.; Tonin, L.T.; Foglio, M.A.; Madjarof, C.; de Carvalho, J.E.; da Costa, W.F.; Cardoso, F.P.; Sarragiotto, M.H. Synthesis and antitumoral activity of novel 3-(2-substituted-1,3,4-oxadiazol-5-yl) and 3-(5-substituted-1,2,4-triazol-3-yl) beta-carboline derivatives. Bioorg. Med. Chem. 2008, 16, 9660–9667. [Google Scholar] [CrossRef] [PubMed]
- Ishida, J.; Wang, H.K.; Bastow, K.F.; Hu, C.Q.; Lee, K.H. Antitumor agents 201. Cytotoxicity of harmine and beta-carboline analogs. Bioorg. Med. Chem. Lett. 1999, 9, 3319–3324. [Google Scholar] [CrossRef]
- Cao, R.; Chen, H.; Peng, W.; Ma, Y.; Hou, X.; Guan, H.; Liu, X.; Xu, A. Design, synthesis and in vitro and in vivo antitumor activities of novel beta-carboline derivatives. Eur. J. Med. Chem. 2005, 40, 991–1001. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Bi, L.; Wang, W.; Wang, C.; Baudy-Floc’h, M.; Ju, J.; Peng, S. Synthesis and cytotoxic activities of beta-carboline amino acid ester conjugates. Bioorg. Med. Chem. 2006, 14, 6998–7010. [Google Scholar]
- Vaidya, A.; Jain, S.; Jain, A.K.; Prashanthakumar, B.R.; Kashaw, S.K.; Agrawal, R.K. Computational Analysis of Quinoline Derivatives as Potent Topoisomerase-II Inhibitors. Med. Chem. Res. 2015, 24, 383–393. [Google Scholar] [CrossRef]
- Vaidya, A.; Jain, A.K.; Kumar, P.; Kashaw, S.K.; Agrawal, R.K. Predicting anti-cancer activity of quinoline derivatives: CoMFA and CoMSIA approach. J. Enzyme Inhib. Med. Chem. 2011, 26, 854–861. [Google Scholar]
- Agrawal, R.K.; Jain, A.K.; Veerasamy, R.; Vaidya, A.; Kashaw, S.; Mourya, V.K.; Agrawal, R.K. QSAR analysis of B-ring-modified diaryl ether derivatives as a InhA inhibitors. Med. Chem. Res. 2012, 21, 145–151. [Google Scholar]
- Jain, S.; Vaidya, A.; Jain, A.K.; Agrawal, R.K.; Kashaw, S.K. Computational analysis of benzyl vinylogous derivatives as potent PDE3B inhibitors. Arab. J. Chem. 2017, 10, S109–S113. [Google Scholar] [CrossRef]
- Vaidya, A. Discovery of novel 1,2,4-oxadiazole derivatives as potent caspase-3 activator for cancer treatment. Chemistry 2021, 3, 373–381. [Google Scholar] [CrossRef]
- Chourasiya, R.K.; Rao, A.R.; Agrawal, R.K. QSAR and docking studies of novel β-carboline derivatives as anticancer. Med. Chem. Res. 2013, 22, 2991–3001. [Google Scholar] [CrossRef]
- Vaidya, A.; Jain, S.; Kumar, B.R.P.; Singh, S.K.; Kashaw, S.K.; Agrawal, R.K. Synthesis of 1.;2.;4-oxadiazole derivatives: Anticancer and 3D QSAR studies. Mon. Chem. 2020, 151, 385–395. [Google Scholar] [CrossRef]
- Shankaraiah, N.; Jadala, C.; Nekkanti, S.; Senwar, K.R.; Nagesh, N.; Shrivastava, S.; Naidu, V.G.; Sathish, M.; Kamal, A. Design and synthesis of C3-tethered 1,2,3-triazolo-β-carboline derivatives: Anticancer activity, DNA-binding ability, viscosity and molecular modeling studies. Bioorg. Chem. 2016, 64, 42–50. [Google Scholar] [CrossRef]
- Abdelsalam, M.A.; AboulWafa, O.M.; Badawey, M.E.A.; El-Shoukrofy, M.S.; El-Miligy, M.M.; Gouda, N.; Elaasser, M.M. Design, synthesis, anticancer screening, docking studies and in silico ADME prediction of some β-carboline derivatives. Future Med. Chem. 2018, 10, 1159–1175. [Google Scholar] [CrossRef]
- Vaidya, A.; Jain, A.K.; Kumar, B.R.P.; Sastry, G.N.; Kashaw, S.K.; Agrawal, R.K. CoMFA, CoMSIA, kNN MFA and Docking studies of 1,2,4-Oxadiazole derivatives as potent Caspase-3 activators. Arab. J. Chem. 2017, 10, S3936–S3946. [Google Scholar] [CrossRef]
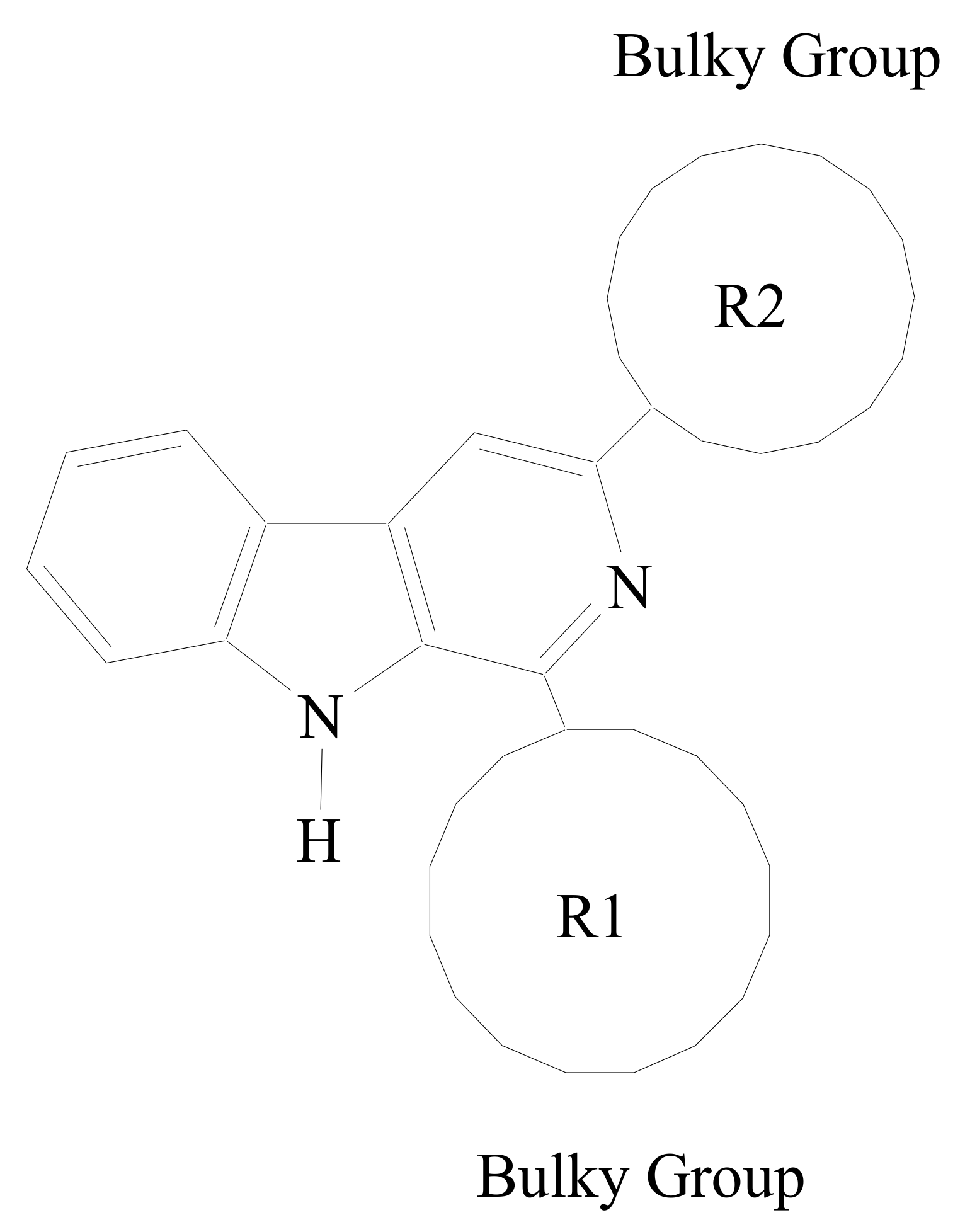

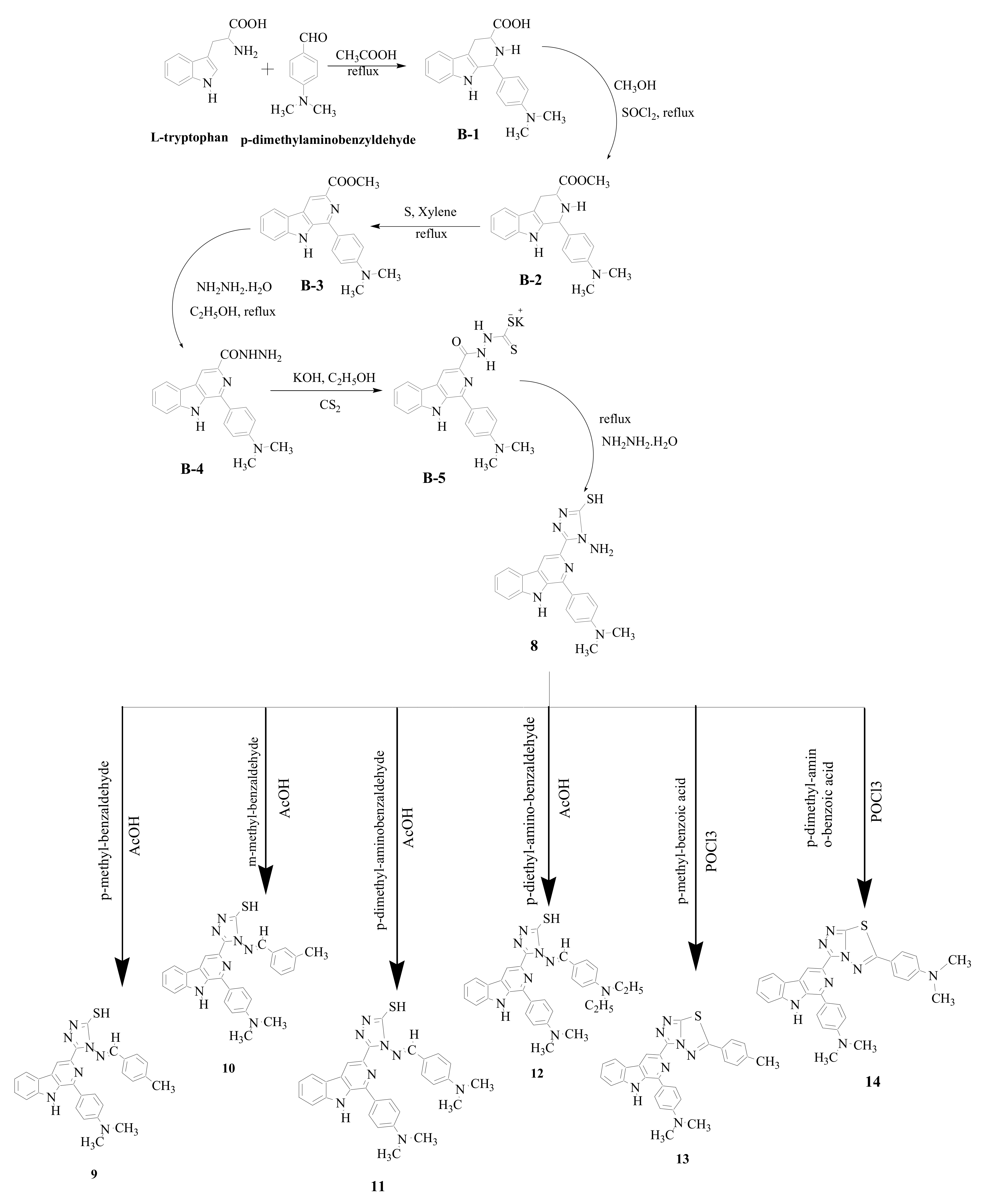

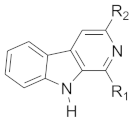
| Compound No. | R1 | R2 |
|---|---|---|
| 1 |  | 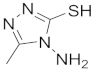 |
| 2 |  |  |
| 3 |  |  |
| 4 |  | 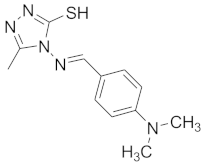 |
| 5 |  | 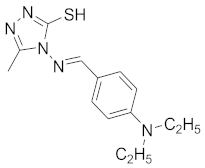 |
| 6 |  | 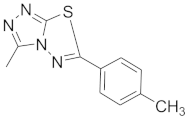 |
| 7 |  | 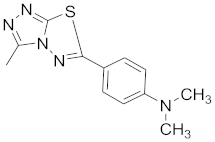 |
| 8 | 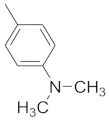 | 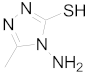 |
| 9 |  | 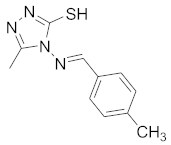 |
| 10 |  | 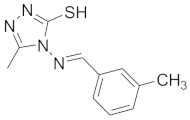 |
| 11 |  | 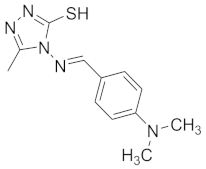 |
| 12 |  | 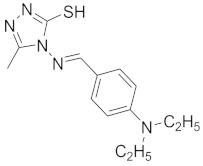 |
| 13 |  | 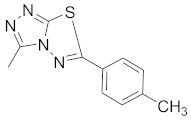 |
| 14 |  | 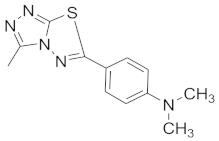 |
| Compound No. | Growth Inhibitory Effects, GI50 (µM) | Dock Score (G-Score) | |
|---|---|---|---|
| HepG2 Cell Line | A549 Cell Line | ||
| 1 | 8.14 | 7.21 | 4.15 |
| 2 | 8.34 | 8.87 | 4.48 |
| 3 | 8.73 | 8.22 | 4.98 |
| 4 | 0.94 | 4.10 | 6.52 |
| 5 | 8.64 | 8.05 | 4.39 |
| 6 | >100 | >100 | 3.27 |
| 7 | >100 | >100 | 3.98 |
| 8 | 8.12 | 7.35 | 4.38 |
| 9 | 0.16 | 0.14 | 7.28 |
| 10 | 0.69 | 6.76 | 6.21 |
| 11 | 5.81 | 6.52 | 5.03 |
| 12 | 7.61 | 4.22 | 4.12 |
| 13 | >100 | >100 | 4.05 |
| 14 | 1.54 | 1.42 | 6.17 |
| Adriamycin | 0.1 | 0.1 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chourasiya, R.K.; Agrawal, R.K.; Vaidya, A. Promising Anticancer Activity of β-Carboline Derivatives: Design, Synthesis, and Pharmacological Evaluation. Chemistry 2022, 4, 1395-1406. https://doi.org/10.3390/chemistry4040091
Chourasiya RK, Agrawal RK, Vaidya A. Promising Anticancer Activity of β-Carboline Derivatives: Design, Synthesis, and Pharmacological Evaluation. Chemistry. 2022; 4(4):1395-1406. https://doi.org/10.3390/chemistry4040091
Chicago/Turabian StyleChourasiya, Ravindra Kumar, Ram Kishore Agrawal, and Ankur Vaidya. 2022. "Promising Anticancer Activity of β-Carboline Derivatives: Design, Synthesis, and Pharmacological Evaluation" Chemistry 4, no. 4: 1395-1406. https://doi.org/10.3390/chemistry4040091
APA StyleChourasiya, R. K., Agrawal, R. K., & Vaidya, A. (2022). Promising Anticancer Activity of β-Carboline Derivatives: Design, Synthesis, and Pharmacological Evaluation. Chemistry, 4(4), 1395-1406. https://doi.org/10.3390/chemistry4040091






