Combining Coordination and Hydrogen Bonds to Develop Discrete Supramolecular Metalla-Assemblies
Abstract
:1. Introduction
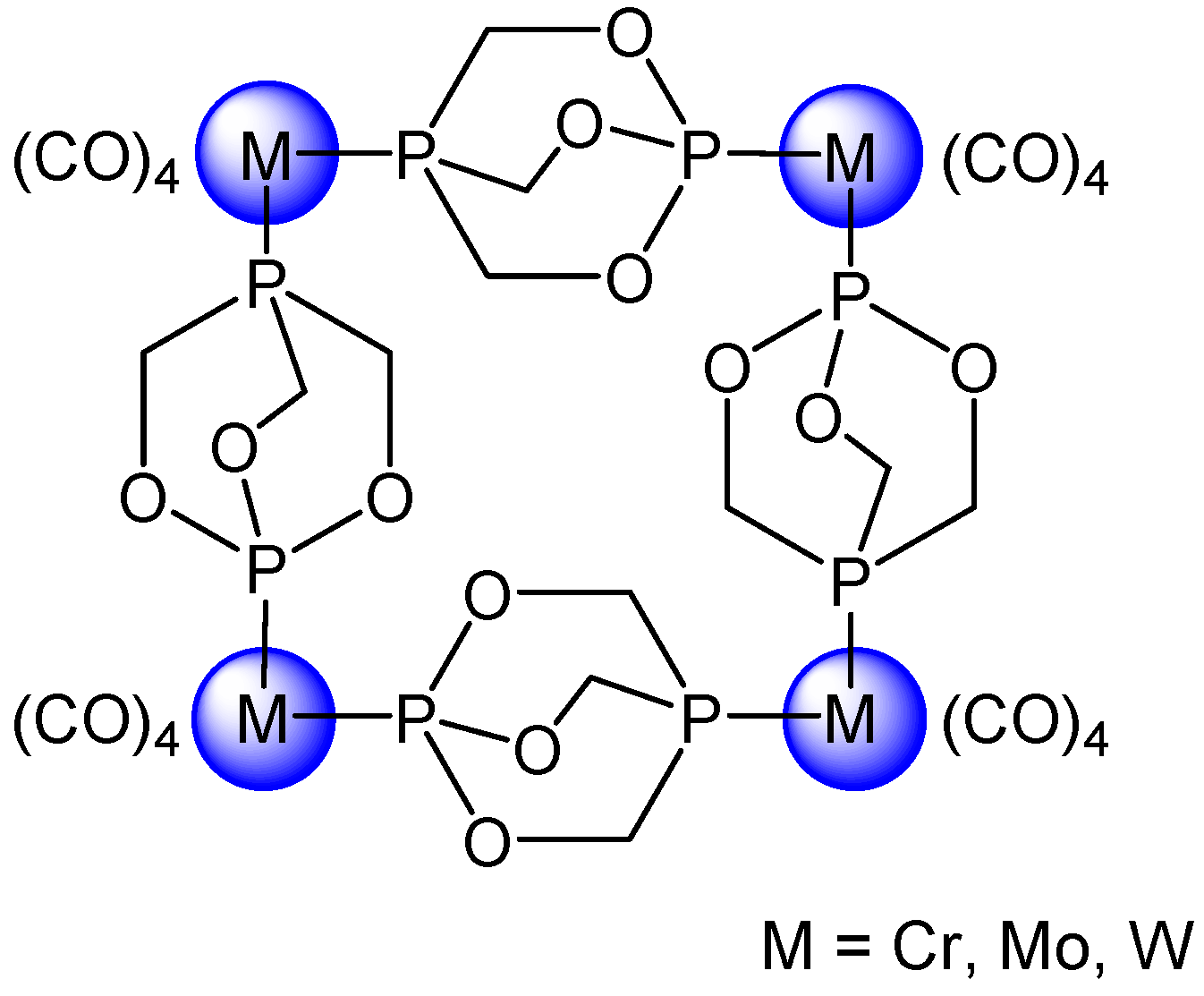
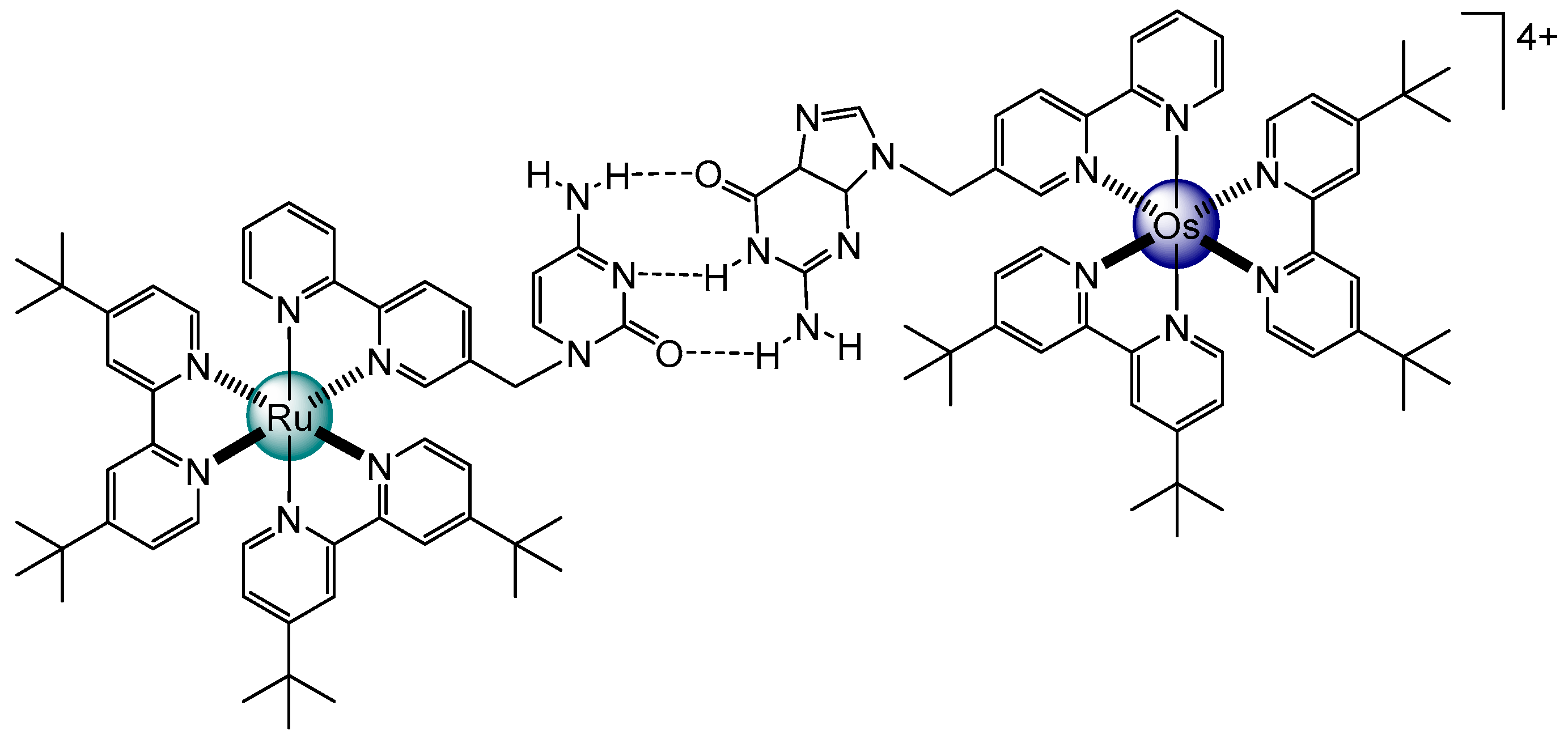
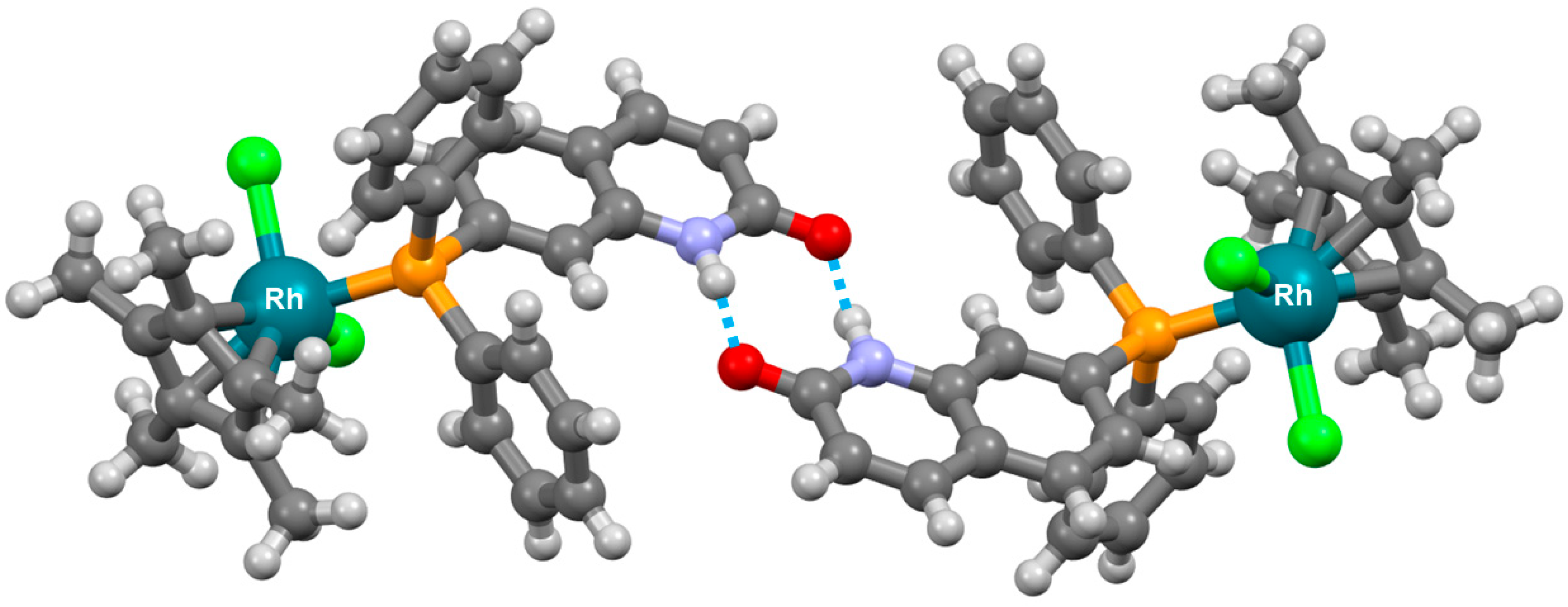
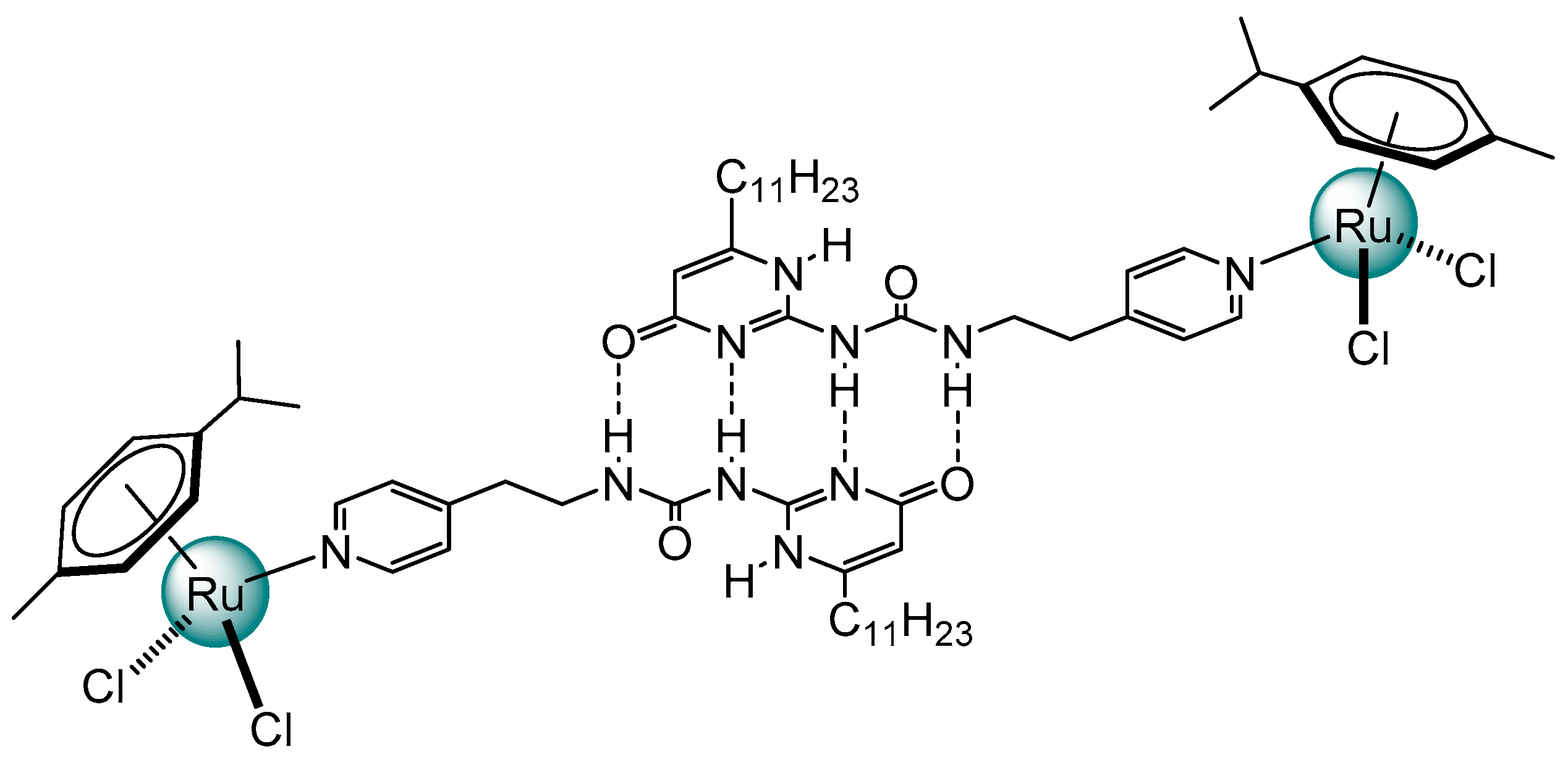
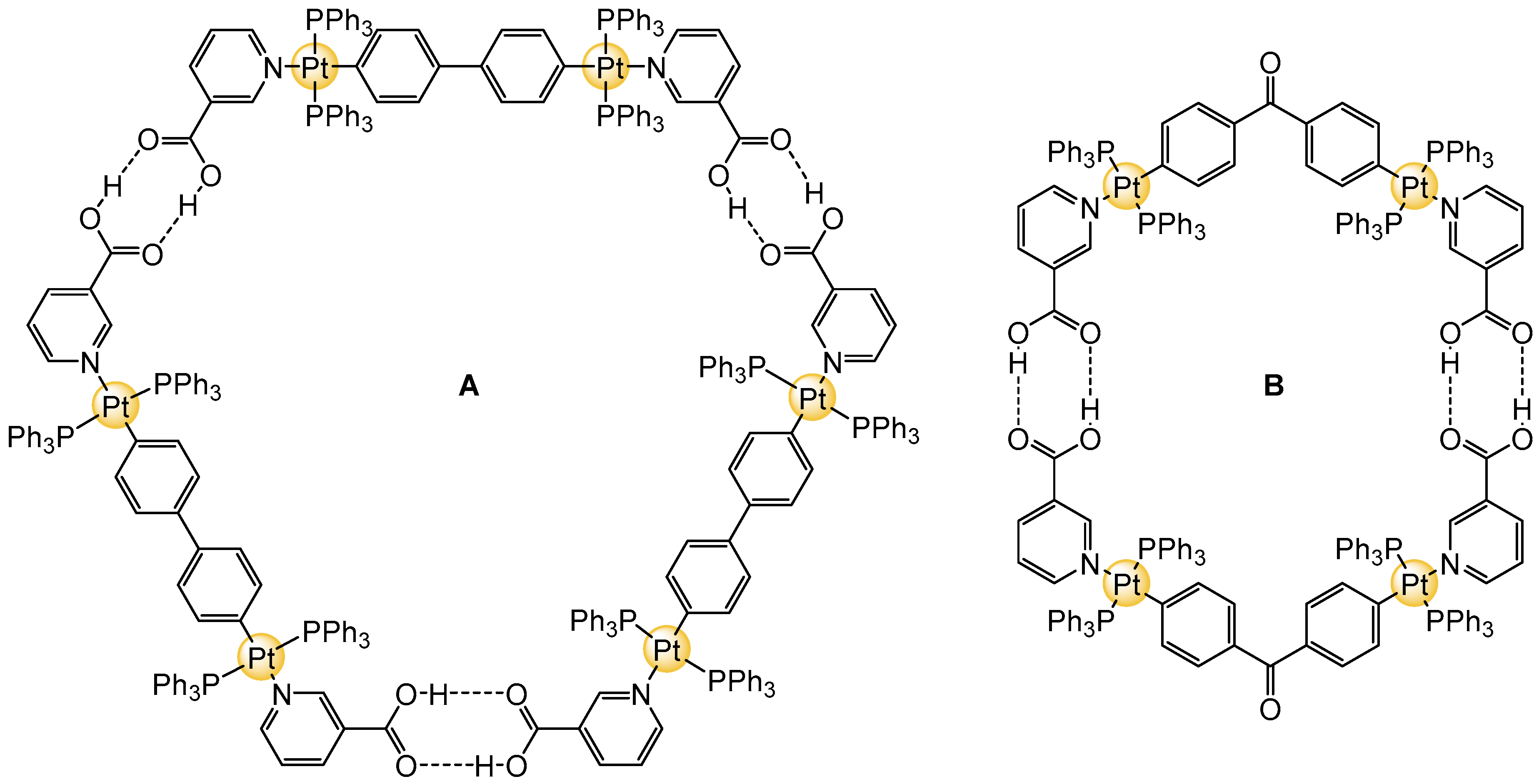
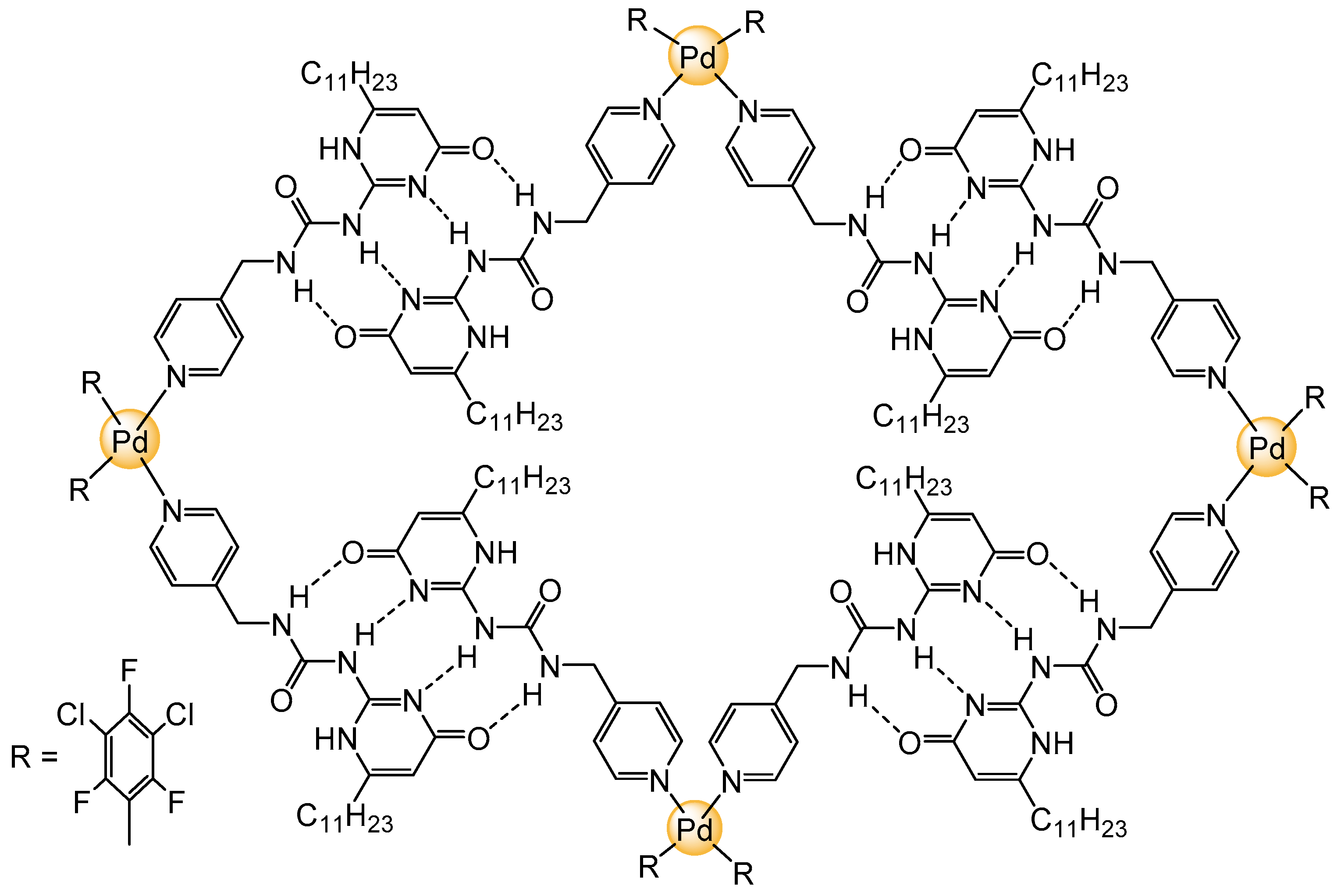
2. Planar and Macrocyclic Assemblies Exploiting Coordination and Hydrogen Bonds
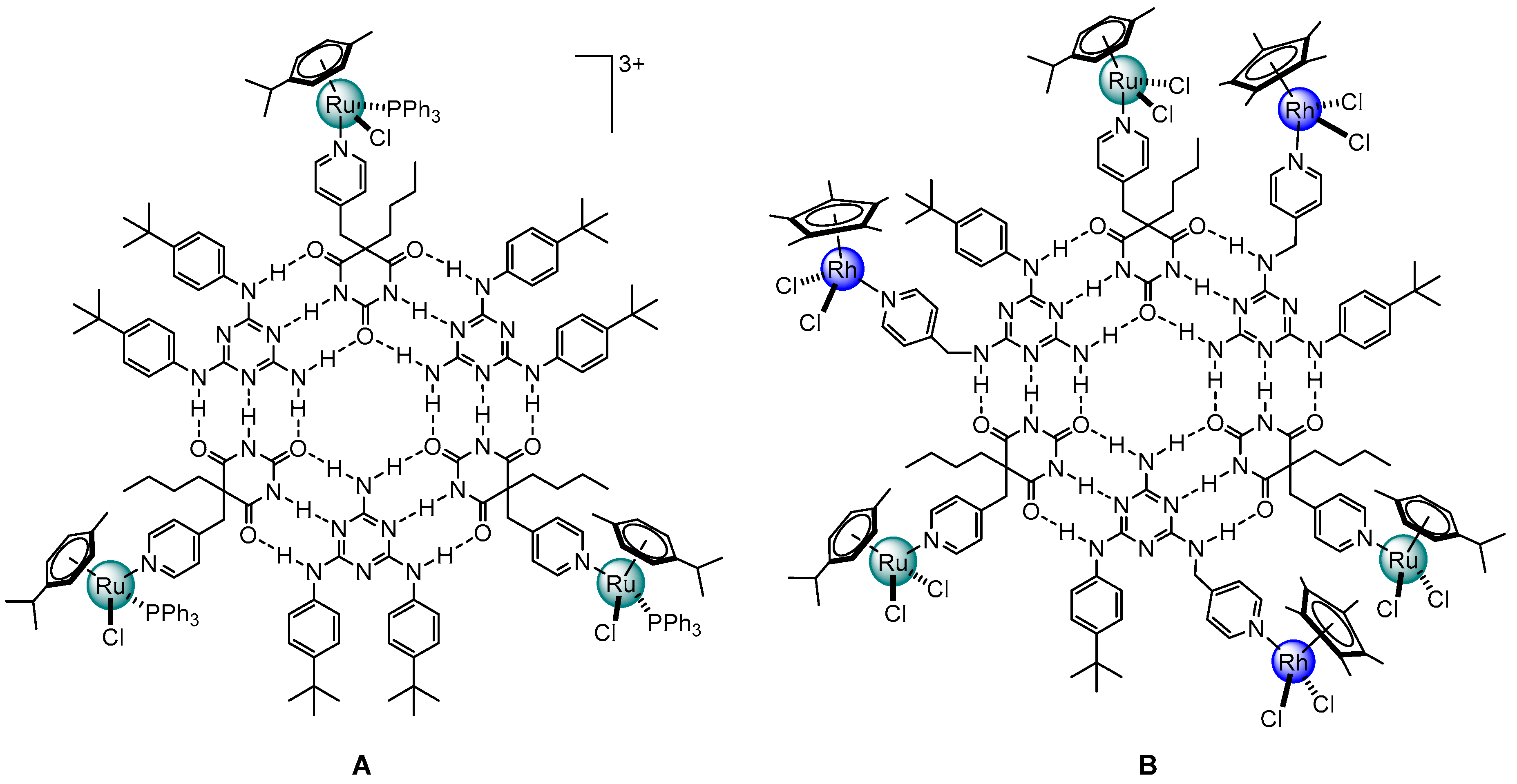
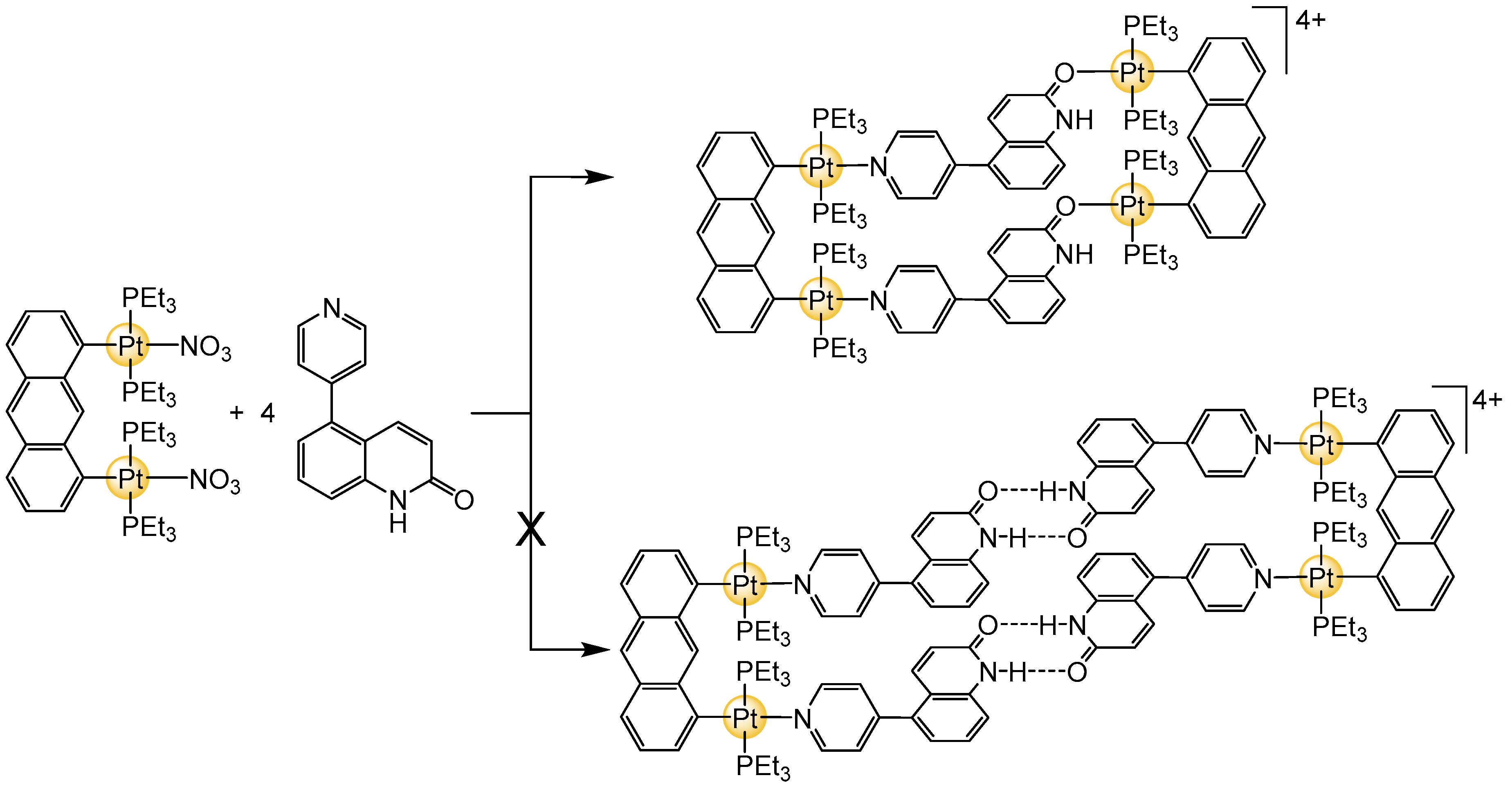
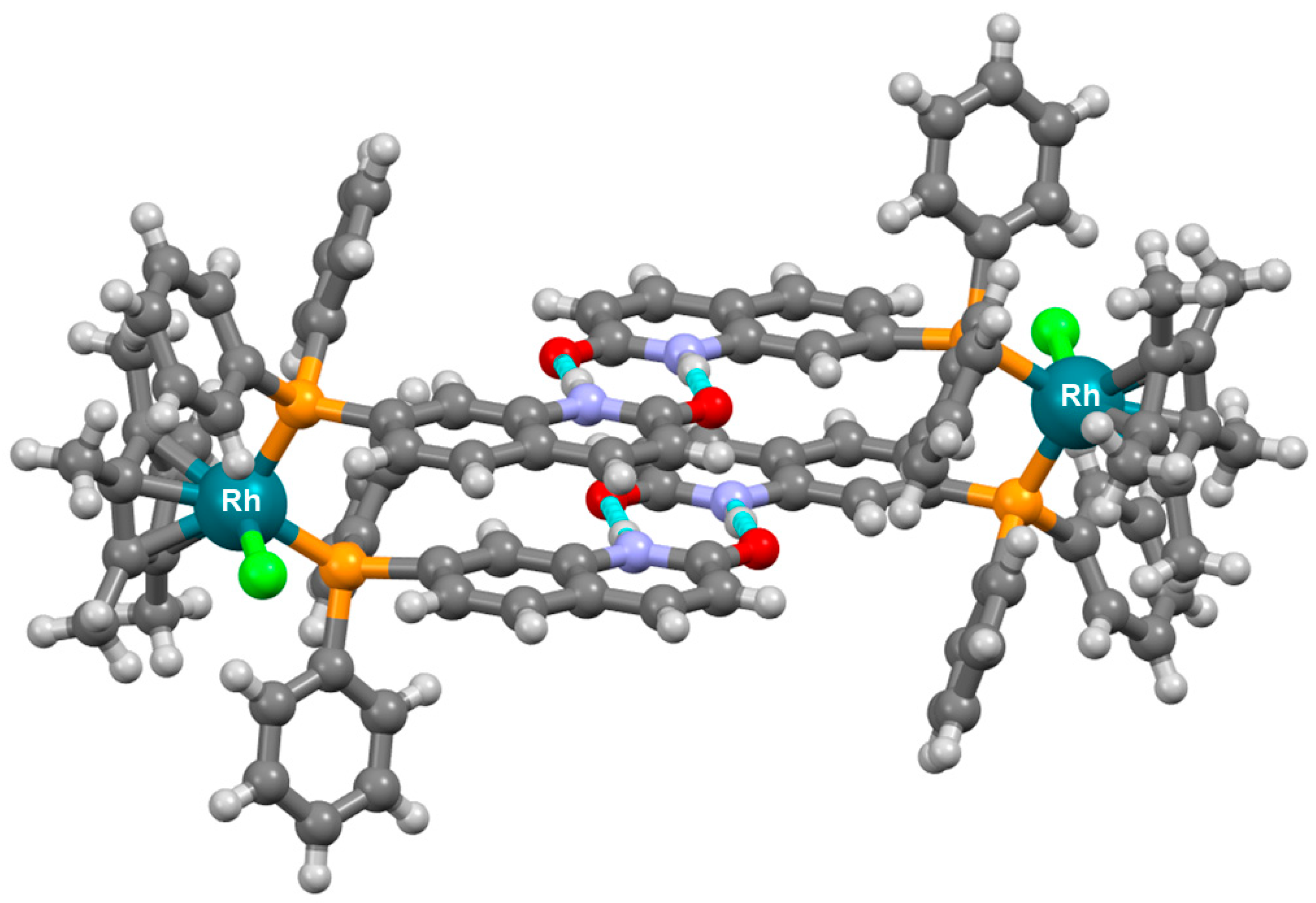
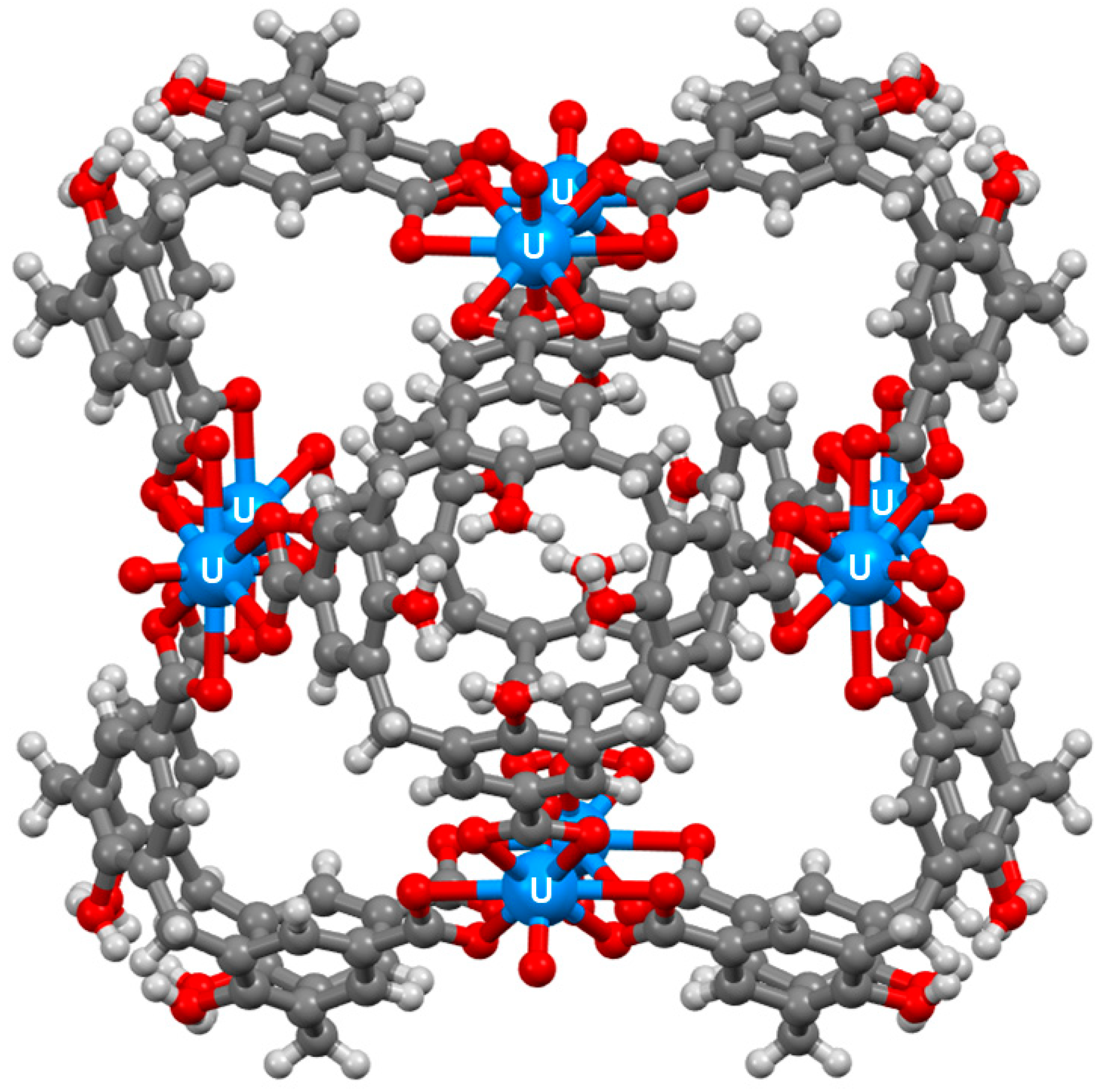
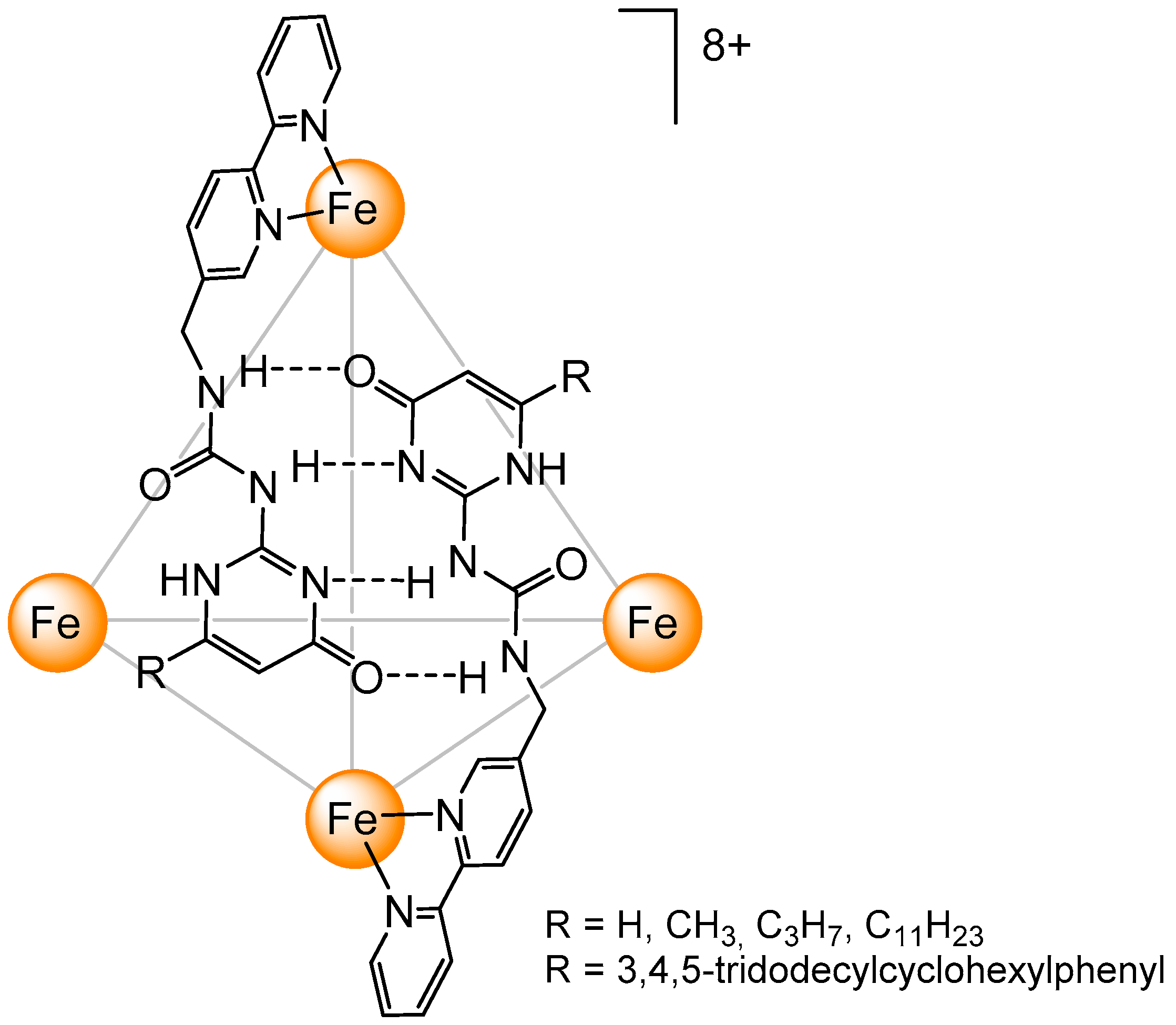
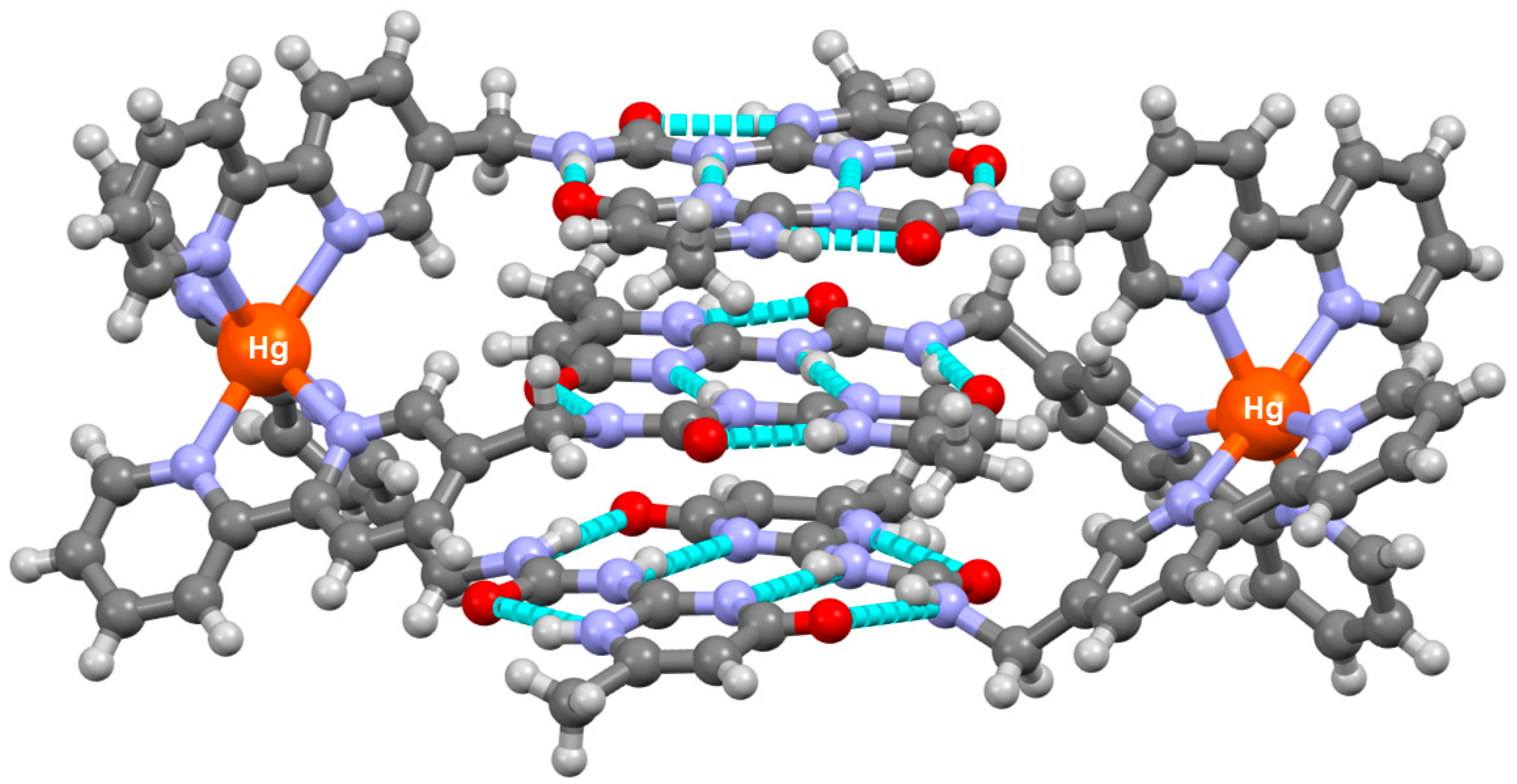
3. Cage-Like Assemblies Exploiting Coordination and Hydrogen Bonds
4. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, L.-J.; Yang, H.-B. Construction of stimuli-responsive functional materials via hierarchical self-assembly involving coordination interactions. Acc. Chem. Res. 2018, 51, 2699–2710. [Google Scholar] [CrossRef] [PubMed]
- Cook, T.R.; Stang, P.J. Recent developments in the preparation and chemistry of metallacycles and metallacages via coordination. Chem. Rev. 2015, 115, 7001–7045. [Google Scholar] [CrossRef] [PubMed]
- Datta, S.; Saha, M.L.; Stang, P.J. Hierarchical assemblies of supramolecular coordination complexes. Acc. Chem. Res. 2018, 51, 2047–2063. [Google Scholar] [CrossRef] [PubMed]
- Stíbal, D.; Therrien, B. Organometallic chemistry in directed assembly. In Comprehensive Supramolecular Chemistry II; Atwood, J.L., Ed.; Elsevier: Oxford, UK, 2017; Volume 6, pp. 305–329. [Google Scholar]
- Hosseini, M.W. Molecular tectonics: From simple tectons to complex molecular networks. Acc. Chem. Res. 2005, 38, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Chen, L.-J.; Zhang, Z.; Luo, Q.; Yang, H.-B.; Tian, H.; Zhu, W.-H. Conformer-dependent self-assembled metallacycles with photo-reversible response. Chem. Sci. 2019, 10, 4896–4904. [Google Scholar] [CrossRef] [PubMed]
- Percástegui, E.G.; Jancik, V. Coordination-driven assemblies based on meso-substituted porphyrins: Metal-organic cages and a new type of meso-metallaporphyrin macrocycles. Coord. Chem. Rev. 2020, 407, 213165. [Google Scholar] [CrossRef]
- Barry, N.P.E.; Therrien, B. Pyrene: The guest of honor. In Organic Nanoreactors; Sadjadi, S., Ed.; Elsevier: London, UK, 2016; pp. 421–461. [Google Scholar]
- Pöthig, A.; Casini, A. Recent developments of supramolecular metal-based structures for applications in cancer therapy and imaging. Theranostics 2019, 9, 93150–93169. [Google Scholar] [CrossRef]
- Sepehrpour, H.; Fu, W.; Sun, Y.; Stang, P.J. Biomedically relevant self-assembled metallacycles and metallacages. J. Am. Chem. Soc. 2019, 141, 14005–14020. [Google Scholar] [CrossRef]
- Amouri, H.; Desmarets, C.; Moussa, J. Confined nanospaces in metallocages: Guest molecules, weakly encapsulated anions, and catalyst sequestration. Chem. Rev. 2012, 112, 2015–2041. [Google Scholar] [CrossRef]
- Wang, G.-L.; Lin, Y.-J.; Berke, H.; Jin, G.-X. Two-step assembly of multinuclear metallacycles with half-sandwich Ir, Rh, and Ru fragments for counteranion encapsulation. Inorg. Chem. 2010, 49, 2193–2201. [Google Scholar] [CrossRef]
- Therrien, B. Biologically relevant arene ruthenium metalla-assemblies. CrystEngComm 2015, 17, 484–491. [Google Scholar] [CrossRef]
- Chen, L.; Chen, C.; Sun, Y.; Lu, S.; Huo, H.; Tan, T.; Li, A.; Li, X.; Ungar, G.; Liu, F.; et al. Luminescent metallacycle-cored liquid crystals induced by metal coordination. Angew. Chem. Int. Ed. 2020, 59. [Google Scholar] [CrossRef]
- Therrien, B. Thermotropic liquid-crystalline materials based on supramolecular coordination complex. Inorganics 2020, 8, 2. [Google Scholar] [CrossRef] [Green Version]
- Cullen, W.; Misuraca, M.C.; Hunter, C.A.; Williams, N.H.; Ward, M.D. Highly efficient catalysis of the Kemp elimination in the cavity of a cubic coordination cage. Nat. Chem. 2016, 8, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, M.; Klosterman, J.K.; Fujita, M. Functional molecular flasks: New properties and reactions within discrete, self-assembled hosts. Angew. Chem. Int. Ed. 2009, 48, 3418–3438. [Google Scholar] [CrossRef]
- Stricklen, P.M.; Volcko, E.J.; Verkade, J.G. Novel homo- and heterometallic coordination macrocycles. J. Am. Chem. Soc. 1983, 105, 2494–2495. [Google Scholar] [CrossRef]
- Fujita, M.; Tominaga, M.; Hori, A.; Therrien, B. Coordination assemblies from a Pd (II)-cornered square complex. Acc. Chem. Res. 2005, 38, 371–380. [Google Scholar] [CrossRef]
- Conn, M.M.; Rebek, J., Jr. Self-assembling capsules. Chem. Rev. 1997, 97, 1647–1668. [Google Scholar] [CrossRef]
- Adriaenssens, L.; Ballester, P. Hydrogen bonded supramolecular capsules with functionalized interiors: The controlled orientation of included guests. Chem. Soc. Rev. 2013, 42, 3261–3277. [Google Scholar] [CrossRef]
- Sigel, R.K.O.; Freisinger, E.; Metzger, S.; Lippert, B. Metalated nucleobase quartets: Dimerization of a metal-modified guanine, cytosine pair of trans-(NH3)2PtII and formation of CH···N hydrogen bonds. J. Am. Chem. Soc. 1998, 120, 12000–12007. [Google Scholar] [CrossRef]
- Young, M.C.; Holloway, L.R.; Johnson, A.M.; Hooley, R.J. A supramolecular sorting hat: Stereocontrol in metal-ligand self-assembly by complementary hydrogen bonding. Angew. Chem. Int. Ed. 2014, 53, 9832–9836. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, C.K.; Hamblin, G.D.; Sleiman, H.F. Supramolecular DNA assembly. Chem. Soc. Rev. 2011, 40, 5647–5656. [Google Scholar] [CrossRef] [PubMed]
- Philp, D.; Stoddart, J.F. Self-assembly in natural and unnatural systems. Angew. Chem. Int. Ed. 1996, 35, 1154–1196. [Google Scholar] [CrossRef]
- Krische, M.J.; Lehn, J.-M. The utilization of persistent H-bonding motifs in the self-assembly of supramolecular architectures. Struct. Bond. 2000, 96, 3–29. [Google Scholar]
- Sijbesma, R.P.; Meijer, E.W. Quadruple hydrogen bonded systems. Chem. Commun. 2003, 5–16. [Google Scholar] [CrossRef] [PubMed]
- Van Manen, H.-J.; Paraschiv, V.; García-López, J.J.; Schönherr, H.; Zapotoczny, S.; Vancso, G.J.; Crego-Calama, M.; Reinhoudt, D.N. Hydrogen-bonded assemblies as a scaffold for metal-containing nanostructures: From zero to two dimensions. Nano Lett. 2004, 4, 441–446. [Google Scholar] [CrossRef]
- Lehn, J.-M. Perspectives in Chemistry—Steps towards complex matter. Angew. Chem. Int. Ed. 2013, 52, 2836–2850. [Google Scholar] [CrossRef]
- Wong, C.-H.; Zimmerman, S.C. Orthogonality in organic, polymer, and supramolecular chemistry: From Merrifield to click chemistry. Chem. Commun. 2013, 49, 1679–1695. [Google Scholar] [CrossRef]
- Krieg, E.; Bastings, M.M.C.; Besenius, P.; Rybtchinski, B. Supramolecular polymers in aqueous media. Chem. Rev. 2016, 116, 2414–2477. [Google Scholar] [CrossRef]
- Ayme, J.-F.; Lehn, J.-M. From coordination chemistry to adaptive chemistry. Adv. Inorg. Chem. 2018, 71, 3–78. [Google Scholar]
- Huck, W.T.S.; Hulst, R.; Timmerman, P.; van Veggel, F.C.J.M.; Reinhoudt, D.N. Noncovalent synthesis of nanostructures: Combining coordination chemistry and hydrogen bonding. Angew. Chem. Int. Ed. 1997, 36, 1006–1008. [Google Scholar] [CrossRef] [Green Version]
- Yan, X.; Li, S.; Pollock, J.B.; Cook, T.R.; Chen, J.; Zhang, Y.; Ji, X.; Yu, Y.; Huang, F.; Stang, P.J. Supramolecular polymers with tunable topologies via hierarchical coordination-driven self-assembly and hydrogen bonding interfaces. Proc. Nat. Acad. Sci. USA 2013, 110, 15585–15590. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Q.; Li, T.; Duan, A.; Dong, S.; Zhao, W.; Stang, P.J. Formation of a supramolecular polymeric adhesive via water-participant hydrogen bond formation. J. Am. Chem. Soc. 2019, 141, 8058–8063. [Google Scholar] [CrossRef] [PubMed]
- Burrows, A.D.; Chan, C.-W.; Chowdhry, M.M.; McGrady, J.E.; Mingos, D.M.P. Multidimensional crystal engineering of bifunctional metal complexes containing complementary triple hydrogen bonds. Chem. Soc. Rev. 1995, 24, 329–339. [Google Scholar] [CrossRef]
- Armaroli, N.; Barigelletti, F.; Calogero, G.; Flamigni, L.; White, C.M.; Ward, M.D. Electronic energy transfer between ruthenium (II) and osmium (II) polypyridyl luminophores in a hydrogen-bonded supramolecular assembly. Chem. Commun. 1997, 2181–2182. [Google Scholar] [CrossRef]
- White, C.M.; Fernandez Gonzalez, M.; Bardwell, D.A.; Rees, L.H.; Jeffery, J.C.; Ward, M.D.; Armaroli, N.; Calogero, G.; Barigelletti, F. Derivatives of luminescent metal-polypyridyl complexes with pendant adenine or thymine groups: Building blocks for supramolecular assemblies based on hydrogen bonding. J. Chem. Soc. Dalton Trans. 1997, 727–735. [Google Scholar] [CrossRef]
- Sommer, S.K.; Zakharov, L.N.; Pluth, M.D. Design, synthesis, and characterization of hybrid metal-ligand hydrogen-bonded (MLHB) supramolecular architectures. Inorg. Chem. 2015, 54, 1912–1918. [Google Scholar] [CrossRef]
- Sommer, S.K.; Henle, E.A.; Zakharov, L.N.; Pluth, M.D. Selection for a single self-assembled macrocycle from a hybrid metal-ligand hydrogen-bonded (MLHB) ligand subunit. Inorg. Chem. 2015, 54, 6910–6916. [Google Scholar] [CrossRef]
- Appavoo, D.; Carnevale, D.; Deschenaux, R.; Therrien, B. Combining coordination and hydrogen-bonds to form arene ruthenium metalla-assemblies. J. Organomet. Chem. 2016, 824, 80–87. [Google Scholar] [CrossRef] [Green Version]
- Appavoo, D.; Raja, N.; Deschenaux, R.; Therrien, B.; Carnevale, D. NMR spectroscopy and DFT calculations of a self-assembled arene ruthenium rectangle obtained from a combination of coordination and hydrogen bonds. Dalton Trans. 2016, 45, 1410–1421. [Google Scholar] [CrossRef] [Green Version]
- Gianneschi, N.C.; Tiekink, E.R.T.; Rendina, L.M. Dinuclear platinum complexes with hydrogen-bonding functionality: Noncovalent assembly of nanoscale cyclic arrays. J. Am. Chem. Soc. 2000, 122, 8474–8479. [Google Scholar] [CrossRef]
- Marshall, L.J.; de Mendoza, J. Self-assembled squares and triangles by simultaneous hydrogen bonding and metal coordination. Org. Lett. 2013, 15, 1548–1551. [Google Scholar] [CrossRef] [PubMed]
- Yagai, S.; Mahesh, S.; Kikkawa, Y.; Unoike, K.; Karatsu, T.; Kitamura, A.; Ajayaghosh, A. Toroidal Nanoobjects from rosette assemblies of melamine-linked oligo(p-phenyleneethynylene)s and cyanurates. Angew. Chem. Int. Ed. 2008, 47, 4691–4694. [Google Scholar] [CrossRef]
- Petelski, A.N.; Peruchena, N.M.; Sosa, G.L. Evolution of the hydrogen-bonding motif in the melamine-cyanuric acid co-crystal: A topological study. J. Mol. Model. 2016, 22, 202. [Google Scholar] [CrossRef]
- Adhikari, B.; Lin, X.; Yamauchi, M.; Ouchi, H.; Aratsu, K.; Yagai, S. Hydrogen-bonded rosettes comprising π-conjugated systems as building blocks for functional one-dimensional assemblies. Chem. Commun. 2017, 53, 9663–9683. [Google Scholar] [CrossRef] [PubMed]
- Ferigán, B.; Folcia, C.L.; Termine, R.; Golemme, A.; Granadino-Roldán, J.M.; Navarro, A.; Serrano, J.L.; Giménez, R.; Sierra, T. Inspecting the electronic architecture and semiconducting properties of a rosette-like supramolecular columnar liquid crystal. Chem. Eur. J. 2018, 24, 17459–17463. [Google Scholar] [CrossRef]
- Igci, C.; Karaman, O.; Fan, Y.; Gonzales III, A.A.; Fenniri, H.; Gumbas, G. Synthesis of N-bridged pyrido[4,3-d]pyrimidines and self-assembly into twin rosette cages and nanotubes in organic media. Sci. Rep. 2018, 8, 15949. [Google Scholar] [CrossRef] [Green Version]
- Whitesides, G.M.; Simanek, E.E.; Mathias, J.P.; Seto, C.T.; Chin, D.N.; Mammen, M.; Gordon, D.M. Noncovalent synthesis: Using physical-organic chemistry to make aggregates. Acc. Chem. Res. 1995, 28, 37–44. [Google Scholar] [CrossRef]
- Zhang, F.; Therrien, B. Coordination of piano-stool complexes to a hydrogen-bonded rosette-type assembly. Eur. J. Inorg. Chem. 2017, 3214–3221. [Google Scholar] [CrossRef]
- Zhang, F.; Therrien, B. Using hydrogen-bonded rosette-type scaffold to generate heteronuclear metalla-assemblies. Eur. J. Inorg. Chem. 2018, 2399–2407. [Google Scholar] [CrossRef]
- Müller, J. Nucleic acid duplexes with metal-mediated base pairs and their structures. Coord. Chem. Rev. 2019, 393, 37–47. [Google Scholar] [CrossRef]
- Park, K.S.; Jung, C.; Park, H.G. “Illusionary” polymerase activity triggered by metal ions: Use for molecular logic-gate operations. Angew. Chem. Int. Ed. 2010, 49, 9757–9760. [Google Scholar] [CrossRef]
- Ono, A.; Togashi, H. Highly selective oligonucleotide-based sensor for mercury (II) in aqueous solutions. Angew. Chem. Int. Ed. 2004, 43, 4300–4302. [Google Scholar] [CrossRef]
- Kobayashi, T.; Takezawa, Y.; Sakamoto, A.; Shionoya, M. Enzymatic synthesis of ligand-bearing DNAs for metal-mediated base pairing utilizing a template-independent polymerase. Chem. Commun. 2016, 52, 3762–3765. [Google Scholar] [CrossRef] [PubMed]
- Hirao, I.; Kimoto, M.; Yamashige, R. Natural versus artificial creation of base pairs in DNA: Origin of nucleobases from the perspectives of unnatural base pair studies. Acc. Chem. Res. 2012, 45, 2055–2065. [Google Scholar] [CrossRef] [PubMed]
- Takezawa, Y.; Shionoya, M. Metal-mediated DNA base pairing: Alternatives to hydrogen-bonded Watson-Crick base pairs. Acc. Chem. Res. 2012, 45, 2066–2076. [Google Scholar] [CrossRef] [PubMed]
- Scharf, P.; Müller, J. Nucleic acids with metal-mediated base pairs and their applications. ChemPlusChem 2013, 78, 20–34. [Google Scholar] [CrossRef]
- Yamanaka, M.; Toyoda, N.; Kobayashi, K. Hybrid cavitand capsule with hydrogen bonds and metal-ligand coordination bonds: Guest encapsulation with anion assistance. J. Am. Chem. Soc. 2009, 131, 9880–9881. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, M.; Kawaharada, M.; Nito, Y.; Takaya, H.; Kobayashi, K. Structural alteration of hybrid supramolecular capsule induced by guest encapsulation. J. Am. Chem. Soc. 2011, 133, 16650–16656. [Google Scholar] [CrossRef] [PubMed]
- Nito, Y.; Adachi, H.; Toyoda, N.; Takaya, H.; Kobayashi, K.; Yamanaka, M. Hydrogen-bond and metal-ligand coordination bond hybrid supramolecular capsules: Identification of hemicapsular intermediate and dual control of guest exchange dynamics. Chem. Asian J. 2014, 9, 1076–1082. [Google Scholar] [CrossRef]
- Pasquale, S.; Sattin, S.; Escudero-Adán, E.C.; Martínez-Belmonte, M.; de Mendoza, J. Giant regular polyhedral from calixarene carboxylates and uranyl. Nat. Commun. 2012, 3, 785. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Zhou, X.; Yuan, W.; Su, X.; Neniškis, A.; Wei, X.; Taujenis, L.; Snarskis, G.; Ward, J.S.; Rissanen, K.; et al. Selective formation of S4- and T-symmetric supramolecular tetrahedral cages and helicates in polar media assembled via cooperative action of coordination and hydrogen bonds. J. Am. Chem. Soc. 2020, 142, 3658–3670. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Therrien, B. Combining Coordination and Hydrogen Bonds to Develop Discrete Supramolecular Metalla-Assemblies. Chemistry 2020, 2, 565-576. https://doi.org/10.3390/chemistry2020034
Therrien B. Combining Coordination and Hydrogen Bonds to Develop Discrete Supramolecular Metalla-Assemblies. Chemistry. 2020; 2(2):565-576. https://doi.org/10.3390/chemistry2020034
Chicago/Turabian StyleTherrien, Bruno. 2020. "Combining Coordination and Hydrogen Bonds to Develop Discrete Supramolecular Metalla-Assemblies" Chemistry 2, no. 2: 565-576. https://doi.org/10.3390/chemistry2020034
APA StyleTherrien, B. (2020). Combining Coordination and Hydrogen Bonds to Develop Discrete Supramolecular Metalla-Assemblies. Chemistry, 2(2), 565-576. https://doi.org/10.3390/chemistry2020034