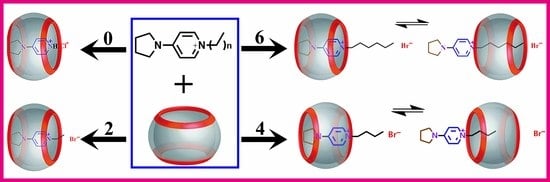
A Study of the Interaction between Cucurbit[7]uril and Alkyl Substituted 4-Pyrrolidinopyridinium Salts
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
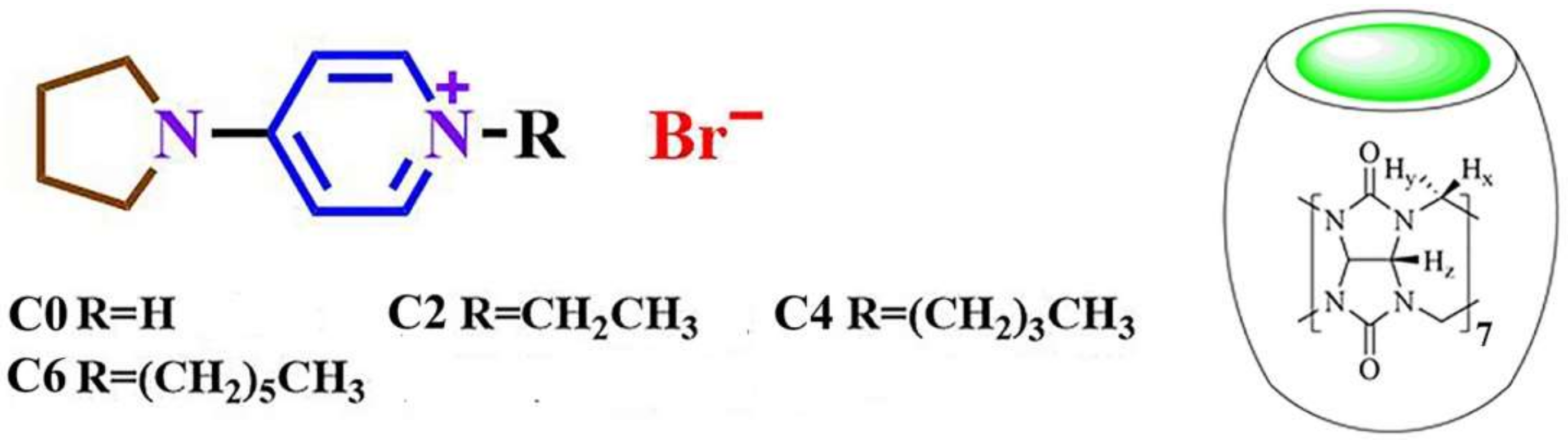
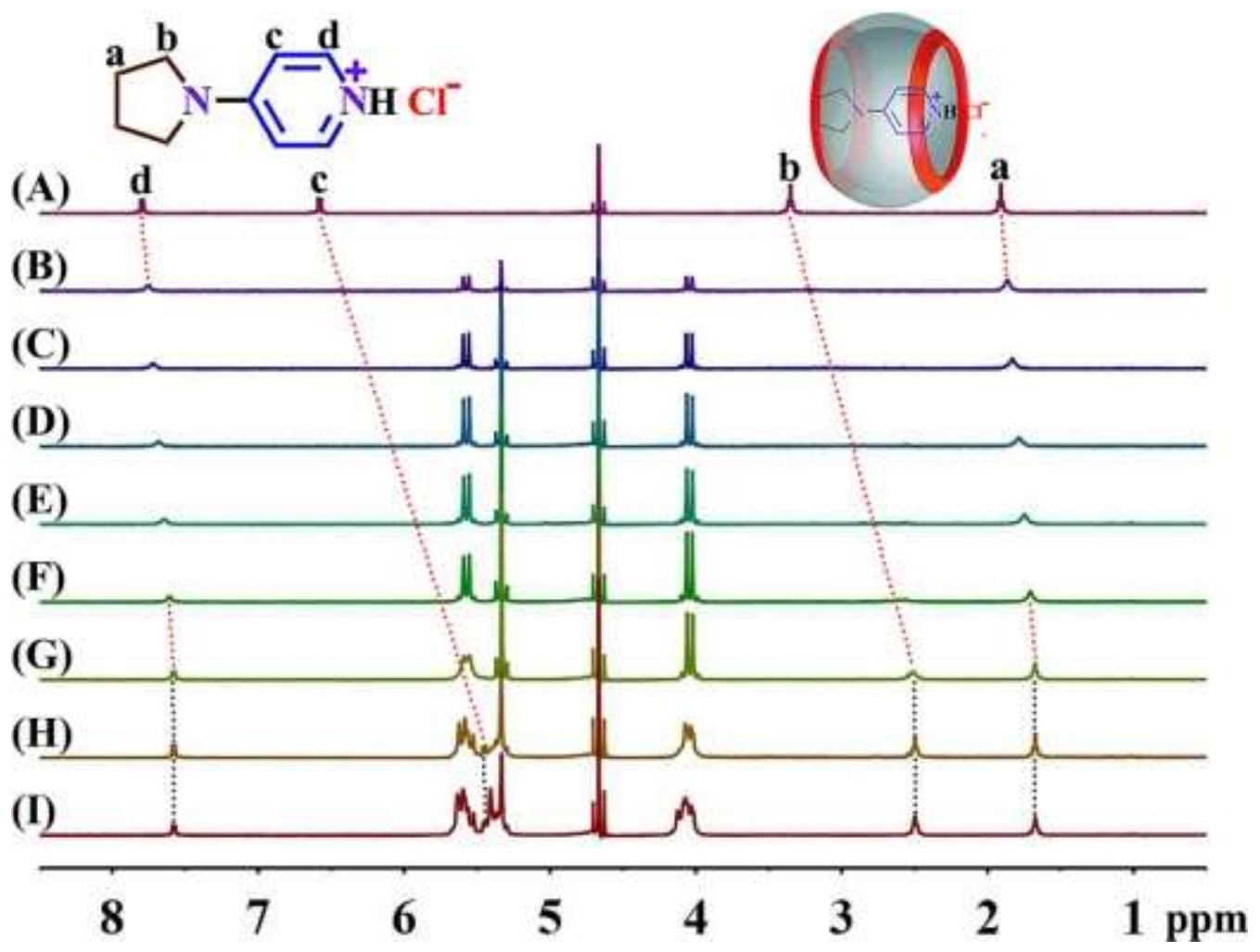
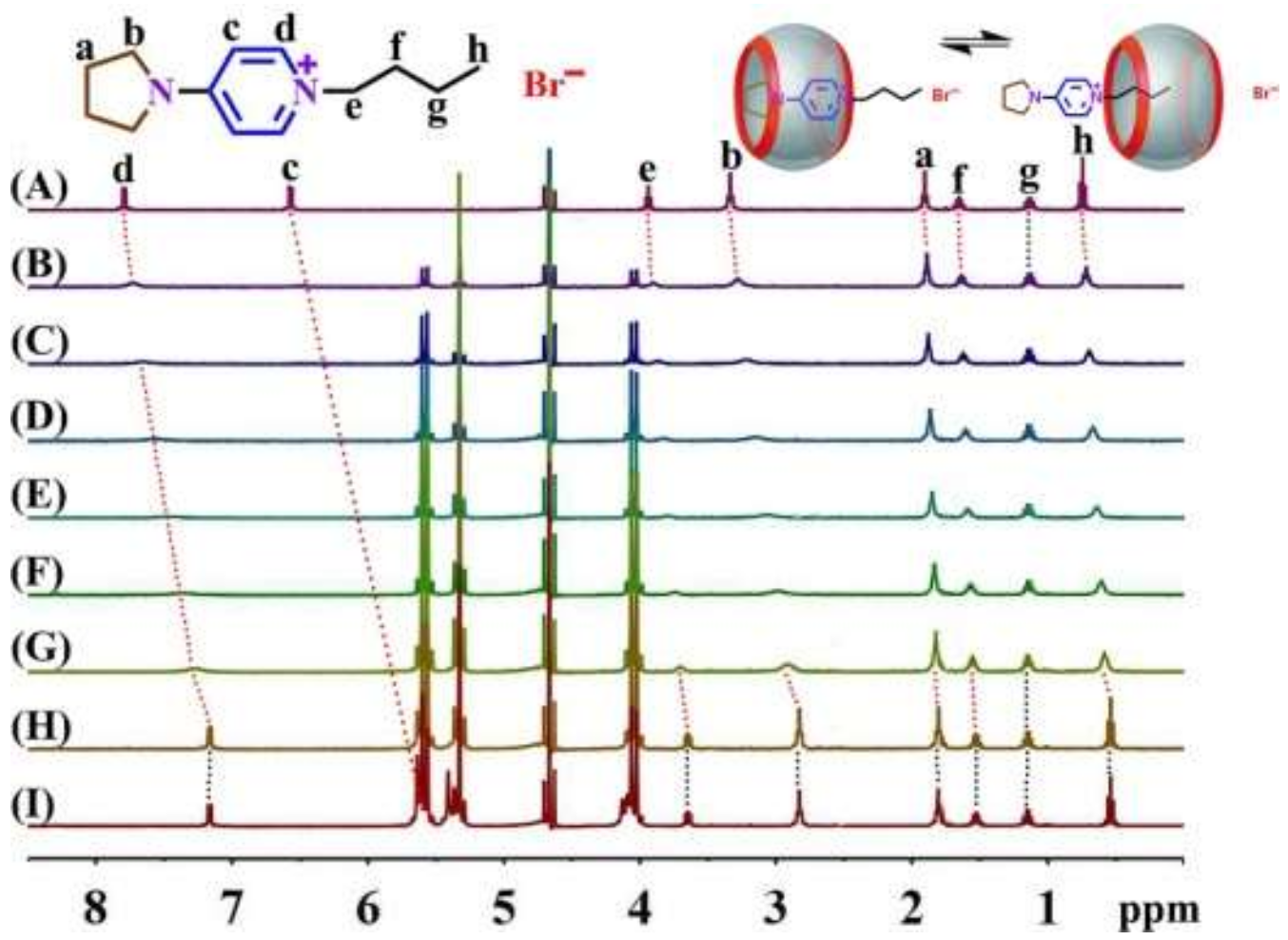
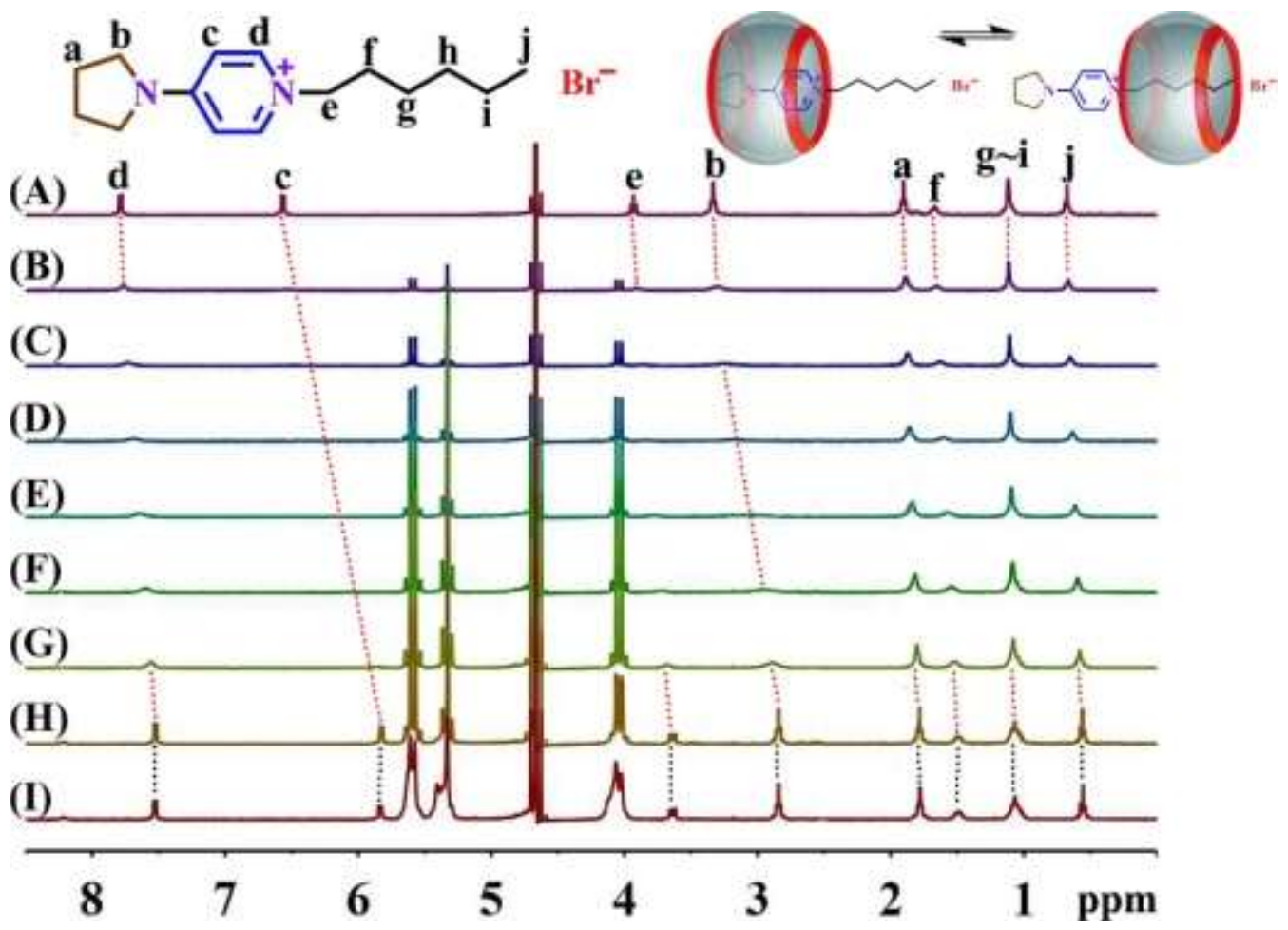
3.1. NMR Spectroscopy
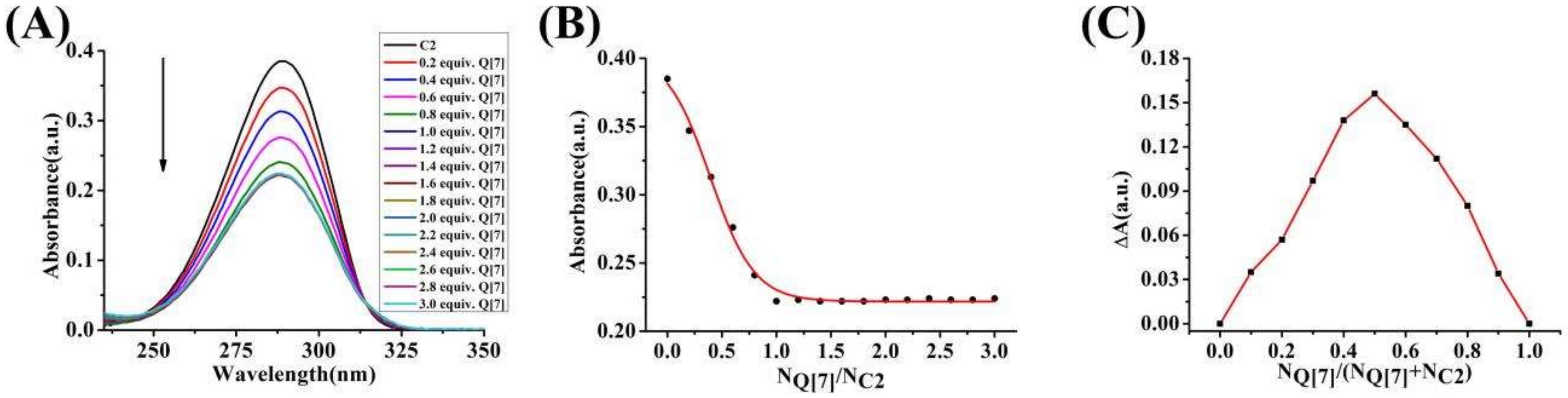
3.2. Ultraviolet (UV) Spectroscopy
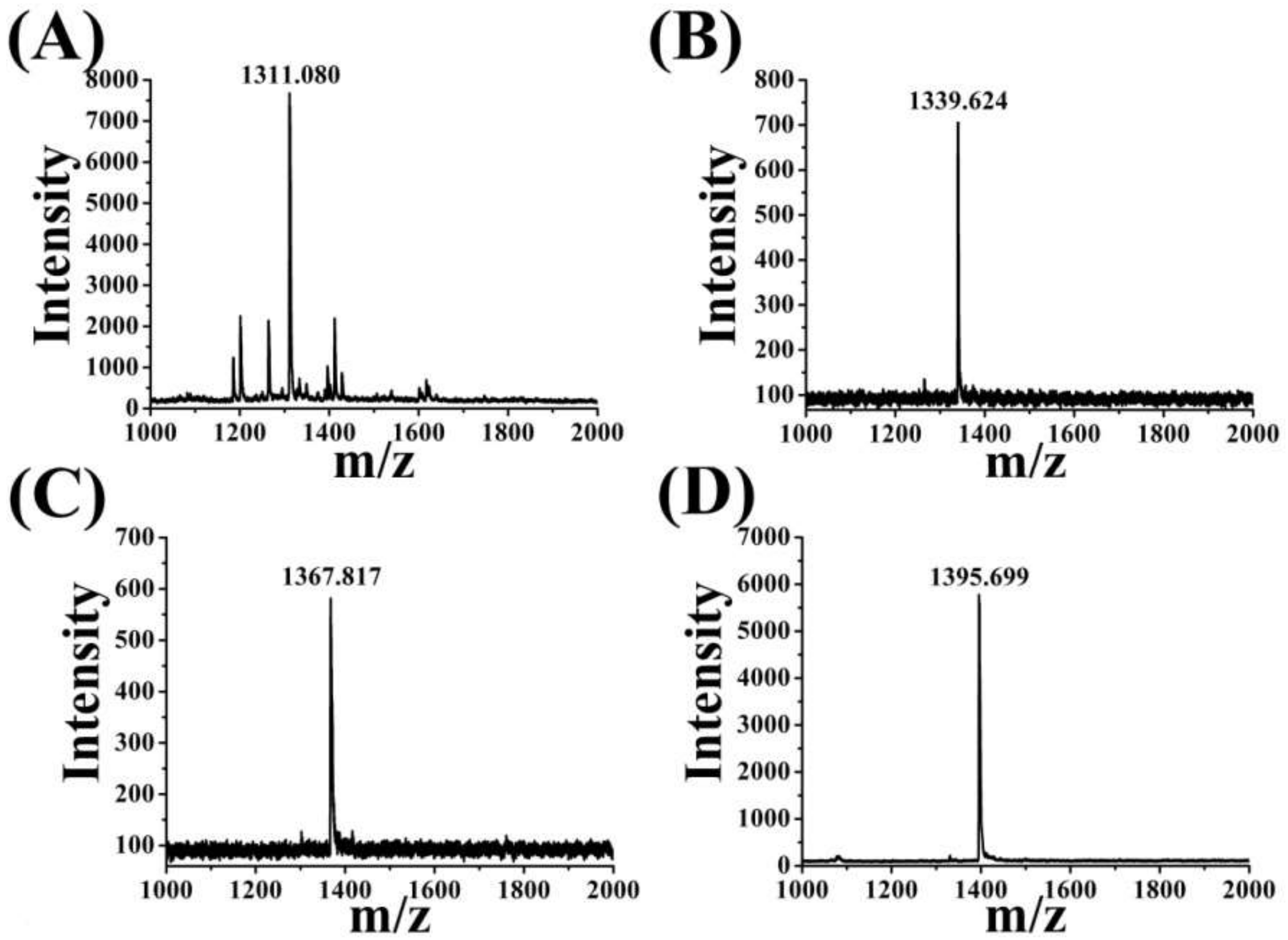
3.3. Mass Spectrometry
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A

| Hd | Hc | Hb | Ha | |
|---|---|---|---|---|
| C0 | 7.80 | 6.58 | 3.35 | 1.91 |
| 0.191 Q[7] | 7.75 | \ | \ | 1.86 |
| 0.326 Q[7] | 7.72 | \ | \ | 1.83 |
| 0.487 Q[7] | 7.68 | \ | \ | 1.79 |
| 0.663 Q[7] | 7.64 | \ | \ | 1.75 |
| 0.838 Q[7] | 7.61 | \ | 2.60 | 1.70 |
| 0.930 Q[7] | 7.58 | \ | 2.51 | 1.67 |
| 1.570 Q[7] | 7.58 | 5.45 | 2.50 | 1.67 |
| 1.939 Q[7] | 7.58 | 5.45 | 2.50 | 1.67 |
| Hd | Hc | He | Hb | Ha | Hf | |
|---|---|---|---|---|---|---|
| C2 | 7.81 | 6.57 | 3.97 | 3.34 | 1.90 | 1.28 |
| 0.133 Q[7] | 7.78 | \ | 3.95 | 3.31 | 1.88 | 1.28 |
| 0.273 Q[7] | \ | \ | 3.93 | \ | 1.86 | 1.28 |
| 0.453 Q[7] | \ | \ | 3.90 | \ | 1.83 | 1.28 |
| 0.595 Q[7] | \ | \ | 3.87 | \ | 1.81 | 1.28 |
| 0.755 Q[7] | 7.32 | \ | 3.85 | 2.69 | 1.79 | 1.28 |
| 0.926 Q[7] | 7.30 | 5.43 | 3.84 | 2.66 | 1.77 | 1.28 |
| 1.415 Q[7] | 7.30 | \ | 3.84 | 2.66 | 1.77 | 1.28 |
| 1.733 Q[7] | 7.30 | \ | 3.84 | 2.66 | 1.77 | 1.28 |

| Hd | Hc | He | Hb | Ha | Hf | Hg | Hh | |
|---|---|---|---|---|---|---|---|---|
| C4 | 7.79 | 6.57 | 3.94 | 3.33 | 1.90 | 1.65 | 1.13 | 0.75 |
| 0.116 Q[7] | 7.73 | \ | 3.90 | 3.28 | 1.89 | 1.63 | 1.13 | 0.72 |
| 0.173 Q[7] | 7.65 | \ | 3.86 | 3.22 | 1.87 | 1.62 | 1.14 | 0.69 |
| 0.331 Q[7] | 7.56 | \ | 3.83 | 3.14 | 1.86 | 1.60 | 1.14 | 0.67 |
| 0.474 Q[7] | 7.46 | \ | 3.78 | 3.06 | 1.85 | 1.58 | 1.14 | 0.64 |
| 0.641 Q[7] | 7.36 | \ | 3.74 | 2.98 | 1.83 | 1.57 | 1.14 | 0.60 |
| 0.750 Q[7] | 7.27 | \ | 3.71 | 2.91 | 1.82 | 1.55 | 1.15 | 0.58 |
| 1.164 Q[7] | 7.16 | \ | 3.64 | 2.82 | 1.80 | 1.53 | 1.15 | 0.54 |
| 1.653 Q[7] | 7.16 | 5.45 | 3.64 | 2.82 | 1.80 | 1.53 | 1.15 | 0.54 |
| Hd | Hc | He | Hb | Ha | Hf | Hg-i | Hj | |
|---|---|---|---|---|---|---|---|---|
| C6 | 7.79 | 6.56 | 3.93 | 3.33 | 1.90 | 1.67 | 1.12 | 0.68 |
| 0.110 Q[7] | 7.76 | 6.54 | 3.91 | 3.30 | 1.89 | 1.65 | 1.11 | 0.66 |
| 0.223 Q[7] | 7.72 | \ | 3.87 | 3.26 | 1.87 | 1.63 | 1.11 | 0.65 |
| 0.363 Q[7] | 7.68 | \ | 3.82 | 3.19 | 1.85 | 1.60 | 1.10 | 0.63 |
| 0.461 Q[7] | 7.64 | \ | 3.77 | 3.04 | 1.84 | 1.57 | 1.09 | 0.62 |
| 0.646 Q[7] | 7.59 | \ | 3.72 | 2.95 | 1.82 | 1.55 | 1.08 | 0.60 |
| 0.739 Q[7] | 7.56 | 5.86 | 3.68 | 2.89 | 1.80 | 1.52 | 1.08 | 0.58 |
| 0.997 Q[7] | 7.52 | 5.81 | 3.64 | 2.84 | 1.78 | 1.49 | 1.07 | 0.56 |
| 1.494 Q[7] | 7.52 | 5.81 | 3.64 | 2.84 | 1.78 | 1.49 | 1.07 | 0.56 |







References
- Zhang, W.; Luo, Y.; Zhou, Y.; Liu, M.; Xu, W.; Bian, B.; Tao, Z.; Xin, X. A highly selective fluorescent chemosensor probe for the detection of Fe3+ and Ag+ based on supramolecular assembly of cucubit[10]uril with a pyrene derivative. Dyes Pigment. 2020, 176, 108235–108242. [Google Scholar] [CrossRef]
- Lucenti, E.; Forni, A.; Botta, C.; Carlucci, L.; Giannini, C.; Marinotto, D.; Previtali, A.; Righetto, S.; Cariati, E. H-aggregates granting crystallization-induced emissive behavior and ultralong phosphorescence from a pure organic molecule. J. Phys. Chem. Lett. 2017, 8, 1894–1898. [Google Scholar] [CrossRef] [Green Version]
- Bian, L.; Shi, H.; Wang, X.; Ling, K.; Ma, H.; Li, M.; Cheng, Z.; Ma, C.; Cai, S.; Wu, Q.; et al. Simultaneously enhancing efficiency and lifetime of ultralong organic phosphorescence materials by molecular self-assembly. J. Am. Chem. Soc. 2018, 140, 10734–10739. [Google Scholar] [CrossRef] [PubMed]
- You, L.; Zha, D.; Anslyn, E.V. Recent advances in supramolecular analytical chemistry using optical sensing. Chem. Rev. 2015, 115, 7840–7892. [Google Scholar] [CrossRef] [PubMed]
- Dale, E.J.; Vermeulen, N.A.; Juricek, M.; Barnes, J.C.; Young, R.M.; Wasielewski, M.R.; Stoddart, J.F. Supramolecular Ex plorations: Ex hibiting the Ex tent of Ex tended cationic cyclophanes. Acc. Chem. Res. 2016, 49, 262–273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barrow, S.J.; Kasera, S.; Rowland, M.J.; del Barrio, J.; Scherman, O.A. Cucurbituril-based molecular recognition. Chem. Rev. 2015, 115, 12320–12406. [Google Scholar] [CrossRef] [Green Version]
- Yu, G.; Jie, K.; Huang, F. Supramolecular amphiphiles based on host–guest molecular recognition motifs. Chem. Rev. 2015, 115, 7240–7303. [Google Scholar] [CrossRef]
- Wu, M.X.; Yang, Y.W. Metal–organic framework (MOF)-based drug/cargo delivery and cancer therapy. Adv. Mater. 2017, 29, 1606134. [Google Scholar] [CrossRef]
- Diederich, F. Complexation of neutral molecules by cyclophane hosts. Angew. Chem. Int. Ed. Engl. 1988, 27, 362–386. [Google Scholar] [CrossRef]
- Xue, M.; Yang, Y.; Chi, X.; Zhang, Z.; Huang, F. Pillararenes, a new class of macrocycles for supramolecular chemistry. Acc. Chem. Res. 2012, 45, 1294–1308. [Google Scholar] [CrossRef]
- Rebek, J. Molecular behavior in small spaces. Acc. Chem. Res. 2009, 42, 1660–1668. [Google Scholar] [CrossRef] [PubMed]
- Zarra, S.; Wood, D.M.; Roberts, D.A.; Nitschke, J.R. Molecular containers in complex chemical systems. Chem. Soc. Rev. 2015, 44, 419–432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szente, L.; Szejtli, J. Highly soluble cyclodextrin derivatives: Chemistry, properties, and trends in development. Adv. Drug Delivery Rev. 1999, 36, 17–28. [Google Scholar] [CrossRef]
- Pedersen, C.J. The discovery of crown ethers (Noble Lecture). Angew. Chem. Int. Ed. Engl. 1988, 27, 1021–1027. [Google Scholar] [CrossRef]
- Masson, E.; Ling, X.; Roymon, J.; Kyeremeh-Mensah, L.; Lu, X. Cucurbituril chemistry: A tale of supramolecular success. RSC Adv. 2012, 2, 1213–1247. [Google Scholar] [CrossRef]
- Kim, K. Mechanically interlocked molecules incorporating cucurbituril and their supramolecular assemblies. Chem. Soc. Rev. 2002, 31, 96–107. [Google Scholar] [CrossRef] [Green Version]
- Assaf, K.I.; Nau, W.M. Cucurbiturils: From synthesis to high-affinity binding and catalysis. Chem. Soc. Rev. 2015, 44, 394–418. [Google Scholar] [CrossRef] [Green Version]
- Lagona, J.; Mukhopadhyay, P.; Chakrabarti, S.; Isaacs, L. The cucurbit[n]uril family. Angew. Chem. Int. Ed. 2005, 44, 4844–4870. [Google Scholar] [CrossRef]
- Gomez-Casado, A.; Jonkheijm, P.; Huskens, J. Recognition properties of cucurbit[7]uril self-assembled monolayers studied with force spectroscopy. Langmuir 2011, 27, 11508–11513. [Google Scholar] [CrossRef]
- Bosmans, R.P.; Briels, J.M.; Milroy, L.G.; de Greef, T.F.; Merkx, M.; Brunsveld, L. Supramolecular Control over Split-Luciferase Complementation. Angew. Chem. Int. Ed. 2016, 55, 8899–8903. [Google Scholar] [CrossRef] [Green Version]
- Mock, W.L.; Shih, N.Y. Host-guest binding capacity of cucurbituril. J. Org. Chem. 1983, 48, 3618. [Google Scholar] [CrossRef]
- Walker, S.; Oun, R.; McInnes, F.J.; Wheate, N.J. The potential of cucurbit[n]urils in drug delivery. Isr. J. Chem. 2011, 51, 616–624. [Google Scholar] [CrossRef]
- Xu, W.; Kan, J.; Yang, B.; Prior, T.J.; Bian, B.; Xiao, X.; Tao, Z.; Redshaw, C. A Study of the Interaction Between Cucurbit[8]uril and Alkyl-Substituted 4-Pyrrolidinopyridinium Salts. Chem. Asian J. 2019, 14, 235–242. [Google Scholar] [CrossRef] [Green Version]
- Xu, W.; Liu, M.; Escanỡ, M.C.; Redshaw, C.; Bian, B.; Fan, Y.; Tao, Z.; Xiao, X. Alkyl substituted 4-pyrrolidinopyridinium salts encapsulated in the cavity of cucurbit[10]uril. New J. Chem. 2019, 43, 7028–7034. [Google Scholar] [CrossRef]
- Ji, L.; Yang, L.; Yu, Z.Y.; Tan, C.S.; Parker, R.M.; Abell, C.; Scherman, O.A. Cucurbit[n]uril-based microcapsules self-assembled within microfluidic droplets: A versatile approach for supramolecular architectures and materials. Accounts Chem. Res. 2017, 50, 208–217. [Google Scholar]
- Yamanaka, M.; Yoshida, U.; Sato, M.; Shigeta, T.; Yoshida, K.; Furuta, T.; Kawabata, T.J. Origin of high E-selectivity in 4-pyrrolidinopyridine-catalyzed tetrasubstituted α, α′-alkenediol: A computational and experimental study. Org. Chem. 2015, 80, 3075–3082. [Google Scholar] [CrossRef] [PubMed]
- Sammaki, T.; Hurley, T.B. 2-formyl-4-pyrrolidinopyridine (FPP): A new catalyst for the hydroxyl-directed methanolysis of esters. J. Am. Chem. Soc. 1996, 118, 8967–8968. [Google Scholar] [CrossRef]
- Nguyen, H.V.; Butler, D.C.; Richards, C.J. A metallocene-pyrrolidinopyridine nucleophilic catalyst for asymmetric synthesis. Org. Lett. 2006, 8, 769–772. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, W.; Zhu, X.; Bian, B.; Xiao, X.; Tao, Z.; Redshaw, C. A Study of the Interaction between Cucurbit[7]uril and Alkyl Substituted 4-Pyrrolidinopyridinium Salts. Chemistry 2020, 2, 262-273. https://doi.org/10.3390/chemistry2020017
Xu W, Zhu X, Bian B, Xiao X, Tao Z, Redshaw C. A Study of the Interaction between Cucurbit[7]uril and Alkyl Substituted 4-Pyrrolidinopyridinium Salts. Chemistry. 2020; 2(2):262-273. https://doi.org/10.3390/chemistry2020017
Chicago/Turabian StyleXu, Weitao, Xinyi Zhu, Bing Bian, Xin Xiao, Zhu Tao, and Carl Redshaw. 2020. "A Study of the Interaction between Cucurbit[7]uril and Alkyl Substituted 4-Pyrrolidinopyridinium Salts" Chemistry 2, no. 2: 262-273. https://doi.org/10.3390/chemistry2020017