Requirements and Architecture of a Cloud Based Insomnia Therapy and Diagnosis Platform: A Smart Cities Approach
Abstract
1. Introduction
2. Background
3. Methods
Requirements Analysis
4. Requirements
4.1. Medical Data Collection
4.2. Patient-Reported Outcomes: ESM/EMA
4.3. Technology-Reported Outcomes
4.4. Smartphones and Smart Meters
4.5. Aetiology and Contextual Factors Influencing Insomnia
4.6. Technical Requirements: Data Exchange and Storage
- Patient for the mapping of the patient;
- Practitioner for the mapping of the HSP;
- Questionnaire for the mapping of the questionnaires and sleep diaries;
- QuestionnaireResponse for the mapping of responses;
- CarePlan for the mapping of the dCBT-I;
- Procedure for the mapping of a therapy step.
4.7. Smart City Requirements
5. Results
5.1. System Requirements
- Functionality and performance;
- Interfaces between the software system and other systems;
- Data security;
- User interfaces;
- Database definition and requirements.
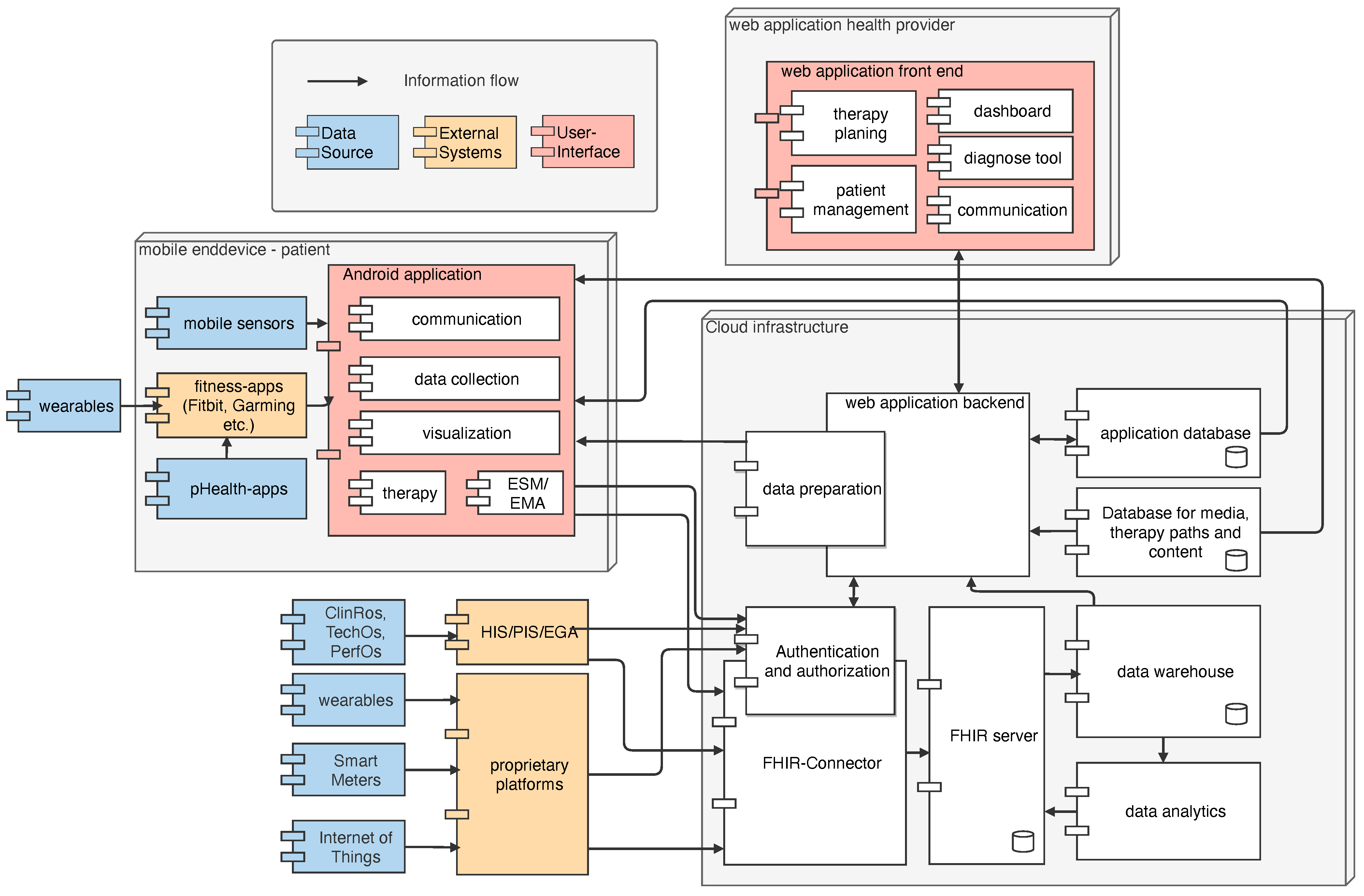
5.2. Software Architecture
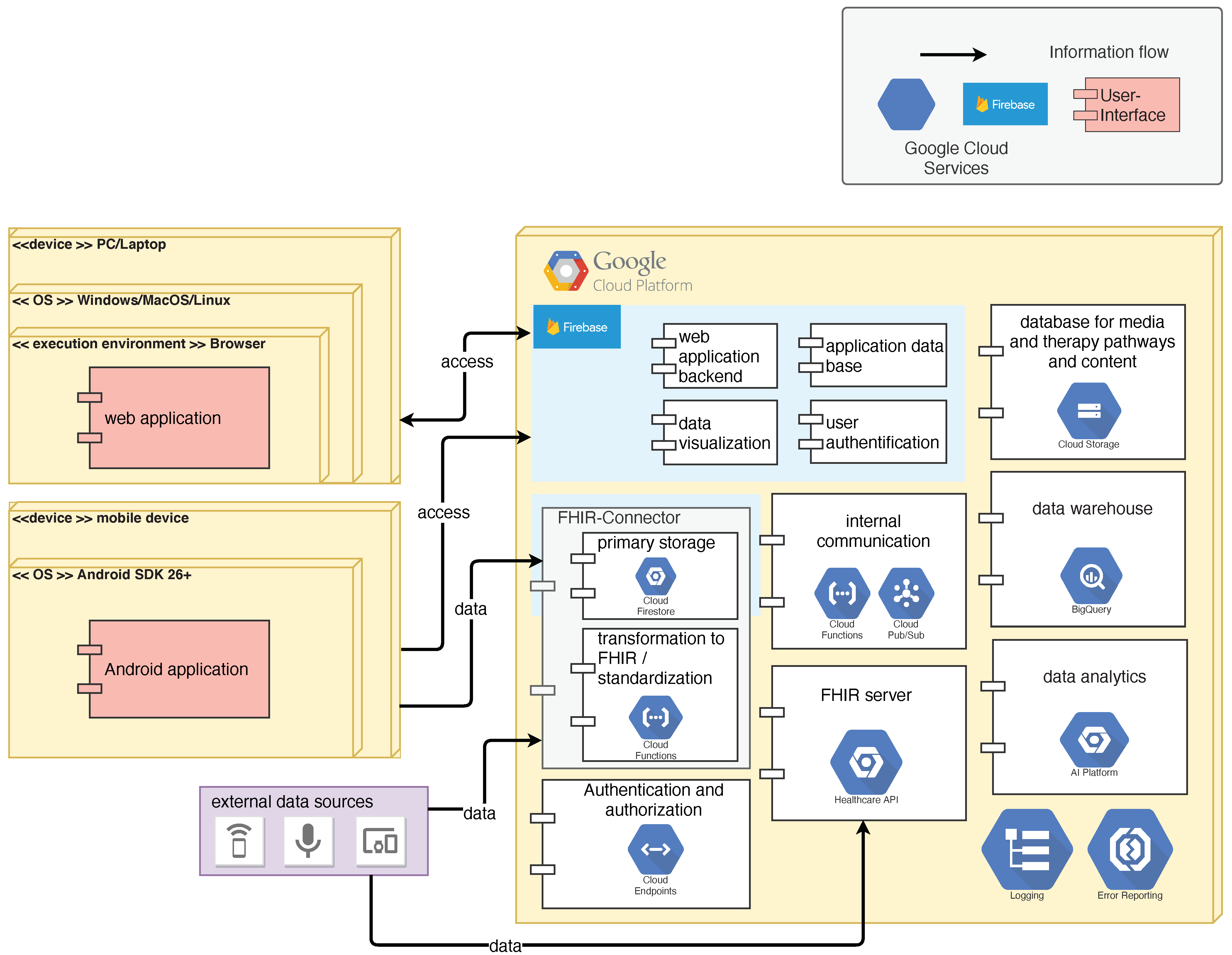
5.3. Implementation Concept
6. Discussion
- Determination of user requirements according to the specifications of ISO 9241-110:2020 (Ergonomics of human-system interaction—Interaction principles);
- Clarification of the aspects of data protection and security, as well as ensuring GDPR conformity, cf. [94];
- Complete profiling of FHIR resources;
- Analysis, evaluation, and selection of cloud service providers, cf. [95];
- Implementation of a prototype for the platform, including cloud, mobile, and web component;
- Detailed cost planning;
- Pilot operation and usability testing.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| API | Application Programming Interface |
| AWMF | Arbeitsgemeinschaft der Wissenschaftlichen Medizinischen Fachgesellschaften |
| (d)CBT-I | (digital) Cognitive Behavioural Therapy for Insomnia |
| CDSS | Clinical Decision Support System |
| DGSM | Deutsche Gesellschaft für Schlafforschung und Schlafmedizin |
| DSM | Diagnostic and Statistical Manual of Mental Disorders |
| EHR | Electronic Health Record |
| EMA | Ecological Momentary Assessment |
| ESM | Experience Sampling Method |
| FHIR | Fast Healthcare Interoperability Resources |
| GCP | Google Cloud Platform |
| GDPR | General Data Protection Regulation |
| HSP | Health Service Provider |
| ICD | International Statistical Classification of Diseases and Related Health Problems |
| IHE | Integrating the Healthcare Enterprise |
| MDR | Medical Device Regulation |
| ÖGSM | Österreichische Gesellschaft für Schlafmedizin und Schlafforschung |
| PHR | Personal Health Record |
| PROs | Patient Reported Outcomes |
| REST | Respresentational State Transfer |
| SaMD | Software as Medical Device |
| SOL | Sleep Onset Latency |
| TechROs | Technology Reported Outcomes |
| TST | Total Sleep Time |
| WASO | Wake After Sleep Onset |
Appendix A. System Requirements
Appendix A.1. Requirements for Functionality and Performance
- 1.
- The system is operated entirely in the cloud by using commercially available services of a cloud provider. Exception: The access point for patients is a native mobile application.
Appendix A.2. Interfaces between the Software System and Other Systems
- 2.
- The system provides a REST interface to submit FHIR resources to the system after successful authorisation;
- 3.
- The system provides a generic API for the transmission of proprietary data formats for other data sources that do not have FHIR functionality;
- 4.
- The system can send requests to Google’s Fused Location Provider API to retrieve the user’s location data;
- 5.
- The system can make requests to Google’s Fit API and thus access activity and lifestyle data;
- 6.
- The system can call the Android system methods BatteryManager and UsageStatsManager to access battery and usage data;
- 7.
- The system receives further data via a public REST API.
Appendix A.3. Requirements for Data Security
- 8.
- The system allows only authorised patients to access the Android application. Users can register themselves and are confirmed by the HSP. Registration is completed by entering an e-mail address and password. Only then can the data collection be started;
- 9.
- The system only allows authorised HSPs to access the web application;
- 10.
- User accounts for HSPs are created and activated by an administrator;
- 11.
- The system only allows authorised systems to transfer data to the system. Authorisation is completed using OAuth 2.0;
- 12.
- The system has functions that allow all administrative activities and accesses to be logged.
Appendix A.4. User Interface Requirements Implemented by Software
- 13.
- The system provides a method for authentication and authorisation of the patient via email and password;
- 14.
- The system allows access to other functions only when the user has been successfully authorised and confirmed;
- 15.
- The system allows the patient to activate and deactivate the collection of personal data depending on the type of data;
- 16.
- The system only collects and transmits personal data if the user agrees to the collection and the collection of the respective data has been activated;
- 17.
- The system allows users to communicate with the relevant HSP via a secure communication channel;
- 18.
- The system offers functions that enable the patient to carry out a digital CBT-I on his or her own responsibility. For this purpose, the system accesses predefined therapy procedures that are assigned by the HSP;
- 19.
- The system logs the user’s therapy progress;
- 20.
- The web application allows a HSP to authenticate and thus log in to the system via email and password;
- 21.
- The system clearly displays all users of a HSP in a list;
- 22.
- The system provides an overview of a patient’s data. This includes status (registered, confirmed, data collection started—diagnosed—in therapy—therapy completed—treatment episode completed), personal data and key figures based on collected data (ClinROs, PROs, TEchROs);
- 23.
- The system allows the HSP to view all raw data collected;
- 24.
- The system enables the HSP to exchange messages with the patient via a secure communication channel;
- 25.
- The system provides an overview of the diagnostic criteria for non-organic insomnia according to DSM-5 and implements a workflow based on the five diagnostic steps. The system is used exclusively for documentation;
- 26.
- The system makes it possible to assign a patient to a previously defined therapy sequence;
- 27.
- The system makes it possible to ask patients to fill in questionnaires or sleep diaries, either once or at regular intervals;
- 28.
- The system allows to inform an administrator about problems that occur.
Administrative Interface
- 29.
- The system can be manually adjusted by administrators;
- 30.
- For manual customisation, tools from the selected cloud provider can be used if available. No additional user interface needs to be designed;
- 31.
- System administrators can create and manage accounts for HSPs;
- 32.
- If available, the identity access management tool of the cloud provider must be used. Roles and permissions are to be assigned according to the principle of minimal rights.
Appendix A.5. Data Definition and Database Requirements
- 33.
- FHIR profiles are specified against FHIR resources for mapping the data;
- 34.
- The system can receive inhomogenous raw data and convert it into standardised FHIR resources (version R4);
- 35.
- The system receives and stores FHIR resources (version R4) on a FHIR server;
- 36.
- The system stores CBT-I media content in a database;
- 37.
- The system stores textual content of the CBT-I and other static data in JSON format;
- 38.
- The system stores user settings and other details in a database;
- 39.
- The system implements an authorisation system to prevent unauthorised access to external data;
- 40.
- The system transfers standardised FHIR resources to a data warehouse;
- 41.
- The system sends calculation results of the machine learning component to the FHIR server.
References
- Ali, O.; Shrestha, A.; Soar, J.; Wamba, S.F. Cloud computing-enabled healthcare opportunities, issues, and applications: A systematic review. Int. J. Inf. Manag. 2018, 43, 146–158. [Google Scholar] [CrossRef]
- Tyagi, S.; Agarwal, A.; Maheshwari, P. A conceptual framework for IoT-based healthcare system using cloud computing. In Proceedings of the 6th International Conference—Cloud System and Big Data Engineering (Confluence), Noida, India, 14–15 January 2016; pp. 503–507. [Google Scholar] [CrossRef]
- Darwish, A.; Hassanien, A.E.; Elhoseny, M.; Sangaiah, A.K.; Muhammad, K. The impact of the hybrid platform of internet of things and cloud computing on healthcare systems: Opportunities, challenges, and open problems. J. Ambient. Intell. Humaniz. Comput. 2019, 10, 4151–4166. [Google Scholar] [CrossRef]
- Solanas, A.; Patsakis, C.; Conti, M.; Vlachos, I.; Ramos, V.; Falcone, F.; Postolache, O.; Perez-Martinez, P.; Pietro, R.; Perrea, D.; et al. Smart health: A context-aware health paradigm within Smart Cities. IEEE Commun. Mag. 2014, 52, 74–81. [Google Scholar] [CrossRef]
- Haghayegh, S.; Khoshnevis, S.; Smolensky, M.H.; Diller, K.R.; Castriotta, R.J. Performance assessment of new-generation Fitbit technology in deriving sleep parameters and stages. Chronobiol. Int. 2020, 37, 47–59. [Google Scholar] [CrossRef]
- Baron, K.G.; Duffecy, J.; Berendsen, M.A.; Cheung Mason, I.; Lattie, E.G.; Manalo, N.C. Feeling validated yet? A scoping review of the use of consumer-targeted wearable and mobile technology to measure and improve sleep. Sleep Med. Rev. 2018, 40, 151–159. [Google Scholar] [CrossRef]
- Fallmann, S.; Chen, L. Computational Sleep Behavior Analysis: A Survey. IEEE Access 2019, 7, 142421–142440. [Google Scholar] [CrossRef]
- Kang, S.G.; Kang, J.M.; Cho, S.J.; Ko, K.P.; Lee, Y.J.; Lee, H.J.; Kim, L.; Winkelman, J.W. Cognitive Behavioral Therapy Using a Mobile Application Synchronizable With Wearable Devices for Insomnia Treatment: A Pilot Study. J. Clin. Sleep Med. JCSM 2017, 13, 633–640. [Google Scholar] [CrossRef] [PubMed]
- Canale, L. Light Pollution, Blue Light, Stress and Insomnia: Dark Sides of the LED Light? Int. J. Electron. Electr. Eng. Syst. 2020, 3, 21–25. [Google Scholar]
- Xu, J.; Zhou, J.; Luo, P.; Mao, D.; Xu, W.; Nima, Q.; Cui, C.; Yang, S.; Ao, L.; Wu, J.; et al. Associations of long-term exposure to ambient air pollution and physical activity with insomnia in Chinese adults. Sci. Total Environ. 2021, 792, 148197. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, G. An investigation of community response to Urban traffic noise. In Global Perspective for Competitive Enterprise, Economy and Ecology, Proceedings of the 16th ISPE International Conference on Concurrent Engineering, Taipei, Taiwan, 20–24 July 2009; Springer: London, UK, 2009; pp. 673–680. [Google Scholar] [CrossRef]
- Bhaskar, S.; Hemavathy, D.; Prasad, S. Prevalence of chronic insomnia in adult patients and its correlation with medical comorbidities. J. Fam. Med. Prim. Care 2016, 5, 780–784. [Google Scholar] [CrossRef]
- Riemann, D.; Baum, E.; Cohrs, S.; Crönlein, T.; Hajak, G.; Hertenstein, E.; Klose, P.; Langhorst, J.; Mayer, G.; Nissen, C.; et al. S3-Leitlinie Nicht erholsamer Schlaf/Schlafstörungen: Kapitel „Insomnie bei Erwachsenen“ (AWMF-Registernummer 063-003), Update 2016. Somnologie 2017, 21, 2–44. [Google Scholar] [CrossRef]
- Mayer, G.; Fietze, I.; Fischer, J.; Penzel, T.; Riemann, D.; Rodenbeck, A.; Sitter, H.; Teschler, H. S3-Leitlinie: Nicht erholsamer Schlaf. Somnologie Schlafforschung Schlafmed. 2009, 13, 1–160. [Google Scholar] [CrossRef]
- Blackwell, T.; Paudel, M.; Redline, S.; Ancoli-Israel, S.; Stone, K.L. A novel approach using actigraphy to quantify the level of disruption of sleep by in-home polysomnography: The MrOS Sleep Study: Sleep disruption by polysomnography. Sleep Med. 2017, 32, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Muche-Borowski, C.; Kopp, I. Wie eine Leitlinie entsteht. Z. Herz Thorax Gefäßchirurgie 2011, 25, 217–223. [Google Scholar] [CrossRef]
- Ibáñez, V.; Silva, J.; Cauli, O. A survey on sleep questionnaires and diaries. Sleep Med. 2018, 42, 90–96. [Google Scholar] [CrossRef]
- Rodenbeck, A. Manual der American Academy of Sleep Medicine. Somnologie Schlafforschung Schlafmed. 2013, 17, 122–130. [Google Scholar] [CrossRef][Green Version]
- Kolla, B.P.; Mansukhani, S.; Mansukhani, M.P. Consumer sleep tracking devices: A review of mechanisms, validity and utility. Expert Rev. Med. Devices 2016, 13, 497–506. [Google Scholar] [CrossRef] [PubMed]
- De Zambotti, M.; Rosas, L.; Colrain, I.M.; Baker, F.C. The Sleep of the Ring: Comparison of the ŌURA Sleep Tracker Against Polysomnography. Behav. Sleep Med. 2019, 17, 124–136. [Google Scholar] [CrossRef]
- Sterr, A.; Ebajemito, J.K.; Mikkelsen, K.B.; Bonmati-Carrion, M.A.; Santhi, N.; della Monica, C.; Grainger, L.; Atzori, G.; Revell, V.; Debener, S.; et al. Sleep EEG Derived From Behind-the-Ear Electrodes (cEEGrid) Compared to Standard Polysomnography: A Proof of Concept Study. Front. Hum. Neurosci. 2018, 12, 452. [Google Scholar] [CrossRef]
- Soh, H.L.; Ho, R.C.; Ho, C.S.; Tam, W.W. Efficacy of digital cognitive behavioural therapy for insomnia: A meta-analysis of randomised controlled trials. Sleep Med. 2020, 75, 315–325. [Google Scholar] [CrossRef]
- Hertenstein, E. (Ed.) Prävention und Psychotherapie der Insomnie: Konzepte, Methoden und Praxis der Freiburger Schlafschule, 1 aufl ed.; Störungsspezifische Psychotherapie, Kohlhammer: Stuttgart, Germany, 2015; OCLC: 908643678. [Google Scholar]
- Spiegelhalder, K.; Backhaus, J.; Riemann, D. Schlafstörungen, Number 7 in Fortschritte der Psychotherapie, 2 überarb. aufl ed.; Hogrefe: Göttingen, Germany, 2011; OCLC: 846061458. [Google Scholar]
- Riemann, D. Ratgeber Schlafstörungen: Informationen für Betroffene und Angehörige, 2 aktualisierte auflage ed.; Number 2 in Ratgeber zur Reihe Fortschritte der Psychotherapie; Hogrefe: Göttingen, Germany, 2016; OCLC: 934812597. [Google Scholar]
- Hauschild, J. Endlich schlafen: Der Ratgeber für einen erholsamen Schlaf; Stiftung Warentest: Berlin, Germany, 2015; OCLC: 909794557. [Google Scholar]
- Mikkelsen, K.B.; Tabar, Y.R.; Kappel, S.L.; Christensen, C.B.; Toft, H.O.; Hemmsen, M.C.; Rank, M.L.; Otto, M.; Kidmose, P. Accurate whole-night sleep monitoring with dry-contact ear-EEG. Sci. Rep. 2019, 9, 16824. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.L.; Jahmunah, V.; Arunkumar, N.; Abdulhay, E.W.; Gururajan, R.; Adib, N.; Ciaccio, E.J.; Cheong, K.H.; Acharya, U.R. A novel automated autism spectrum disorder detection system. Complex Intell. Syst. 2021, 1, 1–15. [Google Scholar] [CrossRef]
- Tang, K.J.W.; Ang, C.K.E.; Constantinides, T.; Rajinikanth, V.; Acharya, U.R.; Cheong, K.H. Artificial Intelligence and Machine Learning in Emergency Medicine. Biocybern. Biomed. Eng. 2021, 41, 156–172. [Google Scholar] [CrossRef]
- Zhao, X.; Ang, C.K.E.; Acharya, U.R.; Cheong, K.H. Application of Artificial Intelligence techniques for the detection of Alzheimer’s disease using structural MRI images. Biocybern. Biomed. Eng. 2021, 41, 456–473. [Google Scholar] [CrossRef]
- Akl, A.; Taati, B.; Mihailidis, A. Autonomous Unobtrusive Detection of Mild Cognitive Impairment in Older Adults. IEEE Trans. Bio-Med. Eng. 2015, 62, 1383. [Google Scholar] [CrossRef]
- John, M. Telemedizinische Assistenzsysteme in der Rehabilitation und Nachsorge – Projekte, Technologien und Funktionen. B&G Bewegungstherapie Gesundheitssport 2017, 33, 197–207. [Google Scholar] [CrossRef]
- Tiemann, M.; Michels, H.; Gedwien, A.; Cordes, M.; Clemens, U.; Pisula, D. DigiTrain—Entwicklung und Evaluation eines digitalen Trainingsprogramms zur Rehabilitationsnachsorge bei chronischen Rückenschmerzen. B&G Bewegungstherapie Gesundheitssport 2017, 33, 235–240. [Google Scholar] [CrossRef]
- Riedl, R.; Robausch, M.; Berghold, A. The Evaluation of the Effectiveness of Austrians Disease Management Program in Patients with Type 2 Diabetes Mellitus—A Population-Based Retrospective Cohort Study. PLoS ONE 2016, 11, 0161429. [Google Scholar] [CrossRef]
- Eichler, S.; Salzwedel, A.; Rabe, S.; Mueller, S.; Mayer, F.; Wochatz, M.; Hadzic, M.; John, M.; Wegscheider, K.; Völler, H. The Effectiveness of Telerehabilitation as a Supplement to Rehabilitation in Patients After Total Knee or Hip Replacement: Randomized Controlled Trial. JMIR Rehabil. Assist. Technol. 2019, 6, e14236. [Google Scholar] [CrossRef]
- Houta, S.; Ameler, T.; Surges, R. Use of HL7 FHIR to structure data in epilepsy self-management applications. In Proceedings of the International Conference on Wireless and Mobile Computing, Networking and Communications (WiMob), Barcelona, Spain, 21–23 October 2019; pp. 111–115, ISSN 2160-4886. [Google Scholar] [CrossRef]
- Unterreiner, M.; Biedermann, C.; El Fahem, R.; John, M.; Klose, S.; Haas, C.T.; Wächter, T. Comparing computer-aided therapy with conventional physiotherapy in Parkinson’s disease: An equivalence study. Neurol. Asia 2019, 24, 309–315. [Google Scholar]
- McConnell, M.V.; Shcherbina, A.; Pavlovic, A.; Homburger, J.R.; Goldfeder, R.L.; Waggot, D.; Cho, M.K.; Rosenberger, M.E.; Haskell, W.L.; Myers, J.; et al. Feasibility of Obtaining Measures of Lifestyle From a Smartphone App: The MyHeart Counts Cardiovascular Health Study. JAMA Cardiol. 2017, 2, 67. [Google Scholar] [CrossRef]
- Lane, N.D.; Lin, M.; Mohammod, M.; Yang, X.; Lu, H.; Cardone, G.; Ali, S.; Doryab, A.; Berke, E.; Campbell, A.T.; et al. BeWell: Sensing Sleep, Physical Activities and Social Interactions to Promote Wellbeing. Mob. Netw. Appl. 2014, 19, 345–359. [Google Scholar] [CrossRef]
- Cornet, R.; Stoicu-Tivadar, L.; Hörbst, A. Digital Healthcare Empowering Europeans: Proceedings of MIE2015; IOS Press: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Höll, B.; Spat, S.; Plank, J.; Schaupp, L.; Neubauer, K.; Beck, P.; Chiarugi, F.; Kontogiannis, V.; Pieber, T.; Holzinger, A. Design of a mobile, safety-critical in-patient glucose management system. Stud. Health Technol. Inform. 2011, 169, 950–954. [Google Scholar] [CrossRef]
- Peter, M.; Yang, Y.; Anthony, M.; Niranjan, B. A Digital Infrastructure for Storing & Sharing Internet of Things, Wearables and App-Based Research Study Data. In Studies in Health Technology and Informatics; IOS Press: Amsterdam, The Netherlands, 2020; pp. 87–96. [Google Scholar] [CrossRef]
- Manea, V.; Wac, K. mQoL: Mobile Quality of Life Lab: From Behavior Change to QoL. In Proceedings of the 2018 ACM International Joint Conference and 2018 International Symposium on Pervasive and Ubiquitous Computing and Wearable Computers, UbiComp’18, Singapore, 8–12 October 2018; Association for Computing Machinery: New York, NY, USA, 2018; pp. 642–647. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, X.; Lee, P.K.C. Improving the effectiveness of online healthcare platforms: An empirical study with multi-period patient-doctor consultation data. Int. J. Prod. Econ. 2019, 207, 70–80. [Google Scholar] [CrossRef]
- IEC. IEC 62304:2006 Medical Device Software—Software Life Cycle Processes; IEC: Geneva, Switzerland, 2006. [Google Scholar]
- ISO/IEC/IEEE. ISO/IEC/IEEE 15288:2015 Systems and Software Engineering—System Life Cycle Processes; ISO/IEC/IEEE: Geneva, Switzerland; Piscataway, NJ, USA, 2015; Available online: www.iso.org (accessed on 19 July 2020).
- OVE Österreichischer Verband für Elektrotechnik. Medizinprodukte—Qualitätsmanagementsysteme—Anforderungen für Regulatorische Zwecke (ISO 13485:2016) (Konsolidierte Fassung); OVE Österreichischer Verband für Elektrotechnik: Vienna, Austria, 2017. [Google Scholar]
- Snyder, H. Literature review as a research methodology: An overview and guidelines. J. Bus. Res. 2019, 104, 333–339. [Google Scholar] [CrossRef]
- Ohayon, M.M. Epidemiology of insomnia: What we know and what we still need to learn. Sleep Med. Rev. 2002, 6, 97–111. [Google Scholar] [CrossRef]
- Ozminkowski, R.J.; Wang, S.; Walsh, J.K. The direct and indirect costs of untreated insomnia in adults in the United States. Sleep 2007, 30, 263–273. [Google Scholar] [CrossRef]
- Daley, M.; Morin, C.M.; LeBlanc, M.; Grégoire, J.P.; Savard, J. The Economic Burden of Insomnia: Direct and Indirect Costs for Individuals with Insomnia Syndrome, Insomnia Symptoms, and Good Sleepers. Sleep 2009, 32, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Ohayon, M. Epidemiological Overview of sleep Disorders in the General Population. Sleep Med. Res. 2011, 2, 1–9. [Google Scholar] [CrossRef]
- Kirshner, B.; Guyatt, G. A methodological framework for assessing health indices. J. Chronic Dis. 1985, 38, 27–36. [Google Scholar] [CrossRef]
- Deshpande, P.; Sudeepthi, B.; Rajan, S.; Abdul Nazir, C. Patient-reported outcomes: A new era in clinical research. Perspect. Clin. Res. 2011, 2, 137. [Google Scholar] [CrossRef]
- Mayo, N.E.; Figueiredo, S.; Ahmed, S.; Bartlett, S.J. Montreal Accord on Patient-Reported Outcomes (PROs) use series – Paper 2: Terminology proposed to measure what matters in health. J. Clin. Epidemiol. 2017, 89, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Liu, J.; Koleva, Y.; Fonseca, V.; Kalsekar, A.; Pawaskar, M. Concordance of Adherence Measurement Using Self-Reported Adherence Questionnaires and Medication Monitoring Devices. PharmacoEconomics 2010, 28, 1097–1107. [Google Scholar] [CrossRef]
- Butte, N.F.; Ekelund, U.; Westerterp, K.R. Assessing physical activity using wearable monitors: Measures of physical activity. Med. Sci. Sport. Exerc. 2012, 44, S5–S12. [Google Scholar] [CrossRef] [PubMed]
- Webber, S.C.; Porter, M.M. Monitoring Mobility in Older Adults Using Global Positioning System (GPS) Watches and Accelerometers: A Feasibility Study. J. Aging Phys. Act. 2009, 17, 455–467. [Google Scholar] [CrossRef] [PubMed]
- Stone, A.A.; Shiffman, S. Ecological momentary assessment (EMA) in behavorial medicine. Ann. Behav. Med. 1994, 16, 199–202. [Google Scholar] [CrossRef]
- Sher, K.J.; Shiffman, S. Ecological Momentary Assessment; Oxford University Press: Oxford, UK, 2014. [Google Scholar]
- Last, J.M. A Dictionary of Epidemiology; Oxford University Press: New York, NY, USA, 2001; OCLC: 654103881. [Google Scholar]
- Verhagen, S.J.W.; Hasmi, L.; Drukker, M.; van Os, J.; Delespaul, P.A.E.G. Use of the experience sampling method in the context of clinical trials. Evid.-Based Ment. Health 2016, 19, 86–89. [Google Scholar] [CrossRef]
- Triantafillou, S.; Saeb, S.; Lattie, E.G.; Mohr, D.C.; Kording, K.P. Relationship Between Sleep Quality and Mood: Ecological Momentary Assessment Study. JMIR Ment. Health 2019, 6, e12613. [Google Scholar] [CrossRef]
- Li, H.; Mukherjee, D.; Krishnamurthy, V.B.; Millett, C.; Ryan, K.A.; Zhang, L.; Saunders, E.F.H.; Wang, M. Use of ecological momentary assessment to detect variability in mood, sleep and stress in bipolar disorder. BMC Res. Notes 2019, 12, 791. [Google Scholar] [CrossRef]
- Liang, Z.; Chapa Martell, M.A. Validity of Consumer Activity Wristbands and Wearable EEG for Measuring Overall Sleep Parameters and Sleep Structure in Free-Living Conditions. J. Healthc. Inform. Res. 2018, 2, 152–178. [Google Scholar] [CrossRef]
- Aledavood, T.; Torous, J.; Triana Hoyos, A.M.; Naslund, J.A.; Onnela, J.P.; Keshavan, M. Smartphone-Based Tracking of Sleep in Depression, Anxiety, and Psychotic Disorders. Curr. Psychiatry Rep. 2019, 21, 49. [Google Scholar] [CrossRef]
- Cornet, V.P.; Holden, R.J. Systematic review of smartphone-based passive sensing for health and wellbeing. J. Biomed. Inform. 2018, 77, 120–132. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, Q.; Hong, T.; Kang, C. Review of Smart Meter Data Analytics: Applications, Methodologies, and Challenges. IEEE Trans. Smart Grid 2019, 10, 3125–3148. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, Q.; Gan, D.; Yang, J.; Kirschen, D.S.; Kang, C. Deep Learning-Based Socio-Demographic Information Identification From Smart Meter Data. IEEE Trans. Smart Grid 2019, 10, 2593–2602. [Google Scholar] [CrossRef]
- Wang, J.; Janson, C.; Lindberg, E.; Holm, M.; Gislason, T.; Benediktsdóttir, B.; Johannessen, A.; Schlünssen, V.; Jogi, R.; Franklin, K.A.; et al. Dampness and mold at home and at work and onset of insomnia symptoms, snoring and excessive daytime sleepiness. Environ. Int. 2020, 139, 105691. [Google Scholar] [CrossRef] [PubMed]
- Dollander, M. Etiologies de l’insomnie chez l’adulte. Encephale 2002, 28, 493–502. [Google Scholar] [PubMed]
- Janson, C.; Norbäck, D.; Omenaas, E.; Gislason, T.; Nyström, L.; Jõgi, R.; Lindberg, E.; Gunnbjörnsdottir, M.; Norrman, E.; Wentzel-Larsen, T.; et al. Insomnia is more common among subjects living in damp buildings. Occup. Environ. Med. 2005, 62, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; Liu, Y.; Liu, H.; Li, M.; Shen, Y.; Gui, L.; Zhang, J.; Luo, Z.; Tao, X.; Yu, J. Factors associated with insomnia among Chinese front-line nurses fighting against COVID-19 in Wuhan: A cross-sectional survey. J. Nurs. Manag. 2020, 28, 1525–1535. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yang, L.; Liu, S.; Ma, S.; Wang, Y.; Cai, Z.; Du, H.; Li, R.; Kang, L. Survey of Insomnia and Related Social Psychological Factors Among Medical Staff Involved in the 2019 Novel Coronavirus Disease Outbreak. Front. Psychiatry 2020, 11, 306. [Google Scholar] [CrossRef] [PubMed]
- Jiaqing, O.; Pugh-Jones, C.; Clark, B.; Trott, J.; Chang, L. The Evolutionarily Mismatched Impact of Urbanization on Insomnia Symptoms: A Short Review of the Recent Literature. Curr. Psychiatry Rep. 2021, 23, 1–9. [Google Scholar] [CrossRef]
- Rajkomar, A.; Oren, E.; Chen, K.; Dai, A.M.; Hajaj, N.; Hardt, M.; Liu, P.J.; Liu, X.; Marcus, J.; Sun, M.; et al. Scalable and accurate deep learning with electronic health records. NPJ Digit. Med. 2018, 1, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Benson, T.; Grieve, G. Principles of Health Interoperability: SNOMED CT, HL7 and FHIR, 3rd ed.; Health Information Technology Standards, Springer International Publishing: Berlin/Heidelberg, Germany, 2016. [Google Scholar] [CrossRef]
- Gopinathan, K.; Kaloumenos, N.A.; Ajmera, K.; Matei, A.; Williams, I.; Davis, A. FHIR FLI: An Open Source Platform for Storing, Sharing and Analysing Lifestyle Data. In Proceedings of the 4th International Conference on Information and Communication Technologies for Ageing Well and e-Health, Madeira, Portugal, 22–23 March 2018; SCITEPRESS—Science and Technology Publications: Setubal, Portugal, 2018; pp. 227–233. [Google Scholar] [CrossRef]
- Massé, M. REST API Design Rulebook; O’Reilly: Sebastopol, CA, USA, 2012; OCLC: 777401536. [Google Scholar]
- Integrating the Healthcare Enterprise. IHE Patient Care Device Technical Framework Supplement: Personal Health Device Observation Upload (POU), HL7® FHIR® R4 Using Resources at FMM Level 2-Normative, Revision 1.1—Trial Implementation; IHE International: Oak Brook, IL, USA, 2020. [Google Scholar]
- Ghani, I. Introduction to PHP Web Services: PHP, JavaScript, MySQL, SOAP, RESTful, JSON, XML, WSDL; eBookit.com: Sudbury MA, USA, 2019; OCLC: 1122454291. [Google Scholar]
- Fuentes, J.M.D.; Gonzalez-manzano, L.; Solanas, A.; Veseli, F. Attribute-Based Credentials for Privacy-Aware Smart Health Services in IoT-Based Smart Cities. Computer 2018, 51, 44–53. [Google Scholar] [CrossRef]
- Ouhbi, S.; Idri, A.; Fernández-Alemán, J.L. Standards-Based Sustainability Requirements for Healthcare Services in Smart Cities. In Smart Cities: Development and Governance Frameworks; Springer International Publishing: Berlin/Heidelberg, Germany, 2018; pp. 299–317. [Google Scholar] [CrossRef]
- Clim, A.; Zota, R.; Constantinescu, R.; Ilie-Nemedi, I. Health services in Smart Cities: Choosing the big data mining based decision support. Int. J. Healthc. Manag. 2019, 13, 79–87. [Google Scholar] [CrossRef]
- Fragidis, L.L.; Chatzoglou, P.D. Implementation of a nationwide electronic health record (EHR): The international experience in 13 countries. Int. J. Health Care Qual. Assur. 2018, 31, 116–130. [Google Scholar] [CrossRef]
- Safdari, R.; Ghazisaeidi, M.; Jebraeily, M. Electronic Health Records: Critical Success Factors in Implementation. Acta Inform. Med. 2015, 23, 102. [Google Scholar] [CrossRef]
- Ahmadi, M.; Aslani, N. Capabilities and Advantages of Cloud Computing in the Implementation of Electronic Health Record. Acta Inform. Med. 2018, 26, 24. [Google Scholar] [CrossRef] [PubMed]
- Akter, S.; Michael, K.; Uddin, M.R.; McCarthy, G.; Rahman, M. Transforming business using digital innovations: The application of AI, blockchain, cloud and data analytics. In Annals of Operations Research; Springer: Berlin/Heidelberg, Germany, 2020; pp. 1–33. [Google Scholar] [CrossRef]
- Turgeman, L.; May, J.H.; Sciulli, R. Insights from a machine learning model for predicting the hospital Length of Stay (LOS) at the time of admission. Expert Syst. Appl. 2017, 78, 376–385. [Google Scholar] [CrossRef]
- Ghobaei-Arani, M.; Souri, A.; Baker, T.; Hussien, A. ControCity: An Autonomous Approach for Controlling Elasticity Using Buffer Management in Cloud Computing Environment. IEEE Access 2019, 7, 106912–106924. [Google Scholar] [CrossRef]
- Bala, R.; Gill, B.; Smith, D. Gartner Magic Quadrant for Cloud Infrastructure. 2019. Available online: cloud.google.com (accessed on 26 February 2020).
- Yuhanna, N. The Forrester Wave™: Data Management For Analytics, Q1 2020; Forrester Research: Cambridge, MA, USA, 2020; p. 17. [Google Scholar]
- Shey, H. The Forrester Wave™: Data Security Portfolio Vendors, Q2 2019; Forrester Research: Cambridge, MA, USA, 2019; p. 16. [Google Scholar]
- Muchagata, J.; Ferreira, A. Translating GDPR into the mHealth Practice. In Proceedings of the International Carnahan Conference on Security Technology (ICCST), Montreal, QC, Canada, 22–25 October 2018; pp. 1–5, ISSN 2153-0742. [Google Scholar] [CrossRef]
- Hyseni, L.N.; Ibrahimi, A. Comparison of the cloud computing platforms provided by Amazon and Google. In Proceedings of the Computing Conference, London, UK, 18–20 July 2017; pp. 236–243. [Google Scholar] [CrossRef]
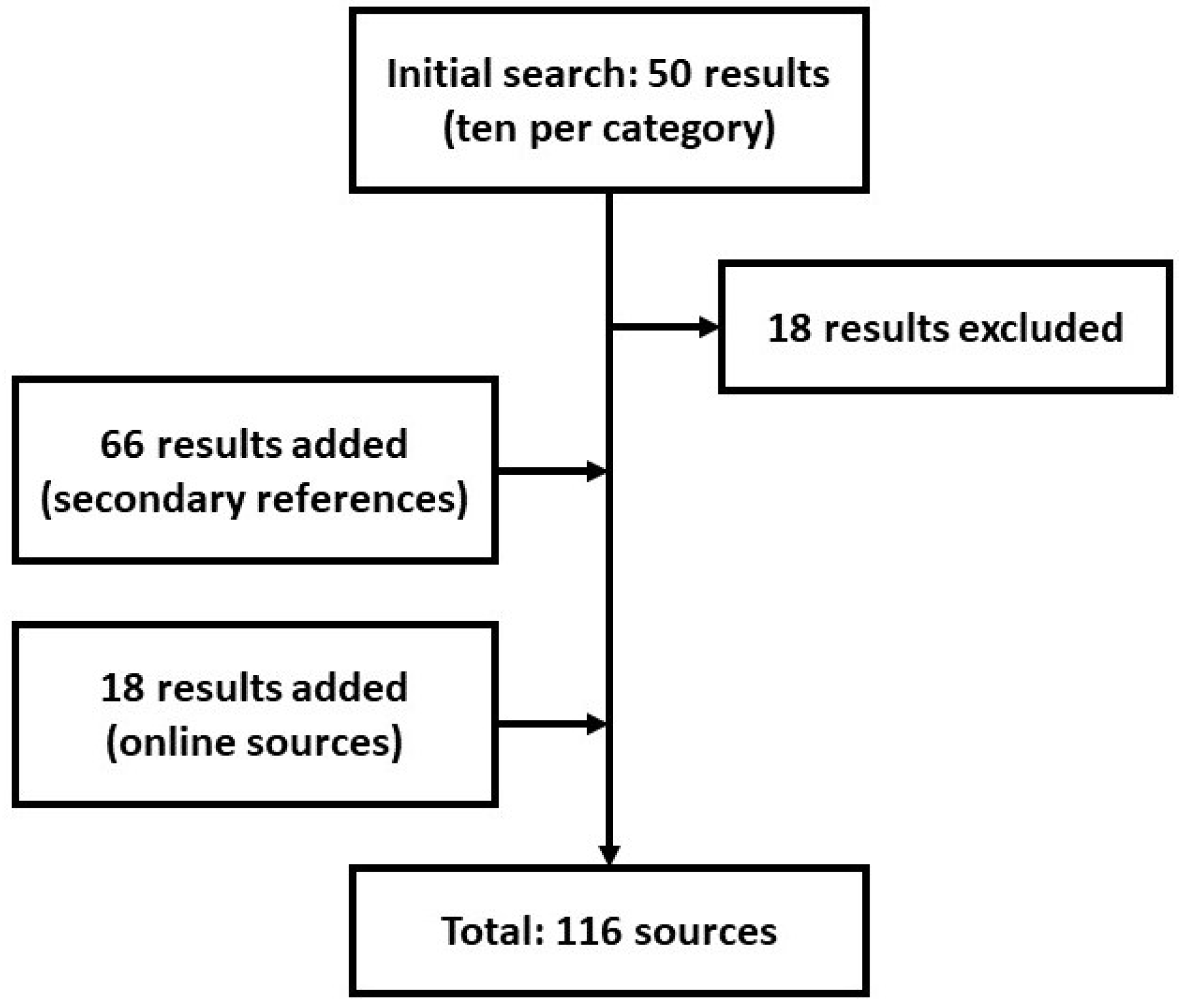
| Category | Search Terms |
|---|---|
| Diagnosis and therapy | Insomnia, Sleep Disorders, Guidelines, Therapy, Polysomnography |
| Data sources and acquisition | Clinical Outcomes, Internet of Things, Sensors, Wearables, Value-based Healthcare |
| Data formats and standardisation | FHIR, Medical Data Exchange, HL7, Standards, REST |
| Legal aspects | MDR, Medical Device, Standards, GDPR |
| State-of-the-art | Therapy Platform, Clinical Application, Disease Management Programme, mHealth |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reichenpfader, D.; Hanke, S. Requirements and Architecture of a Cloud Based Insomnia Therapy and Diagnosis Platform: A Smart Cities Approach. Smart Cities 2021, 4, 1316-1336. https://doi.org/10.3390/smartcities4040070
Reichenpfader D, Hanke S. Requirements and Architecture of a Cloud Based Insomnia Therapy and Diagnosis Platform: A Smart Cities Approach. Smart Cities. 2021; 4(4):1316-1336. https://doi.org/10.3390/smartcities4040070
Chicago/Turabian StyleReichenpfader, Daniel, and Sten Hanke. 2021. "Requirements and Architecture of a Cloud Based Insomnia Therapy and Diagnosis Platform: A Smart Cities Approach" Smart Cities 4, no. 4: 1316-1336. https://doi.org/10.3390/smartcities4040070
APA StyleReichenpfader, D., & Hanke, S. (2021). Requirements and Architecture of a Cloud Based Insomnia Therapy and Diagnosis Platform: A Smart Cities Approach. Smart Cities, 4(4), 1316-1336. https://doi.org/10.3390/smartcities4040070







