1. Introduction
In the current context of climate change and increasing pressure to reduce the carbon footprint of resource-intensive industries, the construction sector faces the challenge of transforming its materials and processes towards more sustainable alternatives [
1,
2]. Ordinary Portland Cement (OPC), the basis of conventional concrete, is responsible for approximately 7–9% of global CO
2 emissions, mainly because of the calcination of calcium carbonate at temperatures nearing 1400 °C, which requires high energy consumption and releases significant amounts of greenhouse gases [
3]. In response, alternative cementitious materials, particularly geopolymers and alkali-activated slag (AAS) systems, have garnered growing interest [
2,
4,
5]. These materials utilise industrial by-products rich in silica and alumina, such as fly ash (FA) and ground granulated blast furnace slag (GGBFS), that, when chemically activated with alkaline solutions, form a cementitious matrix offering excellent mechanical strength, acceptable durability, and a substantial reduction in CO
2 emissions. In addition to their low cost and abundance, these residues help reduce clinker dependency and mitigate the environmental impacts of traditional cement production [
6,
7]. Nevertheless, one of the most critical technical challenges of AAS systems is their poor resistance to carbonation, which limits their use in structures exposed to urban or industrial environments with elevated CO
2 concentrations [
7,
8]. Carbonation occurs when atmospheric CO
2 penetrates the concrete, reacts with alkaline compounds in the matrix, and progressively reduces the pH—disrupting the passive film that protects the embedded steel and initiating corrosion processes [
9,
10,
11].
Unlike Portland concrete, which contains substantial amounts of calcium hydroxide (CH), AAS matrices possess a limited alkaline reserve based on C–A–S–H or N–A–S–H gels, offering a reduced buffering capacity against CO
2 [
12]. Furthermore, their porous structure may facilitate gas diffusion if not properly controlled [
13]. This loss of alkalinity and the onset of corrosion present significant technical risks for structural durability and lead to elevated maintenance costs, contradicting the sustainability goals that motivate the use of such materials [
12].
To address this issue, strategies such as special curing regimes, hybrid activators, or functional additives have been proposed [
13]. A promising option is the use of anion-intercalating lamellar materials, such as calcined magnesium–aluminium layered double hydroxide (CLDH) [
14]. This material, a part of the LDH family, is characterised by its anion-capturing ability, structural rehydration (“memory effect”), and controlled release of OH
−, thus helping preserve internal alkalinity and hinder CO
2 ingress [
15].
When incorporated into the AAS matrix, CLDH acts not only as an alkaline buffer but also modifies the microstructure, reduces pore connectivity, and limits the penetration rate of aggressive gases [
16,
17]. Upon rehydration in humid and carbonated environments, CLDH partially restores its lamellar structure, releasing ions that help maintain a pH conducive to steel protection [
18,
19]. This regenerative capability and synergy with alkaline activators make CLDH an attractive additive from both technical and economic perspectives [
20].
Moreover, recent investigations emphasize that the corrosion behaviour of alkali-activated concretes is highly dependent on the balance between the pore structure, ionic transport, and availability of alkaline species in the matrix [
21]. Electrochemical studies have shown that the lack of portlandite and the prevalence of low-pH C–A–S–H gels accelerate steel depassivation under carbonation [
22]. Consequently, the incorporation of functional additives capable of simultaneously refining the microstructure and stabilizing the alkaline environment has become a critical research focus. CLDH, with its anion-exchange and rehydration capabilities, not only mitigates carbonation effects but also synergistically improves the electrochemical stability of the steel–concrete interface, as confirmed by polarization, EIS, and SEM–EDS analyses in recent works [
23].
The present study investigates the incorporation of increasing CLDH dosages (2%, 4%, 6%, and 8%) as a partial slag replacement in AAS mixtures. These levels are based on previous studies indicating that low dosages may be insufficient to trigger a significant structural reconstruction of CLDH, while higher levels might lead to the oversaturation of lamellar phases, formation of inert residues, or adverse effects on the durability and corrosion resistance of the system [
24,
25].
This approach aims to identify the optimal dosage that maximizes protection against carbonation-induced corrosion without compromising the microstructural integrity of the matrix.
2. Materials and Methods
2.1. Materials
This study employed two primary cementitious materials: ground granulated blast furnace slag (GGBFS) and calcined layered double hydroxide (CLDH). The GGBFS, a by-product of the steel industry supplied by Acerías Paz del Río (Boyacá, Colombia), was obtained via water granulation, ensuring a reactive vitreous phase suitable for alkaline activation. Its chemical composition (
Table 1), determined by X-ray fluorescence (XRF) spectroscopy, revealed high contents of calcium oxide (CaO), silicon dioxide (SiO
2), aluminium oxide (Al
2O
3), and magnesium oxide (MgO), which are characteristic of a basic slag. In addition, the reinforcing bars used in this study were made of carbon steel 1020 according to ASTM A29/A29M, and are commonly employed in reinforced concrete [
26].
The CLDH used was a synthetic hydrotalcite with a lamellar double hydroxide structure primarily composed of magnesium and aluminium. This material was pre-calcined at temperatures above 500 °C to induce crystalline restructuring, enhancing its memory effect in alkaline environments and promoting the adsorption of carbonate species. The CLDH featured a Mg/Al molar ratio close to 3:1, making it compatible with highly alkaline cementitious matrices.
Two types of mixtures were prepared depending on the analysis objective. Alkali-activated pastes were used for physico-chemical, mineralogical, and microstructural characterization and composed only of GGBFS, CLDH, and activator solution, without any aggregates; and alkali-activated concrete was used for durability testing related to reinforcement corrosion. These mixes incorporated the same binder matrix along with coarse and fine aggregates. The coarse aggregate was crushed granite with a nominal maximum size of 19 mm and a bulk density of 2.65 g/cm
3. The fine aggregate consisted of natural siliceous sand with a fineness modulus of 2.70 and a controlled moisture content of 2%. Both aggregates met ASTM C33 standards [
27]. The concrete also included embedded reinforcing steel bars that were cleaned and surface-treated prior to casting to ensure a proper bond. CLDH contents of 0%, 2%, 4%, 6%, and 8% by total binder mass (GGBFS + CLDH) were assessed in both mixture types. Water/activator and activator/binder ratios were held constant across all systems.
The chemical activation of the mixtures was carried out using a biphasic alkaline solution comprising 85% sodium hydroxide (NaOH) by mass and 15% sodium silicate (Na2SiO3). The sodium hydroxide was dissolved in deionised water to achieve a concentration of 14 mol/L, ensuring a high availability of OH− ions to promote the initial dissolution of the slag. This reagent, with a purity of 98% and supplied in pellet form, was obtained from Sigma-Aldrich (St. Louis, MO, USA). The liquid sodium silicate used had a typical composition of 9.1% Na2O, 27.5% SiO2, and 63.4% H2O, corresponding to a SiO2/Na2O molar ratio of 3.02. This ratio was selected to favour the formation of (C,N)–A–S–H-type gels during hydration, balancing the availability of reactive silica with the system’s alkalinity.
The ratio between the total mass of the activator solution and the cementitious material (slag + CLDH) was fixed at 0.55, which was optimised to ensure sufficient workability and efficient system activation. Initial curing of the pastes was performed in an oven at a constant temperature of 85 °C for 24 h, aiming to accelerate the dissolution–precipitation reactions characteristic of alkali-activated binders. After this period, samples were allowed to cool to ambient temperature and subsequently stored under stable conditions.
Before its incorporation into the activator solution, CLDH was mechanically dispersed in a 14 M NaOH solution for 30 min to reduce agglomeration and ensure a homogeneous distribution within the matrix.
2.2. Methods
The mechanical characterisation of alkali-activated slag concrete (AASC) focused on 28-day compressive strength. Cylindrical specimens (150 mm × 300 mm) were prepared using a binder matrix composed of ground granulated blast furnace slag and calcined layered double hydroxide (CLDH) at 0%, 2%, 4%, 6%, and 8% by total binder mass. Compressive strength tests were conducted in accordance with ASTM C39/C39M-15A [
28,
29] using an automated hydraulic press with a loading rate of 0.25 MPa/s. Steel and neoprene caps were employed to ensure a uniform stress distribution during the tests.
The identification of mineral phases present in the samples was carried out using X-ray diffraction (XRD) with a PANalytical X’Pert PRO MPD instrument (Malvern PANalytical, Almelo, The Netherlands) configured in reflection mode with Cu-Kα radiation (λ = 1.5406 Å). Measurements were conducted over a 2θ angle range from 5° to 65°, with a step size of 0.02° and a counting time of 0.5 s per interval.
To assess the atomic environment of silicon and aluminium, magic angle spinning nuclear magnetic resonance (MAS-NMR) spectroscopy was performed using a JEOL ECZ600R spectrometer operating at 119.2 MHz for 29Si and 156.4 MHz for 27Al, under a magnetic field with a strength of 14.1 T. A spinning frequency of 12 kHz was used, alongside external references such as tetramethylsilane (TMS) and Al(H2O)63+. A pulse length of 3.5 μs and a relaxation delay of 5 s were applied to minimise saturation effects.
The microstructural analysis was complemented by scanning electron microscopy (SEM) using a Hitachi SU8230 field emission system.
To investigate the behaviour under carbonation-induced corrosion, cylindrical concrete specimens (10 cm in diameter and 20 cm in height) were prepared, each incorporating a single longitudinally embedded ribbed steel bar. The reinforcing bars had a diameter of 12 mm and a total length of 15 cm, of which 10 cm were embedded in the cementitious matrix, while the remaining 5 cm were left exposed for electrical connection during testing. The exposed section of the bars was coated with epoxy paint to ensure electrical insulation, thereby restricting the active area to the embedded segment.
Prior to casting, the bars were mechanically cleaned to remove surface oxides and to ensure a proper bond with the matrix. These specimens were specifically used for electrochemical impedance spectroscopy (EIS) testing.
Concrete samples were subjected to accelerated carbonation conditions in order to replicate, within a shorter time span, the effects of CO2 ingress into the cementitious matrix. Exposure took place in a controlled chamber with a 3% CO2 concentration, 65% relative humidity, and a constant temperature of 25 °C over a period of 90 days.
The carbonation depth was determined using an alcoholic phenolphthalein solution as a pH indicator. Upon application to the fractured surface of the specimens, carbonated areas (colourless) and still alkaline regions (pink) were visually differentiated, and the average depth of carbonation front was measured using a millimetre-scale ruler.
The concrete specimens were not immersed in any brine solution prior to the accelerated carbonation test. The corrosion state of embedded steel was exclusively assessed using electrochemical impedance spectroscopy (EIS). A Gamry Reference 3000 potentiostat/galvanostat was employed, configured with a three-electrode electrochemical cell: the embedded steel bar as the working electrode, a saturated calomel electrode (SCE) as the reference, and a stainless steel mesh as the counter-electrode. A sinusoidal perturbation of ±10 mV was applied across a frequency range from 100 kHz to 10 mHz. The resulting data were presented in Nyquist plots, allowing for the determination of system polarisation resistance and indirect estimation of corrosion current density.
3. Results
Figure 1 presents the 28-day compressive strength results for mixtures containing 0–8% CLDH by binder mass. The reference mixture (0% CLDH) reached 62 MPa, whereas the mixtures with 2% and 4% achieved 58.5 MPa and 52.8 MPa, respectively, maintaining 85–95% of the strength of the control mix. For 6% and 8% CLDH, the strength decreased to 48.3 MPa and 41.1 MPa, reflecting a progressive dilution effect of the reactive slag [
30]. These experimental values are higher than previous projections, which predicted 44–46 MPa for 2% CLDH and 32–35 MPa for 8% CLDH, indicating that the filler and nucleation effects of CLDH promoted the formation of C-A-S-H/N-A-S-H gels and helped to maintain satisfactory mechanical performance.
3.1. Composition Analysis
3.1.1. XRD
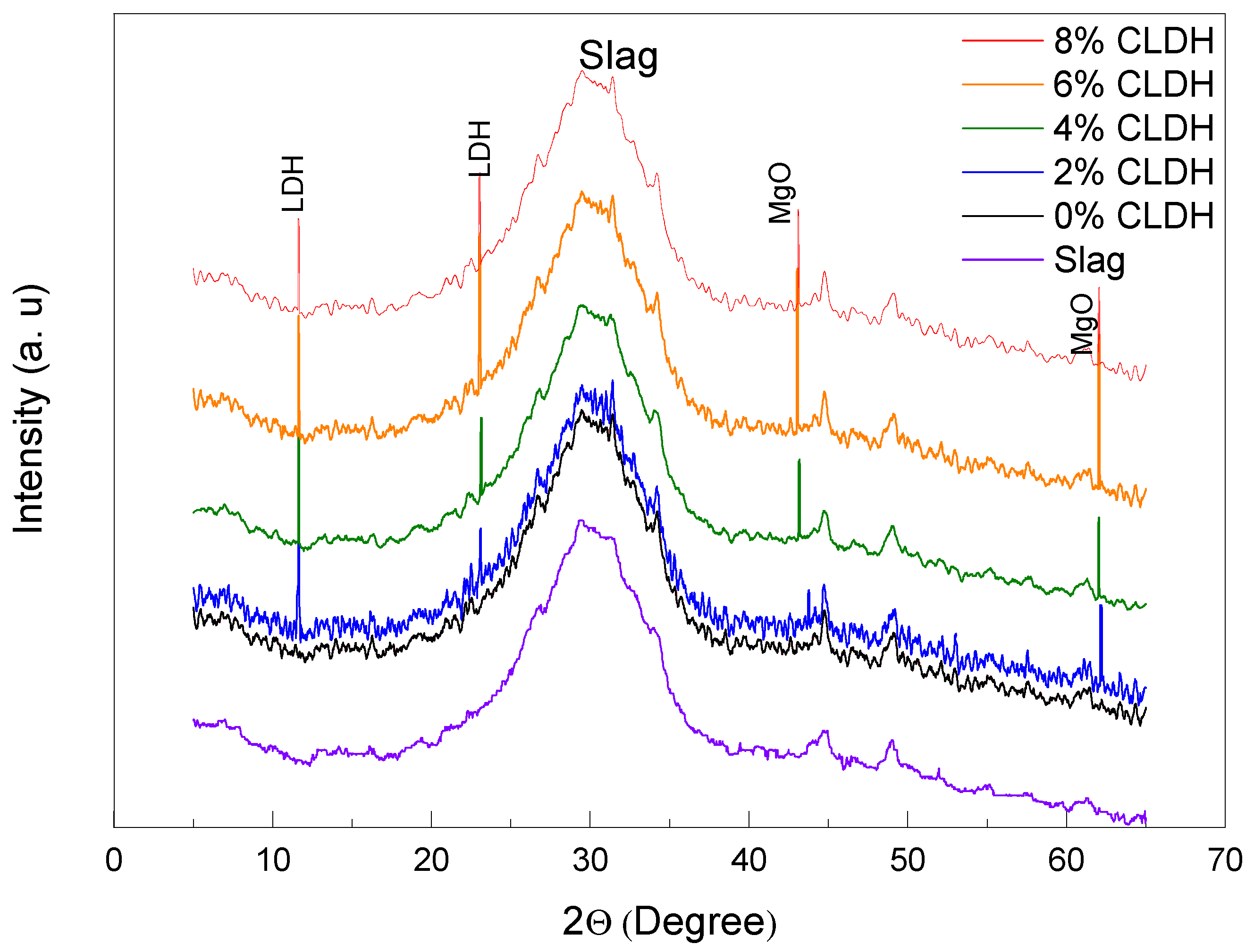
Figure 2 displays the X-ray diffraction (XRD) patterns of alkali-activated slag (AAS) pastes incorporating varying amounts of calcined layered double hydroxide (CLDH): 0%, 2%, 4%, 6%, and 8%. The reference mix (0% CLDH) exhibits a typical diffraction profile for AAS materials, characterised by a broad amorphous halo centred around 30° indicative of C–A–S–H gel formation, which lacks long-range crystalline order detectable by XRD [
31].
With the addition of 2% CLDH, weak peaks emerge near 11.6° and 23.2°, corresponding to the (003) and (006) planes of hydrotalcite-like (LDH) phases. These reflections are consistent with a lamellar structure of trigonal–rhombohedral symmetry (space group R-3m) typical of LDHs reformed from mixed Mg–Al oxides [
32]. Their low intensity suggests a limited degree of structural reconstitution at this dosage.
Increasing the CLDH content to 4% and more markedly to 6% leads to a pronounced intensification of the LDH peaks, confirming the active reconstruction of the lamellar structure via interlayer anion (CO
32− and OH
−) uptake. This reflects effective activation of the memory effect, whereby the calcined oxides regenerate the LDH structure in an alkaline environment [
33]. From 4% CLDH onwards, additional reflections near 43° and 62° appear, with the MgO peak becoming most intense at 6% CLDH. This does not necessarily denote a higher residual MgO content: an analogous behaviour has been observed in AAS systems where CLDH reconverts into LDH-type phases, influencing reaction kinetics and phase assemblage [
34]. The possibilities of preferred crystal orientation or local reprecipitation of Mg species may also contribute to the enhanced diffraction signal; these are conceptual interpretations and should be explicitly stated as such.
A dosage of 6% CLDH is thus identified as optimal, providing a microstructure with a high LDH peak intensity, balanced slag hydration, and electrochemical stability. This behaviour suggests that the MgO detected at 6% CLDH is largely associated with surface reprecipitation rather than unreacted core particles, resulting in a coherent matrix that combines significant LDH regeneration with high polarisation resistance.
In contrast, the 8% CLDH mixture exhibits a relative decrease in LDH peak intensity and a broader MgO signal, indicating that excess CLDH primarily behaves as an inert filler, diluting reactive slag and limiting further cementitious product formation.
A dosage of 6% CLDH is identified as optimal, as it yields the highest intensity of LDH peaks without significant interference from residual phases. This balance ensures sufficient reactivity to promote slag hydration while maintaining robust C–A–S–H gel formation. In contrast, the 8% CLDH mixture exhibits a relative decrease in LDH peak intensity alongside a marked increase in MgO signals, suggesting a threshold beyond which further CLDH addition becomes ineffective [
35]. The excess CLDH may behave as an inert filler, diluting the highly reactive slag and potentially hindering the formation of cementitious products, with adverse effects on microstructural densification and mechanical performance.
Figure 2 shows the X-ray diffraction (XRD) patterns of alkali-activated slag (AAS) pastes with varying proportions of calcined layered double hydroxides (CLDH) as a partial slag replacement (0%, 2%, 4%, 6%, and 8%).
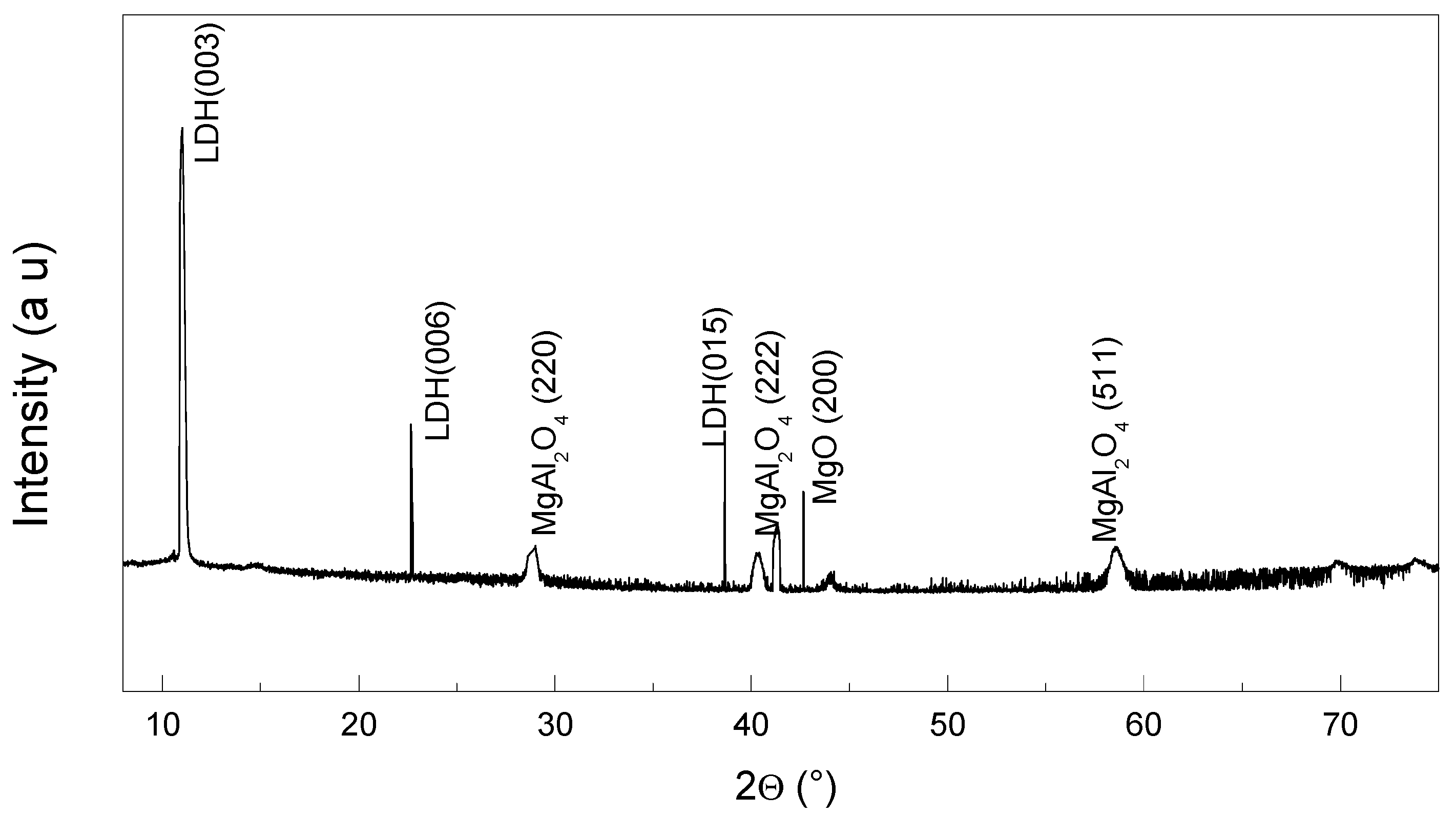
The X-ray diffraction (XRD) pattern (
Figure 3) confirms the presence of layered double hydroxide (LDH), magnesium oxide (MgO), and magnesium aluminate spinel (MgAl
2O
4) phases. The main reflections associated with LDH are clearly identified at (003), (006), and (015), indicating the preservation of the layered structure after synthesis. Peaks corresponding to MgO are indexed at the (200) plane, while MgAl
2O
4 spinel phases appear at (220), (222), and (511), confirming the partial transformation of the LDH structure during calcination. The coexistence of LDH and spinel peaks suggests an incomplete dehydroxylation process, which can contribute to the reactivity of the calcined LDH (CLDH) when incorporated into alkali-activated slag systems. This crystalline composition aligns with the expected behaviour of CLDH as a nucleating agent in promoting C–A–S–H gel formation.
3.1.2. NMR
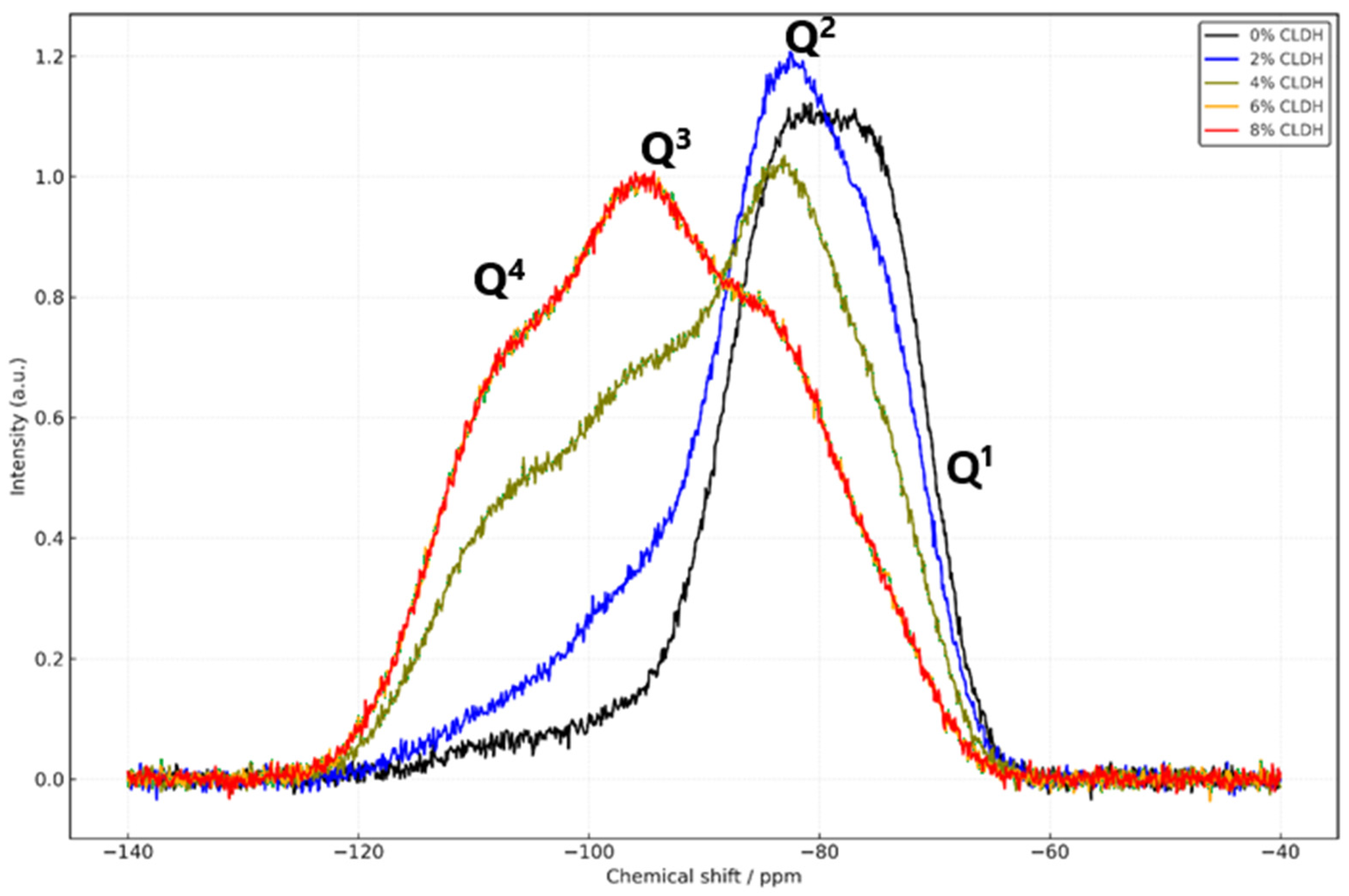
Figure 4 and
Figure 5 present the magic angle spinning nuclear magnetic resonance (MAS-NMR) spectra for
29Si and
27Al nuclei in alkali-activated slag concrete (AASC) mixtures containing different proportions of calcined layered double hydroxide (CLDH): 0%, 2%, 4%, 6%, and 8%. These spectra provide insight into the structural evolution of the cementitious gel as a function of the CLDH content and its integration into the alkali-activated matrix.
The
29Si spectra reveal that the reference mix (0% CLDH) is dominated by Q
1 and Q
2 units, located between −74 and −83 ppm [
36]. This distribution reflects a C–A–S–H gel structure with a low degree of polymerisation, limited connectivity between silicate tetrahedra, and greater susceptibility to carbonation. With the incorporation of 2% CLDH, Q
2 signals intensify and traces of Q
3 (−95 ppm) appear, suggesting an initial rearrangement of the silicate environment. At 4%, the Q
3 intensity increases significantly and Q
4 signals (−103 to −110 ppm) emerge, indicating the development of a denser and more polymerised network. The highest level of structural development is reached at 6% CLDH, where Q
3 dominates and Q
4 is well-defined, pointing to a highly interlinked three-dimensional matrix [
37]. At 8%, these signals persist but without further improvement, suggesting that a functional saturation threshold has been reached.
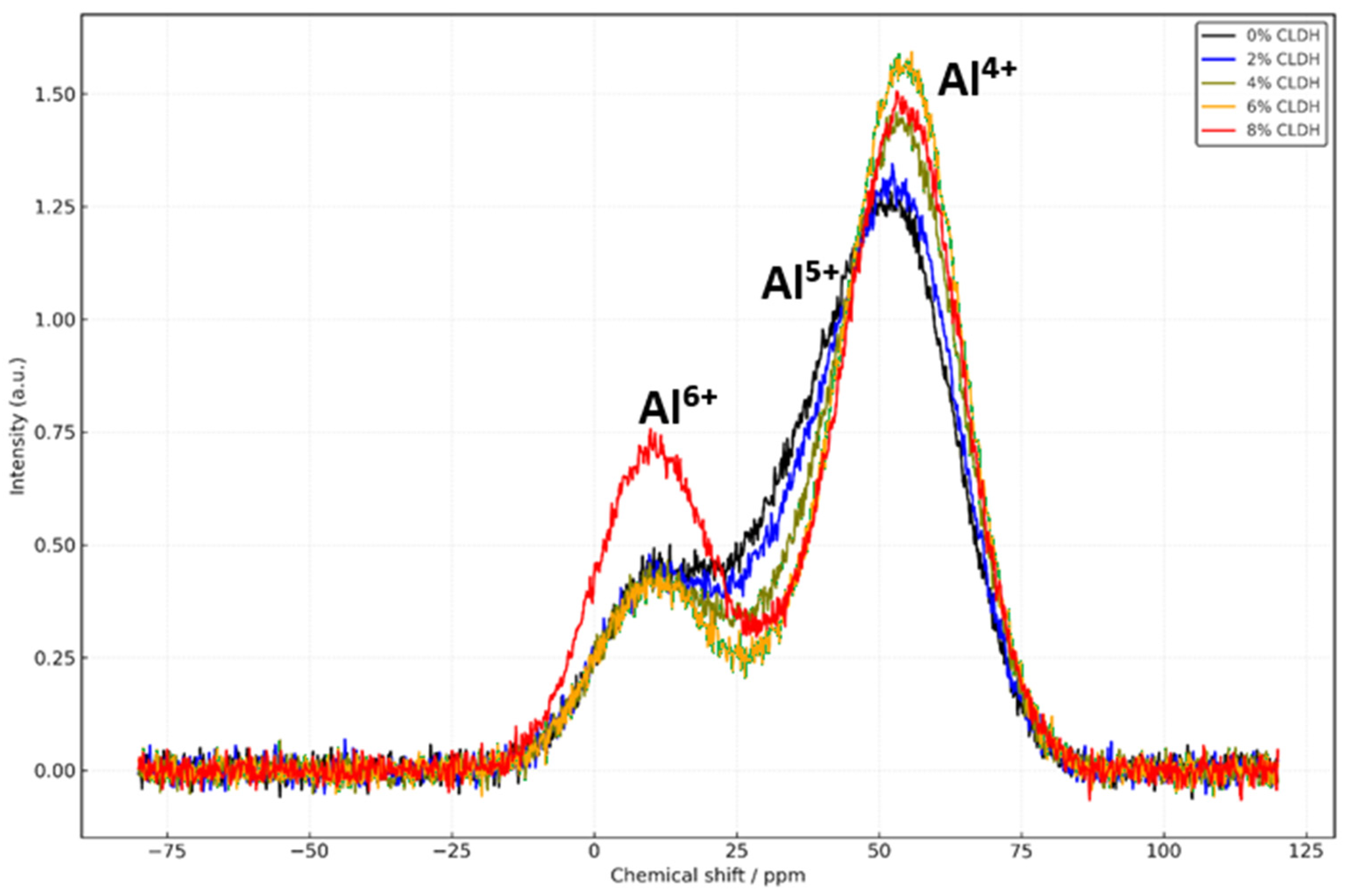
The 27Al spectra consistently show a strong signal near +55 ppm across all mixtures, corresponding to tetrahedrally coordinated aluminium (Al4+) integrated into the C–A–S–H or N–A–S–H gel network. As the CLDH content increases up to 6%, this signal becomes more pronounced, while the signal associated with penta-coordinated aluminium (+38 ppm) decreases, indicating the effective incorporation of Al released from the rehydrated CLDH. In the mix with 8% CLDH, a relative increase in the Al6+ signal at +10 ppm is observed, associated with residual phases such as MgO or spinels, suggesting incomplete CLDH rehydration and poor integration into the principal gel.
Collectively, the results shown in
Figure 4 confirm that CLDH significantly alters the molecular architecture of both silicate and aluminate gels [
38]. An optimal addition of 6% enables maximum silicate network polymerisation and efficient structural incorporation of aluminium. Below this threshold, reinforcement is progressive yet limited; above it, the system loses efficiency because of the formation of inert secondary phases. The CLDH-free sample (0%) reflects a poorly connected and less dense structure, whereas the 8% mixture exhibits signs of structural overload and chemical imbalance. These structural transformations, evidenced by shifts and intensity changes in the MAS-NMR spectra, are directly correlated with the mechanical performance, carbonation resistance, and overall durability of the material.
3.1.3. SEM
Figure 6 presents SEM micrographs at 1000× magnification of alkali-activated slag pastes (AASC) modified with varying proportions of calcined layered double hydroxide (CLDH): 0%, 2%, 4%, 6%, and 8%. This analysis allows for correlations between the surface morphology and matrix density with hydration dynamics, structural reorganisation, and the formation of secondary products induced by CLDH, enabling the evaluation of the additive’s efficiency in enhancing microstructural development [
39].
In the absence of CLDH (
Figure 6a, 0%), the microstructure exhibits high porosity, with poorly consolidated fragments, disaggregated zones, and limited C–A–S–H gel formation. EDS analysis confirms a low concentration of elements associated with hydration products, resulting in a heterogeneous, weakly bonded network with a poor gel distribution [
40]. This configuration correlates with low mechanical strength and higher susceptibility to carbonation. With 2% CLDH (
Figure 6b), a slight improvement in densification is observed, characterized by more integrated particles and a partial reduction in visible voids. The EDS spectra show moderate increases in the Si, Ca, and Al contents, indicating the onset of a more active hydration process. Nonetheless, discontinuous regions persist, suggesting that hydration is still incomplete. At 4% CLDH (
Figure 6c), the microstructural enhancement becomes more evident: the surface exhibits partial continuity, globular hydration products, and overall increased compactness. EDS results reveal a significant rise in the elemental composition associated with C–A–S–H gels, supporting the hypothesis that CLDH acts as a nucleating agent, promoting hydration in previously inert zones. The most favourable behaviour occurs at 6% CLDH (
Figure 6d), where a homogeneous, densely packed matrix with negligible porosity is achieved [
41]. Globular hydration products are fully integrated into a continuous network, indicating an advanced degree of hydration and polymerization. EDS spectra confirm a composition rich in Ca, Si, and Al, consistent with the maximum formation of C–A–S–H gels, which correlates with the highest mechanical strength and durability for this formulation. However, at 8% CLDH (
Figure 6e), a decline in matrix continuity is observed. Although the globular morphology partially persists, isolated or weakly bonded spheres and agglomerated regions appear [
42,
43]. EDS analysis shows a less homogeneous distribution, suggesting the formation of poorly reactive secondary gels or interference with complete slag hydration, which negatively affects the integration of reaction products. In conclusion, the combined SEM-EDS analysis confirms that CLDH exerts a decisive influence on the internal microstructure of AASC pastes. A 6% dosage is identified as optimal for maximizing densification, minimizing porosity, and promoting a homogeneous, continuous matrix. In contrast, mixtures containing 0% and 8% CLDH exhibit suboptimal microstructural configurations, which could compromise durability and corrosion resistance.
3.1.4. Carbonation Front Penetration Under Accelerated CO2
The carbonation depth was assessed in AASC mixtures with varying proportions of CLDH (0%, 2%, 4%, 6%, and 8%) after 87 days of accelerated exposure under controlled conditions (3% CO
2, 65% relative humidity, 25 °C). Measurements were conducted using a 1% alcoholic phenolphthalein solution, which turns magenta in regions with pH above 9.0 and remains colourless when the pH falls below this threshold [
34,
44]. The visual results of the carbonation tests are presented in
Figure 7, where the progression of the carbonation front is clearly observed for each mixture: (a) 0%, (b) 2%, (c) 4%, (d) 6%, and (e) 8% CLDH.
The mixture without CLDH (0%) exhibited a carbonation depth of 50 mm, reflecting a rapid loss of alkalinity and high susceptibility to CO
2 ingress due to the absence of additional chemical buffering mechanisms. As the CLDH content increased, a systematic reduction in the carbonation depth was observed. At 2% CLDH, the carbonation front decreased to 38.2 mm, indicating a moderate improvement attributed to slight microstructural densification. Mixtures containing 4% and 6% CLDH demonstrated significantly enhanced performance, reaching depths of 31.2 mm and 25.4 mm, respectively [
45]. These results confirm that CLDH serves as a dual-functional agent: it provides a chemical buffer through the release of OH
− and CO
32− anions and simultaneously promotes matrix densification, effectively limiting CO
2 penetration.
At 8%, the additional reduction was marginal (23.7 mm), suggesting a functional saturation of the system. In this case, part of the CLDH may not rehydrate effectively and could act as an inert or dilutive filler, reducing overall efficiency.
In summary, a 6% CLDH dosage is identified as optimal, maximising carbonation resistance without compromising the system’s microstructural integrity [
46]. This result is consistent with supporting physicochemical evidence obtained from XRD and NMR analyses, where this proportion also exhibited the highest reactivity and structural stability.
3.1.5. EIS
Figure 8 shows the electrochemical impedance response of the AASC mixtures after accelerated carbonation, including the Nyquist (a) and Bode (b) plots. The progressive incorporation of calcined layered double hydroxide (CLDH) significantly modifies the system’s electrochemical behaviour. In the Nyquist diagrams, an increase in the CLDH content leads to larger semicircular arcs, which reflects a higher polarization resistance (Rp) and indicates improved protection of the embedded steel against corrosion. This behaviour is consistent with the Bode plots, where higher CLDH dosages result in greater impedance magnitudes at low frequencies and a more stable phase angle response, confirming the enhancement of the passive state in the alkali-activated matrix.
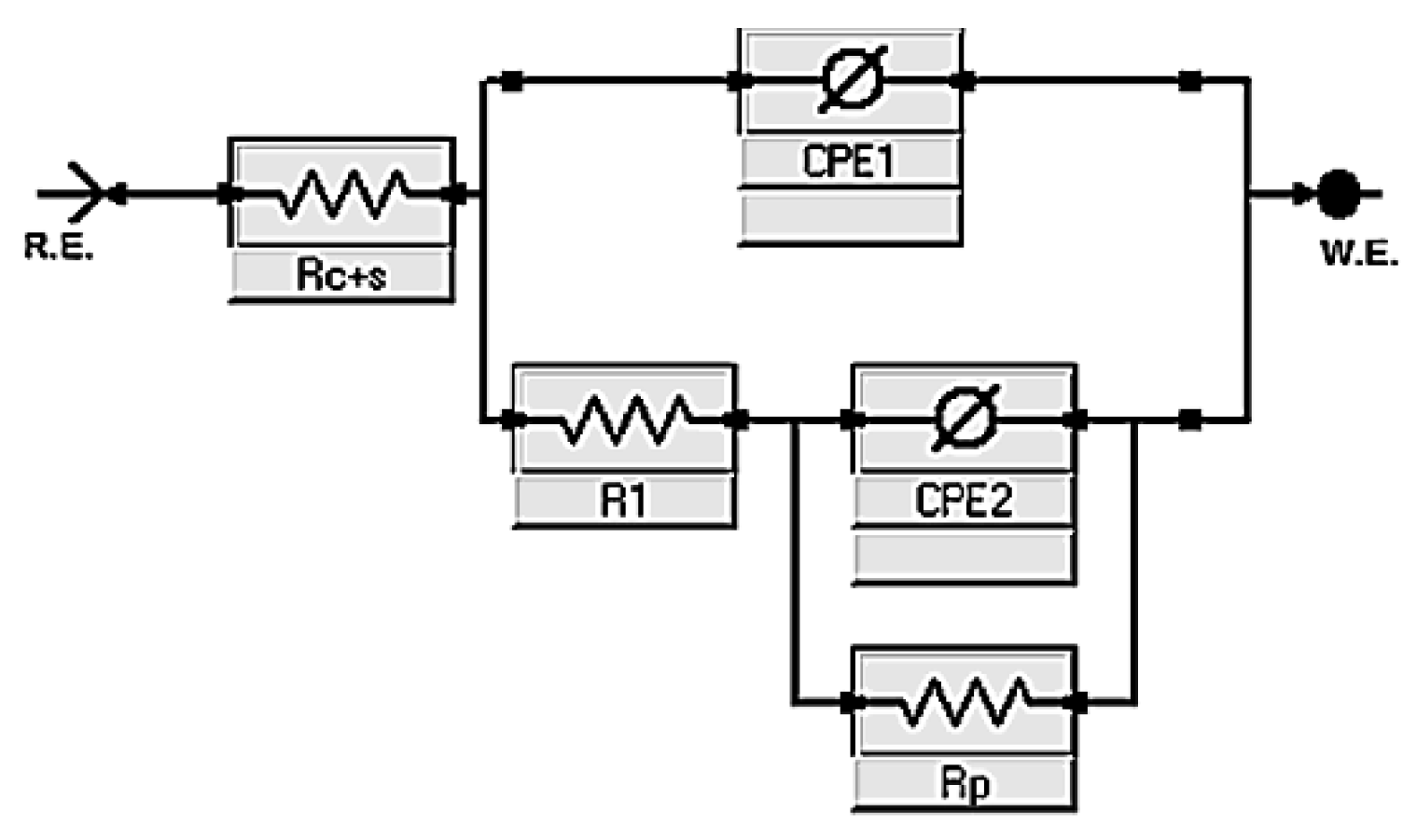
To interpret these findings, the equivalent circuit shown in
Figure 9 was employed. It comprises a solution resistance (Rₛ), two constant phase elements (CPE
1 and CPE
2), and two resistances associated with the interface and diffusion processes (R
1 and Rp). This model enabled the fitting of the impedance data and the extraction of representative system parameters.
The parameters obtained from fitting the equivalent circuit reveal a clear trend with increasing CLDH content. The progressive rise in R
1 and Rp from values below 6 kΩ·cm
2 to over 50 kΩ·cm
2 in the mixture containing 6% CLDH indicates a notable enhancement in polarisation resistance. Simultaneously, the exponents n
1 and n
2 approach values near 0.9, reflecting a transition towards more ideal capacitive behaviour typically associated with homogeneous interfaces and a dense microstructure [
47]. The reductions in C
1 and C
2 values further suggest a decrease in the system’s distributed capacitance, likely because of reductions in structural defects and active pores. In mixtures containing 2% and 4% CLDH, a gradual increase in Rp was observed (reaching approximately 40 kΩ·cm
2) along with a delayed onset of complete carbonation (120 and 160 days, respectively). At these dosages, denser microstructures developed, featuring globular gels and fewer unreacted particles, as confirmed by SEM. MAS-NMR data showed increased gel polymerisation with the emergence of Q
3 units, while XRD detected characteristic peaks of LDH phases [
39]. This structural synergy suggests a marked improvement in system durability, attributed to the “memory effect” of CLDH and its ability to release protective anions such as OH
− and CO
32−.
The optimal content was found to be 6% CLDH, where the Nyquist semicircles were largest and Rp exceeded 55 kΩ·cm
2, indicating a stable passive state. At this level, the carbonation depth was the lowest observed. However, the 8% CLDH mix did not show any significant additional benefit. While the Nyquist semicircle was comparable to that of the 6% mix, with Rp around 56 kΩ·cm
2, complementary analyses indicated a slight reduction in efficiency [
48,
49]. MAS-NMR showed a decrease in the proportion of Q
4 units, XRD revealed the presence of undesirable phases such as unreacted MgO, and SEM displayed a slight decline in compactness. This suggests that beyond 6%, CLDH acts as an inert filler, oversaturating the system without providing further improvements in carbonation resistance or electrochemical performance.
Table 2 presents the values extracted from the equivalent circuit for the various CLDH contents, allowing the quantification of the additive’s effect on the system’s resistive and capacitive components.
4. Discussion
The results of this study provide an integrated understanding of how the incorporation of calcined Mg–Al hydrotalcite (CLDH) influences the mechanical, microstructural, and electrochemical performance of alkali-activated slag concretes (AASC), as well as their resistance to carbonation and the corrosion of embedded steel. This section presents an in-depth analysis that integrates the experimental findings, compares them with the scientific literature, and highlights both the novelty and applicability of the work.
As shown in
Figure 1, the 28-day compressive strength gradually decreases with increasing CLDH content, from 62 MPa in the control mix (0% CLDH) to 41 MPa at 8% CLDH. This trend can be attributed to the dilution effect of the primary reactive precursor (ground granulated blast furnace slag), which reduces the formation of cementitious products. However, the 6% CLDH mix maintains a compressive strength of 48 MPa, equivalent to 75% of the control, making it suitable for moderate structural and non-structural applications.
XRD and MAS-NMR analyses confirm that 6% CLDH optimises the regeneration of lamellar LDH-type phases, promoting the polymerisation of C-A-S-H and N-A-S-H gels. At higher dosages (8%), functional saturation occurs, with the appearance of residual phases such as MgO and a loss of efficiency in generating cementitious products. This behaviour is consistent with recent studies that identify an efficiency threshold for CLDH within the 4–6% range, maximising structural regeneration without inducing inert filler effects [
24,
25]
Moreover, the densification observed in SEM images indicates that CLDH acts as a nucleating agent and promoter of secondary gel formation, which initially favours matrix compaction. However, an excessive dosage leads to local accumulations of unreacted MgO, impairing structural continuity. SEM micrographs show a progressive densification of the matrix as the CLDH content increases, directly correlating with the reduction in the carbonation depth. The 6% CLDH mix exhibits the most homogeneous and compact microstructure, with reduced porosity and interconnected globular gels, which explains its lower CO2 penetration.
The protective mechanism of CLDH is dual: the physical barrier reduces pore connectivity and limits CO2 diffusion, while the controlled release of OH− and CO32− ions restores the local pH and promotes the rehydration of lamellar phases through the “memory effect”.
Unlike the mix without CLDH, which exhibits the highest carbonation after 87 days, the 6% CLDH mix demonstrates a clear improvement in durability, whereas 8% CLDH does not provide significant additional benefits due to functional saturation.
Finally, impedance diagrams and the parameters obtained from the equivalent circuit (
Table 2) confirm a substantial increase in polarisation resistance (Rp) with increasing CLDH content. The optimal 6% mix exceeds 55 kΩ·cm
2, indicating a stable passive state of the embedded steel. The electrochemical enhancement is attributed to improved homogeneity at the steel–paste interface and reduced interconnecting porosity.
These findings have high practical relevance for precast or non-structural elements exposed to urban or industrial environments rich in CO2, as well as for AASC systems where carbonation resistance is critical to minimise maintenance costs.
To strengthen knowledge in this field, future studies will evaluate intermediate dosages (1%, 3%, 5%, and 7%) to more precisely map the effect of CLDH, as well as conduct long-term durability tests under natural conditions, including wet–dry cycles and combined exposure to CO2 and chlorides.
While the results presented in this study provide valuable insights into the influence of calcined Mg–Al hydrotalcite (CLDH) on the mechanical, microstructural, and electrochemical performance of alkali-activated slag concretes (AASC), several limitations should be acknowledged. First, the experimental programme was restricted to a specific range of CLDH dosages (0–8%) and a single activator type, which may limit the generalisability of the findings to other binder compositions or activation systems. Second, all tests were carried out under controlled laboratory curing and accelerated carbonation conditions, which do not fully replicate the variability of natural environmental exposures. Third, microstructural characterisation was performed at selected ages, and intermediate microstructural changes during carbonation were not monitored in detail. Finally, this study did not investigate other deterioration mechanisms, such as chloride ingress, freeze–thaw damage, or combined actions, which could interact with carbonation in real service conditions. Addressing these aspects in future work will help provide a more comprehensive understanding of the long-term performance of CLDH-modified AASC.
5. Conclusions
The incorporation of CLDH slightly affects the mechanical performance of AASC. The 28-day compressive strength decreased progressively from 62 MPa for the reference mixture to 41 MPa at 8% CLDH, reflecting the dilution of reactive slag. Nevertheless, the 6% CLDH mixture maintained 48 MPa, representing more than 75% of the control strength, which is sufficient for non-structural and moderate structural applications. These results confirm that the durability improvements provided by CLDH can be achieved without a critical loss of mechanical integrity.
The progressive incorporation of CLDH into AASC pastes significantly reduces the susceptibility of embedded steel to carbonation-induced corrosion. This is evidenced by the continuous increase in polarisation resistance (Rp), exceeding 55 kΩ·cm2 with 6% CLDH, and the marked reduction in the carbonation depth (down to 25.4 mm after 87 days of exposure).
A dosage of 6% CLDH is identified as optimal, as it results in a dense and continuous microstructure that restricts CO2 diffusion and maintains a high pH in the vicinity of the reinforcement. This prevents depassivation and promotes a stable electrochemical passive state, as shown by the wide Nyquist semicircles.
The parameters extracted from the equivalent circuit model (notably the increases in R1 and Rp, and n1 and n2 values approaching 0.9) reflect a transition towards more homogeneous and less defective interfaces, indicating reduced structural degradation and enhanced electrochemical stability under aggressive environmental conditions.
Although 8% CLDH maintains a high Rp (~56 kΩ·cm2), MAS-NMR and SEM results suggest a slight loss in structural efficiency. Residual phases such as unreacted MgO and less compact microstructures were detected, which may compromise long-term durability under advanced degradation processes.
CLDH functions not only as a physical barrier to CO2 ingress but also as a source of protective anions (OH− and CO32−) through its memory effect. This dual action enhances the system’s chemical resilience and mitigates degradation induced by carbonation agents under simulated accelerated exposure conditions.
Author Contributions
Conceptualization, W.A. and J.C.C.; methodology, W.A.; software, J.A.; validation, W.A., J.A. and J.C.C.; formal analysis, W.A.; investigation, W.A.; resources, J.C.C.; data curation, J.A.; writing—original draft preparation, W.A.; writing—review and editing, J.C.C.; visualization, J.A.; supervision, J.C.C.; project administration, J.C.C.; funding acquisition, J.C.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The data presented in this study are available on request from the corresponding author due to data volume and confidentiality.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Lloyd, N.; Collins, F.G. Carbon Dioxide Equivalent (CO2-e) Emissions: A Comparison between Geopolymer and OPC Cement Concrete. Constr. Build. Mater. 2013, 43, 125–130. [Google Scholar] [CrossRef]
- Jawad, U.; Ali, M.; Cheema, A.; Sultan, S.M.; Akhtar, M.J. Green Cement Valuation: An Optimistic Approach to Carbon Dioxide Reduction. J. Appl. Eng. Sci. 2023, 13, 259–268. [Google Scholar] [CrossRef]
- Nagajothi, S.; Elavenil, S.; Angalaeswari, S.; Natrayan, L.; Mammo, W.D. Durability Studies on Fly Ash Based Geopolymer Concrete Incorporated with Slag and Alkali Solutions. Adv. Civ. Eng. 2022, 2022, 7196446. [Google Scholar] [CrossRef]
- Rao, M.T. Fresh and Mechanical Characteristics of Alkali Activated Fly Ash Slag Concrete Activated with Neutral Grade Liquid Glass. Int. J. Res. Appl. Sci. Eng. Technol. 2023, 11, 1136–1145. [Google Scholar] [CrossRef]
- Luga, E.; Mustafaraj, E.; Corradi, M.; Atiş, C.D. Alkali-Activated Binders as Sustainable Alternatives to Portland Cement and Their Resistance to Saline Water. Materials 2024, 17, 4408. [Google Scholar] [CrossRef]
- Nguyen, Q.D.; Castel, A. Developing Geopolymer Concrete by Using Ferronickel Slag and Ground-Granulated Blast-Furnace Slag. Ceramics 2023, 6, 114. [Google Scholar] [CrossRef]
- Barragán-Ramírez, R.; González-Hernández, A.; Bautista-Ruiz, J.; Ospina, M.; Aperador, W. Enhancing Concrete Durability and Strength with Fly Ash, Steel Slag, and Rice Husk Ash for Marine Environments. Materials 2024, 17, 3001. [Google Scholar] [CrossRef]
- Shagñay, S.; Bautista, A.; Velasco, F.; Ramón-Álvarez, I.; Torres-Carrasco, M. Influence of the Early-Age Length Change of Alkali-Activated Slag Mortars on the Corrosion of Embedded Steel. J. Sustain. Cem.-Based Mater. 2023, 13, 178–195. [Google Scholar] [CrossRef]
- Behfarnia, K.; Rostami, M. An Assessment on Parameters Affecting the Carbonation of Alkali-Activated Slag Concrete. J. Clean. Prod. 2017, 157, 1–9. [Google Scholar] [CrossRef]
- Montoya, R.; Aperador, W.; Bastidas, D.M. Influence of Conductivity on Cathodic Protection of Reinforced Alkali-Activated Slag Mortar. Sustainability 2023, 15, 12404. [Google Scholar] [CrossRef]
- Saha, T. Comparison of Testing Methods for Evaluating the Resistance of Alkali-Activated Blast Furnace Slag Systems to Sulfur Dioxide. Materials 2022, 15, 1344. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Yongning, L.; Ji, T.; Lu, Y.; Lin, X. Effect of activator types and concentration of CO2 on the steel corrosion in the carbonated alkali-activated slag concrete. Constr. Build. Mater. 2020, 262, 120044. [Google Scholar] [CrossRef]
- Al-Duais, I.N.A.; Ahmad, S.; Al-Osta, M.A.; Maslehuddin, M.; Ibrahim, M.M. Durability of Alkali-Activated Concrete Made Using Multiple Precursors as Primary Binders. J. Sustain. Cem.-Based Mater. 2024, 13, 1483–1501. [Google Scholar] [CrossRef]
- Chen, S.J.; Li, W.G.; Ruan, C.K.; Sagoe-Crentsil, K.; Duan, W.H. Pore shape analysis using centrifuge driven metal intrusion: Indication on porosimetry equations, hydration and packing. Constr. Build. Mater. 2017, 154, 95–104. [Google Scholar] [CrossRef]
- Chen, Y.; Xiong, X.; Wang, D.; Zhou, X.; Xu, Z. The Risk of Alkali–Carbonate Reaction and the Freeze–Thaw Resistance of Waste Dolomite Slag-Based Concrete. Buildings 2024, 14, 1664. [Google Scholar] [CrossRef]
- Zhu, Y.; Jia, Y.; Xie, X.S.; Xu, J. Study on the Macroscopic Properties and Microstructure of High Fly Ash Content Alkali-Activated Fly Ash Slag Concrete Cured at Room Temperature. Materials 2025, 18, 547. [Google Scholar] [CrossRef]
- Quan, X.; Zhao, G.; Quan, X.; Liu, X.; Bu, M. Investigation on the Sodium Silicate Modulus-Affected Macroscopic and Microscopic Characteristics of Alkali-Activated Slag Concrete. Mater. Sci. Technol. 2025, 1, 1–16. [Google Scholar] [CrossRef]
- Kaliyavaradhan, S.K.; Unnikrishnan, A.K.; Ambily, P.S. CO2 sequestration through alkali-activated steel slag: A review. J. Sustain. Cem.-Based Mater. 2024, 14, 1042–1058. [Google Scholar] [CrossRef]
- Tsai, C.-J.; Hua, C.; Fazeldehkordi, L.; Shyu, W.-S. Exploring the Potential and Strength Characteristics of Waste Sodium Silicate-Bonded Sand for Sustainable Application in Alkali-Activated Slag Concrete. J. Sustain. Cem.-Based Mater. 2024, 14, 265–277. [Google Scholar] [CrossRef]
- Iqbal, M.A.; Ojala, T.; Al-Neshawy, F.; Punkki, J. Effect of Slag Content and Carbonation/Ageing on Freeze–Thaw Resistance of Concrete. Nord. Concr. Res. 2024, 71, 47–67. [Google Scholar] [CrossRef]
- Criado, M.; Sobrados, I.; Sanz, J. Alkali Activated Slag Cement Pastes: Effect of the Activation Conditions on the Microstructure. Cem. Concr. Compos. 2018, 92, 68–76. [Google Scholar] [CrossRef]
- Vu, D.H.; Dang, L.C.; Kang, G.; Sirivivatnanon, V. Chloride-Induced Corrosion of Steel Reinforcement in Alkali-Activated Materials: A Review. Constr. Build. Mater. 2022, 345, 128354. [Google Scholar] [CrossRef]
- Lamaa, G.; Duarte, A.P.; Silva, R.V.; de Brito, J. Carbonation of Alkali-Activated Materials: A Review. Materials 2023, 16, 3086. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, F.; Long, Y.; Yu, L.; Yang, C.; Wen, M.; Yu, X.; Tian, Y.; Zhao, M. Improving the Carbonation Resistance of Alkali-Activated Slag Mortars by Calcined Mg/Al Layered Double Hydroxides. Appl. Clay Sci. 2022, 216, 106379. [Google Scholar] [CrossRef]
- Zhang, J.; Ma, Y.; Zhang, Z.; Yang, X.; Nong, X.; Wang, H. Effects of Mg-Based Admixtures on Chloride Diffusion in Alkali-Activated Fly Ash-Slag Mortars. Case Stud. Constr. Mater. 2024, 21, e03659. [Google Scholar] [CrossRef]
- ASTM A29/A29M–20; Standard Specification for General Requirements for Steel Bars, Carbon and Alloy, Hot-Wrought. ASTM International: West Conshohocken, PA, USA, 2020. Available online: https://www.astm.org/a0029_a0029m-20.html (accessed on 28 March 2025).
- ASTM C33/C33M–18; Standard Specification for Concrete Aggregates. ASTM International: West Conshohocken, PA, USA, 2018. Available online: https://www.astm.org/c0033_c0033m-18.html (accessed on 5 April 2025).
- ASTM C39/C39M-15a; Standard Test Method for Compressive Strength of Cylindrical Concrete Specimens. ASTM International: West Conshohocken, PA, USA, 2015. Available online: https://www.astm.org/c0039_c0039m-15a.html (accessed on 3 June 2025).
- Aperador, W.; Martínez-Bastidas, D.; Bautista-Ruiz, J.H. Mechanical Properties and Absorption of Chlorides in Alkali-Activated Slag Concrete Exposed to Carbonation. Rev. Fac. Ing. Univ. Antioq. 2012, 62, 189–195. [Google Scholar] [CrossRef]
- Ramezanianpour, A.A.; Moeini, M.A. Mechanical and Durability Properties of Alkali Activated Slag Coating Mortars Containing Nanosilica and Silica Fume. Constr. Build. Mater. 2018, 163, 611–621. [Google Scholar] [CrossRef]
- Zhang, Z.; Huang, Z.; Shi, C.; Zhu, Y.; Zhou, Z.; Jia, X.; Ren, Q.; Jiang, Z. Early Hydration and Microstructure Formation of Ultra-Rapid Hardening Alkali-Activated Slag Cement (URHA) in the Presence of MgO. SSRN 2025, 160, 106057. [Google Scholar] [CrossRef]
- Pavel, O.D.; Manyar, H. Editorial: Layered Double Hydroxides and Their Use as Catalysts in Sustainable Processes. Front. Chem. Eng. 2024, 6, 1534838. [Google Scholar] [CrossRef]
- Nieto-Zambrano, S.; Ramos-Ramirez, E.E.; Tzompantzi, F.; Gutiérrez Ortega, N.L. A XRD Study of the CuAl Layered Double Hydroxide Synthesis Evolution. Mater. Devices 2024, 8, 1408–1416. [Google Scholar] [CrossRef]
- Ke, X.; Bernal, S.A.; Provis, J.L. Controlling the reaction kinetics of sodium carbonate-activated slag cements using calcined layered double hydroxides. Cem. Concr. Res. 2016, 81, 24–37. [Google Scholar] [CrossRef]
- Chen, W.; Li, B.; Thom, N. Effects of Alkali Dosage and Silicate Modulus on Autogenous Shrinkage of Alkali-Activated Slag Cement Paste. Cem. Concr. Res. 2021, 140, 106322. [Google Scholar] [CrossRef]
- Fang, G.; Wang, Q.; Zhang, M. Micromechanical Analysis of Interfacial Transition Zone in Alkali-Activated Fly Ash-Slag Concrete. Cem. Concr. Compos. 2021, 119, 103990. [Google Scholar] [CrossRef]
- Krisková, L.; Ducman, V.; Lončar, M.; Tesovnik, A.; Žibret, G.; Skentzou, D.; Georgopoulos, C. Alkali-Activated Mineral Residues in Construction: Case Studies on Bauxite Residue and Steel Slag Pavement Tiles. Materials 2025, 18, 257. [Google Scholar] [CrossRef]
- Li, L.; Darquennes, A.; Hannawi, K.; Che, C. Effect of the Alkali-Sulphate Activators on the Hydration Process of Blast-Furnace Slag Mortars and Pastes. Materials 2025, 18, 514. [Google Scholar] [CrossRef]
- Han, B.; Cai, L.; Chi, F.; Zhan, W. Impact of Metakaolin to Partially Replace Granulated Blast Furnace Slag on the Performance of Alkali-Activated Slag Grouting Materials and Evaluation of Grouting Effectiveness. Mater. Res. Express 2025, 12, 025507. [Google Scholar] [CrossRef]
- Chen, L.; Wei, L.; Zheng, J.; Zhou, J. Study on the Inhibition Effect of Fly Ash on Alkali–Silica Reaction and Its Influence on Building Energy Performance. Buildings 2025, 15, 392. [Google Scholar] [CrossRef]
- Kong, L.; Sun, S.; Wang, X.; Liu, Y. Mechanism of chloride binding in alkali-activated materials exposed to combined chloride, sulfate and carbonation environment. J. Sustain. Cem.-Based Mater. 2025, 14, 613–626. [Google Scholar] [CrossRef]
- Bai, W.; Ye, D.; Song, Y.-M.; Yuan, C.; Guan, J.; Yang, G.; Xie, C. Study on Mechanical Properties and Damage Mechanism of Alkali-Activated Slag Concrete. J. Build. Eng. 2024, 96, 110357. [Google Scholar] [CrossRef]
- Hussain, Z.; Lin, Z.; Pan, H.; Huang, Y.; Tang, F.; Jiang, L. Synergizing Empirical and AI Methods to Examine Nano-Silica’s Microscale Contribution to Epoxy Coating Corrosion Resistance. Ceram. Int. 2024, 50, 47172–47191. [Google Scholar] [CrossRef]
- Ulubeyli, G.Ç.; Artır, R. Synthesis and Characterization of an Alkali-Activated Binder from Blast Furnace Slag and Marble Waste. Materials 2024, 17, 5248. [Google Scholar] [CrossRef] [PubMed]
- Wen, N.; Hallet, V.; Peys, A.; Pontikes, Y. Degradation of Alkali-Activated Fe-Rich Slag in Magnesium Sulfate Solution. J. Am. Ceram. Soc. 2025, 108, e20211. [Google Scholar] [CrossRef]
- Liang, Q.; Huang, X.; Zhang, L.; Yang, H. A Review on Research Progress of Corrosion Resistance of Alkali-Activated Slag Cement Concrete. Materials 2024, 17, 5065. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, Y.; Zhang, J.; Li, Y. Compressive Strength and Resistance to Sulphate Attack of Ground Granulated Blast Furnace Slag, Lithium Slag, and Steel Slag Alkali-Activated Materials. Buildings 2024, 14, 2320. [Google Scholar] [CrossRef]
- Aperador, W.; Bautista-Ruiz, J.; Sánchez-Molina, J. Effect of Immersion Time in Chloride Solution on the Properties of Structural Rebar Embedded in Alkali-Activated Slag Concrete. Metals 2022, 12, 1952. [Google Scholar] [CrossRef]
- Liu, P.; Chen, G.; Liu, G.; Liu, H.; Zhang, J.; Chen, P.; Su, Y. Study on Impact Resistance of Alkali-Activated Slag Cementitious Material with Steel Fiber. Buildings 2024, 14, 3442. [Google Scholar] [CrossRef]
Figure 1.
Twenty-eight day compressive strength of alkali-activated slag concrete with 0–8% CLDH replacement by binder mass.
Figure 1.
Twenty-eight day compressive strength of alkali-activated slag concrete with 0–8% CLDH replacement by binder mass.
Figure 2.
X-ray diffraction (XRD) patterns of alkali-activated slag (AAS) pastes with varying proportions of calcined layered double hydroxides (CLDH) as a partial slag replacement (0%, 2%, 4%, 6%, and 8%).
Figure 2.
X-ray diffraction (XRD) patterns of alkali-activated slag (AAS) pastes with varying proportions of calcined layered double hydroxides (CLDH) as a partial slag replacement (0%, 2%, 4%, 6%, and 8%).
Figure 3.
X-ray diffraction (XRD) pattern of the sample, with the annotated peaks corresponding to layered double hydroxide (LDH), magnesium oxide (MgO), and magnesium aluminate spinel (MgAl2O4). The primary LDH reflections are observed at (003), (006), and (015), while MgO and MgAl2O4 peaks are indexed at their respective crystallographic planes.
Figure 3.
X-ray diffraction (XRD) pattern of the sample, with the annotated peaks corresponding to layered double hydroxide (LDH), magnesium oxide (MgO), and magnesium aluminate spinel (MgAl2O4). The primary LDH reflections are observed at (003), (006), and (015), while MgO and MgAl2O4 peaks are indexed at their respective crystallographic planes.
Figure 4.
29Si MAS-NMR spectra of alkali-activated slag (AAS) pastes with varying contents of calcined layered double hydroxide (CLDH): 0%, 2%, 4%, 6%, and 8%.
Figure 4.
29Si MAS-NMR spectra of alkali-activated slag (AAS) pastes with varying contents of calcined layered double hydroxide (CLDH): 0%, 2%, 4%, 6%, and 8%.
Figure 5.
27Al MAS-NMR spectra of alkali-activated slag (AAS) pastes with varying proportions of calcined layered double hydroxide (CLDH): 0%, 2%, 4%, 6%, and 8%.
Figure 5.
27Al MAS-NMR spectra of alkali-activated slag (AAS) pastes with varying proportions of calcined layered double hydroxide (CLDH): 0%, 2%, 4%, 6%, and 8%.
Figure 6.
SEM micrographs and corresponding EDS spectra of the fractured surfaces of alkali-activated slag (AAS) pastes with varying contents of calcined layered double hydroxide (CLDH): (a) 0%, (b) 2%, (c) 4%, (d) 6%, and (e) 8%.
Figure 6.
SEM micrographs and corresponding EDS spectra of the fractured surfaces of alkali-activated slag (AAS) pastes with varying contents of calcined layered double hydroxide (CLDH): (a) 0%, (b) 2%, (c) 4%, (d) 6%, and (e) 8%.
Figure 7.
Phenolphthalein-stained cross-sections of AASC pastes with varying CLDH contents after 87 days of accelerated carbonation: (a) 0%, (b) 2%, (c) 4%, (d) 6%, and (e) 8%.
Figure 7.
Phenolphthalein-stained cross-sections of AASC pastes with varying CLDH contents after 87 days of accelerated carbonation: (a) 0%, (b) 2%, (c) 4%, (d) 6%, and (e) 8%.
Figure 8.
Electrochemical impedance spectroscopy (EIS) results for alkali-activated slag (AAS) concretes with varying proportions of calcined layered double hydroxide (CLDH): 0%, 2%, 4%, 6%, and 8%. (
a) Nyquist plots. (
b) Corresponding Bode plots showing the variation in the impedance magnitude and phase angle with frequency. In the reference mixture (0% CLDH), a small semicircle is observed, with polarisation resistance (Rp) values below 6 kΩ·cm
2, indicating a condition of active corrosion. This behaviour aligns with a carbonation depth of 50 mm after just 87 days, suggesting a chemically degraded environment [
37]. MAS-NMR results revealed a poorly polymerised structure dominated by Q
1–Q
2 units, while XRD analysis showed a matrix with minimal formation of protective phases, such as hydrotalcites or well-developed gels. SEM micrographs confirmed significant porosity and discontinuous zones, which facilitated CO
2 ingress and the depassivation of the embedded steel.
Figure 8.
Electrochemical impedance spectroscopy (EIS) results for alkali-activated slag (AAS) concretes with varying proportions of calcined layered double hydroxide (CLDH): 0%, 2%, 4%, 6%, and 8%. (
a) Nyquist plots. (
b) Corresponding Bode plots showing the variation in the impedance magnitude and phase angle with frequency. In the reference mixture (0% CLDH), a small semicircle is observed, with polarisation resistance (Rp) values below 6 kΩ·cm
2, indicating a condition of active corrosion. This behaviour aligns with a carbonation depth of 50 mm after just 87 days, suggesting a chemically degraded environment [
37]. MAS-NMR results revealed a poorly polymerised structure dominated by Q
1–Q
2 units, while XRD analysis showed a matrix with minimal formation of protective phases, such as hydrotalcites or well-developed gels. SEM micrographs confirmed significant porosity and discontinuous zones, which facilitated CO
2 ingress and the depassivation of the embedded steel.
Figure 9.
Proposed equivalent circuit used to fit the electrochemical impedance spectroscopy (EIS) spectra for alkali-activated slag (AAS) pastes containing CLDH, comprising resistive elements (Rc+s, R1, and Rp) and constant phase elements (CPE1 and CPE2) representing transport and polarization processes.
Figure 9.
Proposed equivalent circuit used to fit the electrochemical impedance spectroscopy (EIS) spectra for alkali-activated slag (AAS) pastes containing CLDH, comprising resistive elements (Rc+s, R1, and Rp) and constant phase elements (CPE1 and CPE2) representing transport and polarization processes.
Table 1.
Chemical compositions of both materials analysed via XRF.
Table 1.
Chemical compositions of both materials analysed via XRF.
| | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | SO3 | MnO | K2O | P2O5 |
|---|
| GBFS | 34.8 | 15.5 | 2.4 | 37.4 | 2.3 | 1.4 | 3.8 | 0.4 | 0.2 |
| CLDH | 1.5 | 24.5 | - | 0.5 | 65.0 | - | - | - | - |
Table 2.
Parameters extracted from the equivalent electrical circuit model for AASC mixtures with varying CLDH contents, including solution resistance (Rc+s), interface resistance (R1), polarization resistance (Rp), and constant phase elements (CPE1 and CPE2).
Table 2.
Parameters extracted from the equivalent electrical circuit model for AASC mixtures with varying CLDH contents, including solution resistance (Rc+s), interface resistance (R1), polarization resistance (Rp), and constant phase elements (CPE1 and CPE2).
| Material | Rc+s
Ω cm2 | R1
Ω cm2 | CPE1
μF cm−2 | n1 | Rp
Ω cm2 | CPe2
μF cm−2 | n2 |
|---|
| 0% CLDH | 5 | 489 | 60 | 0.82 | 68,211 | 484 | 0.72 |
| 2% CLDH | 12 | 1421 | 51 | 0.84 | 13,852 | 425 | 0.81 |
| 4% CLDH | 20 | 2359 | 42 | 0.87 | 32,654 | 385 | 0.86 |
| 6% CLDH | 30 | 9542 | 36 | 0.91 | 54,657 | 354 | 0.92 |
| 8% CLDH | 42 | 12,058 | 34 | 0.93 | 57,874 | 332 | 0.91 |
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).