Data Mining Applied to the Electrochemical Noise Technique in the Time/Frequency Domain for Stress Corrosion Cracking Recognition
Abstract
:1. Introduction
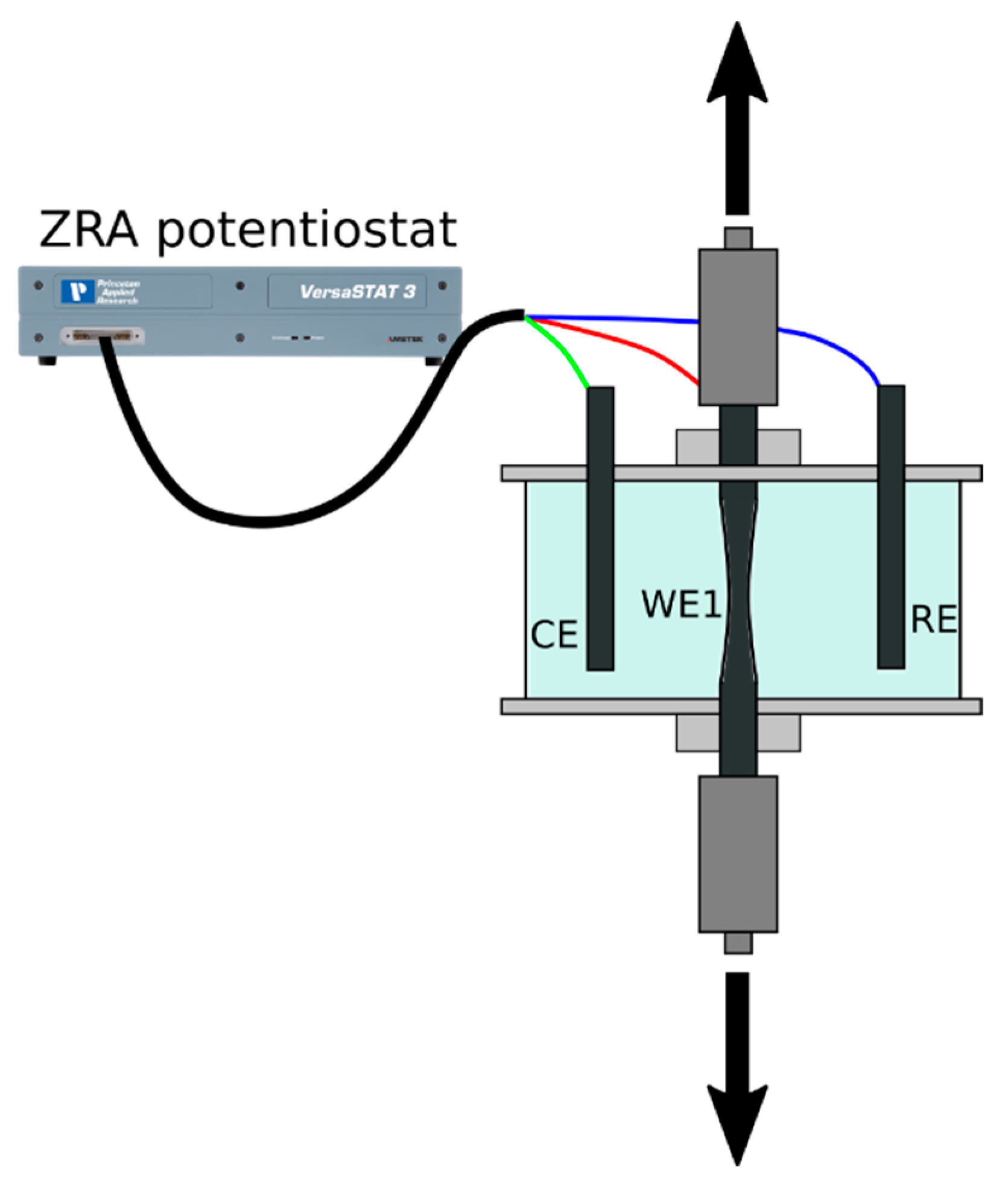
2. Materials and Methods
2.1. Materials and Testing Conditions
2.2. Hilbert–Huang Transform Approach
- Executing the sifting process on the s(t), obtaining an IMF denominated as dn(t).
- Calculating the residual r(t) = sn−1(t) − dn(t) (then, this residual is considered a new s(t)).
- Extracting relative extremes, obtaining two functions: v1(t) is the interpolation of the maxima point, and v2(t) is the interpolation of the minima point
- Determining the m(t) function as m(t) = 1/2(v1(t) + v2(t))
- Extracting d(t) = s(t) − m(t)
3. Results and Discussion
3.1. Time Domain Analysis
3.2. Frequency Domain Analysis
3.3. Time-Frequency Domain Analysis
- At first, up to 1500 s, a low electrochemical activity region can be identified. As previously discussed, this stage could be related with the pre-activation corrosion phase. At very short times the electrochemical activity is not representative. However, with increasing time, the intensity of the IMF progressively increases. The first intensity and wide peak, point A in Figure 10, occurs at about 1000 s, in correspondence with the triggering of metastable pits [61]. Consequently, this phase can be associated with the superficial electrochemical activation of the specimen [62].
- The pre-activation stage continues to evolve during time, with the presence of several sub-peaks (point B in Figure 10), up to about 2000 s, in correspondence with the activation and propagation of superficial pits. Subsequently the IMF signal undergoes a sharp reduction in intensity, during which there are only occasional signal spikes (point B’ in Figure 10). During this time, in the time interval 2000–10,000 s, predominantly, the pit-to-crack transition takes place. The triggering of the SCC phenomenon obviously stimulates the defect evolution partly by mechanical contribution making therefore the electrochemical one less significant. The occasional secondary IMF peaks, identified by point B’, may be associated with the local formation and propagation of additional surface pits that evolve simultaneously during the stress corrosion triggering phase.
- After about 10,000 s a third stage was defined as short to long range crack propagation can be identified. During this stage several fluctuations in IMS signal can be highlighted. In particular After a long electrochemical quiescence step (point C in Figure 10) a significant peak can be identified at about 16,000–18,000 s (point C’ in Figure 10). This trend related to stabilization and abrupt increase steps in IMF noise current signal is periodically identifiable. This behaviour is caused by the crack evolution by SCC [36]. At this stage, the size of the crack can reach a length sufficient to assume that the stress concentration at the crack tip becomes significant, thus implying that there is a large area of plastic deformation, leading to a more extensive blunt at the crack tip. Due to the crack reshape, the mechanical crack evolution mechanism is inhibited, despite electrochemical dissolution [48]. Consequently, a phase of electrochemical stabilization associated with the mechanical evolution of SCC damage is complementary to a phase of metal dissolution at the crack tip, and, therefore, complementary to an electrochemical activity phase. Depending on various electrochemical dissolution factors, the current transient generated by the micro-crack opening and re-passivation can last for a few seconds [63]. These sub-steps alternate cyclically during this stage (points C* in Figure 10), where the sub-critical propagation of the medium and long-range crack takes place.
- After about 45,000 s, after a transient region (coded as pre-quiescence), a large electrochemical plateau in the IMF signal can be identified (above 48,000 s). During this step, no relevant electrochemical vents can be identified. This stage is the prelude for the critical failure of the sample that took place after about 58,000s.
- Stabilization: at first, in the rage 0–100 s (step I in Figure 11), the noise signal is not characterised by any significant fluctuations. EN transient related to this phenomenon is characterised by the absence of high-frequency events: only some events at about 10−3 Hz can be observed. In this phase, the interaction between the electrolyte and specimen surface takes place, with an unstable alternation of general corrosion and passivation phases. The aggressive ions locally destabilize the passive oxide layer of the sample, causing a local thinning of the oxide. This process has a short duration (approximately 100 s) due to the high aggressiveness of the environment conditions of the test.
- Electrochemical activation: in the range 100–1500 s (step II in Figure 11), in correspondence with depassivated surface regions, the electrochemical activity is enhanced and the pit initiation stage extends to micrometre-sized, large dissolved holes. In this phase, an increase in the cumulative charge trend was also identified. This sub-stage is identified by a high activity at medium and low frequencies. In particular, at increasing time (after about 500 s) and with a progressive increment in the number of pits on the surface, the signal activity evolves towards a higher frequency (from 2 × 10−2 to 5 × 10−1 Hz, defining two substep: IIa and Iib). This stage can be related with the electrochemical activation phase (Iia) and the subsequent triggering of metastable pits (Iib) on the sample surface [14].
- SCC Activation and Propagation: This region, ranging from 1500 to 10,000 s, can be related mainly with the SSC triggering by short range crack activation. At this stage, the electrochemical activity becomes significant. Some sub-clusters can be identified. At first, region IIIa, as shown in Figure 11, suggests that high-frequency contribution can be identified. At this stage, the aggressive ions locally destabilize the passive oxide layer of the sample, causing a local thinning of the oxide. In correspondence with depassivated surface regions, the electrochemical activity is enhanced and the pit initiation stage extends to micrometre-sized, large dissolved holes (see pit in Figure 12d). This process leads to the formation of preferential areas of localized attacks by pitting, identifiable by a high magnitude of the IMF. Progressively, the signal activity evolves towards a lower frequency (from 5 × 10−3 to 5 × 10−2 Hz) and magnitude (from 100.4 to 10−0.4 dB), as shown via region IIIb in Figure 11. In this low frequency phase, the mechanical contribution is more relevant than the electrochemical one, as evidenced by the low EN signal magnitude, indicating that SCC short-range activation of the cracks was triggered [14].
- SCC propagation. At about 10,000 s, a new step begins. During this step, the SCC damage evolution phase of the sample occurs (in Figure 12c, it is possible to note a secondary crack originating from a pit). The progressive crack growth (region III) leads to an increase in the region of plastic deformation at the crack tip [64]. This implies that a more severe anodic dissolution is needed by the crack to re-sharpen before inducing a further propagation. At this phase, in fact, the dissolution within the crack is still taking place. When increasing the crack length, an increase in stress concentration occurs, which induces a larger plastic zone ahead of the crack tips. As a result, the crack point becomes more blunted. A larger plastic zone suggests that it will take more time for the fracture to be re-sharpened via dissolving for future crack propagation. This phase was indeed indicated as a mechanical quiescence [53] and the driving force in crack propagation. This stage shows metal dissolution at the crack tip. This region could correspond to the high-frequency activity in the first IMF. The electrochemical dissolution (sub-steps IVa in Figure 11), identifiable via the step-wise increase in cumulative charge (Figure 5), stimulates the mechanical instability of the crack (sub-steps IVb in Figure 11), primed for future subsonic propagation. Consequently, this stage identifies the prelude to the catastrophic failure of the specimen. In the topological map, this region is identified by medium-low amplitude valleys alternating with high-frequency peaks (5 × 10−2–3 × 10−1 Hz). Afterwards, for times above 45,000 s, the Hilbert spectrum shows a low magnitude region where an electrochemical quiescence occurs. Finally, for catastrophic failure, the sample takes place after about 58,000s.
4. Conclusions
- Stabilization: at first, in the rage 0–100 s. The EN transient is characterised by the absence of high-frequency events. Only some events at about 10−3 Hz were observed.
- Electrochemical activation: in the range 100–1500 s. This stage was identified by a high activity at medium and low frequencies (from 2 × 10−2 to 5 × 10−1 Hz,). This stage can be related with electrochemical activation and the subsequent triggering of metastable pits on the sample surface.
- SCC Activation and Propagation: ranging from 1500 to 10,000 s. This is mainly related to the SSC triggering by short-range crack activation. At this stage, the electrochemical activity became significantly identifiable by a high magnitude of IMF. Then, progressively, the signal activity evolved towards a lower frequency (from 5 × 10−3 to 5 × 10−2 Hz) and magnitude (from 100.4 to 10−0.4 dB), because the mechanical contribution was more relevant than the electrochemical.
- SCC propagation. Above 10,000 s. This is related to progressive crack growth. This region was identified by medium–low amplitude valleys alternating with high-frequency peaks (5 × 10−2–3 × 10−1 Hz). For times above 45,000 s a low magnitude region, related to an electrochemical quiescence, was identified before catastrophic failure of the samples.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Benavides, S. Corrosion Control in the Aerospace Industry; Woodhead Publishing: Cambridge, UK, 2009. [Google Scholar]
- Rao, B.P.C.; Raj, B. NDE Methods for Monitoring Corrosion and Corrosion-Assisted Cracking. In Non-Destructive Evaluation of Corrosion and Corrosion-Assisted Cracking; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2019; pp. 101–121. ISBN 9781118987735. [Google Scholar]
- Calabrese, L.; Proverbio, E. A Review on the Applications of Acoustic Emission Technique in the Study of Stress Corrosion Cracking. Corros. Mater. Degrad. 2021, 2, 1–30. [Google Scholar] [CrossRef]
- Cai, W.; Jomdecha, C.; Zhao, Y.; Wang, L.; Xie, S.; Chen, Z. Quantitative Evaluation of Electrical Conductivity inside Stress Corrosion Crack with Electromagnetic NDE Methods. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2020, 378, 20190589. [Google Scholar] [CrossRef] [PubMed]
- Xia, D.-H.; Song, S.-Z.; Behnamian, Y. Detection of Corrosion Degradation Using Electrochemical Noise (EN): Review of Signal Processing Methods for Identifying Corrosion Forms. Corros. Eng. Sci. Technol. 2016, 51, 527–544. [Google Scholar] [CrossRef]
- Lv, J.; Yue, Q.; Ding, R.; Wang, X.; Gui, T.; Zhao, X. The Application of Electrochemical Noise for the Study of Metal Corrosion and Organic Anticorrosion Coatings: A Review. ChemElectroChem 2021, 8, 337–351. [Google Scholar] [CrossRef]
- Xia, D.-H.; Song, S.; Behnamian, Y.; Hu, W.; Cheng, Y.F.; Luo, J.-L.; Huet, F. Review—Electrochemical Noise Applied in Corrosion Science: Theoretical and Mathematical Models towards Quantitative Analysis. J. Electrochem. Soc. 2020, 167, 081507. [Google Scholar] [CrossRef]
- Xia, D.-H.; Deng, C.-M.; Macdonald, D.; Jamali, S.; Mills, D.; Luo, J.-L.; Strebl, M.G.; Amiri, M.; Jin, W.; Song, S.; et al. Electrochemical Measurements Used for Assessment of Corrosion and Protection of Metallic Materials in the Field: A Critical Review. J. Mater. Sci. Technol. 2022, 112, 151–183. [Google Scholar] [CrossRef]
- Lau, K.; Permeh, S. Assessment of Durability and Zinc Activity of Zinc-Rich Primer Coatings by Electrochemical Noise Technique. Prog. Org. Coat. 2022, 167, 106840. [Google Scholar] [CrossRef]
- Obot, I.B.; Onyeachu, I.B.; Zeino, A.; Umoren, S.A. Electrochemical Noise (EN) Technique: Review of Recent Practical Applications to Corrosion Electrochemistry Research. J. Adhes. Sci. Technol. 2019, 33, 1453–1496. [Google Scholar] [CrossRef]
- Zhang, Z.; Yuan, X.; Zhao, Z.; Li, X.; Liu, B.; Bai, P. Electrochemical Noise Comparative Study of Pitting Corrosion of 316L Stainless Steel Fabricated by Selective Laser Melting and Wrought. J. Electroanal. Chem. 2021, 894, 115351. [Google Scholar] [CrossRef]
- Hoseinieh, S.M.; Homborg, A.M.; Shahrabi, T.; Mol, J.M.C.; Ramezanzadeh, B. A Novel Approach for the Evaluation of Under Deposit Corrosion in Marine Environments Using Combined Analysis by Electrochemical Impedance Spectroscopy and Electrochemical Noise. Electrochim. Acta 2016, 217, 226–241. [Google Scholar] [CrossRef]
- Xia, D.-H.; Ma, C.; Behnamian, Y.; Ao, S.; Song, S.; Xu, L. Reliability of the Estimation of Uniform Corrosion Rate of Q235B Steel under Simulated Marine Atmospheric Conditions by Electrochemical Noise (EN) Analyses. Measurement 2019, 148, 106946. [Google Scholar] [CrossRef]
- Calabrese, L.; Bonaccorsi, L.; Galeano, M.; Proverbio, E.; Di Pietro, D.; Cappuccini, F. Identification of Damage Evolution during SCC on 17-4 PH Stainless Steel by Combining Electrochemical Noise and Acoustic Emission Techniques. Corros. Sci. 2015, 98, 573–584. [Google Scholar] [CrossRef]
- Zhao, R.; Xia, D.-H.; Song, S.-Z.; Hu, W. Detection of SCC on 304 Stainless Steel in Neutral Thiosulfate Solutions Using Electrochemical Noise Based on Chaos Theory. Anti-Corros. Methods Mater. 2017, 64, 241–251. [Google Scholar] [CrossRef]
- Wei, Y.-J.; Xia, D.-H.; Song, S.-Z. Detection of SCC of 304 NG Stainless Steel in an Acidic NaCl Solution Using Electrochemical Noise Based on Chaos and Wavelet Analysis. Russ. J. Electrochem. 2016, 52, 560–575. [Google Scholar] [CrossRef]
- Calabrese, L.; Galeano, M.; Proverbio, E.; Di Pietro, D.; Donato, A.; Cappuccini, F. Advanced Signal Analysis of Acoustic Emission Data to Discrimination of Different Corrosion Forms. Int. J. Microstruct. Mater. Prop. 2017, 12, 147–164. [Google Scholar] [CrossRef]
- Carmona-Hernández, A.; Orozco-Cruz, R.; Carpio-Santamaria, F.A.; Campechano-Lira, C.; López-Huerta, F.; Mejía-Sánchez, E.; Contreras, A.; Galván-Martínez, R. Electrochemical Noise Analysis of the X70 Pipeline Steel under Stress Conditions Using Symmetrical and Asymmetrical Electrode Systems. Metals 2022, 12, 1545. [Google Scholar] [CrossRef]
- Benzaid, A.; Huet, F.; Jérôme, M.; Wenger, F.; Gabrielli, C.; Galland, J. Electrochemical Noise Analysis of Cathodically Polarised AISI 4140 Steel. I. Characterisation of Hydrogen Evolution on Vertical Unstressed Electrodes. Electrochim. Acta 2002, 47, 4315–4323. [Google Scholar] [CrossRef]
- Li, X.; Liu, C.; Zhang, T.; Lv, C.; Liu, J.; Ding, R.; Gao, Z.; Wang, R.; Liu, Y. Effect of PH and Applied Stress on Hydrogen Sulfide Stress Corrosion Behavior of HSLA Steel Based on Electrochemical Noise Analysis. J. Iron Steel Res. Int. 2023. [Google Scholar] [CrossRef]
- van der Merwe, J.W.; du Toit, M.; Klenam, D.E.P.; Bodunrin, M.O. Prediction of Stress-Corrosion Cracking Using Electrochemical Noise Measurements: A Case Study of Carbon Steels Exposed to H2O-CO-CO2 Environment. Eng. Fail. Anal. 2023, 144, 106948. [Google Scholar] [CrossRef]
- Jáquez-Muñoz, J.M.; Gaona-Tiburcio, C.; Cabral-Miramontes, J.; Nieves-Mendoza, D.; Maldonado-Bandala, E.; Olguín-Coca, J.; Estupinán-López, F.; López-León, L.D.; Chacón-Nava, J.; Almeraya-Calderón, F. Frequency Analysis of Transients in Electrochemical Noise of Superalloys Waspaloy and Ultimet. Metals 2021, 11, 702. [Google Scholar] [CrossRef]
- Oppenheim, A.V.; Schafer, R.W.; Buck, J.R. Discrete Time Signal Processing; Prentice-Hall: Edinburg, Scotland, 1999; Volume 1999, ISBN 0137549202. [Google Scholar]
- Rhif, M.; Ben Abbes, A.; Farah, I.R.; Martínez, B.; Sang, Y. Wavelet Transform Application for/in Non-Stationary Time-Series Analysis: A Review. Appl. Sci. 2019, 9, 1345. [Google Scholar] [CrossRef]
- Jacobsen, E.; Lyons, R. The Sliding DFT. IEEE Signal Process. Mag. 2003, 20, 74–80. [Google Scholar] [CrossRef]
- Allen, J.B. Short Term Spectral Analysis, Synthesis, and Modification by Discrete Fourier Transform. IEEE Trans. Acoust. Speech Signal Process. 1977, 25, 235–238. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, X.; Zhao, Z.; Bai, P.; Liu, B.; Tan, J.; Wu, X. In-Situ Monitoring of Pitting Corrosion of Q235 Carbon Steel by Electrochemical Noise: Wavelet and Recurrence Quantification Analysis. J. Electroanal. Chem. 2020, 879, 114776. [Google Scholar] [CrossRef]
- Huang, N.E.; Wu, Z. A Review on Hilbert-Huang Transform: Method and Its Applications. October 2008, 46, 1–23. [Google Scholar] [CrossRef]
- Huang, N.E.; Shen, Z.; Long, S.R.; Wu, M.C.; Shih, H.H.; Zheng, Q.; Yen, N.-C.; Tung, C.C.; Liu, H.H. The Empirical Mode Decomposition and the Hilbert Spectrum for Nonlinear and Non-Stationary Time Series Analysis. Proc. R. Soc. A Math. Phys. Eng. Sci. 1998, 454, 903–995. [Google Scholar] [CrossRef]
- Calabrese, L.; Galeano, M.; Proverbio, E. Identifying Corrosion Forms on Synthetic Electrochemical Noise Signals by the Hilbert–Huang Transform Method Hilbert–Huang Transform Method. Corros. Eng. Sci. Technol. 2018, 53, 492–501. [Google Scholar] [CrossRef]
- Homborg, A.M.; van Westing, E.P.M.; Tinga, T.; Zhang, X.; Oonincx, P.J.; Ferrari, G.M.; de Wit, J.H.W.; Mol, J.M.C. Novel Time-Frequency Characterization of Electrochemical Noise Data in Corrosion Studies Using Hilbert Spectra. Corros. Sci. 2013, 66, 97–110. [Google Scholar] [CrossRef]
- Jáquez-Muñoz, J.M.; Gaona-Tiburcio, C.; Cabral-Miramontes, J.; Nieves-Mendoza, D.; Maldonado-Bandala, E.; Olguín-Coca, J.; López-Léon, L.D.; Flores-De los Rios, J.P.; Almeraya-Calderón, F. Electrochemical Noise Analysis of the Corrosion of Titanium Alloys in NaCl and H2SO4 Solutions. Metals 2021, 11, 105. [Google Scholar] [CrossRef]
- Moradi, M.; Rezaei, M. Electrochemical Noise Analysis to Evaluate the Localized Anti-Corrosion Properties of PP/Graphene Oxide Nanocomposite Coatings. J. Electroanal. Chem. 2022, 921, 116665. [Google Scholar] [CrossRef]
- Cottis, R.A. 5—Electrochemical noise for corrosion monitoring. In Techniques for Corrosion Monitoring; Woodhead Publishing: Cambridge, UK, 2001. [Google Scholar] [CrossRef]
- Delgadillo, R.M.; Tenelema, F.J.; Casas, J.R. Marginal Hilbert Spectrum and Instantaneous Phase Difference as Total Damage Indicators in Bridges under Operational Traffic Loads. Struct. Infrastruct. Eng. 2023, 19, 824–844. [Google Scholar] [CrossRef]
- Huang, N.E. An Adaptive Data Analysis Method for Nonlinear and Nonstationary Time Series: The Empirical Mode Decomposition and Hilbert Spectral Analysis. In Applied and Numerical Harmonic Analysis; Birkhäuser Verlag: Basel, Switzerland, 2006. [Google Scholar] [CrossRef]
- Li, Y.; Lin, J.; Niu, G.; Wu, M.; Wei, X. A Hilbert–Huang Transform-Based Adaptive Fault Detection and Classification Method for Microgrids. Energies 2021, 14, 5040. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhao, Z.; Bai, P.; Li, X.; Liu, B.; Tan, J.; Wu, X. In-Situ Monitoring of Pitting Corrosion of AZ31 Magnesium Alloy by Combining Electrochemical Noise and Acoustic Emission Techniques. J. Alloys Compd. 2021, 878, 160334. [Google Scholar] [CrossRef]
- Lentka, Ł.; Smulko, J. Methods of Trend Removal in Electrochemical Noise Data–Overview. Measurement 2019, 131, 569–581. [Google Scholar] [CrossRef]
- Havashinejadian, E.; Danaee, I.; Eskandari, H.; Nikmanesh, S. Investigation on Trend Removal in Time Domain Analysis of Electrochemical Noise Data Using Polynomial Fitting and Moving Average Removal Methods. J. Electrochem. Sci. Technol. 2017, 8, 115–123. [Google Scholar] [CrossRef]
- Hu, J.; Deng, J.; Deng, P.; Wang, G. Corrosion Monitoring Method of 304 Stainless Steel in a Simulated Marine–Industrial Atmospheric Environment: Electrochemical Noise Method. Anti-Corros. Methods Mater. 2022, 69, 629–635. [Google Scholar] [CrossRef]
- Kearns, J.R.; Scully, J.R.; Roberge, P.R.; Reichert, D.L.; Dawson, J.L. Electrochemical Noise Measurement for Corrosion Applications-STP 1277; ASTM International: Philadelphia, PA, USA, 1996. [Google Scholar]
- Gabrielli, C.; Huet, F.; Keddam, M.; Oltra, R. Review of the Probabilistic Aspects of Localized Corrosion. Corrosion 1990, 46, 266–278. [Google Scholar] [CrossRef]
- Calabrese, L.; Galeano, M.; Proverbio, E.; Di Pietro, D.; Donato, A. Topological Neural Network of Combined AE and EN Signals for Assessment of SCC Damage. Nondestruct. Test. Eval. 2020, 35, 98–119. [Google Scholar] [CrossRef]
- Ramezanzadeh, B.; Arman, S.Y.; Mehdipour, M.; Markhali, B.P. Analysis of Electrochemical Noise (ECN) Data in Time and Frequency Domain for Comparison Corrosion Inhibition of Some Azole Compounds on Cu in 1.0 M H2SO4 Solution. Appl. Surf. Sci. 2014, 289, 129–140. [Google Scholar] [CrossRef]
- Fabre-Pulido, J.; Orozco-Cruz, R.; Mejía-Sánchez, E.; Contreras-Cuevas, A.; Galvan-Martinez, R. Stress Corrosion Cracking Process of X60 Steel Immersed in a Brine Solution with Corrosion Inhibitor through the Electrochemical Noise Technique. ECS Trans. 2019, 94, 189. [Google Scholar] [CrossRef]
- Nagiub, A.M. Comparative Electrochemical Noise Study of the Corrosion of Different Alloys Exposed to Chloride Media. Engineering 2014, 6, 1007–1016. [Google Scholar] [CrossRef]
- Cottis, R.A.; Al-Awadhi, M.A.A.; Al-Mazeedi, H.; Turgoose, S. Measures for the Detection of Localized Corrosion with Electrochemical Noise. Electrochim. Acta 2001, 46, 3665–3674. [Google Scholar] [CrossRef]
- Mansfeld, F.; Sun, Z. Technical Note: Localization Index Obtained from Electrochemical Noise Analysis. Corrosion 1999, 55, 915–918. [Google Scholar] [CrossRef]
- Sanchez-Amaya, J.M.; Cottis, R.A.; Botana, F.J. Shot Noise and Statistical Parameters for the Estimation of Corrosion Mechanisms. Corros. Sci. 2005, 47, 3280–3299. [Google Scholar] [CrossRef]
- Cottis, R.A. Interpretation of Electrochemical Noise Data. Corrosion 2001, 57, 265–284. [Google Scholar] [CrossRef]
- Permeh, S.; Lau, K. Identification of Steel Corrosion Associated with Sulfate-Reducing Bacteria by Electrochemical Noise Technique. Mater. Corros. 2023, 74, 20–32. [Google Scholar] [CrossRef]
- Calabrese, L.; Campanella, G.; Proverbio, E. Identification of Corrosion Mechanisms by Univariate and Multivariate Statistical Analysis during Long Term Acoustic Emission Monitoring on a Pre-Stressed Concrete Beam. Corros. Sci. 2013, 73, 161–171. [Google Scholar] [CrossRef]
- Shaikh, H.; Amirthalingam, R.; Anita, T.; Sivaibharasi, N.; Jaykumar, T.; Manohar, P.; Khatak, H.S. Evaluation of Stress Corrosion Cracking Phenomenon in an AISI Type 316LN Stainless Steel Using Acoustic Emission Technique. Corros. Sci. 2007, 49, 740–765. [Google Scholar] [CrossRef]
- Nguyen, T.-T.; Bolivar, J.; Shi, Y.; Réthoré, J.; King, A.; Fregonese, M.; Adrien, J.; Buffiere, J.-Y.; Baietto, M.-C. A Phase Field Method for Modeling Anodic Dissolution Induced Stress Corrosion Crack Propagation. Corros. Sci. 2018, 132, 146–160. [Google Scholar] [CrossRef]
- Tan, Y. Sensing localised corrosion by means of electrochemical noise detection and analysis. Sens. Actuators B Chem. 2009, 139, 688–698. [Google Scholar] [CrossRef]
- Ganjalizadeh, V.; Meena, G.G.; Wall, T.A.; Stott, M.A.; Hawkins, A.R.; Schmidt, H. Fast Custom Wavelet Analysis Technique for Single Molecule Detection and Identification. Nat. Commun. 2022, 13, 1035. [Google Scholar] [CrossRef]
- Kim, J.J. Wavelet Analysis of Potentiostatic Electrochemical Noise. Mater. Lett. 2007, 61, 4000–4002. [Google Scholar] [CrossRef]
- Smith, M.T.; Macdonald, D.D. Wavelet Analysis of Electrochemical Noise Data. Corrosion 2009, 65, 438–448. [Google Scholar] [CrossRef]
- Shoji, T.; Lu, Z.; Murakami, H. Formulating Stress Corrosion Cracking Growth Rates by Combination of Crack Tip Mechanics and Crack Tip Oxidation Kinetics. Corros. Sci. 2010, 52, 769–779. [Google Scholar] [CrossRef]
- Homborg, A.M.; Tinga, T.; Zhang, X.; van Westing, E.P.M.; Oonincx, P.J.; Ferrari, G.M.; de Wit, J.H.W.; Mol, J.M.C. Transient Analysis through Hilbert Spectra of Electrochemical Noise Signals for the Identification of Localized Corrosion of Stainless Steel. Electrochim. Acta 2013, 104, 84–93. [Google Scholar] [CrossRef]
- Guan, L.; Zhang, B.; Yong, X.P.; Wang, J.Q.; Han, E.-H.; Ke, W. Effects of Cyclic Stress on the Metastable Pitting Characteristic for 304 Stainless Steel under Potentiostatic Polarization. Corros. Sci. 2015, 93, 80–89. [Google Scholar] [CrossRef]
- Zhang, Z.; Wu, X. Interpreting Electrochemical Noise Signal Arising from Stress Corrosion Cracking of 304 Stainless Steel in Simulated PWR Primary Water Environment by Coupling Acoustic Emission. J. Mater. Res. Technol. 2022, 20, 3807–3817. [Google Scholar] [CrossRef]
- Calabrese, L.; Galeano, M.; Proverbio, E.; Di Pietro, D.; Cappuccini, F.; Donato, A. Monitoring of 13% Cr Martensitic Stainless Steel Corrosion in Chloride Solution in Presence of Thiosulphate by Acoustic Emission Technique. Corros. Sci. 2016, 111, 151–161. [Google Scholar] [CrossRef]
- Aoyagi, Y.; Kaji, Y. Crystal Plasticity Simulation Considering Oxidation along Grain Boundary and Effect of Grain Size on Stress Corrosion Cracking. Mater. Trans. 2012, 53, 161–166. [Google Scholar] [CrossRef]
- Kawasaki, Y.; Fukui, S.; Fukuyama, T. Phenomenological Process of Rebar Corrosion in Reinforced Concrete Evaluated by Acoustic Emission and Electrochemical Noise. Constr. Build. Mater. 2022, 352, 128829. [Google Scholar] [CrossRef]
- Kovac, J.; Alaux, C.; Marrow, T.J.; Govekar, E.; Legat, A. Correlations of Electrochemical Noise, Acoustic Emission and Complementary Monitoring Techniques during Intergranular Stress-Corrosion Cracking of Austenitic Stainless Steel. Corros. Sci. 2010, 52, 2015–2025. [Google Scholar] [CrossRef]












| Composition [wt %] | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| C | Ni | Cr | S | P | Cu | Mn | Si | Nb | Fe |
| 0.042 | 4.43 | 15.10 | 0.005 | 0.02 | 3.31 | 0.44 | 0.50 | 0.22 | balance |
| Mean Mechanical Properties | ||
|---|---|---|
| Yield stress fp 0.2k (MPa) | Ultimate tensile stress UTS fpk (MPa) | Elongation εuk (%) |
| 745 | 1010 | 24 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calabrese, L.; Galeano, M.; Proverbio, E. Data Mining Applied to the Electrochemical Noise Technique in the Time/Frequency Domain for Stress Corrosion Cracking Recognition. Corros. Mater. Degrad. 2023, 4, 659-679. https://doi.org/10.3390/cmd4040034
Calabrese L, Galeano M, Proverbio E. Data Mining Applied to the Electrochemical Noise Technique in the Time/Frequency Domain for Stress Corrosion Cracking Recognition. Corrosion and Materials Degradation. 2023; 4(4):659-679. https://doi.org/10.3390/cmd4040034
Chicago/Turabian StyleCalabrese, Luigi, Massimiliano Galeano, and Edoardo Proverbio. 2023. "Data Mining Applied to the Electrochemical Noise Technique in the Time/Frequency Domain for Stress Corrosion Cracking Recognition" Corrosion and Materials Degradation 4, no. 4: 659-679. https://doi.org/10.3390/cmd4040034
APA StyleCalabrese, L., Galeano, M., & Proverbio, E. (2023). Data Mining Applied to the Electrochemical Noise Technique in the Time/Frequency Domain for Stress Corrosion Cracking Recognition. Corrosion and Materials Degradation, 4(4), 659-679. https://doi.org/10.3390/cmd4040034








