Concussion Disrupts Sleep in Adolescents: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Literature Search Strategy
2.3. Main Outcomes
2.4. Data Extraction and Curation
2.5. Quality Assessment
2.6. Data Analysis
3. Results
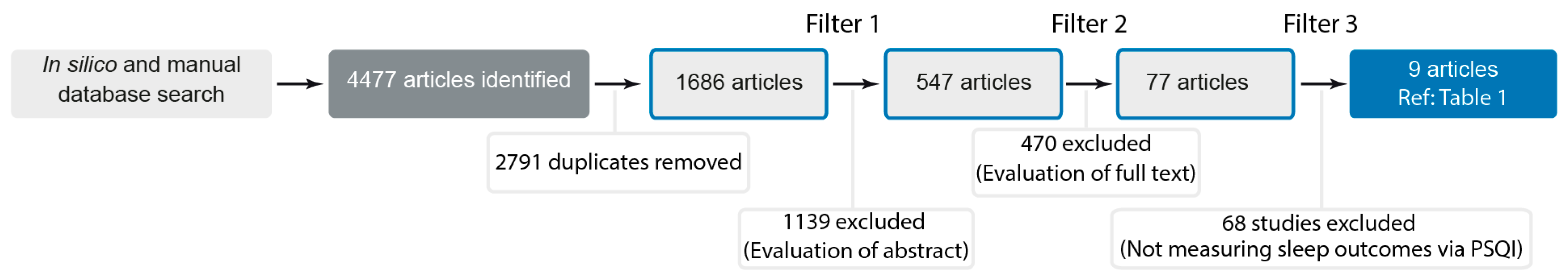
3.1. Study Inclusion and Filtering
3.2. Study Setting
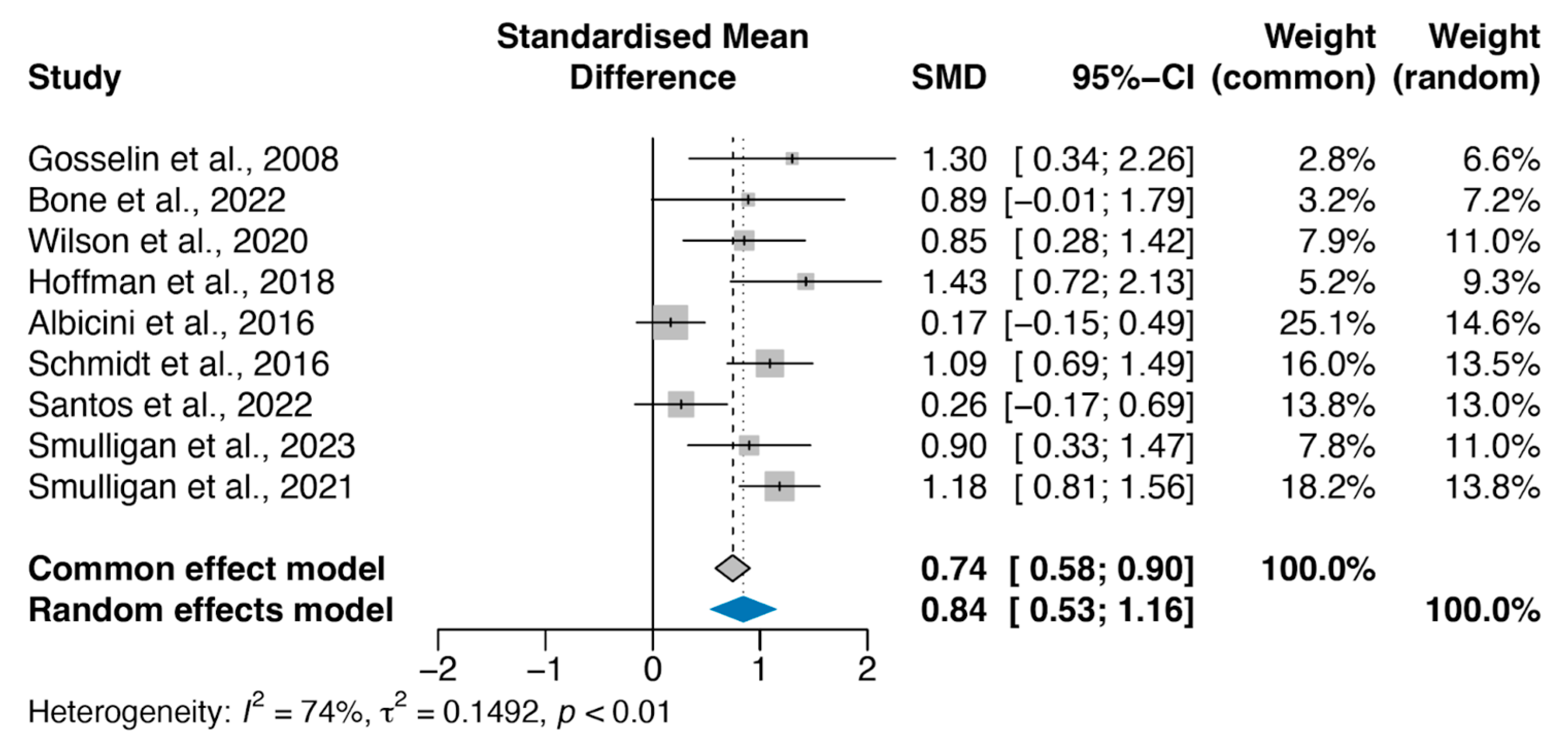
3.3. Meta-Analysis on the Effect of Concussion on Sleep Quality
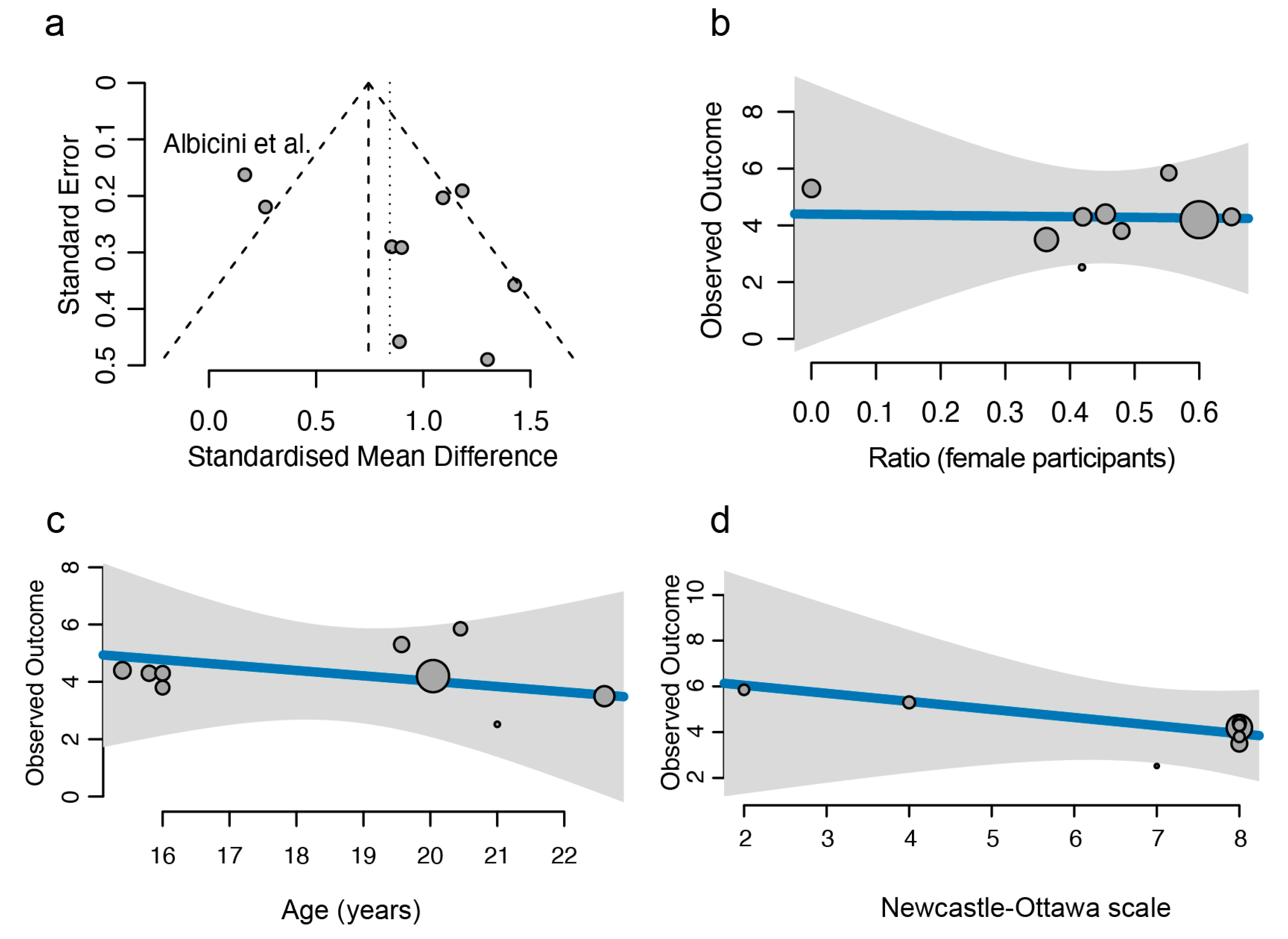
3.4. Risk of Bias, Heterogeneity, and Confounder Analyses
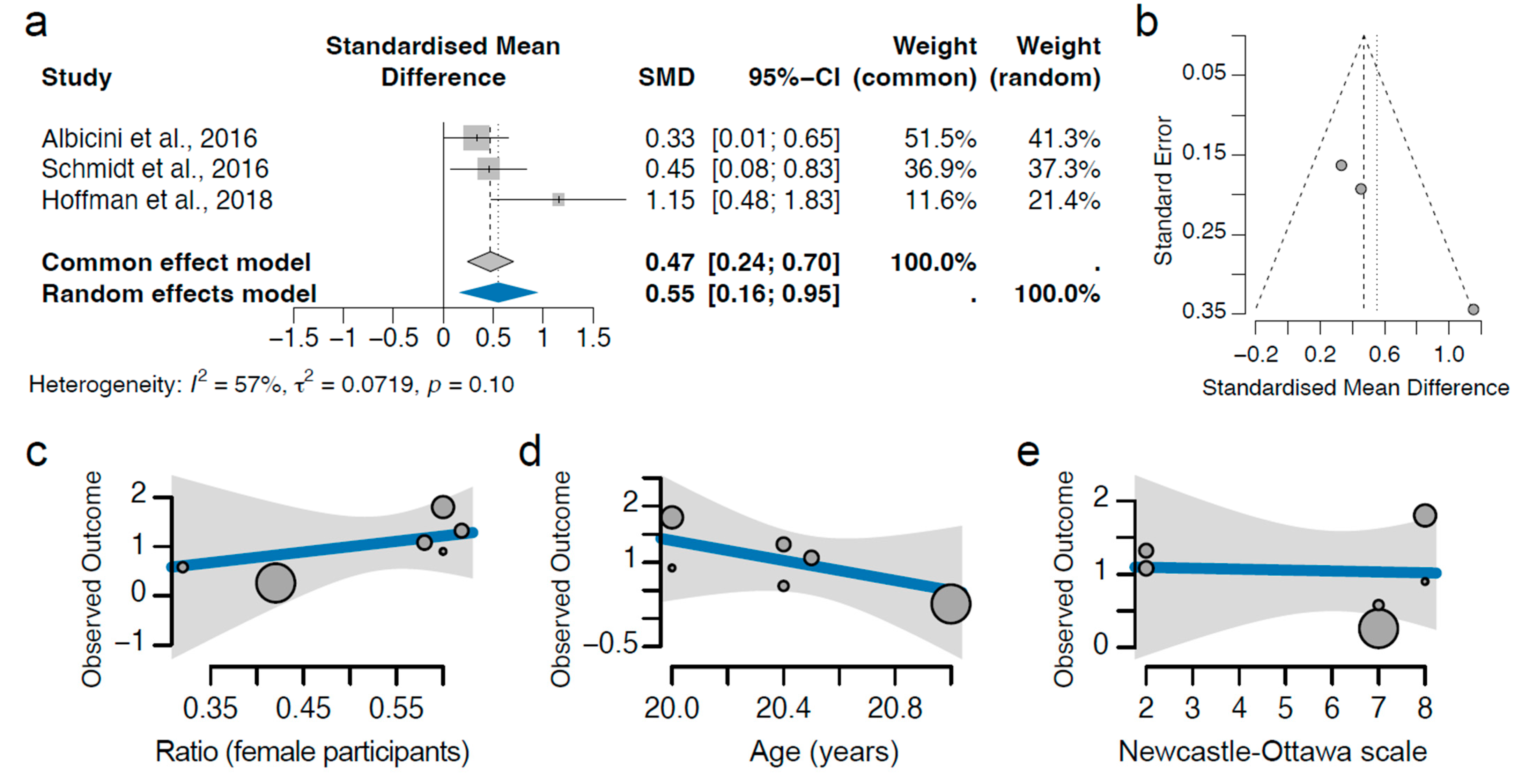
3.5. Concussion Impacts Daytime Dysfunction
4. Discussion
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PSQI | Pittsburgh Sleep Quality Index |
| PCSS | Post-Concussion Symptom Scale |
| ImPACT | Immediate Post-Concussion Assessment and Cognitive Testing |
| ESS | Epworth Sleepiness Scale |
| NOS | Newcastle–Ottawa Scale |
| RCT | Randomised Controlled Trial |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
References
- Yu, Y. Links between Sleep Apnoea and Insomnia in a British Cohort. Clocks Sleep 2023, 5, 552–565. [Google Scholar] [CrossRef]
- Johnson, D.A.; Jackson, C.L.; Williams, N.J.; Alcántara, C. Are Sleep Patterns Influenced by Race/Ethnicity—A Marker of Relative Advantage or Disadvantage? Evidence to Date. Nat. Sci. Sleep 2019, 11, 79–95. [Google Scholar] [CrossRef]
- Grandner, M.A. Epidemiology of Insufficient Sleep and Poor Sleep Quality. In Sleep and Health; Elsevier: Amsterdam, The Netherlands, 2019; pp. 11–20. ISBN 978-0-12-815374-1. [Google Scholar]
- Mosti, C.; Spiers, M.V.; Kloss, J.D. A Practical Guide to Evaluating Sleep Disturbance in Concussion Patients. Neurol. Clin. Pract. 2016, 6, 129–137. [Google Scholar] [CrossRef]
- Theadom, A.; Cropley, M.; Parmar, P.; Barker-Collo, S.; Starkey, N.; Jones, K.; Feigin, V.L. BIONIC Research Group Sleep Difficulties One Year Following Mild Traumatic Brain Injury in a Population-Based Study. Sleep Med. 2015, 16, 926–932. [Google Scholar] [CrossRef] [PubMed]
- Kazl, C.; Torres, A. Definition, Classification, and Epidemiology of Concussion. Semin. Pediatr. Neurol. 2019, 30, 9–13. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, K.L.; Baker, M.M.; Dalton, S.L.; Dompier, T.P.; Broglio, S.P.; Kerr, Z.Y. Epidemiology of Sport-Related Concussions in High School Athletes: National Athletic Treatment, Injury and Outcomes Network (NATION), 2011–2012 Through 2013–2014. J. Athl. Train. 2017, 52, 175–185. [Google Scholar] [CrossRef]
- Chen, Y.; Huang, W.; Constantini, S. The Differences between Blast-Induced and Sports-Related Brain Injuries. Front. Neurol. 2013, 4, 119. [Google Scholar] [CrossRef]
- Hoffman, N.L.; O’Connor, P.J.; Schmidt, M.D.; Lynall, R.C.; Schmidt, J.D. Differences in Sleep between Concussed and Nonconcussed College Students: A Matched Case–Control Study. Sleep 2019, 42, zsy222. [Google Scholar] [CrossRef]
- Wilk, J.E. Mild Traumatic Brain Injury (Concussion) During Combat: Lack of Association of Blast Mechanism with Persistent Postconcussive Symptoms—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/20051900/ (accessed on 19 February 2025).
- Lah, S.; Bogdanov, S.; Menzies, B. Sleep in Children Following Brain Concussion. In Pediatric Sleep Medicine: Mechanisms and Comprehensive Guide to Clinical Evaluation and Management; Gozal, D., Kheirandish-Gozal, L., Eds.; Springer International Publishing: Cham, Switherland, 2021; pp. 681–690. ISBN 978-3-030-65574-7. [Google Scholar]
- Dufour, S.C.; Adams, R.S.; Brody, D.L.; Puente, A.N.; Gray, J.C. Prevalence and Correlates of Concussion in Children: Data from the Adolescent Brain Cognitive Development Study. Cortex J. Devoted Study Nerv. Syst. Behav. 2020, 131, 237–250. [Google Scholar] [CrossRef]
- Jaffee, M.S.; Winter, W.C.; Jones, C.C.; Ling, G. Sleep Disturbances in Athletic Concussion. Brain Inj. 2015, 29, 221–227. [Google Scholar] [CrossRef]
- Saigal, R.; Berger, M.S. The Long-Term Effects of Repetitive Mild Head Injuries in Sports. Neurosurgery 2014, 75 (Suppl. 4), S149–S155. [Google Scholar] [CrossRef]
- George, K.K.; Heithoff, B.P.; Shandra, O.; Robel, S. Mild Traumatic Brain Injury/Concussion Initiates an Atypical Astrocyte Response Caused by Blood-Brain Barrier Dysfunction. J. Neurotrauma 2022, 39, 211–226. [Google Scholar] [CrossRef] [PubMed]
- Bellesi, M.; de Vivo, L.; Chini, M.; Gilli, F.; Tononi, G.; Cirelli, C. Sleep Loss Promotes Astrocytic Phagocytosis and Microglial Activation in Mouse Cerebral Cortex. J. Neurosci. Off. J. Soc. Neurosci. 2017, 37, 5263–5273. [Google Scholar] [CrossRef] [PubMed]
- Alrousan, G.; Hassan, A.; Pillai, A.A.; Atrooz, F.; Salim, S. Early Life Sleep Deprivation and Brain Development: Insights from Human and Animal Studies. Front. Neurosci. 2022, 16, 833786. [Google Scholar] [CrossRef]
- Daneshvar, D.H.; Riley, D.O.; Nowinski, C.J.; McKee, A.C.; Stern, R.A.; Cantu, R.C. Long-Term Consequences: Effects on Normal Development Profile after Concussion. Phys. Med. Rehabil. Clin. N. Am. 2011, 22, 683–700. [Google Scholar] [CrossRef]
- Telzer, E.H.; Goldenberg, D.; Fuligni, A.J.; Lieberman, M.D.; Gálvan, A. Sleep Variability in Adolescence Is Associated with Altered Brain Development. Dev. Cogn. Neurosci. 2015, 14, 16–22. [Google Scholar] [CrossRef]
- Shekleton, J.A.; Parcell, D.L.; Redman, J.R.; Phipps-Nelson, J.; Ponsford, J.L.; Rajaratnam, S.M.W. Sleep Disturbance and Melatonin Levels Following Traumatic Brain Injury. Neurology 2010, 74, 1732–1738. [Google Scholar] [CrossRef]
- Haydon, P.G. Astrocytes and the Modulation of Sleep. Curr. Opin. Neurobiol. 2017, 44, 28–33. [Google Scholar] [CrossRef]
- Bozdemir, E.; Vigil, F.A.; Chun, S.H.; Espinoza, L.; Bugay, V.; Khoury, S.M.; Holstein, D.M.; Stoja, A.; Lozano, D.; Tunca, C.; et al. Neuroprotective Roles of the Adenosine A3 Receptor Agonist AST-004 in Mouse Model of Traumatic Brain Injury. Neurother. J. Am. Soc. Exp. Neurother. 2021, 18, 2707–2721. [Google Scholar] [CrossRef]
- Leddy, J.J.; Burma, J.S.; Toomey, C.M.; Hayden, A.; Davis, G.A.; Babl, F.E.; Gagnon, I.; Giza, C.C.; Kurowski, B.G.; Silverberg, N.D.; et al. Rest and Exercise Early after Sport-Related Concussion: A Systematic Review and Meta-Analysis. Br. J. Sports Med. 2023, 57, 762–770. [Google Scholar] [CrossRef]
- Tang, S.; Sours Rhodes, C.; Jiang, L.; Chen, H.; Roys, S.; Badjatia, N.; Raghavan, P.; Zhuo, J.; Gullapalli, R.P. Association between Sleep Disturbances at Subacute Stage of Mild Traumatic Brain Injury and Long-Term Outcomes. Neurotrauma Rep. 2022, 3, 276–285. [Google Scholar] [CrossRef]
- Ludwig, R.; D’Silva, L.; Vaduvathiriyan, P.; Rippee, M.A.; Siengsukon, C. Sleep Disturbances in the Acute Stage of Concussion Are Associated With Poorer Long-Term Recovery: A Systematic Review. PM&R 2020, 12, 500–511. [Google Scholar] [CrossRef]
- Kontos, A.P. A Revised Factor Structure for the Post-Concussion Symptom Scale: Baseline and Postconcussion Factors—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/22904209/ (accessed on 19 February 2025).
- Schatz, P.; Pardini, J.E.; Lovell, M.R.; Collins, M.W.; Podell, K. Sensitivity and Specificity of the ImPACT Test Battery for Concussion in Athletes. Arch. Clin. Neuropsychol. Off. J. Natl. Acad. Neuropsychol. 2006, 21, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Stevens, D.J.; Alghwiri, A.; Appleton, S.L.; Rogers, J.M.; Plummer, S.L.; Grant, C.; Bickley, K.; Alvaro, P.K.; Kennett, S.; Adams, R.; et al. Should We Lose Sleep Over Sleep Disturbances After Sports-Related Concussion? A Scoping Review of the Literature. J. Head Trauma Rehabil. 2022, 37, E206–E219. [Google Scholar] [CrossRef] [PubMed]
- Noordeen, S.; Ergisi, M.; Popovic, R.; Mahendran, A.; Memon, M.; Hegde, S.; Wang, P.; Qiu, C.; Yu, Y. 49 A Pilot Meta-Analysis on Concussion-Related Sleep Disturbances. BMJ Open Respir. Res. 2021, 8 (Suppl 1), A1–A31, A23. [Google Scholar] [CrossRef]
- Johns, M.W. A New Method for Measuring Daytime Sleepiness: The Epworth Sleepiness Scale. Sleep J. Sleep Res. Sleep Med. 1991, 14, 540–545. [Google Scholar] [CrossRef] [PubMed]
- Buysse, D.J.; Reynolds, C.F.; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A New Instrument for Psychiatric Practice and Research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Carpi, M. The Pittsburgh Sleep Quality Index: A Brief Review. Occup. Med. Oxf. Engl. 2025, 75, 14–15. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. Syst. Rev. 2021, 10, 89. [Google Scholar] [CrossRef]
- Booth, A.; Clarke, M.; Dooley, G.; Ghersi, D.; Moher, D.; Petticrew, M.; Stewart, L. The Nuts and Bolts of PROSPERO: An International Prospective Register of Systematic Reviews. Syst. Rev. 2012, 1, 2. [Google Scholar] [CrossRef]
- Riley, R.D.; Moons, K.G.M.; Snell, K.I.E.; Ensor, J.; Hooft, L.; Altman, D.G.; Hayden, J.; Collins, G.S.; Debray, T.P.A. A Guide to Systematic Review and Meta-Analysis of Prognostic Factor Studies. BMJ 2019, 364, k4597. [Google Scholar] [CrossRef]
- Wells, G. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. In Proceedings of the 3rd Symposium on Systemic Review Basics, Oxford, UK, 3–5 July 2000. [Google Scholar]
- Luchini, C.; Veronese, N.; Nottegar, A.; Shin, J.I.; Gentile, G.; Granziol, U.; Soysal, P.; Alexinschi, O.; Smith, L.; Solmi, M. Assessing the Quality of Studies in Meta-Research: Review/Guidelines on the Most Important Quality Assessment Tools. Pharm. Stat. 2021, 20, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Viechtbauer, W. Conducting Meta-Analyses in R with the Metafor Package. J. Stat. Softw. 2010, 36, 1–48. [Google Scholar] [CrossRef]
- Balduzzi, S.; Rücker, G.; Schwarzer, G. How to Perform a Meta-Analysis with R: A Practical Tutorial. Evid. Based Ment. Health 2019, 22, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Harrer, M.; Cuijpers, P.; Furukawa, T.; Ebert, D. Doing Meta-Analysis with R: A Hands-On Guide; Chapman and Hall/CRC: New York, NY, USA, 2021; ISBN 978-1-003-10734-7. [Google Scholar]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring Inconsistency in Meta-Analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Smith, G.D.; Schneider, M.; Minder, C. Bias in Meta-Analysis Detected by a Simple, Graphical Test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Bone, T.; Konz, S.M.; Garrett, W.; Gilliland, C.A. The Effects of Concussion on Quantity and Quality of Sleep in Football Athletes. Neurology 2022, 98, S22. [Google Scholar] [CrossRef]
- Wilson, J.C.; Kirkwood, M.W.; Provance, A.J.; Walker, G.A.; Wilson, P.E.; Howell, D.R. ADOLESCENTS REPORT SLEEP QUALITY IMPAIRMENTS ACUTELY POST-CONCUSSION. Orthop. J. Sports Med. 2020, 8, 2325967120S00223. [Google Scholar] [CrossRef]
- Gosselin, N.; Lassonde, M.; Petit, D.; Leclerc, S.; Mongrain, V.; Collie, A.; Montplaisir, J. Sleep Following Sport-Related Concussions. Sleep Med. 2009, 10, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Smulligan, K.L.; Wilson, J.C.; Seehusen, C.N.; Wingerson, M.J.; Magliato, S.N.; Howell, D.R. Post-Concussion Dizziness, Sleep Quality, and Postural Instability: A Cross-Sectional Investigation. J. Athl. Train. 2021, 57, 1072–1078. [Google Scholar] [CrossRef] [PubMed]
- Smulligan, K.L.; Wingerson, M.J.; Little, C.C.; Wilson, J.C.; Howell, D.R. Early Physical Activity after Concussion Is Associated with Sleep Quality but Not Dizziness among Adolescent Athletes. J. Sci. Med. Sport 2023, 26, 183–188. [Google Scholar] [CrossRef]
- Lima Santos, J.P.; Kontos, A.P.; Holland, C.L.; Stiffler, R.S.; Bitzer, H.B.; Caviston, K.; Shaffer, M.; Suss, S.J., Jr.; Martinez, L.; Manelis, A.; et al. The Role of Sleep Quality on White Matter Integrity and Concussion Symptom Severity in Adolescents. NeuroImage Clin. 2022, 35, 103130. [Google Scholar] [CrossRef]
- Albicini, M.S.; Lee, J.; McKinlay, A. Ongoing Daytime Behavioural Problems in University Students Following Childhood Mild Traumatic Brain Injury. Int. J. Rehabil. Res. 2016, 39, 77. [Google Scholar] [CrossRef]
- Schmidt, A.T.; Li, X.; Hanten, G.R.; McCauley, S.R.; Faber, J.; Levin, H.S. A Longitudinal Investigation of Sleep Quality in Adolescents and Young Adults After Mild Traumatic Brain Injury. Cogn. Behav. Neurol. 2015, 28, 53–62. [Google Scholar] [CrossRef]
- Martens, G.; Khosravi, M.H.; Lejeune, N.; Kaux, J.-F.; Thibaut, A. Gender Specificities in Sleep Disturbances Following Mild Traumatic Brain Injury: A Preliminary Study. Brain Sci. 2023, 13, 323. [Google Scholar] [CrossRef]
- Ji, W.; Shi, L.; Lin, X.; Shen, Z.; Chen, Q.; Song, D.; Huang, P.; Zhao, Z.; Fan, J.; Hu, Y.; et al. The Relationship between Sleep Quality and Daytime Dysfunction among College Students in China during COVID-19: A Cross-Sectional Study. Front. Public Health 2023, 11, 1253834. [Google Scholar] [CrossRef]
- Altena, E.; Ellis, J.G. How Sleep Affects Daytime Functioning: The Latest Insights from Different Patient and Age Groups. Brain Sci. 2021, 11, 1163. [Google Scholar] [CrossRef]
- Blake, M.J.; Trinder, J.A.; Allen, N.B. Mechanisms Underlying the Association between Insomnia, Anxiety, and Depression in Adolescence: Implications for Behavioral Sleep Interventions. Clin. Psychol. Rev. 2018, 63, 25–40. [Google Scholar] [CrossRef]
- Charest, J.; Grandner, M.A. Sleep and Athletic Performance: Impacts on Physical Performance, Mental Performance, Injury Risk and Recovery, and Mental Health: An Update. Sleep Med. Clin. 2022, 17, 263–282. [Google Scholar] [CrossRef]
- Werner, J.K.; Shahim, P.; Pucci, J.U.; Lai, C.; Raiciulescu, S.; Gill, J.M.; Nakase-Richardson, R.; Diaz-Arrastia, R.; Kenney, K. Poor Sleep Correlates with Biomarkers of Neurodegeneration in Mild Traumatic Brain Injury Patients: A CENC Study. Sleep 2021, 44, zsaa272. [Google Scholar] [CrossRef]
- Yu, Y.; Fedele, G.; Celardo, I.; Loh, S.H.Y.; Martins, L.M. Parp Mutations Protect from Mitochondrial Toxicity in Alzheimer’s Disease. Cell Death Dis. 2021, 12, 651. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Chen, C.Z.; Celardo, I.; Tan, B.W.Z.; Hurcomb, J.D.; Leal, N.S.; Popovic, R.; Loh, S.H.Y.; Martins, L.M. Enhancing Mitochondrial One-Carbon Metabolism Is Neuroprotective in Alzheimer’s Disease Models. Cell Death Dis. 2024, 15, 856. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Fedele, G.; Celardo, I.; Zhou, L.; Tan, B.W.Z.; Loh, S.H.Y.; Martins, L.M. Distinct Forms of Amyloid-β Moderate Sleep Duration through NAD+-Linked Redox Metabolism in Alzheimer’s Disease. bioRxiv 2025. [Google Scholar] [CrossRef]
- Legarreta, A.D.; Monk, S.H.; Kirby, P.W.; Brett, B.L.; Yengo-Kahn, A.M.; Bhatia, A.; Solomon, G.S.; Zuckerman, S.L. Long-Term Neuroimaging Findings in American Football Players: Systematic Review. World Neurosurg. 2018, 120, e365–e379. [Google Scholar] [CrossRef]
- Covassin, T.; Moran, R.; Wilhelm, K. Concussion Symptoms and Neurocognitive Performance of High School and College Athletes Who Incur Multiple Concussions. Am. J. Sports Med. 2013, 41, 2885–2889. [Google Scholar] [CrossRef]
- Oyegbile, T.O.; Delasobera, B.E.; Zecavati, N. Gender Differences in Sleep Symptoms after Repeat Concussions. Sleep Med. 2017, 40, 110–115. [Google Scholar] [CrossRef]
- Olds, T.; Blunden, S.; Petkov, J.; Forchino, F. The Relationships between Sex, Age, Geography and Time in Bed in Adolescents: A Meta-Analysis of Data from 23 Countries. Sleep Med. Rev. 2010, 14, 371–378. [Google Scholar] [CrossRef]
- Lax, I.D.; Paniccia, M.; Agnihotri, S.; Reed, N.; Garmaise, E.; Azadbakhsh, M.; Ng, J.; Monette, G.; Wiseman-Hakes, C.; Taha, T.; et al. Developmental and Gender Influences on Executive Function Following Concussion in Youth Hockey Players. Brain Inj. 2015, 29, 1409–1419. [Google Scholar] [CrossRef]
- Murdaugh, D.L.; Ono, K.E.; Morris, S.O.; Burns, T.G. Effects of Developmental Age on Symptom Reporting and Neurocognitive Performance in Youth After Sports-Related Concussion Compared to Control Athletes. J. Child Neurol. 2018, 33, 474–481. [Google Scholar] [CrossRef]
- Vitale, K.C.; Owens, R.; Hopkins, S.R.; Malhotra, A. Sleep Hygiene for Optimizing Recovery in Athletes: Review and Recommendations. Int. J. Sports Med. 2019, 40, 535–543. [Google Scholar] [CrossRef]
| Reference | Authors | Title | Year | Total | Non-Concussed | Concussed |
|---|---|---|---|---|---|---|
| [9] | Hoffman, N. L., O’Connor, P. J., Schmidt, M. D., Lynall, R. C., & Schmidt, J. D | “Differences in sleep between concussed and non concussed college students: a matched case–control study.” | 2019 | 40 | 20 | 20 |
| [43] | Bone, T., Konz, S. M., Garrett, W., & Gilliland, C. A. | “The Effects of Concussion on Quantity and Quality of Sleep in Football Athletes.” * | 2022 | 27 | 20 | 7 |
| [44] | Wilson, J. C., Kirkwood, M. W., Provance, A. J., Walker, G. A., Wilson, P. E., & Howell, D. R. | “Adolescents report sleep quality impairments acutely postconcussion.” * | 2020 | 68 | 51 | 17 |
| [45] | Gosselin, N., Lassonde, M., Petit, D., Leclerc, S., Mongrain, V., Collie, A., & Montplaisir, J. | “Sleep following sport-related concussions.” | 2008 | 21 | 11 | 10 |
| [46] | Smulligan KL, Wilson JC, Seehusen CN, Wingerson MJ, Magliato SN, Howell DR | “Post-Concussion Dizziness, Sleep Quality, and Postural Instability: A Cross-Sectional Investigation” | 2021 | 131 | 73 | 58 |
| [47] | Smulligan KL, Wingerson MJ, Little CC, Wilson JC, Howell DR. | “Early physical activity after concussion is associated with sleep quality but not dizziness among adolescent athletes” | 2023 | 55 | 21 | 34 |
| [48] | Lima Santos JP, Kontos AP, Holland CL, Stiffler RS, Bitzer HB, Caviston K, Shaffer M, Suss SJ Jr, Martinez L, Manelis A, Iyengar S, Brent D, Ladouceur CD, Collins MW, Phillips ML, Versace A | “The role of sleep quality on white matter integrity and concussion symptom severity in adolescents” | 2022 | 90 | 33 | 57 |
| [49] | Albicini, M. S., Lee, J., & McKinlay, A | “Ongoing daytime behavioural problems in university students following childhood mild traumatic brain injury” | 2016 | 244 | 197 | 47 |
| [50] | Schmidt A. T., Li X., Hanten G. R., McCauley S. R., Faber J. & Levin H. S. | “A Longitudinal Investigation of Sleep Quality in Adolescents and Young Adults After Mild Traumatic Brain Injury” | 2016 | 43 | 77 | |
| Total: | 796 | 456 | 340 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noordeen, S.; Wang, P.; Strazda, A.E.; Arany, E.S.; Ergisi, M.; Yeo, L.J.; Popovic, R.; Mahendran, A.; Khawaja, M.; Grover, K.; et al. Concussion Disrupts Sleep in Adolescents: A Systematic Review and Meta-Analysis. Clocks & Sleep 2025, 7, 46. https://doi.org/10.3390/clockssleep7030046
Noordeen S, Wang P, Strazda AE, Arany ES, Ergisi M, Yeo LJ, Popovic R, Mahendran A, Khawaja M, Grover K, et al. Concussion Disrupts Sleep in Adolescents: A Systematic Review and Meta-Analysis. Clocks & Sleep. 2025; 7(3):46. https://doi.org/10.3390/clockssleep7030046
Chicago/Turabian StyleNoordeen, Suleyman, Poh Wang, Anna E. Strazda, Eszter Sara Arany, Mehmet Ergisi, Linghui Janice Yeo, Rebeka Popovic, Abinayan Mahendran, Mikail Khawaja, Kritika Grover, and et al. 2025. "Concussion Disrupts Sleep in Adolescents: A Systematic Review and Meta-Analysis" Clocks & Sleep 7, no. 3: 46. https://doi.org/10.3390/clockssleep7030046
APA StyleNoordeen, S., Wang, P., Strazda, A. E., Arany, E. S., Ergisi, M., Yeo, L. J., Popovic, R., Mahendran, A., Khawaja, M., Grover, K., Memon, M., Hegde, S., Qiu, C. S., Oulton, C., & Yu, Y. (2025). Concussion Disrupts Sleep in Adolescents: A Systematic Review and Meta-Analysis. Clocks & Sleep, 7(3), 46. https://doi.org/10.3390/clockssleep7030046






