The Role of Daylight for Humans: Gaps in Current Knowledge
Abstract
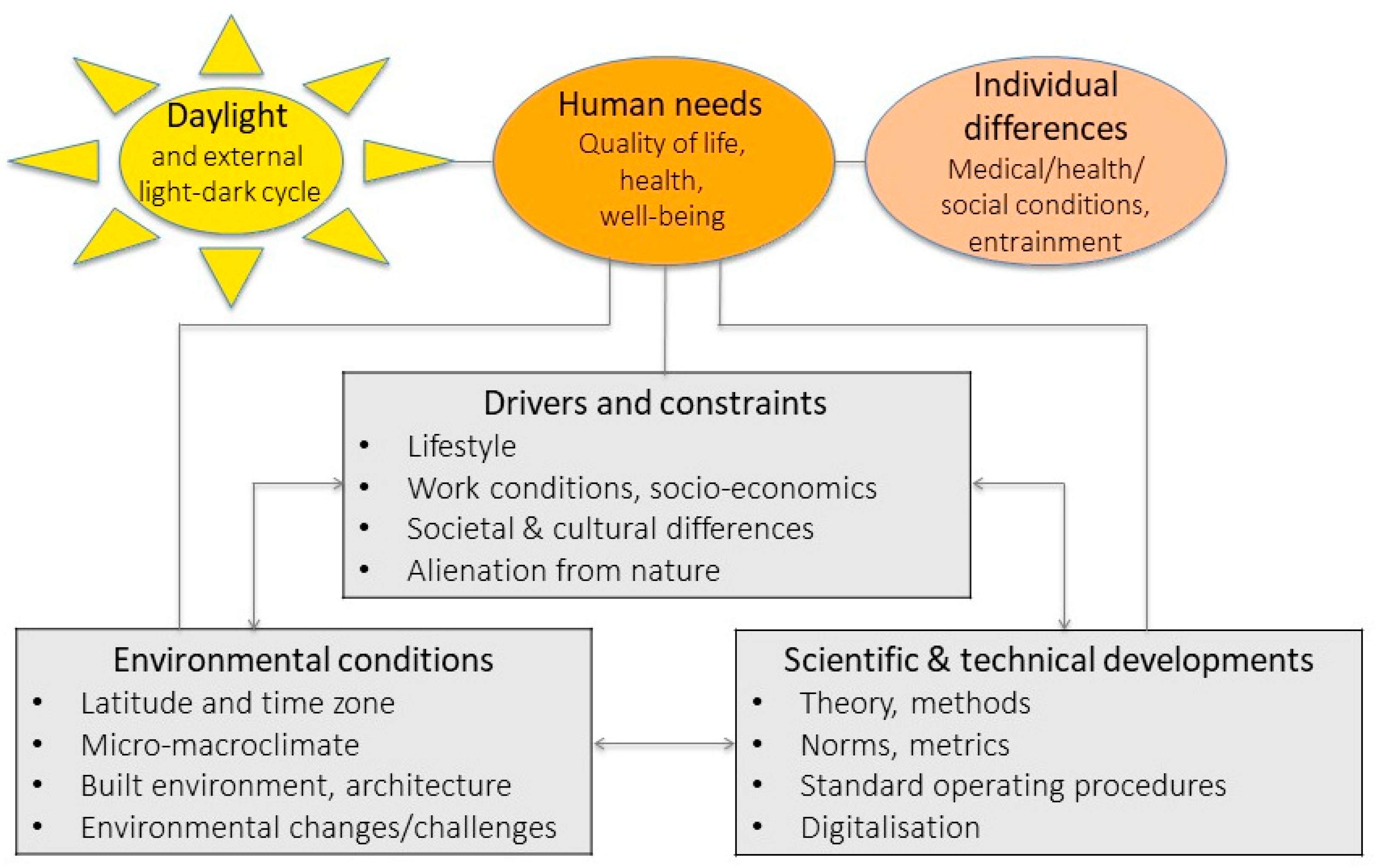
1. Introduction: The Need to Identify Current Gaps of Knowledge in Daylight Research
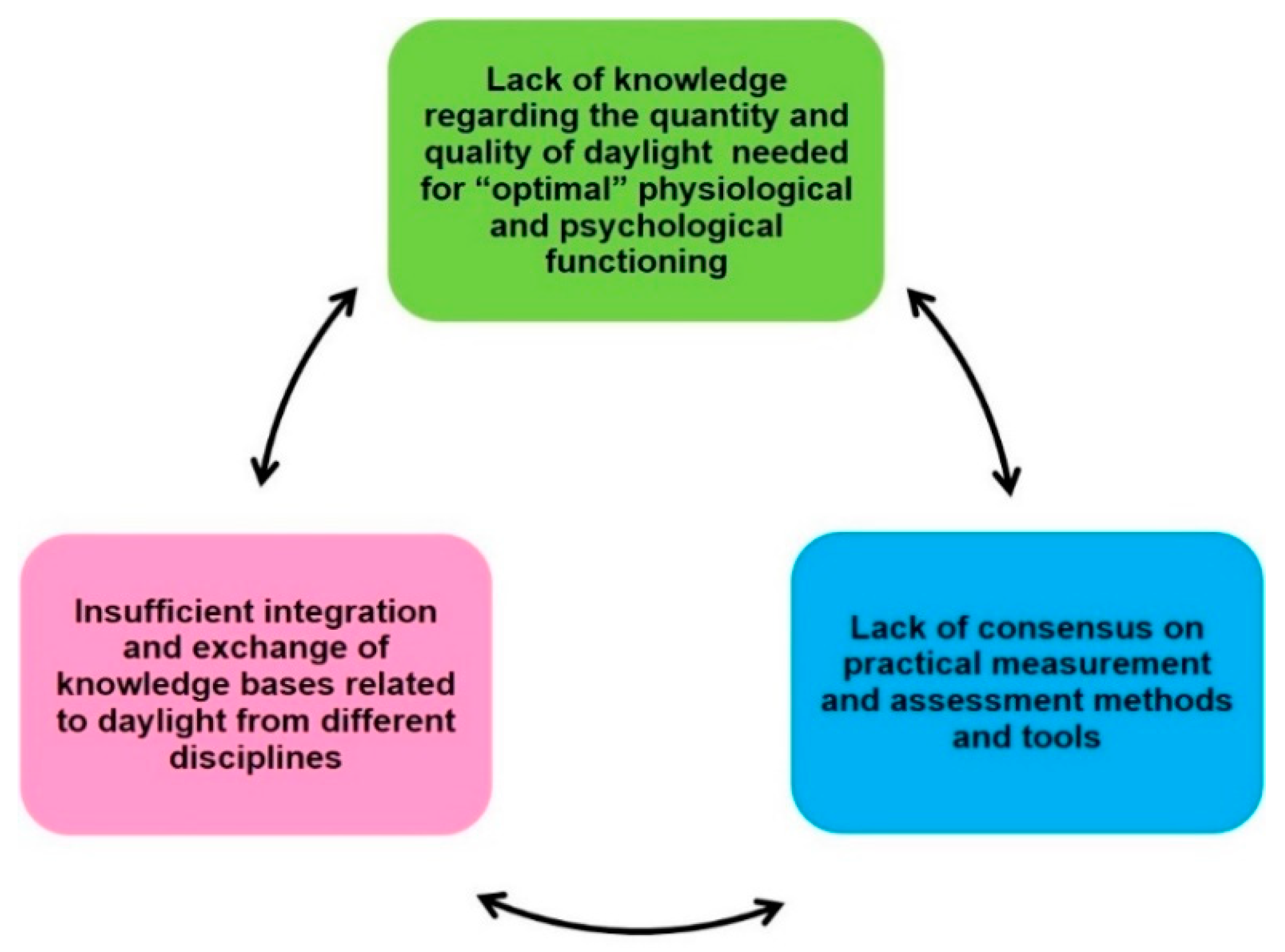
2. Gaps Group I: Lack of Knowledge Regarding Quantity and Quality of Daylight Needed for “Optimal” Physiological and Psychological Functioning
2.1. Open Questions for the Effects of Daylight on Vision and Visual Comfort
- There is considerable literature on visual comfort and glare evaluations with daylight inside buildings, e.g., [39], but very little is known about mixed/synergistic effects of high visual comfort together with non-visual functions such as alertness, mood [40,41,42] and indoor temperature perception [43,44]. These mixed/synergistic effects are missing not only for daylight but also for mixed electric/daylight conditions.
2.2. How Does Daylight Impact on Physiology and Behaviour Beyond Vision?
2.3. Gaps of Knowledge on Circadian Entrainment
2.4. What Is the “Optimal” Dose of Daylight with Respect to Intensity, Spectrum and Timing?
2.5. How Does Daylight Influence Light Sensitivity Functions?
2.6. Is Daylight Exposure Effective as a ‘Treatment’?
2.7. Are There Risks of Daylight Exposures (e.g., Systemic Diseases, Dermatology, Ophthalmology)?
2.8. Gaps of Knowledge Related to Individual Differences
2.9. Gaps of Knowledge in Daylight Research for Work Conditions
2.10. Gaps in Knowledge of Daylight Research Related to Environmental Factors
3. Gaps Group II: Lack of Consensus on Practical Measurement and Assessment Methods and Tools for Monitoring Real (Day-) Light Exposure Across Multiple Time Scales
4. Gaps Group III: Insufficient Integration and Exchange of Daylight Knowledge Bases from Different Disciplines
5. What Is Needed to Fill These Gaps and Achieve the Goals in Daylight Research?
6. Summary
7. Highlights
- Recognising the importance of daylight for human health and well-being.
- Daylight research needs to define requirements for optimal physiological and psychological functioning.
- New techniques are required to monitor and assess (day-) light exposure in the field.
- Interdisciplinary exchange of daylight knowledge is the key to integrating findings into practice, whether architectural or medical.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zehring, W.; Wheeler, D.; Reddy, P.; Konopka, R.; Kyriacou, C.P.; Rosbash, M.; Hall, J. P-element transformation with period locus DNA restores rhythmicity to mutant, arrhythmic Drosophila melanogaster. Cell 1984, 39, 369–376. [Google Scholar] [CrossRef]
- Bargiello, T.; Jackson, F.; Young, M. Restoration of circadian behavioural rhythms by gene transfer in Drosophila. Nature 1984, 312, 752–754. [Google Scholar] [CrossRef] [PubMed]
- Vitaterna, M.; King, D.; Chang, A.-M.; Kornhauser, J.; Lowrey, P.; McDonald, J.; Dove, W.; Pinto, L.; Turek, F.; Takahashi, J. Mutagenesis and mapping of a mouse gene, clock, essential for circadian behavior. Science 1994, 264, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Shichida, Y.; Matsuyama, T. Evolution of opsins and phototransduction. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 2881–2895. [Google Scholar] [CrossRef]
- Nilsson, D. Eye evolution and its functional basis. Vis. Neurosci. 2013, 30, 5–20. [Google Scholar] [CrossRef]
- Bellingham, J.; Foster, R.G. Opsins and mammalian photoentrainment. Cell Tissue Res. 2002, 309, 57–71. [Google Scholar] [CrossRef]
- Koyanagi, M.; Kubokawa, K.; Tsukamoto, H.; Shichida, Y.; Terakita, A. Cephalochordate melanopsin: Evolutionary linkage between invertebrate visual cells and vertebrate photosensitive retinal ganglion cells. Curr. Biol. 2005, 15, 1065–1069. [Google Scholar] [CrossRef]
- Peirson, S.; Halford, S.; Foster, R. The evolution of irradiance detection: Melanopsin and the non-visual opsins. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 2849–2865. [Google Scholar] [CrossRef]
- Provencio, I.; Jiang, G.; De Grip, W.J.; Hayes, W.P.; Rollag, M.D. Melanopsin: An opsin in melanophores, brain, and eye. Proc. Natl. Acad. Sci. USA 1998, 95, 340–345. [Google Scholar] [CrossRef]
- Hattar, S.; Liao, H.; Takao, M.; Berson, D.; Yau, K. Melanopsin-containing retinal ganglion cells: Architecture, projections, and intrinsic photosensitivity. Science 2002, 295, 1065–1070. [Google Scholar] [CrossRef]
- Berson, D.M.; Dunn, F.A.; Takao, M. Phototransduction by retinal ganglion cells that set the circadian clock. Science 2002, 295, 1070–1073. [Google Scholar] [CrossRef] [PubMed]
- Ruby, N.; Brennan, T.; Xie, X.; Cao, V.; Franken, P.; Heller, H.; O’Hara, B. Role of melanopsin in circadian responses to light. Science 2002, 298, 2211–2213. [Google Scholar] [CrossRef] [PubMed]
- Provencio, I.; Rodriguez, I.R.; Jiang, G.; Hayes, W.P.; Moreira, E.F.; Rollag, M.D. A novel human opsin in the inner retina. J. Neurosci. 2000, 20, 600–605. [Google Scholar] [CrossRef] [PubMed]
- Davies, T.; Smyth, T. Why artificial light at night should be a focus for global change research in the 21st century. Glob. Chang. Biol. 2018, 24, 872–882. [Google Scholar] [CrossRef]
- Aschoff, J.; Wever, J. The Circadian System of Man. In Biological Rhythms; Aschoff, J., Ed.; Springer: Boston, MA, USA, 1981; pp. 311–331. [Google Scholar]
- Pittendrigh, C.S. Circadian Systems: Entrainment. In Biological Rhythms; Aschoff, J., Ed.; Springer: Boston, MA, USA, 1981; pp. 95–124. [Google Scholar]
- Roenneberg, T.; Daan, S.; Merrow, M. The art of entrainment. J. Biol. Rhythm. 2003, 18, 183–194. [Google Scholar] [CrossRef]
- Vetter, C. Circadian disruption: What do we actually mean? Eur. J. Neurosci. 2018, 51, 531–550. [Google Scholar] [CrossRef]
- Abbott, S.; Malkani, R.; Zee, P. Circadian disruption and human health: A bidirectional relationship. Eur. J. Neurosci. 2018, 51, 567–583. [Google Scholar] [CrossRef]
- Zimmet, P.; Alberti, K.; Stern, N.; Bilu, C.; El-Osta, A.; Einat, H.; Kronfeld-Schor, N. The circadian syndrome: Is the metabolic syndrome and much more! J. Intern. Med. 2019, 286, 181–191. [Google Scholar] [CrossRef]
- Logan, R.; McClung, C. Rhythms of life: Circadian disruption and brain disorders across the lifespan. Nat. Rev. Neurosci. 2019, 20, 49–65. [Google Scholar] [CrossRef]
- Masri, S.; Sassone-Corsi, P. The emerging link between cancer, metabolism, and circadian rhythms. Nat. Med. 2018, 24, 1795–1803. [Google Scholar] [CrossRef]
- Kervezee, L.; Kosmadopoulos, A.; Boivin, D. Metabolic and cardiovascular consequences of shift work: The role of circadian disruption and sleep disturbances. Eur. J. Neurosci 2018, 51, 396–412. [Google Scholar] [CrossRef] [PubMed]
- McHill, A.; Wright, K.J. Role of sleep and circadian disruption on energy expenditure and in metabolic predisposition to human obesity and metabolic disease. Obes Rev. 2017, 18, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Eckel, R.H.; Depner, C.; Perreault, L.; Markwald, R.; Smith, M.; McHill, A.; Higgins, J.; Melanson, E.; Wright, K.J. Morning circadian misalignment during short sleep duration impacts insulin sensitivity. Curr. Biol. 2015, 25, 3004–3010. [Google Scholar] [CrossRef] [PubMed]
- Depner, C.; Melanson, E.; Eckel, R.; Snell-Bergeon, J.; Perreault, L.; Bergman, B.; Higgins, J.; Guerin, M.; Stothard, E.; Morton, S.; et al. Ad libitum weekend recovery sleep fails to prevent metabolic dysregulation during a repeating pattern of insufficient sleep and weekend recovery sleep. Curr. Biol. 2019, 29, 957–967. [Google Scholar] [CrossRef] [PubMed]
- Foster, R.G.; Wulff, K. The rhythm of rest and excess. Nat. Rev. Neurosci. 2005, 6, 407–414. [Google Scholar] [CrossRef]
- Rijo-Ferreira, F.; Takahashi, J. Genomics of circadian rhythms in health and disease. Genome Med. 2019, 11, 82. [Google Scholar] [CrossRef]
- Cederroth, C.; Albrecht, U.; Bass, J.; Brown, S.; Dyhrfjeld-Johnsen, J.; Gachon, F.; Green, C.; Hastings, M.; Helfrich-Forster, C.; Hogenesch, J.; et al. Medicine in the fourth dimension. Cell Metab. 2019, 30, 238–250. [Google Scholar] [CrossRef]
- Solt, J. Changing perspectives on daylight: Science, technology and culture. In Sponsored Supplement to Science/AAS; Science/AAAS Custom Publishing Office: Washington, DC, USA, 2017; pp. 1–45. [Google Scholar]
- Walker, W.; Melendez-Fernandez, O.; Nelson, R.; Reiter, R. Global climate change and invariable photoperiods: A mismatch that jeopardizes animal fitness. Ecol. Evol. 2019, 9, 10044–10054. [Google Scholar] [CrossRef]
- Knoop, M.; Stefani, O.; Bueno, B.; Matusiak, B.; Hobday, R.; Wirz-Justice, A.; Martiny, K.; Kantermann, T.; Aarts, M.; Zemmouri, N.; et al. Daylight: What makes the difference? Lighting Res. Technol. 2019. [Google Scholar] [CrossRef]
- Bartley, S.; Ball, R. Effects of intermittent photic input on brightness, hue, saturation, visual acuity, and stereopsis. Am. J. Optom. Arch. Am. Acad. Optom. 1969, 46, 315–318. [Google Scholar] [CrossRef]
- Guth, S.; Eastman, A. Brightness difference in seeing. Am. J. Optom. Arch. Am. Acad. Optom. 1954, 31, 567–577. [Google Scholar] [CrossRef] [PubMed]
- Nelson, J. Ideal seeing conditions; the study of the human visual system as a basis for prescribing lighting. Br. Ind. Med. 1945, 2, 224–232. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hunt, R. The effects of daylight and tungsten light-adaptation on color perception. J. Opt. Soc. Am. 1950, 40, 362–371. [Google Scholar] [CrossRef]
- Luckiesh, M. A note on color preference. Am. J. Psychol. 1916, 27, 251–255. [Google Scholar] [CrossRef]
- Boyce, P. Human Factors in Lighting, 2nd ed.; CRC Press: London, UK; New York, NY, USA, 2003. [Google Scholar]
- Hopkinson, R. Glare from daylighting in buildings. Appl. Ergonom. 1972, 3, 206–215. [Google Scholar] [CrossRef]
- Borisuit, A.; Linhart, F.; Scartezzini, J.-L.; Münch, M. Effects of realistic office day- and electric lighting conditions on visual comfort, alertness and mood. Lighting Res. Technol. 2014, 47, 192–209. [Google Scholar] [CrossRef]
- Pierson, C.; Wienold, J.; Bodart, M. Review of factors influencing discomfort glare perception from daylight. Leukos 2018, 14, 111–148. [Google Scholar] [CrossRef]
- Veitch, J.; Newsham, G.; Boyce, P.; Jones, C. Lighting appraisal, well-being, and performance in open-plan offices: A linked mechansisms approach. Lighting Res. Technol. 2008, 40, 133–151. [Google Scholar] [CrossRef]
- Chinazzo, G.; Wienold, J.; Andersen, M. Daylight affects human thermal perception. Sci. Rep. 2019, 9, 13690. [Google Scholar] [CrossRef]
- Te Kulve, M.; Schlangen, L.; van Marken Lichtenbelt, W. Interactions between the perception of light and temperature. Indoor Air 2018, 28, 881–891. [Google Scholar] [CrossRef]
- Cajochen, C.; Münch, M.; Kobialka, S.; Kräuchi, K.; Steiner, R.; Oelhafen, P.; Orgül, S.; Wirz-Justice, A. High sensitivity of human melatonin, alertness, thermoregulation and heart rate to short wavelength light. J. Clin. Endocrinol. Metab. 2005, 90, 1311–1316. [Google Scholar] [CrossRef] [PubMed]
- Lockley, S.W.; Evans, E.E.; Scheer, F.A.J.L.; Brainard, G.C.; Czeisler, C.A.; Aeschbach, D. Short-wavelength sensitivity for the direct effects of light on alertness, vigilance, and the waking electroencephalogram in humans. Sleep 2006, 29, 161–168. [Google Scholar] [PubMed]
- Revell, V.; Skene, D. Light-induced melatonin suppression in humans with polychromatic and monochromatic light. Chronobiol. Int. 2007, 24, 1125–1137. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.; Flynn-Evans, E.; Aeschbach, D.; Brainard, G.; Czeisler, C.; Lockley, S. Diurnal spectral sensitivity of the acute alerting effects of light. Sleep 2014, 37, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Souman, J.; Tinga, A.; te Pas, S.; van Ee, R.; Vlaskamp, B. Acute alerting effects of light: A systematic literature review. Behav. Brain Res. 2018, 337, 228–239. [Google Scholar] [CrossRef] [PubMed]
- Cajochen, C.; Jud, C.; Münch, M.; Kobialka, S.; Wirz-Justice, A.; Albrecht, U. Evening exposure to blue light stimulates the expression of the clock gene PER2 in humans. Eur. J. Neurosci. 2006, 23, 1082–1086. [Google Scholar] [CrossRef] [PubMed]
- Santhi, N.; Groeger, J.; Archer, S.; Gimenez, M.; Schlangen, L.; Dijk, D. Morning sleep inertia in alertness and performance: Effect of cognitive domain and white light conditions. PLoS ONE 2013, 8, e79688. [Google Scholar] [CrossRef]
- Prayag, A.; Jost, S.; Avouac, P.; Dumortier, D.; Gronfier, C. Dynamics of non-visual responses in humans: As fast as lightning? Front. Neurosci. 2019, 13, 126. [Google Scholar] [CrossRef]
- Vandewalle, G.; Maquet, P.; Dijk, D. Light as a modulator of cognitive brain function. Trends Cogn. Sci. 2009, 13, 429–438. [Google Scholar] [CrossRef]
- Münch, M.; Scheuermaier, K.; Zhang, R.; Dunne, S.; Guzik, A.; Silva, E.J.; Ronda, J.; Duffy, J. Effects on subjective and objective alertness and sleep in response to evening light exposure in older subjects. Behav. Brain Res. 2011, 224, 272–278. [Google Scholar] [CrossRef]
- Chellappa, S.L.; Steiner, R.; Blattner, P.; Oelhafen, P.; Götz, T.; Cajochen, C. Non-visual effects of light on melatonin, alertness and cognitive performance: Can blue-enriched light keep us alert? PLoS ONE 2011, 6, e16429. [Google Scholar] [CrossRef] [PubMed]
- Rüger, M.; Gordijn, M.C.M.; Beersma, D.G.M.; de Vries, B.; Daan, S. Time-of-day-dependent effects of bright light exposure on human psychophysiology: Comparison of daytime and nighttime exposure. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 290, R1413–R1420. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.M.; Khalsa, S.B.S.; Scheer, F.A.J.L.; Cajochen, C.; Lockley, S.W.; Czeisler, C.A.; Wright, K. Acute effects of bright light exposure on cortisol levels. J. Biol. Rhythms 2010, 25, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Münch, M.; Kobialka, S.; Steiner, R.; Oelhafen, P.; Wirz-Justice, A.; Cajochen, C. Wavelength-dependent effects of evening light exposure on sleep architecture and sleep EEG power density in men. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 290, R1421–R1428. [Google Scholar] [CrossRef] [PubMed]
- Brainard, G.; Hanifin, J.; Greeson, J.; Byrne, B.; Glickman, G.; Gerner, E.; Rollag, M. Action spectrum for melatonin regulation in humans: Evidence for a novel circadian photoreceptor. J. Neurosci. 2001, 21, 6405–6412. [Google Scholar] [CrossRef]
- Thapan, K.; Arendt, J.; Skene, D.J. An action spectrum for melatonin suppression: Evidence for a novel non-rod, non-cone photoreceptor system in humans. J. Physiol. London 2001, 535, 261–267. [Google Scholar] [CrossRef]
- Gooley, J.; Rajaratnam, S.; Brainard, G.; Kronauer, R.; Czeisler, C.; Lockley, S. Spectral responses of the human circadian system depend on the irradiance and duration of exposure to light. Sci. Transl. Med. 2010, 2, 31ra33. [Google Scholar] [CrossRef]
- Zaidi, F.; Hull, J.; Peirson, S.; Wulff, K.; Aeschbach, D.; Gooley, J.; Brainard, G.; Gregory-Evans, K.; Rizzo Iii, J.; Czeisler, C.; et al. Short-wavelength light sensitivity of circadian, pupillary, and visual awareness in humans lacking an outer retina. Curr. Biol. 2007, 17, 2122–2128. [Google Scholar] [CrossRef]
- Boubekri, M.; Cheung, I.N.; Reid, K.; Wang, C.; Zee, P. Impact of windows and daylight exposure on overall health and sleep quality of office workers: A case-control pilot study. J. Clin. Sleep Med. 2014, 10, 603–611. [Google Scholar] [CrossRef]
- Figueiro, M.; Rea, M. Office lighting and personal light exposures in two seasons: Impact on sleep and mood. Lighting Res. Technol. 2016, 48, 352–364. [Google Scholar] [CrossRef]
- Viola, A.U.; James, L.; Schlangen, L.; Dijk, D. Blue-enriched white light in the workplace improves self-reported alertness, performance and sleep quality. Scand. J. Work Environ. Health 2008, 34, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Vetter, C.; Juda, M.; Lang, D.; Wojtysiak, A.; Roenneberg, T. Blue-enriched office light competes with natural light as a zeitgeber. Scand J. Work Environ. Health 2011, 34, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Maierova, L.; Borisuit, A.; Scartezzini, J.-L.; Jaeggi, S.; Schmidt, C.; Munch, M. Diurnal variations of hormonal secretion, alertness and cognition in extreme chronotypes under different lighting conditions. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Begeman, S.; Beld, G.; Tenner, A. Daylight, artificial light and people in an office environment, overview of visual and biological responses. Int. J. Ind. Ergon. 1997, 20, 231–239. [Google Scholar] [CrossRef]
- Beute, F.; de Kort, Y. The natural context of wellbeing: Ecological momentary assessment of the influence of nature and daylight on affect and stress for individuals with depression levels varying from none to clinical. Health Place 2018, 49, 7–18. [Google Scholar] [CrossRef] [PubMed]
- Veitch, J.; Bisegna, F.; Hubalek, S.; Knoop, M.; Koga, Y.; Noguchi, H.; Schierz, C.; Thorns, P.; Vries, A. Research Roadmap for Healthful Interior Lighting Applications; CIE: Vienna, Austria, 2016. [Google Scholar]
- Heschong, L. Daylighting and human performance. ASHRAE J. 2002, 44, 65–67. [Google Scholar]
- Cheung, I.; Zee, P.; Shalman, D.; Malkani, R.; Kang, J.; Reid, K. Morning and evening blue-enriched light exposure alters metabolic function in normal weight adults. PLoS ONE 2016, 11, e0155601. [Google Scholar] [CrossRef]
- Melanson, E.; Ritchie, H.; Dear, T.; Catenacci, V.; Shea, K.; Connick, E.; Moehlman, T.; Stothard, E.; Higgins, J.; McHill, A.; et al. Daytime bright light exposure, metabolism, and individual differences in wake and sleep energy expenditure during circadian entrainment and misalignment. Neurobiol. Sleep Circadian Rhythm. 2018, 4, 49–56. [Google Scholar] [CrossRef]
- Danilenko, K.; Mustafina, S.; Pechenkina, E. Bright light for weight loss: Results of a controlled crossover trial. Obes Facts 2013, 6, 28–38. [Google Scholar] [CrossRef]
- Wams, E.; Woelders, T.; Marring, I.; van Rosmalen, L.; Beersma, D.; Gordijn, M.; Hut, R. Linking light exposure and subsequent sleep: A field polysomnography study in humans. Sleep 2017, 40. [Google Scholar] [CrossRef]
- Cajochen, C.; Freyburger, M.; Basishvili, T.; Garbazza, C.; Rudzik, F.; Renz, C.; Kobayashi, K.; Shirakawa, Y.; Stefani, O.; Weibel, J. Effect of daylight LED on visual comfort, melatonin, mood, waking performance and sleep. Lighting Res. Technol. 2019. [Google Scholar] [CrossRef]
- Cajochen, C.; Reichert, C.; Maire, M.; Schlangen, L.; Schmidt, C.; Viola, A.; Gabel, V. Evidence that homeostatic sleep regulation depends on ambient lighting conditions during wakefulness. Clocks Sleep 2019, 1, 517–531. [Google Scholar] [CrossRef]
- Wright, K., Jr.; McHill, A.; Birks, B.; Griffin, B.; Rusterholz, T.; Chinoy, E. Entrainment of the human circadian clock to the natural light-dark cycle. Curr. Biol. 2013, 23, 1554–1558. [Google Scholar] [CrossRef] [PubMed]
- Stothard, E.; McHill, A.; Depner, C.; Birks, B.; Moehlman, T.; Ritchie, H.; Guzzetti, J.; Chinoy, E.; LeBourgeois, M.; Axelsson, J.; et al. Circadian entrainment to the natural light-dark cycle across seasons and the weekend. Curr. Biol. 2017, 27, 508–513. [Google Scholar] [CrossRef]
- Münch, M.; Nowozin, C.; Regente, J.; Bes, F.; De Zeeuw, J.; Hadel, S.; Wahnschaffe, A.; Kunz, D. Blue-enriched morning light as a countermeasure to light at the wrong time: Effects on cognition, sleepiness, sleep, and circadian phase. Neuropsychobiology 2016, 74, 207–218. [Google Scholar] [CrossRef]
- Cajochen, C.; Zeitzer, J.M.; Czeisler, C.A.; Dijk, D. Dose-response relationship for light intensity and alertness and its ocular and EEG correlates. Sleep Res. Onl. 1999, 2, 517. [Google Scholar]
- Smolders, K.; Peeters, S.; Vogels, I.; de Kort, Y. Investigation of dose-response relationships for effects of white light exposure on correlates of alertness and executive control during regular daytime working hours. J. Biol. Rhythm. 2018, 33, 649–661. [Google Scholar] [CrossRef]
- Sahin, L.; Wood, B.; Plitnick, B.; Figueiro, M. Daytime light exposure: Effects on biomarkers, measures of alertness, and performance. Behav. Brain Res. 2014, 274, 176–185. [Google Scholar] [CrossRef]
- Allen, A.; Hazelhoff, E.; Martial, F.; Cajochen, C.; Lucas, R. Exploiting metamerism to regulate the impact of a visual display on alertness and melatonin suppression independent of visual appearance. Sleep 2018, 41, zsy100. [Google Scholar] [CrossRef]
- Rahman, S.A.; St Hilaire, M.A.; Lockley, S. The effects of spectral tuning of evening ambient light on melatonin suppression, alertness and sleep. Physiol. Behav. 2017, 177, 221–229. [Google Scholar] [CrossRef]
- De Zeeuw, J.; Papakonstantinou, A.; Nowozin, C.; Stotz, S.; Zaleska, M.; Hädel, S.; Bes, F.; Münch, M.; Kunz, D. Living in biological darkness: Objective sleepiness and the pupillary light responses are affected by different metameric lighting conditions during daytime. J. Biol. Rhythm. 2019, 34, 410–431. [Google Scholar] [CrossRef] [PubMed]
- Youngstedt, S.; Elliott, J.; Kripke, D. Human circadian phase-response curves for exercise. J. Physiol. London 2019, 597, 2253–2268. [Google Scholar] [CrossRef] [PubMed]
- Barger, L.; Wright, K.J.; Hughes, R.; Czeisler, C. Daily exercise facilitates phase delays of circadian melatonin rhythm in very dim light. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004, 286, R1077–R1084. [Google Scholar] [CrossRef] [PubMed]
- Knaier, R.; Meister, S.; Aeschbacher, T.; Gemperle, D.; Rossmeissl, A.; Cajochen, C.; Schmidt-Trucksass, A. Dose-response relationship between light exposure and cycling performance. Scand. J. Med. Sci. Sports 2016, 26, 794–801. [Google Scholar] [CrossRef]
- Aschoff, J.; Wever, R. Spontanperiodik des Menschen bei Auschluss aller Zeitgeber. Die Naturwissenschaften 1962, 49, 337–342. [Google Scholar] [CrossRef]
- Gronfier, C.; Wright, K.P., Jr.; Kronauer, R.E.; Czeisler, C.A. Entrainment of the human circadian pacemaker to longer-than-24-h days. Proc. Natl. Acad. Sci. USA 2007, 104, 9081–9086. [Google Scholar] [CrossRef]
- Wright, K.P., Jr.; Hughes, R.J.; Kronauer, R.E.; Dijk, D.J.; Czeisler, C.A. Intrinsic near-24-h pacemaker period determines limits of circadian entrainment to a weak synchronizer in humans. Proc. Natl. Acad. Sci. USA 2001, 98, 14027–14032. [Google Scholar] [CrossRef]
- Boivin, D.B.; Duffy, J.F.; Kronauer, R.E.; Czeisler, C.A. Dose-response relationships for resetting of human circadian clock by light. Nature 1996, 379, 540–542. [Google Scholar] [CrossRef]
- Scheer, F.A.; Wright, K.P., Jr.; Kronauer, R.E.; Czeisler, C.A. Plasticity of the intrinsic period of the human circadian timing system. PLoS ONE 2007, 2, e721. [Google Scholar] [CrossRef]
- Eastman, C.; Miescke, K. Entrainment of circadian rhythms with 26-h bright light and sleep-wake schedules. Am J. Physiol. 1990, 259, R1189–R1197. [Google Scholar] [CrossRef]
- Wever, R.A. The Circadian System of Man: Results of Experiments under Temporal Isolation; Springer: New York, NY, USA, 1979. [Google Scholar]
- Czeisler, C.A.; Richardson, G.S.; Zimmerman, J.Z.; Moore-Ede, M.C.; Weitzman, E.D. Entrainment of human circadian rhythms by light-dark cycles: A reassessment. Photochem. Photobiol. 1981, 34, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Emens, J.S.; Yuhas, K.; Rough, J.; Kochar, N.; Peters, D.; Lewy, A. Phase angle of entrainment in morning- and evening-types under naturalistic conditions. Chronobiol. Int. 2009, 26, 474–493. [Google Scholar] [CrossRef] [PubMed]
- Moreno, C.; Vasconcelos, S.; Marqueze, E.; Lowden, A.; Middleton, B.; Fischer, F.; Louzada, F.; Skene, D. Sleep patterns in Amazon rubber tappers with and without electric light at home. Sci. Rep. 2015, 5, 14074. [Google Scholar] [CrossRef] [PubMed]
- Woelders, T.; Wams, E.J.; Gordijn, M.; Beersma, D.; Hut, R. Integration of color and intensity increases time signal stability for the human circadian system when sunlight is obscured by clouds. Sci. Rep. 2018, 8, 15214. [Google Scholar] [CrossRef]
- Roenneberg, T.; Foster, R.G. Twilight times: Light and the circadian system. Photochem. Photobiol. 1997, 66, 549–561. [Google Scholar] [CrossRef]
- Danilenko, K.V.; Wirz-Justice, A.; Kräuchi, K.; Weber, J.M.; Terman, M. The human circadian pacemaker can see by the dawn`s early light. J. Biol. Rhythm. 2000, 15, 437–446. [Google Scholar] [CrossRef]
- Walmsley, L.; Hanna, L.; Mouland, J.; Martial, F.; West, A.; Smedley, A.; Bechtold, D.; Webb, A.; Lucas, R.; Brown, T. Colour as a signal for entraining the mammalian circadian clock. PLoS Biol. 2015, 13, e1002127. [Google Scholar] [CrossRef]
- Mouland, J.W.; Martial, F.; Watson, A.; Lucas, R.; Brown, T. Cones support alignment to an inconsistent world by suppressing mouse circadian responses to the blue colors associated with twilight. Curr. Biol. 2019, 29, 4260–4267. [Google Scholar] [CrossRef]
- Brainard, G.; Rollag, M.; Hanifin, J.; van den Beld, G.; Sanford, B. The effect of polarized versus nonpolarized light on melatonin regulation in humans. Photochem. Photobiol. 2000, 71, 766–770. [Google Scholar] [CrossRef]
- Dacey, D.; Liao, H.; Peterson, B.; Robinson, F.; Smith, V.; Pokorny, J.; Yau, K.; Gamlin, P. Melanopsin-expressing ganglion cells in primate retina signal colour and irradiance and project to the LGN. Nature 2005, 433, 749–754. [Google Scholar] [CrossRef]
- Rüger, M.; Gordijn, M.C.M.; Beersma, D.G.M.; DeVries, B.; Daan, S. Nasal versus temporal illumination of the human retina: Effects on core body temperature, melatonin, and circadian phase. J. Biol. Rhythm. 2005, 20, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Visser, E.K.; Beersma, D.G.M.; Daan, S. Melatonin suppression by light in humans is maximal when the nasal part of the retina is illuminated. J. Biol. Rhythm. 1999, 14, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Glickman, G.; Hanifin, J.; Rollag, M.; Wang, J.; Cooper, H.; Brainard, G. Inferior retinal light exposure is more effective than superior retinal exposure in suppressing melatonin in humans. J. Biol. Rhythm. 2003, 18, 71–79. [Google Scholar] [CrossRef]
- Cajochen, C.; Di Biase, R.; Imai, M. Interhemispheric EEG asymmetries during unilateral bright-light exposure and subsequent sleep in humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R1053–R1060. [Google Scholar] [CrossRef] [PubMed]
- Wehr, T.A. Effect of seasonal changes in daylength on human neuroendocrine function. Horm. Res. 1998, 49, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Rabstein, S.; Burek, K.; Lehnert, M.; Beine, A.; Vetter, C.; Harth, V.; Putzke, S.; Kantermann, T.; Walther, J.; Wang-Sattler, R.; et al. Differences in twenty-four-hour profiles of blue-light exposure between day and night shifts in female medical staff. Sci. Total Environ. 2019, 653, 1025–1033. [Google Scholar] [CrossRef]
- Lowden, A.; Ozturk, G.; Reynolds, A.; Bjorvatn, B. Working Time society consensus statements: Evidence based interventions using light to improve circadian adaptation to working hours. Ind. Health 2019, 57, 213–227. [Google Scholar] [CrossRef]
- Boivin, D.B.; Boudreau, P.; James, F.O.; Kin, N.M. Photic resetting in night-shift work: Impact on nurses’ sleep. Chronobiol. Int. 2012, 29, 619–628. [Google Scholar] [CrossRef]
- Nowozin, C.; Wahnschaffe, A.; Rodenbeck, A.; de Zeeuw, J.; Hadel, S.; Kozakov, R.; Schopp, H.; Munch, M.; Kunz, D. Applying melanopic lux to measure biological light effects on melatonin suppression and subjective sleepiness. Curr. Alzheimer Res. 2017, 14, 1042–1052. [Google Scholar] [CrossRef]
- Lok, R.; Woelders, T.; Gordijn, M.; Hut, R.; Beersma, D.G. White light during daytime does not improve alertness in well-rested individuals. J. Biol. Rhythm. 2018, 33, 637–648. [Google Scholar] [CrossRef]
- Mure, L.; Vinberg, F.; Hanneken, A.; Panda, S. Functional diversity of human intrinsically photosensitive retinal ganglion cells. Science 2019, 366, 1251–1255. [Google Scholar] [CrossRef] [PubMed]
- Kaynezhad, P.; Tachtsidis, I.; Jeffery, G. Optical monitoring of retinal respiration in real time: 670 nm light increases the redox state of mitochondria. Exp. Eye Res. 2016, 152, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Sivapathasuntharam, C.; Sivaprasad, S.; Hogg, C.; Jeffery, G. Improving mitochondrial function significantly reduces the rate of age related photoreceptor loss. Exp. Eye Res. 2019, 185, 107691. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.; Santhi, N.; St Hilaire, M.; Gronfier, C.; Bradstreet, D.; Duffy, J.; Lockley, S.; Kronauer, R.; Czeisler, C. Human responses to bright light of different durations. J. Physiol. London 2012, 590, 3103–3112. [Google Scholar] [CrossRef] [PubMed]
- Spitschan, M.; Bock, A.; Ryan, J.; Frazzetta, G.; Brainard, D.; Aguirre, G. The human visual cortex response to melanopsin-directed stimulation is accompanied by a distinct perceptual experience. Proc. Natl. Acad. Sci. USA 2017, 114, 12291–12296. [Google Scholar] [CrossRef]
- Faulkner, S.; Bee, P.; Meyer, N.; Dijk, D.; Drake, R. Light therapies to improve sleep in intrinsic circadian rhythm sleep disorders and neuro-psychiatric illness: A systematic review and meta-analysis. Sleep Med. Rev. 2019, 46, 108–123. [Google Scholar] [CrossRef]
- Geoffroy, P.; Schroder, C.; Reynaud, E.; Bourgin, P. Efficacy of light therapy versus antidepressant drugs, and of the combination versus monotherapy, in major depressive episodes: A systematic review and meta-analysis. Sleep Med. Rev. 2019, 48, 101213. [Google Scholar] [CrossRef]
- Gottlieb, J.; Benedetti, F.; Geoffroy, P.; Henriksen, T.; Lam, R.; Murray, G.; Phelps, J.; Sit, D.; Swartz, H.; Crowe, M.; et al. The chronotherapeutic treatment of bipolar disorders: A systematic review and practice recommendations from the ISBD task force on chronotherapy and chronobiology. Bipolar. Disord. 2019, 21, 741–773. [Google Scholar] [CrossRef]
- Penders, T.; Stanciu, C.; Schoemann, A.; Ninan, P.; Bloch, R.; Saeed, S. Bright light therapy as augmentation of pharmacotherapy for treatment of depression: A systematic review and meta-analysis. Prim. Care Companion CNS Disord. 2016, 18. [Google Scholar] [CrossRef]
- Beaulieu, C.; Rufiange, M.; Dumont, M.; Lachapelle, P. Modulation of ERG retinal sensitivity parameters with light environment and photoperiod. Doc. Ophthalmol. 2009, 118, 89–99. [Google Scholar] [CrossRef]
- Danilenko, K.; Plisov, I.; Wirz-Justice, A.; Hebert, M. Human retinal light sensitivity and melatonin rhythms following four days in near darkness. Chronobiol. Int. 2009, 26, 93–107. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, A.; Wisniewski, S.; Healey, B.; Pattyn, N.; Kunz, D.; Basner, M.; Munch, M. Impact of long-term daylight deprivation on retinal light sensitivity, circadian rhythms and sleep during the Antarctic winter. Sci. Rep. 2018, 8, 16185. [Google Scholar] [CrossRef] [PubMed]
- Owen, J.; Arendt, J. Melatonin suppression in human subjects by bright and dim light in antarctica: Time and season-dependent effects. Neurosci. Lett. 1992, 137, 181–184. [Google Scholar] [CrossRef]
- Jasser, S.A.; Hanifin, J.; Rollag, M.; Brainard, G. Dim light adaptation attenuates acute melatonin suppression in humans. J. Biol. Rhythm. 2006, 21, 394–404. [Google Scholar] [CrossRef] [PubMed]
- Hébert, M.; Martin, S.K.; Eastman, C.I. The effects of prior light history on the suppression of melatonin by light in humans. J. Pineal Res. 2002, 33, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.; Scheer, F.; Czeisler, C.; Aeschbach, D. Direct effects of light on alertness, vigilance, and the waking electroencephalogram in humans depend on prior light history. Sleep 2013, 36, 1239–1246. [Google Scholar] [CrossRef]
- Chakraborty, R.; Ostrin, L.; Nickla, D.; Iuvone, P.; Pardue, M.; Stone, R. Circadian rhythms, refractive development, and myopia. Ophthalmic Physiol. Opt. 2018, 38, 217–245. [Google Scholar] [CrossRef]
- Riemersma-van der Lek, R.F.; Swaab, D.F.; Twisk, J.; Hol, E.M.; Hoogendijk, W.J.G.; Van Someren, E.J.W. Effect of bright light and melatonin on cognitive and noncognitive function in elderly residents of group care facilities: A randomized controlled trial. JAMA 2008, 299, 2642–2655. [Google Scholar] [CrossRef]
- Bromundt, V.; Wirz-Justice, A.; Boutellier, M.; Winter, S.; Haberstroh, M.; Terman, M.; Munch, M. Effects of a dawn-dusk simulation on circadian rest-activity cycles, sleep, mood and well-being in dementia patients. Exp. Gerontol. 2019, 124, 110641. [Google Scholar] [CrossRef]
- Wahnschaffe, A.; Nowozin, C.; Haedel, S.; Rath, A.; Appelhof, S.; Munch, M.; Kunz, D. Implementation of dynamic lighting in a nursing home: Impact on agitation but not on rest-activity patterns. Curr. Alzheimer Res. 2017, 14, 1076–1083. [Google Scholar] [CrossRef]
- Figueiro, M.; Plitnick, B.; Roohan, C.; Sahin, L.; Kalsher, M.; Rea, M. Effects of a tailored lighting intervention on sleep quality, rest-activity, mood, and behavior in older adults with Alzheimer disease and related dementias: A randomized clinical trial. Clin. Sleep Med. 2019, 15, 1757–1767. [Google Scholar] [CrossRef] [PubMed]
- Figueiro, M.; Plitnick, B.; Lok, A.; Jones, G.; Higgins, P.; Hornick, T.; Rea, M.S. Tailored lighting intervention improves measures of sleep, depression, and agitation in persons with Alzheimer’s disease and related dementia living in long-term care facilities. Clin. Interv. Aging 2014, 9, 1527–1537. [Google Scholar] [CrossRef] [PubMed]
- Fargason, R.; Fobian, A.; Hablitz, L.; Paul, J.; White, B.; Cropsey, K.; Gamble, K. Correcting delayed circadian phase with bright light therapy predicts improvement in ADHD symptoms: A pilot study. J. Psychiatr. Res. 2017, 91, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Bromundt, V.; Wirz-Justice, A.; Kyburz, S.; Opwis, K.; Dammann, G.; Cajochen, C. Circadian sleep-wake cyles, well-being, and light therapy in borderline personality disorder. J. Pers. Disord. 2012, 27, 680–696. [Google Scholar] [CrossRef] [PubMed]
- Rutten, S.; Vriend, C.; van den Heuvel, O.; Smit, J.; Berendse, H.; van der Werf, Y. Bright light therapy in Parkinson’s disease: An overview of the background and evidence. Parkinson’s Dis. 2012, 2012, 767105. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Smilowska, K.; van Wamelen, J.; Schoutens, A.; Meinders, M.; Bloem, B. Blue light therapy glasses in Parkinson’s disease: patients’ experience. Parkinson’s Dis. 2019, 2019, 1906271. [Google Scholar] [CrossRef] [PubMed]
- Burkhalter, H.; Wirz-Justice, A.; Denhaerynck, K.; Fehr, T.; Steiger, J.; Venzin, R.; Cajochen, C.; Weaver, T.; De Geest, S. The effect of bright light therapy on sleep and circadian rhythms in renal transplant recipients: A pilot randomized, multicentre wait-list controlled trial. Transpl. Int. 2015, 28, 59–70. [Google Scholar] [CrossRef]
- Johnson, J.; Garland, S.; Carlson, L.; Savard, J.; Simpson, J.; Ancoli-Israel, S.; Campbell, T. Bright light therapy improves cancer-related fatigue in cancer survivors: A randomized controlled trial. J. Cancer Surviv. 2018, 12, 206–215. [Google Scholar] [CrossRef]
- Wirz-Justice, A.; Graw, P.; Kräuchi, K.; Sarrafzadeh, A.; English, J.; Arendt, J.; Sand, L. Natural’ light treatment of seasonal affective disorder. J. Affect. Disord. 1996, 37, 109–120. [Google Scholar] [CrossRef]
- Gbyl, K.; Ostergaard Madsen, H.; Dunker Svendsen, S.; Petersen, P.; Hageman, I.; Volf, C.; Martiny, K. Depressed patients hospitalized in southeast-facing rooms are discharged earlier than patients in Northwest-facing rooms. Neuropsychobiology 2016, 74, 193–201. [Google Scholar] [CrossRef]
- Gbyl, K.; Ostergaard Madsen, H.; Dunker Svendsen, S.; Petersen, P.; Hageman, I.; Volf, C.; Martiny, K. Erratum. Neuropsychobiology 2017, 75, 52. [Google Scholar] [CrossRef]
- Benedetti, F.; Colombo, C.; Barbini, B.; Campori, E.; Smeraldi, E. Morning sunlight reduces length of hospitalization in bipolar depression. J. Affect. Disord. 2001, 62, 221–223. [Google Scholar] [CrossRef]
- Gimenez, M.; Geerdinck, L.; Versteylen, M.; Leffers, P.; Meekes, G.; Herremans, H.; de Ruyter, B.; Bikker, J.; Kuijpers, P.; Schlangen, L. Patient room lighting influences on sleep, appraisal and mood in hospitalized people. J. Sleep Res. 2017, 26, 236–246. [Google Scholar] [CrossRef] [PubMed]
- Luetz, A.; Grunow, J.; Morgeli, R.; Rosenthal, M.; Weber-Carstens, S.; Weiss, B.; Spies, C. Innovative ICU solutions to prevent and reduce delirium and post-intensive care unit syndrome. Semin. Respir. Crit. Care Med. 2019, 40, 673–686. [Google Scholar] [CrossRef]
- Beauchemin, K.M.; Hays, P. Dying in the dark: Sunshine, gender and outcomes in myocardial infarction. J. R. Soc. Med. 1998, 91, 352–354. [Google Scholar] [CrossRef]
- Ritchie, H.; Stothard, E.; Wright, K.J. Entrainment of the human circadian clock to the light-dark cycle and its impact on patients in the ICU and nursing home settings. Curr. Pharma. Des. 2015, 21, 3438–3442. [Google Scholar] [CrossRef]
- Munch, M.; Schmieder, M.; Bieler, K.; Goldbach, R.; Fuhrmann, T.; Zumstein, N.; Vonmoos, P.; Scartezzini, J.; Wirz-Justice, A.; Cajochen, C. Bright light delights: Effects of daily light exposure on emotions, rest-activity cycles, sleep and melatonin secretion in severely demented patients. Curr. Alzheimer Res. 2017, 14, 1063–1075. [Google Scholar]
- Gies, P.; van Deventer, E.; Green, A.; Sinclair, C.; Tinker, R. Review of the global solar UV index 2015 workshop report. Health Phys. 2018, 114, 84–90. [Google Scholar] [CrossRef]
- Lanca, C.; Teo, A.; Vivagandan, A.; Htoon, H.; Najjar, R.; Spiegel, D.; Pu, S.; Saw, S. The effects of different outdoor environments, sunglasses and hats on light levels: Implications for myopia prevention. Transl. Vis. Sci. Technol. 2019, 8, 7. [Google Scholar] [CrossRef]
- Phillips, A.; Vidafar, P.; Burns, A.; McGlashan, E.; Anderson, C.; Rajaratnam, S.; Lockley, S.; Cain, S. High sensitivity and interindividual variability in the response of the human circadian system to evening light. Proc. Natl. Acad. Sci. USA 2019, 116, 12019–12024. [Google Scholar] [CrossRef]
- McGlashan, E.; Poudel, G.; Vidafar, P.; Drummond, S.; Cain, S. Imaging individual differences in the response of the human suprachiasmatic area to light. Front. Neurol. 2018, 9, 1022. [Google Scholar] [CrossRef] [PubMed]
- Watson, L.; Phillips, A.K.; Hosken, I.; McGlashan, E.; Anderson, C.; Lack, L.; Lockley, S.; Rajaratnam, S.M.; Cain, S. Increased sensitivity of the circadian system to light in delayed sleep-wake phase disorder. J. Physiol. London 2018, 596, 6249–6261. [Google Scholar] [CrossRef] [PubMed]
- Akacem, L.; Wright, K., Jr.; LeBourgeois, M. Sensitivity of the circadian system to evening bright light in preschool-age children. Physiol. Rep. 2018, 6, e13617. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, S.; Nagafuchi, Y.; Lee, S.I.; Harada, T. Influence of light at night on melatonin suppression in children. J. Clin. Endocrinol. Metab. 2014, 99, 3298–3303. [Google Scholar] [CrossRef]
- Nagare, R.; Rea, M.S.; Plitnick, B.; Figueiro, M. Nocturnal melatonin suppression by adolescents and adults for different levels, spectra, and durations of light exposure. J. Biol. Rhythm. 2019, 34, 178–194. [Google Scholar] [CrossRef]
- Chellappa, S.; Bromundt, V.; Frey, S.; Steinemann, A.; Schmidt, C.; Schlote, T.; Goldblum, D.; Cajochen, C. Association of intraocular cataract lens replacement with circadian rhythms, cognitive function, and sleep in older adults. JAMA Ophthalmol. 2019, 137, 878–885. [Google Scholar] [CrossRef]
- Turner, P.L.; Mainster, M.A. Circadian photoreception: Ageing and the eye’s important role in systemic health. Br. J. Ophthalmol. 2008, 92, 1439–1444. [Google Scholar] [CrossRef]
- Münch, M.; Ladaique, M.; Roemer, S.; Hashemi, K.; Kawasaki, A. Melanopsin-mediated acute light responses measured in winter and in summer: Seasonal variations in adults with and without cataracts. Front. Neurol. 2017, 8, 464. [Google Scholar] [CrossRef]
- Najjar, R.; Chiquet, C.; Teikari, P.; Cornut, P.; Claustrat, B.; Denis, P.; Cooper, H.; Gronfier, C. Aging of non-visual spectral sensitivity to light in humans: Compensatory mechanisms? PLoS ONE 2014, 9, e85837. [Google Scholar] [CrossRef]
- Daneault, V.; Vandewalle, G.; Hébert, M.; Teikari, P.; Mure, L.; Doyon, J.; Gronfier, C.; Cooper, H.; Dumont, M.; Carrier, J. Does pupil constriction under blue and green monochromatic light exposure change with age? J. Biol. Rhythm. 2012, 27, 257–264. [Google Scholar] [CrossRef]
- Park, J.; Moura, A.; Raza, A.; Rhee, D.; Kardon, R.; Hood, D. Toward a clinical protocol for assessing rod, cone, and melanopsin contributions to the human pupil response. Investig. Ophthalmol. Vis. Sci. 2011, 52, 6624–6635. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Shen, C.; Zhang, L.; Qi, L.; Yao, L.; Chen, J.; Yang, G.; Chen, T.; Zhang, Z. Dark adaptation-induced changes in rod, cone and intrinsically photosensitive retinal ganglion cell (ipRGC) sensitivity differentially affect the pupil light response (PLR). Graefes Arch. Clin. Exp. Ophthalmol. 2015, 253, 1997–2005. [Google Scholar] [CrossRef] [PubMed]
- Yuhas, P.; Shorter, P.; McDaniel, E.; Earley, M.; Hartwick, A. Blue and red light-evoked pupil responses in photophobic subjects with TBI. Optom. Vis. Sci. 2017, 94, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Kardon, R.; Hong, S.; Kawasaki, A. Entrance pupil size predicts retina illumination in darkly pigmented eyes but not lightly pigmented eyes. Investig. Ophthalmol. Vis. Sci. 2013, 54, 5559–5567. [Google Scholar] [CrossRef][Green Version]
- Kankipati, L.; Girkin, C.; Gamlin, P. The post-illumination pupil response is reduced in glaucoma patients. Investig. Ophthalmol. Vis. Sci. 2011, 52, 2287–2292. [Google Scholar] [CrossRef]
- Münch, M.; Léon, L.; Collomb, S.; Kawasaki, A. Comparison of acute non-visual bright light responses in patients with optic nerve disease, glaucoma and healthy controls. Sci. Rep. 2015, 5, 15185. [Google Scholar] [CrossRef]
- Rukmini, A.; Milea, D.; Baskaran, M.; How, A.; Perera, S.; Aung, T.; Gooley, J. Pupillary responses to high-irradiance blue light correlate with glaucoma severity. Ophthalmology 2015, 122, 1777–1785. [Google Scholar] [CrossRef]
- Perez-Rico, C.; de la Villa, P.; Arribas-Gomez, I.; Blanco, R. Evaluation of functional integrity of the retinohypothalamic tract in advanced glaucoma using multifocal electroretinography and light-induced melatonin suppression. Exp. Eye Res. 2010, 91, 578–583. [Google Scholar] [CrossRef]
- Maynard, M.; Zele, A.; Feigl, B. Melanopsin-mediated post-illumination pupil response in early age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2015, 56, 6906–6913. [Google Scholar] [CrossRef]
- Barboni, M.T.S.; Bueno, C.; Nagy, B.; Maia, P.; Vidal, K.; Alves, R.; Reiter, R.J.; do Amaral, F.; Cipolla-Neto, J.; Ventura, D. Melanopsin system dysfunction in Smith-Magenis syndrome patients. Investig. Ophthalmol. Vis. Sci. 2018, 59, 362–369. [Google Scholar] [CrossRef]
- Bullock, B.; McGlashan, E.M.; Burns, A.; Lu, B.; Cain, S. Traits related to bipolar disorder are associated with an increased post-illumination pupil response. Psychiatry Res. 2019, 278, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Roecklein, K.; Wong, P.; Ernecoff, N.; Miller, M.; Donofry, S.; Kamarck, M.; Wood-Vasey, W.; Franzen, P. The post illumination pupil response is reduced in seasonal affective disorder. Psychiatry Res. 2013, 210, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Dumpala, S.; Zele, A.; Feigl, B. Outer retinal structure and function deficits contribute to circadian disruption in patients with type 2 diabetes. Investig. Ophthalmol. Vis. Sci. 2019, 60, 1870–1878. [Google Scholar] [CrossRef] [PubMed]
- McGlashan, E.; Nandam, L.; Vidafar, P.; Mansfield, D.; Rajaratnam, S.; Cain, S. The SSRI citalopram increases the sensitivity of the human circadian system to light in an acute dose. Psychopharmacology 2018, 235, 3201–3209. [Google Scholar] [CrossRef] [PubMed]
- Turco, M.; Biscontin, A.; Corrias, M.; Caccin, L.; Bano, M.; Chiaromanni, F.; Salamanca, M.; Mattei, D.; Salvoro, C.; Mazzotta, G.; et al. Diurnal preference, mood and the response to morning light in relation to polymorphisms in the human clock gene PER3. Sci. Rep. 2017, 7, 6967. [Google Scholar] [CrossRef] [PubMed]
- Azzi, A.; Dallmann, R.; Casserly, A.; Rehrauer, H.; Patrignani, A.; Maier, B.; Kramer, A.; Brown, S. Circadian behavior is light-reprogrammed by plastic DNA methylation. Nat. Neurosci. 2014, 17, 377–382. [Google Scholar] [CrossRef]
- Roecklein, K.; Rohan, K.; Duncan, W.; Rollag, M.; Rosenthal, N.; Lipsky, R.; Provencio, I. A missense variant (P10L) of the melanopsin (OPN4) gene in seasonal affective disorder. J. Affect. Disord. 2009, 114, 279–285. [Google Scholar] [CrossRef][Green Version]
- Lee, S.; Hida, A.; Tsujimura, S.; Morita, T.; Mishima, K.; Higuchi, S. Association between melanopsin gene polymorphism (I394T) and pupillary light reflex is dependent on light wavelength. J. Physiol. Anthropol. 2013, 32, 16. [Google Scholar] [CrossRef]
- Pereira, D.; Tufik, S.; Louzada, F.; Benedito-Silva, A.; Lopez, A.R.; Lemos, N.; Korczak, A.; D’Almeida, V.; Pedrazzoli, M. Association of the length polymorphism delayed sleep-phase syndrome: Does in the human Per3 gene with the latitude have an influence upon it? Sleep 2005, 28, 29–32. [Google Scholar]
- Mendes, M.; Darling, A.; Hart, K.; Morse, S.; Murphy, R.J.; Lanham-New, S. Impact of high latitude, urban living and ethnicity on 25-hydroxyvitamin D status: A need for multidisciplinary action? J. Steroid Biochem. Mol. Biol. 2019, 188, 95–102. [Google Scholar] [CrossRef]
- Grant, W.; Bhattoa, H.; Pludowski, P. Determinants of Vitamin D Deficiency From Sun Exposure: A Global Perspective. In Vitamin D, 4th ed.; Feldman, D., Ed.; Academic Press: New York, NY, USA, 2018; pp. 79–90. [Google Scholar]
- Darling, A.; Hart, K.; Arber, S.; Berry, J.; Morgan, P.; Middleton, B.; SA, L.-N.; Skene, D. 25-Hydroxyvitamin D status, light exposure and sleep quality in UK dwelling South Asian and Caucasian postmenopausal women. J. Steroid Biochem. Mol. Biol. 2019, 189, 265–273. [Google Scholar] [CrossRef] [PubMed]
- van der Meijden, W.; Van Someren, J.; Te Lindert, B.; Bruijel, J.; van Oosterhout, F.; Coppens, J.; Kalsbeek, A.; Cajochen, C.; Bourgin, P.; Van Someren, E. Individual differences in sleep timing relate to melanopsin-based phototransduction in healthy adolescents and young adults. Sleep 2016, 39, 1305–1310. [Google Scholar] [CrossRef] [PubMed]
- Leger, D.; Bayon, V.; Elbaz, M.; Philip, P.; Choudat, D. Underexposure to light at work and its dissociation to insomnia and sleepiness: A cross-sectional study of 13 296 workers of one transportation company. J. Psychosom. Res. 2011, 70, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Rufiange, M.; Beaulieu, C.; Lachapelle, P.; Dumont, M. Circadian light sensitivity and rate of retinal dark adaptation in indoor and outdoor workers. J. Biol. Rhythm. 2007, 22, 454–457. [Google Scholar] [CrossRef]
- Daugaard, S.; Markvart, J.; Bonde, J.; Christoffersen, J.; Garde, A.; Hansen, A.; Schlunssen, V.; Vestergaard, J.; Vistisen, H.; Kolstad, H. Light exposure during days with night, outdoor, and indoor work. Ann. Work Expo. Health 2019, 63, 651–665. [Google Scholar] [CrossRef]
- Chellappa, S.; Steiner, R.; Oelhafen, P.; Cajochen, C. Sex differences in light sensitivity impact on brightness perception, vigilant attention and sleep in humans. Sci. Rep. 2017, 7, 14215. [Google Scholar] [CrossRef]
- Abramov, I.; Gordon, J.; Feldman, O.; Chavarga, A. Sex and vision II: Color appearance of monochromatic lights. Biol. Sex Differ. 2012, 3, 21. [Google Scholar] [CrossRef]
- Rao, S.; Chun, C.; Fan, J.; Kofron, J.; Yang, M.B.; Hegde, R.; Ferrara, N.; Copenhagen, D.; Lang, R. A direct and melanopsin-dependent fetal light response regulates mouse eye development. Nature 2013, 494, 243–246. [Google Scholar] [CrossRef]
- Cambras, T.; Canal, M.; Cernuda-Cernuda, R.; Garcia-Fernandez, J.; Diez-Noguera, A. Darkness during early postnatal development is required for normal circadian patterns in the adult rat. Chronobiol. Int. 2015, 32, 178–186. [Google Scholar] [CrossRef]
- Kerkhof, G.; Korving, H.; Willemse-vd Geest, H.; Rietveld, W. Diurnal differences between morning-type and evening-type subjects in self-rated alertness, body temperature and the visual and auditory evoked potential. Neurosci. Lett. 1980, 1, 11–15. [Google Scholar] [CrossRef]
- Horne, J.A.; Östberg, O. A self-assessment questionnaire to determine morningness-eveningness in human circadian rhythms. Int. J. Chronobiol. 1976, 4, 97–110. [Google Scholar] [PubMed]
- Porcheret, K.; Wald, L.; Fritschi, L.; Gerkema, M.; Gordijn, M.; Merrrow, M.; Rajaratnam, S.; Rock, D.; Sletten, T.; Warman, G.; et al. Chronotype and environmental light exposure in a student population. Chronobiol. Int. 2018, 35, 1365–1374. [Google Scholar] [CrossRef] [PubMed]
- Roenneberg, T. Having trouble typing? What on earth Is chronotype? J. Biol. Rhythm. 2015, 30, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Roenneberg, T.; Wirz-Justice, A.; Merrow, M. Life between clocks: Daily temporal patterns of human chronotypes. J. Biol. Rhythm. 2003, 18, 80–90. [Google Scholar] [CrossRef]
- Goulet, G.; Mongrain, V.; Desrosiers, C.; Paquet, J.; Dumont, M. Daily light exposure in morning-type and evening-type individuals. J. Biol. Rhythm. 2007, 22, 151–158. [Google Scholar] [CrossRef]
- Shochat, T.; Santhi, N.; Herer, P.; Flavell, S.; Skeldon, A.; Dijk, D. Sleep timing in late autumn and late spring associates with light exposure rather than sun time in college students. Front. Neurosci. 2019, 13, 882. [Google Scholar] [CrossRef]
- Dumont, M.; Blais, H.; Roy, J.; Paquet, J. Controlled patterns of daytime light exposure improve circadian adjustment in simulated night work. J. Biol. Rhythm. 2009, 24, 427–437. [Google Scholar] [CrossRef]
- Matusiak, B.S.; Klöckner, C.A. How we evaluate the view out through the window. Archit. Sci. Rev. 2016, 59, 203–211. [Google Scholar] [CrossRef]
- Ulrich, R. Biophilia, biophobia, and natural landscapes. In The Biophilia Hypothesis; Kellert, S.R., Wilson, E., Eds.; Island Press: Washington, DC, USA, 1993; pp. 73–137. [Google Scholar]
- Vondrasova, D.; Hajek, I.; Illnerova, H. Exposure to long summer days affects the human melatonin and cortisol rhythms. Brain Res. 1997, 759, 166–170. [Google Scholar] [CrossRef]
- Hadlow, N.; Brown, S.; Wardrop, R.; Conradie, J.; Henley, D. Where in the world? Latitude, longitude and season contribute to the complex co-ordinates determining cortisol levels. Clin. Endocrinol. Oxf 2018, 89, 299–307. [Google Scholar] [CrossRef]
- Eastman, C.I. Natural summer and winter sunlight exposure patterns in seasonal affective disorder. Physiol. Behav. 1990, 48, 611–616. [Google Scholar] [CrossRef]
- Graw, P.; Recker, S.; Sand, L.; Kräuchi, K.; Wirz-Justice, A. Winter and summer outdoor light exposure in women with and without seasonal affective disorder. J. Affect. Disord. 1999, 56, 163–169. [Google Scholar] [CrossRef]
- Hadlow, N.; Brown, S.; Wardrop, R.; Henley, D. The effects of season, daylight saving and time of sunrise on serum cortisol in a large population. Chronobiol. Int. 2014, 31, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Roenneberg, T.; Wirz-Justice, A.; Skene, D.J.; Ancoli-Israel, S.; Wright, K.P.; Dijk, D.J.; Zee, P.; Gorman, M.R.; Winnebeck, E.; Klerman, E. Why should we abolish daylight saving time? J. Biol. Rhythm. 2019, 34, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Roenneberg, T.; Winnebeck, E.; Klerman, E. Daylight saving time and artificial time zones-a battle between biological and social times. Front. Physiol. 2019, 10, 944. [Google Scholar] [CrossRef] [PubMed]
- Kantermann, T.; Juda, M.; Merrow, M.; Roenneberg, T. The human circadian clock’s seasonal adjustment is disrupted by daylight saving time. Curr. Biol. 2007, 17, 1996–2000. [Google Scholar] [CrossRef] [PubMed]
- Borisenkov, M.; Tserne, T.; Panev, A.; Kuznetsova, E.; Petrova, N.; Timonin, V.; Kolomeichuk, S.; Vinogradova, I.; Kovyazina, M.; Khokhlov, N.; et al. Seven-year survey of sleep timing in Russian children and adolescents: Chronic 1-h forward transition of social clock is associated with increased social jetlag and winter pattern of mood seasonality. Biol. Rhythm. Res. 2017, 48, 3–12. [Google Scholar] [CrossRef]
- Andersen, M. Unweaving the human response in daylighting design. Build. Environ. 2015, 91, 101–117. [Google Scholar] [CrossRef]
- van Duijnhoven, J.; Aarts, M.P.; Aries, M.B.; Rosemann, A.; Kort, H. Systematic review on the interaction between office light conditions and occupational health: Elucidating gaps and methodological issues. Indoor Built Environ. 2019, 28, 152–174. [Google Scholar] [CrossRef]
- Dubois, M.; Cantin, F.; Johnsen, K. The effect of coated glazing on visual perception: A pilot study using scaleamodels. Lighting Res. Technol. 2007, 39, 283–304. [Google Scholar] [CrossRef]
- Dong, B.; Prakash, V.; Feng, F.; O’Neill, Z. A review of smart building sensing system for better indoor environment control. Energy Build. 2019, 199, 29–46. [Google Scholar] [CrossRef]
- Pasanen, T.; Tyrvainen, L.; Korpela, K. The relationship between perceived health and physical activity indoors, outdoors in built environments, and outdoors in nature. Appl. Psychol. Health Well Being 2014, 6, 324–346. [Google Scholar] [CrossRef] [PubMed]
- Takayama, N.; Korpela, K.; Lee, J.; Morikawa, T.; Tsunetsugu, Y.; Park, B.J.; Li, Q.; Tyrvainen, L.; Miyazaki, Y.; Kagawa, T. Emotional, restorative and vitalizing effects of forest and urban environments at four sites in Japan. Int. J. Environ. Res. Public Health 2014, 11, 7207–7230. [Google Scholar] [CrossRef] [PubMed]
- Donovan, G.; Gatziolis, D.; Douwes, J. Relationship between exposure to the natural environment and recovery from hip or knee arthroplasty: A New Zealand retrospective cohort study. BMJ. Open 2019, 9, e029522. [Google Scholar] [CrossRef]
- Nang, E.; Abuduxike, G.; Posadzki, P.; Divakar, U.; Visvalingam, N.; Nazeha, N.; Dunleavy, G.; Christopoulos, G.; Soh, C.; Jarbrink, K.; et al. Review of the potential health effects of light and environmental exposures in underground workplaces. Tunn. Undergr. Space Technol. 2019, 84, 201–209. [Google Scholar] [CrossRef]
- Spitschan, M.; Stefani, O.; Blattner, P.; Gronfier, C.; Lockley, S.; Lucas, R. How to report light exposure in human chronobiology and sleep research experiments. Clocks Sleep 2019, 1, 280–289. [Google Scholar] [CrossRef]
- International Commissoin on Illumination. CIE System for Metrology of Optical Radiation for ipRGC-Influenced Responses to Light; CIE: Vienna, Austria, 2018. [Google Scholar]
- World Health Organization. Implementation framework for phase VII (2019–2014) of the WHO European Healthy Cities Network: Goals, Requirements and Strategic Approaches. Available online: http://www.euro.who.int/en/health-topics/environment-and-health/urban-health/who-european-healthy-cities-network (accessed on 10 October 2019).
- World Health Organization. Health 21-Health for all in the 21st Century: An Introduction; World Health Organization Europe: Copenhagen, Denmark, 2019. [Google Scholar]
- Lucas, R.; Peirson, S.; Berson, D.; Brown, T.; Cooper, H.; Czeisler, C.; Figueiro, M.; Gamlin, P.; Lockley, S.; O’Hagan, J.; et al. Measuring and using light in the melanopsin age. Trends Neurosci. 2014, 37, 1–9. [Google Scholar] [CrossRef]
- Markvart, J.; Hansen, Å.; Christoffersen, J. Comparison and correction of the light sensor output from 48 wearable light exposure devices by using a side-by-side field calibration method. Leukos 2015, 11, 155–171. [Google Scholar] [CrossRef]
- Figueiro, M.; Hamner, R.; Bierman, A.; Rea, M. Comparisons of three practical field devices used to measure personal light exposures and activity levels. Lighting Res. Technol. 2013, 45, 421–434. [Google Scholar] [CrossRef]
| (i) Metabolic functions | |
| How does electric light and/or daylight affect metabolic functions? | This question is closely related to meal timing, caloric intake and meal composition, or weight loss. A few studies have investigated these aspects under laboratory conditions [72,73,74], but not yet under daylight conditions. The question is whether daylight exposure specifically affects the timing of meals, post-prandial responses, temperature regulation, metabolism, body composition and the gut microbiome? |
| (ii)Sleep | |
| Does daylight and/or mixed daylight/electric light conditions during the day mediate better sleep quality at night, and if yes, how? | There is some evidence that indoor and/or outdoor bright light exposure during the day leads to longer sleep duration and increases sleep quality as was shown in laboratory and field studies [63,64,75]. Electric light exposure (with a spectral power distribution closer to daylight than standard LED-light) during daytime enhances slow wave sleep (=‘deep sleep’) [76] and brighter ambient light (compared to dim light) increased homeostatic sleep pressure during wakefulness [77]. The following questions relate to daylight and sleep:
|
| (iii) Alertness andcognition | |
| How do daylight-specific properties affect alertness and cognitive functions? | Many laboratory studies with steady state electric lighting showed light-dependent alertness [45,46,49,81,82,83] and cognitive repercussions [46,53,55]. A few studies have looked at spectrally tuned electrical light conditions and alertness and cognition [76,84,85,86]. |
| (iv)Physicalactivity | |
| Does physical activity and daylight interact to induce larger phase shifts of the circadian clock? | From studies in the laboratory there is a phase-response curve of physical activity with both phase delays and advances at specific times of day [87]. Activity facilitates phase delays in very dim light [88]. The question relates to the interaction of exercise with (day-) light exposure as shown with cycling performance [89]. |
| Daylight and Circadian Entrainment | |
| (i) Daylight properties: Quality/quantity | Which combinations of daylight qualities and quantities are relevant for circadian entrainment (alone and in combination with electric light sources) and dependent on time of day? |
| Spectrum/colour | Which frequency bands of the electromagnetic spectrum are relevant when and how important is the integration of colour [100]? |
| Dynamics | Do ultradian variations/dynamics of daylight play a role in circadian entrainment? |
| Twilight | What is the role of twilight (dawn and dusk) for circadian entrainment in humans [101,102]? For example, is the colour signal of the “blue hour” as important for humans as it is for mice [103,104]? |
| Polarisation of daylight | What is the role of polarised daylight? Direct sunlight is not polarised but daylight from a particular region of the sky is partially polarised. In contrast to bees, humans can hardly perceive polarised light, however a physiological influence of polarised light on humans cannot yet be excluded. A study by Brainard et al. showed no difference between unpolarised and polarised light on melatonin suppression [105]. |
| Role of light distribution in the visual field, direction of light | A further characteristic of daylight is its large-area expansion with an unobstructed view of the sky. Non-human primates have ipRGcs distributed over the entire retina with a density of 3-5 cells/mm², and a maximum concentration of 20-25 cells/mm² around the fovea [106]. Since rods, cones and the ipRGCs in the eye are distributed over large areas of the retina, it is assumed that the non-visual effect of light is greatest when the light comes from a large source, such as an indirect illumination of a large bright area. In nature, this light comes from the sky. If only a small area of the retina is illuminated, as is the case with directed light from a spot, a weaker non-visual effect is assumed. The direction of light seems to be crucial at least for electric light that was shown to affect melatonin suppression [107,108,109] and sleep [110]. |
| Season, latitude and day length | Does photoperiod length impact on circadian entrainment? From the literature it is known that there are differences in humans under purely natural seasonal lighting conditions when compared to mixed natural/electric light conditions [78,79,111]. |
| (ii) Effect on peripheral tissues | How does electric light and/or daylight (indirectly) influence peripheral clock tissue function (e.g., metabolism, immune function, cardiovascular function, cell repair, detoxification, mitochondrial turnover)? |
| (iii) Circadian misalignment/disruption | How can electric and/or daylight help to mitigate circadian misalignment/circadian disruption (e.g., in shift workers [112,113,114], adolescents)? |
| Spectral range | Does the fact that the spectral power distribution of daylight goes beyond the visible range make a difference for non-visual functions, when compared to electric light? For example, the infrared (IR) portion that is always present in daylight is non-existent in electric light (except for banned incandescent light sources). It might have an important role for the retina, since a large body of literature suggests connections between long-wavelength radiation and (beneficial) physiological functions in the retina (see e.g., [118,119]). In addition, there is probably a specific role of the spectral power distribution and colour of twilight, as shown in rodents [103,104]. |
| Duration and thresholds |
|
| Dose-response relationships | What are the dose-response relationships for daylight and mixed daylight/electric light exposures for non-visual functions? In particular, with regard to the interaction of circadian and homeostatic processes with environmental conditions – that include electric light. |
| Irradiance and spectral composition | Daylight is spatially and temporally variable. These changes take place over very wide frequency bands:
|
| 24 h-Dynamics | How do the 24-h dynamics of daylight impact on non-visual functions? |
| Light history | How can prior light history (see Table 4) of any light exposure be incorporated into the above-mentioned outcomes? |
| Therapeutic use of light | Light treatment has been established for winter and other depressive disorders and circadian sleep disturbances [122,123,124,125]. What should the daylight exposure recommendations be for these different therapeutic interventions (regarding intensity, spectrum, timing and prior light exposure)? |
(i) Physiological differences:
|
(ii) Genetic differences:
|
| (iii) Cultural, behavioural differences: |
(iv) Mixed physiological/behavioural effects:
|
| Daylight conditions for individuals at workplaces | How much and which qualities of daylight do different individuals/groups receive at their work- and living places (24/7), and how does this relate to their health status? |
| Daylight exposure as a countermeasure (for shift workers) | Can daylight be a means to counteract the detrimental effects of “light at the wrong time of day”, such as occurs with light in the evening or with (night) shift work? The reason might be that light during the day has a desensitisation effect for light exposure in the evening (see Table 3 and Table 4: light history), and in night shift workers bright light exposure after daytime sleep could help readjustment to the daytime work hours [204], see also recent recommendations of the ‘Working Time Society’ (WTS/ICOH; [113]). |
| Daylight and visual comfort and non-visual functions at workplaces | Are existing workspace regulations on glare and visual comfort at workplaces sufficient to concomitantly provide good and ‘biologically relevant’ daylight conditions [192]? |
| View/window | How important is the view out of the window and the environment outside the window [205] for workplace-related variables such as performance and alertness? |
| Impactof latitude, time zones, climate | |
| Seasonal changes | Does exposure to seasonal changes in day length have consequences for human physiology and health, as some of the existing literature suggests [79,111,207,208]? |
| Latitude | In those most vulnerable e.g., living at high latitudes, light therapy and improved home/work lighting has been shown to be useful for winter depression and sleep disorders, given that there is insufficient daylight for some months of the year [209,210]. |
| Location within time zone | Does the location within a time zone modify daylight’s effect on an individual’s circadian phase [208,211]? |
| Daylight saving time (DST) | Is there a long-term effect of daylight saving time [212,213,214,215] on wake and sleep physiology? |
| Micro/macroclimate | How does ambient temperature, humidity, and air pollution modulate daylight’s effects on physiology? |
| Rural vs. urban environments | Is there a difference in daylight exposure dose between those living in rural vs. urban environments? |
| Architecture, urban design | |
| Daylight conditions in buildings | What role do the complex daylight conditions in buildings, such as building orientation, window positioning, glazing, play on visual comfort/adversity (glare), productivity and performance, circadian entrainment and health in general [32,216,217]? Could architecture be deployed to accentuate spatial-temporal modulation of daylight to stimulate retinal photoreception? |
| Glazing | How do single vs. double vs. triple glazing systems, electrochromic windows, and new technologies such as smart windows with integrated micro-daylighting systems influence health [218]? How effective is dynamic glazing in order to address heat emission and light? |
| Floor orientation, spatial distribution of daylight | How does the location of apartments (floor and geographical orientation) within a given building, and the indoor spatial distribution of daylight, affect human health? |
| Daylight enhanced qualities | What design can provide daylight-enhanced qualities in single buildings and urban settings? |
| Complementation with electrical light/daylight systems | How can the geographical orientation of a building, which determines its indoor light intensity, be compensated/complemented with daylight systems/electric light? |
| (Day-) light control systems | How should modern (light) sensor and controller technology be used to support health, performance, and well-being of its inhabitants? |
| Self-control of (day-) light | How much self-control over sensor control is necessary and desired [219]? |
| Alienation from nature | What are the consequences of losses of daylight/weather/seasonal effects due to urban densification, loss of daylight recreation areas, daylight restriction due to high buildings etc. on mood, health and quality of life [69,220,221,222,223]? |
| Role of view | Low daylight exposure and daylight deprivation usually also means deprivation from a view. This could have additional and far-reaching negative consequences, although research to date is scarce [223]. |
| Automated integration of daylight and electric light in buildings | Lack of sophisticated and automated integration of daylight and electric light in buildings and algorithms to detect the two separately (this would also support reduction of lighting derived energy costs) |
| Monitoring spectral irradiance | Lack of validated, commercially available and affordable wearable devices for continuously monitoring spectral irradiance (at eye level). This could also be used as a “dosimeter” in research, therapies, living/working spaces and for lifestyle applications. |
| Tools for circadian phase assessments | Lack of practical means to make circadian phase assessments in daily life, the clinic, and elsewhere (comparing electric and daylight conditions). |
| Tools for mental health evaluations | Long-term daily mental health evaluations; approaches developed so far are wrist-worn diaries with visual analogue scales (also via mobile derived apps). |
| Standard operation procedures for the use of daylight treatment and daylight exposure (with regards to non-visual light responses) | Lack of standard operation procedures (SOPs) and definitions of daylight treatment responses for different individuals and patients. There is a need for large-scale field studies in schools, institutions (e.g., hospitals, prisons, care homes), shift- and non-shift workplaces, people working underground and people frequently traveling across times zones (with different overlay stays) using the same SOPs. |
| Norms and metrics | No (internationally accepted) consensus on the parameters to be measured and reported, and at what level of accuracy the monitoring tools can achieve this (see Section 2.3). A recent tutorial paper summarises the most important requirements [224]. One question that also arises: What is a suitable light metric to measure “naturalness” of light? How can we compare electric light sources to daylight? Some existing official metrics are summarised in the most recent international CIE standard, even though the D65/D55 parameters do not reflect the spectrum of daylight [225] and need to be revised to incorporate seasonal and geographical changes. |
| Large scale lighting digitalisation | Lack of large scale/practical biomedical digital techniques to design, monitor, predict and validate individually tailored daylight exposure/electric light regimens. |
| Criteria | Define criteria for (day) light measures (see e.g., the new CIE standard S026). Universally agree to use this new standard, which is facilitated by the CIE-S-026-EDI-Toolbox-beta version E1.051.xls based on Ref. [228] and a more recent Tutorial [224]. |
| Daytime biomarkers for physiology and behaviour | Replicate physiological, cognitive and emotional outcome (bio-) markers, and validate more than once in different laboratories, in order that they be implemented as reliable markers for describing the effect of daylight on physiology and behaviour. |
| Devices to monitor daylight | Develop robust, validated and commercially affordable devices to monitor spectral daylight exposure (representing light exposure at eye level in a vertical direction) along with temperature and humidity continuously indoors and outdoors. The need to calibrate and correct outputs from light recordings has been shown mostly for wrist worn devices [229,230]. |
| Application in different populations | The questions discussed in Section 2 and Section 3 additionally need to be carried out in men and women, different ages (children, elderly) and ethnic groups, monitored across different environments (school/workplace/home); seasons; as well as in different patient groups (such as disorders of the nervous system, eyes). |
| Standard/exemplary data sets | Collect and evaluate different data sets to assess how much and which qualities of daylight different groups of individuals/patients receive at their work- and living places (24/7). |
| Status quo in real life | Define the status quo in daylight: irradiance measurements (including melanopic irradiance), interviews/focus groups with target populations (e.g., care home residents, prison inmates, dermatologists, ophthalmologists, shift- and night workers, tourist industry, people working underground (at e.g., train stations, miners), and related disciplines. |
| Laboratory and field studies | Do prospective longitudinal and multicentre studies using the established SOPs under field and laboratory conditions in a sufficiently large sample, performed in both males and females. |
| Combined day- and electric light interventions | Compare light interventions with daylight exposure, and not only electric light OR daylight but also the (dynamic) mixture of both should be studied, since this is the norm. Additionally, a suitable scheduled length of daily light exposure (daily accumulation) requires monitoring over a long duration (chronic/seasonal light exposure). |
| Guidelines/recommendations for research and clinical studies | Propose guidelines/recommendations on how to set-up daylight research studies and clinical trials with daylight interventions. |
| Guidelines/recommendations for short- and long-term interventions | Propose guidelines/recommendations on how to measure/monitor daylight interventions and related physiological and behavioural outcomes over time. |
| Tools for inter-individual differences and circadian phase | Develop reliable tools to assess inter-individual differences and internal circadian phase for users and practitioners. |
| Instruments for light monitoring in the field | Further validate instruments which monitor individual light and colour perception/preferences under daylight conditions in the field. |
| Daylight recommendations for task and biological requirements | Develop daylight recommendations, which relate to both task requirements and physiological/psychological aspects. |
| Education | Educate professionals, government policy makers, and the public in ‘light hygiene’ e.g., sufficient vs. inadequate vs. too much daylight exposure. |
| Architecture/building science | Early stage planning of building/window positioning optimised for better daylight exposure and spatial-temporal modulation. Develop real mixed electric light/daylight simulation tools. Multimodal approach for optimised light exposure, heat emission, noise, air quality. |
| Database | Create an open access daylight database (wiki/online depository). |
| Modelling | Develop mathematical models that help predict biologically appropriate daylight exposure (e.g., characteristics such as timing, dose, spectral composition and light exposure pattern) for promoting circadian entrainment, sleep, performance and health in addition to the required standards for visual function at the level of the individual as well as in different populations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Münch, M.; Wirz-Justice, A.; Brown, S.A.; Kantermann, T.; Martiny, K.; Stefani, O.; Vetter, C.; Wright, K.P., Jr.; Wulff, K.; Skene, D.J. The Role of Daylight for Humans: Gaps in Current Knowledge. Clocks & Sleep 2020, 2, 61-85. https://doi.org/10.3390/clockssleep2010008
Münch M, Wirz-Justice A, Brown SA, Kantermann T, Martiny K, Stefani O, Vetter C, Wright KP Jr., Wulff K, Skene DJ. The Role of Daylight for Humans: Gaps in Current Knowledge. Clocks & Sleep. 2020; 2(1):61-85. https://doi.org/10.3390/clockssleep2010008
Chicago/Turabian StyleMünch, Mirjam, Anna Wirz-Justice, Steven A. Brown, Thomas Kantermann, Klaus Martiny, Oliver Stefani, Céline Vetter, Kenneth P. Wright, Jr., Katharina Wulff, and Debra J. Skene. 2020. "The Role of Daylight for Humans: Gaps in Current Knowledge" Clocks & Sleep 2, no. 1: 61-85. https://doi.org/10.3390/clockssleep2010008
APA StyleMünch, M., Wirz-Justice, A., Brown, S. A., Kantermann, T., Martiny, K., Stefani, O., Vetter, C., Wright, K. P., Jr., Wulff, K., & Skene, D. J. (2020). The Role of Daylight for Humans: Gaps in Current Knowledge. Clocks & Sleep, 2(1), 61-85. https://doi.org/10.3390/clockssleep2010008








